Outpatient Cancer Care Delivery in the Context of E-Oncology: A French Perspective on “Cancer outside the Hospital Walls”
Abstract
1. Introduction
2. Changes in Cancer Care
2.1. Epidemiological and Economic Constraints
2.2. Medical-Scientific Advances and Changes in Cancer Management
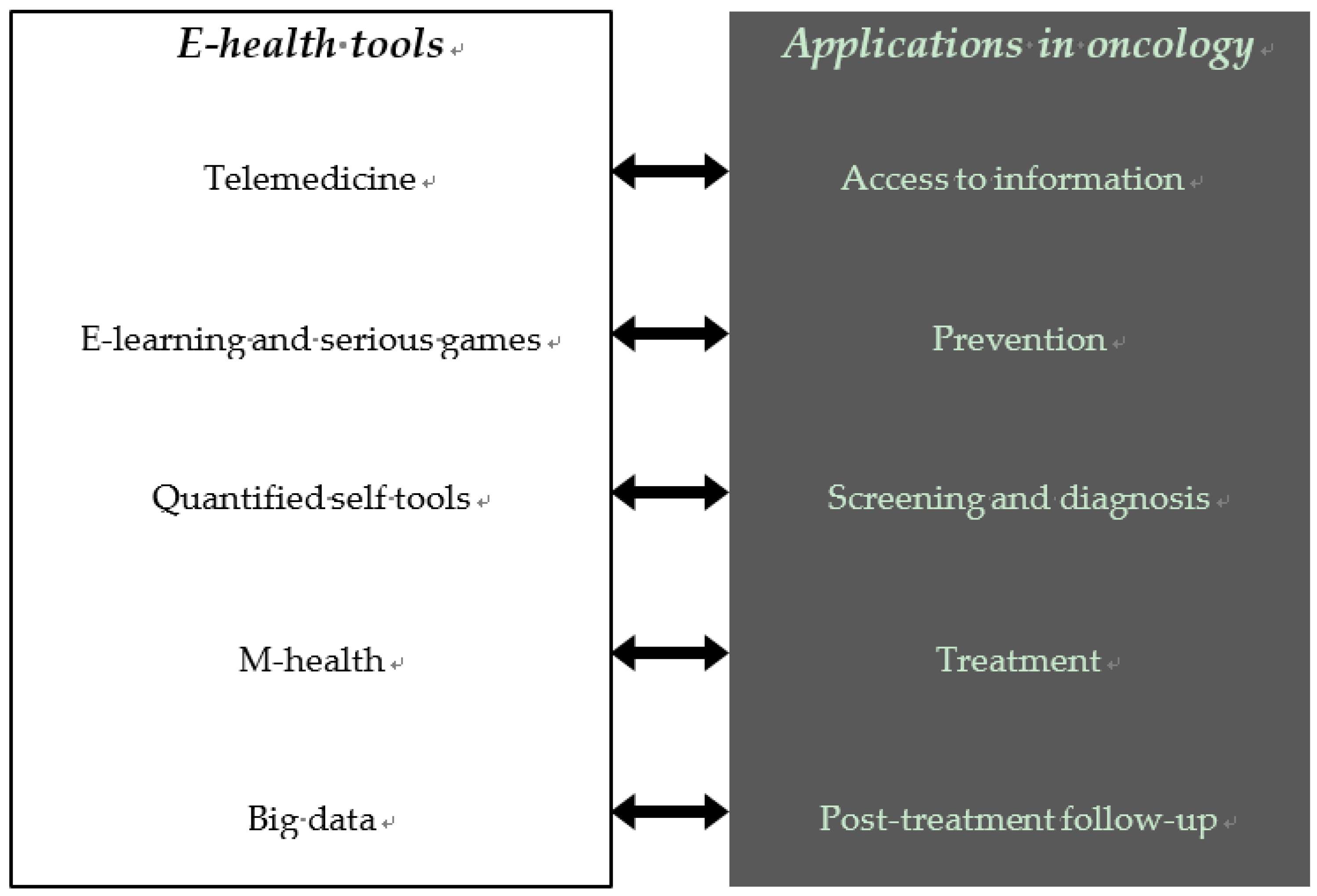
3. E-Health
4. Potential Impacts of E-Health in Oncology
4.1. Access to Information and Education
4.2. Prevention
4.3. Screening and Diagnosis
4.4. Treatment
4.5. Post-Treatment Follow-Up
5. Issues and Limits of “Cancer Outside the Hospital Walls”
5.1. For the Patients
5.2. For Health Care Professionals and Informal Caregivers
5.3. For Health Facilities
5.4. For the Public Authorities
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Heuckmann, J.M.; Thomas, R.K. A new generation of cancer genome diagnostics for routine clinical use: Overcoming the roadblocks to personalized cancer medicine. Ann. Oncol. 2015, 26, 1830–1837. [Google Scholar] [CrossRef]
- Siegel, R.L.; Jemal, A.; Wender, R.C.; Gansler, T.; Ma, J.; Brawley, O.W. An assessment of progress in cancer control. CA Cancer J. Clin. 2018, 68, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Giulia, F.; Luffer, S.; Kumar, R.; Wu, B.; Tavallai, M.; Bekele, R.T.; Birrer, M.J. How can molecular abnormalities influence our clinical approach. Ann. Oncol. 2017, 28, viii16–viii24. [Google Scholar] [CrossRef] [PubMed]
- Jordan, K.; Aapro, M.; Kaasa, S.; Ripamonti, C.I.; Scotte, F.; Strasser, F.; Young, A.; Bruera, E.; Herrstedt, J.; Keefe, D.; et al. European Society for Medical Oncology (ESMO) position paper on supportive and palliative care. Ann. Oncol. 2018, 29, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Hird, N.; Ghosh, S.; Kitano, H. Digital health revolution: Perfect storm or perfect opportunity for pharmaceutical R&D? Drug Discov. Today 2016, 21, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Kimberly, J.; Cronk, I. Making value a priority: How this paradigm shift is changing the landscape in health care. Ann. N. Y. Acad. Sci. 2016, 1381, 162–167. [Google Scholar] [CrossRef]
- Mooney, S.J.; Pejaver, V. Big Data in Public Health: Terminology, Machine Learning, and Privacy. Annu. Rev. Public Health 2018, 39, 95–112. [Google Scholar] [CrossRef]
- Munoz, R.F.; Chavira, D.A.; Himle, J.A.; Koerner, K.; Muroff, J.; Reynolds, J.; Rose, R.D.; Ruzek, J.I.; Teachman, B.A.; Schueller, S.M. Digital apothecaries: A vision for making health care interventions accessible worldwide. Mhealth 2018, 4, 18. [Google Scholar] [CrossRef]
- Escriva Boulley, G.; Leroy, T.; Bernetiere, C.; Paquienseguy, F.; Desfriches-Doria, O.; Preau, M. Digital health interventions to help living with cancer: A systematic review of participants’ engagement and psychosocial effects. Psychooncology 2018, 27, 2677–2686. [Google Scholar] [CrossRef]
- Binder-Foucard, F.; Bossard, N.; Delafosse, P.; Belot, A.; Woronoff, A.S.; Remontet, L.; French Network of Cancer Registries. Cancer incidence and mortality in France over the 1980–2012 period: Solid tumors. Rev. Epidemiol. Sante Publique 2014, 62, 95–108. [Google Scholar] [CrossRef]
- Gaujal, L.; Renou, M.; Dujaric, M.E.; Baffert, S.; Tardivon, A.; Kriegel, I.; Buecher, B.; Girod, A.; Grosset, L.; Asselain, B.; et al. First outpatient satisfaction questionnaire with day-surgery in a French comprehensive cancer center. Bull. Cancer 2016, 103, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Bekelman, J.E.; Sylwestrzak, G.; Barron, J.; Liu, J.; Epstein, A.J.; Freedman, G.; Malin, J.; Emanuel, E.J. Uptake and costs of hypofractionated vs conventional whole breast irradiation after breast conserving surgery in the United States, 2008–2013. JAMA 2014, 312, 2542–2550. [Google Scholar] [CrossRef]
- Thariat, J.; Creisson, A.; Chamignon, B.; Dejode, M.; Gastineau, M.; Hebert, C.; Boissin, F.; Topfer, C.; Gilbert, E.; Grondin, B.; et al. Integrating patient education in your oncology practice. Bull. Cancer 2016, 103, 674–690. [Google Scholar] [CrossRef]
