Impact of FAK Expression on the Cytotoxic Effects of CIK Therapy in Triple-Negative Breast Cancer
Abstract
1. Introduction
2. Results
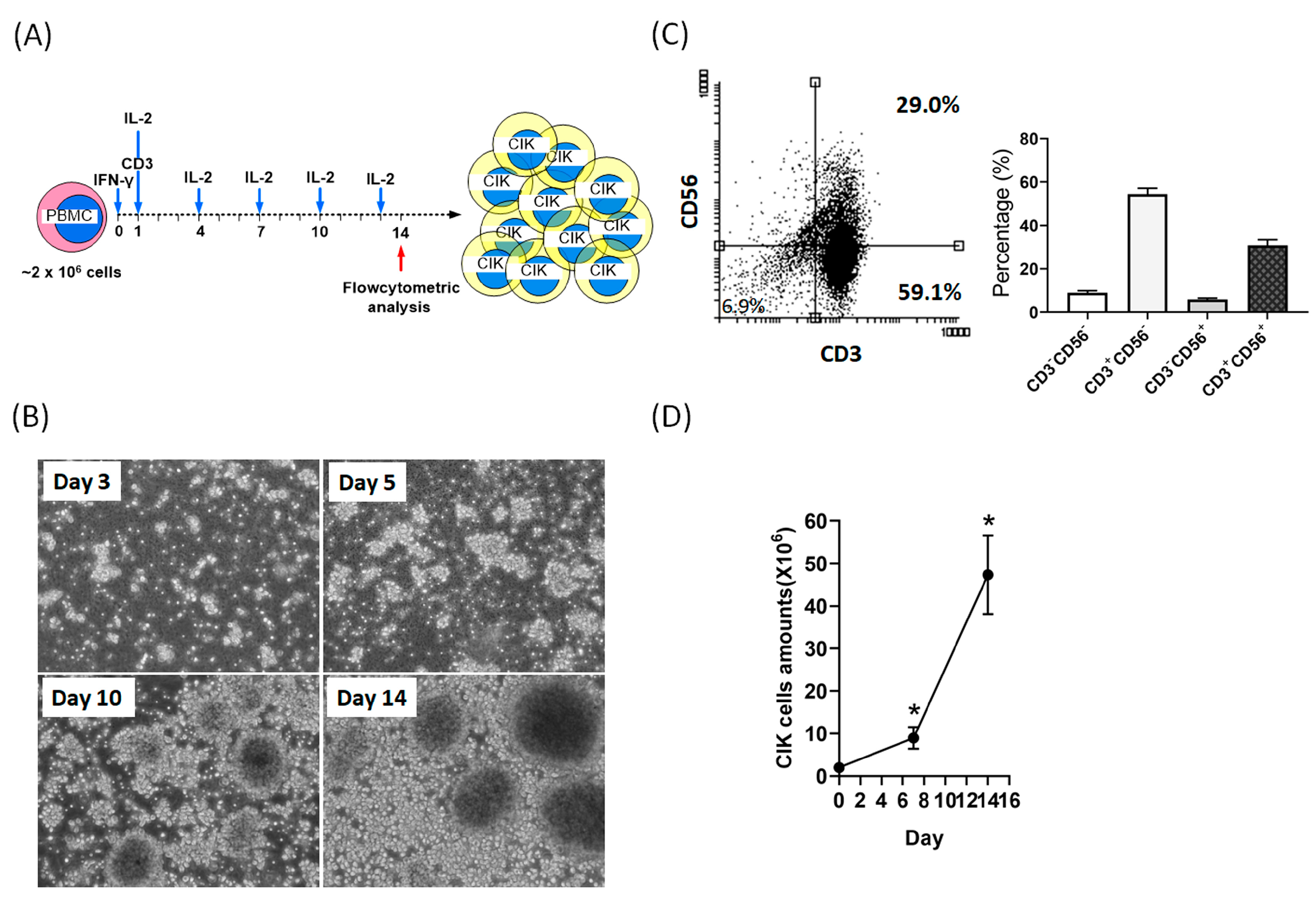
2.1. Activation and Expansion of CIK Cells
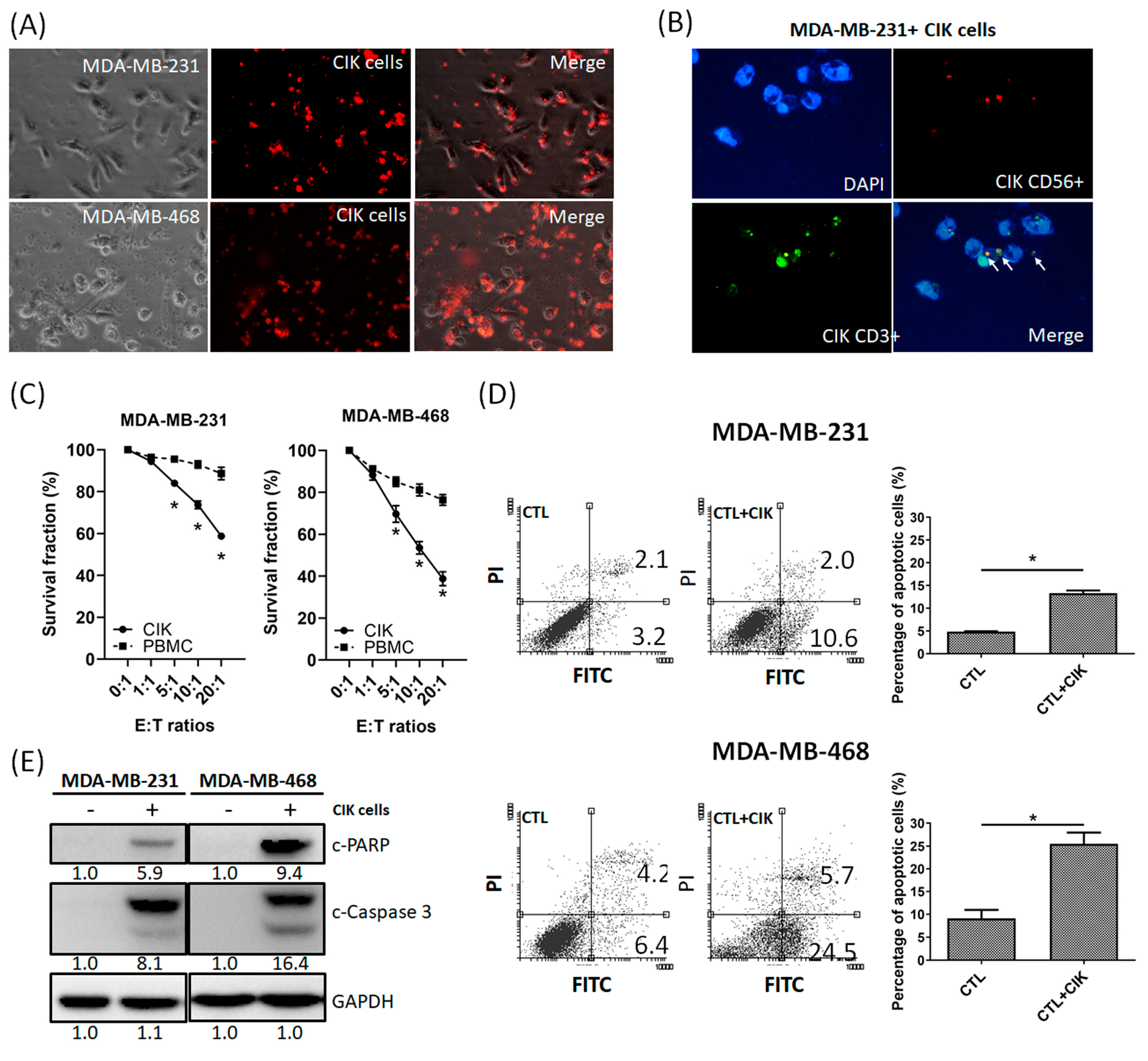
2.2. Anti-Tumor Effects of CIK Cells on MDA-MB-231 and MDA-MB-468 TNBC Cells
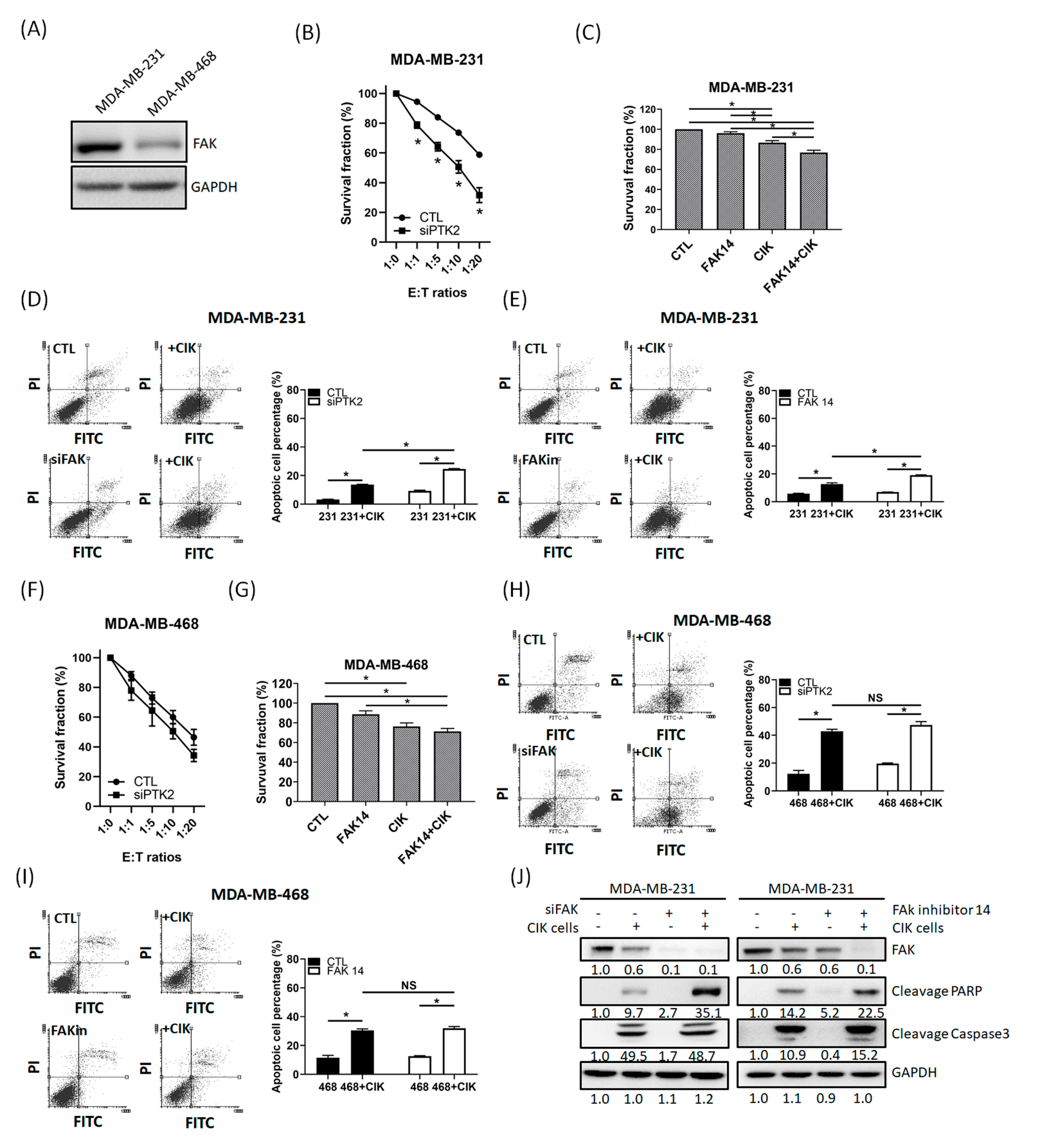
2.3. FAK Inhibition of TNBC Cells Promotes the Cytotoxic Effects of CIK Cells towards TNBC Cells
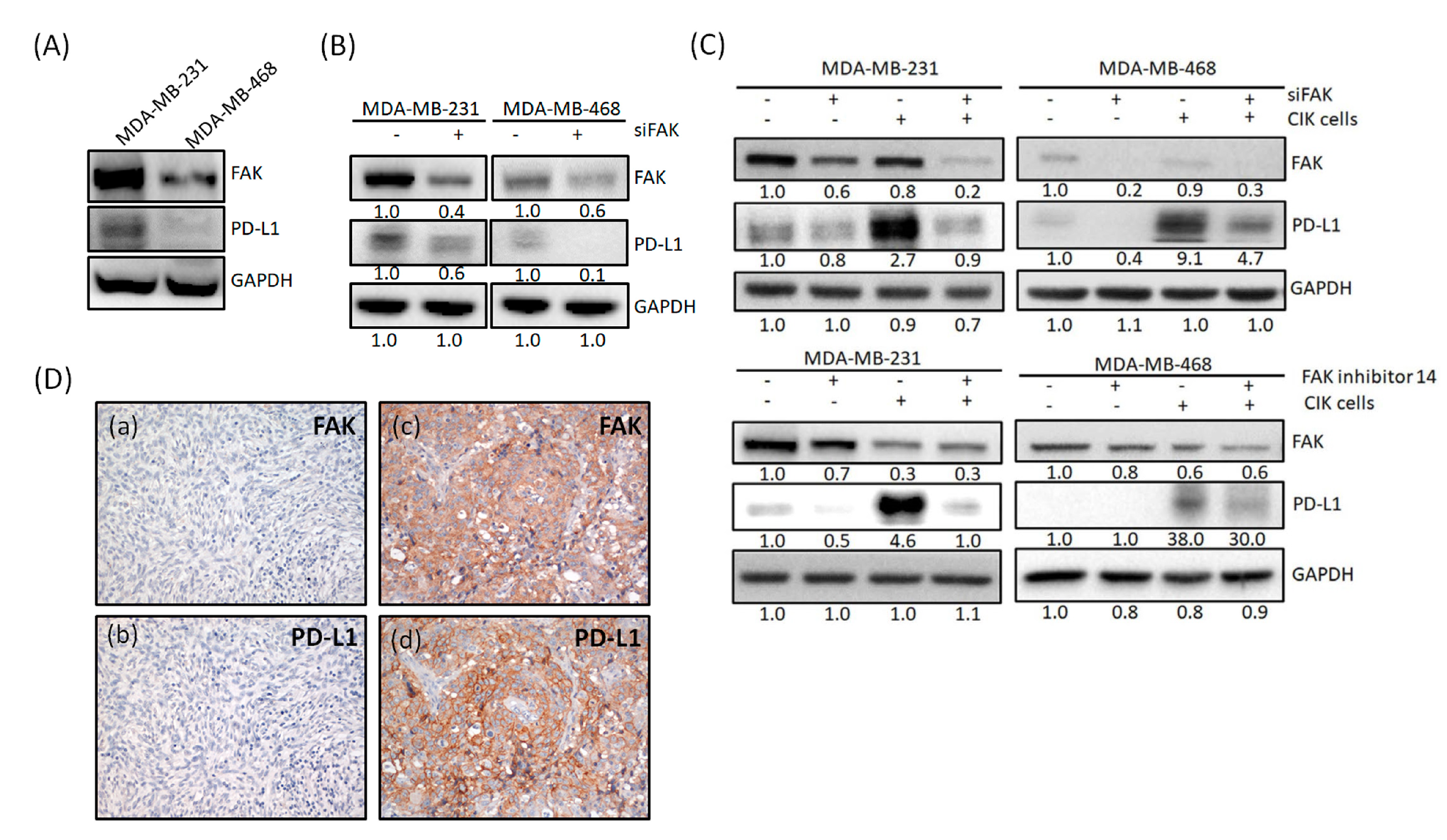
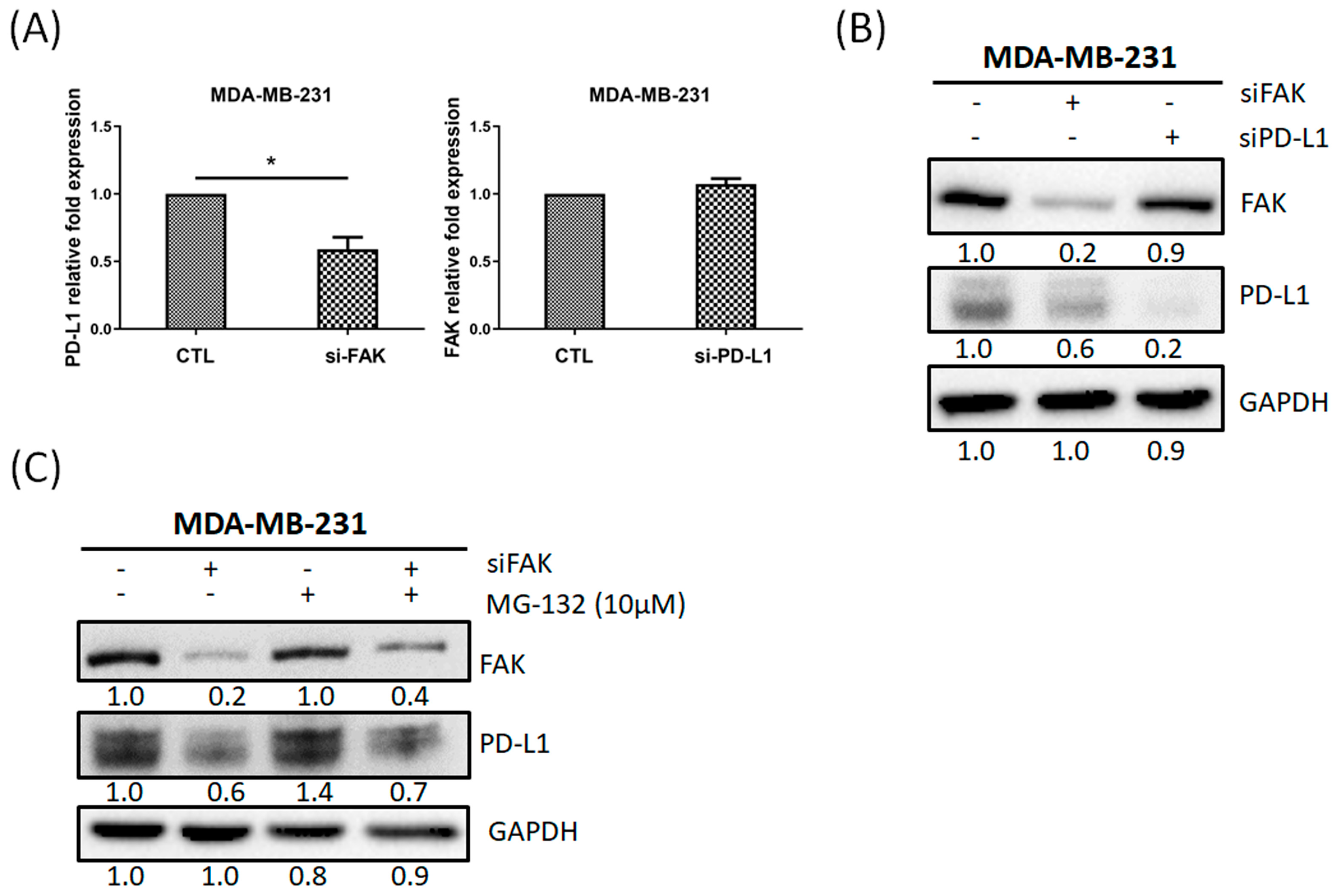
2.4. FAK Expression Is Correlated with PD-L1 Expression
2.5. PD-L1 Is Regulated through Gene Expression of FAK in TNBC Cells
3. Discussion
4. Materials and Methods
4.1. Blood Collection
4.2. CIK Cell Preparation
4.3. Cell Lines
4.4. Flow Cytometry Analysis
4.5. Cell Proliferation Assay
4.6. Immunofluorescent (IFC) Staining
4.7. RNA Interference, FAK Inhibitor and Proteasome Inhibitor
4.8. Quantitative Real-Time Polymerase Chain Reaction (q-PCR)
4.9. Western Blotting
4.10. Human Specimens
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Ou-Yang, F.; Pan, M.R.; Chang, S.J.; Wu, C.C.; Fang, S.Y.; Li, C.L.; Hou, M.F.; Luo, C.W. Identification of CHD4-beta1 integrin axis as a prognostic marker in triple-negative breast cancer using next-generation sequencing and bioinformatics. Life Sci. 2019, 238, 116963. [Google Scholar] [CrossRef]
- Kennecke, H.; Yerushalmi, R.; Woods, R.; Cheang, M.C.; Voduc, D.; Speers, C.H.; Nielsen, T.O.; Gelmon, K. Metastatic behavior of breast cancer subtypes. J. Clin. Oncol. 2010, 28, 3271–3277. [Google Scholar] [CrossRef]
- O’Reilly, E.A.; Gubbins, L.; Sharma, S.; Tully, R.; Guang, M.H.; Weiner-Gorzel, K.; McCaffrey, J.; Harrison, M.; Furlong, F.; Kell, M.; et al. The fate of chemoresistance in triple negative breast cancer (TNBC). BBA Clin. 2015, 3, 257–275. [Google Scholar] [CrossRef]
- Aysola, K.; Desai, A.; Welch, C.; Xu, J.; Qin, Y.; Reddy, V.; Matthews, R.; Owens, C.; Okoli, J.; Beech, D.J.; et al. Triple Negative Breast Cancer—An Overview. Hered. Genet. 2013, 2013. [Google Scholar] [CrossRef]
