Are ENT1/ENT1, NOTCH3, and miR-21 Reliable Prognostic Biomarkers in Patients with Resected Pancreatic Adenocarcinoma Treated with Adjuvant Gemcitabine Monotherapy?
Abstract
:1. Introduction
2. Results
2.1. Clinical–Pathological Characteristics of Patients
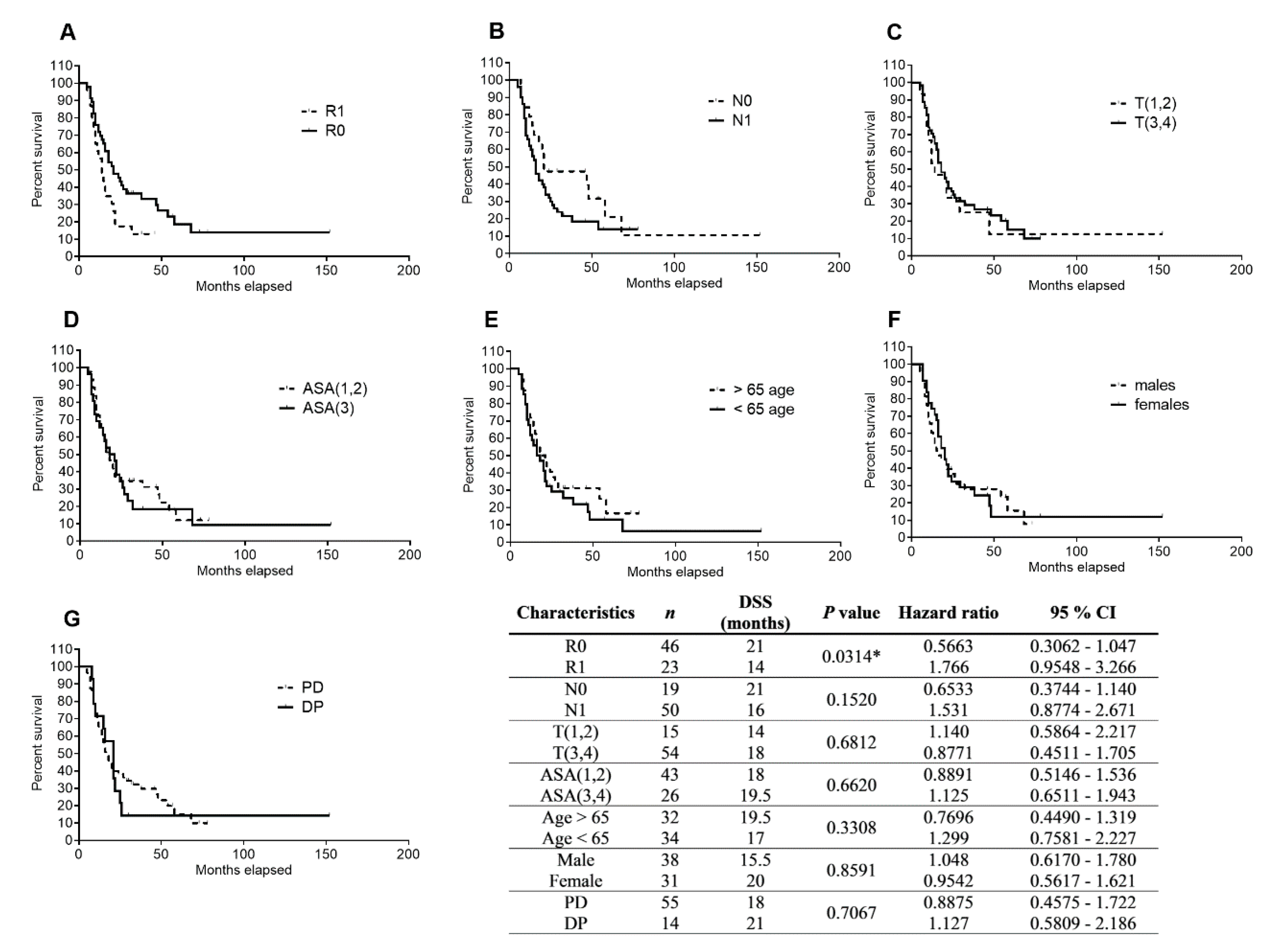
2.2. Clinical–Pathological Factors and Chemotherapy Response as Survival Markers
2.3. Analysis of ENT1, NOTCH3 mRNA, and miR-21 Levels in PDAC Tissue and Their Association with DSS
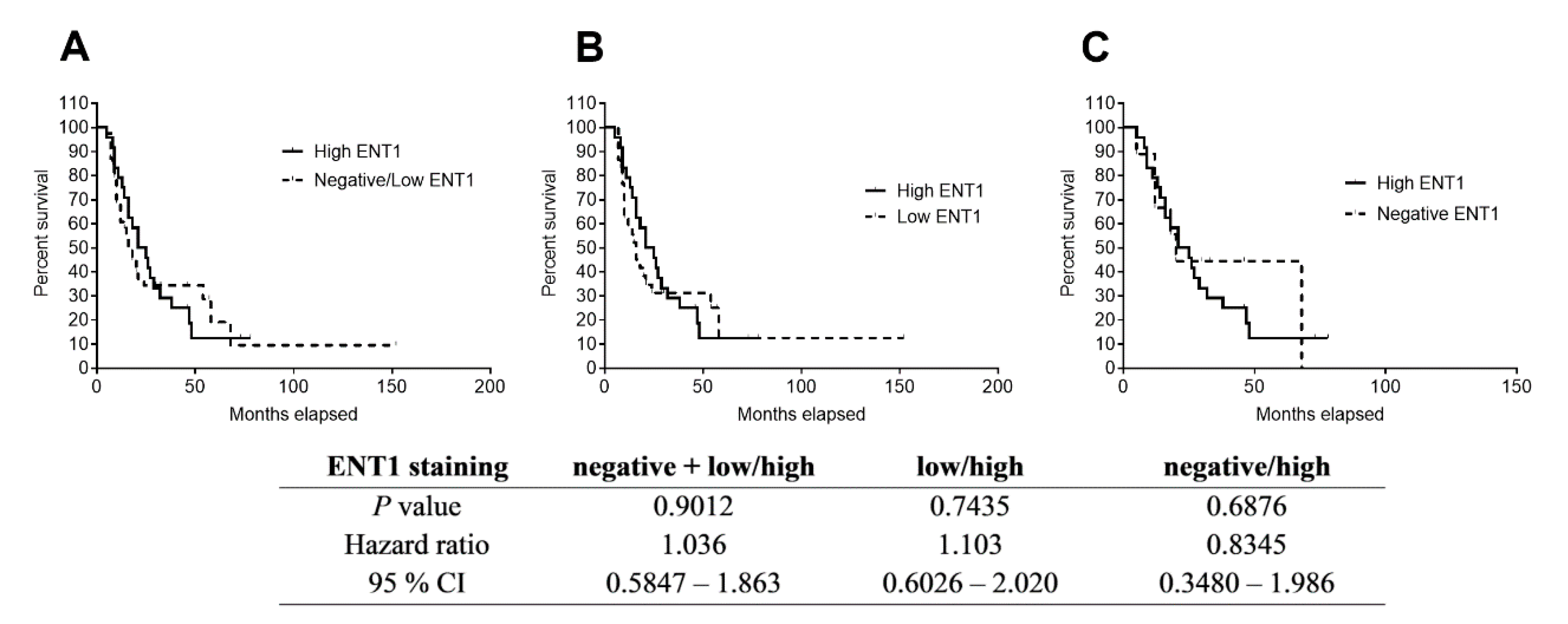
2.4. Immunostaining of ENT1 in FFPE Samples of PDAC
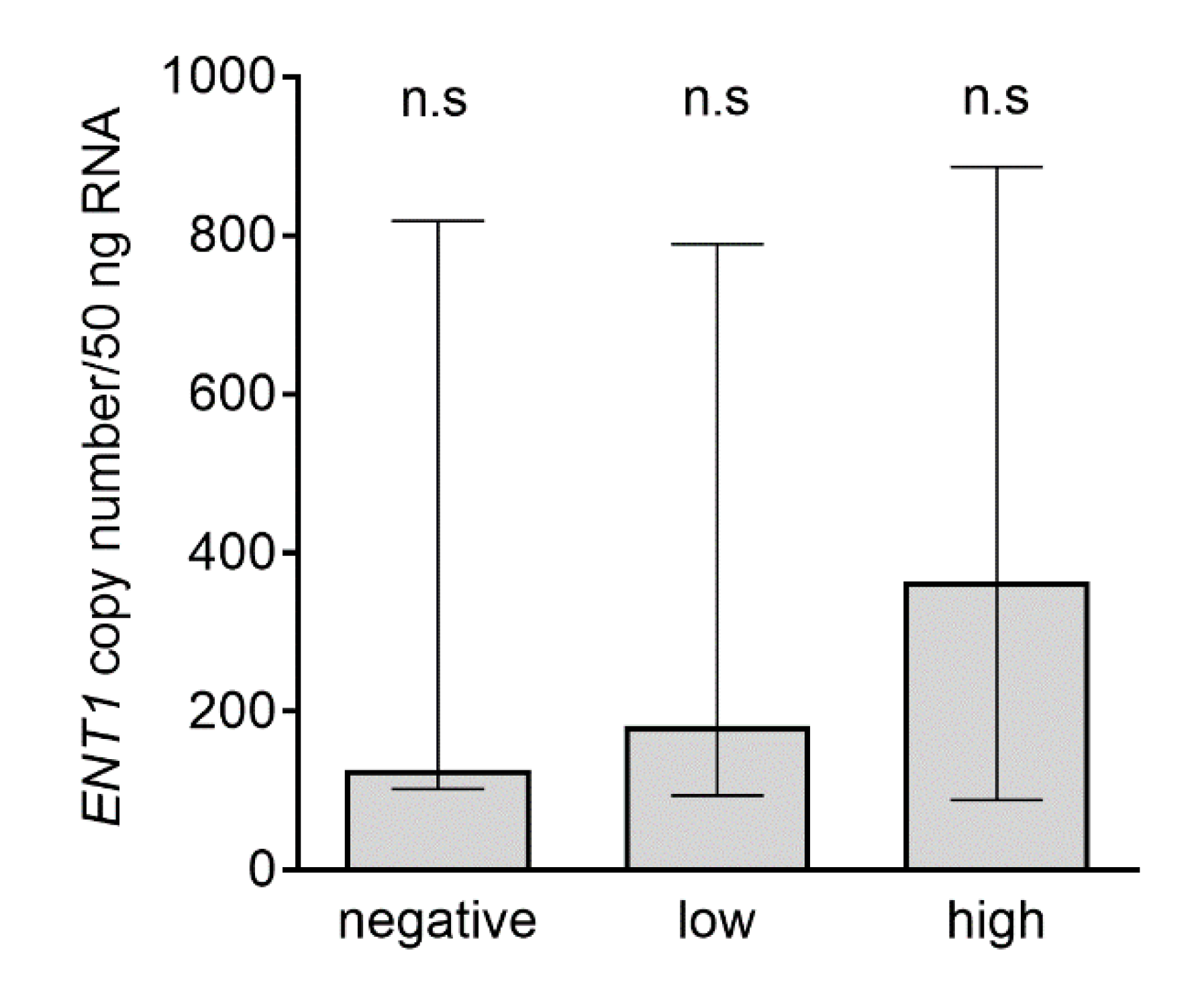
2.5. Analysis of ENT1 Transcripts in Subgroups with Negative, Low, and High ENT1 Protein Expression Analyzed by Immunohistochemistry
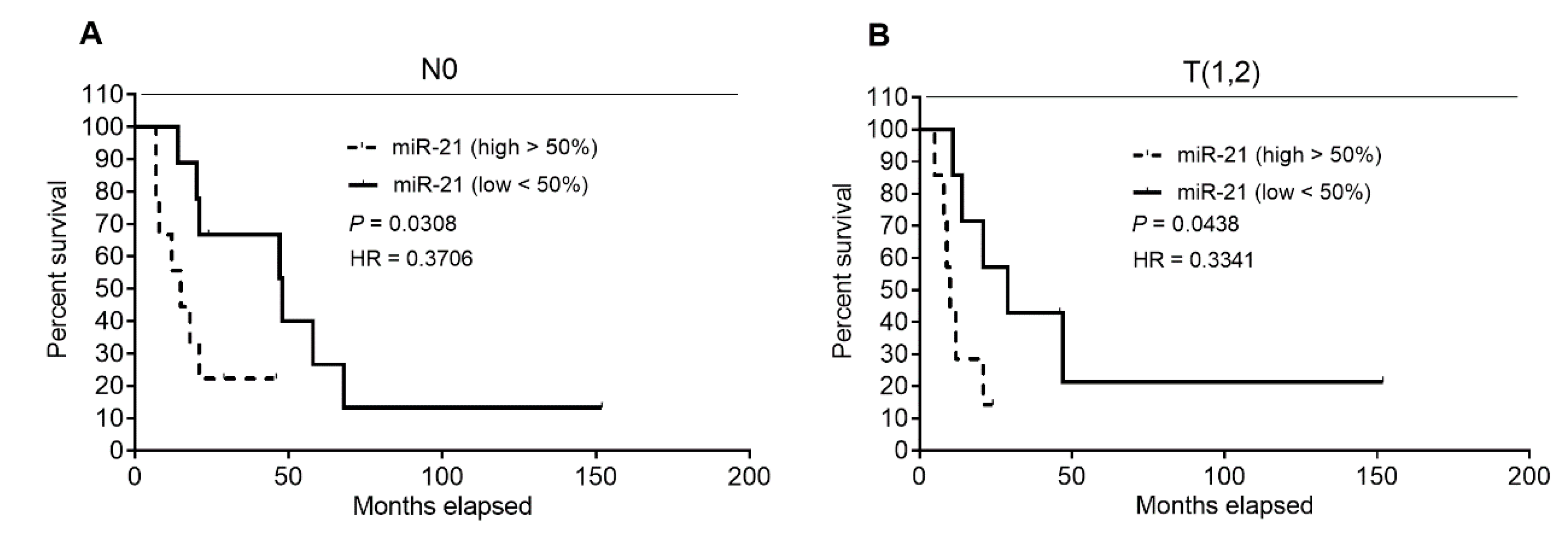
2.6. Analysis of Patients’ DSS Association with ENT1, NOTCH3 mRNA, and miR-21 Levels in Patients’ Subgroups, Defined by Clinical–Pathological Characteristics
2.7. Quantitative RT-PCR Analysis of ENT1, NOTCH3 mRNA, and miR-21 Expression in FFPE Samples of PDAC
3. Discussion
4. Materials and Methods
4.1. Patients and Pancreatic Cancer Staging
4.2. Preparation of Formalin-Fixed Paraffin-Embedded Samples of Pancreas
4.3. Extraction of mRNA from FFPE Samples and Reverse Transcription
4.4. Quantitative Analysis of ENT1, NOTCH3, and miR-21 Expression
4.5. Immunohistochemical Analysis of ENT1 Expression
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Garrido-Laguna, I.; Hidalgo, M. Pancreatic cancer: From state-of-the-art treatments to promising novel therapies. Nat. Rev. Clin. Oncol. 2015, 12, 319–334. [Google Scholar] [CrossRef]
- Kleeff, J.; Korc, M.; Apte, M.; la Vecchia, C.; Johnson, C.D.; Biankin, A.V.; Neale, R.E.; Tempero, M.; Tuveson, D.A.; Hruban, R.H.; et al. Pancreatic cancer. Nat. Rev. Dis. Primers 2016, 2, 16022. [Google Scholar] [CrossRef]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef]
- Kommalapati, A.; Tella, S.H.; Goyal, G.; Ma, W.W.; Mahipal, A. Contemporary management of localized resectable pancreatic cancer. Cancers 2018, 10, 24. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Dunn, J.A.; Stocken, D.D.; Almond, J.; Link, K.; Beger, H.; Bassi, C.; Falconi, M.; Pederzoli, P.; Dervenis, C.; et al. Adjuvant chemoradiotherapy and chemotherapy in resectable pancreatic cancer: A randomised controlled trial. Lancet 2001, 358, 1576–1585. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Friess, H.; Bassi, C.; Dunn, J.A.; Hickey, H.; Beger, H.; Fernandez-Cruz, L.; Dervenis, C.; Lacaine, F.; et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N. Engl. J. Med. 2004, 350, 1200–1210. [Google Scholar] [CrossRef]
- Oettle, H.; Neuhaus, P.; Hochhaus, A.; Hartmann, J.T.; Gellert, K.; Ridwelski, K.; Niedergethmann, M.; Zulke, C.; Fahlke, J.; Arning, M.B.; et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: The CONKO-001 randomized trial. JAMA 2013, 310, 1473–1481. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Smith, C.T.; Bassi, C.; Ghaneh, P.; Owen, E.; Moore, M.; Padbury, R.; Doi, R.; Smith, D.; et al. Adjuvant 5-fluorouracil and folinic acid vs observation for pancreatic cancer: Composite data from the ESPAC-1 and-3(v1) trials. Br. J. Cancer 2009, 100, 246–250. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Palmer, D.H.; Ghaneh, P.; Psarelli, E.E.; Valle, J.W.; Halloran, C.M.; Faluyi, O.; O’Reilly, D.A.; Cunningham, D.; Wadsley, J.; et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet 2017, 389, 1011–1024. [Google Scholar] [CrossRef]
- Conroy, T.; Hammel, P.; Hebbar, M.; Abdelghani, M.B.; Wei, A.C.; Raoul, J.L.; Chone, L.; Francois, E.; Artru, P.; Biagi, J.J.; et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N. Engl. J. Med. 2018, 379, 2395–2406. [Google Scholar] [CrossRef]
