Key Role of Obesity in Genitourinary Tumors with Emphasis on Urothelial and Prostate Cancers
Abstract
1. Introduction
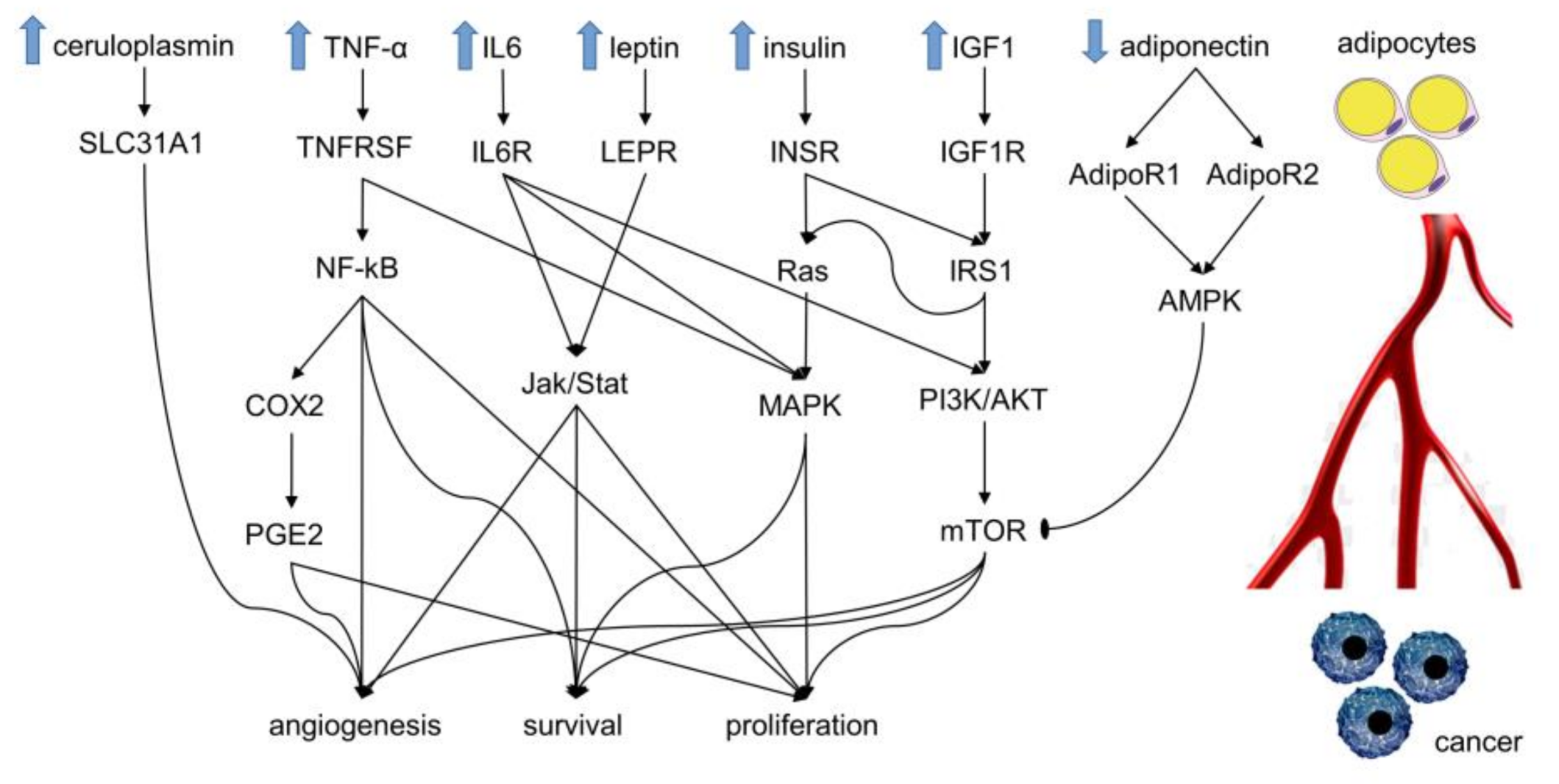
2. Molecular Mechanisms Linking Obesity and Cancer
3. Obesity and Risk of Urothelial Cancer
4. BMI and Response of Urothelial Cancer to Chemotherapy and Immunotherapy
5. Key Issues on Obesity and Urothelial Cancer
6. Obesity and Prostate Cancer
7. Obesity and Risk of Prostate Cancer
8. Correlation between Obesity and Response to Therapy in Prostate Cancer
8.1. Docetaxel
8.2. Cabazitaxel
8.3. Abiraterone Acetate and Enzalutamide
8.4. Radium 223
9. Key Issues on Obesity and Prostate Cancer
10. Conclusions
Funding
Conflicts of Interest
References
- Budny, A.; Grochowski, C.; Kozłowski, P.; Kolak, A.; Kamińska, M.; Budny, B.; Abramiuk, M.; Burdan, F. Obesity as a tumour development triggering factor. Ann. Agric. Environ. Med. 2019, 26, 13–23. [Google Scholar] [CrossRef]
- Al-Zalabani, A.H.; Stewart, K.F.; Wesselius, A.; Schols, A.M.; Zeegers, M.P. Modifiable risk factors for the prevention of bladder cancer: A systematic review of meta-analyses. Eur. J. Epidemiol. 2016, 31, 811–851. [Google Scholar] [CrossRef]
- Pietrzyk, L.; Torres, A.; Maciejewski, R.; Torres, K. Obesity and Obese-related Chronic Low-grade Inflammation in Promotion of Colorectal Cancer Development. Asian Pac. J. Cancer Prev. 2015, 16, 4161–4168. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [PubMed]
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar] [CrossRef]
- Stone, T.W.; McPherson, M.; Gail Darlington, L. Obesity and Cancer: Existing and New Hypotheses for a Causal Connection. EBioMedicine. 2018, 30, 14–28. [Google Scholar] [CrossRef]
- van Kruijsdijk, R.C.; van der Wall, E.; Visseren, F.L. Obesity and cancer: The role of dysfunctional adipose tissue. Cancer Epidemiol. Biomark. Prev. 2009, 18, 2569–2578. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, C.M.; Himbert, C.; Holowatyj, A.N.; Hursting, S.D. Energy balance and gastrointestinal cancer: Risk, interventions, outcomes and mechanisms. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 683–698. [Google Scholar] [CrossRef]
- Massari, F.; Ciccarese, C.; Santoni, M.; Iacovelli, R.; Mazzucchelli, R.; Piva, F.; Scarpelli, M.; Berardi, R.; Tortora, G.; Lopez-Beltran, A.; et al. Metabolic phenotype of bladder cancer. Cancer Treat. Rev. 2016, 45, 46–57. [Google Scholar] [CrossRef]
- Iyengar, N.M.; Gucalp, A.; Dannenberg, A.J.; Hudis, C.A. Obesity and Cancer Mechanisms: Tumor Microenvironment and Inflammation. J. Clin. Oncol. 2016, 34, 4270–4276. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zhao, M.; Li, J.; Lou, G.; Yuan, Y.; Bu, S.; Xi, Y. Obesity-associated digestive cancers: A review of mechanisms and interventions. Tumour Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Zheng, J.; Yao, X.; Peng, B. Adiponectin inhibits VEGF-A in prostate cancer cells. Tumour Biol. 2015, 36, 4287–4292. [Google Scholar] [CrossRef] [PubMed]
- Liberti, M.V.; Locasale, J.W. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 2016, 41, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Porstmann, T.; Santos, C.R.; Griffiths, B.; Cully, M.; Wu, M.; Leevers, S.; Griffiths, J.R.; Chung, Y.L.; Schulze, A. SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metabolism. 2008, 8, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Flavin, R.; Peluso, S.; Nguyen, P.L.; Loda, M. Fatty acid synthase as a potential therapeutic target in cancer. Future Oncol. 2010, 6, 551–562. [Google Scholar] [CrossRef]
- Azrad, M.; Turgeon, C.; Demark-Wahnefried, W. Current evidence linking polyunsaturated Fatty acids with cancer risk and progression. Front. Oncol. 2013, 3, 224. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Shan, K.; Chen, H.; Chen, Y.Q. n-3 Polyunsaturated Fatty Acids and their Role in Cancer Chemoprevention. Curr. Pharmacol. Rep. 2015, 1, 283. [Google Scholar] [CrossRef]