- Berrada, N.; Delaloge, S.; Andre, F. Treatment of triple-negative metastatic breast cancer: Toward individualized targeted treatments or chemosensitization? Ann. Oncol. 2010, 21 (Suppl. 7), vii30–vii35. [Google Scholar] [CrossRef]
- Wahba, H.A.; El-Hadaad, H.A. Current approaches in treatment of triple-negative breast cancer. Cancer Biol. Med. 2015, 12, 106–116. [Google Scholar] [CrossRef]
- Anders, C.K.; Carey, L.A. Biology, metastatic patterns, and treatment of patients with triple-negative breast cancer. Clin. Breast Cancer 2009, 9 (Suppl. 2), S73–S81. [Google Scholar] [CrossRef]
- Bonotto, M.; Gerratana, L.; Poletto, E.; Driol, P.; Giangreco, M.; Russo, S.; Minisini, A.M.; Andreetta, C.; Mansutti, M.; Pisa, F.E.; et al. Measures of outcome in metastatic breast cancer: Insights from a real-world scenario. Oncologist 2014, 19, 608–615. [Google Scholar] [CrossRef]
- Agarwal, G.; Nanda, G.; Lal, P.; Mishra, A.; Agarwal, A.; Agrawal, V.; Krishnani, N. Outcomes of Triple-Negative Breast Cancers (TNBC) Compared with Non-TNBC: Does the Survival Vary for All Stages? World J. Surg. 2016, 40, 1362–1372. [Google Scholar] [CrossRef]
- Yin, H.L.; Wu, C.C.; Lin, C.H.; Chai, C.Y.; Hou, M.F.; Chang, S.J.; Tsai, H.P.; Hung, W.C.; Pan, M.R.; Luo, C.W. beta1 Integrin as a Prognostic and Predictive Marker in Triple-Negative Breast Cancer. Int. J. Mol. Sci. 2016, 17, 1432. [Google Scholar] [CrossRef]
- O’Hanlon, L.H. Natural born killers: NK cells drafted into the cancer fight. J. Natl. Cancer Inst. 2004, 96, 651–653. [Google Scholar] [CrossRef][Green Version]
- Cao, Z.; Zhang, Z.; Huang, Z.; Wang, R.; Yang, A.; Liao, L.; Du, J. Antitumor and immunomodulatory effects of low-dose 5-FU on hepatoma 22 tumor-bearing mice. Oncol. Lett. 2014, 7, 1260–1264. [Google Scholar] [CrossRef]
- Mittendorf, E.A.; Philips, A.V.; Meric-Bernstam, F.; Qiao, N.; Wu, Y.; Harrington, S.; Su, X.; Wang, Y.; Gonzalez-Angulo, A.M.; Akcakanat, A.; et al. PD-L1 expression in triple-negative breast cancer. Cancer Immunol. Res. 2014, 2, 361–370. [Google Scholar] [CrossRef]
