Antiplatelet Therapy Improves the Prognosis of Patients with Hepatocellular Carcinoma
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
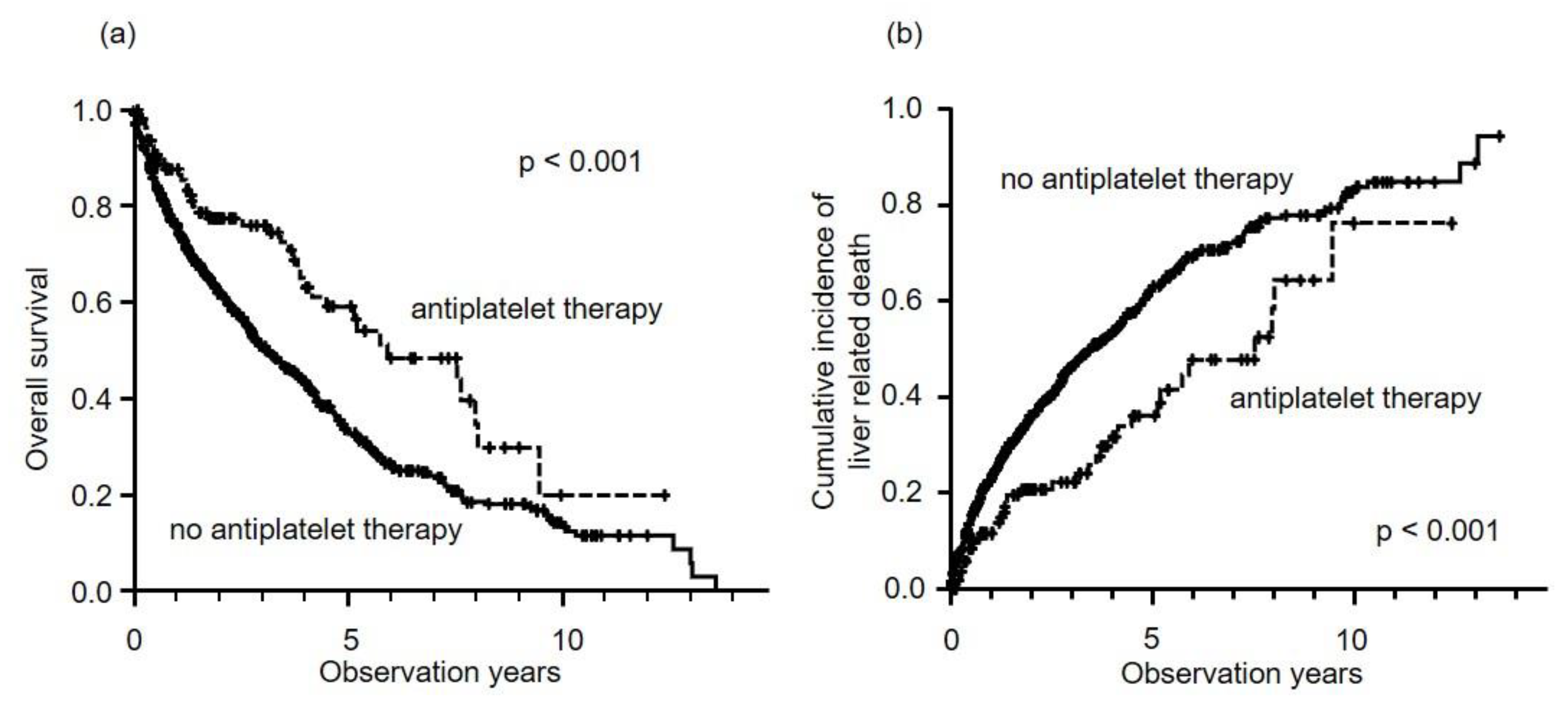
2.2. Prognosis of HCC Patients
2.3. Propensity Score Matching
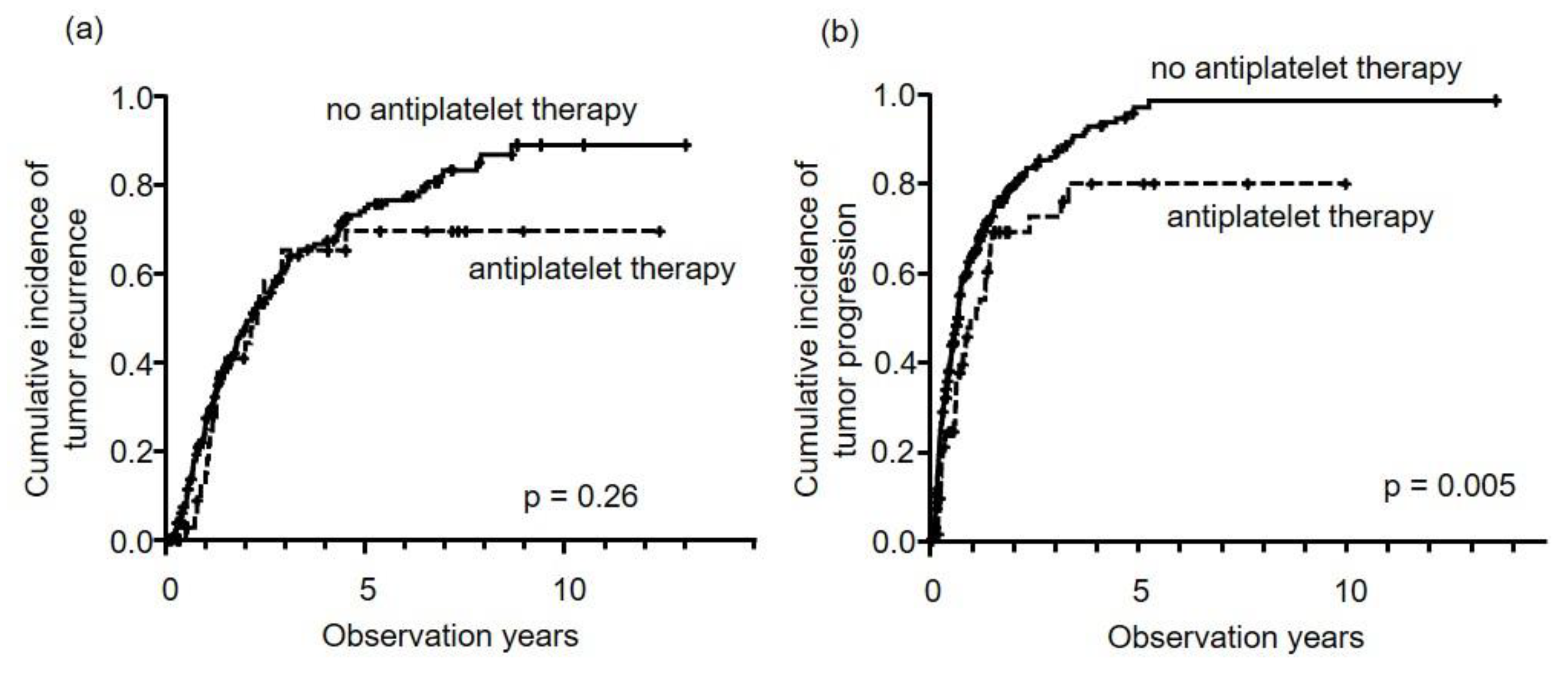
2.4. HCC Recurrence and Time to Progression
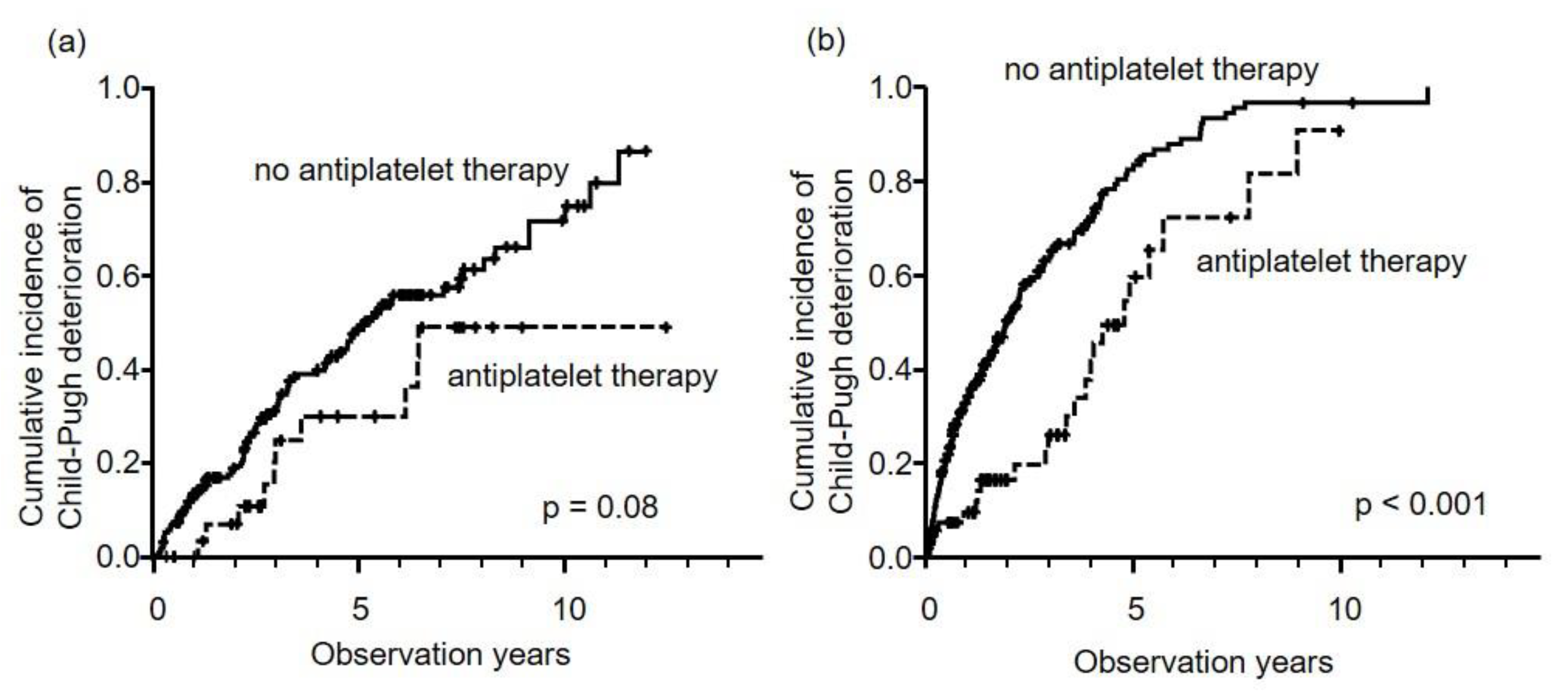
2.5. Deterioration of Liver Function
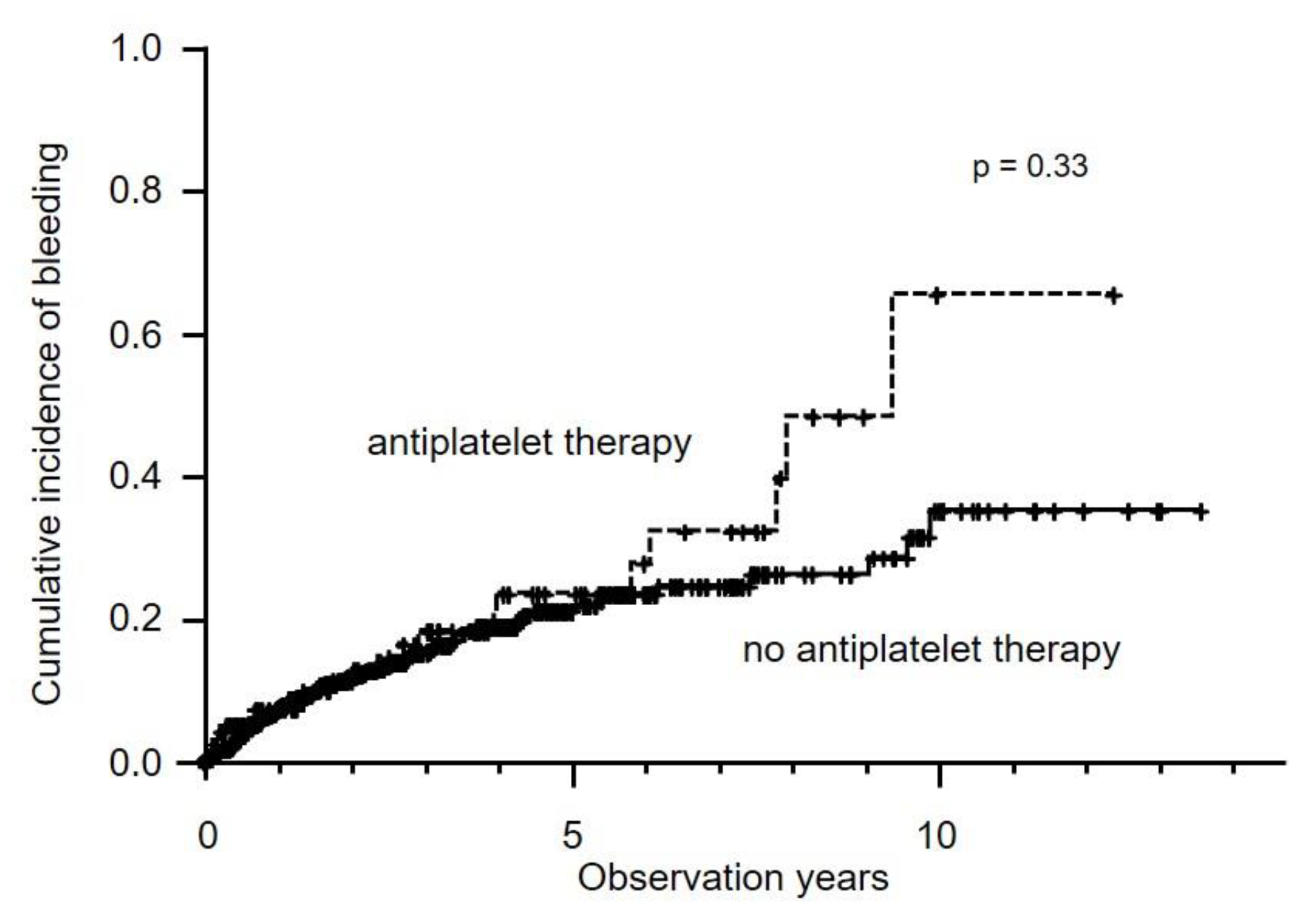
2.6. Hemorrhagic Events
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Definition of Antiplatelet Therapy
4.3. Endpoint of this Study
4.4. Statistical Analysis
4.5. Propensity Score Matching
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Yasui, Y.; Tsuchiya, K.; Kurosaki, M.; Takeguchi, T.; Takeguchi, Y.; Okada, M.; Wang, W.; Kubota, Y.; Goto, T.; Komiyama, Y.; et al. Up-to-seven criteria as a useful predictor for tumor downstaging to within Milan criteria and Child-Pugh grade deterioration after initial conventional transarterial chemoembolization. Hepatol. Res. 2018, 48, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, U.; Dennis, B.B.; Li, A.A.; Cholankeril, G.; Kim, D.; Khan, M.A.; Ahmed, A. Use of anti-platelet agents in the prevention of hepatic fibrosis in patients at risk for chronic liver disease: A systematic review and meta-analysis. Hepatol. Int. 2019, 13, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-Y.; Hsu, Y.-C.; Tseng, H.-C.; Yu, S.-H.; Lin, J.-T.; Wu, M.-S.; Wu, C.-Y. Association of daily aspirin therapy with risk of hepatocellular carcinoma in patients with chronic hepatitis B. JAMA Intern. Med. 2019, 179, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Chung, G.E.; Lee, J.-H.; Oh, S.; Nam, J.Y.; Chang, Y.; Cho, H.; Ahn, H.; Cho, Y.Y.; Yoo, J.-J.; et al. Antiplatelet therapy and the risk of hepatocellular carcinoma in chronic hepatitis B patients on antiviral treatment. Hepatology 2017, 66, 1556–1569. [Google Scholar] [CrossRef] [PubMed]
