Dose Intensity/Body Surface Area Ratio is a Novel Marker Useful for Predicting Response to Lenvatinib against Hepatocellular Carcinoma
Abstract
1. Introduction
2. Results
2.1. Baseline Characteristics of Patients
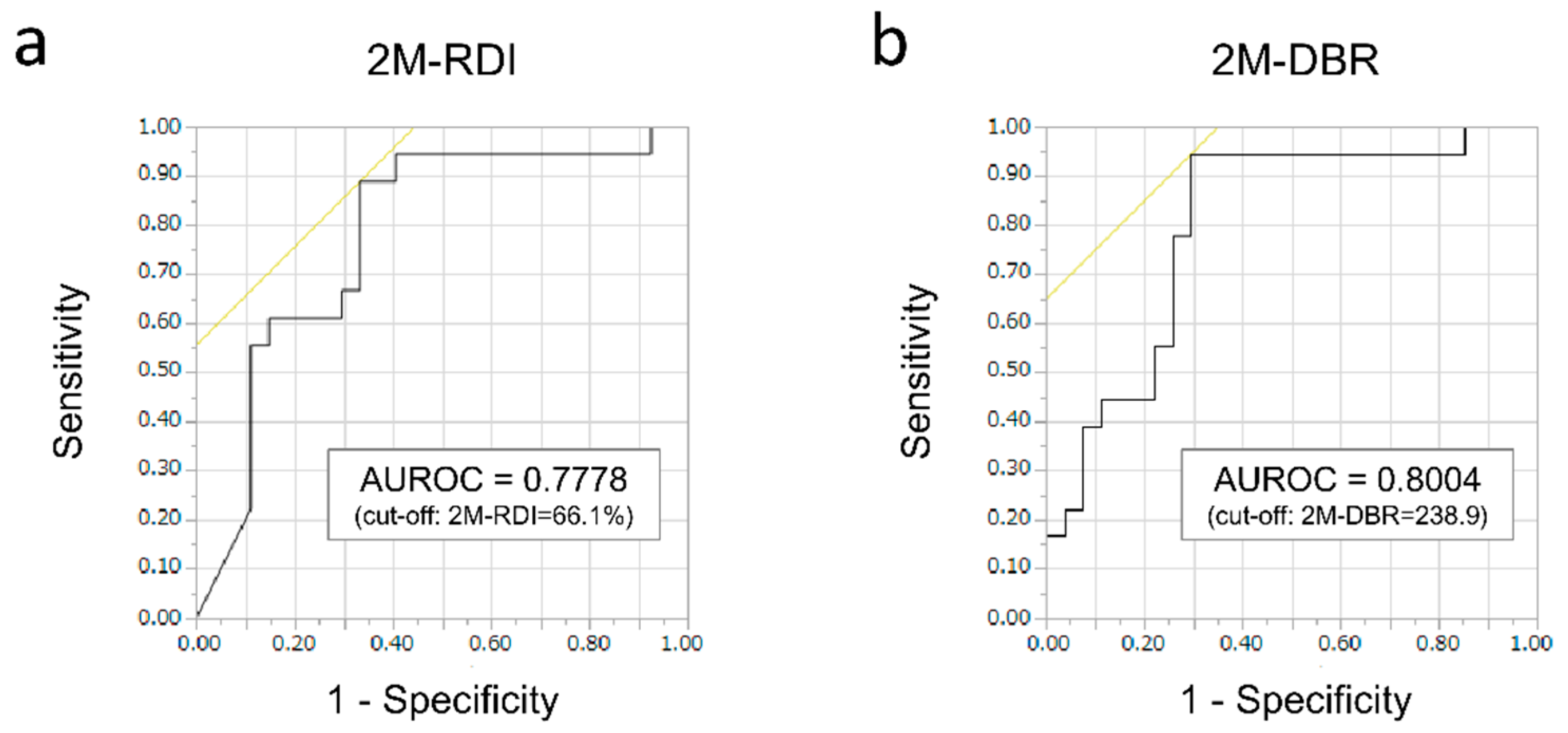
2.2. Therapeutic Response to Lenvatinib According to 2M-RDI or 2M-DBR
2.3. Adverse Events Related to Lenvatinib Therapy
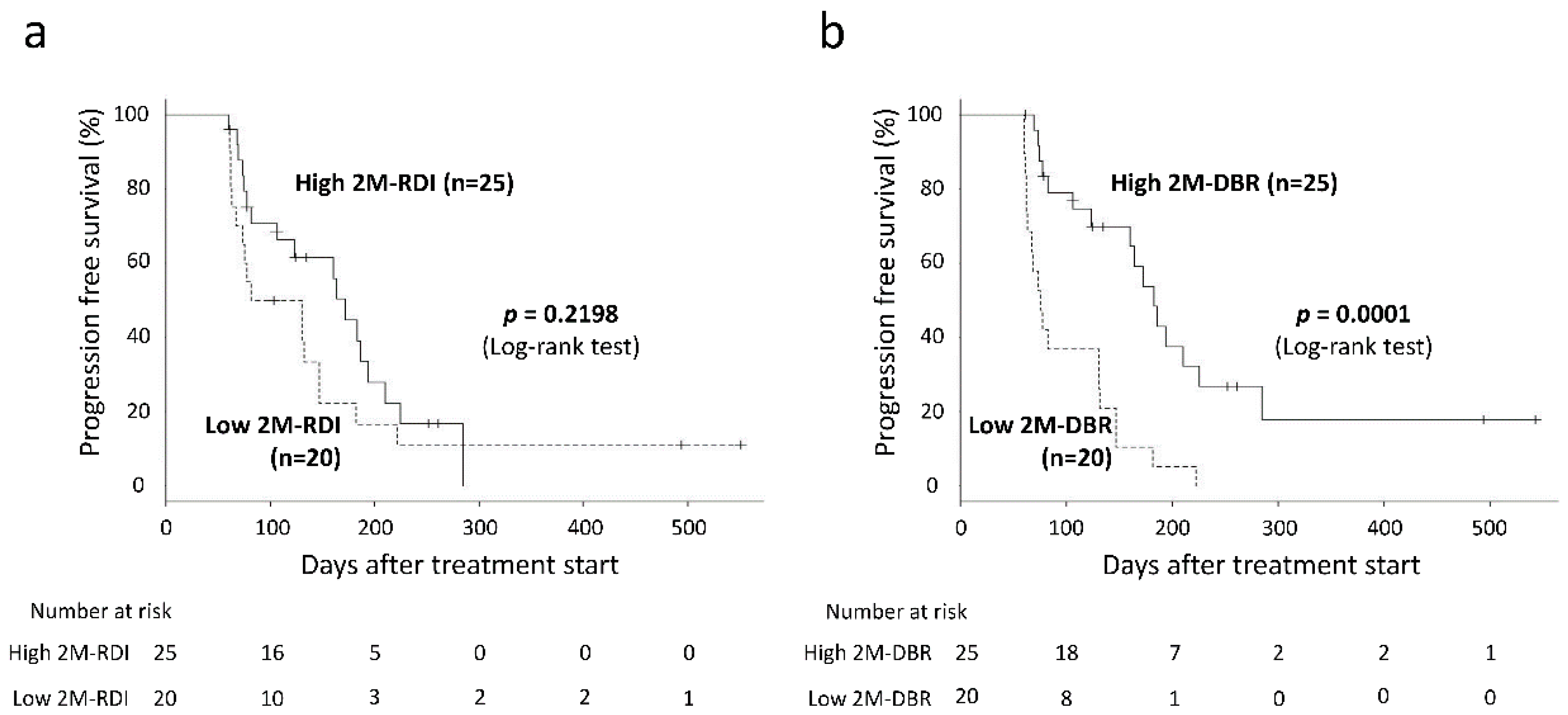
2.4. Relationship Between 2M-RDI or 2M-DBR and Progression-Free Survival
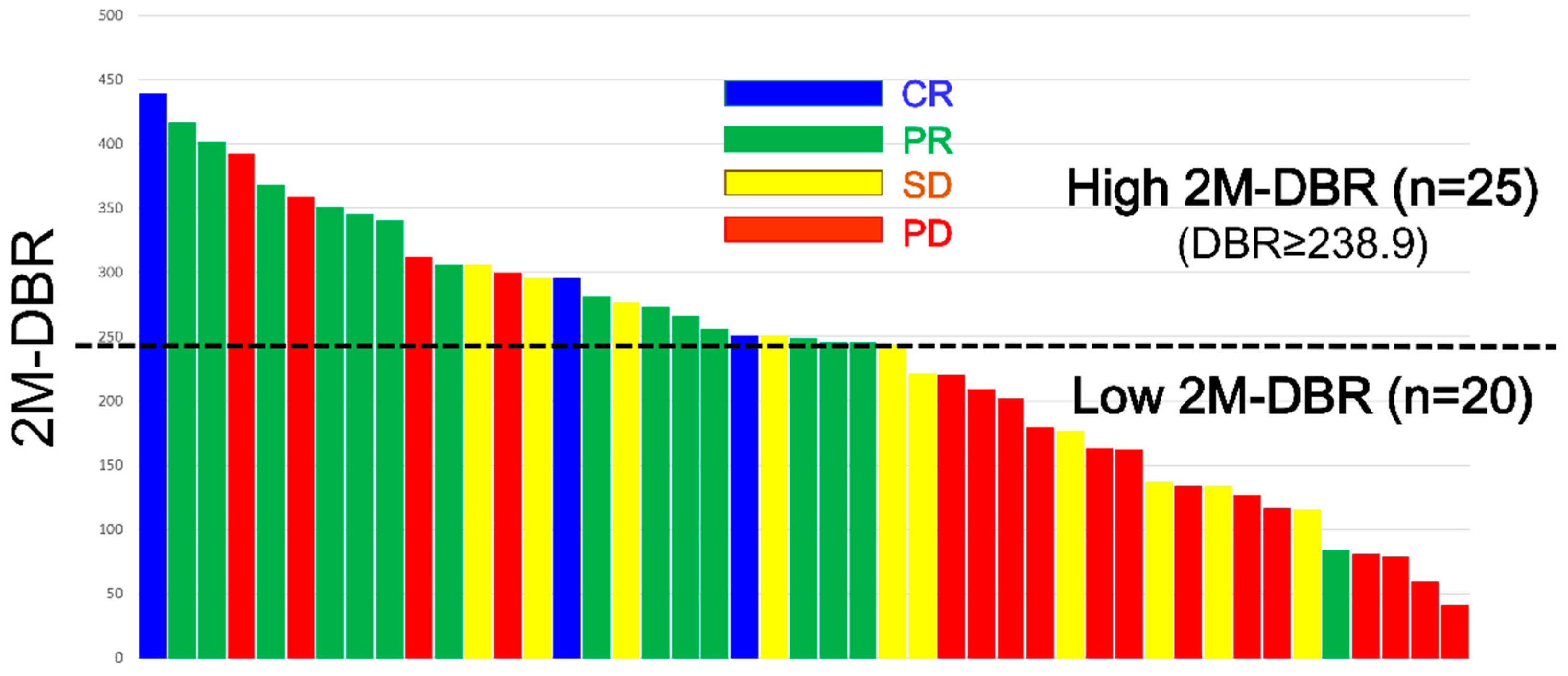
2.5. Comparison between Patients with High 2M-DBR and Low 2M-DBR
2.6. Factors Associated with Progression-Free Survival
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Protocol of Treatment with Lenvatinib
4.3. Calculation of 2M-RDI and 2M-DBR
4.4. Evaluation of Therapeutic Response
4.5. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Akinyemiju, T.; Abera, S.; Ahmed, M.; Alam, N.; Alemayohu, M.A.; Allen, C.; Al-Raddadi, R.; Alvis-Guzman, N.; Amoako, Y.; Artaman, A.; et al. The Burden of Primary Liver Cancer and Underlying Etiologies from 1990 to 2015 at the Global, Regional, and National Level: Results from the Global Burden of Disease Study 2015. JAMA Oncol. 2017, 3, 1683–1691. [Google Scholar] [PubMed]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Eso, Y.; Marusawa, H. Novel approaches for molecular targeted therapy against hepatocellular carcinoma. Hepatol. Res. 2018, 48, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Havrilesky, L.J.; Reiner, M.; Morrow, P.K.; Watson, H.; Crawford, J. A review of relative dose intensity and survival in patients with metastatic solid tumors. Crit Rev. Oncol. Hematol. 2015, 93, 203–210. [Google Scholar] [CrossRef]
- Schraa, S.J.; Frerichs, K.A.; Agterof, M.J.; Hunting, J.C.B.; Los, M.; de Jong, P.C. Relative dose intensity as a proxy measure of quality and prognosis in adjuvant chemotherapy for breast cancer in daily clinical practice. Eur. J. Cancer 2017, 79, 152–157. [Google Scholar] [CrossRef]