- D’Andrea, L.D.; Romanelli, A.; Di Stasi, R.; Pedone, C. Bioinorganic aspects of angiogenesis. Dalton Trans. 2010, 39, 7625–7636. [Google Scholar] [CrossRef]
- Wang, D.; DuBois, R.N. An inflammatory mediator, prostaglandin E2, in colorectal cancer. Cancer J. 2013, 19, 502–510. [Google Scholar] [CrossRef]
- Chromecki, T.F.; Cha, E.K.; Fajkovic, H.; Rink, M.; Ehdaie, B.; Svatek, R.S.; Karakiewicz, P.I.; Lotan, Y.; Tilki, D.; Bastian, P.J.; et al. Obesity is associated with worse oncological outcomes in patients treated with radical cystectomy. BJU Int. 2013, 111, 249–255. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Cimadamore, A.; Scarpelli, M.; Santoni, M.; Cheng, L.; Lopez-Beltran, A.; Montironi, R. Upper tract urothelial carcinoma and its variants: Transition from morphology to personalized molecular characterization in diagnosis, prognosis and therapy. Exp. Rev. Mol. Diagn. 2018, 18, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Dabi, Y.; El Mrini, M.; Duquesnes, I.; Delongchamps, N.B.; Sibony, M.; Zerbib, M.; Xylinas, E. Impact of body mass index on the oncological outcomes of patients treated with radical nephroureterectomy for upper tract urothelial carcinoma. World J. Urol. 2018, 36, 65–71. [Google Scholar] [CrossRef]
- Koebnick, C.; Michaud, D.; Moore, S.C.; Park, Y.; Hollenbeck, A.; Bal- lard-Barbash, R.; Schatzkin, A.; Leitzmann, M.F. Body mass index, physical activity, and bladder cancer in a large prospective study. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored. Am. Soc. Prev. Oncol. 2008, 17, 1214–1221. [Google Scholar] [CrossRef]
- Ishioka, J.; Masuda, H.; Kijima, T.; Tatokoro, M.; Yoshida, S.; Yokoyama, M.; Matsuoka, Y.; Numao, N.; Koga, F.; Saito, K.; et al. Bimodal pattern of the impact of body mass index on cancer-specific survival of upper urinary tract urothelial carcinoma patients. Anticancer Res. 2014, 34, 5683–5688. [Google Scholar] [PubMed]
- Gierth, M.; Zeman, F.; Denzinger, S.; Vetterlein, M.W.; Fisch, M.; Bastian, P.J.; Syring, I.; Ellinger, J.; Müller, S.C.; Herrmann, E.; et al. Influence of Body Mass Index on Clinical Outcome Parameters, Complication Rate and Survival after Radical Cystectomy: Evidence from a Prospective European Multicentre Study. Urol. Int. 2018, 101, 16–24. [Google Scholar] [CrossRef]
- Holick, C.N.; Giovannucci, E.L.; Stampfer, M.J.; Michaud, D.S. Prospective study of body mass index, height, physical activity and incidence of bladder cancer in US men and women. Int. J. Cancer J. Int. Cancer 2007, 120, 140–146. [Google Scholar] [CrossRef]
- Murakami, Y.; Matsumoto, K.; Ikeda, M.; Utsunomiya, T.; Hirayama, T.; Koguchi, D.; Matsuda, D.; Okuno, N.; Taoka, Y.; Irie, A.; et al. Impact of body mass index on the oncological outcomes of patients with upper and lower urinary tract cancers treated with radical surgery: A multi-institutional retrospective study. Asia Pac. J. Clin. Oncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Jeong, C.W.; Kwak, C.; Kim, H.H.; Ku, J.H. Can body mass index predict survival outcomes in patients treated with radical nephroureterectomy for upper-tract urothelial carcinoma? Int. Urol. Nephrol. 2015, 47, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, J.Y.; Kim, J.W.; Hah, Y.S.; Cho, K.S. Effect of neoadjuvant chemotherapy on locally advanced upper tract urothelial carcinoma: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2019, 135, 59–65. [Google Scholar] [CrossRef]
- Massari, F.; Santoni, M.; di Nunno, V.; Cheng, L.; Lopez-Beltran, A.; Cimadamore, A.; Gasparrini, S.; Scarpelli, M.; Battelli, N.; Montironi, R. Adjuvant and neoadjuvant approaches for urothelial cancer: Updated indications and controversies. Cancer Treat. Rev. 2018, 68, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Joshi, M.; Meijer, R.P.; Glantz, M.; Holder, S.; Harvey, H.A.; Kaag, M.; Fransen van de Putte, E.E.; Horenblas, S.; Drabick, J.J. Neoadjuvant chemotherapy for muscle invasive bladder cancer: A systematic Review and Two-Step Meta-Analysis. Oncologist 2016, 21, 708–715. [Google Scholar] [CrossRef]