- Huang, J.; Liao, W.; Zhou, J.; Zhang, P.; Wen, F.; Wang, X.; Zhang, M.; Zhou, K.; Wu, Q.; Li, Q. Cost-effectiveness analysis of adjuvant treatment for resected pancreatic cancer in China based on the ESPAC-4 trial. Cancer Manag. Res. 2018, 10, 4065–4072. [Google Scholar] [CrossRef]
- Fujita, H.; Ohuchida, K.; Mizumoto, K.; Itaba, S.; Ito, T.; Nakata, K.; Yu, J.; Kayashima, T.; Souzaki, R.; Tajiri, T.; et al. Gene expression levels as predictive markers of outcome in pancreatic cancer after gemcitabine-based adjuvant chemotherapy. Neoplasia 2010, 12, 807–817. [Google Scholar] [CrossRef]
- Greenhalf, W.; Ghaneh, P.; Neoptolemos, J.P.; Palmer, D.H.; Cox, T.F.; Lamb, R.F.; Garner, E.; Campbell, F.; Mackey, J.R.; Costello, E.; et al. Pancreatic cancer hENT1 expression and survival from gemcitabine in patients from the ESPAC-3 trial. J. Natl. Cancer Inst. 2014, 106, djt347. [Google Scholar] [CrossRef]
- Marechal, R.; Bachet, J.B.; Mackey, J.R.; Dalban, C.; Demetter, P.; Graham, K.; Couvelard, A.; Svrcek, M.; Bardier-Dupas, A.; Hammel, P.; et al. Levels of gemcitabine transport and metabolism proteins predict survival times of patients treated with gemcitabine for pancreatic adenocarcinoma. Gastroenterology 2012, 143, 664–674. [Google Scholar] [CrossRef]
- Morinaga, S.; Nakamura, Y.; Watanabe, T.; Mikayama, H.; Tamagawa, H.; Yamamoto, N.; Shiozawa, M.; Akaike, M.; Ohkawa, S.; Kameda, Y.; et al. Immunohistochemical analysis of human equilibrative nucleoside transporter-1 (hENT1) predicts survival in resected pancreatic cancer patients treated with adjuvant gemcitabine monotherapy. Ann. Surg. Oncol. 2012, 19 (Suppl. 3), S558–S564. [Google Scholar] [CrossRef]
- Andersson, R.; Aho, U.; Nilsson, B.I.; Peters, G.J.; Pastor-Anglada, M.; Rasch, W.; Sandvold, M.L. Gemcitabine chemoresistance in pancreatic cancer: Molecular mechanisms and potential solutions. Scand. J. Gastroenterol. 2009, 44, 782–786. [Google Scholar] [CrossRef]
- Garcia-Manteiga, J.; Molina-Arcas, M.; Casado, F.J.; Mazo, A.; Pastor-Anglada, M. Nucleoside transporter profiles in human pancreatic cancer cells: Role of hCNT1 in 2′,2′-difluorodeoxycytidine-induced cytotoxicity. Clin. Cancer Res. 2003, 9, 5000–5008. [Google Scholar]
- Farrell, J.J.; Elsaleh, H.; Garcia, M.; Lai, R.; Ammar, A.; Regine, W.F.; Abrams, R.; Benson, A.B.; Macdonald, J.; Cass, C.E.; et al. Human equilibrative nucleoside transporter 1 levels predict response to gemcitabine in patients with pancreatic cancer. Gastroenterology 2009, 136, 187–195. [Google Scholar] [CrossRef]
- Giovannetti, E.; del Tacca, M.; Mey, V.; Funel, N.; Nannizzi, S.; Ricci, S.; Orlandini, C.; Boggi, U.; Campani, D.; del Chiaro, M.; et al. Transcription analysis of human equilibrative nucleoside transporter-1 predicts survival in pancreas cancer patients treated with gemcitabine. Cancer Res. 2006, 66, 3928–3935. [Google Scholar] [CrossRef]
- Svrcek, M.; Cros, J.; Marechal, R.; Bachet, J.B.; Flejou, J.F.; Demetter, P. Human equilibrative nucleoside transporter 1 testing in pancreatic ductal adenocarcinoma: A comparison between murine and rabbit antibodies. Histopathology 2015, 66, 457–462. [Google Scholar] [CrossRef]
- Sinn, M.; Riess, H.; Sinn, B.V.; Stieler, J.M.; Pelzer, U.; Striefler, J.K.; Oettle, H.; Bahra, M.; Denkert, C.; Blaker, H.; et al. Human equilibrative nucleoside transporter 1 expression analysed by the clone SP 120 rabbit antibody is not predictive in patients with pancreatic cancer treated with adjuvant gemcitabine—Results from the CONKO-001 trial. Eur. J. Cancer 2015, 51, 1546–1554. [Google Scholar] [CrossRef]