- Yabusaki, N.; Fujii, T.; Yamada, S.; Murotani, K.; Sugimoto, H.; Kanda, M.; Nakayama, G.; Koike, M.; Fujiwara, M.; Kodera, Y. The significance of relative dose intensity in adjuvant chemotherapy of pancreatic ductal adenocarcinoma-including the analysis of clinicopathological factors influencing relative dose intensity. Med. (Baltim.) 2016, 95, e4282. [Google Scholar] [CrossRef]
- Ishihara, H.; Takagi, T.; Kondo, T.; Iwamoto, K.; Tachibana, H.; Yoshida, K.; Omae, K.; Iizuka, J.; Kobayashi, H.; Tanabe, K. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn. J. Clin. Oncol. 2018, 48, 667–672. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Hirakawa, T.; Inokuchi, K. Importance of relative dose intensity in chemotherapy for diffuse large B-cell lymphoma. J. Clin. Exp. Hematop. 2011, 51, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tsuchiya, K.; Kurosaki, M.; Yasui, Y.; Inada, K.; Kirino, S.; Yamashita, K.; Sekiguchi, S.; Hayakawa, Y.; Osawa, L.; et al. Sorafenib-Regorafenib Sequential Therapy in Japanese Patients with Unresectable Hepatocellular Carcinoma-Relative Dose Intensity and Post-Regorafenib Therapies in Real World Practice. Cancers 2019, 11, 1517. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Kudo, M.; Kawazoe, S.; Osaki, Y.; Ikeda, M.; Okusaka, T.; Tamai, T.; Suzuki, T.; Hisai, T.; Hayato, S.; et al. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J. Gastroenterol. 2017, 52, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Okusaka, T.; Mitsunaga, S.; Ueno, H.; Tamai, T.; Suzuki, T.; Hayato, S.; Kadowaki, T.; Okita, K.; Kumada, H. Safety and Pharmacokinetics of Lenvatinib in Patients with Advanced Hepatocellular Carcinoma. Clin. Cancer Res. 2016, 22, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Tamai, T.; Hayato, S.; Hojo, S.; Suzuki, T.; Okusaka, T.; Ikeda, K.; Kumada, H. Dose Finding of Lenvatinib in Subjects With Advanced Hepatocellular Carcinoma Based on Population Pharmacokinetic and Exposure-Response Analyses. J. Clin. Pharm. 