Identification of KIF11 as a Novel Target in Meningioma
Abstract
:1. Introduction
2. Results
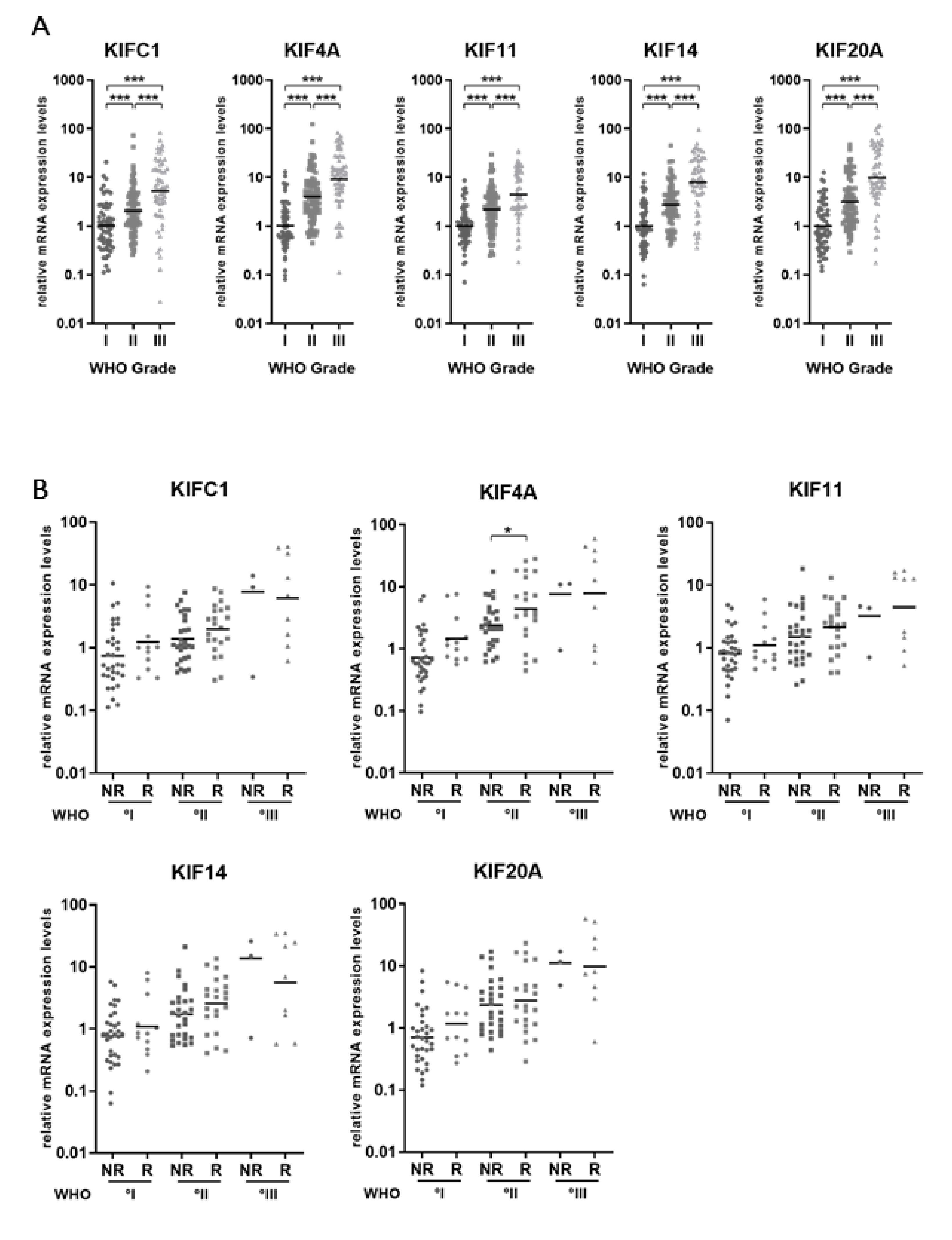
2.1. Kinesin mRNA Levels Increase with WHO Grade of Meningiomas
2.2. Subgroup Analysis Reveals Elevated KIF4A Levels in MGMs Experiencing Future Recurrence or Malignant Progression
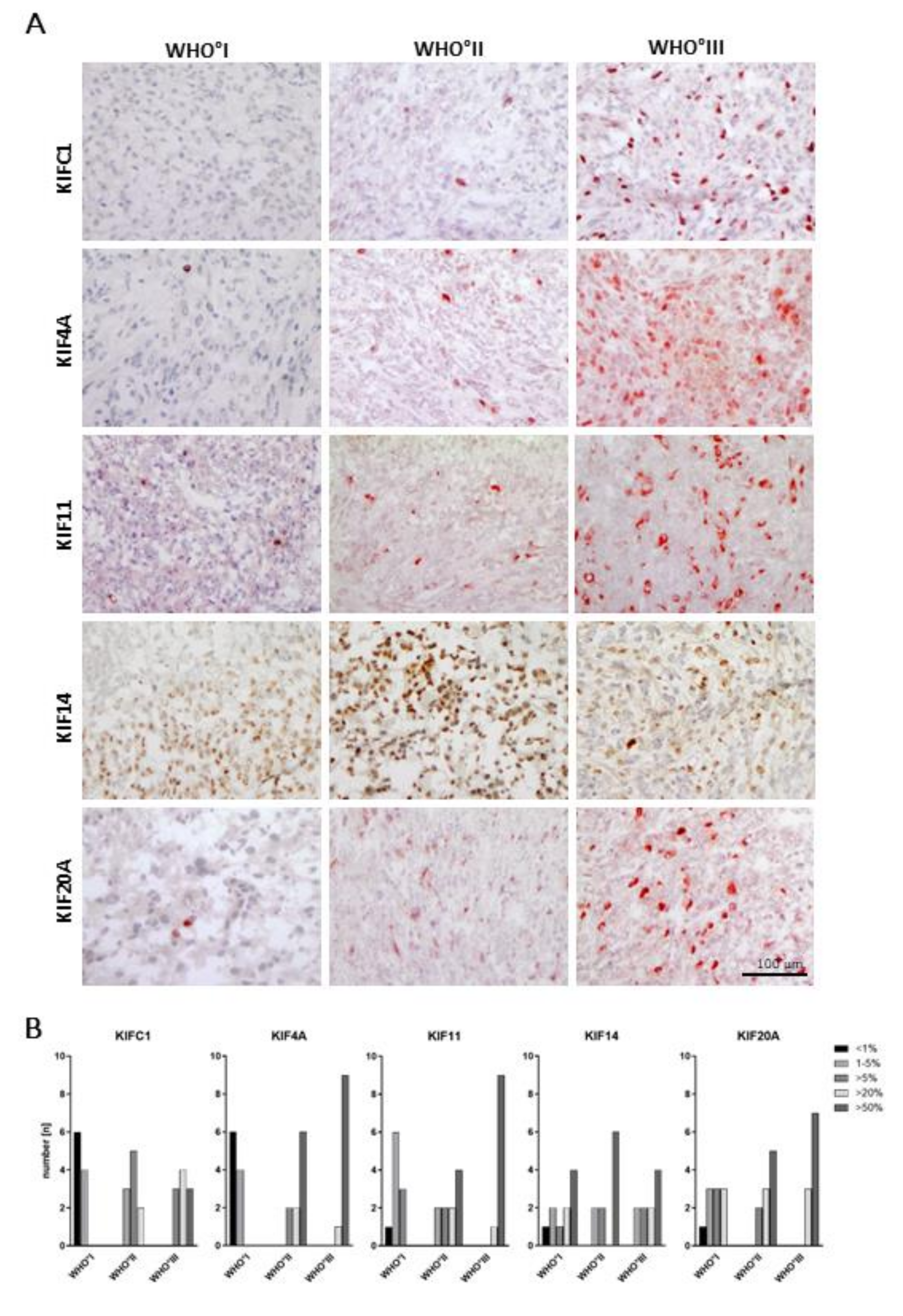
2.3. WHO Grade-Specific Increase of Kinesin Protein Levels in Meningioma Tissue Samples
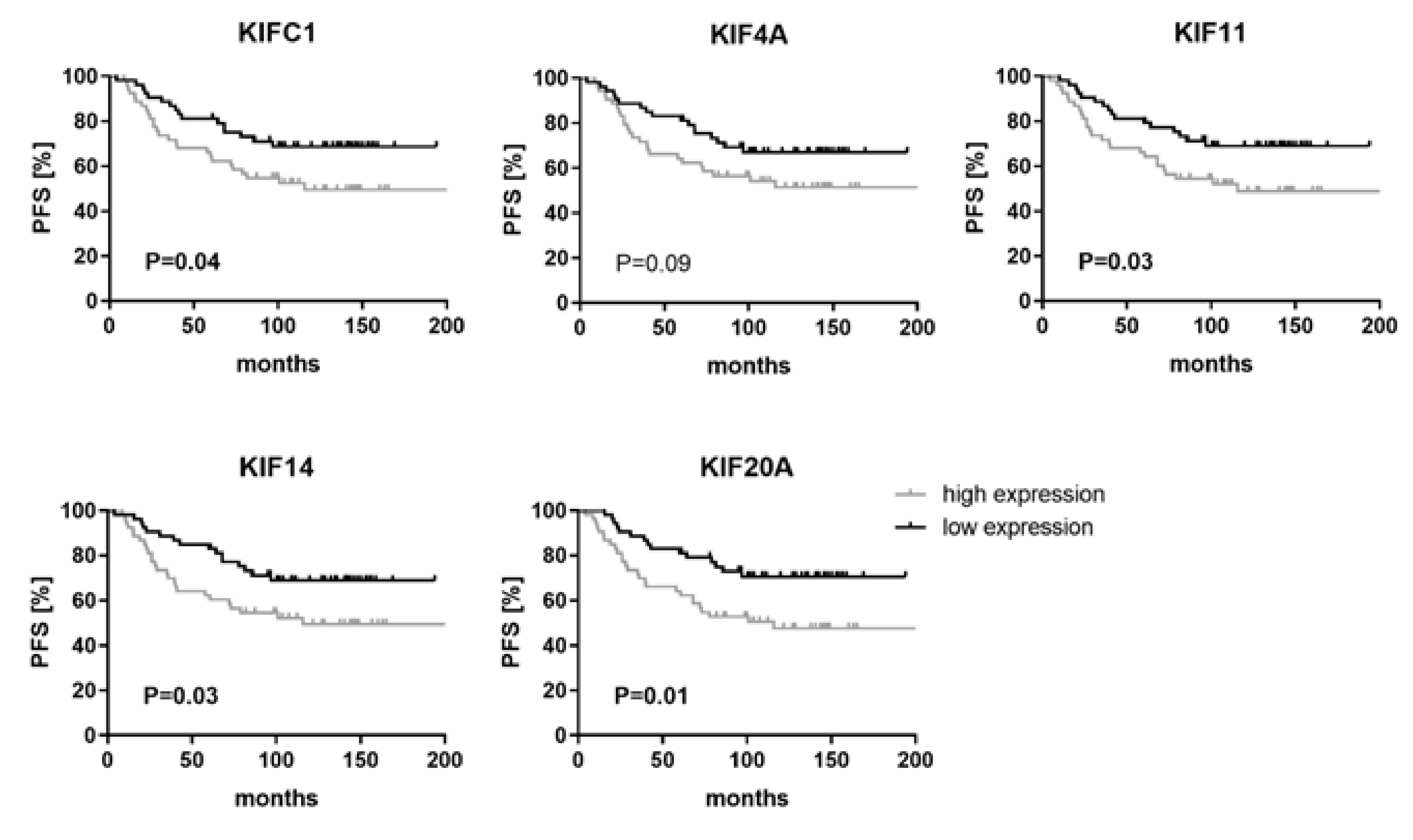
2.4. High Levels of KIFC1, KIF11, KIF14 and KIF20A Are Associated with Shorter Progression-Free Survival
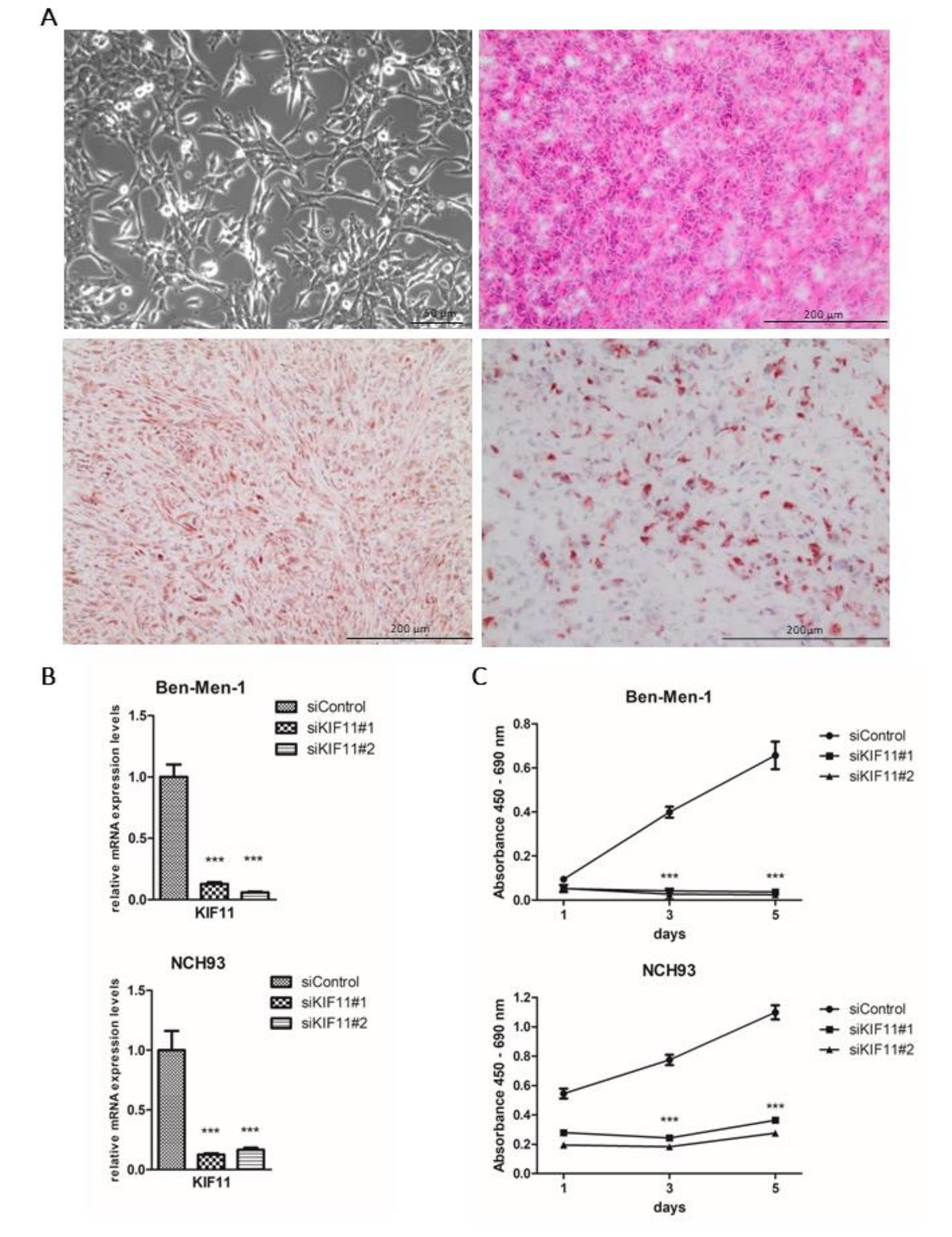
2.5. NCH93 as a Novel Anaplastic Meningioma Cell Line for Functional Testing
2.6. Depletion of KIF11 via siRNA Inhibits Meningioma Proliferation In Vitro
3. Discussion
4. Materials and Methods
4.1. Tumor Samples and RNA Isolation
4.2. Quantitative Real-Time PCR
4.3. Immunohistochemical Staining
4.4. Cell Lines
4.5. Panel Sequencing
4.6. siRNA-Mediated Knockdown
4.7. Cell Proliferation Assay
4.8. Xenograft Mouse Model
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mawrin, C.; Perry, A. Pathological classification and molecular genetics of meningiomas. J. Neurooncol. 2010, 99, 379–391. [Google Scholar] [CrossRef]
- Louis, D.N.; Ohgaki, H.; Wiestler, O.D.; Cavenee, W.K.; Burger, P.C.; Jouvet, A.; Scheithauer, B.W.; Kleihues, P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007, 114, 97–109. [Google Scholar] [CrossRef]
- Baldi, I.; Engelhardt, J.; Bonnet, C.; Bauchet, L.; Berteaud, E.; Gruber, A.; Loiseau, H. Epidemiology of meningiomas. Neurochirurgie 2014. [Google Scholar] [CrossRef] [PubMed]
- Van Alkemade, H.; de Leau, M.; Dieleman, E.M.; Kardaun, J.W.; van Os, R.; Vandertop, W.P.; van Furth, W.R.; Stalpers, L.J. Impaired survival and long-term neurological problems in benign meningioma. Neuro Oncol. 2012, 14, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Durand, A.; Labrousse, F.; Jouvet, A.; Bauchet, L.; Kalamarides, M.; Menei, P.; Deruty, R.; Moreau, J.J.; Fevre-Montange, M.; Guyotat, J. WHO grade II and III meningiomas: A study of prognostic factors. J. Neuro Oncol. 2009, 95, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Vranic, A.; Popovic, M.; Cor, A.; Prestor, B.; Pizem, J. Mitotic count, brain invasion, and location are independent predictors of recurrence-free survival in primary atypical and malignant meningiomas: A study of 86 patients. Neurosurgery 2010, 67, 1124–1132. [Google Scholar] [CrossRef]
- Gousias, K.; Schramm, J.; Simon, M. The Simpson grading revisited: Aggressive surgery and its place in modern meningioma management. J. Neurosurg. 2016, 125, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Alexiou, G.A.; Gogou, P.; Markoula, S.; Kyritsis, A.P. Management of meningiomas. Clin. Neurol Neurosurg. 2010, 112, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Clark, V.E.; Erson-Omay, E.Z.; Serin, A.; Yin, J.; Cotney, J.; Ozduman, K.; Avsar, T.; Li, J.; Murray, P.B.; Henegariu, O.; Yilmaz, S.; et al. Genomic analysis of non-NF2 meningiomas reveals mutations in TRAF7, KLF4, AKT1, and SMO. Science 2013, 339, 1077–1080. [Google Scholar] [CrossRef] [PubMed]