- Brouard, B.; Bardo, P.; Vignot, M.; Bonnet, C.; Vignot, S. eHealth and mHealth: Current developments in 2014 and perspectives in oncology. Bull. Cancer 2014, 101, 940–950. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Topol, E.J. State of Telehealth. N. Engl. J. Med. 2016, 375, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Pipame: E-Santé, Faire Emerger L’offre Française en Répondant Aux Besoins Présents et Futurs Des Acteurs de Santé. 2016. Available online: http://www.entreprises.gouv.fr/files/files/directions_services/etudes-et-statistiques/prospective/Numerique/2016-02-Pipame-e-sante.pdf (accessed on 30 January 2019).
- Hazin, R.; Qaddoumi, I. Teleoncology: Current and future applications for improving cancer care globally. Lancet Oncol. 2010, 11, 204–210. [Google Scholar] [CrossRef]
- Sabesan, S.; Kelly, J. Are teleoncology models merely about avoiding long distance travel for patients? Eur. J. Cancer Care (Engl.) 2014, 23, 745–749. [Google Scholar] [CrossRef]
- Satcher, R.L.; Bogler, O.; Hyle, L.; Lee, A.; Simmons, A.; Williams, R.; Hawk, E.; Matin, S.; Brewster, A.M. Telemedicine and telesurgery in cancer care: Inaugural conference at MD Anderson Cancer Center. J. Surg. Oncol. 2014, 110, 353–359. [Google Scholar] [CrossRef]
- Abaza, H.; Marschollek, M. mHealth Application Areas and Technology Combinations*. A Comparison of Literature from High and Low/Middle Income Countries. Methods Inf. Med. 2017, 56, e105–e122. [Google Scholar] [CrossRef]
- Fairhurst, K.; Sheikh, A. Texting appointment reminders to repeated non-attenders in primary care: Randomised controlled study. Qual. Saf. Health Care 2008, 17, 373–376. [Google Scholar] [CrossRef]
- Gerber, B.S.; Stolley, M.R.; Thompson, A.L.; Sharp, L.K.; Fitzgibbon, M.L. Mobile phone text messaging to promote healthy behaviors and weight loss maintenance: A feasibility study. Health Inform. J. 2009, 15, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Holtz, B.; Whitten, P. Managing asthma with mobile phones: A feasibility study. Telemed. J. E. Health 2009, 15, 907–909. [Google Scholar] [CrossRef]
- Berger, M.L.; Curtis, M.D.; Smith, G.; Harnett, J.; Abernethy, A.P. Opportunities and challenges in leveraging electronic health record data in oncology. Future Oncol. 2016, 12, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, S.D.; Koch, S.B.; Williford, P.M.; Feldman, S.R.; Pearce, D.J. Web App- and Text Message-Based Patient Education in Mohs Micrographic Surgery-A Randomized Controlled Trial. Dermatol. Surg. 2018, 44, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Micheel, C.M.; Anderson, I.A.; Lee, P.; Chen, S.C.; Justiss, K.; Giuse, N.B.; Ye, F.; Kusnoor, S.V.; Levy, M.A. Internet-Based Assessment of Oncology Health Care Professional Learning Style and Optimization of Materials for Web-Based Learning: Controlled Trial With Concealed Allocation. J. Med. Internet Res. 2017, 19, e265. [Google Scholar] [CrossRef] [PubMed]
- Buller, D.B.; Berwick, M.; Lantz, K.; Buller, M.K.; Shane, J.; Kane, I.; Liu, X. Smartphone mobile application delivering personalized, real-time sun protection advice: A randomized clinical trial. JAMA Dermatol. 2015, 151, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Free, C.; Knight, R.; Robertson, S.; Whittaker, R.; Edwards, P.; Zhou, W.; Rodgers, A.; Cairns, J.; Kenward, M.G.; Roberts, I. Smoking cessation support delivered via mobile phone text messaging (txt2stop): A single-blind, randomised trial. Lancet 2011, 378, 49–55. [Google Scholar] [CrossRef]
- Mannino, R.G.; Myers, D.R.; Tyburski, E.A.; Caruso, C.; Boudreaux, J.; Leong, T.; Clifford, G.D.; Lam, W.A. Smartphone app for non-invasive detection of anemia using only patient-sourced photos. Nat. Commun. 2018, 9, 4924. [Google Scholar] [CrossRef]
- Sahin, D.; Hacisalihoglu, U.P.; Kirimlioglu, S.H. Telecytology: Is it possible with smartphone images? Diagn. Cytopathol. 2018, 46, 40–46. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, S.M.; Shin, H.; Jang, J.S.; Kim, Y.I.; Han, D.H. A Mobile Game for Patients With Breast Cancer for Chemotherapy Self-Management and Quality-of-Life Improvement: Randomized Controlled Trial. J. Med. Internet. Res. 2018, 20, e273. [Google Scholar] [CrossRef]