- Hwang, I.C.; Chang, J.; Kim, K.; Park, S.M. Aspirin use and risk of hepatocellular carcinoma in a national cohort study of korean adults. Sci. Rep. 2018, 8, 4968. [Google Scholar] [CrossRef]
- Simon, T.G.; Duberg, A.S.; Aleman, S.; Chung, R.T.; Chan, A.T.; Ludvigsson, J.F. Association of aspirin with hepatocellular carcinoma and liver-related mortality. N. Engl. J. Med. 2020, 382, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Muhlestein, J.B. Effect of antiplatelet therapy on inflammatory markers in atherothrombotic patients. Thromb. Haemost. 2010, 103, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Brouquet, A.; Benoist, S.; Julie, C.; Penna, C.; Beauchet, A.; Rougier, P.; Nordlinger, B. Risk factors for chemotherapy-associated liver injuries: A multivariate analysis of a group of 146 patients with colorectal metastases. Surgery 2009, 145, 362–371. [Google Scholar] [CrossRef]
- Brown, J.R.; DuBois, R.N. COX-2: A molecular target for colorectal cancer prevention. J. Clin. Oncol. 2005, 23, 2840–2855. [Google Scholar] [CrossRef]
- Kopp, E.; Ghosh, S. Inhibition of NF-kappa B by sodium salicylate and aspirin. Science 1994, 265, 956–959. [Google Scholar] [CrossRef]
- Schwenger, P.; Bellosta, P.; Vietor, I.; Basilico, C.; Skolnik, E.Y.; Vilcek, J. Sodium salicylate induces apoptosis via p38 mitogen-activated protein kinase but inhibits tumor necrosis factor-induced c-Jun N-terminal kinase/stress-activated protein kinase activation. Proc. Natl. Acad. Sci. USA 1997, 94, 2869–2873. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.T.; Ogino, S.; Fuchs, C.S. Aspirin use and survival after diagnosis of colorectal cancer. JAMA 2009, 302, 649–658. [Google Scholar] [CrossRef]
