Post-Surgery Circulating Tumor Cells and AXL Overexpression as New Poor Prognostic Biomarkers in Resected Lung Adenocarcinoma
Abstract
1. Background
2. Patients and Methods
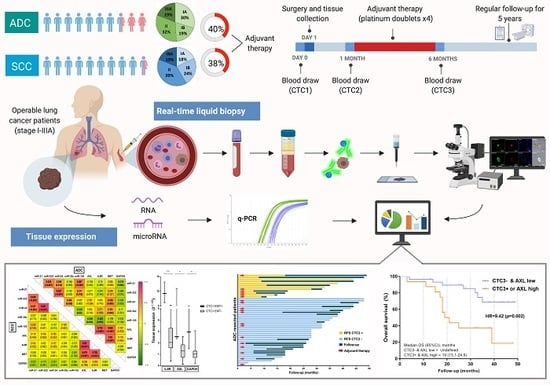
Study Design and Patients
3. Materials and Methods
3.1. Isolation and Characterization of CTCs
3.2. Tissues Total RNA Extraction and cDNA Synthesis
3.3. Quantitative (Real Time) Polymerase Chain Reaction for mRNA and miRNA Expression
3.4. Statistical Analyses
4. Results
4.1. Isolation and Characterization of CTCs
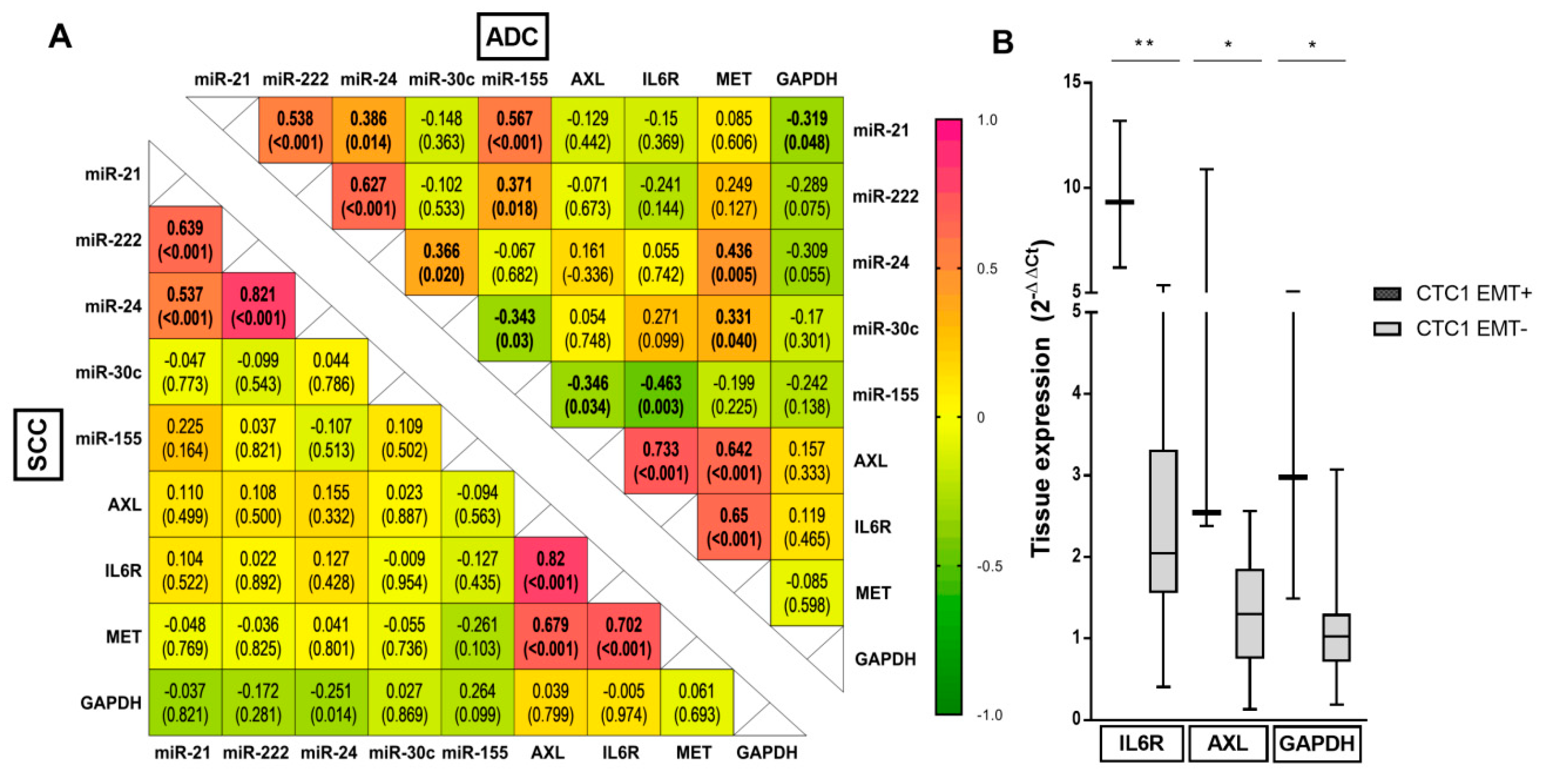
4.2. Genetic and miRNA Tissue Profiling and CTC Subpopulations
4.3. Prognostic Markers of Relapse-Free Survival
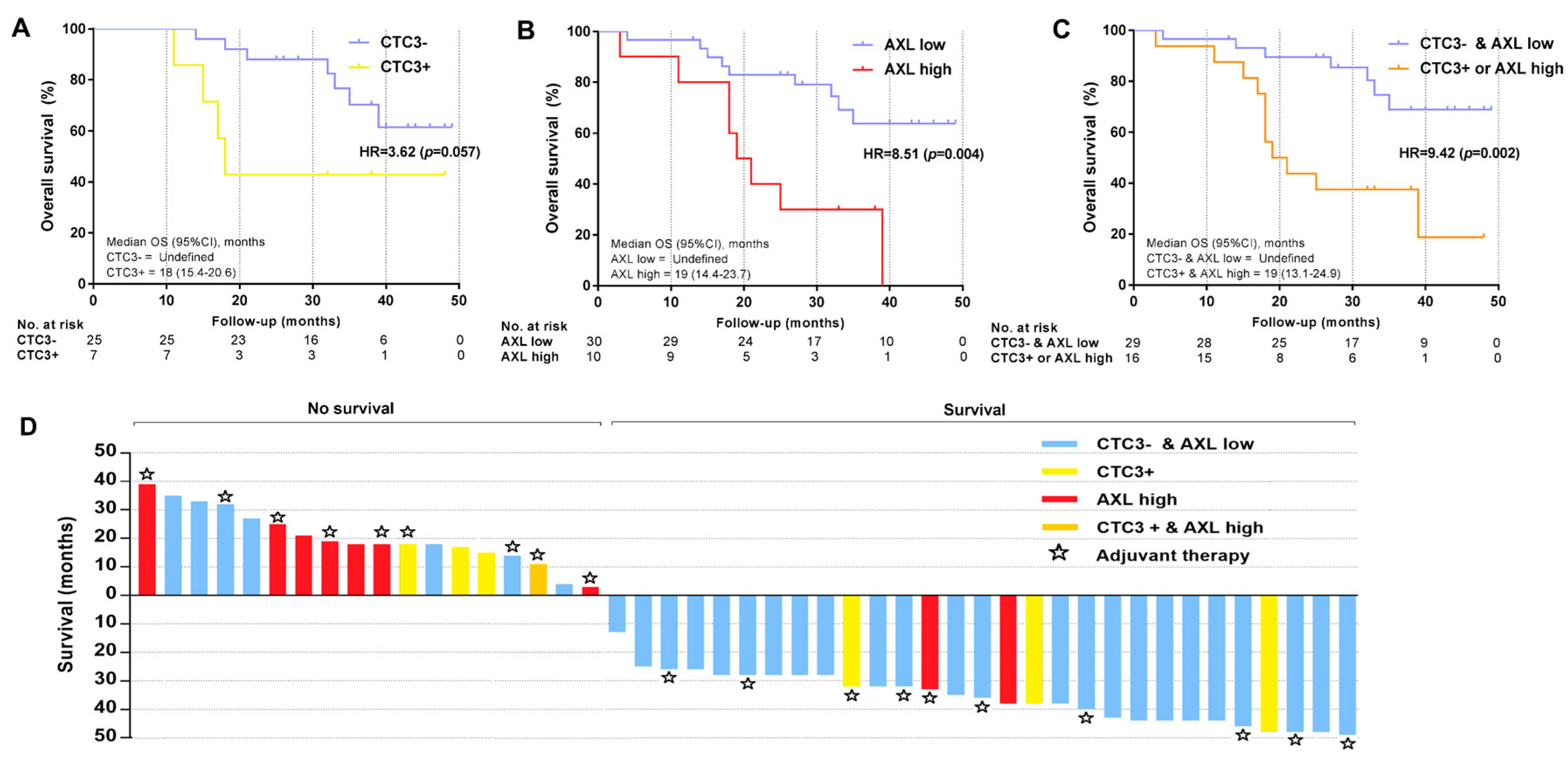
4.4. Prognostic Markers of Overall Survival: Elucidating the Role of AXL
5. Discussion
6. Conclusions
7. Limitations of the Study
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Burdett, S.; Pignon, J.P.; Tierney, J.; Tribodet, H.; Stewart, L.; Le Pechoux, C.; Aupérin, A.; Le Chevalier, T.; Stephens, R.J.; Arriagada, R.; et al. Adjuvant chemotherapy for resected early-stage non-small cell lung cancer. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Novello, S.; Asamura, H.; Bazan, J.; Carbone, D.; Goldstraw, P.; Grunenwald, D.; Ricardi, U.; Vansteenkiste, J. Early Stage Lung Cancer: Progress in the Last 40 Years. J. Thorac. Oncol. 2014, 9, 1434–1442. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.D.; Alexandrov, A.; Kim, J.; Wala, J.; Berger, A.H.; Pedamallu, C.S.; Shukla, S.A.; Guo, G.; Brooks, A.N.; Murray, B.A.; et al. Distinct patterns of somatic genome alterations in lung adenocarcinomas and squamous cell carcinomas. Nat. Genet. 2016, 48, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Jamal-Hanjani, M.; Wilson, G.A.; McGranahan, N.; Birkbak, N.J.; Watkins, T.B.K.; Veeriah, S.; Shafi, S.; Johnson, D.H.; Mitter, R.; Rosenthal, R.; et al. Tracking the Evolution of Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 376, 2109–2121. [Google Scholar] [CrossRef] [PubMed]