2017, 57, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Moriguchi, M.; Seko, Y.; Ishikawa, H.; Yo, T.; Kimura, H.; Fujii, H.; Shima, T.; Mitsumoto, Y.; Ishiba, H.; et al. Impact of Relative Dose Intensity of Early-phase Lenvatinib Treatment on Therapeutic Response in Hepatocellular Carcinoma. Anticancer Res. 2019, 39, 5149–5156. [Google Scholar] [CrossRef]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Prognostic factor of lenvatinib for unresectable hepatocellular carcinoma in real-world conditions-Multicenter analysis. Cancer Med. 2019, 8, 3719–3728. [Google Scholar] [CrossRef]
- Ueshima, K.; Nishida, N.; Hagiwara, S.; Aoki, T.; Minami, T.; Chishina, H.; Takita, M.; Minami, Y.; Ida, H.; Takenaka, M.; et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers 2019, 11, 952. [Google Scholar] [CrossRef]
- Asama, H.; Suzuki, R.; Takagi, T.; Sugimoto, M.; Konno, N.; Watanabe, K.; Nakamura, J.; Kikuchi, H.; Takasumi, M.; Sato, Y.; et al. Evaluation of inflammation-based markers for predicting the prognosis of unresectable pancreatic ductal adenocarcinoma treated with chemotherapy. Mol. Clin. Oncol. 2018, 9, 408–414. [Google Scholar] [CrossRef]
- Daitoku, N.; Miyamoto, Y.; Tokunaga, R.; Sakamoto, Y.; Hiyoshi, Y.; Iwatsuki, M.; Baba, Y.; Iwagami, S.; Yoshida, N.; Baba, H. Controlling Nutritional Status (CONUT) Score Is a Prognostic Marker in Metastatic Colorectal Cancer Patients Receiving First-line Chemotherapy. Anticancer Res. 2018, 38, 4883–4888. [Google Scholar] [CrossRef] [PubMed]
- Hikage, M.; Taniyama, Y.; Sakurai, T.; Sato, C.; Takaya, K.; Okamoto, H.; Konno, T.; Ujiie, N.; Naitoh, T.; Unno, M.; et al. The Influence of the Perioperative Nutritional Status on the Survival Outcomes for Esophageal Cancer Patients with Neoadjuvant Chemotherapy. Ann. Surg. Oncol. 2019, 26, 4744–4753. [Google Scholar] [CrossRef] [PubMed]
