Treatment Options for Pancreatic Neuroendocrine Tumors
Abstract
1. Introduction
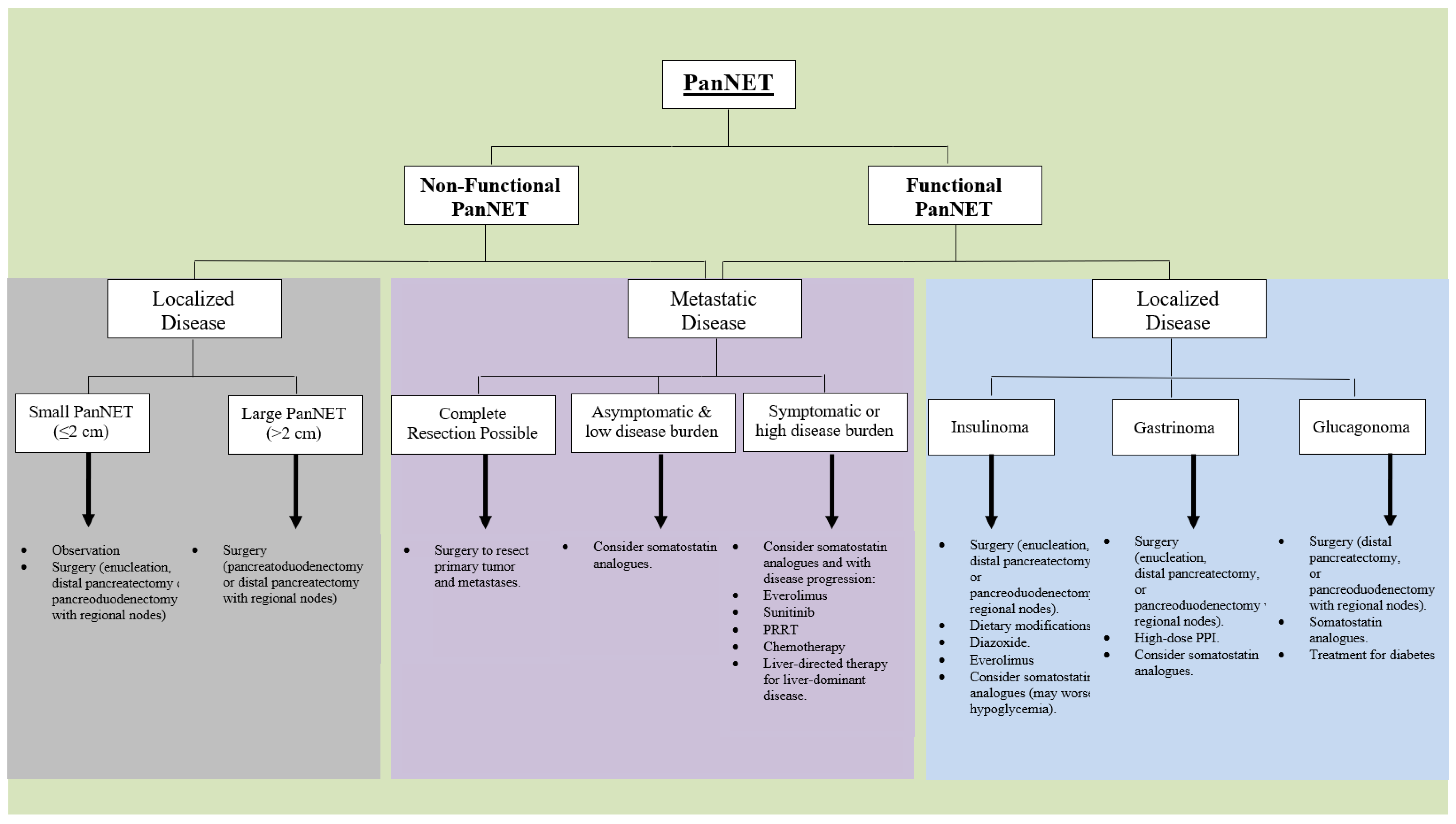
2. Surgical Treatment
2.1. Surgically Resectable Disease
2.2. Surgical Treatment for Advanced Disease
2.3. Post-Surgery Follow-Up
3. Medical Therapy
3.1. Symptoms-Directed Therapy
3.2. Control of Tumor Growth
3.2.1. Somatostatin Analogues
3.2.2. Molecularly Targeted Therapies
Everolimus
Sunitinib
Pazopanib, Sorafenib, Surafitinib, and Axitinib
Temisirolimus and Bevacizumab
3.2.3. Cytotoxic Chemotherapy
Streptozocin-Based Chemotherapy
Temozolomide-Based Chemotherapy
Platinum-Based Chemotherapy
4. Radiation Therapy
4.1. External Beam Radiotherapy (EBRT)
4.2. Peptide Receptor Radionuclide Therapy (PRRT)
5. Liver-Directed Therapies
6. Future Directions
7. Conclusions
Funding
Conflicts of Interest
References
- Lawrence, B.; Bjorn, G.I.; Anthony, C.; Bernhard, S.; Mark, K.; Irvin, M.M. Epidemiology of gastroenteropancreatic neuroendocrine tumours. Endocrinol. Metab. Clin. 2011, 40, 1–18. [Google Scholar] [CrossRef] [PubMed]
- McKenna, L.R.; Edil, B.H. Update on pancreatic neuroendocrine tumors. Gland Surg. 2014, 3, 258–275. [Google Scholar] [PubMed]
- Bartolini, I.; Bencini, L.; Risaliti, M.; Ringressi, M.N.; Moraldi, L.; Taddei, A. Current Management of Pancreatic Neuroendocrine Tumors: From Demolitive Surgery to Observation. Gastroenterol. Res. Pract. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, R.V.; Osamura, R.; Kloppel, G.; Rosai, J. WHO Classification of Tumours of Endocrine Organs, 4th ed.; IARC Press: Lyon, France, 2017. [Google Scholar]
- Halfdanarson, T.R.; Rabe, K.G.; Rubin, J.; Petersen, G.M. Pancreatic neuroendocrine tumors (PNETs): Incidence, prognosis and recent trend toward improved survival. Ann. Oncol. 2008, 19, 1727–1733. [Google Scholar] [CrossRef] [PubMed]
- Crippa, S.; Partelli, S.; Zamboni, G.; Scarpa, A.; Tamburrino, D.; Bassi, C.; Pederzoli, P.; Falconi, M. Incidental diagnosis as prognostic factor in different tumor stages of nonfunctioning pancreatic endocrine tumors. Surgery 2014, 155, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Gaujoux, S.; Partelli, S.; Maire, F.; D’Onofrio, M.; Larroque, B.; Tamburrino, D.; Sauvanet, A.; Falconi, M.; Ruszniewski, P. Observational study of natural history of small sporadic nonfunctioning pancreatic neuroendocrine tumors. J. Clin. Endocrinol. Metab. 2013, 98, 4784–4789. [Google Scholar] [CrossRef] [PubMed]
- Falconi, M.; Eriksson, B.; Kaltsas, G.; Bartsch, D.; Capdevila, J.; Koskudla, B.; Kwekkeboom, D.; Rindi, G.; Kloppel, G.; Reed, N.; et al. Consensus guidelines update for the management of functional p-NETs (F-p-NETs) and non-functional p-NETs (NF-p-NETs). Neuroendocrinology 2016, 103, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Oberg, K.; Knigge, U.; Kwekkeboom, D.; Perren, A. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2012, 23, 124–130. [Google Scholar] [CrossRef]