- Clark, V.E.; Harmanci, A.S.; Bai, H.; Youngblood, M.W.; Lee, T.I.; Baranoski, J.F.; Ercan-Sencicek, A.G.; Abraham, B.J.; Weintraub, A.S.; Hnisz, D.; et al. Recurrent somatic mutations in POLR2A define a distinct subset of meningiomas. Nat. Genet. 2016, 48, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Abedalthagafi, M.; Bi, W.L.; Aizer, A.A.; Merrill, P.H.; Brewster, R.; Agarwalla, P.K.; Listewnik, M.L.; Dias-Santagata, D.; Thorner, A.R.; Van Hummelen, P.; et al. Oncogenic PI3K mutations are as common as AKT1 and SMO mutations in meningioma. Neuro Oncol. 2016, 18, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Brastianos, P.K.; Horowitz, P.M.; Santagata, S.; Jones, R.T.; McKenna, A.; Getz, G.; Ligon, K.L.; Palescandolo, E.; Van Hummelen, P.; Ducar, M.D.; et al. Genomic sequencing of meningiomas identifies oncogenic SMO and AKT1 mutations. Nat. Genet. 2013, 45, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Sahm, F.; Schrimpf, D.; Olar, A.; Koelsche, C.; Reuss, D.; Bissel, J.; Kratz, A.; Capper, D.; Schefzyk, S.; Hielscher, T.; et al. TERT promoter mutations and risk of recurrence in meningioma. J. Natl. Cancer Inst. 2016, 108. [Google Scholar] [CrossRef] [PubMed]
- Reuss, D.E.; Piro, R.M.; Jones, D.T.; Simon, M.; Ketter, R.; Kool, M.; Becker, A.; Sahm, F.; Pusch, S.; Meyer, J.; et al. Secretory meningiomas are defined by combined KLF4 K409Q and TRAF7 mutations. Acta Neuropathol. 2013, 125, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Pachow, D.; Andrae, N.; Kliese, N.; Angenstein, F.; Stork, O.; Wilisch-Neumann, A.; Kirches, E.; Mawrin, C. mTORC1 inhibitors suppress meningioma growth in mouse models. Clin. Cancer Res. 2013, 19, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Gogineni, V.R.; Nalla, A.K.; Gupta, R.; Gujrati, M.; Klopfenstein, J.D.; Mohanam, S.; Rao, J.S. alpha3beta1 integrin promotes radiation-induced migration of meningioma cells. Int. J. Oncol. 2011, 38, 1615–1624. [Google Scholar] [CrossRef] [PubMed]
- Weller, M.; Roth, P.; Sahm, F.; Burghardt, I.; Schuknecht, B.; Rushing, E.J.; Regli, L.; Lindemann, J.P.; von Deimling, A. Durable control of metastatic AKT1-mutant WHO Grade 1 meningothelial meningioma by the AKT inhibitor, AZD5363. J. Natl. Cancer Inst. 2017, 109, 1–4. [Google Scholar] [CrossRef]
- Hirokawa, N.; Tanaka, Y. Kinesin superfamily proteins (KIFs): Various functions and their relevance for important phenomena in life and diseases. Exp. Cell Res. 2015, 334, 16–25. [Google Scholar] [CrossRef]
- Miki, H.; Setou, M.; Hirokawa, N.; Group, R.G.; Members, G.S.L. Kinesin superfamily proteins (KIFs) in the mouse transcriptome. Genome Res. 2003, 13, 1455–1465. [Google Scholar] [CrossRef]
- Lawrence, C.J.; Dawe, R.K.; Christie, K.R.; Cleveland, D.W.; Dawson, S.C.; Endow, S.A.; Goldstein, L.S.; Goodson, H.V.; Hirokawa, N.; Howard, J.; et al. A standardized kinesin nomenclature. J. Cell Biol. 2004, 167, 19–22. [Google Scholar] [CrossRef]
- Vale, R.D.; Reese, T.S.; Sheetz, M.P. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 1985, 42, 39–50. [Google Scholar] [CrossRef]
- Wordeman, L. How kinesin motor proteins drive mitotic spindle function: Lessons from molecular assays. Semin. Cell Dev. Biol. 2010, 21, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Hirokawa, N. Kinesin and dynein superfamily proteins and the mechanism of organelle transport. Science 1998, 279, 519–526. [Google Scholar] [CrossRef]
