Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
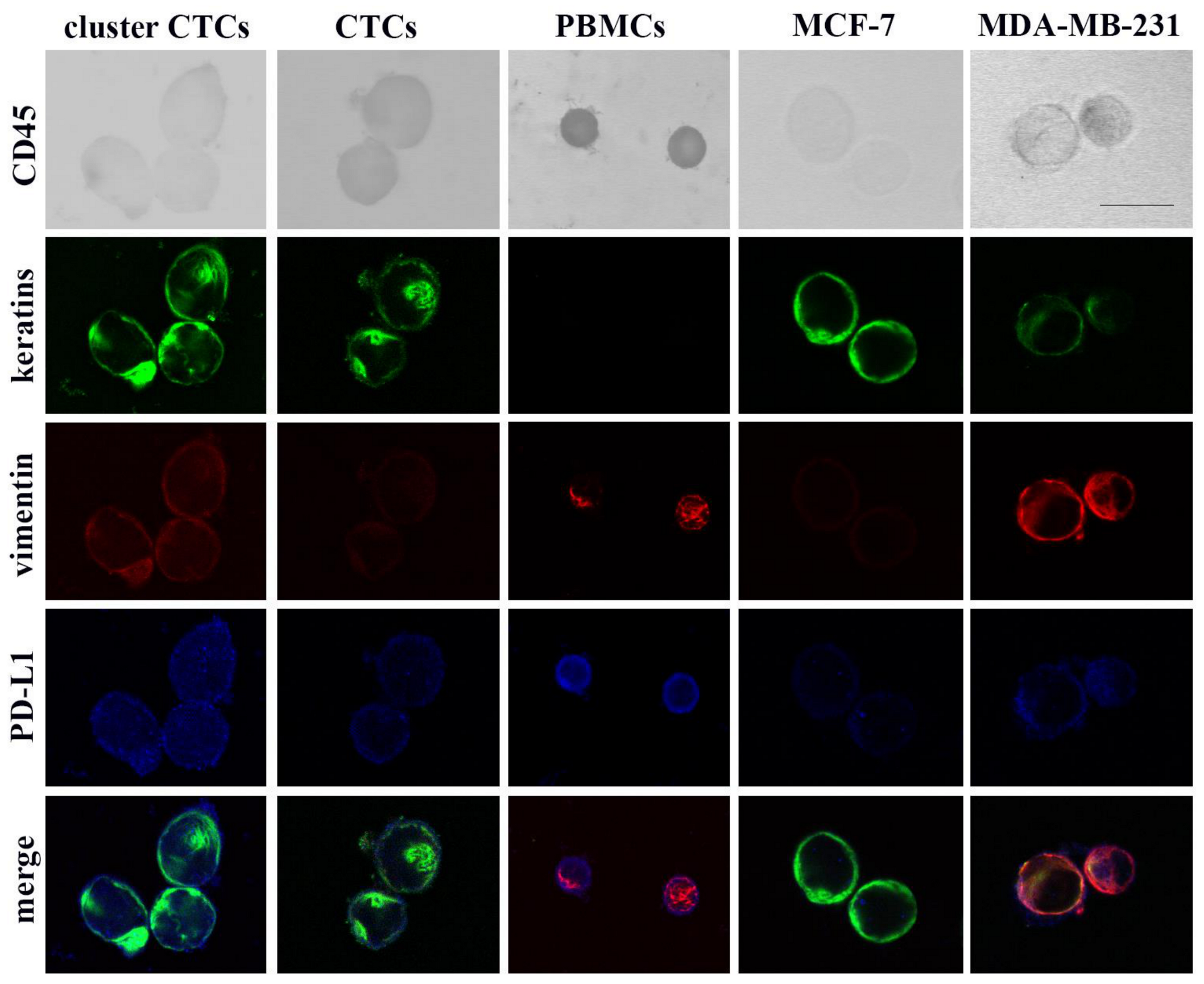
2.1. Expression Levels of Keratins, Vimentin and PD-L1 in Breast Cancer Cell Lines
2.2. Patient and Disease Characteristics
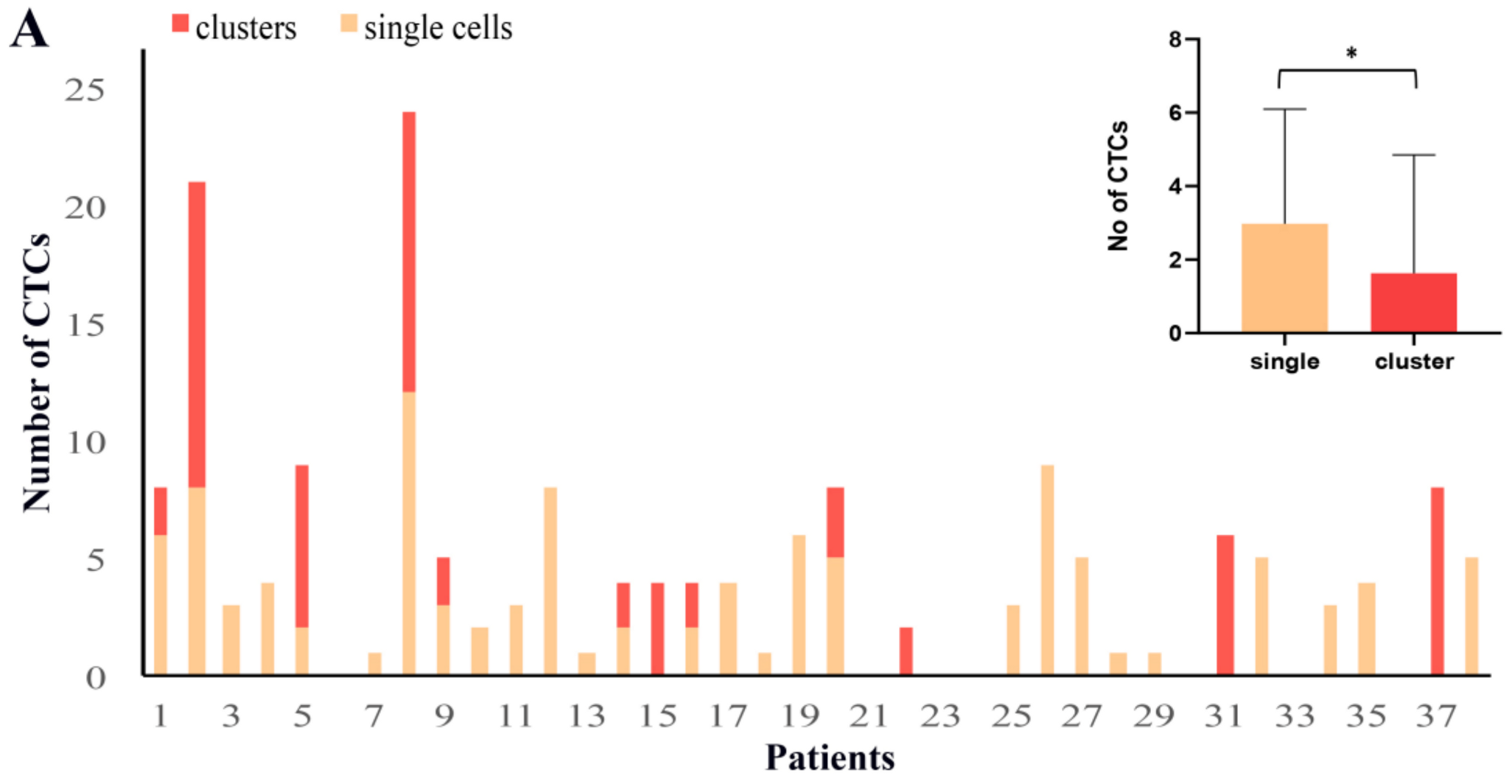
2.3. Detection and Characterization of CTCs Acording to EMT Status and PD-L1 Expression at Baseline
2.4. Detection and Characterization of CTCs Acording to EMT Status and PD-L1 Expression on Day 8 of Eribulin Administration
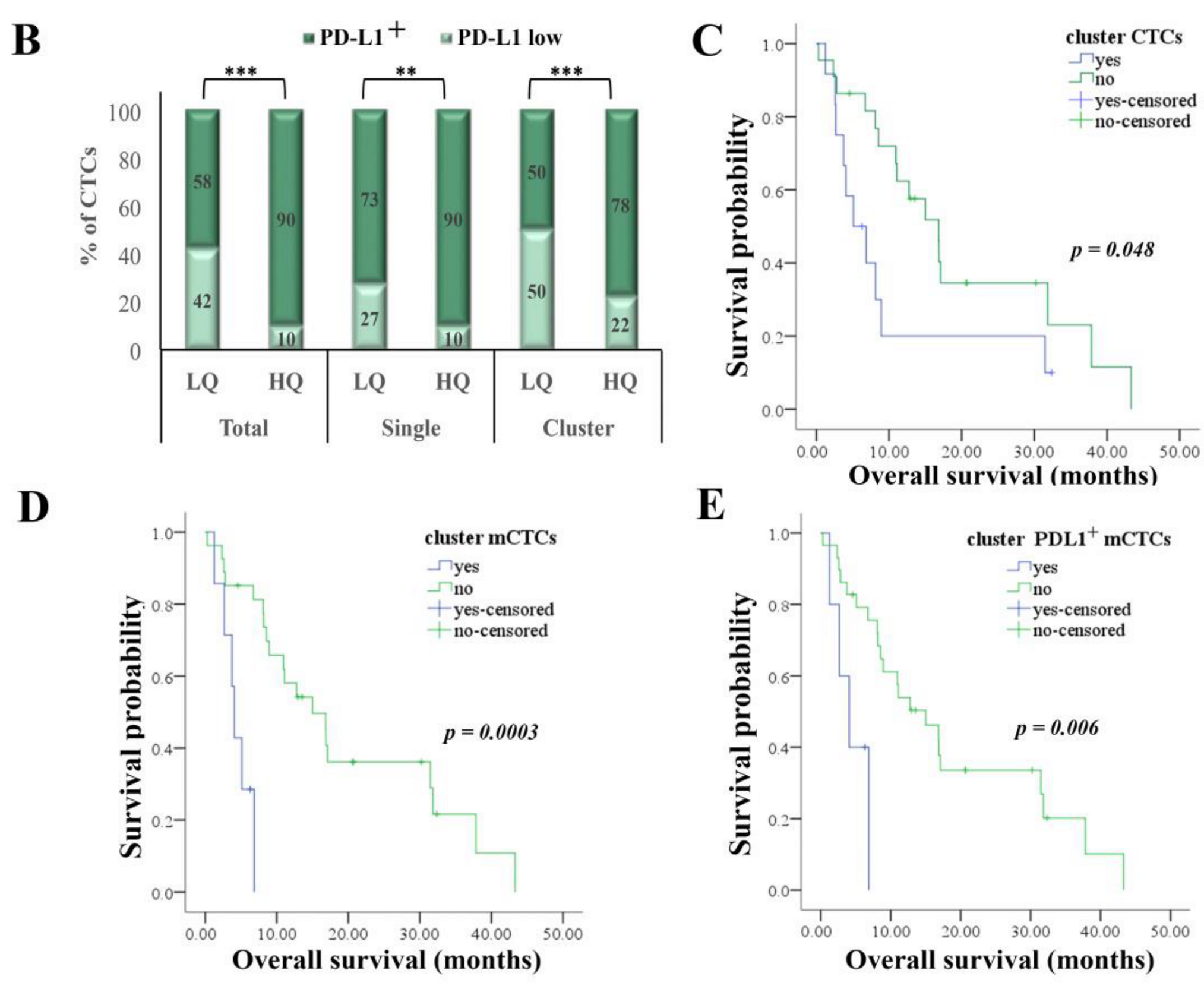
2.5. Correlation of CTC Monitoring with Clinicopathological Characteristics and Patient Outcome
3. Discussion
4. Material and Methods
4.1. Study Design
4.2. Cell Lines and Treatments
4.2.1. Culture Conditions
4.2.2. Cytospin Preparations of Cultured Cells
4.3. Confocal Microscopy
4.3.1. Patients and Cytospin Preparations
4.3.2. Quadruple Immunostaining
4.3.3. Identification and Phenotypic Characterization of CTCs
4.4. Image Analysis
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.M.M.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef]
- Liu, M.C.; Shields, P.G.; Warren, R.D.; Cohen, P.; Wilkinson, M.; Ottaviano, Y.L.; Rao, S.B.; Eng-Wong, J.; Seillier-Moiseiwitsch, F.; Noone, A.M.; et al. Circulating tumor cells: A useful predictor of treatment efficacy in metastatic breast cancer. J. Clin. Oncol. 2009, 27, 5153–5159. [Google Scholar] [CrossRef]
- Bidard, F.C.; Peeters, D.J.; Fehm, T.; Nolé, F.; Gisbert-Criado, R.; Mavroudis, D.; Grisanti, S.; Generali, D.; Garcia-Saenz, J.A.; Stebbing, J.; et al. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: A pooled analysis of individual patient data. Lancet Oncol. 2014, 15, 406–414. [Google Scholar] [CrossRef]
- Yu, M.; Bardia, A.; Wittner, B.S.; Stott, S.L.; Smas, M.E.; Ting, D.T.; Isakoff, S.J.; Ciciliano, J.C.; Wells, M.N.; Shah, A.M.; et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 2013, 339, 580–584. [Google Scholar] [CrossRef]
- Abreu, M.; Abalo, A.; Lago-lest, M.; Rafael, L. Analysis of a real-world cohort of metastatic breast cancer patients shows circulating tumor cell. Cancers 2020, 12, 1111. [Google Scholar]
- Hong, Y.; Fang, F.; Zhang, Q. Circulating tumor cell clusters: What we know and what we expect. Int. J. Oncol. 2016, 49, 2206–2216. [Google Scholar] [CrossRef] [PubMed]