- Brunetti, O.; Russo, A.; Scarpa, A.; Santini, D.; Reni, M.; Bittoni, A.; Azzariti, A.; Aprile, G.; Delcuratolo, S.; Signorile, M.; et al. MicroRNA in pancreatic adenocarcinoma: Predictive/prognostic biomarkers or therapeutic targets? Oncotarget 2015, 6, 23323–23341. [Google Scholar] [CrossRef]
- Chan, J.A.; Krichevsky, A.M.; Kosik, K.S. MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res. 2005, 65, 6029–6033. [Google Scholar] [CrossRef]
- Dhayat, S.A.; Abdeen, B.; Kohler, G.; Senninger, N.; Haier, J.; Mardin, W.A. MicroRNA-100 and microRNA-21 as markers of survival and chemotherapy response in pancreatic ductal adenocarcinoma UICC stage II. Clin. Epigenet. 2015, 7, 132. [Google Scholar] [CrossRef]
- Giovannetti, E.; Funel, N.; Peters, G.J.; del Chiaro, M.; Erozenci, L.A.; Vasile, E.; Leon, L.G.; Pollina, L.E.; Groen, A.; Falcone, A.; et al. MicroRNA-21 in pancreatic cancer: Correlation with clinical outcome and pharmacologic aspects underlying its role in the modulation of gemcitabine activity. Cancer Res. 2010, 70, 4528–4538. [Google Scholar] [CrossRef]
- Kim, R.; Tan, A.; Lai, K.K.; Jiang, J.; Wang, Y.; Rybicki, L.A.; Liu, X. Prognostic roles of human equilibrative transporter 1 (hENT-1) and ribonucleoside reductase subunit M1 (RRM1) in resected pancreatic cancer. Cancer 2011, 117, 3126–3134. [Google Scholar] [CrossRef]
- Vychytilova-Faltejskova, P.; Kiss, I.; Klusova, S.; Hlavsa, J.; Prochazka, V.; Kala, Z.; Mazanec, J.; Hausnerova, J.; Kren, L.; Hermanova, M.; et al. MiR-21, miR-34a, miR-198 and miR-217 as diagnostic and prognostic biomarkers for chronic pancreatitis and pancreatic ductal adenocarcinoma. Diagn. Pathol. 2015, 10, 38. [Google Scholar] [CrossRef]
- Kalloger, S.E.; Riazy, M.; Tessier-Cloutier, B.; Karasinska, J.M.; Gao, D.; Peixoto, R.D.; Samimi, S.; Chow, C.; Wong, H.L.; Mackey, J.R.; et al. A predictive analysis of the SP120 and 10D7G2 antibodies for human equilibrative nucleoside transporter 1 (hENT1) in pancreatic ductal adenocarcinoma treated with adjuvant gemcitabine. J. Pathol. Clin. Res. 2017, 3, 179–190. [Google Scholar] [CrossRef]
- Dang, L.; Fan, X.; Chaudhry, A.; Wang, M.; Gaiano, N.; Eberhart, C.G. Notch3 signaling initiates choroid plexus tumor formation. Oncogene 2006, 25, 487–491. [Google Scholar] [CrossRef]
- Ashida, R.; Nakata, B.; Shigekawa, M.; Mizuno, N.; Sawaki, A.; Hirakawa, K.; Arakawa, T.; Yamao, K. Gemcitabine sensitivity-related mRNA expression in endoscopic ultrasound-guided fine-needle aspiration biopsy of unresectable pancreatic cancer. J. Exp. Clin. Cancer Res. 2009, 28, 83. [Google Scholar] [CrossRef]
- Eto, K.; Kawakami, H.; Kuwatani, M.; Kudo, T.; Abe, Y.; Kawahata, S.; Takasawa, A.; Fukuoka, M.; Matsuno, Y.; Asaka, M.; et al. Human equilibrative nucleoside transporter 1 and Notch3 can predict gemcitabine effects in patients with unresectable pancreatic cancer. Br. J. Cancer 2013, 108, 1488–1494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dillhoff, M.; Liu, J.; Frankel, W.; Croce, C.; Bloomston, M. MicroRNA-21 is overexpressed in pancreatic cancer and a potential predictor of survival. J. Gastrointest. Surg. 2008, 12, 2171–2176. [Google Scholar] [CrossRef] [PubMed]
- Mann, C.D.; Bastianpillai, C.; Neal, C.P.; Masood, M.M.; Jones, D.J.; Teichert, F.; Singh, R.; Karpova, E.; Berry, D.P.; Manson, M.M. Notch3 and HEY-1 as prognostic biomarkers in pancreatic adenocarcinoma. PLoS ONE 2012, 7, e51119. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.X.; Zhou, L.; Li, Q.J.; Feng, W.; Wang, P.M.; Li, E.F.; Gong, W.J.; Kou, M.W.; Gou, W.T.; Yang, Y.L. Association between high levels of Notch3 expression and high invasion and poor overall survival rates in pancreatic ductal adenocarcinoma. Oncol. Rep. 2016, 36, 2893–2901. [Google Scholar] [CrossRef] [PubMed]
