Risk of Contralateral Breast Cancer in Women with and without Pathogenic Variants in BRCA1, BRCA2, and TP53 Genes in Women with Very Early-Onset (<36 Years) Breast Cancer
Abstract
1. Introduction
2. Methods
- A population-based study of 287 women aged 30 or under and diagnosed with breast cancer between January 1980 and December 1997 and recorded in the North-West of England cancer registry. Living participants provided consent for blood samples to be tested for BRCA1, BRCA2, and TP53 PVs (n = 125; 39 PV carriers).
- women that were proven carriers (n = 358) from 854 BRCA1, 851 BRCA2, and 74 TP53 positive families referred to the Genomic Medicine Department at St Mary’s Hospital, Manchester from 1990–2018, and not identified from 1.
- women without known PVs in the three genes were ascertained from (1) and the Family History Clinic at Wythenshawe Hospital South Manchester after testing negative for PVs in all three genes following a diagnosis of breast cancer aged 35 years and under (n = 14).
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ford, D.; Easton, D.F.; Stratton, M.; Narod, S.; Goldgar, D.; Devilee, P.; Bishop, D.T.; Weber, B.; Lenoir, G.; Chang-Claude, J.; et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am. J. Hum. Genet. 1998, 62, 676–689. [Google Scholar] [CrossRef]
- Amadou, A.; Achatz, M.I.W.; Hainaut, P. Revisiting tumor patterns and penetrance in germline TP53 mutation carriers: Temporal phases of Li-Fraumeni syndrome. Curr. Opin. Oncol. 2018, 30, 23–29. [Google Scholar] [CrossRef]