- Rimar, K.J.; Glaser, A.P.; Kundu, S.; Schaeffer, E.M.; Meeks, J.; Psutka, S.P. Changes in Lean Muscle Mass Associated with Neoadjuvant Platinum-Based Chemotherapy in Patients with Muscle Invasive Bladder Cancer. Bladder Cancer 2018, 4, 411–418. [Google Scholar] [CrossRef] [PubMed]
- von der Maase, H.; Sengelov, L.; Roberts, J.T.; Ricci, S.; Dogliotti, L.; Oliver, T.; Moore, M.J.; Zimmermann, A.; Arning, M. Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin with methotrexate vinblastine doxorubicin plus cisplatin in patients with bladder cancer. J. Clin. Oncol. 2005, 23, 4602–4608. [Google Scholar] [CrossRef] [PubMed]
- von der Maase, H.; Sengelov, L.; Roberts, J.T.; Ricci, S.; Dogliotti, L.; Oliver, T.; Moore, M.J.; Zimmermann, A.; Arning, M. Randomized phase II/III trial assessing gemcitabine/carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer who are unfit for cisplatin-based chemotherapy EORTC study 30986. J. Clin. Oncol. 2012, 30, 191–199. [Google Scholar] [CrossRef]
- Galsky, M.D.; Chen, G.J.; Oh, W.K.; Bellmunt, J.; Roth, B.J.; Petrioli, R.; Dogliotti, L.; Dreicer, R.; Sonpavde, G. Comparative effectiveness of cisplatin-based and carboplatin-based chemotherapy for treatment of advanced urothelial carcinoma. Ann. Oncol. 2012, 23, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Locke, J.A.; Pond, G.R.; Sonpavde, G.; Necchi, A.; Giannatempo, P.; Paluri, R.K.; Niegisch, G.; Albers, P.; Buonerba, C.; Di Lorenzo, G.; et al. Efficacy and safety of gemcitabine plus either taxane or carboplatin in the first- line setting of metastatic urothelial carcinoma: A systematic review and meta- analysis. Clin. Genitourin. Cancer 2016, 14, 331–340. [Google Scholar] [CrossRef]
- Bellmunt, J.; Fougeray, R.; Rosenberg, J.E.; von der Maase, H.; Schutz, F.A.; Salhi, Y.; Culine, S.; Choueiri, T.K. Long-term survival results of a randomized phase III trial of vinflunine plus best supportive care versus best supportive care alone in advanced urothelial carcinoma patients after failure of platinum- based chemotherapy. Ann Oncol. 2013, 24, 1466–1472. [Google Scholar] [CrossRef] [PubMed]
- Costantini, C.; Millard, F. Update on chemotherapy in the treatment of urothelial carcinoma. Sci. World J. 2011, 11, 1981–1994. [Google Scholar] [CrossRef] [PubMed]
- Albers, P.; Park, S.I.; Niegisch, G.; Fechner, G.; Steiner, U.; Lehmann, J.; Heimbach, D.; Heidenreich, A.; Fimmers, R.; Siener, R. AUO Bladder Cancer Group. Randomized phase III trial of 2nd line gemcitabine and paclitaxel chemotherapy in patients with advanced bladder cancer: Short-term versus prolonged treatment [German Association of Urological Oncology (AUO) trial AB 20/99]. Ann. Oncol. 2011, 22, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Naiki, T.; Kawai, N.; Hashimoto, Y.; Okamura, T.; Ando, R.; Yasui, T.; Okada, A.; Etani, T.; Tozawa, K.; Kohri, K. Gemcitabine and docetaxel, an effective second-line chemotherapy for lung metastasis of urothelial carcinoma. Int. J. Clin. Oncol. 2014, 19, 516–522. [Google Scholar] [CrossRef]
- Schepisi, G.; Santoni, M.; Massari, F.; Gurioli, G.; Salvi, S.; Conteduca, V.; Montironi, R.; De Giorgi, U. Urothelial cancer: Inflammatory mediators and implications for immunotherapy. BioDrugs 2016, 30, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Massari, F.; Ciccarese, C.; Vau, N.; Santoni, M.; Montironi, R.; Cheng, L.; Marques, R.C.; Scarpelli, M.; Fonseca, J.; Matrana, M.R.; et al. Emerging immunotargets in bladder cancer. Curr. Drug Targets 2016, 17, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Bellmunt, J.; de Wit, R.; Vaughn, D.J.; Fradet, Y.; Lee, J.L.; Fong, L.; Vogelzang, N.J.; Climent, M.A.; Petrylak, D.P.; Choueiri, T.K.; et al. KEYNOTE-045 Investigators. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N. Engl. J. Med. 2017, 376, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Durán, I.; van der Heijden, M.S.; Loriot, Y.; Vogelzang, N.J.; De Giorgi, U.; Oudard, S.; Retz, M.M.; Castellano, D.; Bamias, A.; et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): A multicentre, open-label, phase 3 randomised controlled trial. Lancet 2018, 391, 748–757. [Google Scholar] [CrossRef]