- Kawase, A.; Yoshida, J.; Ishii, G.; Nakao, M.; Aokage, K.; Hishida, T.; Nishimura, M.; Nagai, K. Differences between squamous cell carcinoma and adenocarcinoma of the lung: Are adenocarcinoma and squamous cell carcinoma prognostically equal? Jpn. J. Clin. Oncol. 2012, 42, 189–195. [Google Scholar] [CrossRef]
- Devarakonda, S.; Rotolo, F.; Tsao, M.-S.; Lanc, I.; Brambilla, E.; Masood, A.; Olaussen, K.A.; Fulton, R.; Sakashita, S.; McLeer-Florin, A.; et al. Tumor Mutation Burden as a Biomarker in Resected Non–Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 2995–3006. [Google Scholar] [CrossRef]
- Nadal, R.; Lorente, J.A.; Rosell, R.; Serrano, M.J. Relevance of molecular characterization of circulating tumor cells in breast cancer in the era of targeted therapies. Expert Rev. Mol. Diagn. 2013, 13, 295–307. [Google Scholar] [CrossRef]
- Barradas, A.M.C.; Terstappen, L.W.M.M. Towards the Biological Understanding of CTC: Capture Technologies, Definitions and Potential to Create Metastasis. Cancers (Basel) 2013, 5, 1619–1642. [Google Scholar] [CrossRef]
- Hofman, V.; Ilie, M.I.; Long, E.; Selva, E.; Bonnetaud, C.; Molina, T.; Vénissac, N.; Mouroux, J.; Vielh, P.; Hofman, P. Detection of circulating tumor cells as a prognostic factor in patients undergoing radical surgery for non-small-cell lung carcinoma: Comparison of the efficacy of the CellSearch AssayTM and the isolation by size of epithelial tumor cell method. Int. J. Cancer 2011, 129, 1651–1660. [Google Scholar] [CrossRef]
- Bayarri-Lara, C.; Ortega, F.G.; Cueto Ladrón de Guevara, A.; Puche, J.L.; Ruiz Zafra, J.; de Miguel-Pérez, D.; Ramos, A.S.-P.; Giraldo-Ospina, C.F.; Navajas Gómez, J.A.; Delgado-Rodriguez, M.; et al. Circulating Tumor Cells Identify Early Recurrence in Patients with Non-Small Cell Lung Cancer Undergoing Radical Resection. PLoS ONE 2016, 11, e0148659. [Google Scholar] [CrossRef]