- Mooney, K.H.; Beck, S.L.; Wong, B.; Dunson, W.; Wujcik, D.; Whisenant, M.; Donaldson, G. Automated home monitoring and management of patient-reported symptoms during chemotherapy: Results of the symptom care at home RCT. Cancer Med. 2017, 6, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Deal, A.M.; Dueck, A.C.; Scher, H.I.; Kris, M.G.; Hudis, C.; Schrag, D. Overall Survival Results of a Trial Assessing Patient-Reported Outcomes for Symptom Monitoring During Routine Cancer Treatment. JAMA 2017, 318, 197–198. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.; Lethrosne, C.; Pourel, N.; Molinier, O.; Pointreau, Y.; Domont, J.; Bourgeois, H.P.; Senellart, H.; Tremolieres, P.; Lizee, T.; et al. Randomized Trial Comparing a Web-Mediated Follow-up With Routine Surveillance in Lung Cancer Patients. J. Natl. Cancer Inst. 2017, 109, 1–8. [Google Scholar] [CrossRef]
- Protiere, C.; Viens, P.; Genre, D.; Cowen, D.; Camerlo, J.; Gravis, G.; Alzieu, C.; Bertucci, F.; Resbeut, M.; Maraninchi, D.; et al. Patient participation in medical decision-making: A French study in adjuvant radio-chemotherapy for early breast cancer. Ann. Oncol. 2000, 11, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Dow, L.A.; Virago, E.; Khatcheressian, J.; Lyckholm, L.J.; Matsuyama, R. Giving honest information to patients with advanced cancer maintains hope. Oncology (Williston Park) 2010, 24, 521–525. [Google Scholar] [PubMed]
- Cluzeau, T.; Mounier, N. Patients and the Web. Bull. Cancer 2010, 97, 1133–1136. [Google Scholar] [CrossRef] [PubMed]
- Owens, O.L.; Beer, J.M.; Reyes, L.I.; Thomas, T.L. Systematic Review of Commercially Available Mobile Phone Applications for Prostate Cancer Education. Am. J. Mens Health 2018. [Google Scholar] [CrossRef]
- Pandey, A.; Hasan, S.; Dubey, D.; Sarangi, S. Smartphone apps as a source of cancer information: Changing trends in health information-seeking behavior. J. Cancer Educ. 2013, 28, 138–142. [Google Scholar] [CrossRef]
- Rozati, H.; Shah, S.P.; Shah, N. Smartphone Applications for the Clinical Oncologist in UK Practice. J. Cancer Educ. 2015, 30, 367–373. [Google Scholar] [CrossRef]
- Senior, K. Smart phones: New clinical tools in oncology? Lancet Oncol. 2011, 12, 429–430. [Google Scholar] [CrossRef]
- Khan, Z.; Darr, U.; Khan, M.A.; Nawras, M.; Khalil, B.; Abdel-Aziz, Y.; Alastal, Y.; Barnett, W.; Sodeman, T.; Nawras, A. Improving Internal Medicine Residents’ Colorectal Cancer Screening Knowledge Using a Smartphone App: Pilot Study. JMIR Med. Educ. 2018, 4, e10. [Google Scholar] [CrossRef]
- Nioi, A.; Wendelboe-Nelson, C.; Cowan, S.; Cowie, H.; Rashid, S.; Ritchie, P.; Cherrie, M.; Lansdown, T.C.; Cherrie, J.W. A randomised control crossover trial of a theory based intervention to improve sun-safe and healthy behaviours in construction workers: Study protocol. BMC Public Health 2018, 18, 259. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; Sniehotta, F.F.; Birch-Machin, M.A.; Olivier, P.; Araujo-Soares, V. Systematic and Iterative Development of a Smartphone App to Promote Sun-Protection Among Holidaymakers: Design of a Prototype and Results of Usability and Acceptability Testing. JMIR Res. Protoc. 2017, 6, e112. [Google Scholar] [CrossRef]
- Hacker, E.; Horsham, C.; Vagenas, D.; Jones, L.; Lowe, J.; Janda, M. A Mobile Technology Intervention With Ultraviolet Radiation Dosimeters and Smartphone Apps for Skin Cancer Prevention in Young Adults: Randomized Controlled Trial. JMIR Mhealth Uhealth 2018, 6, e199. [Google Scholar] [CrossRef] [PubMed]
- Cupertino, A.P.; Cartujano-Barrera, F.; Perales, J.; Formagini, T.; Rodriguez-Bolanos, R.; Ellerbeck, E.F.; Ponciano-Rodriguez, G.; Reynales-Shigematsu, L.M. “Vive Sin Tabaco... inverted exclamation markDecidete!” Feasibility and Acceptability of an e-Health Smoking Cessation Informed Decision-Making Tool Integrated in Primary Healthcare in Mexico. Telemed. J. E. Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, S.S.; Thind, H.; Liu, B.; Wilson, L.C. Towards research-tested smartphone applications for preventing breast cancer. Mhealth 2016, 2, 26. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.A.; Whitehead, M.S.; Sheats, J.; Mastromonico, J.; Yoo, W.; Coughlin, S.S. A Community-Engaged Approach to Developing a Mobile Cancer Prevention App: The mCPA Study Protocol. JMIR Res. Protoc. 2016, 5, e34. [Google Scholar] [CrossRef]
- Garnett, C.; Crane, D.; Michie, S.; West, R.; Brown, J. Evaluating the effectiveness of a smartphone app to reduce excessive alcohol consumption: Protocol for a factorial randomised control trial. BMC Public Health 2016, 16, 536. [Google Scholar] [CrossRef]