- Venere, M.; Horbinski, C.; Crish, J.F.; Jin, X.; Vasanji, A.; Major, J.; Burrows, A.C.; Chang, C.; Prokop, J.; Wu, Q.; et al. The mitotic kinesin KIF11 is a driver of invasion, proliferation, and self-renewal in glioblastoma. Sci. Transl. Med. 2015, 7, 304ra143. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Gong, H.; Huang, K. Oncogenic role of kinesin proteins and targeting kinesin therapy. Cancer Sci. 2013, 104, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Wang, X.; Zhu, H.; Wang, W.; Zhang, S.; Wang, Z. KIF2A overexpression and its association with clinicopathologic characteristics and unfavorable prognosis in colorectal cancer. Tumour Biol. 2015, 36, 8895–8902. [Google Scholar] [CrossRef]
- Manning, C.S.; Hooper, S.; Sahai, E.A. Intravital imaging of SRF and Notch signalling identifies a key role for EZH2 in invasive melanoma cells. Oncogene 2015, 34, 4320–4332. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, J.; Mitra, M.; Mallikarjuna, K.; Pranav, O.; Srinivasan, R.; Nagpal, A.; Venkatesan, P.; Kumaramanickavel, G. KIF14 and E2F3 mRNA expression in human retinoblastoma and its phenotype association. Mol. Vis. 2009, 15, 235–240. [Google Scholar] [PubMed]
- Li, Y.; Lu, W.; Chen, D.; Boohaker, R.J.; Zhai, L.; Padmalayam, I.; Wennerberg, K.; Xu, B.; Zhang, W. KIFC1 is a novel potential therapeutic target for breast cancer. Cancer Biol. 2015, 16, 1316–1322. [Google Scholar] [CrossRef]
- Kato, T.; Wada, H.; Patel, P.; Hu, H.P.; Lee, D.; Ujiie, H.; Hirohashi, K.; Nakajima, T.; Sato, M.; Kaji, M.; et al. Overexpression of KIF23 predicts clinical outcome in primary lung cancer patients. Lung Cancer 2016, 92, 53–61. [Google Scholar] [CrossRef]
- Suzuki, N.; Hazama, S.; Ueno, T.; Matsui, H.; Shindo, Y.; Iida, M.; Yoshimura, K.; Yoshino, S.; Takeda, K.; Oka, M. A phase I clinical trial of vaccination with KIF20A-derived peptide in combination with gemcitabine for patients with advanced pancreatic cancer. J. Immunother. 2014, 37, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Markowski, J.; Oczko-Wojciechowska, M.; Gierek, T.; Jarzab, M.; Paluch, J.; Kowalska, M.; Wygoda, Z.; Pfeifer, A.; Tyszkiewicz, T.; Jarzab, B.; et al. Gene expression profile analysis in laryngeal cancer by high-density oligonucleotide microarrays. J. Physiol. Pharm. 2009, 60 (Suppl. 1), 57–63. [Google Scholar]
- Sun, X.; Jin, Z.; Song, X.; Wang, J.; Li, Y.; Qian, X.; Zhang, Y.; Yin, Y. Evaluation of KIF23 variant 1 expression and relevance as a novel prognostic factor in patients with hepatocellular carcinoma. BMC Cancer 2015, 15, 961. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, C.; Wang, Q.; Liu, J.; Tian, M.; Yuan, Y.; Li, X.; Qu, X. Role of KIF2A in the progression and metastasis of human glioma. Mol. Med. Rep. 2016, 13, 1781–1787. [Google Scholar] [CrossRef]
- Takahashi, S.; Fusaki, N.; Ohta, S.; Iwahori, Y.; Iizuka, Y.; Inagawa, K.; Kawakami, Y.; Yoshida, K.; Toda, M. Downregulation of KIF23 suppresses glioma proliferation. J. Neurooncol. 2012, 106, 519–529. [Google Scholar] [CrossRef]
- Chen, S.; Han, M.; Chen, W.; He, Y.; Huang, B.; Zhao, P.; Huang, Q.; Gao, L.; Qu, X.; Li, X. KIF1B promotes glioma migration and invasion via cell surface localization of MT1-MMP. Oncol. Rep. 2016, 35, 971–977. [Google Scholar] [CrossRef]
- Schmidt, M.; Mock, A.; Jungk, C.; Sahm, F.; Ull, A.T.; Warta, R.; Lamszus, K.; Gousias, K.; Ketter, R.; Roesch, S.; et al. Transcriptomic analysis of aggressive meningiomas identifies PTTG1 and LEPR as prognostic biomarkers independent of WHO grade. Oncotarget 2016, 7, 14551–14568. [Google Scholar] [CrossRef]
- Steiner, H.H.; Bonsanto, M.M.; Beckhove, P.; Brysch, M.; Geletneky, K.; Ahmadi, R.; Schuele-Freyer, R.; Kremer, P.; Ranaie, G.; Matejic, D.; et al. Antitumor vaccination of patients with glioblastoma multiforme: A pilot study to assess feasibility, safety, and clinical benefit. J. Clin. Oncol. 2004, 22, 4272–4281. [Google Scholar] [CrossRef]