- Song, H.Y.; Wang, Y.; Lan, H.; Zhang, Y.X. Expression of Notch receptors and their ligands in pancreatic ductal adenocarcinoma. Exp. Ther. Med. 2018, 16, 53–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volinia, S.; Calin, G.A.; Liu, C.G.; Ambs, S.; Cimmino, A.; Petrocca, F.; Visone, R.; Iorio, M.; Roldo, C.; Ferracin, M.; et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA 2006, 103, 2257–2261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bloomston, M.; Frankel, W.L.; Petrocca, F.; Volinia, S.; Alder, H.; Hagan, J.P.; Liu, C.G.; Bhatt, D.; Taccioli, C.; Croce, C.M. MicroRNA expression patterns to differentiate pancreatic adenocarcinoma from normal pancreas and chronic pancreatitis. JAMA 2007, 297, 1901–1908. [Google Scholar] [CrossRef]
- Gerard, C.; Fagnoni, P.; Vienot, A.; Borg, C.; Limat, S.; Daval, F.; Calais, F.; Vardanega, J.; Jary, M.; Nerich, V. A systematic review of economic evaluation in pancreatic ductal adenocarcinoma. Eur. J. Cancer 2017, 86, 207–216. [Google Scholar] [CrossRef]
- Ansari, D.; Tingstedt, B.; Andersson, R. Pancreatic cancer—Cost for overtreatment with gemcitabine. Acta Oncol. 2013, 52, 1146–1151. [Google Scholar] [CrossRef]
- Bordeaux, J.; Welsh, A.; Agarwal, S.; Killiam, E.; Baquero, M.; Hanna, J.; Anagnostou, V.; Rimm, D. Antibody validation. Biotechniques 2010, 48, 197–209. [Google Scholar] [CrossRef] [Green Version]
- Voskuil, J.L. The challenges with the validation of research antibodies. F1000Research 2017, 6, 161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uson, P.L.S.J.; Macarenco, R.; Oliveira, F.N.; Smaletz, O. Impact of pathology review for decision therapy in localized prostate cancer. Clin. Med. Insights Pathol. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Middleton, L.P.; Feeley, T.W.; Albright, H.W.; Walters, R.; Hamilton, S.H. Second-opinion pathologic review is a patient safety mechanism that helps reduce error and decrease waste. J. Oncol. Pract. 2014, 10, 275–280. [Google Scholar] [CrossRef]
- Hidalgo, M.; Cascinu, S.; Kleeff, J.; Labianca, R.; Lohr, J.M.; Neoptolemos, J.; Real, F.X.; van Laethem, J.L.; Heinemann, V. Addressing the challenges of pancreatic cancer: Future directions for improving outcomes. Pancreatology 2015, 15, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ansari, D.; Tingstedt, B.; Andersson, B.; Holmquist, F.; Sturesson, C.; Williamsson, C.; Sasor, A.; Borg, D.; Bauden, M.; Andersson, R. Pancreatic cancer: Yesterday, today and tomorrow. Future Oncol. 2016, 12, 1929–1946. [Google Scholar] [CrossRef] [PubMed]
- Spratlin, J.; Sangha, R.; Glubrecht, D.; Dabbagh, L.; Young, J.D.; Dumontet, C.; Cass, C.; Lai, R.; Mackey, J.R. The absence of human equilibrative nucleoside transporter 1 is associated with reduced survival in patients with gemcitabine-treated pancreas adenocarcinoma. Clin. Cancer Res. 2004, 10, 6956–6961. [Google Scholar] [CrossRef]
- Nordh, S.; Ansari, D.; Andersson, R. hENT1 expression is predictive of gemcitabine outcome in pancreatic cancer: A systematic review. World J. Gastroenterol. 2014, 20, 8482–8490. [Google Scholar] [CrossRef]
- Marechal, R.; Mackey, J.R.; Lai, R.; Demetter, P.; Peeters, M.; Polus, M.; Cass, C.E.; Young, J.; Salmon, I.; Deviere, J.; et al. Human equilibrative nucleoside transporter 1 and human concentrative nucleoside transporter 3 predict survival after adjuvant gemcitabine therapy in resected pancreatic adenocarcinoma. Clin. Cancer Res. 2009, 15, 2913–2919. [Google Scholar] [CrossRef]