- Shah, M.; Goldner, W.; Halfdanarson, T.; Bergsland, E.; Berlin, J.; Halperin, D.; Chan, J.; Kulke, M.; Benson, A.; Blaszkowsky, L.; et al. NCCN Clinical Practice Guidelines in Oncology: Neuroendocrine and Adrenal Tumors. J. Natl. Compr. Cancer Netw. 2018, 16, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Kulke, M.H.; Hörsch, D.; Caplin, M.E.; Anthony, L.B.; Bergsland, E.; Öberg, K.; Welin, S.; Warner, R.R.P.; Lombard-Bohas, C.; Kunz, P.L.; et al. Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome. J. Clin. Oncol. 2017, 35, 14–23. [Google Scholar] [CrossRef]
- Caplin, M.E.; Pavel, M.; Ćwikła, J.B.; Phan, A.T.; Raderer, M.; Sedláčková, E.; Cadiot, G.; Wolin, E.M.; Capdevila, J.; Wall, L.; et al. Lanreotide in Metastatic Enteropancreatic Neuroendocrine Tumors. N. Engl. J. Med. 2014, 371, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Carbonero, R.; Sorbye, H.; Baudin, E.; Raymond, E.; Wiedenmann, B.; Niederle, B.; Sedlackova, E.; Toumpanakis, C.; Anlauf, M.; Cwikla, J.B.; et al. ENETS consensus guidelines for high-grade gastroenteropancreatic neuroendocrine tumors and neuroendocrine carcinomas. Neuroendocrinology 2016, 103, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Kunz, P.; Reidy-Lagunes, D.; Anthony, L.; Bertino, E.; Brendtro, K.; Chan, J.; Chen, H.; Jensen, R.; Kang Kim, M.; Klimstra, D.; et al. Consensus Guidelines for the Management and Treatment of Neuroendocrine Tumors. Pancreas 2013, 42, 557–577. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Shah, M.; Ito, T.; Bohas, C.; Wolin, E.M.; Van Custem, E.; Hobday, T.; Okusaka, T.; Capdevila, J.; de Vries, E.; et al. Everolimus for Advanced Pancreatic Neuroendocrine Tumors. N. Engl. J. Med. 2011, 364, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Pavel, M.E.; Hainsworth, J.D.; Baudin, E.; Peeters, M.; Hörsch, D.; Winkler, R.E.; Klimovsky, J.; Lebwohl, D.; Jehl, V.; Wolin, E.M.; et al. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): A randomised, placebo-controlled, phase 3 study. Lancet 2011, 378, 2005–2012. [Google Scholar] [CrossRef]
- Yao, J.C.; Fazio, N.; Singh, S.; Buzzoni, R.; Carnaghi, C.; Wolin, E.; Tomasek, J.; Raderer, M.; Lahner, H.; Voi, M.; et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): A randomised, placebo-controlled, phase 3 study. Lancet 2016, 387, 968–977. [Google Scholar] [CrossRef]