- Massari, F.; Di Nunno, V.; Cubelli, M.; Santoni, M.; Fiorentino, M.; Montironi, R.; Cheng, L.; Lopez-Beltran, A.; Battelli, N.; Ardizzoni, A. Immune checkpoint inhibitors for metastatic bladder cancer. Cancer Treat. Rev. 2018, 64, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Massari, F.; Di Nunno, V. Atezolizumab for platinum-treated metastatic urothelial carcinoma. Lancet 2018, 391, 716–718. [Google Scholar] [CrossRef]
- Fukushima, H.; Yokoyama, M.; Nakanishi, Y.; Tobisu, K.; Koga, F. Sarcopenia as a prognostic biomarker of advanced urothelial carcinoma. PLoS ONE 2015, 10, e0115895. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Takei, K.; Uematsu, T.; Tokura, Y.; Suzuki, I.; Sakamoto, K.; Nishihara, D.; Yamaguchi, Y.; Mizuno, T.; Nukui, A.; et al. Significance of sarcopenia as a prognostic factor for metastatic urothelial carcinoma patients treated with systemic chemotherapy. Int. J. Clin. Oncol. 2018, 23, 338–346. [Google Scholar] [CrossRef]
- Leiter, A.; Doucette, J.; Krege, S.; Lin, C.C.; Hahn, N.; Ecke, T.; Sonpavde, G.; Bamias, A.; Oh, W.K.; Galsky, M.D. Obesity and Outcomes in Patients with Metastatic Urothelial Carcinoma. Bl. Cancer 2016, 2, 341–349. [Google Scholar] [CrossRef]
- Cortellini, A.; Bersanelli, M.; Buti, S.; Cannita, K.; Santini, D.; Perrone, F.; Giusti, R.; Tiseo, M.; Michiara, M.; Di Marino, P.; et al. A multicenter study of body mass index in cancer patients treated with anti-PD-1/PD-L1 immune checkpoint inhibitors: When overweight becomes favorable. J. Immunother. Cancer 2019, 7, 57. [Google Scholar] [CrossRef]
- Lopez-Beltran, A.; Santoni, M.; Massari, F.; Ciccarese, C.; Tortora, G.; Cheng, L.; Moch, H.; Scarpelli, M.; Montironi, R. Bladder cancer: Determinants of personalized therapy. Curr. Drug Targets 2015, 16, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Massari, F.; Ciccarese, C.; Santoni, M.; Brunelli, M.; Conti, A.; Modena, A.; Montironi, R.; Santini, D.; Cheng, L.; Martignoni, G.; et al. The route to personalized medicine in bladder cancer: Where do we stand? Target. Oncol. 2015, 10, 325–336. [Google Scholar] [CrossRef]
- Buti, S.; Ciccarese, C.; Zanoni, D.; Santoni, M.; Modena, A.; Maines, F.; Gilli, G.; Bria, E.; Brunelli, M.; Rimanti, A.; et al. Prognostic and predictive factors in patients treated with chemotherapy for advanced urothelial cancer: Where do we stand? Future Oncol. 2015, 11, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Massari, F.; Santoni, M.; Ciccarese, C.; Brunelli, M.; Conti, A.; Santini, D.; Montironi, R.; Cascinu, S.; Tortora, G. Emerging concepts on drug resistance in bladder cancer: Implications for future strategies. Crit. Rev. Oncol. Hemat. 2015, 96, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Alam, J.; Venkatesan, M.I.; Eiguren-Fernandez, A.; Schmitz, D.; Di Stefano, E.; Slaughter, N.; Killeen, E.; Wang, X.; Huang, A.; et al. Nrf2 is a key transcription factor that regulates antioxidant defense in macrophages and epithelial cells: Protecting against the proinflammatory and oxidizing effects of diesel exhaust chemicals. J. Immunol. 2004, 173, 3467–3481. [Google Scholar] [CrossRef]
- Hayden, A.; Douglas, J.; Sommerlad, M.; Andrews, L.; Gould, K.; Hussain, S.; Thomas, G.J.; Packham, G.; Crabb, S.J. The Nrf2 transcription factor contributes to resistance to cisplatin in bladder cancer. Urol Oncol. 2014, 32, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.A.; Lee, I.K. The role of Nrf2: Adipocyte differentiation, obesity, and insulin resistance. Oxid. Med. Cell. Longev. 2013, 2013, 184598. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhou, S.; Jiang, X.; Wang, Y.H.; Li, F.; Wang, Y.G.; Zheng, Y.; Cai, L. The role of the Nrf2/Keap1 pathway in obesity and metabolic syndrome. Rev. Endocr. Metab. Disord. 2015, 16, 35–45. [Google Scholar] [CrossRef]
