Trending Towards Safer Breast Cancer Surgeries? Examining Acute Complication Rates from A 13-Year NSQIP Analysis
Abstract
1. Introduction
2. Methods and Materials
2.1. Data Collection
2.2. Complications and Outcome Measures
2.3. NSQIP Variable Definitions
2.4. Statistics
3. Results
3.1. Participant Pool and Demographics
3.2. Overall Complication Rate and Trend Analysis
3.3. Independent Factors Associated with Complications (p < 0.05)
3.4. Adjusted Predictive Factors
3.4.1. Predicting Factors Associated with Lower Complication Rates
3.4.2. Predicting Factors Associated with Higher Complication Rates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Abbrev. | Definition |
| BCT | Breast Conservative Therapy |
| IvBC | Invasive Breast Cancer |
| DCIS | Ductal Carcinoma in situ |
| PM | Partial Mastectomy |
| M | Mastectomy |
| M + R | Mastectomy with reconstruction |
| OPS | Oncoplastic Surgery |
| M + I | Mastectomy with breast prosthesis, delayed-insertion or tissue expander for implant placement |
| M + MF | Mastectomy with Muscular Flap |
| MLR | Multivariable Logistical Regression |
| R2 | Linear Regression |
| ACS | American College of Surgeons |
| NSQIP | National Surgical Quality Improvement Program |
| CPT | Current Procedural Terminology |
| ICD-9 | International Classification of Diseases Ninth Revision |
| CG | Categories |
References
- Jonczyk, M.M.; Jean, J.; Graham, R.; Chatterjee, A. Surgical trends in breast cancer: A rise in novel operative treatment options over a 12 year analysis. Breast Cancer Res. Treat. 2018. [Google Scholar] [CrossRef] [PubMed]
- Cil, T.D.; Cordeiro, E. Complications of oncoplastic breast surgery involving soft tissue transfer versus breast-conserving surgery: An analysis of the NSQIP database. Ann. Surg. Oncol. 2016, 23, 3266–3271. [Google Scholar] [CrossRef] [PubMed]
- Chand, N.D.; Browne, V.; Paramanathan, N.; Peiris, L.J.; Laws, S.A.; Rainsbury, R.M. Patient-reported outcomes are better after oncoplastic breast conservation than after mastectomy and autologous reconstruction. Plast. Reconstr. Surg. Glob. Open 2017, 5, e1419. [Google Scholar] [CrossRef] [PubMed]
- Bazzarelli, A.; Zhang, J.; Arnaout, A. Patient-Reported Satisfaction Following Oncoplastic Breast-Conserving Therapy. In Proceedings of the American Society of Breast Surgeons Annual Meeting, Dallas, TX, USA, 13 April 2016. [Google Scholar]
- Carlson, G.W. Trends in autologous breast reconstruction. Semin. Plast. Surg. 2004, 18, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Schmauss, D.; Machens, H.G.; Harder, Y. Breast reconstruction after mastectomy. Front. Surg. 2015, 71. [Google Scholar] [CrossRef]
- Anker, C.J.; Hymas, R.V.; Ahluwalia, R.; Kokeny, K.E.; Avizonis, V.; Boucher, K.M.; Neumayer, L.A.; Agarwal, J.P. The effect of radiation on complication rates and patient satisfaction in breast reconstruction using temporary tissue expanders and permanent implants. Breast J. 2015, 21, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S.A.; Jeevaratnam, J.A.; Agrawal, A.; Cutress, R.I. Mastectomy skin flap necrosis: Challenges and solutions. Breast Cancer 2017, 9, 141–152. [Google Scholar] [CrossRef]
- Carlson, G.W.; Bostwick, J., 3rd; Styblo, T.M.; Moore, B.; Bried, J.T.; Murray, D.R.; Wood, W.C. Skin-sparing mastectomy. Oncologic and reconstructive considerations. Ann. Surg. 1997, 225, 570–575. [Google Scholar] [CrossRef]
- Angarita, F.A.; Acuna, S.A.; Cordeiro, E.; Elnahas, A.; Sutradhar, S.; Jackson, T.; Cil, T.D. Thirty-day postoperative morbidity and mortality in elderly women with breast cancer: An analysis of the NSQIP database. Breast Cancer Res. Treat. 2018, 170, 373–379. [Google Scholar] [CrossRef]
- Sotheran, W.J.; Rainsbury, R.M. Skin-sparing mastectomy in the UK—A review of current practice. Ann. R. Coll. Surg. Engl. 2004, 86, 82–86. [Google Scholar] [CrossRef]
- O’Brien, W.; Hasselgren, P.O.; Hummel, R.P.; Coith, R.; Hyams, D.; Kurtzman, L.; Neale, H.W. Comparison of postoperative wound complications and early cancer recurrence between patients undergoing mastectomy with or without immediate breast reconstruction. Am. J. Surg. 1993, 166, 1–5. [Google Scholar] [CrossRef]
- Services USDoHH. Body Mass Index (BMI); Center for Disease Control and Prevention: Atlanta, GA, USA, 2015. Available online: https://www.cdc.gov/healthyweight/assessing/bmi/index.html (accessed on 1 November 2018).