- Evans, D.G.; Shenton, A.; Woodward, E.; Lalloo, F.; Howell, A.; Maher, E.R. Penetrance estimates for BRCA1 and BRCA2 based on genetic testing in a Clinical Cancer Genetics service setting: Risks of breast/ovarian cancer quoted should reflect the cancer burden in the family. BMC Cancer 2008, 8, 155. [Google Scholar] [CrossRef]
- Mavaddat, N.; Peock, S.; Frost, D.; Ellis, S.; Platte, R.; Fineberg, E.; Evans, D.G.; Izatt, L.; Eeles, R.A.; Adlard, J.; et al. Cancer risks for BRCA1 and BRCA2 mutation carriers: Results from prospective analysis of EMBRACE. J. Natl. Cancer Inst. 2013, 105, 812–822. [Google Scholar] [CrossRef]
- Lalloo, F.; Varley, J.; Ellis, D.; Moran, A.; O’Dair, L.; Pharoah, P.; Evans, D.G.; Early Onset Breast Cancer Study Group. Prediction of pathogenic mutations in patients with early-onset breast cancer by family history. Lancet 2003, 361, 1101–1102. [Google Scholar] [CrossRef]
- Lalloo, F.; Varley, J.; Moran, A.; Ellis, D.; O’Dair, L.; Pharoah, P.; Antoniou, A.; Hartley, R.; Shenton, A.; Seal, S.; et al. BRCA1, BRCA2 and TP53 mutations in very early-onset breast cancer with associated risks to relatives. Eur. J. Cancer 2006, 42, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Molina-Montes, E.; Pérez-Nevot, B.; Pollán, M.; Sánchez-Cantalejo, E.; Espín, J.; Sánchez, M.J. Cumulative risk of second primary contralateral breast cancer in BRCA1/BRCA2 mutation carriers with a first breast cancer: A systematic review and meta-analysis. Breast 2014, 23, 721–742. [Google Scholar] [CrossRef]
- Evans, D.G.R.; Moran, A.; Hartley, R.; Dawson, J.; Bulman, B.; Knox, F.; Howell, A.; Lalloo, F. Long-term outcomes of breast cancer in women aged 30 years or younger, based on family history, pathology and BRCA1/BRCA2/TP53 status. Br. J. Cancer 2010, 102, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Schon, K.; Tischkowitz, M. Clinical implications of germline mutations in breast cancer: TP53. Breast Cancer Res. Treat. 