- Amintas, S.; Bedel, A.; Moreau-Gaudry, F.; Boutin, J.; Buscail, L.; Merlio, J.P.; Vendrely, V.; Dabernat, S.; Buscail, E. Circulating tumor cell clusters: United we stand divided we fall. Int. J. Mol. Sci. 2020, 21, 2653. [Google Scholar] [CrossRef] [PubMed]
- Zeinali, M.; Lee, M.; Nadhan, A.; Mathur, A.; Hedman, C.; Lin, E.; Harouaka, R.; Wicha, M.S.; Zhao, L.; Palanisamy, N.; et al. High-throughput label-free isolation of heterogeneous circulating tumor cells and CTC clusters from non-small-cell lung cancer patients. Cancers 2020, 12, 127. [Google Scholar] [CrossRef] [PubMed]
- Lecharpentier, A.; Vielh, P.; Perez-Moreno, P.; Planchard, D.; Soria, J.C.; Farace, F. Detection of circulating tumour cells with a hybrid (epithelial/mesenchymal) phenotype in patients with metastatic non-small cell lung cancer. Br. J. Cancer 2011, 105, 1338–1341. [Google Scholar] [CrossRef] [PubMed]
- Aktas, B.; Tewes, M.; Fehm, T.; Hauch, S.; Kimmig, R.; Kasimir-Bauer, S. Stem cell and epithelial-mesenchymal transition markers are frequently overexpressed in circulating tumor cells of metastatic breast cancer patients. Breast Cancer Res. 2009, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, C.; Gradilone, A.; Naso, G.; Vincenzi, B.; Petracca, A.; Nicolazzo, C.; Palazzo, A.; Saltarelli, R.; Spremberg, F.; Cortesi, E.; et al. Epithelial-mesenchymal transition and stemness features in circulating tumor cells from breast cancer patients. Breast Cancer Res. Treat. 2011, 130, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Polioudaki, H.; Agelaki, S.; Chiotaki, R.; Politaki, E.; Mavroudis, D.; Matikas, A.; Georgoulias, V.; Theodoropoulos, P.A. Variable expression levels of keratin and vimentin reveal differential EMT status of circulating tumor cells and correlation with clinical characteristics and outcome of patients with metastatic breast cancer. BMC Cancer 2015, 15, 399. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.M.; Krebs, M.; Ward, T.; Sloane, R.; Priest, L.; Hughes, A.; Clack, G.; Ranson, M.; Blackhall, F.; Dive, C. Circulating tumor cells as a window on metastasis biology in lung cancer. Am. J. Pathol. 2011, 178, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Mani, S.A.; Guo, W.; Liao, M.; Eaton, E.N.