- Zhou, Y.; Simmons, J.; Jordan, C.D.; Sonbol, M.B.; Maihle, N.; Tang, S.C. Aspirin treatment effect and association with PIK3CA mutation in breast cancer: A biomarker analysis. Clin. Breast Cancer 2019, 19, 354–362.e7. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, M. Aspirin and clopidogrel: Efficacy, safety, and the issue of drug resistance. Arter. Thromb. Vasc. Biol. 2004, 24, 1980–1987. [Google Scholar] [CrossRef]
- Rodriguez-Miguel, A.; Garcia-Rodriguez, L.A.; Gil, M.; Montoya, H.; Rodriguez-Martin, S.; de Abajo, F.J. Clopidogrel and low-dose aspirin, alone or together, reduce risk of colorectal cancer. Clin. Gastroenterol. Hepatol. 2019, 17, 2024–2033.e2. [Google Scholar] [CrossRef] [PubMed]
- Mezouar, S.; Darbousset, R.; Dignat-George, F.; Panicot-Dubois, L.; Dubois, C. Inhibition of platelet activation prevents the P-selectin and integrin-dependent accumulation of cancer cell microparticles and reduces tumor growth and metastasis in vivo. Int. J. Cancer 2015, 136, 462–475. [Google Scholar] [CrossRef]
- Symeonidis, D.; Koukoulis, G.; Christodoulidis, G.; Mamaloudis, I.; Chatzinikolaou, I.; Tepetes, K. Impact of antiplatelet treatment on colorectal cancer staging characteristics. World J. Gastrointest. Endosc. 2012, 4, 409–413. [Google Scholar] [CrossRef]
- Choe, K.S.; Correa, D.; Jani, A.B.; Liauw, S.L. The use of anticoagulants improves biochemical control of localized prostate cancer treated with radiotherapy. Cancer 2010, 116, 1820–1826. [Google Scholar] [CrossRef]
- Starley, B.Q.; Calcagno, C.J.; Harrison, S.A. Nonalcoholic fatty liver disease and hepatocellular carcinoma: A weighty connection. Hepatology 2010, 51, 1820–1832. [Google Scholar] [CrossRef]