- Font, A.; Taron, M.; Gago, J.L.; Costa, C.; Sánchez, J.J.; Carrato, C.; Mora, M.; Celiz, P.; Perez, L.; Rodríguez, D.; et al. BRCA1 mRNA expression and outcome to neoadjuvant cisplatin-based chemotherapy in bladder cancer. Ann. Oncol. 2011, 22, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Manders, P.; Pijpe, A.; Hooning, M.J.; Kluijt, I.; Vasen, H.F.; Hoogerbrugge, N.; van Asperen, C.J.; Meijers-Heijboer, H.; Ausems, M.G.; van Os, T.A.; et al. Body weight and risk of breast cancer in BRCA1/2 mutation carriers. Breast Cancer Res. Treat. 2011, 126, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Santoni, M.; Scarpelli, M.; Mazzucchelli, R.; Lopez-Beltran, A.; Cheng, L.; Epstein, J.I.; Cascinu, S.; Briganti, A.; Catto, J.W.; Montorsi, F.; et al. Current Histopathologic and Molecular Characterizations of Prostate cancer: Towards Individualized Prognosis and Therapies. Eur. Urol. 2015, 69, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Santoni, M.; Piva, F.; Scarpelli, M.; Cheng, L.; Lopez-Beltran, A.; Massari, F.; Iacovelli, R.; Berardi, R.; Santini, D.; Montironi, R. The origin of prostate metastases: Emerging insights. Cancer Metastasis Rev. 2015, 34, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Piva, F.; Santoni, M.; Scarpelli, M.; Briganti, A.; Montorsi, F.; Montironi, R. “Tracking the Origin of Metastatic Prostate Cancer”. Role of Evolutionary algorithms, Adaptive therapy, and Single cell analysis. Eur. Urol. 2015, 68, e134–e135. [Google Scholar] [CrossRef]
- Baca, S.C.; Prandi, D.; Lawrence, M.S.; Mosquera, J.M.; Romanel, A.; Drier, Y.; Park, K.; Kitabayashi, N.; MacDonald, T.Y.; Ghandi, M.; et al. Punctuated evolution of prostate cancer genomes. Cell 2013, 153, 666–677. [Google Scholar] [CrossRef] [PubMed]
- Ciccarese, C.; Santoni, M.; Massari, F.; Modena, A.; Piva, F.; Conti, A.; Mazzucchelli, R.; Cheng, L.; Lopez-Beltran, A.; Scarpelli, M.; et al. Metabolic alterations in Renal and Prostate cancer. Curr. Drug Metab. 2016, 17, 150–155. [Google Scholar] [CrossRef]
- Santoni, M.; Cheng, L.; Conti, A.; Mariani, C.; Lopez-Beltran, A.; Montironi, R.; Battelli, N. Activity and functions of tumor-associated macrophages in prostate carcinogenesis. Eur. Urol. Suppl. 2017, 16, 301–308. [Google Scholar] [CrossRef]
- Minardi, D.; Scartozzi, M.; Montesi, L.; Santoni, M.; Burattini, L.; Bianconi, M.; Lacetera, V.; Milanese, G.; Cascinu, S.; Muzzonigro, G. Neutrophil-to-lymphocyte ratio may be associated with the outcome in patients with prostate cancer. Springerplus 2015, 4, 255. [Google Scholar] [CrossRef] [PubMed]
- Montironi, R.; Santoni, M.; Sotte, V.; Cheng, L.; Lopez-Beltran, A.; Massari, F.; Matrana, M.R.; Moch, H.; Berardi, R.; Scarpelli, M. Emerging immunotargets and immunotherapies in prostate cancer. Curr. Drug Targets 2016, 20, 111–117. [Google Scholar] [CrossRef]
- Santoni, M.; Conti, A.; Burattini, L.; Berardi, R.; Scarpelli, M.; Cheng, L.; Lopez-Beltran, A.; Cascinu, S.; Montironi, R. Neuroendocrine differentiation in prostate cancer: Novel morphological insights and future therapeutic perspectives. BBA Cancer Rev. 2014, 1846, 630–637. [Google Scholar] [CrossRef]
- Santoni, M.; Aurilio, G.; Maccioni, A.; Nolè, F.; Battelli, N. Key players of neuroendocrine differentiation in prostate cancer. Ann. Transl. Med. 2019, 7. In press. [Google Scholar] [CrossRef]
- Massari, F.; Mollica, V.; Di Nunno, V.; Gatto, L.; Santoni, M.; Scarpelli, M.; Cimadamore, A.; Lopez-Beltran, A.; Cheng, L.; Battelli, N.; et al. The human microbiota and prostate cancer: Friend or foe? Cancers 2019, 11, 459. [Google Scholar] [CrossRef]