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Campbell, L.L.; Polyak, K.; et al. EMT creates cells with the properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Morel, A.P.; Lièvre, M.; Thomas, C.; Hinkal, G.; Ansieau, S.; Puisieux, A. Generation of breast cancer stem cells through epithelial-mesenchymal transition. PLoS ONE 2008, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, M.A.; Stoupis, G.; Theodoropoulos, P.A.; Mavroudis, D.; Georgoulias, V.; Agelaki, S. Circulating tumor cells with stemness and epithelial-to-mesenchymal transition features are chemoresistant and predictive of poor outcome in metastatic breast cancer. Mol. Cancer Ther. 2019, 18, 437–447. [Google Scholar] [CrossRef]
- Polioudaki, H.; Chantziou, A.; Kalyvianaki, K.; Malamos, P.; Notas, G.; Mavroudis, D.; Kampa, M.; Castanas, E.; Theodoropoulos, P.A. Nuclear localization of PD-L1: Artifact or reality? Cell. Oncol. 2019, 42, 237–242. [Google Scholar] [CrossRef]
- Dong, H.; Strome, S.E.; Salomao, D.R.; Tamura, H.; Hirano, F.; Flies, D.B.; Roche, P.C.; Lu, J.; Zhu, G.; Tamada, K.; et al. Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nat. Med. 2002, 8, 793–800. [Google Scholar] [CrossRef]
- Garcia-diaz, A.; Shin, D.S.; Moreno, B.H.; Saco, J.; Escuin-ordinas, H.; Rodriguez, G.A.; Zaretsky, J.M.; Sun, L.; Wang, X.; Parisi, G.; et al. Interferon receptor signaling pathways regulating PD-L1 and PD-L2. Cell Rep. 2017, 19, 1189–1201. [Google Scholar] [CrossRef]
- Voutsadakis, I.A. Expression and function of immune ligand-receptor pairs in NK cells and cancer stem cells: Therapeutic implications. Cell. Oncol. 2018, 41, 107–121. [Google Scholar] [CrossRef]
- Papadaki, M.A.; Sotiriou, A.I.; Vasilopoulou, C.; Filika, M.; Aggouraki, D.; Tsoulfas, P.G.; Apostolopoulou, C.A.; Rounis, K.; Mavroudis, D.; Agelaki, S. Optimization of the enrichment of circulating tumor cells for downstream phenotypic analysis in patients with non-small cell lung cancer treated with anti-pd-1 immunotherapy. Cancers 2020, 12, 1556. [Google Scholar] [CrossRef] [PubMed]
- Kloten, V.; Lampignano, R.; Krahn, T.; Schlange, T. Circulating tumor cell PD-L1 expression as biomarker for therapeutic efficacy of immune checkpoint inhibition in NSCLC. Cells 2019, 8, 809. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, M.A.; Koutsopoulos, A.V.; Tsoulfas, P.G.; Lagoudaki, E.; Aggouraki, D.; Monastirioti, A.; Koutoulaki, C.; Apostolopoulou, C.A.; Merodoulaki, A.C.; Papadaki, C.; et al. Clinical relevance of immune checkpoints on circulating tumor cells in breast cancer. Cancers 2020, 12, 376. [Google Scholar] [CrossRef] [PubMed]