2018, 167, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Paluch-Shimon, S.; Cardoso, F.; Sessa, C.; Balmana, J.; Cardoso, M.J.; Gilbert, F.; Senkus, E.; ESMO Guidelines Committee. Prevention and screening in BRCA mutation carriers and other breast/ovarian hereditary cancer syndromes: ESMO clinical practice guidelines for cancer prevention and screening. Ann. Oncol. 2016, 27 (Suppl. 5), v103–v110. [Google Scholar] [CrossRef]
- Mai, P.L.; Best, A.F.; Peters, J.A.; DeCastro, R.M.; Khincha, P.P.; Loud, J.T.; Bremer, R.C.; Rosenberg, P.S.; Savage, S.A. Risks of first and subsequent cancers among TP53 mutation carriers in the National Cancer Institute Li-Fraumeni syndrome cohort. Cancer 2016, 122, 3673–3681. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef] [PubMed]
- Hisada, M.; Garber, J.E.; Li, F.P.; Fung, C.Y.; Fraumeni, J.F. Multiple Primary Cancers in Families with Li-Fraumeni Syndrome. J. Natl. Cancer Inst. 1998, 90, 606–611. [Google Scholar] [CrossRef] [PubMed]
- National Collaborating Centre for Cancer (UK). Familial Breast Cancer: Classification and Care of People at Risk of Familial Breast Cancer and Management of Breast Cancer and Related Risks in People with a Family History of Breast Cancer; National Collaborating Centre for Cancer (National Institute for Health and Clinical Excellence: Guidance): Cardiff, UK, 2013. Available online: http://www.ncbi.nlm.nih.gov/books/NBK247567/ (accessed on 5 November 2019).