- Tamaki, N.; Higuchi, M.; Kurosaki, M.; Kirino, S.; Osawa, L.; Watakabe, K.; Wang, W.; Okada, M.; Shimizu, T.; Takaura, K.; et al. Wisteria floribunda agglutinin-positive mac-2 binding protein as an age-independent fibrosis marker in nonalcoholic fatty liver disease. Sci. Rep. 2019, 9, 10109. [Google Scholar] [CrossRef] [PubMed]
- Nishio, T.; Hatano, E.; Sakurai, T.; Taura, K.; Okuno, M.; Kasai, Y.; Seo, S.; Yasuchika, K.; Mori, A.; Kaido, T.; et al. Impact of hepatic steatosis on disease-free survival in patients with non-B non-C hepatocellular carcinoma undergoing hepatic resection. Ann. Surg. Oncol. 2015, 22, 2226–2234. [Google Scholar] [CrossRef]
- Fujita, K.; Nozaki, Y.; Wada, K.; Yoneda, M.; Endo, H.; Takahashi, H.; Iwasaki, T.; Inamori, M.; Abe, Y.; Kobayashi, N.; et al. Effectiveness of antiplatelet drugs against experimental non-alcoholic fatty liver disease. Gut 2008, 57, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Chávez, E.; Castro-Sánchez, L.; Shibayama, M.; Tsutsumi, V.; Salazar, E.P.; Moreno, M.G.; Muriel, P. Effects of acetyl salycilic acid and ibuprofen in chronic liver damage induced by CCl4. J. Appl. Toxicol. 2012, 32, 51–59. [Google Scholar] [CrossRef]
- Lee, P.-C.; Yeh, C.-M.; Hu, Y.-W.; Chen, C.-C.; Liu, C.-J.; Su, C.-W.; Huo, T.-I.; Huang, Y.-H.; Chao, Y.; Chen, T.-J.; et al. Antiplatelet therapy is associated with a better prognosis for patients with hepatitis B virus-related hepatocellular carcinoma after liver resection. Ann. Surg. Oncol. 2016, 23, 874–883. [Google Scholar] [CrossRef]
- Boas, F.E.; Brown, K.T.; Ziv, E.; Yarmohammadi, H.; Sofocleous, C.T.; Erinjeri, J.P.; Harding, J.J.; Solomon, S.B. Aspirin is associated with improved liver function after embolization of hepatocellular carcinoma. AJR Am. J. Roentgenol. 2019, 213, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, N.; Rani, B.; Gerwins, P.; Heindryckx, F. Platelets as Key Factors in Hepatocellular Carcinoma. Cancers 2019, 11, 1022. [Google Scholar] [CrossRef] [PubMed]