- El-Tamer, M.B.; Ward, B.M.; Schifftner, T.; Neumayer, L.; Khuri, S.; Henderson, W. Morbidity and mortality following breast cancer surgery in women: National benchmarks for standards of care. Ann. Surg. 2007, 245, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Losken, A.; Styblo, T.M.; Carlson, G.W.; Jones, G.E.; Amerson, B.J. Management algorithm and outcome evaluation of partial mastectomy defects treated using reduction or mastopexy techniques. Ann. Plast. Surg. 2007, 59, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Tenofsky, P.L.; Dowell, P.; Topalovski, T.; Helmer, S.D. Surgical, oncologic, and cosmetic differences between oncoplastic and nononcoplastic breast conserving surgery in breast cancer patients. Am. J. Surg. 2014, 207, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, D.A.; Upchurch, G.R., Jr.; DeMonner, S.; Stanley, J.C.; Khuri, S.; Daley, J.; Henderson, W.G.; Hayward, R. Perioperative cardiovascular risk stratification of patients with diabetes who undergo elective major vascular surgery. J. Vasc. Surg. 2002, 35, 894–901. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Breast Cancer Facts & Figures 2017–2018; American Cancer Society, Inc.: Atlanta, GA, USA, 2017; Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2017-2018.pdf (accessed on 10 October 2018).
- Labor USdo. Health Plans & Benefits: Womens’ Health and Cancer Rights; United States department of Labor: Washington, DC, USA, 2018. Available online: https://www.dol.gov/general/topic/health-plans/womens (accessed on 11 December 2018).
- Bertozzi, N.; Pesce, M.; Santi, P.L.; Raposio, E. Oncoplastic breast surgery: Comprehensive review. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2572–2585. [Google Scholar] [PubMed]
- Carter, S.A.; Lyons, G.R.; Kuerer, H.M.; Bassett, R.L., Jr.; Oates, S.; Thompson, A.; Caudle, A.S.; Mittendorf, E.A.; Bedrosian, I.; Lucci, A.; et al. Operative and oncologic outcomes in 9861 patients with operable breast cancer: Single-institution analysis of breast conservation with oncoplastic reconstruction. Ann. Surg. Oncol. 2016, 23, 3190–3198. [Google Scholar] [CrossRef]
- Haloua, M.H.; Krekel, N.M.; Winters, H.A.; Rietveld, D.H.; Meijer, S.; Bloemers, F.W.; van den Tol, M.P. A systematic review of oncoplastic breast-conserving surgery: Current weaknesses and future prospects. Ann. Surg. 2013, 257, 609–620. [Google Scholar] [CrossRef]
- Campbell, E.J.; Romics, L. Oncological safety and cosmetic outcomes in oncoplastic breast conservation surgery, a review of the best level of evidence literature. Breast Cancer 2017, 9, 521–530. [Google Scholar] [CrossRef]
- Manning, A.T.; Sacchini, V.S. Conservative mastectomies for breast cancer and risk-reducing surgery: The Memorial Sloan Kettering Cancer Center experience. Gland. Surg. 2016, 5, 55–62. [Google Scholar] [CrossRef]
- Bennett, K.G.; Qi, J.; Kim, H.M.; Hamill, J.B.; Pusic, A.L.; Wilkins, E.G. Comparison of 2-year complication rates among common techniques for postmastectomy breast reconstruction. JAMA Surgery 2018, 153, 901–908. [Google Scholar] [CrossRef]