- Baldwin, S.; Kukar, M.; Gabriel, E.; Attwood, K.; Wilkinson, N.; Hochwald, S.N.; Kuvshinoff, B. Pancreatic cancer metastatic to a limited number of lymph nodes has no impact on outcome. Hpb 2016, 18, 523–528. [Google Scholar] [CrossRef] [Green Version]
- Demir, I.E.; Jager, C.; Schlitter, A.M.; Konukiewitz, B.; Stecher, L.; Schorn, S.; Tieftrunk, E.; Scheufele, F.; Calavrezos, L.; Schirren, R.; et al. R0 versus R1 resection matters after pancreaticoduodenectomy, and less after distal or total pancreatectomy for pancreatic cancer. Ann. Surg. 2018, 268, 1058–1068. [Google Scholar] [CrossRef]
- Valsangkar, N.P.; Bush, D.M.; Michaelson, J.S.; Ferrone, C.R.; Wargo, J.A.; Lillemoe, K.D.; Fernandez-del Castillo, C.; Warshaw, A.L.; Thayer, S.P. N0/N1, PNL, or LNR? The effect of lymph node number on accurate survival prediction in pancreatic ductal adenocarcinoma. J. Gastrointest. Surg. 2013, 17, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Cho, J.H.; Lee, S.J.; Lee, D.K.; Yoon, D.S.; Cho, J.Y. Gemcitabine combined with capecitabine compared to gemcitabine with or without erlotinib as first-line chemotherapy in patients with advanced pancreatic cancer. Cancer Res. Treat. 2015, 47, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; de Preter, K.; Pattyn, F.; Poppe, B.; van Roy, N.; de Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, research0034-1. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Zhang, H.; Miranda, L.; Lin, S. Serious overestimation in quantitative PCR by circular (supercoiled) plasmid standard: Microalgal pcna as the model gene. PLoS ONE 2010, 5, e9545. [Google Scholar] [CrossRef]
- Pfaffl, M.W.; Hageleit, M. Validities of mRNA quantification using recombinant RNA and recombinant DNA external calibration curves in real-time RT-PCR. Biotechnol. Lett. 2001, 23, 275–282. [Google Scholar] [CrossRef]
- Lin, C.H.; Chen, Y.C.; Pan, T.M. Quantification bias caused by plasmid DNA conformation in quantitative real-time PCR assay. PLoS ONE 2011, 6, e29101. [Google Scholar] [CrossRef]
- Cecka, F.; Jon, B.; Subrt, Z.; Ferko, A. Solid pseudopapillary tumour of the pancreas: Diagnosis, treatment, and prognosis. Acta Chir. Belg. 2014, 114, 58–62. [Google Scholar] [CrossRef]
- Hlavsa, J.; Cecka, F.; Zaruba, P.; Zajak, J.; Gurlich, R.; Strnad, R.; Pavlik, T.; Kala, Z.; Lovecek, M. Tumor grade as significant prognostic factor in pancreatic cancer: Validation of a novel TNMG staging system. Neoplasma 2018, 65, 637–643. [Google Scholar] [CrossRef]
- Lidsky, M.E.; Sun, Z.; Nussbaum, D.P.; Adam, M.A.; Speicher, P.J.; Blazer, D.G., 3rd. Going the extra mile: Improved survival for pancreatic cancer patients traveling to high-volume centers. Ann. Surg. 2017, 266, 333–338. [Google Scholar] [CrossRef]
- Blackford, A.; Serrano, O.K.; Wolfgang, C.L.; Parmigiani, G.; Jones, S.; Zhang, X.; Parsons, D.W.; Lin, J.C.; Leary, R.J.; Eshleman, J.R.; et al. SMAD4 gene mutations are associated with poor prognosis in pancreatic cancer. Clin. Cancer Res. 2009, 15, 4674–4679. [Google Scholar] [CrossRef]
- Donahue, T.R.; Tran, L.M.; Hill, R.; Li, Y.; Kovochich, A.; Calvopina, J.H.; Patel, S.G.; Wu, N.; Hindoyan, A.; Farrell, J.J.; et al. Integrative survival-based molecular profiling of human pancreatic cancer. Clin. Cancer Res. 2012, 18, 1352–1363. [Google Scholar] [CrossRef] [PubMed]
- Newhook, T.E.; Blais, E.M.; Lindberg, J.M.; Adair, S.J.; Xin, W.; Lee, J.K.; Papin, J.A.; Parsons, J.T.; Bauer, T.W. A thirteen-gene expression signature predicts survival of patients with pancreatic cancer and identifies new genes of interest. PLoS ONE 2014, 9, e105631. [Google Scholar] [CrossRef] [PubMed]