- Raymond, E.; Dahan, L.; Raoul, J.-L.; Bang, Y.-J.; Borbath, I.; Lombard-Bohas, C.; Valle, J.; Metrakos, P.; Smith, D.; Vinik, A.; et al. Sunitinib Malate for the Treatment of Pancreatic Neuroendocrine Tumors. N. Engl. J. Med. 2011, 364, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, C.; Han, J.; Luan, Y.; Cui, Y.; Shen, R.; Sha, D.; Cong, L.; Zhang, Z.; Wang, W. Therapeutic effect of sunitinib malate and its influence on blood glucose concentrations in a patient with metastatic insulinoma. Expert Rev. Anticancer Ther. 2013, 13, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Fountas, A.; Tigas, S.; Giotaki, Z.; Petrakis, D.; Pentheroudakis, G.; Tsatsoulis, A. Severe resistant hypoglycemia in a patient with a pancreatic neuroendocrine tumor on sunitinib treatment. Hormones 2015, 14, 438–441. [Google Scholar] [CrossRef] [PubMed]
- De Mestier, L.; Walter, T.; Brixi, H.; Lombard-Bohas, C.; Cadiot, G. Sunitinib achieved fast and sustained control of VIPoma symptoms. Eur. J. Endocrinol. 2015, 172, K1–K3. [Google Scholar] [CrossRef]
- Phan, A.T.; Halperin, D.M.; Chan, J.A.; Fogelman, D.R.; Hess, K.R.; Malinowski, P.; Regan, E.; Ng, C.S.; Yao, J.C.; Kulke, M.H. Pazopanib and depot octreotide in advanced, well-differentiated neuroendocrine tumours: A multicentre, single-group, phase 2 study. Lancet Oncol. 2015, 16, 695–703. [Google Scholar] [CrossRef]
- Xu, J.M.; Li, J.; Bai, C.-M.; Xu, N.; Zhou, Z.; Li, Z.; Zhou, C.; Jia, R.; Lu, M.; Cheng, Y.; et al. Surufatinib in advanced well-differentiated neuroendocrine tumors: A multicenter, single-arm, open-label, phase Ib/II trial. Clin. Cancer Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hobday, T.J.; Qin, R.; Reidy-lagunes, D.; Moore, M.J.; Strosberg, J.; Kaubisch, A.; Shah, M.; Kindler, H.L.; Lenz, H.; Chen, H.; et al. Multicenter Phase II Trial of Temsirolimus and Bevacizumab in Pancreatic Neuroendocrine Tumors. J. Clin. Oncol. 2015, 33, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Phan, A.T.; Hess, K.; Fogelman, D.; Jacobs, C.; Dagohoy, C.; Leary, C.; Xie, K.; Ng, C.S. Perfusion computed tomography as functional biomarker in randomized run-in study of Bevacizumab and Everolimus in well-differentiated neuroendocrine tumors. Pancreas 2015, 44, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Guthrie, K.A.; Moran, C.; Strosberg, J.R.; Kulke, M.H.; Chan, J.A.; Loconte, N.; Mcwilliams, R.R.; Wolin, E.M.; Mattar, B.; et al. Phase III Prospective Randomized Comparison Trial of Depot Octreotide Plus Interferon Alfa-2b Versus Depot Octreotide Plus Bevacizumab in Patients With Advanced Carcinoid Tumors: SWOG S0518. J. Clin. Oncol. 2017, 35, 1695–1704. [Google Scholar] [CrossRef] [PubMed]
- Moertel, C.; Lefkoupoulo, M.; Lipsitz, S.; Hahn, R.; Klaasen, D. Streptozocin-doxorubicin, streptozocin-fluorouracil or chlorozotocin in the treatment of advanced islet-cell carcinoma. N. Engl. J. Med. 1992, 326, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Kouvaraki, M.A.; Ajani, J.A.; Hoff, P.; Wolff, R.; Evans, D.B.; Lozano, R.; Yao, J.C. Fluorouracil, Doxorubicin, and Streptozocin in the Treatment of Patients with Locally Advanced and Metastatic Pancreatic Endocrine Carcinomas. J. Clin. Oncol. 2004, 22, 4762–4771. [Google Scholar] [CrossRef]