- Bayarri-Lara, C.I.; De Miguel Pérez, D.; Cueto, A.; On De Guevara, L.; Rodriguez Fern Andez, D.A.; Puche, J.L.; Ramos, A.S.A.-P.; Zafra, J.R.; Giraldo Ospina, C.F.; Delgado-Rodr Iguez, M.; et al. Association of circulating tumour cells with early relapse and 18 F-fluorodeoxyglucose positron emission tomography uptake in resected non-small-cell lung cancers. Cardiothorac. Surg. 2017, 52, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, M.; Ise, N.; Omi, K.; Goishi, K.; Higashiyama, S. Cisplatin influences acquisition of resistance to molecular-targeted agents through epithelial-mesenchymal transition-like changes. Cancer Sci. 2013, 104, 904–911. [Google Scholar] [CrossRef] [PubMed]
- Satelli, A.; Mitra, A.; Brownlee, Z.; Xia, X.; Bellister, S.; Overman, M.J.; Kopetz, S.; Ellis, L.M.; Meng, Q.H.; Li, S. Epithelial-Mesenchymal Transitioned Circulating Tumor Cells Capture for Detecting Tumor Progression. Clin. Cancer Res. 2015, 21, 899–906. [Google Scholar] [CrossRef]
- Gurtan, A.M.; Sharp, P.A. The Role of miRNAs in Regulating Gene Expression Networks. J. Mol. Biol. 2013, 425, 3582–3600. [Google Scholar] [CrossRef]
- Wu, K.-L.; Tsai, Y.-M.; Lien, C.-T.; Kuo, P.-L.; Hung, J.-Y.; Wu, K.-L.; Tsai, Y.-M.; Lien, C.-T.; Kuo, P.-L.; Hung, J.-Y. The Roles of MicroRNA in Lung Cancer. Int. J. Mol. Sci. 2019, 20, 1611. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, H.; Nishida, N.; Calin, G.A.; Pantel, K. Clinical relevance of circulating cell-free microRNAs in cancer. Nat. Rev. Clin. Oncol. 2014, 11, 145–156. [Google Scholar] [CrossRef]
- Goldstraw, P.; Crowley, J.; Chansky, K.; Giroux, D.J.; Groome, P.A.; Rami-Porta, R.; Postmus, P.E.; Rusch, V.; Sobin, L.; International Association for the Study of Lung Cancer International Staging Committee; et al. The IASLC Lung Cancer Staging Project: Proposals for the Revision of the TNM Stage Groupings in the Forthcoming (Seventh) Edition of the TNM Classification of Malignant Tumours. J. Thorac. Oncol. 2007, 2, 706–714. [Google Scholar]
- Kris, M.G.; Gaspar, L.E.; Chaft, J.E.; Kennedy, E.B.; Azzoli, C.G.; Ellis, P.M.; Lin, S.H.; Pass, H.I.; Seth, R.; Shepherd, F.A.; et al. Adjuvant Systemic Therapy and Adjuvant Radiation Therapy for Stage I to IIIA Completely Resected Non–Small-Cell Lung Cancers: American Society of Clinical Oncology/Cancer Care Ontario Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 2960–2974. [Google Scholar] [CrossRef] [PubMed]