- Chakravorty, A.; Shrestha, A.K.; Sanmugalingam, N.; Rapisarda, F.; Roche, N.; Querci Della Rovere, G.; Macneill, F.A. How safe is oncoplastic breast conservation? Comparative analysis with standard breast conserving surgery. Eur. J. Surg. Oncol. 2012, 38, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Asban, A.; Homsy, C.; Chen, L.; Fisher, C.; Losken, A.; Chatterjee, A. A cost-utility analysis comparing large volume displacement oncoplastic surgery to mastectomy with single stage implant reconstruction in the treatment of breast cancer. Breast 2018, 41, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Offodile, A.C.; Asban, A.; Minasian, R.A.; Losken, A.; Graham, R.; Chen, L.; Czerniecki, B.J.; Fisher, C. A cost-utility analysis comparing oncoplastic breast surgery to standard lumpectomy in large breasted women. Adv. Breast Cancer Res. 2018, 7, 14. [Google Scholar] [CrossRef]
- Jonczyk, M.M.; Patel, K.; Graham, R.; Naber, S.; Erban, J.; Chen, L.; Chatterjee, A. Does Oncoplastic Surgery Offer Low Positive Margin Rates Using New SSO/ASBrS/ASTRO Margin Guidelines? In Proceedings of the San Antonio Breast Cancer Symposium, San Antonio, TX, USA, 5–9 December 2017. Tufts Medical Center: Poster Presentation. [Google Scholar]
- Chang, E.I.; Vaca, L.; DaLio, A.L.; Festekjian, J.H.; Crisera, C.A. Assessment of advanced age as a risk factor in microvascular breast reconstruction. Ann. Plast. Surg. 2011, 67, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, E.; Jackson, T.D.; Elnahas, A.; Cil, T. Higher rate of breast surgery complications in patients with metastatic breast cancer: An analysis of the NSQIP database. Ann. Surg. Oncol. 2014, 21, 3167–3172. [Google Scholar] [CrossRef] [PubMed]
- Lopez-de-Andres, A.; Jimenez-Trujillo, I.; Hernandez-Barrera, V.; de Miguel-Diez, J.; Mendez-Bailon, M.; de Miguel-Yanes, J.M.; Perez-Farinos, N.; Salinero-Fort, M.A.; Del Barrio, J.L.; Romero-Maroto, M.; et al. Association of type 2 diabetes with in-hospital complications among women undergoing breast cancer surgical procedures. A retrospective study using the Spanish National Hospital Discharge Database, 2013–2014. BMJ Open 2017, 7, e017676. [Google Scholar] [CrossRef]
- Tanaka, S.; Hayek, G.; Jayapratap, P.; Yerrasetti, S.; Hilaire, H.S.; Sadeghi, A.; Corsetti, R.; Fuhrman, G. The impact of chemotherapy on complications associated with mastectomy and immediate autologous tissue reconstruction. Am. Surg. 2016, 82, 713–717. [Google Scholar]
- Cheng, H.; Clymer, J.W.; Po-Han Chen, B.; Sadeghirad, B.; Ferko, N.C.; Cameron, C.G.; Hinoul, P. Prolonged operative duration is associated with complications: A systematic review and meta-analysis. J. Surg. Res. 2018, 229, 134–144. [Google Scholar] [CrossRef]
- Hackett, N.J.; De Oliveira, G.S.; Jain, U.K.; Kim, J.Y. ASA class is a reliable independent predictor of medical complications and mortality following surgery. Int. J. Surg. 2015, 18, 184–190. [Google Scholar] [CrossRef]