- Zimmer, J. Immunotherapy: Natural killers take on cancer. Nature 2014, 505, 483. [Google Scholar] [CrossRef]
- Wei, F.; Rong, X.X.; Xie, R.Y.; Jia, L.T.; Wang, H.Y.; Qin, Y.J.; Chen, L.; Shen, H.F.; Lin, X.L.; Yang, J.; et al. Cytokine-induced killer cells efficiently kill stem-like cancer cells of nasopharyngeal carcinoma via the NKG2D-ligands recognition. Oncotarget 2015, 6, 35023–35039. [Google Scholar] [CrossRef]
- Wang, X.; Teng, F.; Kong, L.; Yu, J. PD-L1 expression in human cancers and its association with clinical outcomes. OncoTargets Ther. 2016, 9, 5023–5039. [Google Scholar] [CrossRef]
- de Mello, R.A.; Veloso, A.F.; Esrom Catarina, P.; Nadine, S.; Antoniou, G. Potential role of immunotherapy in advanced non-small-cell lung cancer. OncoTargets Ther. 2017, 10, 21–30. [Google Scholar] [CrossRef]
- Gao, X.; Mi, Y.; Guo, N.; Xu, H.; Xu, L.; Gou, X.; Jin, W. Cytokine-Induced Killer Cells as Pharmacological Tools for Cancer Immunotherapy. Front. Immunol. 2017, 8, 774. [Google Scholar] [CrossRef]
- Guillon, A.; Reckamp, K.L.; Heuze-Vourc’h, N. Immunotherapy improves the prognosis of lung cancer: Do we have to change intensive care unit admission and triage guidelines? Crit. Care 2017, 21, 18. [Google Scholar] [CrossRef]
- Hsu, S.H.; Chen, S.H.; Kuo, C.C.; Chang, J.Y. Ubiquitin-conjugating enzyme E2 B regulates the ubiquitination of O(6)-methylguanine-DNA methyltransferase and BCNU sensitivity in human nasopharyngeal carcinoma cells. Biochem. Pharmacol. 2018, 158, 327–338. [Google Scholar] [CrossRef]
- Wang, Y.; Dai, H.; Li, H.; Lv, H.; Wang, T.; Fu, X.; Han, W. Growth of human colorectal cancer SW1116 cells is inhibited by cytokine-induced killer cells. Clin. Dev. Immunol. 2011, 2011, 621414. [Google Scholar] [CrossRef]
- Berghmans, T.; Meert, A.P. Immunotherapy and non-small cell lung cancer: A (r)evolution. Rev. Med. Brux. 2017, 38, 175–177. [Google Scholar]
- Introna, M. CIK as therapeutic agents against tumors. J. Autoimmun. 2017, 85, 32–44. [Google Scholar] [CrossRef]
- Dal Bello, M.G.; Alama, A.; Coco, S.; Vanni, I.; Grossi, F. Understanding the checkpoint blockade in lung cancer immunotherapy. Drug Discov. Today 2017, 22, 1266–1273. [Google Scholar] [CrossRef]
