Mining Prognosis Index of Brain Metastases Using Artificial Intelligence
Abstract
1. Introduction
2. Results
2.1. Clinical Characteristics and Overall Survival
2.2. Qualitative Feature Analysis
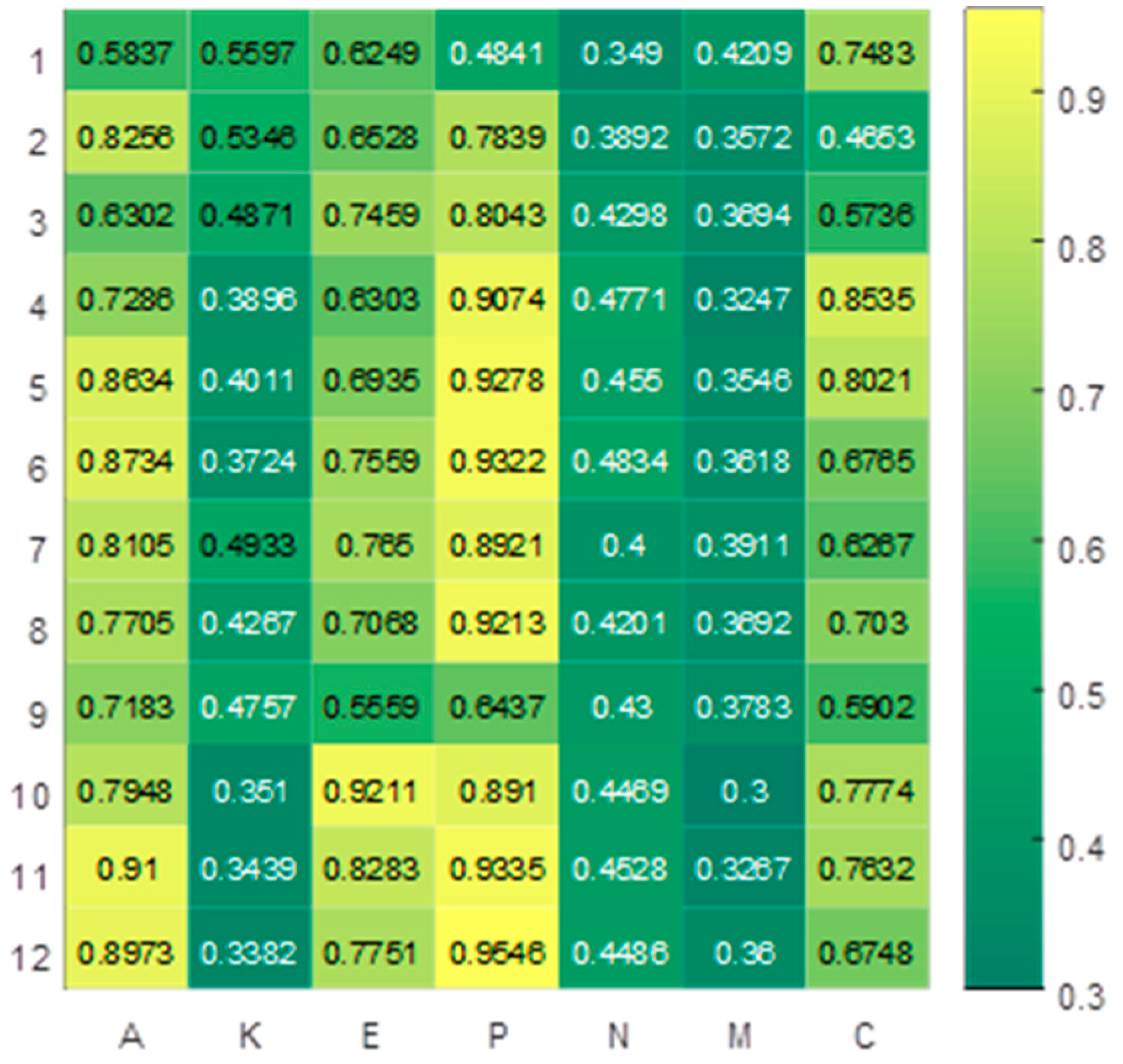
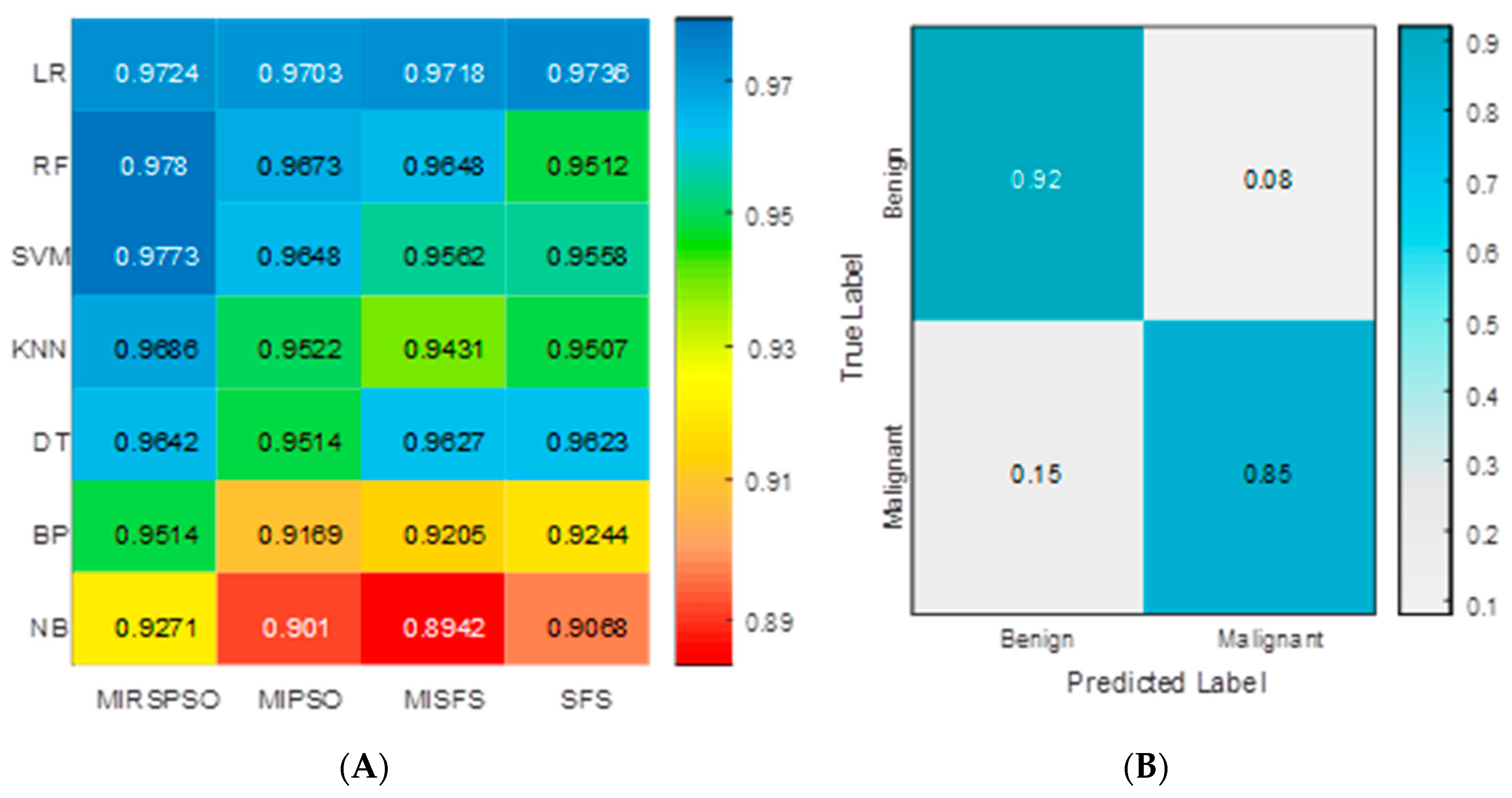
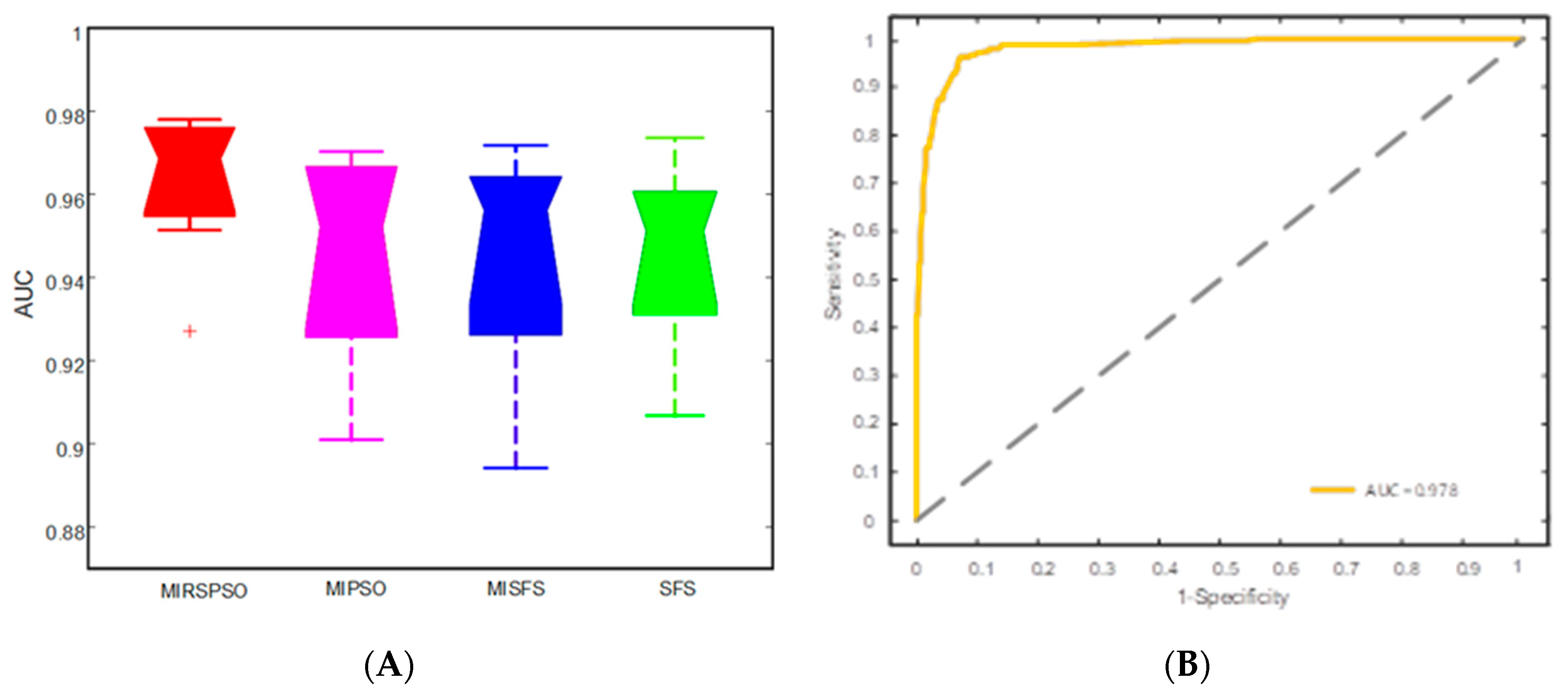
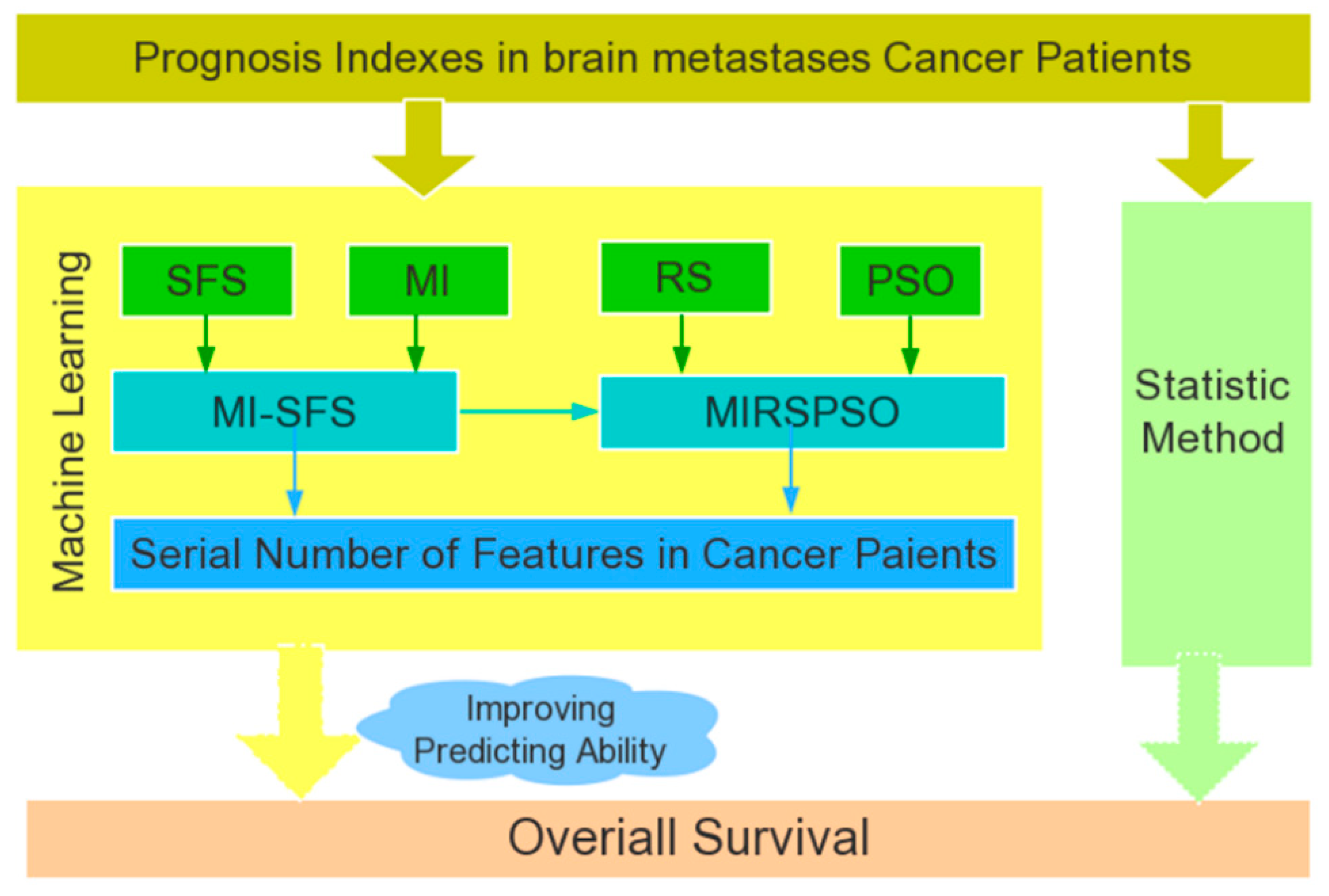
2.3. Feature Selection and Prognosis Prediction
3. Discussion
4. Materials and Methods
4.1. Patient Enrollment
4.2. Treatment Process
4.3. Patients Survival and Prognostic Factors
4.4. Feature Selection with MIRSPSO
4.5. Feature Classification Methods
4.6. Identification of Excellent Performance Groups
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PI | Prognostic Index |
| BM | Brain Metastases |
| NSCLC | Non-small Cell Lung Cancer |
| RPA | Recursive Partitioning Analysis |
| SIR | Score Index for Radiosurgery |
| GPA | Graded Prognostic Assessment |
| BSBM | Basic Score for Brain Metastases |
| AUC | Area under the receiver operating characteristic curve |
| SD | Standard deviation |
| LR | Logistic Regression |
| SVM | Support Vector Machine |
| RF | Random Forest |
| DC | Distance Correlation |
| MIRSPSO | Mutual Information and Rough set with Particle Swarm Optimization |
| NB | Naive Bayes |
| MST | Median Survival Time |
| WBRT | Whole Brain Radiotherapy |
| SRS | Stereotactic Radiosurgery |
| MRI | Magnetic Resonance Imaging |
| OS | Overall Survival |
| K-M | Kaplan-Meier |
| KPS | Karnofsky Performance Status |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Kattan, M.W.; Hess, K.R.; Amin, M.B.; Lu, Y.; Moons, K.G.; Gershenwald, J.E.; Gimotty, P.A.; Guinney, J.H.; Halabi, S.; Lazar, A.J.; et al. American Joint Committee on Cancer acceptance criteria for inclusion of risk models for individualized prognosis in the