- Koga, H.; Sakisaka, S.; Ohishi, M.; Kawaguchi, T.; Taniguchi, E.; Sasatomi, K.; Harada, M.; Kusaba, T.; Tanaka, M.; Kimura, R.; et al. Expression of cyclooxygenase-2 in human hepatocellular carcinoma: Relevance to tumor dedifferentiation. Hepatology 1999, 29, 688–696. [Google Scholar] [CrossRef]
- Liang, B.; Zheng, C.-S.; Feng, G.-S.; Wu, H.-P.; Wang, Y.; Zhao, H.; Qian, J.; Liang, H.-M. Correlation of hypoxia-inducible factor 1alpha with angiogenesis in liver tumors after transcatheter arterial embolization in an animal model. Cardiovasc. Interv. Radiol. 2010, 33, 806–812. [Google Scholar] [CrossRef]
- Rahman, M.; Selvarajan, K.; Hasan, M.R.; Chan, A.P.; Jin, C.; Kim, J.; Chan, S.K.; Le, N.D.; Kim, Y.-B.; Tai, I.T. Inhibition of COX-2 in colon cancer modulates tumor growth and MDR-1 expression to enhance tumor regression in therapy-refractory cancers in vivo. Neoplasia 2012, 14, 624–633. [Google Scholar] [CrossRef]
- Xie, Z.Y.; Liu, M.S.; Zhang, C.; Cai, P.C.; Xiao, Z.H.; Wang, F.F. Aspirin enhances the sensitivity of hepatocellular carcinoma side population cells to doxorubicin via miR-491/ABCG2. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef]
- Dong, H.; Liu, G.; Jiang, B.; Guo, J.; Tao, G.; Yiu, W.; Zhou, J.; Li, G. The effects of aspirin plus cisplatin on SGC7901/CDDP cells in vitro. Biomed. Rep. 2014, 2, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Khan, P.; Bhattacharya, A.; Sengupta, D.; Banerjee, S.; Adhikary, A.; Das, T. Aspirin enhances cisplatin sensitivity of resistant non-small cell lung carcinoma stem-like cells by targeting mTOR-Akt axis to repress migration. Sci. Rep. 2019, 9, 16913. [Google Scholar] [CrossRef] [PubMed]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR‘ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef]