- Smetana, G.W.; Lawrence, V.A.; Cornell, J.E. Preoperative pulmonary risk stratification for noncardiothoracic surgery: Systematic review for the American College of Physicians. Ann. Intern. Med. 2006, 144, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; MacKenzie, C.R.; Gold, J.P.; Ales, K.L.; Topkins, M.; Shires, G.T. Intraoperative blood pressure. What patterns identify patients at risk for postoperative complications? Ann. Surg. 1990, 212, 5675–5680. [Google Scholar] [CrossRef]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Colvin, M.M.; Drazner, M.H.; Filippatos, G.S.; Fonarow, G.C.; Givertz, M.M.; et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines and the Heart Failure Society of America. Circulation 2017, 136, e137–e161. [Google Scholar] [CrossRef] [PubMed]
- Hammill, B.G.; Curtis, L.H.; Bennett-Guerrero, E.; O’Connor, C.M.; Jollis, J.G.; Schulman, K.A.; Hernandez, A.F. Impact of heart failure on patients undergoing major noncardiac surgery. Anesthesiology 2008, 108, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Smit-Fun, V.; Buhre, W.F. The patient with chronic heart failure undergoing surgery. Curr. Opin. Anaesthesiol. 2016, 29, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Fuentes, G.; Hashmi, H.R.; Venkatram, S. Perioperative evaluation of patients with pulmonary conditions undergoing non-cardiothoracic surgery. Health Serv. Insights 2016, 9 (Suppl. 1), 9–23. [Google Scholar] [CrossRef]
- Bousquet, J.; Clark, T.J.; Hurd, S.; Khaltaev, N.; Lenfant, C.; O’Byrne, P.; Sheffer, A. GINA guidelines on asthma and beyond. Allergy 2007, 62, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Fleisher, L.A.; Fleischmann, K.E.; Auerbach, A.D.; Barnason, S.A.; Beckman, J.A.; Bozkurt, B.; Davila-Roman, V.G.; Gerhard-Herman, M.D.; Holly, T.A.; Kane, G.C.; et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. Circulation 2014, 130, 2215–2245. [Google Scholar] [CrossRef]
- Hobson, C.; Singhania, G.; Bihorac, A. Acute kidney injury in the surgical patient. Crit. Care Clin. 2015, 31, 705–723. [Google Scholar] [CrossRef]
- Mendu, M.L.; Ciociolo, G.R., Jr.; McLaughlin, S.R.; Graham, D.A.; Ghazinouri, R.; Parmar, S.; Grossier, A.; Rosen, R.; Laskowski, K.R.; Riella, L.V.; et al. A decision-making algorithm for initiation and discontinuation of RRT in severe AKI. Clin. J. Am. Soc. Nephrol CJASN 2017, 12, 228–236. [Google Scholar] [CrossRef]
- Stevens, P.E.; Levin, A. Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline. Ann. Inter. Med. 2013, 158, 825–830. [Google Scholar] [CrossRef] [PubMed]
| Clusters | Individual Complication with NSQIP Code |
|---|---|
| Wound Complications | Superficial Incisional Infection SSI: SUPINFEC Deep incisional Infection DSI: WNDINFD |
| Infectious Complications | Organ/Space SSI: ORGSPCSSI Urinary Tract Infection UTI: URNINFEC Sepsis: OTHSYSEP Septic Shock: OTHSESHOCK |
| Respiratory Complications | Pneumonia: OUPNEUMO Unplanned re-intubation: REINTUB |
| Thromboembolic Complications | Pulmonary Embolism: PULEMBOL DVT requiring Therapy: OTHDVT |