- Antony, J.; Huang, R.Y.-J. AXL-Driven EMT State as a Targetable Conduit in Cancer. Cancer Res. 2017, 77, 3725–3732. [Google Scholar] [CrossRef] [PubMed]
- Puzone, R.; Savarino, G.; Salvi, S.; Bello, M.G.D.; Barletta, G.; Genova, C.; Rijavec, E.; Sini, C.; Esposito, A.I.; Ratto, G.B.; et al. Glyceraldehyde-3-phosphate dehydrogenase gene over expression correlates with poor prognosis in non small cell lung cancer patients. Mol. Cancer 2013, 12, 97. [Google Scholar] [CrossRef] [PubMed]
- Van der Wekken, A.J.; Saber, A.; Hiltermann, T.J.N.; Kok, K.; van den Berg, A.; Groen, H.J.M. Resistance mechanisms after tyrosine kinase inhibitors afatinib and crizotinib in non-small cell lung cancer, a review of the literature. Crit. Rev. Oncol. Hematol. 2016, 100, 107–116. [Google Scholar] [CrossRef]
- Ishikawa, M.; Sonobe, M.; Nakayama, E.; Kobayashi, M.; Kikuchi, R.; Kitamura, J.; Imamura, N.; Date, H. Higher Expression of Receptor Tyrosine Kinase Axl, and Differential Expression of its Ligand, Gas6, Predict Poor Survival in Lung Adenocarcinoma Patients. Ann. Surg. Oncol. 2013, 20, 467–476. [Google Scholar] [CrossRef]
- Fortunato, O.; Boeri, M.; Verri, C.; Moro, M.; Sozzi, G. Therapeutic use of microRNAs in lung cancer. BioMed Res. Int. 2014, 2014, 756975. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gao, Y.; Zhang, K.; Li, C.; Pan, Y.; Chen, J.; Wang, R.; Chen, L. MicroRNAs as Regulators of Cisplatin Resistance in Lung Cancer. Cell. Physiol. Biochem. 2015, 37, 1869–1880. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.L.; Jensen, J.L.; Ørntoft, T.F. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004, 64, 5245–5250. [Google Scholar] [CrossRef]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, research0034.1. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C (T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Budczies, J.; Klauschen, F.; Sinn, B.V.; Győrffy, B.; Schmitt, W.D. Cutoff Finder: A Comprehensive and Straightforward Web Application Enabling Rapid Biomarker Cutoff Optimization. PLoS ONE 2012, 7, e51862. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, G.; Greenland, S. Simulation study of confounder-selection strategies. Am. J. Epidemiol. 1993, 138, 923–936. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Tanaka, T.; Kishimoto, T. Therapeutic uses of anti-interleukin-6 receptor antibody. Int. Immunol. 2015, 27, 21–29. [Google Scholar] [CrossRef]
- Liu, R.-Y.; Zeng, Y.; Lei, Z.; Wang, L.; Yang, H.; Liu, Z.; Zhao, J.; Zhang, H.-T. JAK/STAT3 signaling is required for TGF-β-induced epithelial-mesenchymal transition in lung cancer cells. Int. J. Oncol. 2014, 44, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Tang, Z.; Huang, A.; Chen, P.; Liu, P.; Yang, J.; Lu, W.; Liao, J.; Sun, Y.; Wen, S.; et al. Glyceraldehyde-3-phosphate dehydrogenase promotes cancer growth and metastasis through upregulation of SNAIL expression. Int. J. Oncol. 2017, 50, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Murlidhar, V.; Reddy, R.M.; Fouladdel, S.; Zhao, L.; Ishikawa, M.K.; Grabauskiene, S.; Zhang, Z.; Lin, J.; Chang, A.C.; Carrott, P.; et al. Poor Prognosis Indicated by Venous Circulating Tumor Cell Clusters in Early-Stage Lung Cancers. Cancer Res. 2017, 77, 5194–5206. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Wang, W. Clinical significance of circulating tumor cells in squamous cell lung cancer patients. Cancer Biomark. 2017, 18, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Gao, X.; Cheng, K.; Yu, L. Correlation between the presence of circulating tumor cells and the pathologic type and staging of non-small cell lung cancer during the early postoperative period. Oncol. Lett. 2017, 14, 5825–5830. [Google Scholar] [CrossRef]
- Manjunath, Y.; Upparahalli, S.V.; Avella, D.M.; Deroche, C.B.; Kimchi, E.T.; Staveley-O’Carroll, K.F.; Smith, C.J.; Li, G.; Kaifi, J.T. PD-L1 Expression with Epithelial Mesenchymal Transition of Circulating Tumor Cells Is Associated with Poor Survival in Curatively Resected Non-Small Cell Lung Cancer. Cancers (Basel) 2019, 11, 806. [Google Scholar] [CrossRef]