practice of precision medicine. CA Cancer J.Clin. 2016, 66, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.; Wolfe, R.; Ball, D.; Conron, M.; Stirling, R.G.; Solomon, B.; MacManus, M.; Officer, A.; Karnam, S.; Burbury, K.; et al. Lung cancer prognostic index: A risk score to predict overall survival after the diagnosis of non-small-cell lung cancer. Br. J. Cancer 2017, 117, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Fujima, N.; Shimizu, Y.; Yoshida, D.; Kano, S.; Mizumachi, T.; Homma, A.; Yasuda, K.; Onimaru, R.; Sakai, O.; Kudo, K.; et al. Machine-Learning-Based Prediction of Treatment Outcomes Using MR Imaging-Derived Quantitative Tumor Information in Patients with Sinonasal Squamous Cell Carcinomas: A Preliminary Study. Cancers 2019, 11, 800. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.K. Primary tumor location found to impact prognosis and response to therapy in patients with metastatic colorectal cancer. CA Cancer J.Clin. 2017, 67, 259–260. [Google Scholar] [CrossRef] [PubMed]
- Sparano, J.A.; Gray, R.J.; Ravdin, P.M.; Makower, D.F.; Pritchard, K.I.; Albain, K.S.; Hayes, D.F.; Geyer, J.C.; Dees, E.C.; Goetz, M.P. Clinical and Genomic Risk to Guide the Use of Adjuvant Therapy for Breast Cancer. N. Engl.J. Med. 2019, 380, 2395–2405. [Google Scholar] [CrossRef]
- You, R.; Liu, Y.P.; Lin, M.; Huang, P.Y.; Tang, L.Q.; Zhang, Y.N.; Pan, Y.; Liu, W.L.; Guo, W.B.; Zou, X.; et al. Relationship of circulating tumor cells and Epstein-Barr virus DNA to progression-free survival and overall survival in metastatic nasopharyngeal carcinoma patients. Int. J. Cancer 2019. [Google Scholar] [CrossRef]
- Lan, K.; Wang, D.T.; Fong, S.; Liu, L.S.; Wong, K.K.L.; Dey, N. A Survey of Data Mining and Deep Learning in Bioinformatics. J.Med. Syst. 2018, 42, 139. [Google Scholar] [CrossRef]