- Lucanus, A.J.; Yip, G.W. Kinesin superfamily: Roles in breast cancer, patient prognosis and therapeutics. Oncogene 2018, 37, 833–838. [Google Scholar] [CrossRef]
- Rath, O.; Kozielski, F. Kinesins and cancer. Nat. Rev. Cancer 2012, 12, 527–539. [Google Scholar] [CrossRef]
- Baas, P.W.; Matamoros, A.J. Inhibition of kinesin-5 improves regeneration of injured axons by a novel microtubule-based mechanism. Neural Regen. Res. 2015, 10, 845–849. [Google Scholar] [CrossRef]
- Jiang, M.; Zhuang, H.; Xia, R.; Gan, L.; Wu, Y.; Ma, J.; Sun, Y.; Zhuang, Z. KIF11 is required for proliferation and self-renewal of docetaxel resistant triple negative breast cancer cells. Oncotarget 2017, 8, 92106–92118. [Google Scholar] [CrossRef]
- Daigo, K.; Takano, A.; Thang, P.M.; Yoshitake, Y.; Shinohara, M.; Tohnai, I.; Murakami, Y.; Maegawa, J.; Daigo, Y. Characterization of KIF11 as a novel prognostic biomarker and therapeutic target for oral cancer. Int. J. Oncol. 2018, 52, 155–165. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, W.R.; Yang, L.C.; Wang, J.; Sun, J.Y.; Zhang, W.W.; He, Z.Y.; Wu, S.G. KIF11 functions as an oncogene and is associated with poor outcomes from breast cancer. Cancer Res. Treat. 2018. [Google Scholar] [CrossRef]
- Schneider, M.A.; Christopoulos, P.; Muley, T.; Warth, A.; Klingmueller, U.; Thomas, M.; Herth, F.J.; Dienemann, H.; Mueller, N.S.; Theis, F.; et al. AURKA, DLGAP5, TPX2, KIF11 and CKAP5: Five specific mitosis-associated genes correlate with poor prognosis for non-small cell lung cancer patients. Int. J. Oncol. 2017, 50, 365–372. [Google Scholar] [CrossRef]
- Imai, T.; Oue, N.; Nishioka, M.; Mukai, S.; Oshima, T.; Sakamoto, N.; Sentani, K.; Matsusaki, K.; Yoshida, K.; Yasui, W. Overexpression of KIF11 in gastric cancer with intestinal mucin phenotype. Pathobiology 2017, 84, 16–24. [Google Scholar] [CrossRef]
- Shah, J.J.; Kaufman, J.L.; Zonder, J.A.; Cohen, A.D.; Bensinger, W.I.; Hilder, B.W.; Rush, S.A.; Walker, D.H.; Tunquist, B.J.; Litwiler, K.S.; et al. A Phase 1 and 2 study of Filanesib alone and in combination with low-dose dexamethasone in relapsed/refractory multiple myeloma. Cancer 2017, 123, 4617–4630. [Google Scholar] [CrossRef]
- Hernandez-Garcia, S.; San-Segundo, L.; Gonzalez-Mendez, L.; Corchete, L.A.; Misiewicz-Krzeminska, I.; Martin-Sanchez, M.; Lopez-Iglesias, A.A.; Algarin, E.M.; Mogollon, P.; Diaz-Tejedor, A.; et al. The kinesin spindle protein inhibitor filanesib enhances the activity of pomalidomide and dexamethasone in multiple myeloma. Haematologica 2017, 102, 2113–2124. [Google Scholar] [CrossRef]
- LoRusso, P.M.; Goncalves, P.H.; Casetta, L.; Carter, J.A.; Litwiler, K.; Roseberry, D.; Rush, S.; Schreiber, J.; Simmons, H.M.; Ptaszynski, M.; et al. First-in-human phase 1 study of filanesib (ARRY-520), a kinesin spindle protein inhibitor, in patients with advanced solid tumors. Investig. New Drugs 2015, 33, 440–449. [Google Scholar] [CrossRef]
- Chari, A.; Htut, M.; Zonder, J.A.; Fay, J.W.; Jakubowiak, A.J.; Levy, J.B.; Lau, K.; Burt, S.M.; Tunquist, B.J.; Hilder, B.W.; et al. A phase 1 dose-escalation study of filanesib plus bortezomib and dexamethasone in patients with recurrent/refractory multiple myeloma. Cancer 2016, 122, 3327–3335. [Google Scholar] [CrossRef]