- Tsukita, Y.; Fujino, N.; Miyauchi, E.; Saito, R.; Fujishima, F.; Itakura, K.; Kyogoku, Y.; Okutomo, K.; Yamada, M.; Okazaki, T.; et al. Axl kinase drives immune checkpoint and chemokine signalling pathways in lung adenocarcinomas. Mol. Cancer 2019, 18, 24. [Google Scholar] [CrossRef]
- Raimondi, C.; Carpino, G.; Nicolazzo, C.; Gradilone, A.; Gianni, W.; Gelibter, A.; Gaudio, E.; Cortesi, E.; Gazzaniga, P. PD-L1 and epithelial-mesenchymal transition in circulating tumor cells from non-small cell lung cancer patients: A molecular shield to evade immune system? Oncoimmunology 2017, 6, e1315488. [Google Scholar] [CrossRef]
- Aguilera, T.A.; Rafat, M.; Castellini, L.; Shehade, H.; Kariolis, M.S.; Hui, A.B.-Y.; Stehr, H.; von Eyben, R.; Jiang, D.; Ellies, L.G.; et al. Reprogramming the immunological microenvironment through radiation and targeting Axl. Nat. Commun. 2016, 7, 13898. [Google Scholar] [CrossRef]
- Forde, P.M.; Chaft, J.E.; Smith, K.N.; Anagnostou, V.; Cottrell, T.R.; Hellmann, M.D.; Zahurak, M.; Yang, S.C.; Jones, D.R.; Broderick, S.; et al. Neoadjuvant PD-1 Blockade in Resectable Lung Cancer. N. Engl. J. Med. 2018, 378, 1976–1986. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | NSCLC n = 97 (%) | ADC n = 47 (%) | SCC n = 50 (%) | p |
|---|---|---|---|---|
| Gender | ||||
| Men | 84 (86.6%) | 36 (76.6%) | 48 (96%) | 0.005 |
| Women | 13 (13.4%) | 11 (23.4%) | 2 (4%) | |
| Age (years) | ||||
| Mean ± SD | 66.13 ± 8.65 | 65.49 ± 9.64 | 66.7 ± 7.6 | |
| <70 | 58 (59.8%) | 28 (59.6%) | 30 (60%) | 0.966 |
| ≥70 | 39 (40.2%) | 19 (40.4%) | 20 (40%) | |
| Smoking habits | ||||
| Never smoker | 9 (9.3%) | 7 (14.9%) | 2 (4%) | 0.168 |
| Ex-smoker | 62 (63.9%) | 11 (23.4%) | 15 (30%) | |
| Current smoker | 26 (26.8%) | 29 (61.7%) | 33 (66%) | |
| Stage | ||||
| I | 44 (45.4%) | 23 (48.9%) | 21 (42%) | 0.700 |
| II | 25 (36.1%) | 15 (31.9%) | 20 (40%) | |
| III | 18 (18.6%) | 9 (19.1%) | 9 (18%) | |
| N status | ||||
| N0 | 71 (73.2%) | 34 (72.3%) | 37 (74%) | 0.909 |
| N1 | 15 (15.5%) | 8 (17%) | 7 (14%) | |
| N2 | 11 (11.3%) | 5 (10.6%) | 6 (12%) | |
| Tumor size (cm) | ||||
| Mean ± SD | 4.03 ± 2.13 | 3.52 ± 2.1 | 4.5 ± 2.1 | |