| Factor | Antiplatelet Therapy (n = 111) | No Antiplatelet Therapy (n = 661) | p-Value |
|---|---|---|---|
| Age (IQR), years | 77 (69–82) | 71 (64–78) | <0.01 |
| Male, % | 96 (86.5) | 455 (68.9) | <0.01 |
| Etiology, HBV/HCV/NBNC | 14/46/51 | 84/345/231 | 0.13 |
| Albumin (IQR), g/dL | 3.9 (3.5–4.1) | 3.6 (3.2–4.0) | <0.01 |
| Bilirubin (IQR), mg/dL | 0.6 (0.5–0.8) | 0.8 (0.6–1.2) | <0.01 |
| ALT (IQR), IU/L | 29 (24–59) | 36 (19–47) | <0.01 |
| PLT (IQR), ×104/μL | 15.9 (11.4–19.0) | 12.5 (8.7–17.8) | <0.01 |
| Prothrombin time (IQR), % | 85.2 (78.9–96.7) | 79.3 (68.2–91.5) | <0.01 |
| Child–Pugh grade (A/B/C) | 97/11/3 | 450/176/35 | <0.01 |
| Stage, I/II/III/IVa/IVb | 18/55/29/2/7 | 125/269/160/65/42 | 0.053 |
| Tumor number (IQR) | 1 (1–2) | 1(1–3) | 0.01 |
| Tumor size (IQR), cm | 3.3 (2.2–5.5) | 2.8 (1.8–5.3) | 0.05 |
| Vascular invasion (%) | 6 (5.4) | 79 (12.0) | 0.06 |
| Extrahepatic metastasis (%) | 7 (6.3) | 43 (6.5) | 1.0 |
| AFP (IQR), ng/mL | 8.8 (4.6–36.3) | 15.3 (5.6–121.8) | 0.01 |
| DCP (IQR), mAU/mL | 113.0 (29.5–951.5) | 71.5 (25.0–845.0) | 0.22 |
| First treatment, curative/TACE/others/BSC | 37/66/5/3 | 235/357/24/45 | 0.33 |
| Factor | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Age, years | ||||||
| ≤70 | 1 | |||||
| >70 | 1.065 | 0.873–1.30 | 0.533 | |||
| Gender | ||||||
| Male | 1 | |||||
| Female | 0.979 | 0.796–1.20 | 0.841 | |||
| Child–Pugh score | 1.48 | 1.41–1.56 | <0.001 | 1.37 | 1.29–1.47 | <0.001 |
| HCC stage | ||||||
| I | 1 | 1 | ||||
| II | 1.33 | 0.988–1.78 | <0.001 | 1.20 | 0.847–1.71 | 0.303 |
| III | 2.75 | 2.03–3.73 | <0.001 | 2.05 | 1.40–3.00 | <0.001 |
| IVa | 13.8 | 9.45–20.0 | <0.001 | 6.36 | 3.91–10.3 | <0.001 |
| IVb | 21.3 | 14.1–32.1 | <0.001 | 8.77 | 5.2–14.8 | <0.001 |