- Stratford, J.K.; Bentrem, D.J.; Anderson, J.M.; Fan, C.; Volmar, K.A.; Marron, J.S.; Routh, E.D.; Caskey, L.S.; Samuel, J.C.; Der, C.J.; et al. A six-gene signature predicts survival of patients with localized pancreatic ductal adenocarcinoma. PLoS Med. 2010, 7, e1000307. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Beyer, A.; Aebersold, R. On the dependency of cellular protein levels on mRNA abundance. Cell 2016, 165, 535–550. [Google Scholar] [CrossRef]
- Aust, D.E.; Terdiman, J.P.; Willenbucher, R.F.; Chew, K.; Ferrell, L.; Florendo, C.; Molinaro-Clark, A.; Baretton, G.B.; Lohrs, U.; Waldman, F.M. Altered distribution of β-catenin, and its binding proteins E-cadherin and APC, in ulcerative colitis-related colorectal cancers. Mod. Pathol. 2001, 14, 29–39. [Google Scholar] [CrossRef]
- Sajic, T.; Ciuffa, R.; Lemos, V.; Xu, P.; Leone, V.; Li, C.; Williams, E.G.; Makris, G.; Banaei-Esfahani, A.; Heikenwalder, M.; et al. A new class of protein biomarkers based on subcellular distribution: Application to a mouse liver cancer model. Sci. Rep. 2019, 9, 6913. [Google Scholar] [CrossRef] [PubMed]
- Emanuelli, A.; Ayyathan, D.M.; Koganti, P.; Shah, P.A.; Apel-Sarid, L.; Paolini, B.; Detroja, R.; Frenkel-Morgenstern, M.; Blank, M. Altered expression and localization of tumor suppressive E3 ubiquitin ligase SMURF2 in human prostate and breast cancer. Cancers 2019, 11, 556. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Maitra, A.; Ghosh, B.; Zechner, U.; Argani, P.; Iacobuzio-Donahue, C.A.; Sriuranpong, V.; Iso, T.; Meszoely, I.M.; Wolfe, M.S.; et al. Notch mediates TGFα-induced changes in epithelial differentiation during pancreatic tumorigenesis. Cancer Cell 2003, 3, 565–576. [Google Scholar] [CrossRef]
- Vo, K.; Amarasinghe, B.; Washington, K.; Gonzalez, A.; Berlin, J.; Dang, T.P. Targeting notch pathway enhances rapamycin antitumor activity in pancreas cancers through PTEN phosphorylation. Mol. Cancer 2011, 10, 138. [Google Scholar] [CrossRef]
- Gnoni, A.; Licchetta, A.; Scarpa, A.; Azzariti, A.; Brunetti, A.E.; Simone, G.; Nardulli, P.; Santini, D.; Aieta, M.; Delcuratolo, S.; et al. Carcinogenesis of pancreatic adenocarcinoma: Precursor lesions. Int. J. Mol. Sci. 2013, 14, 19731–19762. [Google Scholar] [CrossRef]
- Cerveny, L.; Ptackova, Z.; Ceckova, M.; Karahoda, R.; Karbanova, S.; Jiraskova, L.; Greenwood, S.L.; Glazier, J.D.; Staud, F. Equilibrative nucleoside transporter 1 (ENT1, SLC29A1) facilitates transfer of the antiretroviral drug abacavir across the placenta. Drug Metab. Dispos. 2018, 46, 1817–1826. [Google Scholar] [CrossRef] [PubMed]
- del Santo, B.; Valdes, R.; Mata, J.; Felipe, A.; Casado, F.J.; Pastor-Anglada, M. Differential expression and regulation of nucleoside transport systems in rat liver parenchymal and hepatoma cells. Hepatology 1998, 28, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Jiraskova, L.; Cerveny, L.; Karbanova, S.; Ptackova, Z.; Staud, F. Expression of concentrative nucleoside transporters (SLC28A) in the human placenta: effects of gestation age and prototype differentiation-affecting agents. Mol. Pharm. 2018, 15, 2732–2741. [Google Scholar] [CrossRef]
- Hulse, A.M.; Cai, J.J. Genetic variants contribute to gene expression variability in humans. Genetics 2013, 193, 95–108. [Google Scholar] [CrossRef]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef] [PubMed]