- Lu, T.P.; Kuo, K.T.; Chen, C.H.; Chang, M.C.; Lin, H.P.; Hu, Y.H.; Chiang, Y.C.; Cheng, W.F.; Chen, C.A. Developing a Prognostic Gene Panel of Epithelial Ovarian Cancer Patients by a Machine Learning Model. Cancers 2019, 11, 270. [Google Scholar] [CrossRef]
- Sun, Y.; Goodison, S.; Li, J.; Liu, L.; Farmerie, W. Improved breast cancer prognosis through the combination of clinical and genetic markers. Bioinformatics 2007, 23, 30–37. [Google Scholar] [CrossRef]
- Smith, A.K.; White, D.B.; Arnold, R.M. Uncertainty—The other side of prognosis. N. Engl. J. Med. 2013, 368, 2448–2450. [Google Scholar] [CrossRef] [PubMed]
- Qian, D.; Liu, H.; Wang, X.; Ge, J.; Luo, S.; Patz, E.F., Jr.; Moorman, P.G.; Su, L.; Shen, S.; Christiani, D.C.; et al. Potentially functional genetic variants in the complement-related immunity gene-set are associated with non-small cell lung cancer survival. Int.J. Cancer 2019, 144, 1867–1876. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.F.; Hsu, F.T.; Hsieh, K.L.; Kao, Y.J.; Cheng, S.J.; Hsu, J.B.; Tsai, P.H.; Chen, R.J.; Huang, C.C.; Yen, Y.; et al. Machine Learning-Based Radiomics for Molecular Subtyping of Gliomas. Clin. Cancer Res. 2018, 24, 4429–4436. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, E.D.; Simunovic, M.; Jhaveri, K.; Kirsch, R.; Brierley, J.; Drolet, S.; Brown, C.; Vos, P.M.; Xiong, W.; MacLean, T. Safety and feasibility of using magnetic resonance imaging criteria to identify patients with “good prognosis” rectal cancer eligible for primary surgery: The phase 2 nonrandomized QuickSilver clinical trial. JAMA Oncol. 2019, 5, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Wilensky, G.R. Bedside Computer Vision—Moving Artificial Intelligence from Driver Assistance to Patient Safety. N. Engl.J.Med. 2018, 378, 1269–1271. [Google Scholar] [CrossRef] [PubMed]