- Tada, T.; Kumada, T.; Toyoda, H.; Kiriyama, S.; Tanikawa, M.; Hisanaga, Y.; Kanamori, A.; Kitabatake, S.; Yama, T. Impact of the branched-chain amino acid to tyrosine ratio and branched-chain amino acid granule therapy in patients with hepatocellular carcinoma: A propensity score analysis. J. Gastroenterol. Hepatol. 2015, 30, 1412–1419. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Imai, M.; Ko, M.; Sato, H.; Nozawa, Y.; Sano, T.; Iwanaga, A.; Seki, K.; Honma, T.; Yoshida, T. Evaluation of the branched-chain amino acid-to-tyrosine ratio prior to treatment as a prognostic predictor in patients with liver cirrhosis. Oncotarget 2017, 8, 79480–79490. [Google Scholar] [CrossRef]
- Tada, T.; Kumada, T.; Toyoda, H.; Yasuda, S.; Koyabu, T.; Nakashima, M. Impact of Branched-Chain Amino Acid Granule Therapy in Patients with Hepatocellular Carcinoma Who Have Normal Albumin Levels and Low Branched-Chain Amino Acid to Tyrosine Ratios. Nutr. Cancer 2019, 71, 1132–1141. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Hirooka, M.; Tsuji, K.; Itobayashi, E.; Kariyama, K.; Ishikawa, T.; Tajiri, K.; Ochi, H.; Tada, T.; et al. A better method for assessment of hepatic function in hepatocellular carcinoma patients treated with radiofrequency ablation: Usefulness of albumin-bilirubin grade. Hepatol. Res. 2018, 48, E61–E67. [Google Scholar] [CrossRef]
- Ignacio de Ulíbarri, J.; González-Madroño, A.; de Villar, N.G.; González, P.; González, B.; Mancha, A.; Rodríguez, F.; Fernández, G. CONUT: a tool for controlling nutritional status. First validation in a hospital population. Nutr. Hosp. 2005, 20, 38–45. [Google Scholar]
- Eso, Y.; Takai, A.; Taura, K.; Takahashi, K.; Ueda, Y.; Marusawa, H.; Seno, H. Association of Mac-2-binding protein glycosylation isomer level with nutritional status in chronic liver disease. J. Gastroenterol. Hepatol. 2018, 33, 1649–1658. [Google Scholar] [CrossRef]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; S Sulkowski, M.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Yamasaki, K.; Tateyama, M.; Abiru, S.; Komori, A.; Nagaoka, S.; Saeki, A.; Hashimoto, S.; Sasaki, R.; Bekki, S.; Kugiyama, Y.; et al. Elevated serum levels of Wisteria floribunda agglutinin-positive human Mac-2 binding protein predict the development of hepatocellular carcinoma in hepatitis C patients. Hepatology 2014, 60, 1563–1570. [Google Scholar] [CrossRef]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