- Uy, C.; Lopez, J.; Trinh-Shevrin, C.; Kwon, S.C.; Sherman, S.E.; Liang, P.S. Text Messaging Interventions on Cancer Screening Rates: A Systematic Review. J. Med. Internet Res. 2017, 19, e296. [Google Scholar] [CrossRef]
- Tran, P.L.; Benski, C.; Viviano, M.; Petignat, P.; Combescure, C.; Jinoro, J.; Herinianasolo, J.L.; Vassilakos, P. Performance of Smartphone-Based Digital Images for Cervical Cancer Screening in a Low-Resource Context. Int. J. Technol. Assess. Health Care 2018, 34, 337–342. [Google Scholar] [CrossRef]
- Linde, D.S.; Andersen, M.S.; Mwaiselage, J.D.; Manongi, R.; Kjaer, S.K.; Rasch, V. Text messages to increase attendance to follow-up cervical cancer screening appointments among HPV-positive Tanzanian women (Connected2Care): Study protocol for a randomised controlled trial. Trials 2017, 18, 555. [Google Scholar] [CrossRef] [PubMed]
- Quercia, K.; Tran, P.L.; Jinoro, J.; Herniainasolo, J.L.; Viviano, M.; Vassilakos, P.; Benski, C.; Petignat, P. A Mobile Health Data Collection System for Remote Areas to Monitor Women Participating in a Cervical Cancer Screening Campaign. Telemed. J. E. Health 2018, 24, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Rohilla, L.; Bagga, R.; Srinivasan, R.; Jindal, H.A.; Sharma, N.; Kankaria, A.; Jamir, L.; Suri, V.; Singh, R.K.; et al. Feasibility of implementing cervical cancer screening program using smartphone imaging as a training aid for nurses in rural India. Public Health Nurs. 2018, 35, 526–533. [Google Scholar] [CrossRef]
- Hitt, W.C.; Low, G.M.; Lynch, C.E.; Gauss, C.H.; Magann, E.F.; Lowery, C.L.; Eswaran, H. Application of a Telecolposcopy Program in Rural Settings. Telemed. J. E. Health 2016, 22, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Brittain, K.; Kamp, K.; Cassandras, C.; Salaysay, Z.; Gomez-Marquez, J. A Mobile App to Increase Informed Decisions About Colorectal Cancer Screening Among African American and Caucasian Women: A Pilot Study. Gastroenterol. Nurs. 2018, 41, 297–303. [Google Scholar] [CrossRef]
- Choi, J.; Cho, Y.; Woo, H. mHealth Approaches in Managing Skin Cancer: Systematic Review of Evidence-Based Research Using Integrative Mapping. JMIR Mhealth Uhealth 2018, 6, e164. [Google Scholar] [CrossRef]
- Chuchu, N.; Takwoingi, Y.; Dinnes, J.; Matin, R.N.; Bassett, O.; Moreau, J.F.; Bayliss, S.E.; Davenport, C.; Godfrey, K.; O’Connell, S.; et al. Smartphone applications for triaging adults with skin lesions that are suspicious for melanoma. Cochrane Database Syst. Rev. 2018, 12, CD013192. [Google Scholar] [CrossRef] [PubMed]
- Maier, T.; Kulichova, D.; Schotten, K.; Astrid, R.; Ruzicka, T.; Berking, C.; Udrea, A. Accuracy of a smartphone application using fractal image analysis of pigmented moles compared to clinical diagnosis and histological result. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 663–667. [Google Scholar] [CrossRef]
- Gou, T.; Hu, J.; Wu, W.; Ding, X.; Zhou, S.; Fang, W.; Mu, Y. Smartphone-based mobile digital PCR device for DNA quantitative analysis with high accuracy. Biosens. Bioelectron. 2018, 120, 144–152. [Google Scholar] [CrossRef]
- Prasad, A.; Hasan, S.M.A.; Grouchy, S.; Gartia, M.R. DNA microarray analysis using a smartphone to detect the BRCA-1 gene. Analyst 2018, 144, 197–205. [Google Scholar] [CrossRef]
- Turner, R.; Madsen, J.; Herrera, P.D.S.; Wallace, J.; Madrigal, J.; Hinestrosa, J.P.; Dobrovolskaia, I.; Krishnan, R. Cancer Detection at your Fingertips: Smartphone-Enabled DNA Testing. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018, 2018, 5418–5421. [Google Scholar] [CrossRef]
- Al-Barrak, J.; Cheung, W.Y. Adherence to imatinib therapy in gastrointestinal stromal tumors and chronic myeloid leukemia. Support. Care Cancer 2013, 21, 2351–2357. [Google Scholar] [CrossRef] [PubMed]
- Despas, F.; Roche, H.; Laurent, G. Anticancer drug adherence. Bull. Cancer 2013, 100, 473–484. [Google Scholar] [CrossRef]
- Ahmed, I.; Ahmad, N.S.; Ali, S.; Ali, S.; George, A.; Saleem Danish, H.; Uppal, E.; Soo, J.; Mobasheri, M.H.; King, D.; et al. Medication Adherence Apps: Review and Content Analysis. JMIR Mhealth Uhealth 2018, 6, e62. [Google Scholar] [CrossRef]
- Becker, S.; Kribben, A.; Meister, S.; Diamantidis, C.J.; Unger, N.; Mitchell, A. User profiles of a smartphone application to support drug adherence—experiences from the iNephro project. PLoS ONE 2013, 8, e78547. [Google Scholar] [CrossRef] [PubMed]