- Gomez, H.L.; Philco, M.; Pimentel, P.; Kiyan, M.; Monsalvo, M.L.; Conlan, M.G.; Saikali, K.G.; Chen, M.M.; Seroogy, J.J.; Wolff, A.A.; et al. Phase I dose-escalation and pharmacokinetic study of ispinesib, a kinesin spindle protein inhibitor, administered on days 1 and 15 of a 28-day schedule in patients with no prior treatment for advanced breast cancer. Anticancer Drugs 2012, 23, 335–341. [Google Scholar] [CrossRef]
- Burris, H.A., 3rd; Jones, S.F.; Williams, D.D.; Kathman, S.J.; Hodge, J.P.; Pandite, L.; Ho, P.T.; Boerner, S.A.; Lorusso, P. A phase I study of ispinesib, a kinesin spindle protein inhibitor, administered weekly for three consecutive weeks of a 28-day cycle in patients with solid tumors. Investig. New Drugs 2011, 29, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.A.; Siu, L.L.; Chen, E.X.; Hotte, S.J.; Chia, S.; Schwarz, J.K.; Pond, G.R.; Johnson, C.; Colevas, A.D.; Synold, T.W.; et al. Phase II study of ispinesib in recurrent or metastatic squamous cell carcinoma of the head and neck. Investig. New Drugs 2008, 26, 257–264. [Google Scholar] [CrossRef]
- Lee, C.W.; Belanger, K.; Rao, S.C.; Petrella, T.M.; Tozer, R.G.; Wood, L.; Savage, K.J.; Eisenhauer, E.A.; Synold, T.W.; Wainman, N.; et al. A phase II study of ispinesib (SB-715992) in patients with metastatic or recurrent malignant melanoma: A National Cancer Institute of Canada Clinical Trials Group trial. Investig. New Drugs 2008, 26, 249–255. [Google Scholar] [CrossRef]
- Blagden, S.P.; Molife, L.R.; Seebaran, A.; Payne, M.; Reid, A.H.; Protheroe, A.S.; Vasist, L.S.; Williams, D.D.; Bowen, C.; Kathman, S.J.; et al. A phase I trial of ispinesib, a kinesin spindle protein inhibitor, with docetaxel in patients with advanced solid tumours. Br. J. Cancer 2008, 98, 894–899. [Google Scholar] [CrossRef]
- Holen, K.D.; Belani, C.P.; Wilding, G.; Ramalingam, S.; Volkman, J.L.; Ramanathan, R.K.; Vasist, L.S.; Bowen, C.J.; Hodge, J.P.; Dar, M.M.; et al. A first in human study of SB-743921, a kinesin spindle protein inhibitor, to determine pharmacokinetics, biologic effects and establish a recommended phase II dose. Cancer Chemother. Pharm. 2011, 67, 447–454. [Google Scholar] [CrossRef]
- Bongero, D.; Paoluzzi, L.; Marchi, E.; Zullo, K.M.; Neisa, R.; Mao, Y.; Escandon, R.; Wood, K.; O’Connor, O.A. The novel kinesin spindle protein (KSP) inhibitor SB-743921 exhibits marked activity in in vivo and in vitro models of aggressive large B-cell lymphoma. Leuk. Lymphoma 2015, 56, 2945–2952. [Google Scholar] [CrossRef]
- O’Connor, O.A.; Gerecitano, J.; Van Deventer, H.; Hainsworth, J.; Zullo, K.M.; Saikali, K.; Seroogy, J.; Wolff, A.; Escandon, R. The addition of granulocyte-colony stimulating factor shifts the dose limiting toxicity and markedly increases the maximum tolerated dose and activity of the kinesin spindle protein inhibitor SB-743921 in patients with relapsed or refractory lymphoma: Results of an international, multicenter phase I/II study. Leuk. Lymphoma 2015, 56, 2585–2591. [Google Scholar] [CrossRef]
- Hu, J.R.; Liu, M.; Wang, D.H.; Hu, Y.J.; Tan, F.Q.; Yang, W.X. Molecular characterization and expression analysis of a KIFC1-like kinesin gene in the testis of Eumeces chinensis. Mol. Biol. Rep. 2013. [Google Scholar] [CrossRef]
- Tan, F.Q.; Ma, X.X.; Zhu, J.Q.; Yang, W.X. The expression pattern of the C-terminal kinesin gene kifc1 during the spermatogenesis of Sepiella maindroni. Gene 2013, 532, 53–62. [Google Scholar] [CrossRef]