- Du, L.; Herbst, R.S.; Morgensztern, D. Immunotherapy in Lung Cancer. Hematol. Oncol. Clin. N. Am. 2017, 31, 131–141. [Google Scholar] [CrossRef]
- Durrieu, L.; Lemieux, W.; Dieng, M.M.; Fontaine, F.; Duval, M.; Le Deist, F.; Haddad, E. Implication of different effector mechanisms by cord blood-derived and peripheral blood-derived cytokine-induced killer cells to kill precursor B acute lymphoblastic leukemia cell lines. Cytotherapy 2014, 16, 845–856. [Google Scholar] [CrossRef]
- Gammaitoni, L.; Giraudo, L.; Macagno, M.; Leuci, V.; Mesiano, G.; Rotolo, R.; Sassi, F.; Sanlorenzo, M.; Zaccagna, A.; Pisacane, A.; et al. Cytokine-Induced Killer Cells Kill Chemo-surviving Melanoma Cancer Stem Cells. Clin. Cancer Res. 2017, 23, 2277–2288. [Google Scholar] [CrossRef]
- Luo, H.; Gong, L.; Zhu, B.; Huang, Y.; Tang, C.; Yu, S.; Yang, Z.; Zhou, X. Therapeutic outcomes of autologous CIK cells as a maintenance therapy in the treatment of lung cancer patients: A retrospective study. Biomed. Pharmacother. 2016, 84, 987–993. [Google Scholar] [CrossRef]
- Nardone, V.; Botta, C.; Caraglia, M.; Martino, E.C.; Ambrosio, M.R.; Carfagno, T.; Tini, P.; Semeraro, L.; Misso, G.; Grimaldi, A.; et al. Tumor infiltrating T lymphocytes expressing FoxP3, CCR7 or PD-1 predict the outcome of prostate cancer patients subjected to salvage radiotherapy after biochemical relapse. Cancer Biol. Ther. 2016, 17, 1213–1220. [Google Scholar] [CrossRef]
- Reynders, K.; De Ruysscher, D. Tumor infiltrating lymphocytes in lung cancer: A new prognostic parameter. J. Thorac. Dis. 2016, 8, E833–E835. [Google Scholar] [CrossRef]
- Ahn, M.J.; Sun, J.M.; Lee, S.H.; Ahn, J.S.; Park, K. EGFR TKI combination with immunotherapy in non-small cell lung cancer. Expert Opin. Drug Saf. 2017, 16, 465–469. [Google Scholar] [CrossRef]
- Li, D.P.; Li, W.; Feng, J.; Chen, K.; Tao, M. Adjuvant chemotherapy with sequential cytokine-induced killer (CIK) cells in stage IB non-small cell lung cancer. Oncol. Res. 2015, 22, 67–74. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, L.; Du, H.; He, X.; Yin, Y.; Gu, Y.; Liu, L.; Lu, K.; Guo, R.; Liu, P.; et al. Autologous cytokine-induced killer cell therapy in lung cancer patients: A retrospective study. Biomed. Pharmacother. 2015, 70, 248–252. [Google Scholar] [CrossRef]