- Spoelstra, S.L.; Given, C.W.; Sikorskii, A.; Coursaris, C.K.; Majumder, A.; DeKoekkoek, T.; Schueller, M.; Given, B.A. Proof of Concept of a Mobile Health Short Message Service Text Message Intervention That Promotes Adherence to Oral Anticancer Agent Medications: A Randomized Controlled Trial. Telemed. J. E. Health 2016, 22, 497–506. [Google Scholar] [CrossRef]
- Chalela, P.; Munoz, E.; Inupakutika, D.; Kaghyan, S.; Akopian, D.; Kaklamani, V.; Lathrop, K.; Ramirez, A. Improving adherence to endocrine hormonal therapy among breast cancer patients: Study protocol for a randomized controlled trial. Contemp. Clin. Trials Commun. 2018, 12, 109–115. [Google Scholar] [CrossRef]
- Meguerditchian, A.; Tamblyn, R.; Meterissian, S.; Law, S.; Prchal, J.; Winslade, N.; Stern, D. Adjuvant Endocrine Therapy in Breast Cancer: A Novel e-Health Approach in Optimizing Treatment for Seniors (OPTIMUM): A Two-Group Controlled Comparison Pilot Study. JMIR Res. Protoc. 2016, 5, e199. [Google Scholar] [CrossRef]
- Homsi, J.; Walsh, D.; Rivera, N.; Rybicki, L.A.; Nelson, K.A.; Legrand, S.B.; Davis, M.; Naughton, M.; Gvozdjan, D.; Pham, H. Symptom evaluation in palliative medicine: Patient report vs systematic assessment. Support. Care Cancer 2006, 14, 444–453. [Google Scholar] [CrossRef]
- Coolbrandt, A.; Van den Heede, K.; Vanhove, E.; De Bom, A.; Milisen, K.; Wildiers, H. Immediate versus delayed self-reporting of symptoms and side effects during chemotherapy: Does timing matter? Eur. J. Oncol. Nurs. 2011, 15, 130–136. [Google Scholar] [CrossRef]
- Weaver, A.; Young, A.M.; Rowntree, J.; Townsend, N.; Pearson, S.; Smith, J.; Gibson, O.; Cobern, W.; Larsen, M.; Tarassenko, L. Application of mobile phone technology for managing chemotherapy-associated side-effects. Ann. Oncol. 2007, 18, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- McCann, L.; Maguire, R.; Miller, M.; Kearney, N. Patients’ perceptions and experiences of using a mobile phone-based advanced symptom management system (ASyMS) to monitor and manage chemotherapy related toxicity. Eur. J. Cancer Care (Engl.) 2009, 18, 156–164. [Google Scholar] [CrossRef]
- Kotronoulas, G.; Kearney, N.; Maguire, R.; Harrow, A.; Di Domenico, D.; Croy, S.; MacGillivray, S. What is the value of the routine use of patient-reported outcome measures toward improvement of patient outcomes, processes of care, and health service outcomes in cancer care? A systematic review of controlled trials. J. Clin. Oncol. 2014, 32, 1480–1501. [Google Scholar] [CrossRef] [PubMed]
- Kluetz, P.G.; Chingos, D.T.; Basch, E.M.; Mitchell, S.A. Patient-Reported Outcomes in Cancer Clinical Trials: Measuring Symptomatic Adverse Events With the National Cancer Institute’s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Am. Soc. Clin. Oncol. Educ. Book 2016, 35, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Borosund, E.; Cvancarova, M.; Moore, S.M.; Ekstedt, M.; Ruland, C.M. Comparing effects in regular practice of e-communication and Web-based self-management support among breast cancer patients: Preliminary results from a randomized controlled trial. J. Med. Internet Res. 2014, 16, e295. [Google Scholar] [CrossRef] [PubMed]
- Graetz, I.; Anderson, J.N.; McKillop, C.N.; Stepanski, E.J.; Paladino, A.J.; Tillmanns, T.D. Use of a web-based app to improve postoperative outcomes for patients receiving gynecological oncology care: A randomized controlled feasibility trial. Gynecol. Oncol. 2018, 150, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Okera, M.; Baker, N.A.; Hayward, A.M.; Selva-Nayagam, S. Oncology workforce issues: The challenge of the outpatient clinic. Intern. Med. J. 2011, 41, 499–503. [Google Scholar] [CrossRef]
- Green, J.; Murchie, P.; Lee, A.J. Does patients’ place of residence affect the type of physician performing primary excision of cutaneous melanoma in northern Scotland? J. Rural Health 2013, 29, S35–S42. [Google Scholar] [CrossRef]
- Hall, S.J.; Samuel, L.M.; Murchie, P. Toward shared care for people with cancer: Developing the model with patients and GPs. Fam. Pract. 2011, 28, 554–564. [Google Scholar] [CrossRef]