- Mazel, M.; Jacot, W.; Pantel, K.; Bartkowiak, K.; Topart, D.; Cayrefourcq, L.; Rossille, D.; Maudelonde, T.; Fest, T.; Alix-Panabières, C. Frequent expression of PD-L1 on circulating breast cancer cells. Mol. Oncol. 2015, 9, 1773–1782. [Google Scholar] [CrossRef]
- Po, J.W.; Ma, Y.; Balakrishna, B.; Brungs, D.; Azimi, F.; De Souza, P.; Becker, T.M. Immunomagnetic isolation of circulating melanoma cells and detection of PD-L1 status. PLoS ONE 2019, 14, e0211866. [Google Scholar] [CrossRef]
- Cheng, B.; Tong, G.; Wu, X.; Cai, W.; Li, Z.; Tong, Z.; He, L.; Yu, S.; Wang, S. Enumeration and characterization of circulating tumor cells and its application in advanced gastric cancer. Onco. Targets. Ther. 2019, 12, 7887–7896. [Google Scholar] [CrossRef]
- Yue, C.; Jiang, Y.; Li, P.; Wang, Y.; Xue, J.; Li, N.; Li, D.; Wang, R.; Dang, Y.; Hu, Z.; et al. Dynamic change of PD-L1 expression on circulating tumor cells in advanced solid tumor patients undergoing PD-1 blockade therapy. Oncoimmunology 2018, 7, 1–12. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhan, H. Communication between EMT and PD-L1 signaling: New insights into tumor immune evasion. Cancer Lett. 2020, 468, 72–81. [Google Scholar] [CrossRef]
- Mak, M.P.; Tong, P.; Diao, L.; Cardnell, R.J.; Gibbons, D.L.; William, W.N.; Skoulidis, F.; Parra, E.R.; Rodriguez-Canales, J.; Wistuba, I.I.; et al. A patient-derived, pan-cancer EMT signature identifies global molecular alterations and immune target enrichment following epithelial-to-mesenchymal transition. Clin. Cancer Res. 2016, 22, 609–620. [Google Scholar] [CrossRef]
- Dongre, A.; Rashidian, M.; Reinhardt, F.; Bagnato, A.; Keckesova, Z.; Ploegh, H.L.; Weinberg, R.A. Epithelial-to-mesenchymal transition contributes to immunosuppression in breast carcinomas. Cancer Res. 2017, 77, 3982–3989. [Google Scholar] [CrossRef]
- Manjunath, Y.; Upparahalli, S.V.; Avella, D.M.; Deroche, C.B.; Kimchi, E.T.; Staveley-O’carroll, K.F.; Smith, C.J.; Li, G.; Kaifi, J.T. PD-L1 expression with epithelial mesenchymal transition of circulating tumor cells is associated with poor survival in curatively resected non-small cell lung cancer. Cancers 2019, 11, 806. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, C.; Carpino, G.; Nicolazzo, C.; Gradilone, A.; Gianni, W.; Gelibter, A.; Gaudio, E.; Cortesi, E.; Gazzaniga, P. PD-L1 and epithelial-mesenchymal transition in circulating tumor cells from non-small cell lung cancer patients: A molecular shield to evade immune system? Oncoimmunology 2017, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Kamath, K.; Manna, T.; Okouneva, T.; Miller, H.P.; Davis, C.; Littlefield, B.A.; Wilson, L. The primary antimitotic mechanism of action of the synthetic halichondrin E7389 is suppression of microtubule growth. Mol. Cancer Ther. 2005, 4, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Okouneva, T.; Azarenko, O.; Wilson, L.; Littlefield, B.A.; Jordan, M.A. Inhibition of centromere dynamics by eribulin (E7389) during mitotic metaphase. Mol Cancer Ther 2008, 7, 2001–2003. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Wilson, L.; Azarenko, O.; Zhu, X.; Lewis, B.M.; Littlefield, B.A.; Jordan, M.A. Eribulin binds at microtubule ends to a single site on tubulin to suppress dynamic instability. Biochemistry 2010, 49, 1331–1337. [Google Scholar] [CrossRef]
- Towle, M.J.; Salvato, K.A.; Wels, B.F.; Aalfs, K.K.; Zheng, W.; Seletsky, B.M.; Zhu, X.; Lewis, B.M.; Kishi, Y.; Yu, M.J.; et al. Eribulin induces irreversible mitotic blockade: Implications of cell-based pharmacodynamics for in vivo efficacy under intermittent dosing conditions. Cancer Res. 2011, 71, 496–505. [Google Scholar] [CrossRef]
- Dybdal-Hargreaves, N.F.; Risinger, A.L.; Moobery, S.L. Eribulin mesylate: Mechanism of action of a unique microtubule targeting agent. Clin. Cancer Res. 2015, 21, 2445–2452. [Google Scholar] [CrossRef]
- Yoshida, T.; Ozawa, Y.; Kimura, T.; Sato, Y.; Kuznetsov, G.; Xu, S.; Uesugi, M.; Agoulnik, S.; Taylor, N.; Funahashi, Y.; et al. Eribulin mesilate suppresses experimental metastasis of breast cancer cells by reversing phenotype from epithelial-mesenchymal transition (EMT) to mesenchymal-epithelial transition (MET) states. Br. J. Cancer 2014, 110, 1497–1505. [Google Scholar] [CrossRef]
- Kashiwagi, S.; Asano, Y.; Goto, W.; Takada, K.; Takahashi, K.; Hatano, T.; Tanaka, S.; Takashima, T.; Tomita, S.; Motomura, H.; et al. Mesenchymal-epithelial transition and tumor vascular remodeling in eribulin chemotherapy for breast cancer. Anticancer Res. 2018, 38, 401–410. [Google Scholar] [CrossRef]
- Cortes, J.; Schöffski, P.; Littlefield, B.A. Multiple modes of action of eribulin mesylate: Emerging data and clinical implications. Cancer Treat. Rev. 2018, 70, 190–198. [Google Scholar] [CrossRef]