- Farina, F.; Pierobon, P.; Delevoye, C.; Monnet, J.; Dingli, F.; Loew, D.; Quanz, M.; Dutreix, M.; Cappello, G. Kinesin KIFC1 actively transports bare double-stranded DNA. Nucleic Acids Res. 2013, 41, 4926–4937. [Google Scholar] [CrossRef] [PubMed]
- Sati, L.; Seval-Celik, Y.; Unek, G.; Korgun, E.T.; Demir, R. The presence of kinesin superfamily motor proteins KIFC1 and KIF17 in normal and pathological human placenta. Placenta 2009, 30, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.X.; Sperry, A.O. C-terminal kinesin motor KIFC1 participates in acrosome biogenesis and vesicle transport. Biol. Reprod. 2003, 69, 1719–1729. [Google Scholar] [CrossRef]
- Han, J.; Wang, F.; Lan, Y.; Wang, J.; Nie, C.; Liang, Y.; Song, R.; Zheng, T.; Pan, S.; Pei, T.; et al. KIFC1 regulated by miR-532-3p promotes epithelial-to-mesenchymal transition and metastasis of hepatocellular carcinoma via gankyrin/AKT signaling. Oncogene 2018. [Google Scholar] [CrossRef]
- Fu, X.; Zhu, Y.; Zheng, B.; Zou, Y.; Wang, C.; Wu, P.; Wang, J.; Chen, H.; Du, P.; Liang, B.; et al. KIFC1, a novel potential prognostic factor and therapeutic target in hepatocellular carcinoma. Int. J. Oncol. 2018, 52, 1912–1922. [Google Scholar] [CrossRef]
- Xiao, Y.X.; Shen, H.Q.; She, Z.Y.; Sheng, L.; Chen, Q.Q.; Chu, Y.L.; Tan, F.Q.; Yang, W.X. C-terminal kinesin motor KIFC1 participates in facilitating proper cell division of human seminoma. Oncotarget 2017, 8, 61373–61384. [Google Scholar] [CrossRef]
- Imai, T.; Oue, N.; Yamamoto, Y.; Asai, R.; Uraoka, N.; Sentani, K.; Yoshida, K.; Yasui, W. Overexpression of KIFC1 and its association with spheroid formation in esophageal squamous cell carcinoma. Pathol. Res. Pr. 2017, 213, 1388–1393. [Google Scholar] [CrossRef]
- Xiao, Y.X.; Yang, W.X. KIFC1: A promising chemotherapy target for cancer treatment? Oncotarget 2016, 7, 48656–48670. [Google Scholar] [CrossRef]
- Oue, N.; Mukai, S.; Imai, T.; Pham, T.T.; Oshima, T.; Sentani, K.; Sakamoto, N.; Yoshida, K.; Yasui, W. Induction of KIFC1 expression in gastric cancer spheroids. Oncol. Rep. 2016, 36, 349–355. [Google Scholar] [CrossRef]
- Liu, Y.; Zhan, P.; Zhou, Z.; Xing, Z.; Zhu, S.; Ma, C.; Li, Q.; Zhu, Q.; Miao, Y.; Zhang, J.; et al. The overexpression of KIFC1 was associated with the proliferation and prognosis of non-small cell lung cancer. J. Thorac. Dis. 2016, 8, 2911–2923. [Google Scholar] [CrossRef]
- Li, G.; Chong, T.; Yang, J.; Li, H.; Chen, H. Kinesin motor protein KIFC1 is a target protein of miR-338-3p and associated with poor prognosis and progression of renal cell carcinoma. Oncol. Res. 2018. [Google Scholar] [CrossRef]
- Sekino, Y.; Oue, N.; Shigematsu, Y.; Ishikawa, A.; Sakamoto, N.; Sentani, K.; Teishima, J.; Matsubara, A.; Yasui, W. KIFC1 induces resistance to docetaxel and is associated with survival of patients with prostate cancer. Urol. Oncol. 2017, 35, 31 e13–31 e20. [Google Scholar] [CrossRef]
- Xue, D.; Cheng, P.; Han, M.; Liu, X.; Xue, L.; Ye, C.; Wang, K.; Huang, J. An integrated bioinformatical analysis to evaluate the role of KIF4A as a prognostic biomarker for breast cancer. Onco Targets 2018, 11, 4755–4768. [Google Scholar] [CrossRef]
- Gao, H.; Chen, X.; Cai, Q.; Shang, Z.; Niu, Y. Increased KIF4A expression is a potential prognostic factor in prostate cancer. Oncol Lett. 2018, 15, 7941–7947. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Saito, M.; Saito, K.; Kanke, Y.; Watanabe, Y.; Onozawa, H.; Hayase, S.; Sakamoto, W.; Ishigame, T.; Momma, T.; et al. Enhanced expression of KIF4A in colorectal cancer is associated with lymph node metastasis. Oncol Lett. 2018, 15, 2188–2194. [Google Scholar] [CrossRef]