- Ciccarese, C.; Santoni, M.; Brunelli, M.; Buti, S.; Modena, A.; Nabissi, M.; Artibani, W.; Martignoni, G.; Montironi, R.; Tortora, G.; et al. AR-V7 and prostate cancer: The watershed for treatment selection? Cancer Treat. Rev. 2015, 43, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Mollica, V.; Di Nunno, V.; Cimadamore, A.; Lopez-Beltran, A.; Cheng, L.; Santoni, M.; Scarpelli, M.; Montironi, R.; Massari, F. Molecular mechanisms related to hormone inhibition resistance in prostate cancer. Cells 2019, 8, 43. [Google Scholar] [CrossRef]
- Montironi, R.; Santoni, M.; Mazzucchelli, R.; Burattini, L.; Berardi, R.; Galosi, A.B.; Cheng, L.; Lopez-Beltran, A.; Briganti, A.; Montorsi, F.; et al. Prostate cancer: From Gleason scoring to prognostic grade grouping. Exp. Rev. Anticancer Ther. 2016, 16, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Riondino, S.; Roselli, M.; Palmirotta, R.; Della-Morte, D.; Ferroni, P.; Guadagni, F. Obesity and colorectal cancer: Role of adipokines in tumor initiation and progression. World J. Gastroenterol. 2014, 20, 5177–5190. [Google Scholar] [CrossRef] [PubMed]
- Conteduca, V.; Caffo, O.; Galli, L.; Maugeri, A.; Scarpi, E.; Maines, F.; Chiuri, V.E.; Lolli, C.; Kinspergher, S.; Schepisi, G.; et al. Association among metabolic syndrome, inflammation, and survival in prostate cancer. Urol. Oncol. 2018, 35, 240. [Google Scholar] [CrossRef]
- Conteduca, V.; Caffo, O.; Derosa, L.; Veccia, A.; Petracci, E.; Chiuri, V.E.; Santoni, M.; Santini, D.; Fratino, L.; Maines, F.; et al. Metabolic Syndrome in Castration-Resistant Prostate Cancer Patients Treated with Abiraterone. Prostate 2015, 75, 1329–1338. [Google Scholar] [CrossRef]
- Ellem, S.J.; Wang, H.; Poutanen, M.; Risbridger, G.P. Increased endogenous estrogen synthesis leads to the sequential induction of prostatic inflammation (prostatitis) and prostatic pre-malignancy. Am. J. Pathol. 2009, 175, 1187–1199. [Google Scholar] [CrossRef]
- Sfanos, K.S.; Wilson, B.A.; De Marzo, A.M.; Isaacs, W.B. Acute inflammatory proteins constitute the organic matrix of prostatic corpora amylacea and calculi in men with prostate cancer. Proc. Natl. Acad. Sci. USA 2009, 106, 3443–3448. [Google Scholar] [CrossRef]
- Nakai, Y.; Nonomura, N. Inflammation and prostate carcinogenesis. Int. J. Urol. 2013, 20, 150–160. [Google Scholar] [CrossRef]
- Saltiel, A.R.; Olefsky, J.M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Invest. 2017, 127, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.H.; LeRoith, D. Obesity, type 2 diabetes, and cancer: The insulin and IGF connection. Endocr. Relat. Cancer 2012, 19, F27–F45. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Narita, S.; Inoue, T.; Tsuchiya, N.; Satoh, S.; Nanjo, H.; Sasaki, T.; Habuchi, T. Diet-induced macrophage inhibitory cytokine 1 promotes prostate cancer progression. Endocr. Relat. Cancer 2013, 21, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Tseng, C.; Zhang, Y.; Sirin, O.; Corn, P.G.; Li-Ning-Tapia, E.M.; Troncoso, P.; Davis, J.; Pettaway, C.; Ward, J.; et al. CXCL1 mediates obesity-associated adipose stromal cell trafficking and function in the tumour microenvironment. Nat. Commun. 2016, 7, 11674. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Catalano, S.; Gelsomino, L.; Marsico, S.; Giordano, C.; Panza, S.; Bonofiglio, D.; Bossi, G.; Covington, K.R.; Fuqua, S.A.; et al. Leptin mediates tumor-stromal interactions that promote the invasive growth of breast cancer cells. Cancer Res. 2012, 72, 1416–1427. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E.; Makowski, L.; Di Giovanni, J.; Kolonin, M.G. Cancer as a Matter of Fat: The Crosstalk between Adipose Tissue and Tumors. Trends Cancer 2018, 4, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Ahn, S.; Saha, A.; DiGiovanni, J.; Kolonin, M.G. Adipose stromal cell targeting suppresses prostate cancer epithelial-mesenchymal transition and chemoresistance. Oncogene 2019, 38, 1979–1988. [Google Scholar] [CrossRef]