- Verbeke, C.S. Resection margins and R1 rates in pancreatic cancer—Are we there yet? Histopathology 2008, 52, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Menon, K.V.; Gomez, D.; Smith, A.M.; Anthoney, A.; Verbeke, C.S. Impact of margin status on survival following pancreatoduodenectomy for cancer: The Leeds Pathology Protocol (LEEPP). Hpb 2009, 11, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Kotorashvili, A.; Ramnauth, A.; Liu, C.; Lin, J.; Ye, K.; Kim, R.; Hazan, R.; Rohan, T.; Fineberg, S.; Loudig, O. Effective DNA/RNA co-extraction for analysis of microRNAs, mRNAs, and genomic DNA from formalin-fixed paraffin-embedded specimens. PLoS ONE 2012, 7, e34683. [Google Scholar] [CrossRef]
- Abrahamsen, H.N.; Steiniche, T.; Nexo, E.; Hamilton-Dutoit, S.J.; Sorensen, B.S. Towards quantitative mRNA analysis in paraffin-embedded tissues using real-time reverse transcriptase-polymerase chain reaction: A methodological study on lymph nodes from melanoma patients. J. Mol. Diagn. 2003, 5, 34–41. [Google Scholar] [CrossRef]
- Antonov, J.; Goldstein, D.R.; Oberli, A.; Baltzer, A.; Pirotta, M.; Fleischmann, A.; Altermatt, H.J.; Jaggi, R. Reliable gene expression measurements from degraded RNA by quantitative real-time PCR depend on short amplicons and a proper normalization. Lab. Investig. 2005, 85, 1040–1050. [Google Scholar] [CrossRef] [Green Version]
- Szafranska, A.E.; Davison, T.S.; Shingara, J.; Doleshal, M.; Riggenbach, J.A.; Morrison, C.D.; Jewell, S.; Labourier, E. Accurate molecular characterization of formalin-fixed, paraffin-embedded tissues by microRNA expression profiling. J. Mol. Diagn. 2008, 10, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Ueno, H.; Ikeda, M.; Kojima, Y.; Hagihara, A.; Kondo, S.; Morizane, C.; Okusaka, T. Do recurrent and metastatic pancreatic cancer patients have the same outcomes with gemcitabine treatment? Oncology 2009, 77, 217–223. [Google Scholar] [CrossRef]
- Metindir, J.; Dilek, G.B.; Pak, I. Staining characterization by immunohistochemistry of tumor cancer antigen in patients with endometrial cancer. Eur. J. Gynaecol. Oncol. 2008, 29, 489–492. [Google Scholar]
- Clark, T.G.; Bradburn, M.J.; Love, S.B.; Altman, D.G. Survival analysis part I: Basic concepts and first analyses. Br. J. Cancer 2003, 89, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Bradburn, M.J.; Clark, T.G.; Love, S.B.; Altman, D.G. Survival analysis part II: Multivariate data analysis—An introduction to concepts and methods. Br. J. Cancer 2003, 89, 431–436. [Google Scholar] [CrossRef] [PubMed]



| Number of Patients | 69 |
| Gender (females/males) | 31/38 |
| Age (years) | |
| Median | 65 |
| Range | 39–80 |
| Surgery (type of resection) | |
| PD | 55 |
| DP | 14 |
| Resection margin status | |
| R0 | 46 |
| R1 | 23 |
| T: stage of primary tumor | |
| T1 | 3 |
| T2 | 12 |
| T3 | 53 |
| T4 | 1 |
| N: regional lymph nodes | |
| N0 | 19 |
| N1 | 50 |
| M: distant metastasis | |
| M0 | 68 |
| M1 | 1 |
| DSS from surgery (months) | |
| Median | 21 |
| Range | 5–152 |
| ASA (I-III) | |
| I | 1 |
| II | 42 |
| III | 26 |
| Patient Stratification | p Value | Hazard Ratio | 95.0% CI | |
|---|---|---|---|---|
| Gender (male) | 0.918 | 0.970 | 0.543 | 1.731 |
| ENT1 (above median) | 0.821 | 0.934 | 0.514 | 1.695 |
| Resection margin status (R1) | 0.135 | 0.624 | 0.336 | 1.159 |
| ASA score | ||||
| ASA III (reference value) | 0.601 | |||
| ASA I | 0.421 | 2.395 | 0.286 | 20.063 |
| ASA II | 0.639 | 0.869 | 0.484 | 1.561 |
| Primary tumor stage | ||||
| T4 (reference value) | 0.272 | |||
| T1 | 0.434 | 0.354 | 0.026 | 4.786 |
| T2 | 0.182 | 0.212 | 0.022 | 2.068 |
| T3 | 0.106 | 0.169 | 0.019 | 1.463 |
| Regional lymph nodes (N1) | 0.149 | 0.591 | 0.289 | 1.207 |
| Resection type (DP) | 0.675 | 1.159 | 0.582 | 2.309 |