- Wang, H.; Lu, C.; Li, Q.; Xie, J.; Chen, T.; Tan, Y.; Wu, C.; Jiang, J. The role of Kif4A in doxorubicin-induced apoptosis in breast cancer cells. Mol. Cells 2014, 37, 812–818. [Google Scholar] [CrossRef]
- Taniwaki, M.; Takano, A.; Ishikawa, N.; Yasui, W.; Inai, K.; Nishimura, H.; Tsuchiya, E.; Kohno, N.; Nakamura, Y.; Daigo, Y. Activation of KIF4A as a prognostic biomarker and therapeutic target for lung cancer. Clin. Cancer Res. 2007, 13, 6624–6631. [Google Scholar] [CrossRef]
- Yang, Z.; Li, C.; Yan, C.; Li, J.; Yan, M.; Liu, B.; Zhu, Z.; Wu, Y.; Gu, Q. KIF14 promotes tumor progression and metastasis and is an independent predictor of poor prognosis in human gastric cancer. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 181–192. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, Y.; Liang, P.; Zhang, Z.; Guo, X.; Xia, L.; Zhao, Y.; Shu, X.S.; Sun, S.; Ying, Y.; et al. Overexpression of a novel candidate oncogene KIF14 correlates with tumor progression and poor prognosis in prostate cancer. Oncotarget 2017, 8, 45459–45469. [Google Scholar] [CrossRef]
- Qiu, H.L.; Deng, S.Z.; Li, C.; Tian, Z.N.; Song, X.Q.; Yao, G.D.; Geng, J.S. High expression of KIF14 is associated with poor prognosis in patients with epithelial ovarian cancer. Eur. Rev. Med. Pharm. Sci. 2017, 21, 239–245. [Google Scholar]
- O’Hare, M.; Shadmand, M.; Sulaiman, R.S.; Sishtla, K.; Sakisaka, T.; Corson, T.W. Kif14 overexpression accelerates murine retinoblastoma development. Int. J. Cancer 2016, 139, 1752–1758. [Google Scholar] [CrossRef]
- Sheng, Y.; Wang, W.; Hong, B.; Jiang, X.; Sun, R.; Yan, Q.; Zhang, S.; Lu, M.; Wang, S.; Zhang, Z.; et al. Upregulation of KIF20A correlates with poor prognosis in gastric cancer. Cancer Manag. Res. 2018, 10, 6205–6216. [Google Scholar] [CrossRef]
- Kawai, Y.; Shibata, K.; Sakata, J.; Suzuki, S.; Utsumi, F.; Niimi, K.; Sekiya, R.; Senga, T.; Kikkawa, F.; Kajiyama, H. KIF20A expression as a prognostic indicator and its possible involvement in the proliferation of ovarian clearcell carcinoma cells. Oncol. Rep. 2018, 40, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Huang, X.; Chen, Y.; Fu, Y.; Xu, C.; Xiang, W.; Li, C.; Zhang, S.; Yu, C. Aberrant KIF20A expression might independently predict poor overall survival and recurrence-free survival of hepatocellular carcinoma. IUBMB Life 2018, 70, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Huang, W.; Shi, H. Positive expression of KIF20A indicates poor prognosis of glioma patients. Onco Targets 2016, 9, 6741–6749. [Google Scholar] [CrossRef] [PubMed]
- Puttmann, S.; Senner, V.; Braune, S.; Hillmann, B.; Exeler, R.; Rickert, C.H.; Paulus, W. Establishment of a benign meningioma cell line by hTERT-mediated immortalization. Lab. Invest. 2005, 85, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Mawrin, C. Animal models of meningiomas. Chin. Clin. Oncol. 2017, 6, S6. [Google Scholar] [CrossRef]
- Sahm, F.; Schrimpf, D.; Jones, D.T.; Meyer, J.; Kratz, A.; Reuss, D.; Capper, D.; Koelsche, C.; Korshunov, A.; Wiestler, B.; et al. Next-generation sequencing in routine brain tumor diagnostics enables an integrated diagnosis and identifies actionable targets. Acta Neuropathol. 2016, 131, 903–910. [Google Scholar] [CrossRef]
| Clinical Factors | Group | Patients | |
|---|---|---|---|
| n | (%) | ||
| Sex | Male | 93 | 45 |
| Female | 115 | 55 | |