- Goto, W.; Kashiwagi, S.; Asano, Y.; Takada, K.; Morisaki, T.; Fujita, H.; Takashima, T.; Ohsawa, M.; Hirakawa, K.; Ohira, M. Eribulin promotes antitumor immune responses in patients with locally advanced or metastatic breast cancer. Anticancer Res. 2018, 38, 2929–2938. [Google Scholar] [CrossRef] [PubMed]
- Horimoto, Y.; Tokuda, E.; Murakami, F.; Uomori, T.; Himuro, T.; Nakai, K.; Orihata, G.; Iijima, K.; Togo, S.; Shimizu, H.; et al. Analysis of circulating tumour cell and the epithelial mesenchymal transition (EMT) status during eribulin-based treatment in 22 patients with metastatic breast cancer: A pilot study 11 Medical and Health Sciences 1112 Oncology and Carcinogenesis. J. Transl. Med. 2018, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Horimoto, Y.; Tokuda, E.; Murakami, F.; Uomori, T.; Himuro, T.; Nakai, K.; Orihata, G.; Iijima, K.; Saito, M. Impact of circulating tumour cells on survival of eribulin-treated patients with metastatic breast cancer. Med. Oncol. 2019, 36, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Alsuliman, A.; Colak, D.; Al-Harazi, O.; Fitwi, H.; Tulbah, A.; Al-Tweigeri, T.; Al-Alwan, M.; Ghebeh, H. Bidirectional crosstalk between PD-L1 expression and epithelial to mesenchymal transition: Significance in claudin-low breast cancer cells. Mol. Cancer 2015, 14, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Schott, D.S.; Pizon, M.; Pachmann, U.; Pachmann, K. Sensitive detection of PD-L1 expression on circulating epithelial tumor cells (CETCs) could be a potential biomarker to select patients for treatment with PD-1/PD-L1 inhibitors in early and metastatic solid tumors. Oncotarget 2017, 8, 72755–72772. [Google Scholar] [CrossRef]
- Gkountela, S.; Castro-Giner, F.; Szczerba, B.M.; Vetter, M.; Landin, J.; Scherrer, R.; Krol, I.; Scheidmann, M.C.; Beisel, C.; Stirnimann, C.U.; et al. Circulating tumor cell clustering shapes DNA methylation to enable metastasis seeding. Cell 2019, 176, 98–112.e14. [Google Scholar] [CrossRef]
- Aceto, N.; Bardia, A.; Miyamoto, D.T.; Donaldson, M.C.; Wittner, B.S.; Spencer, J.A.; Yu, M.; Pely, A.; Engstrom, A.; Zhu, H.; et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell 2014, 158, 1110–1122. [Google Scholar] [CrossRef]
- Terry, S.; Savagner, P.; Ortiz-Cuaran, S.; Mahjoubi, L.; Saintigny, P.; Thiery, J.P.; Chouaib, S. New insights into the role of EMT in tumor immune escape. Mol. Oncol. 2017, 11, 824–846. [Google Scholar] [CrossRef]