| Variable | n = 45 |
|---|---|
| Age (years, range) | 70.4 ± 8.61 (50–85) |
| Gender (Male vs. Female) | 37/8 |
| Body weight (<60kg vs. ≥60kg) | 15/30 |
| Body surface area (m2) | 1.72 ± 0.17 |
| Etiology (HBV vs. HCV vs. non-B non-C) | 6/16/23 |
| TNM stage (III vs. IVA vs. IV B) | 16/8/21 |
| BCLC stage (B vs. C) | 16/29 |
| Treatment history (Naïve vs. recurrence) | |
| <Treatment prior to lenvatinib> | 5/40 |
| Surgery | 4 |
| Radiofrequency ablation | 1 |
| TACE | 25 |
| Sorafenib | 9 |
| Regorafenib | 1 |
| AST (IU/L) | 44.3 ± 24.7 |
| ALT (IU/L) | 28.0 ± 15.0 |
| Platelets (x104/μL) | 14.9 ± 7.00 |
| ALB (g/dL) | 3.56 ± 0.44 |
| T-Bil (mg/dL) | 0.90 ± 0.35 |
| PT (%) | 94.0 ± 15.5 |
| Child–Pugh score (5A vs. 6A vs. 7B) | 23/16/6 |
| ALBI score | −2.27 ± 0.40 |
| mALBI grade (1 vs. 2a vs. 2b vs. 3) | 11/13/21/0 |
| BTR | 5.85 ± 2.17 |
| CONUT score | 3.64 ± 1.80 |
| FIB-4 index | 5.17 ± 4.30 |
| M2BPGi (cut-off index) | 2.87 ± 2.93 |
| AFP (ng/mL) | 8844 ± 27760 |
| DCP (mAU/mL) | 5183 ± 9891 |
| Initial dose of lenvatinib (4 mg vs. 8 mg vs. 12 mg) | 3/25/17 |
| Initial dose of lenvatinib (standard vs. reduced) | 29/16 |
| Response | n = 45 |
|---|---|
| Complete response (CR) | 3 |
| Partial response (PR) | 15 |
| Stable disease (SD) | 10 |
| Progressive disease (PD) | 17 |
| Objective response rate (ORR) | 40.0% |
| (18/45) | |
| Disease control rate (DCR) | 62.2% |
| (28/45) |
| Response | High 2M-DBR Group (n = 25) | Low 2M-DBR Group (n = 20) | p Value |
|---|---|---|---|
| Complete response (CR) | 3 | 0 | |
| Partial response (PR) | 14 | 1 | |
| Stable disease (SD) | 4 | 6 | |
| Progressive disease (PD) | 4 | 13 | |
| Objective response rate (ORR) | 68.0% | 5.0% | <0.0001 |
| (17/25) | (1/20) | ||
| Disease control rate (DCR) | 84.0% | 35.0% | 0.0008 |
| (21/25) | (7/20) |
| Adverse Events | Any Grade (%) | Grade ≥3 (%) |
|---|---|---|
| Hypertension | 25 (55.6) | 7 (15.6) |