- Ducreux, M.; Dahan, L.; Smith, D.; Toole, D.O.; Dromain, C.; Vilgrain, V.; Baudin, E.; Lombard-Bohas, C.; Scoazec, J.-Y.; Seitz, J.-F.; et al. Bevacizumab combined with 5-FU/streptozocin in patients with progressive metastatic well-differentiated pancreatic endocrine tumours (BETTER trial)—A phase II non-randomised trial. Eur. J. Cancer 2014, 50, 3098–3106. [Google Scholar] [CrossRef]
- Welin, S.; Sorbye, H.; Sebjornsen, S.; Knappskog, S.; Busch, C.; Öberg, K. Clinical effect of temozolomide-based chemotherapy in poorly differentiated endocrine carcinoma after progression on first-line chemotherapy. Cancer 2011, 117, 4617–4622. [Google Scholar] [CrossRef]
- Strosberg, J.R.; Fine, R.; Choi, J.; Nasir, A.; Coppola, D.; Chen, D.-T.; Helm, J.; Kvols, L. First-Line Chemotherapy With Capecitabine and Temozolomide in Patients With Metastatic Pancreatic Endocrine Carcinomas. Cancer 2011, 117, 268–275. [Google Scholar] [CrossRef]
- Kunz, P.; Catalano, P.; Nimeri, H.; Fisher, G.; Longacre, T.; Suarez, C.; Yao, J.; Kulke, M.H.; Hendifar, A.; Shanks, J.; et al. A randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors: A trial of the ECOG-ACRIN Cancer Research Group (E2211). J. Clin. Oncol. 2018, 36, 4004. [Google Scholar] [CrossRef]
- Rindi, G.; Klimstra, D.S.; Abedi-Ardekani, B.; Asa, S.L.; Bosman, F.T.; Brambilla, E.; Busam, K.J.; de Krijger, R.R.; Dietel, M.; El-Naggar, A.K.; et al. A common classification framework for neuroendocrine neoplasms: An International Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Mod. Pathol. 2018, 31, 1770–1786. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.L.; Thompson, R.; Lam, M.; Pavlakis, N.; Hallet, J.; Law, C.; Singh, S.; Myrehaug, S. External Beam Radiotherapy in the Treatment of Gastroenteropancreatic Neuroendocrine Tumours: A Systematic Review. Clin. Oncol. 2018, 30, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; Bushnell, D.; et al. Phase 3 Trial of 177 Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.; Costa, R.; Bacchi, C.E.; Filho, P.A. Case Report Metastatic Insulinoma Managed with Radiolabeled Somatostatin Analog. Case Rep. Endocrinol. 2013, 2013, 8–11. [Google Scholar]
- Dumont, R.A.; Seiler, D.; Marincek, N.; Brunner, P.; Radojewski, P.; Müller-brand, J.; Maecke, H.R.; Briel, M.; Walter, M.A. Survival after somatostatin based radiopeptide therapy in metastasized gastrinoma. Am. J. Nucl. Med. Mol. Imaging 2015, 5, 46–55. [Google Scholar] [PubMed]