- Oelsner, S.; Wagner, J.; Friede, M.E.; Pfirrmann, V.; Genssler, S.; Rettinger, E.; Buchholz, C.J.; Pfeifer, H.; Schubert, R.; Ottmann, O.G.; et al. Chimeric antigen receptor-engineered cytokine-induced killer cells overcome treatment resistance of pre-B-cell acute lymphoblastic leukemia and enhance survival. Int. J. Cancer 2016, 139, 1799–1809. [Google Scholar] [CrossRef]
- Taliaferro-Smith, L.; Oberlick, E.; Liu, T.; McGlothen, T.; Alcaide, T.; Tobin, R.; Donnelly, S.; Commander, R.; Kline, E.; Nagaraju, G.P.; et al. FAK activation is required for IGF1R-mediated regulation of EMT, migration, and invasion in mesenchymal triple negative breast cancer cells. Oncotarget 2015, 6, 4757–4772. [Google Scholar] [CrossRef]
- Tai, Y.L.; Chen, L.C.; Shen, T.L. Emerging roles of focal adhesion kinase in cancer. Biomed. Res. Int. 2015, 2015, 690690. [Google Scholar] [CrossRef]
- Golubovskaya, V.M. Focal adhesion kinase as a cancer therapy target. Anticancer Agents Med. Chem. 2010, 10, 735–741. [Google Scholar] [CrossRef]
- Li, S.; Hua, Z.C. FAK expression regulation and therapeutic potential. Adv. Cancer Res. 2008, 101, 45–61. [Google Scholar] [CrossRef]
- Agochiya, M.; Brunton, V.G.; Owens, D.W.; Parkinson, E.K.; Paraskeva, C.; Keith, W.N.; Frame, M.C. Increased dosage and amplification of the focal adhesion kinase gene in human cancer cells. Oncogene 1999, 18, 5646–5653. [Google Scholar] [CrossRef]
- Owens, L.V.; Xu, L.; Dent, G.A.; Yang, X.; Sturge, G.C.; Craven, R.J.; Cance, W.G. Focal adhesion kinase as a marker of invasive potential in differentiated human thyroid cancer. Ann. Surg. Oncol. 1996, 3, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Golubovskaya, V.M.; Ylagan, L.; Miller, A.; Hughes, M.; Wilson, J.; Wang, D.; Brese, E.; Bshara, W.; Edge, S.; Morrison, C.; et al. High focal adhesion kinase expression in breast carcinoma is associated with lymphovascular invasion and triple-negative phenotype. BMC Cancer 2014, 14, 769. [Google Scholar] [CrossRef] [PubMed]