- Hana, T.; Tanaka, S.; Nejo, T.; Takahashi, S.; Kitagawa, Y.; Koike, T.; Nomura, M.; Takayanagi, S.; Saito, N. Mining-Guided Machine Learning Analyses Revealed the Latest Trends in Neuro-Oncology. Cancers 2019, 11, 178. [Google Scholar] [CrossRef] [PubMed]
- Ferroni, P.; Zanzotto, F.M.; Riondino, S.; Scarpato, N.; Guadagni, F.; Roselli, M. Breast Cancer Prognosis Using a Machine Learning Approach. Cancers 2019, 11, 328. [Google Scholar] [CrossRef]
- Burki, T.K. Predicting lung cancer prognosis using machine learning. Lancet Oncol. 2016, 17, e421. [Google Scholar] [CrossRef]
- Kourou, K.; Exarchos, T.P.; Exarchos, K.P.; Karamouzis, M.V.; Fotiadis, D.I. Machine learning applications in cancer prognosis and prediction. Comput. Struct. Biotechnol. J. 2015, 138, 8–17. [Google Scholar] [CrossRef]
- Hosny, A.; Parmar, C.; Quackenbush, J.; Schwartz, L.H.; Aerts, H.J. Artificial intelligence in radiology. Nat. Rev. Cancer 2018, 18, 500–510. [Google Scholar] [CrossRef]
- Chang, S.W.; Abdul-Kareem, S.; Merican, A.F.; Zain, R.B. Oral cancer prognosis based on clinicopathologic and genomic markers using a hybrid of feature selection and machine learning methods. BMC Bioinform. 2013, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.X.; Huang, S.G.; Du, J.F.; Zhang, X.C.; Jiang, N.; Kang, W.X.; Mao, J.; Zhao, Q. Comparison of Prognostic Indices in NSCLC Patients with Brain Metastases after Radiosurgery. Int.J. Biol. Sci. 2018, 14, 2065–2072. [Google Scholar] [CrossRef] [PubMed]
- Sepehri, S.; Upadhaya, T.; Desseroit, M.C.; Visvikis, D.; Le Rest, C.C.; Hatt, M. Comparison of machine learning algorithms for building prognostic models in non-small cell lung cancer using clinical and radiomics features from 18F-FDG PET/CT images. J. Nucl. Med. 2018, 59 (Suppl. 1), 328. [Google Scholar]