- Grozinsky-glasberg, S.; Barak, D.; Fraenkel, M.; Walter, M.A.; Mueller-Brand, J.; Eckstein, J.; Applebaum, L.; Shimon, I.; Gross, D. Peptide Receptor Radioligand Therapy Is an Effective Treatment for the Long-Term Stabilization of Malignant Gastrinomas. Cancer 2011, 117, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Van Schaik, E.; Van Vliet, E.I.; Feelders, R.A.; Krenning, E.P.; Khan, S.; Kamp, K.; Valkema, R.; Van Nederveen, F.H.; Teunissen, J.J.M.; Kwekkeboom, D.J.; et al. Improved Control of Severe Hypoglycemia in Patients with Malignant Insulinomas by Peptide Receptor. J. Clin. Endocrinol. Metab. 2011, 96, 3381–3389. [Google Scholar] [CrossRef]
- Brabander, T.; Van Der Zwan, W.A.; Teunissen, J.J.M.; Kam, B.L.R.; Feelders, R.A.; De Herder, W.W.; Van Eijck, C.H.J.; Franssen, G.J.H.; Krenning, E.P.; Kwekkeboom, D.J. Long-Term Efficacy, Survival, and Safety with Gastroenteropancreatic and Bronchial Neuroendocrine Tumors. Clin. Cancer Res. 2017, 23, 4617–4624. [Google Scholar] [CrossRef]
- Zandee, W.T.; Barbander, T.; Blazevic, A.; Kam, B.L.R.; Teunissen, J.J.M.; Feelders, R.A.; Hofland, J.; De Herder, W.W. Symptomatic and radiological response to 177 Lu-DOTATATE for the treatment of functioning pancreatic neuroendocrine tumors. J. Clin. Endocrinol. Metab. 2019, 104, 1336–1344. [Google Scholar] [CrossRef]
- Pavel, M.; O’Toole, D.; Costa, F.; Capdevila, J.; Gross, D.; Kianmanesh, R.; Krenning, E.; Knigge, U.; Salazar, R.; Pape, U.F.; et al. ENETS consensus guidelines update for the management of distant metastatic disease of intestinal, pancreatic, bronchial neuroendocrine neoplasms (NEN) and NEN of unknown primary site. Neuroendocrinology 2016, 103, 172–185. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, D.; Ruszniewski, P. Chemoembolization and other ablative therapies for liver metastases of gastrointestinal endocrine tumours. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.; Bester, L.; Salem, R.; Sharma, R.A.; Parks, R.W.; Ruszniewski, P. Role of hepatic intra-arterial therapies in metastatic neuroendocrine tumours (NET): Guidelines from the NET-Liver-Metastases Consensus Conference. HPB 2015, 17, 29–37. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akirov, A.; Larouche, V.; Alshehri, S.; Asa, S.L.; Ezzat, S. Treatment Options for Pancreatic Neuroendocrine Tumors. Cancers 2019, 11, 828. https://doi.org/10.3390/cancers11060828
Akirov A, Larouche V, Alshehri S, Asa SL, Ezzat S. Treatment Options for Pancreatic Neuroendocrine Tumors. Cancers. 2019; 11(6):828. https://doi.org/10.3390/cancers11060828
Chicago/Turabian StyleAkirov, Amit, Vincent Larouche, Sameerah Alshehri, Sylvia L. Asa, and Shereen Ezzat. 2019. "Treatment Options for Pancreatic Neuroendocrine Tumors" Cancers 11, no. 6: 828. https://doi.org/10.3390/cancers11060828
APA StyleAkirov, A., Larouche, V., Alshehri, S., Asa, S. L., & Ezzat, S. (2019). Treatment Options for Pancreatic Neuroendocrine Tumors. Cancers, 11(6), 828. https://doi.org/10.3390/cancers11060828