- Heymann, S.; Delaloge, S.; Rahal, A.; Caron, O.; Frebourg, T.; Barreau, L.; Pachet, C.; Mathieu, M.C.; Marsiglia, H.; Bourgier, C. Radio-induced malignancies after breast cancer postoperative radiotherapy in patients with Li-Fraumeni syndrome. Radiat. Oncol. Lond. Engl. 2010, 5, 104. [Google Scholar] [CrossRef]
- Malone, K.E.; Begg, C.B.; Haile, R.W.; Borg, A.; Concannon, P.; Tellhed, L.; Xue, S.; Teroka, S.; Bernstein, L.; Capanu, M.; et al. Population-Based Study of the Risk of Second Primary Contralateral Breast Cancer Associated with Carrying a Mutation in BRCA1 or BRCA2. J. Clin. Oncol. 2010, 28, 2404–2410. [Google Scholar] [CrossRef]
- Rhiem, K.; Engel, C.; Graeser, M.; Zachariae, S.; Kast, K.; Kiechle, M.; Ditsch, N.; Janni, W.; Mundhenke, C.; Golatta, M.; et al. The risk of contralateral breast cancer in patients from BRCA1/2 negative high risk families as compared to patients from BRCA1 or BRCA2 positive families: A retrospective cohort study. Breast Cancer Res. 2012, 14, R156. [Google Scholar] [CrossRef]
- Kurian, A.W.; McClure, L.A.; John, E.M.; Horn-Ross, P.L.; Ford, J.M.; Clarke, C.A. Second primary breast cancer occurrence according to hormone receptor status. J. Natl. Cancer Inst. 2009, 101, 1058–1065. [Google Scholar] [CrossRef]
- Brewer, H.R.; Jones, M.E.; Schoemaker, M.J.; Ashworth, A.; Swerdlow, A.J. Family history and risk of breast cancer: An analysis accounting for family structure. Breast Cancer Res. Treat. 2017, 165, 193–200. [Google Scholar] [CrossRef]
- The Hereditary Breast Cancer Clinical Study Group; Kotsopoulos, J.; Lubinski, J.; Lynch, H.T.; Tung, N.; Armel, S.; Senter, L.; Singer, C.F.; Fruscio, R.; Couch, F.; et al. Oophorectomy and risk of contralateral breast cancer among BRCA1 and BRCA2 mutation carriers. Breast Cancer Res. Treat. 2019, 175, 443–449. [Google Scholar] [CrossRef]