- Sun, D.; Wang, M.; Li, A. A multimodal deep neural network for human breast cancer prognosis prediction by integrating multi-dimensional data. IEEE/ACM Trans. Comput. Biol. Bioinform. 2018, 16, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Dang, Y.; Li, F.; Wei, W.; Ma, Y.; Qiao, S.; Wang, Q. Biological intensity-modulated radiotherapy plus neoadjuvant chemotherapy for multiple peritoneal metastases of ovarian cancer: A case report. Oncol. Lett. 2015, 9, 1239–1243. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Long, N.P.; Jung, K.H.; Anh, N.H.; Yan, H.H.; Nghi, T.D.; Park, S.; Yoon, S.J.; Min, J.E.; Kim, H.M.; Lim, J.H.; et al. An Integrative Data Mining and Omics-Based Translational Model for the Identification and Validation of Oncogenic Biomarkers of Pancreatic Cancer. Cancers 2019, 11, 155. [Google Scholar] [CrossRef] [PubMed]
- Oberije, C.; Nalbantov, G.; Dekker, A.; Boersma, L.; Borger, J.; Reymen, B.; van Baardwijk, A.; Wanders, R.; De Ruysscher, D.; Steyerberg, E.; et al. A prospective study comparing the predictions of doctors versus models for treatment outcome of lung cancer patients: A step toward individualized care and shared decision making. Radiother. Oncol. 2014, 112, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Ha, E.J.; Kim, J.H. Application of deep learning to the diagnosis of cervical lymph node metastasis from thyroid cancer with CT. Eur. Radiol. 2019, 1–6. [Google Scholar] [CrossRef]
- Zhang, B.; He, X.; Ouyang, F.; Gu, D.; Dong, Y.; Zhang, L.; Mo, X.; Huang, W.; Tian, J.; Zhang, S. Radiomic machine-learning classifiers for prognostic biomarkers of advanced nasopharyngeal carcinoma. Cancer Lett. 2017, 403, 21–27. [Google Scholar] [CrossRef]