- Kolev, V.N.; Tam, W.F.; Wright, Q.G.; McDermott, S.P.; Vidal, C.M.; Shapiro, I.M.; Xu, Q.; Wicha, M.S.; Pachter, J.A.; Weaver, D.T. Inhibition of FAK kinase activity preferentially targets cancer stem cells. Oncotarget 2017, 8, 51733–51747. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Hegde, S.; Knolhoff, B.L.; Zhu, Y.; Herndon, J.M.; Meyer, M.A.; Nywening, T.M.; Hawkins, W.G.; Shapiro, I.M.; Weaver, D.T.; et al. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nat. Med. 2016, 22, 851–860. [Google Scholar] [CrossRef]
- Tiede, S.; Meyer-Schaller, N.; Kalathur, R.K.R.; Ivanek, R.; Fagiani, E.; Schmassmann, P.; Stillhard, P.; Hafliger, S.; Kraut, N.; Schweifer, N.; et al. The FAK inhibitor BI 853520 exerts anti-tumor effects in breast cancer. Oncogenesis 2018, 7, 73. [Google Scholar] [CrossRef]
- Luo, C.W.; Wu, C.C.; Ch’ang, H.J. Radiation sensitization of tumor cells induced by shear stress: The roles of integrins and FAK. Biochim. Biophys. Acta 2014, 1843, 2129–2137. [Google Scholar] [CrossRef]
- Pan, M.R.; Hou, M.F.; Ou-Yang, F.; Wu, C.C.; Chang, S.J.; Hung, W.C.; Yip, H.K.; Luo, C.W. FAK is Required for Tumor Metastasis-Related Fluid Microenvironment in Triple-Negative Breast Cancer. J. Clin. Med. 2019, 8, 38. [Google Scholar] [CrossRef]
- Serrels, A.; Lund, T.; Serrels, B.; Byron, A.; McPherson, R.C.; von Kriegsheim, A.; Gomez-Cuadrado, L.; Canel, M.; Muir, M.; Ring, J.E.; et al. Nuclear FAK controls chemokine transcription, Tregs, and evasion of anti-tumor immunity. Cell 2015, 163, 160–173. [Google Scholar] [CrossRef]
- Serrels, A.; Frame, M.C. FAK goes nuclear to control antitumor immunity-a new target in cancer immuno-therapy. Oncoimmunology 2016, 5, e1119356. [Google Scholar] [CrossRef]
- Symeonides, S.N.; Anderton, S.M.; Serrels, A. FAK-inhibition opens the door to checkpoint immunotherapy in Pancreatic Cancer. J. Immunother. Cancer 2017, 5, 17. [Google Scholar] [CrossRef]