- Razdan, S.N.; Patel, V.; Jewell, S.; McCarthy, C.M. Quality of life among patients after bilateral prophylactic mastectomy: A systematic review of patient reported outcomes. Qual. Life Res. 2016, 25, 1409–1421. [Google Scholar] [CrossRef]
- Cuzick, J.; Sestak, I.; Cawthorn, S.; Hamed, H.; Holli, K.; Howell, A.; Forbes, J.F.; IBIS-I Investigators. Tamoxifen for prevention of breast cancer: Extended long-term follow-up of the IBIS-I breast cancer prevention trial. Lancet Oncol. 2015, 16, 67–75. [Google Scholar] [CrossRef]
- DeCensi, A.; Puntoni, M.; Guerrieri-Gonzaga, A.; Caviglia, S.; Avino, F.; Cortesi, L.; Taverniti, C.; Pacquola, M.G.; Falcini, F.; Gulisano, M.; et al. Randomized Placebo Controlled Trial of Low-Dose Tamoxifen to Prevent Local and Contralateral Recurrence in Breast Intraepithelial Neoplasia. J. Clin. Oncol. 2019, 37, 1629–1637. [Google Scholar] [CrossRef] [PubMed]

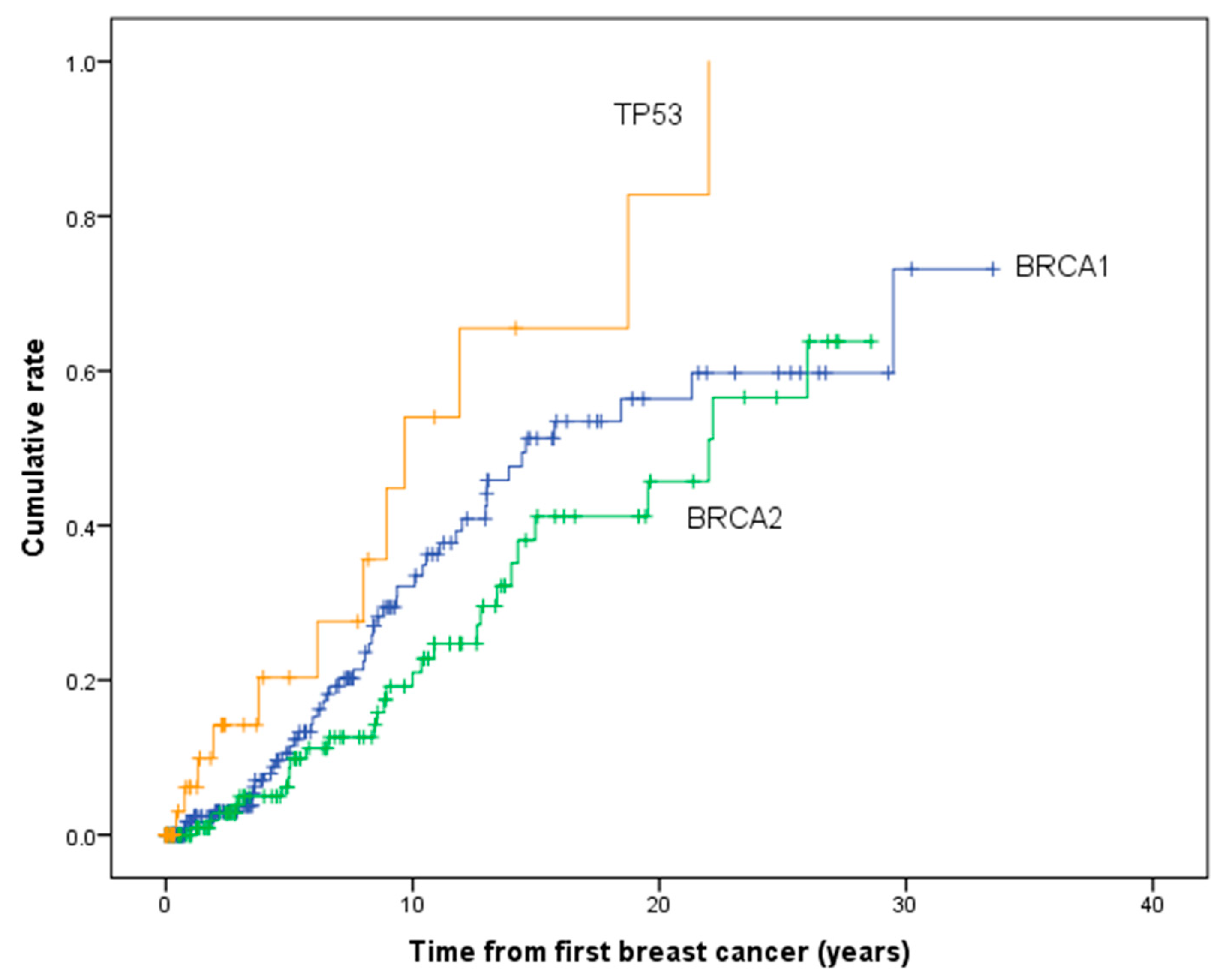
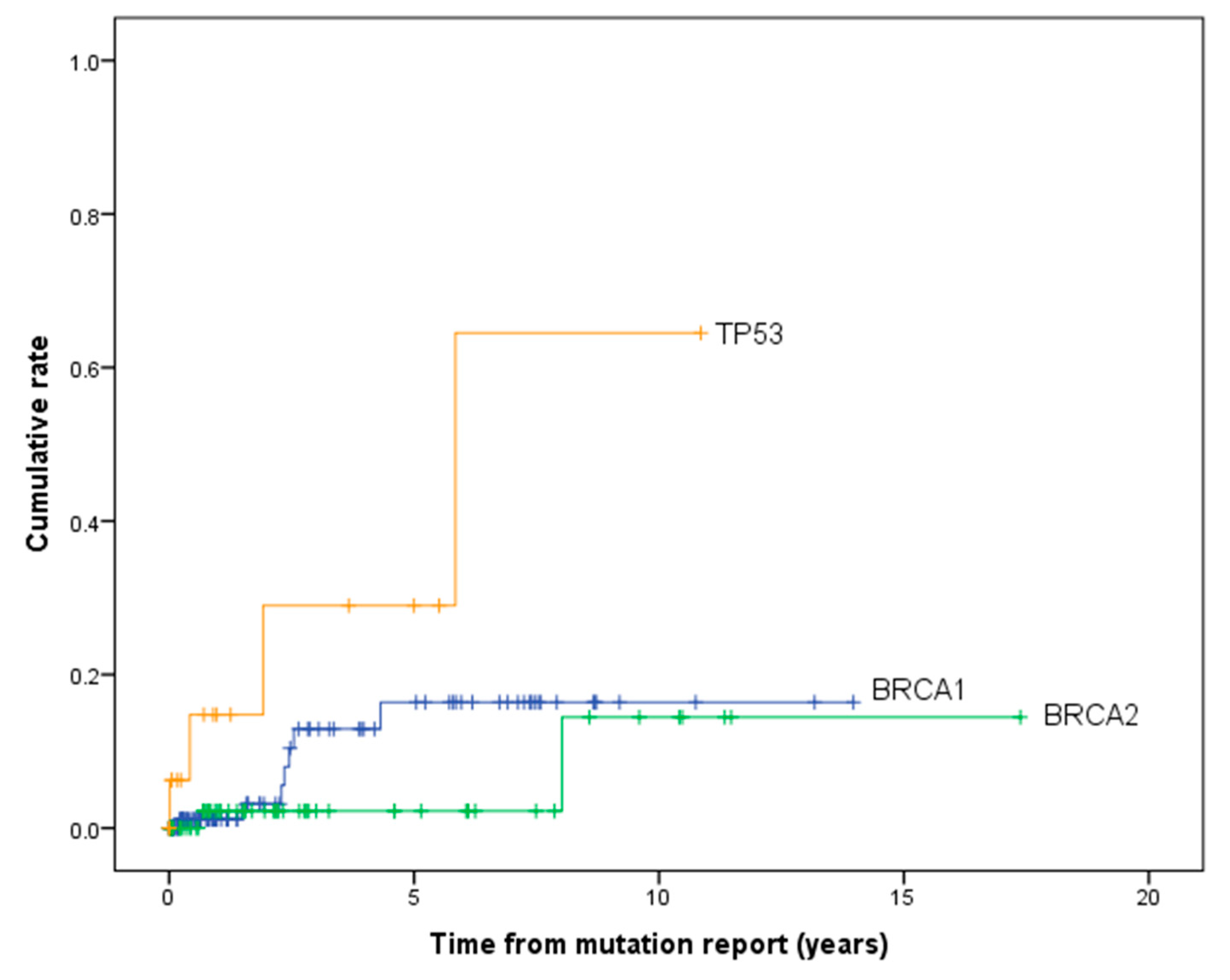
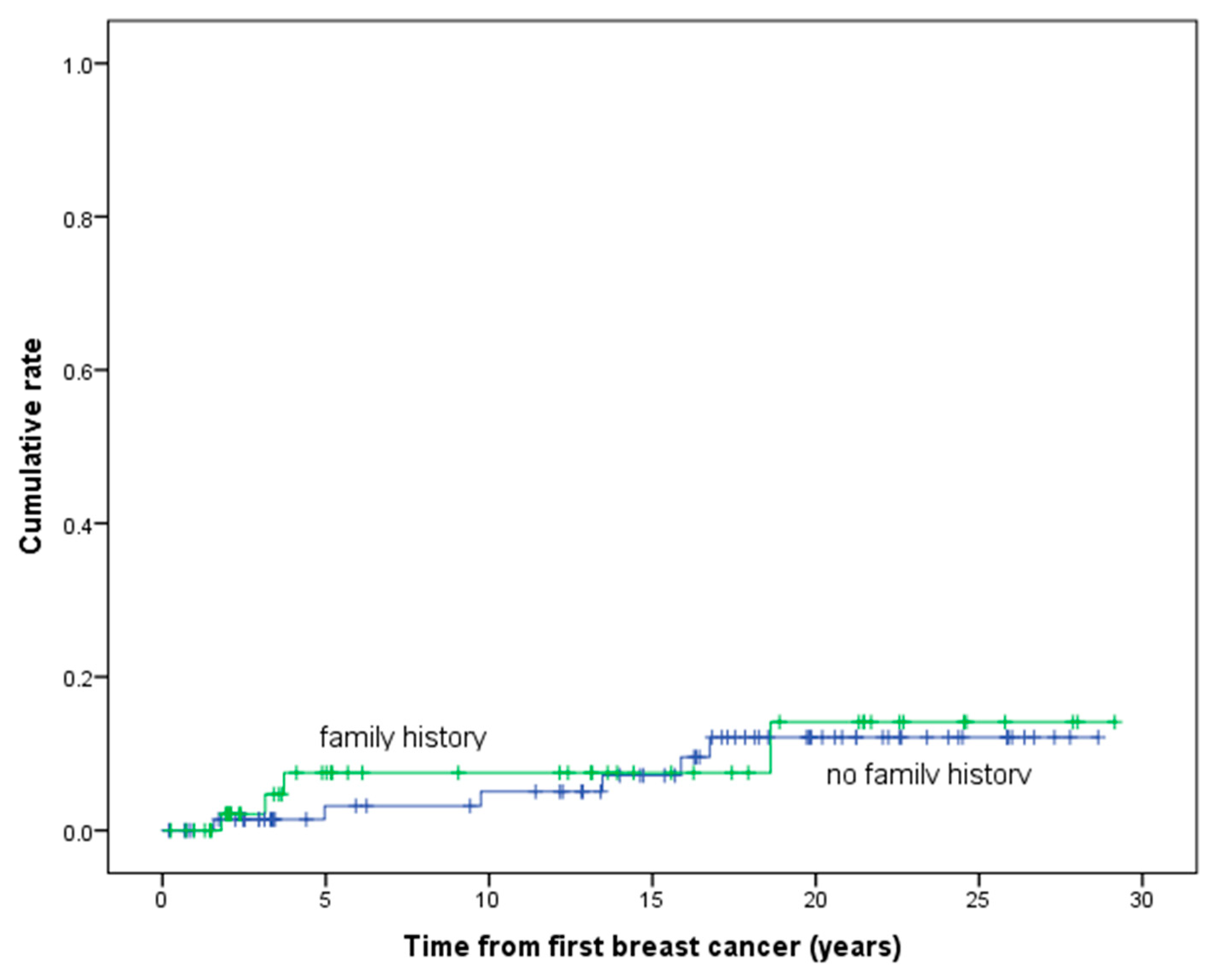
| Genetic Variant * | Total Breast Cancer (n) | Carrier Status | Contralateral Breast Cancer n (%) | Synchronous Breast Cancer n (%) | Risk-Reducing Mastectomy n (%) | Died n (%) | Person Years of Follow-Up | Annual Rate Contralateral Breast Cancer (Excluding Synchronous) (%) | Annual Rate All Contralateral and Synchronous Breast Cancer |
|---|---|---|---|---|---|---|---|---|---|
| BRCA1 | 218 | Y | 54 (24.8) | 4 (1.8) | 36 (16.5) | 48 (22.0) | 1403 | 3.57 | 3.85 |
| BRCA2 | 132 | Y | 30 (22.7) | 2 (1.5) | 23 (17.4) | 35 (26.5) | 1063 | 2.63 | 2.82 |
| TP53 | 47 | Y | 14 (29.8) | 2 (4.3) | 6 (12.8) | 23 (48.9) | 171 | 7.03 | 8.20 |