- Tannock, I.F.; de Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Théodore, C.; James, N.D.; Turesson, I.; et al. TAX 327 Investigators. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.J.; Halabi, S.; de Wit, R.; Tannock, I.F.; Eisenberger, M. The relationship of body mass index and serum testosterone with disease outcomes in men with castration-resistant metastatic prostate cancer. Prostate Cancer Prostatic Dis. 2009, 12, 88–93. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, W.; Liu, X.; Chaftari, P.; Cruz Carreras, M.T.; Gonzalez, C.; Viets-Upchurch, J.; Merriman, K.; Tu, S.M.; Dalal, S.; Yeung, S.C. Association of body composition with outcome of docetaxel chemotherapy in metastatic prostate cancer: A retrospective review. PLoS ONE 2015, 10, e0122047. [Google Scholar] [CrossRef]
- Cushen, S.J.; Power, D.G.; Murphy, K.P.; McDermott, R.; Griffin, B.T.; Lim, M.; Daly, L.; MacEneaney, P.; O’ Sullivan, K.; Prado, C.M.; et al. Impact of body composition parameters on clinical outcomes in patients with metastatic castrate-resistant prostate cancer treated with docetaxel. Clin. Nutr. ESPEN 2016, 13, e39–e45. [Google Scholar] [CrossRef]
- Cavo, A.; Rubagotti, A.; Zanardi, E.; Fabbroni, C.; Zinoli, L.; Di Meglio, A.; Arboscello, E.; Bellodi, A.; Spallarossa, P.; Cattrini, C.; et al. Abiraterone acetate and prednisone in the pre- and post-docetaxel setting for metastatic castration-resistant prostate cancer: A mono-institutional experience focused on cardiovascular events and their impact on clinical outcomes. Ther. Adv. Med. Oncol. 2018, 10. [Google Scholar] [CrossRef]
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.E.; Sternberg, C.N.; Miller, K.; de Wit, R.; Mulders, P.; Chi, K.N.; Shore, N.D.; et al. AFFIRM Investigators. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 2012, 367, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Fizazi, K.; Scher, H.I.; Molina, A.; Logothetis, C.J.; Chi, K.N.; Jones, R.J.; Staffurth, J.N.; North, S.; Vogelzang, N.J.; Saad, F.; et al. COU-AA-301 Investigators. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: Final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012, 13, 983–992. [Google Scholar] [CrossRef]
- Antoun, S.; Bayar, A.; Ileana, E.; Laplanche, A.; Fizazi, K.; di Palma, M.; Escudier, B.; Albiges, L.; Massard, C.; Loriot, Y. High subcutaneous adipose tissue predicts the prognosis in metastatic castration-resistant prostate cancer patients in post chemotherapy setting. Eur. J. Cancer 2015, 51, 2570–2577. [Google Scholar] [CrossRef]
- Frantellizzi, V.; Farcomeni, A.; Follacchio, G.A.; Pacilio, M.; Pellegrini, R.; Pani, R.; De Vincentis, G. 3-variable prognostic score (3-PS) for overall survival prediction in metastatic castration-resistant prostate cancer treated with 223Radium-dichloride. Ann. Nucl. Med. 2018, 32, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Loeb, K.R.; Loeb, L.A. Significance of multiple mutations in cancer. Carcinogenesis 2000, 21, 379–385. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef]
- Hursting, S.D.; Lashinger, L.M.; Colbert, L.H.; Rogers, C.J.; Wheatley, K.W.; Nunez, N.P.; Mahabir, S.; Barrett, J.C.; Forman, M.R.; Perkins, S.N. Energy balance and carcinogenesis: Underlying pathways and targets for intervention. Curr. Cancer Drug Targets 2007, 7, 484–491. [Google Scholar] [CrossRef] [PubMed]
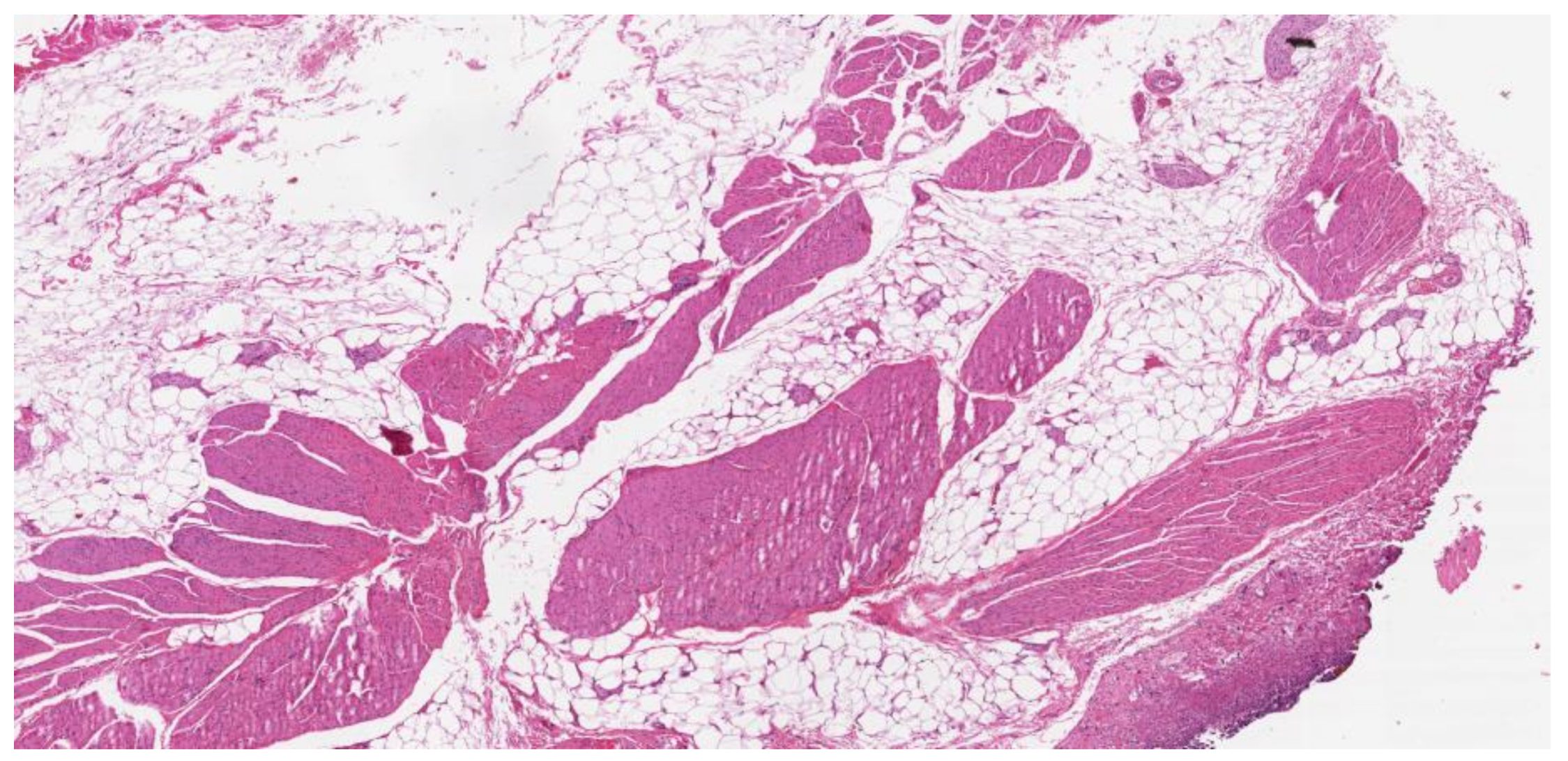
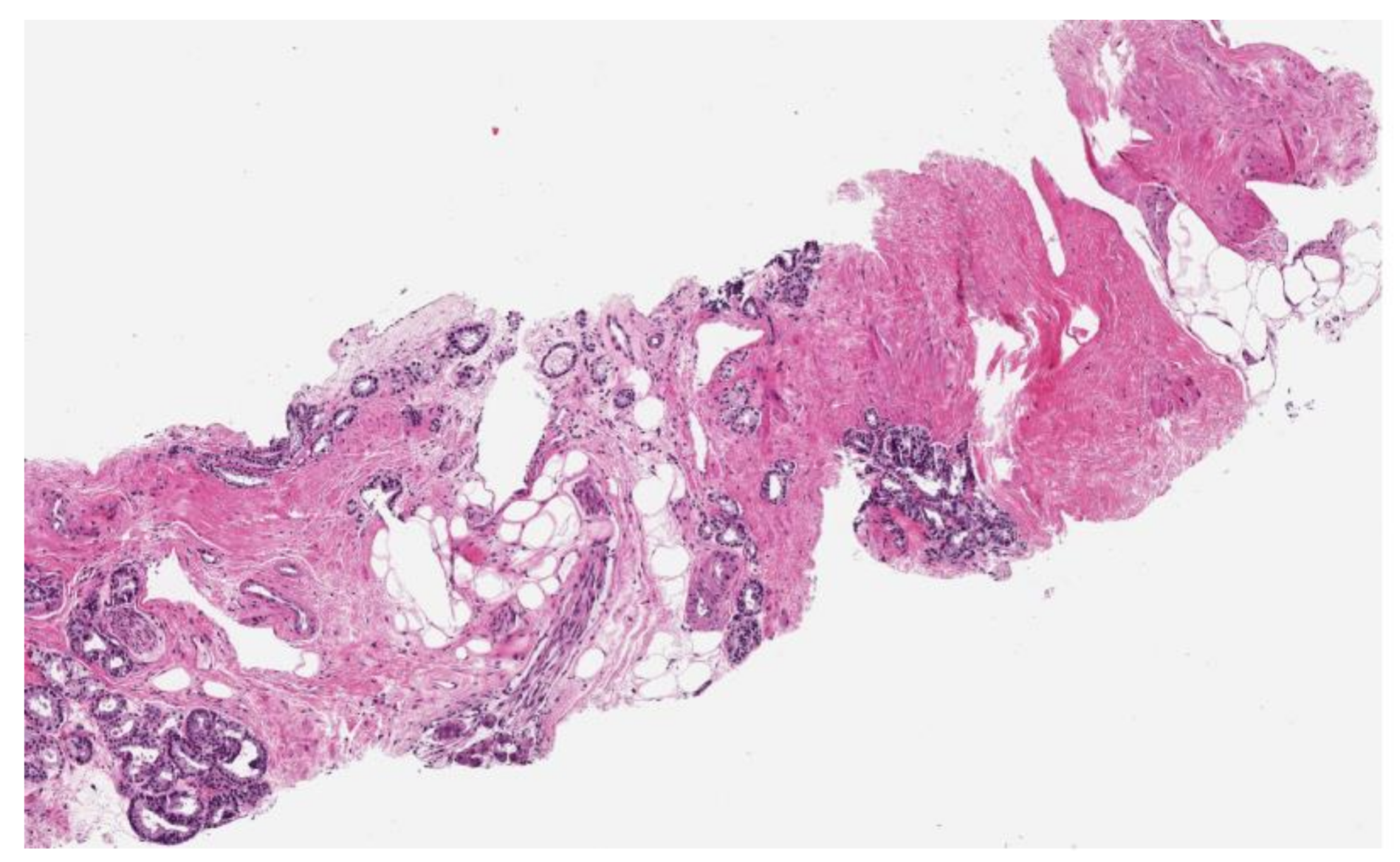
| Patients | Results | Reference |
|---|---|---|
| 88 mUC patients | SMI was a significant and independent predictor of shorter OS (HR 0.90, p < 0.001). Median OS rates were 11 and 31 months for sarcopenic and non-sarcopenic patients; sarcopenia was a significant and independent predictor of shorter OS (HR 3.36, p < 0.001) | Fukushima et al., 2015 [48] |