| General fatigue | 21 (46.7) | 2 (4.4) |
| Diarrhea | 16 (35.6) | 2 (4.4) |
| Appetite loss | 15 (33.3) | 2 (4.4) |
| Hand–foot skin reaction | 12 (26.7) | 1 (2.2) |
| Weight loss | 11 (24.4) | 1 (2.2) |
| Proteinuria | 10 (22.2) | 3 (6.7) |
| Hypothyroidism | 10 (22.2) | 1 (2.2) |
| Hoarseness | 7 (15.6) | 0 |
| High 2M-DBR Group (n = 25) | Low 2M-DBR Group (n = 20) | p Value | |
|---|---|---|---|
| Age (years, range) | 68.5 ± 8.47 | 72.8 ± 8.19 | 0.1018 |
| Gender (male/female) | 22/3 | 15/5 | 0.2226 |
| Body weight (<60kg/≥60kg) | 6/19 | 10/10 | 0.0702 |
| BSA (m2) | 1.76 ± 0.14 | 1.66 ± 0.18 | 0.0309 |
| Etiology (HBV vs. HCV vs. non-B non-C) | 5/8/12 | 1/8/11 | 0.3360 |
| TNM stage (III vs. IVA vs. IV B) | 9/3/13 | 7/5/8 | 0.4960 |
| BCLC stage (B vs. C) | 9/16 | 7/13 | 0.9445 |
| Treatment history (Naïve vs. recurrence) | 3/22 | 2/18 | 0.8320 |
| AST (IU/L) | 40.5 ± 19.9 | 49.0 ± 28.9 | 0.4172 |
| ALT (IU/L) | 27.0 ± 12.0 | 30.0 ± 18.8 | 0.9818 |
| Platelets (×104/μL) | 15.0 ± 7.01 | 14.7 ± 6.95 | 0.9272 |
| ALB (g/dL) | 3.63 ± 0.50 | 3.49 ± 0.35 | 0.1385 |
| T-Bil (mg/dL) | 0.84 ± 0.25 | 0.98 ± 0.43 | 0.2316 |
| PT (%) | 92.1 ± 15.2 | 96.3 ± 15.7 | 0.7975 |
| Child–Pugh score (5A vs. 6A vs. 7B) | 14/9/2 | 9/7/4 | 0.4804 |
| ALBI score | −2.33 ± 0.43 | −2.18 ± 0.35 | 0.2007 |
| mALBI grade (1 vs. 2a or 2b) | 9/16 | 2/18 | 0.0437 |
| BTR | 6.78 ± 2.00 | 4.97 ± 1.94 | 0.0222 |
| CONUT score | 3.12 ± 1.70 | 4.30 ± 1.71 | 0.0281 |
| FIB-4 index | 4.67 ± 4.20 | 5.80 ± 4.33 | 0.2217 |
| M2BPGi (cut-off index) | 2.21 ± 2.61 | 3.60 ± 3.09 | 0.0760 |
| Initial dose of lenvatinib(standard vs. reduced) | 19/6 | 10/10 | 0.0702 |
| Variable | No. of Cases | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | ||
| Age, years | 35/10 | 0.70 (0.34–1.53) | 0.3507 | ||
| (≥64 vs. <64) | |||||
| Gender | 8/37 | 1.69 (1.55–5.57) | 0.2483 | ||
| (Female vs. Male) | |||||
| Body weight | 30/15 | 0.64 (0.32–1.35) | 0.2300 | ||
| (≥ 60kg vs. < 60kg) | |||||
| Body surface area, m2 | 29/16 | 0.53 (0.26–1.09) | 0.0824 | ||
| (≥ 1.6623 vs. < 1.6623) | |||||