- Armfield, N.R.; Bradford, M.; Bradford, N.K. The clinical use of Skype--For which patients, with which problems and in which settings? A snapshot review of the literature. Int. J. Med. Inform. 2015, 84, 737–742. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Vijayaraghavan, S.; Wherton, J.; Shaw, S.; Byrne, E.; Campbell-Richards, D.; Bhattacharya, S.; Hanson, P.; Ramoutar, S.; Gutteridge, C.; et al. Virtual online consultations: Advantages and limitations (VOCAL) study. BMJ Open 2016, 6, e009388. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, R.; Hall, S.; Sinclair, J.E.; Bond, C.; Murchie, P. Using technology to deliver cancer follow-up: A systematic review. BMC Cancer 2014, 14, 311. [Google Scholar] [CrossRef] [PubMed]
- Kearney, N.; McCann, L.; Norrie, J.; Taylor, L.; Gray, P.; McGee-Lennon, M.; Sage, M.; Miller, M.; Maguire, R. Evaluation of a mobile phone-based, advanced symptom management system (ASyMS) in the management of chemotherapy-related toxicity. Support. Care Cancer 2009, 17, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Matthew, A.G.; Currie, K.L.; Irvine, J.; Ritvo, P.; Santa Mina, D.; Jamnicky, L.; Nam, R.; Trachtenberg, J. Serial personal digital assistant data capture of health-related quality of life: A randomized controlled trial in a prostate cancer clinic. Health Qual. Life Outcomes 2007, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.; Viger, L.; Charron, A.; Voog, E.; Dupuis, O.; Pointreau, Y.; Letellier, C. Detection of lung cancer relapse using self-reported symptoms transmitted via an internet web-application: Pilot study of the sentinel follow-up. Support. Care Cancer 2014, 22, 1467–1473. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.; Viger, L.; Charron, A.; Voog, E.; Letellier, C. Detecting lung cancer relapse using self-evaluation forms weekly filled at home: The sentinel follow-up. Support. Care Cancer 2014, 22, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.; Yossi, S.; Septans, A.L.; Charron, A.; Voog, E.; Dupuis, O.; Ganem, G.; Pointreau, Y.; Letellier, C. Improving Survival in Patients Treated for a Lung Cancer Using Self-Evaluated Symptoms Reported Through a Web Application. Am. J. Clin. Oncol. 2015, 40, 464–469. [Google Scholar] [CrossRef]
- Villani, D.; Cognetta, C.; Repetto, C.; Serino, S.; Toniolo, D.; Scanzi, F.; Riva, G. Promoting Emotional Well-Being in Older Breast Cancer Patients: Results From an eHealth Intervention. Front. Psychol. 2018, 9, 2279. [Google Scholar] [CrossRef]
- Akechi, T.; Yamaguchi, T.; Uchida, M.; Imai, F.; Momino, K.; Katsuki, F.; Sakurai, N.; Miyaji, T.; Horikoshi, M.; Furukawa, T.A.; et al. Smartphone problem-solving and behavioural activation therapy to reduce fear of recurrence among patients with breast cancer (SMartphone Intervention to LEssen fear of cancer recurrence: SMILE project): Protocol for a randomised controlled trial. BMJ Open 2018, 8, e024794. [Google Scholar] [CrossRef]
- Douma, J.A.J.; Verheul, H.M.W.; Buffart, L.M. Feasibility, validity and reliability of objective smartphone measurements of physical activity and fitness in patients with cancer. BMC Cancer 2018, 18, 1052. [Google Scholar] [CrossRef]
- Geng, Z.; Wu, F.; Zhang, Y.; Wang, J.; Wang, J.; Liu, H.; Yuan, C. Mobile Physical Activity Intervention for Breast Cancer Patients During Chemotherapy. Stud. Health Technol. Inform. 2018, 250, 236. [Google Scholar] [PubMed]
- Lee, H.; Uhm, K.E.; Cheong, I.Y.; Yoo, J.S.; Chung, S.H.; Park, Y.H.; Lee, J.Y.; Hwang, J.H. Patient Satisfaction with Mobile Health (mHealth) Application for Exercise Intervention in Breast Cancer Survivors. J. Med. Syst. 2018, 42, 254. [Google Scholar] [CrossRef] [PubMed]
- Cheong, I.Y.; An, S.Y.; Cha, W.C.; Rha, M.Y.; Kim, S.T.; Chang, D.K.; Hwang, J.H. Efficacy of Mobile Health Care Application and Wearable Device in Improvement of Physical Performance in Colorectal Cancer Patients Undergoing Chemotherapy. Clin. Colorectal Cancer 2018, 17, e353–e362. [Google Scholar] [CrossRef] [PubMed]
- Ormel, H.L.; van der Schoot, G.G.F.; Westerink, N.L.; Sluiter, W.J.; Gietema, J.A.; Walenkamp, A.M.E. Self-monitoring physical activity with a smartphone application in cancer patients: A randomized feasibility study (SMART-trial). Support. Care Cancer 2018, 26, 3915–3923. [Google Scholar] [CrossRef] [PubMed]
- Orlemann, T.; Reljic, D.; Zenker, B.; Meyer, J.; Eskofier, B.; Thiemt, J.; Herrmann, H.J.; Neurath, M.F.; Zopf, Y. A Novel Mobile Phone App (OncoFood) to Record and Optimize the Dietary Behavior of Oncologic Patients: Pilot Study. JMIR Cancer 2018, 4, e10703. [Google Scholar] [CrossRef] [PubMed]