| Patients (n = 38) | n (%) |
|---|---|
| Age (years), median (range) | 61 (34–78) |
| Performance Status (PS) | |
| 0–1 | 30 (78.9) |
| 2 | 8 (21.1) |
| Menopausal status | |
| pre-menopausal | 13 (34.2) |
| post-menopausal | 25 (65.8) |
| Histology | |
| Ductal | 31 (81.6) |
| Lobular | 3 (7.9) |
| Mixed | 2 (5.3) |
| Other | 2 (5.3) |
| Stage at diagnosis | |
| I–III | 30 (78.9) |
| IV | 8 (21.1) |
| Subtype | |
| ER+ and/or PR+/HER2- | 20 (52.6) |
| Triple-negative | 12 (31.6) |
| HER2+ | 6 (15.8) |
| Surgery * | |
| Yes | 31 (81.6) |
| No | 7 (18.4) |
| Prior chemotherapy for metastatic disease | |
| Taxane-based | 16 (42.1) |
| Anthracycline-based | 1 (2.6) |
| Taxane/Anthracycline -based | 15 (39.5) |
| Other | 3 (7.9) |
| None | 3 (7.9) |
| Prior Trastuzumab for metastatic disease | |
| Yes | 6 (15.8) |
| No | 32 (84.2) |
| Prior Hormonotherapy for metastatic disease | |
| Yes | 20 (52.6) |
| No | 18 (47.4) |
| Disease sites | |
| 1–2 | 20 (52.6) |
| >2 | 18 (47.4) |
| Organs affected | |
| Bones | 24 (63.2) |
| Liver | 23 (60.5) |
| Lung | 18 (47.4) |
| Central Nervous System | 6 (15.8) |
| Lymph nodes | 14 (36.8) |
| Cutis | 5 (13.2) |
| Line of Eribulin treatment | |
| 1–2 | 13 (34.2) |
| >2 | 25 (65.8) |
| Response to treatment at first evaluation | |
| Partial response | 3 (7.9) |
| Stable disease | 9 (23.7) |
| Progressive disease | 20 (52.6) |
| Non-evaluable | 6 (15.8) |
| Disease status at the end of treatment | |
| Partial response | 1 (2.6) |
| Stable disease | 9 (23.7) |
| Progressive disease | 22 (57.9) |
| Non-evaluable | 6 (15.8) |
| Univariate Cox Regression Analysis | Progression-Free Survival (PFS) | Overall Survival (OS) | ||
|---|---|---|---|---|
| Covariates | HR (95% CI) | p Value | HR (95% CI) | p Value |
| Age (>median 59 yrs) | 1.420 (0.648–3.111) | 0.381 | 1.052 (0.420–2.635) | 0.914 |
| Performance status (0–1) | 0.503 (0.199–1.272) | 0.147 | 0.403 (0.147–1.110) | 0.079 |
| Menopausal status (pre) | 1.621 (0.770–3.410) | 0.203 | 1.326 (0.592–2.970) | 0.493 |
| Stage at diagnosis (I–III) | 1.506 (0.618–3.666) | 0.367 | 1.593 (0.540–4.701) | 0.399 |
| Molecular subtype of tumor | ||||
| ER+ and/or PR+/HER2- | 0.349 (0.156–0.778) | 0.010 * | 0.452 (0.200–1.024) | 0.057 |
| HER2-positive | 0.735 (0.250–2.160) | 0.575 | 0.448 (0.103–1.951) | 0.285 |
| Triple-negative | 2.866 (1.285–6.393) | 0.010 * | 2.211 (0.976–5.007) | 0.057 |