| 87 mUC patients who underwent chemotherapy | SMI stratified by the value of the BMI was a significant predictor of shorter OS in univariate analysis (p = 0.037) HR = 3.102; p = 0.026 | Abe et al., 2018 [49] |
| 537 mUC patients treated with cisplatin-based combination therapy | Embolic events and renal failure were higher in patients with an average or higher BSA. Obese patients have similar response rates, survival outcomes, and tolerability of cisplatin-based therapy to non-obese patients. | Leiter et al., 2016 [50] |
| 976 patients with different tumors: NSCLC (65.1%), melanoma (18.7%), RCC (13.8%) and others (2.4%) | Median TTF, PFS and OS were significantly longer for overweight/obese patients in univariate (p < 0.0001, for all the survival intervals) and multivariate models. | Cortellini et al., 2019 [51] |
| Drug | Patients/Cells | Results | Reference |
|---|---|---|---|
| Docetaxel | 1006 CRPC patients | Obesity was associated with younger age, lower PSA and tALP, and higher performance status, primary Gleason sum, testosterone and Hb. In multivariate analysis, neither BMI, presence of obesity, nor baseline testosterone was significantly associated with OS or PSA declines. | Armstrong et al., 2009 [91] |
| 333 mCRPC patients | High VMR, obesity, and weekly regimens were significant predictors of longer survival after docetaxel. | Wu et al., 2015 [92] | |
| 63 mCRPC patients | In multivariate analysis, BMI ≥25 kg/m2 (HR: 0.349, CI: 0.156–0.782, p = 0.010) was a significant predictor of longer OS and both visceral fat index ≥ median 58.7 cm2/m2 (HR: 2.266 CI: 1.066–4.814, p = 0.033) and anaemia (HR: 2.81, CI: 1.297–6.091, p = 0.009) were significant predictors of shorter OS. | Cushen et al., 2016 [93] | |
| Cabazitaxel | Human cell co-culture models | ASC-mediated chemoresistance to cabazitaxel. ASC induce epithelial-mesenchymal transition in prostate cancer cells. | Su et al., 2019 [89] |
| Abiraterone acetate and Enzalutamide | 105 patients | At multivariable analysis BMI >25 kg/m2 resulted significantly associated both with worse progression-free survival and worse OS (p = 0.03 and p = 0.042, respectively) | Cavo et al., 2018 [94] |
| 120 patients mCRPC | High volume of SAT is independently associated with OS. | Antoun et al., 2015 [97] | |
| Radium223 | 92 mCRPC patients | Patients’ weight, BMI, ECOG-PS, Hb and tALP significantly correlated with OS at univariate analysis, while only ECOG-PS and Hb levels were significant predictors of OS at multivariate analysis. | Frantellizzi et al., 2018 [98] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santoni, M.; Cimadamore, A.; Massari, F.; Piva, F.; Aurilio, G.; Martignetti, A.; Scarpelli, M.; Di Nunno, V.; Gatto, L.; Battelli, N.; et al. Key Role of Obesity in Genitourinary Tumors with Emphasis on Urothelial and Prostate Cancers. Cancers 2019, 11, 1225. https://doi.org/10.3390/cancers11091225
Santoni M, Cimadamore A, Massari F, Piva F, Aurilio G, Martignetti A, Scarpelli M, Di Nunno V, Gatto L, Battelli N, et al. Key Role of Obesity in Genitourinary Tumors with Emphasis on Urothelial and Prostate Cancers. Cancers. 2019; 11(9):1225. https://doi.org/10.3390/cancers11091225
Chicago/Turabian StyleSantoni, Matteo, Alessia Cimadamore, Francesco Massari, Francesco Piva, Gaetano Aurilio, Angelo Martignetti, Marina Scarpelli, Vincenzo Di Nunno, Lidia Gatto, Nicola Battelli, and et al. 2019. "Key Role of Obesity in Genitourinary Tumors with Emphasis on Urothelial and Prostate Cancers" Cancers 11, no. 9: 1225. https://doi.org/10.3390/cancers11091225
APA StyleSantoni, M., Cimadamore, A., Massari, F., Piva, F., Aurilio, G., Martignetti, A., Scarpelli, M., Di Nunno, V., Gatto, L., Battelli, N., Cheng, L., Lopez-Beltran, A., & Montironi, R. (2019). Key Role of Obesity in Genitourinary Tumors with Emphasis on Urothelial and Prostate Cancers. Cancers, 11(9), 1225. https://doi.org/10.3390/cancers11091225