| ≤4 cm | 54 (55.8%) | 31 (57.4%) | 23 (42.6%) | 0.048 |
| >4 cm | 43 (44.3%) | 16 (37.2%) | 27 (62.8%) | |
| PET (SUVmax) | ||||
| Mean ± SD | 11.05 ± 5.67 | 9.16 ± 5.06 | 12.74 ± 5.69 | |
| ≤9.4 | 47 (49.5%) | 30 (66.7%) | 17 (34%) | 0.002 |
| >9.4 | 48 (50.5%) | 15 (33.3%) | 33 (66%) | |
| Surgical approach | ||||
| Thoracotomy | 57 (58.8%) | 23 (48.9%) | 34 (68%) | 0.057 |
| VATS | 40 (41.2%) | 24 (51.1%) | 16 (32%) | |
| Type of resection | ||||
| Lobectomy | 80 (82.5%) | 43 (91.5%) | 37 (74%) | 0.024 |
| Pneumonectomy | 17 (17.5%) | 4 (8.5%) | 13 (26%) | |
| Adjuvant chemotherapy | ||||
| No | 59 (60.8%) | 28 (59.6%) | 31 (62%) | 0.807 |
| Yes | 38 (39.2%) | 19 (40.4%) | 19 (38%) | |
| Adjuvant radiotherapy | ||||
| No | 91 (93.8%) | 44 (93.6%) | 47 (94%) | 0.938 |
| Yes | 6 (6.2%) | 3 (6.4%) | 3 (6%) | |
| CTC1 | ||||
| Mean (SD) Range | 2.13 (± 8.7) 0–84 | 1.3 (± 2.6) 0–11 | 2.92 (± 11.9) 0–84 | |
| Absence | 57 (48.8%) | 32 (68.1%) | 25 (50%) | 0.071 |
| Presence | 40 (41.2%) | 15 (31.9%) | 25 (50%) | |
| CTC2 | ||||
| Mean (SD) Range | 0.74 (± 1.6) 0–10 | 0.74 (± 1.9) 0–10 | 0.74 (± 1.2) 0–4 | |
| Absence | 70 (72.2%) | 35 (74.5%) | 35 (70%) | 0.624 |
| Presence | 27 (27.8%) | 12 (25.5%) | 15 (30%) | |
| CTC3 | ||||
| Mean (SD) Range | 0.7 (± 1.8) 0–9 | 0.88 (± 2.1) 0–9 | 0.55 (± 2.8) 0–8 | |
| Absence | 57 (81.4%) | 25 (78.1%) | 32 (84.2%) | 0.514 |
| Presence | 13 (18.6%) | 7 (21.9%) | 6 (15.8%) | |
| CTC1 EMT | NSCLC n = 54 (%) | ADC n = 22 (%) | SCC n = 32 (%) | |
| Mean (SD) Range | 0.3 (± 0.44) 0–3 | 0.14 (± 0.4) 0–1 | 0.41 (± 0.8) 0–3 | |
| Absence | 43 (79.6%) | 19 (86.4%) | 24 (75%) | 0.308 |
| Presence | 11 (20.4%) | 3 (13.6%) | 8 (25%) |
| ADC | |||||||
|---|---|---|---|---|---|---|---|
| RFS | HR | 95% CI | p | OS | HR | 95% CI | p |
| Resection type | Relapse | ||||||
| Pneumonectomy | 4.23 | 1.13–15.8 | 0.032 | Yes | 15.0 | 1.04–216.2 | 0.047 |
| Lobectomy | 1.00 | No | 1.00 | ||||
| CTC2 | PET (SUVmax) | ||||||
| Presence | 2.51 | 1.07–5.87 | 0.034 | >9.4 | 6.4 | 0.96–42.5 | 0.055 |
| Absence | 1.00 | ≤9.4 | 1.00 | ||||
| CTC3 | |||||||