- Qian, Z.; Li, Y.; Wang, Y.; Li, L.; Li, R.; Wang, K.; Li, S.; Tang, K.; Zhang, C.; Fan, X.; et al. Differentiation of glioblastoma from solitary brain metastases using radiomic machine-learning classifiers. Cancer Lett. 2019, 451, 128–135. [Google Scholar] [CrossRef]
- Andrews, D.W.; Scott, C.B.; Sperduto, P.W.; Flanders, A.E.; Gaspar, L.E.; Schell, M.C.; Werner-Wasik, M.; Demas, W.; Ryu, J.; Bahary, J.-P.; et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: Phase III results of the RTOG 9508 randomised trial. Lancet 2004, 363, 1665–1672. [Google Scholar] [CrossRef]
- Zhang, X.; Mei, C.; Chen, D.; Li, J. Feature selection in mixed data: A method using a novel fuzzy rough set-based information entropy. Pattern Recognit. 2016, 56, 1–15. [Google Scholar] [CrossRef]
- Xue, B.; Zhang, M.; Browne, W.N.; Yao, X. A Survey on Evolutionary Computation Approaches to Feature Selection. IEEE Trans. Evol. Comput. 2016, 20, 606–626. [Google Scholar] [CrossRef]

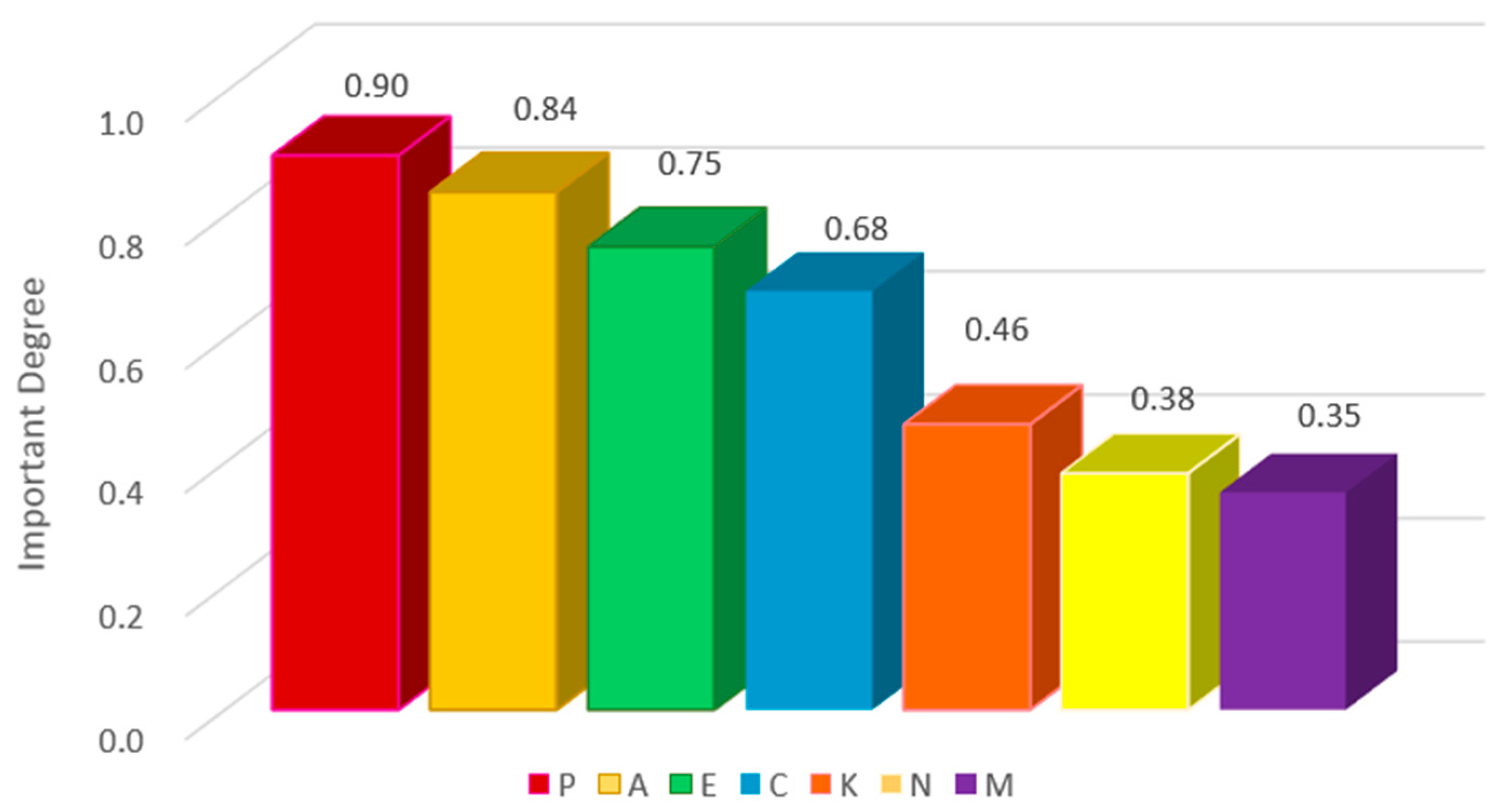
| Cancer Feature | Label |
|---|---|
| Age | A |
| Karnofsky Performance Status | K |
| Extracranial metastasis | E |
| Primary tumor control | P |
| Number of lesions | N |
| Max lesion volume | M |
| If chemical therapy | C |
| No. | Methods | Cancer Features in Patients | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | SFS | C | A | E | K | P | M | N |
| 2 | MISFS | A | P | E | K | C | N | M |
| 3 | MIPSO | P | E | A | C | K | N | M |
| 4 | MIRSPSO | P | C | A | E | N | K | M |
| P | A | C | E | N | K | M | ||
| P | E | C | A | N | K | M | ||
| P | A | E | C | K | N | M | ||
| P | A | E | C | K | N | M | ||
| A | P | C | E | K | N | M | ||
| P | E | A | C | N | K | M | ||
| P | A | E | C | N | K | M | ||
| P | A | E | C | N | K | M | ||
| No. | Cancer Features in Patients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | P | C | A | E | N | K | M | 0.9 | 0.1 |