| Bleeding Complications | Intraoperative or post-operative transfusion: OTHBLEED |
| Renal Complications | Postoperative Renal Failure: OPRENAFL Progressive Renal Insufficiency: RENAFAIL |
| Cardiac Complications | Cardiac arrest requiring CPR: CDARREST Myocardial infarction: CDMI |
| Stroke Complications | Stroke: CNSCVA |
| Demographics (%) | PM n = 95,468 | OPS n = 7279 | M n = 70,616 | M + MF n = 4747 | M + I n = 44,093 | p-Value | |
|---|---|---|---|---|---|---|---|
| Age | Young: <60 | 56.63 | 54.71 | 42.19 | 75.27 | 78.25 | <0.0001 |
| Older: >60 | 43.37 | 45.29 | 57.81 | 24.73 | 21.75 | ||
| Race | White | 72.66 | 74.91 | 68.73 | 73.98 | 76.77 | <0.0001 |
| Black | 10.71 | 11.84 | 11.83 | 12.79 | 7.99 | ||
| Asian/Pacific | 3.88 | 4.09 | 6.13 | 3.71 | 4.39 | ||
| Native | 0.50 | 0.16 | 0.72 | 0.21 | 0.22 | ||
| Unknown | 12.24 | 8.98 | 12.59 | 9.31 | 10.63 | ||
| Pathology | Invasive | 78.46 | 81.04 | 86.58 | 78.83 | 80.29 | <0.0001 |
| DCIS | 21.54 | 19.10 | 13.42 | 21.17 | 19.71 | ||
| Admission Status | Inpatient | 7.15 | 15.11 | 51.81 | 93.66 | 61.99 | <0.0001 |
| Outpatient | 92.85 | 84.89 | 48.19 | 6.34 | 38.01 | ||
| BMI | Underweight | 2.09 | 1.36 | 2.80 | 1.18 | 2.29 | <0.0001 |
| Normal | 27.27 | 27.17 | 29.20 | 30.90 | 41.27 | ||
| Overweight | 30.55 | 29.43 | 29.35 | 33.85 | 28.72 | ||
| Obese | 40.07 | 29.43 | 38.65 | 34.02 | 27.71 | ||
| Diabetic | Non-Diabetic | 86.91 | 88.68 | 84.24 | 93.22 | 94.60 | <0.0001 |
| Diabetic-Insulin | 3.67 | 3.20 | 4.94 | 1.85 | 1.38 | ||
| Diabetic-Oral | 9.42 | 8.12 | 10.82 | 4.93 | 4.02 | ||
| Pregnancy Status | 0.03 | 0.00 | 0.06 | 0.04 | 0.02 | <0.0001 | |
| Steroid Use | 1.87 | 1.91 | 2.70 | 1.56 | 1.76 | <0.0001 | |
| Prior Chemotherapy | 0.83 | 1.20 | 3.37 | 2.53 | 1.66 | <0.0001 | |
| Prior Radiation Therapy | 0.08 | 0.04 | 0.26 | 0.34 | 0.09 | <0.0001 | |
| Recent Weight Loss | 0.27 | 0.34 | 0.72 | 0.40 | 0.29 | <0.0001 | |
| PMH of Angina | 0.07 | 0.07 | 0.11 | 0.04 | 0.02 | <0.0001 | |
| PMH of Hypertension | 0.47 | 0.40 | 0.50 | 0328 | 0.24 | <0.0001 | |
| PMH of CHF | 0.27 | 0.29 | 0.43 | 0.06 | 0.04 | <0.0001 | |
| PMH of Renal Failure | 0.03 | 0.03 | 0.04 | 0.00 | 0.01 | 0.009 | |
| PMH of Bleeding Disorders | 1.64 | 1.14 | 2.25 | 0.57 | 0.64 | <0.0001 | |
| PMH of COPD | 3.05 | 1.81 | 3.96 | 0.93 | 0.80 | <0.0001 | |
| Recent Pneumonia | 0.01 | 0.00 | 0.01 | 0.00 | 0.00 | <0.0001 | |
| Dyspnea | At Rest | 0.29 | 0.08 | 0.42 | 0.06 | 0.06 | <0.0001 |
| Moderate | 5.56 | 3.49 | 7.48 | 3.60 | 2.46 | ||
| None | 94.15 | 96.43 | 92.11 | 96.33 | 97.48 | ||
| ASA Class | 1 | 6.19 | 5.19 | 3.74 | 6.38 | 7.88 | <0.0001 |
| 2 | 58.95 | 60.35 | 51.60 | 69.20 | 69.34 | ||
| 3 | 33.25 | 33.48 | 42.22 | 24.04 | 21.69 | ||
| 4 | 1.36 | 0.80 | 2.33 | 0.32 | 0.30 | ||
| 5 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||
| Open Wound Infection | 0.37 | 0.25 | 1.31 | 1.22 | 0.20 | <0.0001 | |
| Prior Infection | SIRS | 0.20 | 0.23 | 0.42 | 0.55 | 0.23 | <0.0001 |
| (Within 48 h) | Sepsis | 0.01 | 0.01 | 0.04 | 0.06 | 0.00 | |