| Age at 1st diagnosis (years) | Median | 59 | |
| Range | 18–87 | ||
| WHO Grade | WHO°I | 61 | 29.3 |
| WHO°II | 88 | 42.3 | |
| WHO°III | 59 | 28.4 | |
| Subtype | Fibroblastic | 8 | 3.8 |
| Meningothelial | 9 | 4.3 | |
| Transitional | 31 | 14.9 | |
| Psammomatous | 4 | 1.9 | |
| Angiomatous | 1 | 0.5 | |
| Secretory | 3 | 1.4 | |
| Atypical | 75 | 36.1 | |
| Anaplastic | 40 | 19.2 | |
| Rhabdoid | 2 | 1.0 | |
| Papillary | 2 | 1.0 | |
| Mixed/Unknown | 33 | 15.9 | |
| Location | Convexity | 75 | 36.1 |
| Falx | 32 | 15.4 | |
| Tentorial or parasagittal | 42 | 20.2 | |
| Cranial base | 48 | 23.1 | |
| Multiple | 4 | 1.9 | |
| Other/NA | 7 | 3.4 | |
| Primary or recurrent tumor | Primary tumor | 134 | 64 |
| Recurrent tumor | 74 | 36 | |
| Resection grade | Simpson °I | 101 | 48.6 |
| Simpson °II | 59 | 28.4 | |
| Simpson °III | 27 | 13 | |
| Simpson °IV | 18 | 8.7 | |
| Simpson °V | 1 | 0.5 | |
| Unknown | 2 | 1.0 | |
| Postoperative treatment | Radiotherapy | 62 | |
| Chemotherapy | 6 | ||
| Clinical Factors | Group | Patients | |
|---|---|---|---|
| n | (%) | ||
| Sex | Male | 39 | 36.8 |
| Female | 67 | 63.2 | |
| Age at 1st diagnosis (years) | Median | 60 | |
| Range | 24–85 | ||
| WHO Grade | WHO°I | 44 | 41.5 |
| WHO°II | 50 | 47.2 | |
| WHO°III | 12 | 11.3 | |
| Subtype | Fibroblastic | 7 | 6.6 |
| Meningothelial | 7 | 6.6 | |
| Transitional | 20 | 18.9 | |
| Psammomatous | 4 | 3.8 | |
| Angiomatous | 0 | 0.0 | |
| Secretory | 3 | 2.8 | |
| Atypical | 42 | 39.6 | |
| Anaplastic | 9 | 8.5 | |
| Rhabdoid | 0 | 0.0 | |
| Papillary | 1 | 0.9 | |
| Mixed/Unknown | 13 | 12.3 | |
| Location | Convexity | 38 | 35.8 |
| Falx | 16 | 15.1 | |
| Tentorial or parasagittal | 22 | 20.8 | |
| Cranial base | 25 | 23.6 | |
| Multiple | 0 | 0.0 | |
| Other/NA | 5 | 4.7 | |
| Primary or recurrent tumor | Primary tumor | 106 | 100 |
| Recurrent tumor | 0 | 0 | |
| Resection grade | Simpson °I | 59 | 55.7 |
| Simpson °II | 33 | 31.1 | |
| Simpson °III | 14 | 13.2 | |
| Future clinical progression | No future recurrence | 63 | |
| Recurrence with same WHO° | 34 | ||
| Recurrence with higher WHO° | 9 | ||
| Median | |||
| Follow-up time (months) | WHO°I | 135.5 | |
| WHO°II | 100.1 | ||
| WHO°III | 47.9 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jungwirth, G.; Yu, T.; Moustafa, M.; Rapp, C.; Warta, R.; Jungk, C.; Sahm, F.; Dettling, S.; Zweckberger, K.; Lamszus, K.; et al. Identification of KIF11 as a Novel Target in Meningioma. Cancers 2019, 11, 545. https://doi.org/10.3390/cancers11040545
Jungwirth G, Yu T, Moustafa M, Rapp C, Warta R, Jungk C, Sahm F, Dettling S, Zweckberger K, Lamszus K, et al. Identification of KIF11 as a Novel Target in Meningioma. Cancers. 2019; 11(4):545. https://doi.org/10.3390/cancers11040545
Chicago/Turabian StyleJungwirth, Gerhard, Tao Yu, Mahmoud Moustafa, Carmen Rapp, Rolf Warta, Christine Jungk, Felix Sahm, Steffen Dettling, Klaus Zweckberger, Katrin Lamszus, and et al. 2019. "Identification of KIF11 as a Novel Target in Meningioma" Cancers 11, no. 4: 545. https://doi.org/10.3390/cancers11040545
APA StyleJungwirth, G., Yu, T., Moustafa, M., Rapp, C., Warta, R., Jungk, C., Sahm, F., Dettling, S., Zweckberger, K., Lamszus, K., Senft, C., Loehr, M., Keßler, A. F., Ketter, R., Westphal, M., Debus, J., von Deimling, A., Simon, M., Unterberg, A., ... Herold-Mende, C. (2019). Identification of KIF11 as a Novel Target in Meningioma. Cancers, 11(4), 545. https://doi.org/10.3390/cancers11040545