| Presence | 10.8 | 1.54–76.4 | 0.017 | ||||
| Absence | 1.00 | ||||||
| AXL | |||||||
| High | 15.7 | 1.63–150.7 | 0.017 | ||||
| Low | 1.00 | ||||||
| SCC | |||||||
| Size (cm) | Relapse | ||||||
| >4cm | 6.77 | 1.94–23.56 | 0.003 | Yes | 6.42 | 2.17–19.04 | 0.001 |
| ≤4cm | 1.00 | No | 1.00 | ||||
| N status | N status | ||||||
| N0 | 1.00 | 0.192 | N0 | 1.00 | 0.075 | ||
| N1 | 2.72 | 0.92–7.98 | 0.070 | N1 | 1.37 | 0.41–4.60 | 0.609 |
| N2 | 1.26 | 0.28–5.84 | 0.764 | N2 | 3.54 | 1.19–10.59 | 0.024 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Miguel-Pérez, D.; Bayarri-Lara, C.I.; Ortega, F.G.; Russo, A.; Moyano Rodriguez, M.J.; Alvarez-Cubero, M.J.; Maza Serrano, E.; Lorente, J.A.; Rolfo, C.; Serrano, M.J. Post-Surgery Circulating Tumor Cells and AXL Overexpression as New Poor Prognostic Biomarkers in Resected Lung Adenocarcinoma. Cancers 2019, 11, 1750. https://doi.org/10.3390/cancers11111750
de Miguel-Pérez D, Bayarri-Lara CI, Ortega FG, Russo A, Moyano Rodriguez MJ, Alvarez-Cubero MJ, Maza Serrano E, Lorente JA, Rolfo C, Serrano MJ. Post-Surgery Circulating Tumor Cells and AXL Overexpression as New Poor Prognostic Biomarkers in Resected Lung Adenocarcinoma. Cancers. 2019; 11(11):1750. https://doi.org/10.3390/cancers11111750
Chicago/Turabian Stylede Miguel-Pérez, Diego, Clara Isabel Bayarri-Lara, Francisco Gabriel Ortega, Alessandro Russo, María José Moyano Rodriguez, Maria Jesus Alvarez-Cubero, Elizabeth Maza Serrano, José Antonio Lorente, Christian Rolfo, and María José Serrano. 2019. "Post-Surgery Circulating Tumor Cells and AXL Overexpression as New Poor Prognostic Biomarkers in Resected Lung Adenocarcinoma" Cancers 11, no. 11: 1750. https://doi.org/10.3390/cancers11111750
APA Stylede Miguel-Pérez, D., Bayarri-Lara, C. I., Ortega, F. G., Russo, A., Moyano Rodriguez, M. J., Alvarez-Cubero, M. J., Maza Serrano, E., Lorente, J. A., Rolfo, C., & Serrano, M. J. (2019). Post-Surgery Circulating Tumor Cells and AXL Overexpression as New Poor Prognostic Biomarkers in Resected Lung Adenocarcinoma. Cancers, 11(11), 1750. https://doi.org/10.3390/cancers11111750