| Any Operation in Last 30 Days | 1.92 | 0.91 | 1.30 | 2.19 | 0.66 | <0.0001 | |
| Operating Time | Less than 1 h | 0.48 | 0.16 | 0.13 | 0.01 | 0.01 | <0.0001 |
| 1–2 h | 0.42 | 0.37 | 0.47 | 0.04 | 0.11 | ||
| 2–3 h | 0.08 | 0.23 | 0.12 | 0.07 | 0.29 | ||
| 3–5 h | 0.02 | 0.18 | 0.02 | 0.34 | 0.45 | ||
| 5–10 h | 0 | 0.05 | 0 | 0.47 | 0.12 | ||
| 10+ h | 0 | 0 | 0 | 0.06 | 0 | ||
| Return to OR (Within 30 Days) | 0.05 | 0.04 | 0.04 | 0.08 | 0.07 | <0.0001 | |
| Surgery Type | PM **, OPS, M, M + R, M + I |
|---|---|
| Age | Young: <60 Older: >60 ** |
| Race | White **, Black, Asian, Native, Unknown |
| Pathology | Invasive Breast Cancer Ductal Carcinoma in situ ** |
| Admission Status | Inpatient ** or Outpatient |
| BMI | Underweight Normal ** OverweightObese |
| Pregnancy Status | Yes or No ** |
| Smoking Status | Smoker or Non-Smoker ** |
| Smoking Pack Per Day (PPD) | None ** 0–20 PPD 21–50 PPD 50–100 PPD >100 PPD |
| Steroid Use | Yes or No ** |
| Prior Chemotherapy | Yes or No ** |
| Prior Radiation Therapy | Yes or No ** |
| Peri-Operative ASA Class | 1 **, 2, 3, 4, 5 |
| Operative Wound Infection | Yes or No ** |
| Any Operation in Last 30 Days | Yes or No ** |
| Recent Pneumonia | Yes or No ** |
| Diabetic | Non-Diabetic Diabetic on Insulin ** Diabetic on Oral medication |
| Recent Weight Loss | Yes or No ** |
| PMH of Angina | Yes or No ** |
| PMH of Congestive Heart Failure | Yes or No ** |
| PMH of Renal Failure | Yes or No ** |
| PMH of COPD | Yes or No ** |
| History of Bleeding Disorder | Yes or No ** |
| Operating Time | Less than 1 h ** 1–2 h 2–3 h 3–5 h 5–10 h Over 10 h |
| Categories → Complication (n, %) | BCT n = 102,747 | MAST n = 119,456 | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PM n = 95,468 | OPS n = 7279 | M n = 70,616 | M + MF n = 4747 | M + I n = 44,093 | |||||||
| Wound Complication | 1341 | 1.40 | 128 | 1.76 | 2385 | 3.38 | 244 | 5.14 | 1325 | 3.01 | <0.0001 |
| Infectious | 456 | 0.48 | 50 | 0.69 | 823 | 1.17 | 92 | 1.94 | 652 | 1.48 | <0.0001 |
| Respiratory | 90 | 0.09 | 8 | 0.11 | 172 | 0.24 | 12 | 0.25 | 43 | 0.10 | <0.0001 |
| Bleeding | 66 | 0.07 | 28 | 0.38 | 844 | 1.20 | 206 | 4.34 | 277 | 0.63 | <0.0001 |
| Thromboembolic | 99 | 0.10 | 9 | 0.12 | 200 | 0.28 | 55 | 1.16 | 179 | 0.41 | <0.0001 |
| Renal | 21 | 0.02 | 2 | 0.03 | 53 | 0.08 | 3 | 0.06 | 11 | 0.02 | <0.0001 |
| Cardiac | 42 | 0.04 | 4 | 0.05 | 89 | 0.13 | 3 | 0.06 | 8 | 0.02 | <0.0001 |
| Stroke | 29 | 0.03 | 4 | 0.05 | 64 | 0.09 | 4 | 0.08 | 10 | 0.02 | <0.0001 |
| Overall Complication Rate | 2144 | 2.25% | 233 | 3.2% | 4630 | 6.56% | 619 | 13.04% | 2505 | 5.68% | <0.0001 |
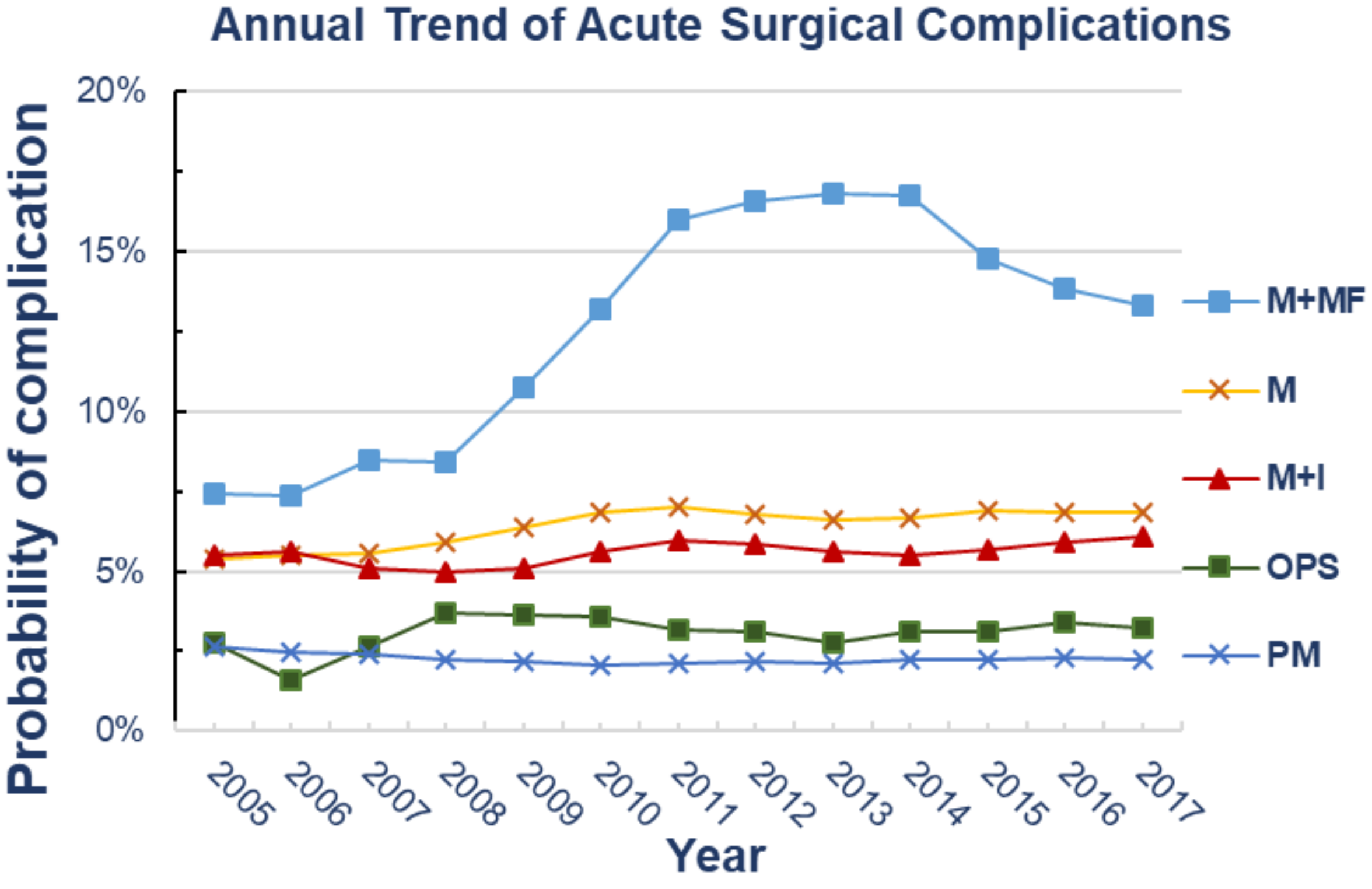
| Year | Trend of Surgical Complication Rates (n, %) * | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PM | OPS | M | M + I | M + MF | |||||||
| 2005 | 22 | 2.7% | 1 | 2.8% | 32 | 5.4% | 11 | 5.5% | 4 | 7.4% | |
| 2006 | 68 | 2.5% | 3 | 1.6% | 112 | 5.5% | 30 | 5.6% | 17 | 7.4% | |
| 2007 | 103 | 2.4% | 1 | 2.7% | 193 | 5.6% | 73 | 5.1% | 24 | 8.5% | |
| 2008 | 110 | 2.3% | 7 | 3.7% | 254 | 5.9% | 91 | 5.0% | 38 | 8.4% | |