- Mohan, N.; Hosain, S.; Zhao, J.; Shen, Y.; Luo, X.; Jiang, J.; Endo, Y.; Wu, W.J. Atezolizumab potentiates Tcell-mediated cytotoxicity and coordinates with FAK to suppress cell invasion and motility in PD-L1(+) triple negative breast cancer cells. Oncoimmunology 2019, 8, e1624128. [Google Scholar] [CrossRef] [PubMed]
- Osipov, A.; Saung, M.T.; Zheng, L.; Murphy, A.G. Small molecule immunomodulation: The tumor microenvironment and overcoming immune escape. J. Immunother. Cancer 2019, 7, 224. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.H.; Tsai, K.W.; Chiu, S.Y.; Kuo, W.H.; Chen, H.Y.; Jiang, S.S.; Chang, K.J.; Hung, W.C.; Wang, L.H. Identification of the Novel Role of CD24 as an Oncogenesis Regulator and Therapeutic Target for Triple-Negative Breast Cancer. Mol. Cancer Ther. 2019, 18, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Lin, F.; Geng, R.; Ge, X.; Tang, W.; Chang, J.; Wu, Z.; Liu, X.; Lin, Y.; Zhang, Z.; et al. Implication of combined PD-L1/PD-1 blockade with cytokine-induced killer cells as a synergistic immunotherapy for gastrointestinal cancer. Oncotarget 2016, 7, 10332–10344. [Google Scholar] [CrossRef] [PubMed]
- Sangiolo, D. Cytokine induced killer cells as promising immunotherapy for solid tumors. J. Cancer 2011, 2, 363–368. [Google Scholar] [CrossRef]
- Zhou, Z.Q.; Zhao, J.J.; Pan, Q.Z.; Chen, C.L.; Liu, Y.; Tang, Y.; Zhu, Q.; Weng, D.S.; Xia, J.C. PD-L1 expression is a predictive biomarker for CIK cell-based immunotherapy in postoperative patients with breast cancer. J. Immunother. Cancer 2019, 7, 228. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, S.; Yang, B.; Lu, S.; Du, Y.; Liu, H. Adjuvant treatment for triple-negative breast cancer: A retrospective study of immunotherapy with autologous cytokine-induced killer cells in 294 patients. Cancer Biol. Med. 2019, 16, 350–360. [Google Scholar] [CrossRef]
- Li, M.; Wang, Y.; Wei, F.; An, X.; Zhang, N.; Cao, S.; Ren, B.; Zhang, X.; Ren, X. Efficiency of Cytokine-Induced Killer Cells in Combination with Chemotherapy for Triple-Negative Breast Cancer. J. Breast Cancer 2018, 21, 150–157. [Google Scholar] [CrossRef]