- Lerebours, F.; Saltel, P.; Bethune-Volters, A.; Nallet, G.; Bourdat, P.; Vesin-Etterlen, F.; Zernik, N.; Flinois, A. Satisfaction of Breast Cancer patients treated by chemotherapy outpatient clinics: Results of the TemporELLES survey. Bull. Cancer 2015, 102, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Corrie, P.G.; Moody, A.M.; Armstrong, G.; Nolasco, S.; Lao-Sirieix, S.H.; Bavister, L.; Prevost, A.T.; Parker, R.; Sabes-Figuera, R.; McCrone, P.; et al. Is community treatment best? A randomised trial comparing delivery of cancer treatment in the hospital, home and GP surgery. Br. J. Cancer 2013, 109, 1549–1555. [Google Scholar] [CrossRef]
- Sabesan, S. Medical models of teleoncology: Current status and future directions. Asia Pac. J. Clin. Oncol. 2014, 10, 200–204. [Google Scholar] [CrossRef]
- Bohme, C.; von Osthoff, M.B.; Frey, K.; Hubner, J. Qualitative evaluation of mobile cancer apps with particular attention to the target group, content, and advertising. J. Cancer Res. Clin. Oncol. 2018, 144, 173–181. [Google Scholar] [CrossRef]
- McKay, F.H.; Cheng, C.; Wright, A.; Shill, J.; Stephens, H.; Uccellini, M. Evaluating mobile phone applications for health behaviour change: A systematic review. J. Telemed. Telecare 2018, 24, 22–30. [Google Scholar] [CrossRef]
- Mobasheri, M.H.; Johnston, M.; King, D.; Leff, D.; Thiruchelvam, P.; Darzi, A. Smartphone breast applications—What’s the evidence? Breast 2014, 23, 683–689. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, S.; Brady, R.R. Colorectal smartphone apps: Opportunities and risks. Colorectal Dis. 2012, 14, e530–e534. [Google Scholar] [CrossRef] [PubMed]
- Giunti, G.; Giunta, D.H.; Guisado-Fernandez, E.; Bender, J.L.; Fernandez-Luque, L. A biopsy of Breast Cancer mobile applications: State of the practice review. Int. J. Med. Inform. 2018, 110, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Abbott, L.M.; Magnusson, R.S.; Gibbs, E.; Smith, S.D. Smartphone use in dermatology for clinical photography and consultation: Current practice and the law. Australas. J. Dermatol. 2018, 59, 101–107. [Google Scholar] [CrossRef]
- van Gurp, J.; van Selm, M.; Vissers, K.; van Leeuwen, E.; Hasselaar, J. How outpatient palliative care teleconsultation facilitates empathic patient-professional relationships: A qualitative study. PLoS ONE 2015, 10, e0124387. [Google Scholar] [CrossRef]
- Chesser, A.; Burke, A.; Reyes, J.; Rohrberg, T. Navigating the digital divide: A systematic review of eHealth literacy in underserved populations in the United States. Inform. Health Soc. Care 2016, 41, 1–19. [Google Scholar] [CrossRef]
- Salovey, P.; Williams-Piehota, P.; Mowad, L.; Moret, M.E.; Edlund, D.; Andersen, J. Bridging the digital divide by increasing computer and cancer literacy: Community technology centers for head-start parents and families. J. Health Commun. 2009, 14, 228–245. [Google Scholar] [CrossRef]
- Pathmanathan, S.; Burgher, B.; Sabesan, S. Is intensive chemotherapy safe for rural cancer patients? Intern. Med. J. 2013, 43, 643–649. [Google Scholar] [CrossRef]
- Marzorati, C.; Renzi, C.; Russell-Edu, S.W.; Pravettoni, G. Telemedicine Use Among Caregivers of Cancer Patients: Systematic Review. J. Med. Internet Res. 2018, 20, e223. [Google Scholar] [CrossRef]
- Shaffer, K.M.; Kim, Y.; Carver, C.S. Physical and mental health trajectories of cancer patients and caregivers across the year post-diagnosis: A dyadic investigation. Psychol. Health 2016, 31, 655–674. [Google Scholar] [CrossRef]
- Slev, V.N.; Mistiaen, P.; Pasman, H.R.; Verdonck-de Leeuw, I.M.; van Uden-Kraan, C.F.; Francke, A.L. Effects of eHealth for patients and informal caregivers confronted with cancer: A meta-review. Int. J. Med. Inform. 2016, 87, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, L.; Buthion, V.; Vidal-Trecan, G.; Briot, P. Impact of the healthcare payment system on patient access to oral anticancer drugs: An illustration from the French and United States contexts. BMC Health Serv. Res. 2014, 14, 274. [Google Scholar] [CrossRef]
- French National Study of Costs. 2012. Available online: http://www.atih.sante.fr/couts-de-prise-en-charge-l-hopital-en-2012-en-mco (accessed on 30 January 2019).