| 2009 | 119 | 2.2% | 11 | 3.7% | 326 | 6.4% | 119 | 5.1% | 35 | 10.7% | |
| 2010 | 115 | 2.1% | 6 | 3.6% | 340 | 6.9% | 147 | 5.6% | 54 | 13.2% | |
| 2011 | 129 | 2.1% | 15 | 3.2% | 331 | 7.0% | 192 | 6.0% | 64 | 16.0% | |
| 2012 | 151 | 2.2% | 16 | 3.1% | 359 | 6.8% | 224 | 5.8% | 58 | 16.5% | |
| 2013 | 177 | 2.1% | 16 | 2.7% | 391 | 6.6% | 249 | 5.6% | 56 | 16.8% | |
| 2014 | 199 | 2.3% | 21 | 3.1% | 422 | 6.7% | 252 | 5.5% | 54 | 16.7% | |
| 2015 | 276 | 2.3% | 37 | 3.1% | 436 | 6.9% | 280 | 5.7% | 47 | 14.7% | |
| 2016 | 278 | 2.3% | 33 | 3.4% | 494 | 6.8% | 320 | 5.9% | 34 | 13.8% | |
| 2017 | 295 | 2.2% | 45 | 3.2% | 434 | 6.8% | 340 | 6.1% | 41 | 13.3% | |
| Overall Trend Analysis | |||||||||||
| p-Value | (R2) | 0.29 | (0.27) | 0.67 | (0.14) | 0.004 | (0.71) | 0.02 | (0.39) | 0.01 | (0.60) |
| Complication | OR (95% CI) | Complication | OR (95% CI) |
|---|---|---|---|
| Wound Complications Protective Diabetic on Oral medication Non-Diabetic | 0.75 (0.63–0.88) ** 0.61 (0.53–0.70) *** | Infectious Complications Protective Diabetic on Oral medication Non-Diabetic | 0.74 (0.56–0.97) * 0.62 (0.49–0.78) *** |
| Risk | Risk | ||
| PMH of Angina PMH of COPD PMH of Bleeding Disorder Smoker BMI Obese BMI: Overweight | 2.22 (1.24–3.98) ** 1.29 (1.09–1.53) ** 1.44 (1.17–1.77) ** 1.61 (1.46–1.77) *** 2.17 (1.97–2.38) *** 1.34 (1.21–1.48) *** | PMH of CHF PMH of COPD PMH of Bleeding Disorder PMH of HTN BMI: Overweight BMI ObeseRecent Weight Loss | 2.13 (1.17–3.89) * 1.34 (1.02–1.76) * 1.73 (1.27–2.35) ** 1.20 (1.06–1.37) ** 1.36 (1.16–1.63) ** 1.62 (1.39–1.90) *** 3.48 (2.24–5.42) *** |
| Respiratory Complications Protective Age <60 | 0.66 (0.47–0.93) * | Bleeding Complications Protective Non-Diabetic | 0.65 (0.49–0.89) ** |
| Risk | Risk | ||
| PMH of COPD BMI: underweight Recent Weight Loss PMH of Bleeding Disorder | 1.80 (1.14–2.85) * 2.25 (1.14–4.42) * 2.76 (1.09–6.99) * 2.70 (1.67–4.36) *** | Recent Weight Loss PMH of CHF Steroid Use PMH of Bleeding Disorder | 1.96 (1.02–3.79) * 2.97 (1.67–5.26) * 1.82 (1.26–2.62) ** 2.62 (1.90–3.62) *** |
| Neuro Complications Protective Age <60 | 0.14 (0.06–0.32) *** | Thromboembolic Complications Risk BMI: Overweight BMI: Obese | 1.66 (1.18–2.36) ** 2.45 (1.77–3.40) *** |
| Renal Complications Protective Non-Diabetic | 0.35 (0.18–0.71) ** | Cardiac Complications Protective Age <60 | 0.53 (0.30–0.91) * |
| Risk | Risk | ||
| PMH of Bleeding Disorder PMH of Renal Failure PMH of CHF Recent Weight Loss | 2.50 (1.05–5.94) * 10.45 (1.19–91.74) * 5.06 (1.64–15.59) ** 8.39 (2.87–24.53) ** | BMI: Underweight PMH of Angina PMH of HTN | 2.94 (1.23–7.03) * 5.20 (1.20–22.46) * 2.34 (1.33–4.12) ** |
| Complication | OR (95% CI) | Complication | OR (95% CI) | Complication | OR (95% CI) |
|---|---|---|---|---|---|
| Wound Complications | Infectious Complications | Bleeding Complications | |||
| Protective | Protective | Protective | |||
| Outpatient | 0.84 (0.77–0.91) *** | Outpatient | 0.74 (0.64–0.85) *** | Outpatient | 0.20 (0.16–0.6) *** |