| ALB | 9/36 | 0.45 (0.15–1.06) | 0.0697 | ||
| (≥ 4.0 g/dL vs. < 4.0 g/dL) | |||||
| Child–Pugh score | 22/23 | 1.32 (0.68–2.60) | 0.4185 | ||
| (6 or 7 vs. 5) | |||||
| mALBI grade | 11/34 | 0.51 (0.19–1.15) | 0.1106 | ||
| (1 vs. 2a or 2b) | |||||
| BTR | 8/25 | 0.36 (0.15–0.95) | 0.0401 | 0.76 (0.27–2.18) | 0.5971 |
| (≥ 4.36 vs. < 4.36) | |||||
| CONUT score | 20/25 | 1.64 (0.83–3.22) | 0.1505 | ||
| (>3 vs. ≤ 3) | |||||
| 2M-RDI | 25/20 | 0.66 (0.34–1.30) | 0.2290 | ||
| (≥ 66.1% vs. < 66.1%) | |||||
| 2M-DBR | 25/20 | 0.26 (0.13–0.54) | 0.0003 | 0.29(0.11–0.76) | 0.0127 |
| (≥ 238.9 vs. < 238.9) | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eso, Y.; Nakano, S.; Mishima, M.; Arasawa, S.; Iguchi, E.; Nakamura, F.; Takeda, H.; Takai, A.; Takahashi, K.; Taura, K.; et al. Dose Intensity/Body Surface Area Ratio is a Novel Marker Useful for Predicting Response to Lenvatinib against Hepatocellular Carcinoma. Cancers 2020, 12, 49. https://doi.org/10.3390/cancers12010049
Eso Y, Nakano S, Mishima M, Arasawa S, Iguchi E, Nakamura F, Takeda H, Takai A, Takahashi K, Taura K, et al. Dose Intensity/Body Surface Area Ratio is a Novel Marker Useful for Predicting Response to Lenvatinib against Hepatocellular Carcinoma. Cancers. 2020; 12(1):49. https://doi.org/10.3390/cancers12010049
Chicago/Turabian StyleEso, Yuji, Shigeharu Nakano, Masako Mishima, Soichi Arasawa, Eriko Iguchi, Fumiyasu Nakamura, Haruhiko Takeda, Atsushi Takai, Ken Takahashi, Kojiro Taura, and et al. 2020. "Dose Intensity/Body Surface Area Ratio is a Novel Marker Useful for Predicting Response to Lenvatinib against Hepatocellular Carcinoma" Cancers 12, no. 1: 49. https://doi.org/10.3390/cancers12010049
APA StyleEso, Y., Nakano, S., Mishima, M., Arasawa, S., Iguchi, E., Nakamura, F., Takeda, H., Takai, A., Takahashi, K., Taura, K., & Seno, H. (2020). Dose Intensity/Body Surface Area Ratio is a Novel Marker Useful for Predicting Response to Lenvatinib against Hepatocellular Carcinoma. Cancers, 12(1), 49. https://doi.org/10.3390/cancers12010049