| AFP, ng/mL | ||||||
| ≤200 | 1 | 1 | ||||
| >200 | 2.90 | 2.33–3.60 | <0.001 | 1.36 | 1.05–1.77 | 0.019 |
| DCP, mAU/mL | ||||||
| ≤400 | 1 | 1 | ||||
| >400 | 3.01 | 2.47–3.66 | <0.001 | 1.58 | 1.22–2.05 | <0.001 |
| First treatment | ||||||
| Curative therapy | 1 | 1 | ||||
| TACE | 3.15 | 2.46–4.01 | <0.001 | 1.85 | 1.40–2.45 | <0.001 |
| Others | 28.7 | 20.2–40.8 | <0.001 | 4.24 | 2.70–6.66 | <0.001 |
| Antiplatelet therapy | ||||||
| Without | 1 | 1 | ||||
| With | 0.550 | 0.398–0.758 | <0.001 | 0.640 | 0.445–0.926 | 0.002 |
| Factor | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Age, years | ||||||
| ≤70 | 1 | |||||
| >70 | 0.916 | 0.730–1.15 | 0.45 | |||
| Gender | ||||||
| Male | 1 | |||||
| Female | 1.22 | 0.943–1.57 | 0.13 | |||
| Child–Pugh score | 1.04 | 0.957–1.13 | 0.352 | |||
| HCC stage | ||||||
| I | 1 | 1 | ||||
| II | 1.39 | 0.895–2.16 | 0.143 | 1.23 | 0.774–1.960 | 0.379 |
| III | 3.06 | 1.96–4.77 | <0.001 | 2.47 | 1.52–4.01 | <0.001 |
| IVa | 6.63 | 3.76–11.7 | <0.001 | 5.59 | 3.00–10.4 | <0.001 |
| IVb | 13.2 | 7.18–24.4 | <0.001 | 8.83 | 4.47–17.4 | <0.001 |
| AFP, ng/mL | ||||||
| ≤200 | 1 | 1 | ||||
| >200 | 1.63 | 1.26–2.11 | <0.001 | 0.959 | 0.720–1.28 | 0.774 |
| DCP, mAU/mL | ||||||
| ≤400 | 1 | 1 | ||||
| >400 | 2.23 | 1.76–2.83 | <0.001 | 1.63 | 1.25–2.12 | <0.001 |
| Antiplatelet therapy | ||||||
| Without | 1 | 1 | ||||
| With | 0.622 | 0.446–0.869 | 0.005 | 0.690 | 0.484–0.983 | 0.040 |
| Factor | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Age, years | ||||||
| ≤70 | 1 | 1 | ||||
| >70 | 1.32 | 1.04–1.67 | 0.023 | 1.69 | 1.29–2.20 | <0.001 |
| Gender | ||||||
| Male | 1 | |||||
| Female | 0.81 | 0.633–1.04 | 0.094 | |||
| Child–Pugh score | ||||||
| 5 | 1 | 1 | ||||
| 6 | 2.82 | 2.23–3.56 | <0.001 | 3.46 | 2.69–4.46 | <0.001 |
| HCC stage | ||||||