| Patient Stratification | p Value | Hazard Ratio | 95.0% CI | |
|---|---|---|---|---|
| Gender (male) | 0.956 | 0.984 | 0.553 | 1.751 |
| NOTCH3 (above median) | 0.383 | 1.286 | 0.731 | 2.263 |
| Resection margin status (R1) | 0.126 | 0.614 | 0.328 | 1.147 |
| ASA score | ||||
| ASA III (reference value) | 0.725 | |||
| ASA I | 0.484 | 2.120 | 0.259 | 17.379 |
| ASA II | 0.773 | 0.915 | 0.501 | 1.671 |
| Primary tumor stage | ||||
| T4 (reference value) | 0.299 | |||
| T1 | 0.507 | 0.424 | 0.034 | 5.358 |
| T2 | 0.267 | 0.275 | 0.028 | 2.686 |
| T3 | 0.144 | 0.200 | 0.023 | 1.728 |
| Regional lymph nodes (N1) | 0.152 | 0.598 | 0.296 | 1.209 |
| Resection type (DP) | 0.661 | 1.163 | 0.593 | 2.283 |
| Patient Stratification | p Value | Hazard Ratio | 95.0% CI | |
|---|---|---|---|---|
| Gender (male) | 0.817 | 0.935 | 0.530 | 1.649 |
| miR-21 (above median) | 0.089 | 0.475 | 0.201 | 1.120 |
| Resection margin status (R1) | 0.113 | 0.604 | 0.324 | 1.126 |
| ASA score | ||||
| ASA III (reference value) | 0.587 | |||
| ASA I | 0.477 | 2.138 | 0.263 | 17.375 |
| ASA II | 0.530 | 0.828 | 0.460 | 1.491 |
| Primary tumor stage | ||||
| T4 (reference value) | 0.091 | |||
| T1 | 0.280 | 0.237 | 0.017 | 3.234 |
| T2 | 0.088 | 0.131 | 0.013 | 1.355 |
| T3 | 0.036 | 0.085 | 0.009 | 0.846 |
| Regional lymph nodes (N1) | 0.175 | 0.619 | 0.310 | 1.238 |
| Resection type (DP) | 0.658 | 1.169 | 0.586 | 2.335 |
| Patient Stratification | p Value | Hazard Ratio | 95.0% CI | |
|---|---|---|---|---|
| Gender (male) | 0.836 | 0.936 | 0.500 | 1.752 |
| ENT1 | ||||
| High (reference value) | 0.277 | |||
| Negative | 0.471 | 1.469 | 0.517 | 4.174 |
| Low | 0.109 | 1.800 | 0.877 | 3.695 |
| Resection margin status (R1) | 0.051 | 0.505 | 0.254 | 1.003 |
| ASA score | ||||
| ASA III (reference value) | 0.592 | |||
| ASA I | 0.768 | 1.385 | 0.159 | 12.091 |
| ASA II | 0.367 | 0.747 | 0.396 | 1.408 |
| Primary tumor stage | ||||
| T4 (reference value) | 0.179 | |||
| T1 | 0.637 | 0.513 | 0.032 | 8.162 |
| T2 | 0.175 | 0.211 | 0.022 | 2.001 |
| T3 | 0.075 | 0.138 | 0.016 | 1.222 |
| Regional lymph nodes (N1) | 0.049 | 0.424 | 0.180 | 0.996 |
| Resection type (DP) | 0.865 | 1.070 | 0.492 | 2.325 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiraskova, L.; Ryska, A.; Duintjer Tebbens, E.J.; Hornychova, H.; Cecka, F.; Staud, F.; Cerveny, L. Are ENT1/ENT1, NOTCH3, and miR-21 Reliable Prognostic Biomarkers in Patients with Resected Pancreatic Adenocarcinoma Treated with Adjuvant Gemcitabine Monotherapy? Cancers 2019, 11, 1621. https://doi.org/10.3390/cancers11111621
Jiraskova L, Ryska A, Duintjer Tebbens EJ, Hornychova H, Cecka F, Staud F, Cerveny L. Are ENT1/ENT1, NOTCH3, and miR-21 Reliable Prognostic Biomarkers in Patients with Resected Pancreatic Adenocarcinoma Treated with Adjuvant Gemcitabine Monotherapy? Cancers. 2019; 11(11):1621. https://doi.org/10.3390/cancers11111621
Chicago/Turabian StyleJiraskova, Lucie, Ales Ryska, Erik Jurjen Duintjer Tebbens, Helena Hornychova, Filip Cecka, Frantisek Staud, and Lukas Cerveny. 2019. "Are ENT1/ENT1, NOTCH3, and miR-21 Reliable Prognostic Biomarkers in Patients with Resected Pancreatic Adenocarcinoma Treated with Adjuvant Gemcitabine Monotherapy?" Cancers 11, no. 11: 1621. https://doi.org/10.3390/cancers11111621
APA StyleJiraskova, L., Ryska, A., Duintjer Tebbens, E. J., Hornychova, H., Cecka, F., Staud, F., & Cerveny, L. (2019). Are ENT1/ENT1, NOTCH3, and miR-21 Reliable Prognostic Biomarkers in Patients with Resected Pancreatic Adenocarcinoma Treated with Adjuvant Gemcitabine Monotherapy? Cancers, 11(11), 1621. https://doi.org/10.3390/cancers11111621