- Coriat, R.; Boudou-Rouquette, P.; Durand, J.P.; Forgeot d’Arc, P.; Martin, I.; Mir, O.; Ropert, S.; Alexandre, J.; Goldwasser, F. Cost effectiveness of integrated medicine in patients with cancer receiving anticancer chemotherapy. J. Oncol. Pract. 2012, 8, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Michaud, T.L.; Zhou, J.; McCarthy, M.A.; Siahpush, M.; Su, D. Costs of Home-Based Telemedicine Programs: A Systematic Review. Int. J. Technol. Assess. Health Care 2018, 34, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Doolittle, G.C.; Williams, A.R.; Spaulding, A.; Spaulding, R.J.; Cook, D.J. A cost analysis of a tele-oncology practice in the United States. J. Telemed. Telecare 2004, 10 (Suppl. 1), 27–29. [Google Scholar] [CrossRef] [PubMed]
- Thaker, D.A.; Monypenny, R.; Olver, I.; Sabesan, S. Cost savings from a telemedicine model of care in northern Queensland, Australia. Med. J. Aust. 2013, 199, 414–417. [Google Scholar] [CrossRef] [PubMed]
| Field | Study Type | Cancer or Subjects Type | Number of Subjects | E-Health Tools Involved | Main Results of the Experimental vs. Control Arms | Reference |
|---|---|---|---|---|---|---|
| Information Access | Randomized clinical trial, 4 arms (web applications vs. text messages vs. both vs. standard care) | Cutaneous cancer before Mohs micrographic surgery | 90 | Web applications and text messages for patient education | Reduction of patients’ preoperative anxiety | [25] |
| Educating Health care professionals | Controlled clinical trial with concealed allocation, 2 arms (educational materials consistent with subjects’ preferences for learning vs. materials typical of the My Cancer Genome website) | Oncology health care professionals | 751 | Web-based | Improved learning with tailored, web-based learning style educational material | [26] |
| Prevention | Randomized clinical trial, 2 arms (mobile application vs. control) | Adults from the Knowledge Panel, 18 years or older who owned an Android smartphone | 604 | Mobile application providing personalized, real-time sun protection advice | Improved sun protection | [27] |
| Prevention | Randomized clinical trial, 2 arms (mobile application vs. control) | UK smokers willing to attempt quitting | 5800 | Periodic, motivational text messages on smartphones | Higher 6-month rate of biochemically-verified tobacco cessation | [28] |
| Diagnosis | Prospective development of a non-invasive anemia screening tool | Patients with anemia of different etiologies and healthy subjects | 337 | Smartphone application and photos | Detection of anemia with an accuracy of ±2.4 g/dL (0.92 after personalized calibration) and a sensitivity of 97% when compared with blood count hemoglobin levels | [29] |
| Diagnosis | Retrospective assessment of smartphone usage in telecytology | Different cytological materials | 172 | Smartphone photos transferred via WhatsApp® | High intraobserver Kappa agreement between microscopic diagnoses and smartphone image diagnoses; change in patient management in 11.4% of cases | [30] |
| Treatment observance and tolerance | Randomized clinical trial, 2 arms (mobile games vs. standard care) | Patients with metastatic breast cancer planning to receive chemotherapy | 76 | Smartphone-based mobile games | Better patient education, improved drug compliance, decreased side effects, and better quality of life | [31] |
| Treatment tolerance | Randomized clinical trial, 2 arms (automated home monitoring and follow-up vs. standard care) | Patients beginning chemotherapy | 358 | Symptom Care at Home (SCH) intervention | Reduction of clinical symptoms | [32] |
| Treatment tolerance | Randomized clinical trial, 2 arms (Patient-Reported Outcomes (PROs)-based symptom monitoring vs. standard care) | Patients receiving outpatient chemotherapy for advanced solid tumors | 766 | PRO tablet computers | Improvements in health-related quality of life at 6 months, fewer admissions to hospitals or emergency rooms, better overall and quality-adjusted survivals | [33] |
| Follow-up and survival | Randomized clinical trial, 2 arms (e-FAP-based follow-up vs. standard follow-up) | Patients with stage III/IV lung cancer | 121 | E-follow-up application (e-FAP) | Improved overall survival (median and 1-year overall survival); similar relapse rates, but better performance status at initial relapse, and better quality of life | [34] |
| Strengths | Patients - More “actors in healthcare” for oral treatments - Downplay dramatization of diseases and treatments - Improved comfort/quality of life at home and work - Decreased time spent in transportation and “scary” waiting rooms - Greater autonomy in managing appointments - Greater equality in caregiver relationships - Broader and more rapid access to: medical files, second opinions, and disease and treatment information - Sharing of disease and treatment-related experiences (social networks, and forums) - Equal access to care Oncologists and Hospitals - Decision-making support (diagnosis, and treatment) - Information exchange between a city and rural medical centers - Optimization of medical resources: improved time management |
| Weaknesses | - Novel and complex organization - Lack of coordination between healthcare professionals - Insufficient training of non-hospital personnel (doctors, pharmacists, nurses, etc.) with no current method of reimbursement - Insufficient digital training - “Bringing cancer back to home or work”, which could place further strain on familial and professional relationships (loss of confidence and trust) - Care for unsupported companions |
| Opportunities | - Health care cost reduction - Increased cooperation between health care facilities (hospitals, cancer centers, rural health care providers, etc.) - Creation of new health care professions (coordination, and follow-up: nurse navigators) - Digital market |
| Threats | - Patient–caregiver estrangement: feelings of loneliness and anxiety concerning the diseases and treatment toxicity - Virtual “less human” relationships - Trivialization of the burden of care - Poor grasp of the risks involved - Failure to comply with oral treatments - Digital divide (elderly, poorly educated, and foreigners) - Overbooking doctors (burnout) - Elimination of certain hospital functions |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertucci, F.; Le Corroller-Soriano, A.-G.; Monneur-Miramon, A.; Moulin, J.-F.; Fluzin, S.; Maraninchi, D.; Gonçalves, A. Outpatient Cancer Care Delivery in the Context of E-Oncology: A French Perspective on “Cancer outside the Hospital Walls”. Cancers 2019, 11, 219. https://doi.org/10.3390/cancers11020219
Bertucci F, Le Corroller-Soriano A-G, Monneur-Miramon A, Moulin J-F, Fluzin S, Maraninchi D, Gonçalves A. Outpatient Cancer Care Delivery in the Context of E-Oncology: A French Perspective on “Cancer outside the Hospital Walls”. Cancers. 2019; 11(2):219. https://doi.org/10.3390/cancers11020219
Chicago/Turabian StyleBertucci, François, Anne-Gaëlle Le Corroller-Soriano, Audrey Monneur-Miramon, Jean-François Moulin, Sylvain Fluzin, Dominique Maraninchi, and Anthony Gonçalves. 2019. "Outpatient Cancer Care Delivery in the Context of E-Oncology: A French Perspective on “Cancer outside the Hospital Walls”" Cancers 11, no. 2: 219. https://doi.org/10.3390/cancers11020219
APA StyleBertucci, F., Le Corroller-Soriano, A.-G., Monneur-Miramon, A., Moulin, J.-F., Fluzin, S., Maraninchi, D., & Gonçalves, A. (2019). Outpatient Cancer Care Delivery in the Context of E-Oncology: A French Perspective on “Cancer outside the Hospital Walls”. Cancers, 11(2), 219. https://doi.org/10.3390/cancers11020219