| Risk | Risk | Risk | |||
| M | 1.79 (1.62–1.98) *** | M | 1.78 (1.48–2.13) *** | OPS | 2.78 (1.20–6.45) * |
| M + MF | 2.24 (1.83–2.74) *** | M + MF | 2.24 (1.60–3.13) *** | M + I | 3.61 (2.28–5.73) *** |
| M + I | 1.72 (1.51–1.96) *** | M + I | 2.17 (1.73–2.72) *** | M | 5.78 (3.83–8.72) *** |
| ASA2 | 1.22 (1.02–1.48) * | ASA2 | 1.66 (1.14–2.40) ** | M + MF | 10.99 (6.73–17.90) *** |
| ASA3 | 1.57 (1.29–1.91) *** | ASA3 | 2.44 (1.67–3.58) *** | ASA3 | 2.18 (1.41–3.37) ** |
| ASA4 | 1.67 (1.23–2.26) ** | ASA4 | 4.09 (2.50–6.67) *** | ASA4 | 5.32 (3.12–9.08) *** |
| Open Wound | 1.83 (1.40–2.39) *** | Open Wound | 1.98 (1.33–2.94) ** | Open Wound | 2.09 (1.36–3.21) ** |
| OPT1 | 1.32 (1.18–1.48) *** | OPT2. | 1.36 (1.09–1.71) * | OPT4. | 2.92 (1.99–4.28) *** |
| OPT2 | 1.51 (1.33–1.72) *** | OPT3. | 1.70 (1.34–2.17) *** | OPT5. | 7.21 (4.22–12.33) *** |
| OPT3 | 1.75 (1.52–2.02) *** | OOPT4. | 2.55 (1.90–3.42) *** | ||
| OPT4 | 2.14 (1.78–2.57) *** | OPT5. | 2.64 (1.30–5.37) * | ||
| OPT5 | 2.81 (1.85–4.27) *** | ||||
| Thromboembolic Complications | Cardiac Complications | Respiratory Complications | |||
| Protective | Protective | Protective | |||
| Outpatient | 0.70 (0.52–0.95) * | Outpatient | 0.20 (0.11–0.35) *** | Outpatient | 0.24 (0.16–0.35) *** |
| Risk | M + I | 0.17 (0.04–0.83) * | Risk | ||
| M | 2.04 (1.37–3.02) ** | OPT1 | 0.59 (0.35–0.97) * | ASA4 | 6.62 (2.14–20.48) ** |
| M + MF | 3.91 (2.16–7.07) *** | OPT2 | 0.47 (0.24–0.93) * | ||
| M + I | 2.15 (1.33–3.47) ** | OPT3 | 0.28 (0.09–0.86) * | ||
| Prior operation within 30 days | 3.20 (1.97–5.20) *** | Risk | |||
| OPT3 | 1.95 (1.16–3.27) * | ASA4 | 10.31 (1.23–86.12) * | ||
| OPT4 | 3.40 (1.89–6.09) *** | ||||
| OPT5 | 6.08 (2.41–15.32) ** | Renal Complications | Neuro Complications | ||
| Protective | Protective | ||||
| Outpatient | 0.26 (0.13–0.52) ** | Outpatient | 0.45 (0.26–0.81) ** | ||
| OPT2 | 0.29 (0.12–0.71) ** | ||||
| Risk | |||||
| OPS | 4.58 (1.28–16.35) * | ||||
| M | 2.61 (1.31–5.21) ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jonczyk, M.M.; Jean, J.; Graham, R.; Chatterjee, A. Trending Towards Safer Breast Cancer Surgeries? Examining Acute Complication Rates from A 13-Year NSQIP Analysis. Cancers 2019, 11, 253. https://doi.org/10.3390/cancers11020253
Jonczyk MM, Jean J, Graham R, Chatterjee A. Trending Towards Safer Breast Cancer Surgeries? Examining Acute Complication Rates from A 13-Year NSQIP Analysis. Cancers. 2019; 11(2):253. https://doi.org/10.3390/cancers11020253
Chicago/Turabian StyleJonczyk, Michael M., Jolie Jean, Roger Graham, and Abhishek Chatterjee. 2019. "Trending Towards Safer Breast Cancer Surgeries? Examining Acute Complication Rates from A 13-Year NSQIP Analysis" Cancers 11, no. 2: 253. https://doi.org/10.3390/cancers11020253
APA StyleJonczyk, M. M., Jean, J., Graham, R., & Chatterjee, A. (2019). Trending Towards Safer Breast Cancer Surgeries? Examining Acute Complication Rates from A 13-Year NSQIP Analysis. Cancers, 11(2), 253. https://doi.org/10.3390/cancers11020253