| 0.9074 | 0.8535 | 0.7286 | 0.6303 | 0.4771 | 0.3896 | 0.3247 | |||
| 2 | P | A | C | E | N | K | M | 0.8 | 0.2 |
| 0.9278 | 0.8634 | 0.8021 | 0.6935 | 0.455 | 0.4011 | 0.3546 | |||
| 3 | P | A | E | C | N | K | M | 0.7 | 0.3 |
| 0.9322 | 0.8734 | 0.7559 | 0.6765 | 0.4834 | 0.3724 | 0.3618 | |||
| 4 | P | A | E | C | K | N | M | 0.6 | 0.4 |
| 0.8921 | 0.8105 | 0.765 | 0.6267 | 0.4933 | 0.4 | 0.3911 | |||
| 5 | P | A | E | C | K | N | M | 0.5 | 0.5 |
| 0.9213 | 0.7705 | 0.7068 | 0.703 | 0.4267 | 0.4201 | 0.3692 | |||
| 6 | A | P | C | E | K | N | M | 0.4 | 0.6 |
| 0.7183 | 0.6437 | 0.5902 | 0.5559 | 0.4757 | 0.43 | 0.3783 | |||
| 7 | P | E | A | C | N | K | M | 0.3 | 0.7 |
| 0.891 | 0.9211 | 0.7948 | 0.7774 | 0.4469 | 0.351 | 0.3 | |||
| 8 | P | A | E | C | N | K | M | 0.2 | 0.8 |
| 0.9335 | 0.91 | 0.8283 | 0.7632 | 0.4528 | 0.3439 | 0.3267 | |||
| 9 | P | A | E | C | N | K | M | 0.1 | 0.9 |
| 0.9546 | 0.8973 | 0.7751 | 0.6748 | 0.4486 | 0.3382 | 0.36 | |||
| Statistic Method | RPA | GPA | SIR | BSBM | Overall Survival | Machine Learning |
|---|---|---|---|---|---|---|
| MST | 19 | 26 | 25 | 23 | 24 | - |
| Sensitivity | 0.67 | 0.71 | 0.59 | 0.88 | - | 0.92 |
| Specificity | 0.39 | 0.33 | 0.43 | 0.46 | - | 0.85 |
| Accuracy | 0.682 | 0.655 | 0.611 | 0.758 | - | 0.885 |
| PPV | - | - | - | - | 0.83 | 0.86 |
| NPV | - | - | - | - | 0.65 | 0.914 |
| P | <0.0001 a | <0.0001 a | <0.0001 a | <0.000 a | - | - |
| Characteristics | N (%) | |
|---|---|---|
| Patients | 700 | |
| Gender | Male | 456 (65.1%) |
| Female | 244 (34.9%) | |
| Age(years) | Median | 55 |
| Range | 48 (16–92) | |
| KPS | Median | 75 |
| Range | 30 (55–95) | |
| Primary tumor type | NSCLC | 635 (90.7%) |
| Breast cancer | 57 (8.1%) | |
| Other | 8 (1.2%) | |
| Primary tumor control | No | 319 (45.6%) |
| Yes | 381 (54.4%) | |