| Total | 397 | 98 (24.7) | 8 (2.0) | 65 (16.4) | 106 (26.7) | 2636 | 3.41 | 3.72 | |
| Family history | 53 | N | 4 (7.5) | - | 26 (49.1) | 576 | - | 0.69 | |
| No family history | 74 | N | 6 (8.1) | - | 23 (31.1) | 1074 | - | 0.56 | |
| Family history unknown | 134 | N | 3 (2.2) | - | 85 (63.4) | 1312 | - | 0.23 | |
| Total | 261 | 13 (5.0) | - | 134 (51.3) | 2963 | - | 0.44 |
| Genetic Variant | Total Breast Cancers (n) | Contralateral Breast Cancers (n) | Synchronous Breast Cancers (n) | Years of Follow-Up | Annual Rate Contralateral Breast Cancer (Excluding Synchronous) (%) | Annual Rate All Contralateral and Synchronous Breast Cancers |
|---|---|---|---|---|---|---|
| BRCA1 | 108 | 7 (6.5) | 0 (0.0) | 280 | 2.50 | 2.50 |
| BRCA2 | 63 | 3 (4.8) | 1 (1.6) | 187.0 | 1.07 | 1.60 |
| TP53 | 19 | 5 (26.3) | 1 (5.3) | 37.6 | 10.65 | 13.31 |
| Total | 190 | 15 (7.9) | 2 (1.1) | 504.4 | 2.58 | 2.97 |
| Genetic Variant | All Women | Post PV Report | ||||
|---|---|---|---|---|---|---|
| Years after Initial Breast Cancer | Cumulative Risk (%) | 95% CI | Years after Initial Breast Cancer | Cumulative Risk (%) | 95% CI | |
| BRCA1 | 5 | 10.5 | 6.37–17.2 | 5 | 16.6 | 8.0–32.5 |
| 10 | 32.3 | 24.0–42.6 | 10 | 16.6 | 8.0–32.5 | |
| 15 | 51.5 | 40.7–63.4 | 15 | 16.6 | 8.0–32.5 | |
| 20 | 56.6 | 44.8–69.1 | 20 | 16.6 | 8.0–32.5 | |
| BRCA2 | 5 | 7.2 | 3.5–14.6 | 5 | 2.0 | 0.3–13.5 |
| 10 | 20.7 | 13.0–32.1 | 10 | 15.1 | 2.9–60.4 | |
| 15 | 40.9 | 28.3–56.4 | 15 | 15.1 | 2.9–60.4 | |
| 20 | 45.3 | 31.4–61.9 | 20 | 15.1 | 2.9–60.4 | |
| TP53 | 5 | 19.1 | 8.0–41.8 | 5 | 27.9 | 9.3–66.6 |
| 10 | 53.1 | 29.6–80.6 | 10 | 56.8 | 20.5–95.3 | |
| 15 | 64.9 | 38.2–89.7 | 15 | 56.8 | 20.5–95.3 | |