| No of organs affected (>2) | 1.188 (0.590–2.391) | 0.629 | 1.787 (0.758–4.211) | 0.184 |
| Metastatic sites | ||||
| Liver | 1.290 (0.634–2.625) | 0.483 | 1.256 (0.534–2.953) | 0.601 |
| Lung | 0.851 (0.424–1.710) | 0.651 | 0.826 (0.374–1.823) | 0.636 |
| Bones | 1.230 (0.574–2.637) | 0.594 | 1.507 (0.664–3.421) | 0.327 |
| Lymph nodes | 0.722 (0.336–1.549) | 0.402 | 0.486 (0.199–1.190) | 0.114 |
| CNS | 1.848 (0.624–5.475) | 0.268 | 5.099 (1.476–17.616) | 0.010 * |
| Skin | 1.096 (0.378–3.178) | 0.867 | 3.399 (0.919–12.569) | 0.067 |
| Line of eribulin treatment (>2) | 1.139 (0.554–2.341) | 0.724 | 0.804 (0.352–1.833) | 0.603 |
| Νew visceral metastases | 2.670 (0.800–8.906) | 0.110 | 1.390 (0.526–3.676) | 0.507 |
| cluster CTCs | 1.119 (0.537–2.334) | 0.764 | 2.266 (0.8986–5.211) | 0.054 |
| cluster mCTCs | 1.728 (0.720–4.147) | 0.221 | 7.273 (2.103–25.147) | 0.002 * |
| cluster PD-L1+ mCTCs | 1.876 (0.705–4.995) | 0.208 | 4.964 (1.418–17.376) | 0.012 * |
| Multivariate Cox Regression Analysis | Overall Survival (OS) | |
|---|---|---|
| Covariates | HR (95% CI) | p Value |
| Central nervous system metastasis | 2.121 (0.502–8.960) | 0.306 |
| Cluster mCTCs | 5.145 (1.240–21.350) | 0.024 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polioudaki, H.; Mala, A.; Gkimprixi, E.; Papadaki, M.A.; Chantziou, A.; Tzardi, M.; Mavroudis, D.; Agelaki, S.; Theodoropoulos, P.A. Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome. Cancers 2020, 12, 3735. https://doi.org/10.3390/cancers12123735
Polioudaki H, Mala A, Gkimprixi E, Papadaki MA, Chantziou A, Tzardi M, Mavroudis D, Agelaki S, Theodoropoulos PA. Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome. Cancers. 2020; 12(12):3735. https://doi.org/10.3390/cancers12123735
Chicago/Turabian StylePolioudaki, Hara, Anastasia Mala, Eleni Gkimprixi, Maria A. Papadaki, Amanda Chantziou, Maria Tzardi, Dimitris Mavroudis, Sofia Agelaki, and Panayiotis A. Theodoropoulos. 2020. "Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome" Cancers 12, no. 12: 3735. https://doi.org/10.3390/cancers12123735
APA StylePolioudaki, H., Mala, A., Gkimprixi, E., Papadaki, M. A., Chantziou, A., Tzardi, M., Mavroudis, D., Agelaki, S., & Theodoropoulos, P. A. (2020). Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome. Cancers, 12(12), 3735. https://doi.org/10.3390/cancers12123735