| Number of lesions | Median | 3 |
| Range | 5 (1–6) | |
| Tumor volume(mL) | Median | 6 |
| Range | 40.4 (0.04–49) | |
| Maximum diameter(mm) | <10 | 28 (4%) |
| 10–20 | 189 (27%) | |
| 21–30 | 245 (35%) | |
| 31–40 | 205 (29.3%) | |
| >40 | 33 (4.7%) | |
| Type of therapy | SRS | 225 (32.1%) |
| Fractionated SRS WBRT | 127 (18.1%) | |
| SBRT | 133 (19%) | |
| Surgical resection | 197 (28.1%) | |
| Extracranial metastasis | No | 470 (67.1%) |
| Yes | 230 (33.9%) | |
| Histology classification | Adenocarcinoma | 170 (24.3%) |
| Squamous cell carcinoma | 120 (17.1%) | |
| Large cell carcinoma | 87 (12.4%) | |
| In situ carcinoma | 195 (27.9%) | |
| Invasive carcinoma | 128 (18.3%) | |
| Molecular classification | EGFR | 23 (3.3%) |
| KRAS | 17 (2.4%) | |
| BRAF | 4 (0.6%) | |
| TP53 | 9 (1.3%) | |
| ALK | 15 (2.1%) | |
| TNBC | 8 (1.1%) | |
| HER2 | 9 (1.3%) | |
| PTEN | 7 (1.0%) | |
| Pattern of dissemination | Blood | 361 (51.6%) |
| Lymph | 255 (36.4%) | |
| Others | 84 (12%) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, S.; Yang, J.; Fong, S.; Zhao, Q. Mining Prognosis Index of Brain Metastases Using Artificial Intelligence. Cancers 2019, 11, 1140. https://doi.org/10.3390/cancers11081140
Huang S, Yang J, Fong S, Zhao Q. Mining Prognosis Index of Brain Metastases Using Artificial Intelligence. Cancers. 2019; 11(8):1140. https://doi.org/10.3390/cancers11081140
Chicago/Turabian StyleHuang, Shigao, Jie Yang, Simon Fong, and Qi Zhao. 2019. "Mining Prognosis Index of Brain Metastases Using Artificial Intelligence" Cancers 11, no. 8: 1140. https://doi.org/10.3390/cancers11081140
APA StyleHuang, S., Yang, J., Fong, S., & Zhao, Q. (2019). Mining Prognosis Index of Brain Metastases Using Artificial Intelligence. Cancers, 11(8), 1140. https://doi.org/10.3390/cancers11081140