| 20 | 82.4 | 49.9–98.8 | 20 | 56.8 | 20.5–95.3 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hyder, Z.; Harkness, E.F.; Woodward, E.R.; Bowers, N.L.; Pereira, M.; Wallace, A.J.; Howell, S.J.; Howell, A.; Lalloo, F.; Newman, W.G.; et al. Risk of Contralateral Breast Cancer in Women with and without Pathogenic Variants in BRCA1, BRCA2, and TP53 Genes in Women with Very Early-Onset (<36 Years) Breast Cancer. Cancers 2020, 12, 378. https://doi.org/10.3390/cancers12020378
Hyder Z, Harkness EF, Woodward ER, Bowers NL, Pereira M, Wallace AJ, Howell SJ, Howell A, Lalloo F, Newman WG, et al. Risk of Contralateral Breast Cancer in Women with and without Pathogenic Variants in BRCA1, BRCA2, and TP53 Genes in Women with Very Early-Onset (<36 Years) Breast Cancer. Cancers. 2020; 12(2):378. https://doi.org/10.3390/cancers12020378
Chicago/Turabian StyleHyder, Zerin, Elaine F. Harkness, Emma R. Woodward, Naomi L. Bowers, Marta Pereira, Andrew J. Wallace, Sacha J. Howell, Anthony Howell, Fiona Lalloo, William G. Newman, and et al. 2020. "Risk of Contralateral Breast Cancer in Women with and without Pathogenic Variants in BRCA1, BRCA2, and TP53 Genes in Women with Very Early-Onset (<36 Years) Breast Cancer" Cancers 12, no. 2: 378. https://doi.org/10.3390/cancers12020378
APA StyleHyder, Z., Harkness, E. F., Woodward, E. R., Bowers, N. L., Pereira, M., Wallace, A. J., Howell, S. J., Howell, A., Lalloo, F., Newman, W. G., Smith, M. J., & Evans, D. G. (2020). Risk of Contralateral Breast Cancer in Women with and without Pathogenic Variants in BRCA1, BRCA2, and TP53 Genes in Women with Very Early-Onset (<36 Years) Breast Cancer. Cancers, 12(2), 378. https://doi.org/10.3390/cancers12020378