- Hu, J.; Hu, J.; Liu, X.; Hu, C.; Li, M.; Han, W. Effect and safety of cytokine-induced killer (CIK) cell immunotherapy in patients with breast cancer: A meta-analysis. Medicine 2017, 96, e8310. [Google Scholar] [CrossRef]
- Sidaway, P. Pancreatic cancer: FAK regulates sensitivity to immunotherapy. Nat. Rev. Clin. Oncol. 2016, 13, 528. [Google Scholar] [CrossRef]
- Taylor, K.; Chen, X.; Tancioni, I.; Kleinschmidt, E.; Barrie, A.; Diaz-Osterman, C.; Fu, G.; Mark, A.; Xu, G.; Fisch, K.; et al. Focal Adhesion Kinase (FAK) Regulation of Programmed Death-1 (PD-1)/Programmed Death Ligand-1 (PD-L1) checkpoint signaling in a mouse model of epithelial ovarian cancer. Gynecol. Oncol. 2019, 154, e24. [Google Scholar] [CrossRef]
| FAK | PD-L1, n (%) | p-Value | |
|---|---|---|---|
| Positive (>1%) | Negative | ||
| High | 17 (26.5) | 20 (31.3) | |
| Low | 5 (7.8) | 22 (34.4) | p = 0.023 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, M.-R.; Wu, C.-C.; Kan, J.-Y.; Li, Q.-L.; Chang, S.-J.; Wu, C.-C.; Li, C.-L.; Ou-Yang, F.; Hou, M.-F.; Yip, H.-K.; et al. Impact of FAK Expression on the Cytotoxic Effects of CIK Therapy in Triple-Negative Breast Cancer. Cancers 2020, 12, 94. https://doi.org/10.3390/cancers12010094
Pan M-R, Wu C-C, Kan J-Y, Li Q-L, Chang S-J, Wu C-C, Li C-L, Ou-Yang F, Hou M-F, Yip H-K, et al. Impact of FAK Expression on the Cytotoxic Effects of CIK Therapy in Triple-Negative Breast Cancer. Cancers. 2020; 12(1):94. https://doi.org/10.3390/cancers12010094
Chicago/Turabian StylePan, Mei-Ren, Cheng-Che Wu, Jung-Yu Kan, Qiao-Lin Li, Shu-Jyuan Chang, Chun-Chieh Wu, Chung-Liang Li, Fu Ou-Yang, Ming-Feng Hou, Hon-Kan Yip, and et al. 2020. "Impact of FAK Expression on the Cytotoxic Effects of CIK Therapy in Triple-Negative Breast Cancer" Cancers 12, no. 1: 94. https://doi.org/10.3390/cancers12010094
APA StylePan, M.-R., Wu, C.-C., Kan, J.-Y., Li, Q.-L., Chang, S.-J., Wu, C.-C., Li, C.-L., Ou-Yang, F., Hou, M.-F., Yip, H.-K., & Luo, C.-W. (2020). Impact of FAK Expression on the Cytotoxic Effects of CIK Therapy in Triple-Negative Breast Cancer. Cancers, 12(1), 94. https://doi.org/10.3390/cancers12010094