| I | 1 | 1 | ||||
| II | 1.15 | 0.834–1.58 | 0.396 | 0.784 | 0.555–1.11 | 0.169 |
| III | 2.26 | 1.60–3.18 | <0.001 | 1.36 | 0.925–1.99 | 0.119 |
| IVa | 12.1 | 7.56–19.3 | <0.001 | 4.56 | 2.61–7.98 | <0.001 |
| IVb | 13.3 | 7.78–22.8 | <0.001 | 5.93 | 3.02–11.7 | <0.001 |
| AFP, ng/mL | ||||||
| ≤200 | 1 | 1 | ||||
| >200 | 2.51 | 1.90–3.30 | <0.001 | 1.80 | 1.31–2.47 | <0.001 |
| DCP, mAU/mL | ||||||
| ≤400 | 1 | 1 | ||||
| >400 | 2.17 | 1.71–2.77 | <0.001 | 1.55 | 1.17–2.06 | <0.001 |
| First treatment | ||||||
| Curative therapy | 1 | 1 | ||||
| TACE | 2.54 | 1.99–3.26 | <0.001 | 1.77 | 1.34–2.36 | <0.001 |
| Others | 24.3 | 13.9–42.4 | <0.001 | 4.57 | 2.38–8.81 | <0.001 |
| Antiplatelet therapy | ||||||
| Without | 1 | 1 | ||||
| With | 0.524 | 0.369–0.746 | <0.001 | 0.467 | 0.321–0.679 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayashi, T.; Shibata, M.; Oe, S.; Miyagawa, K.; Honma, Y.; Harada, M. Antiplatelet Therapy Improves the Prognosis of Patients with Hepatocellular Carcinoma. Cancers 2020, 12, 3215. https://doi.org/10.3390/cancers12113215
Hayashi T, Shibata M, Oe S, Miyagawa K, Honma Y, Harada M. Antiplatelet Therapy Improves the Prognosis of Patients with Hepatocellular Carcinoma. Cancers. 2020; 12(11):3215. https://doi.org/10.3390/cancers12113215
Chicago/Turabian StyleHayashi, Tsuguru, Michihiko Shibata, Shinji Oe, Koichiro Miyagawa, Yuichi Honma, and Masaru Harada. 2020. "Antiplatelet Therapy Improves the Prognosis of Patients with Hepatocellular Carcinoma" Cancers 12, no. 11: 3215. https://doi.org/10.3390/cancers12113215
APA StyleHayashi, T., Shibata, M., Oe, S., Miyagawa, K., Honma, Y., & Harada, M. (2020). Antiplatelet Therapy Improves the Prognosis of Patients with Hepatocellular Carcinoma. Cancers, 12(11), 3215. https://doi.org/10.3390/cancers12113215




