Combination of Different Fecal Immunochemical Tests in Colorectal Cancer Screening: Any Gain in Diagnostic Performance?
Abstract
:1. Introduction
2. Results
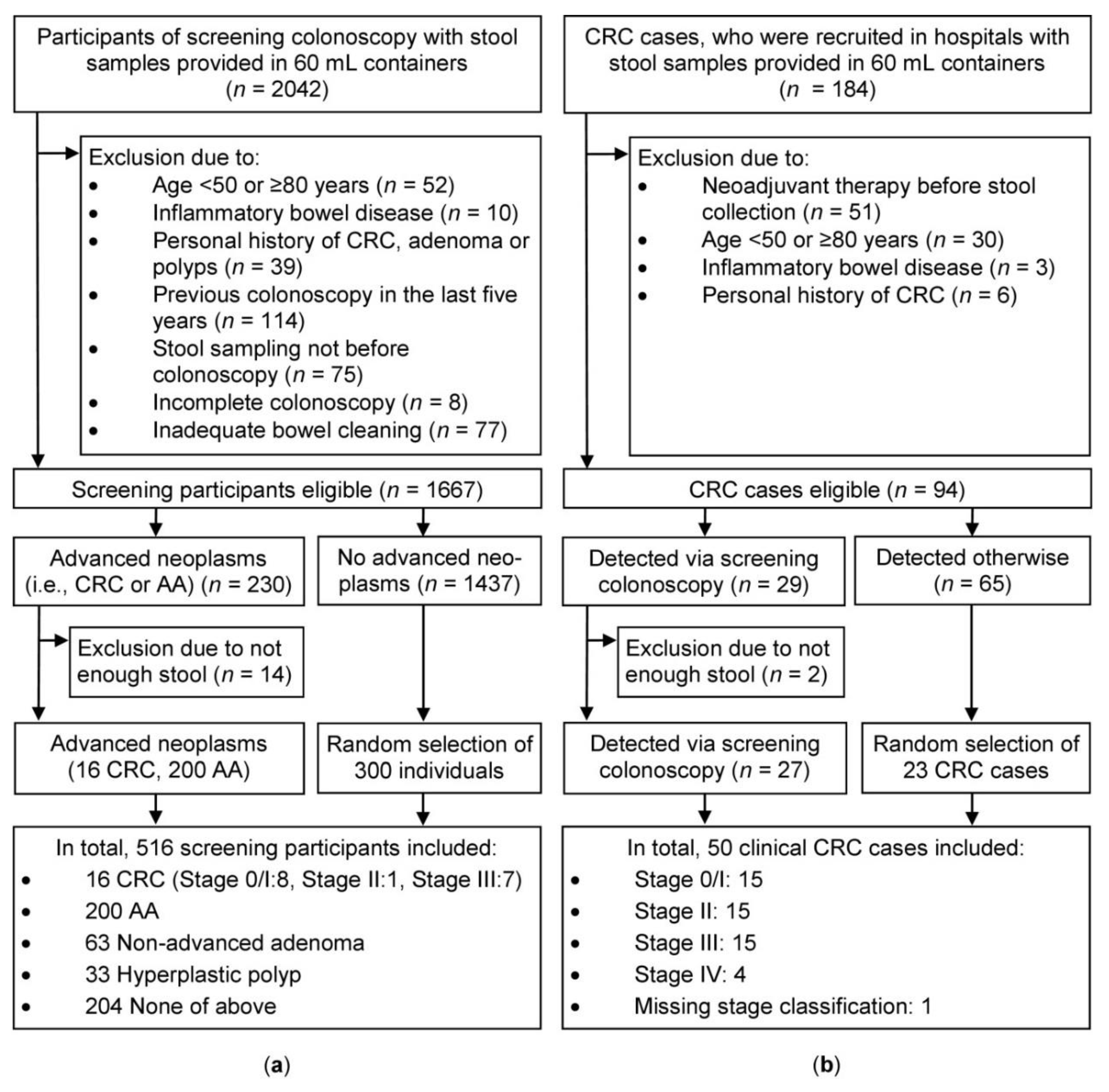
2.1. Study Population
2.2. Sensitivity and Specificity
2.3. Area Under the Curve (AUC)
2.4. Correlation Analyses
2.4.1. Spearman Correlation between Test Measurements
2.4.2. Cohen’s Kappa for Agreement of Test Classification
3. Discussion
4. Materials and Methods
4.1. Study Design and Study Population
4.2. Selection of Study Participants
4.3. Sample and Data Collection
4.4. Fecal Immunochemical Test Analysis
- CARE diagnostica GmbH, Voerde, Germany (CAREprime Hb and immoCARE-C)
- Immundiagnostik AG, Bensheim, Germany (IDK Hb ELISA and QuantOn Hem)
- R‑Biopharm AG, Darmstadt, Germany (RIDASCREEN Hb).
4.5. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Hewitson, P.; Glasziou, P.; Watson, E.; Towler, B.; Irwig, L. Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): An update. Am. J. Gastroenterol. 2008, 103, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Scholefield, J.H.; Moss, S.M.; Mangham, C.M.; Whynes, D.K.; Hardcastle, J.D. Nottingham trial of faecal occult blood testing for colorectal cancer: A 20-year follow-up. Gut 2012, 61, 1036–1040. [Google Scholar] [CrossRef]
- Shaukat, A.; Mongin, S.J.; Geisser, M.S.; Lederle, F.A.; Bond, J.H.; Mandel, J.S.; Church, T.R. Long-term mortality after screening for colorectal cancer. N. Engl. J. Med. 2013, 369, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Halloran, S.P.; Launoy, G.; Zappa, M.; International Agency for Research on Cancer. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition—faecal occult blood testing. Endoscopy 2012, 44, SE65–SE87. [Google Scholar] [PubMed]
- US Preventive Services Task Force. Screening for colorectal cancer: US preventive services task force recommendation statement. JAMA 2016, 315, 2564–2575. [Google Scholar] [CrossRef]
- Wolf, A.M.D.; Fontham, E.T.H.; Church, T.R.; Flowers, C.R.; Guerra, C.E.; LaMonte, S.J.; Etzioni, R.; McKenna, M.T.; Oeffinger, K.C.; Shih, Y.T.; et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J. Clin. 2018, 68, 250–281. [Google Scholar] [CrossRef]
- Benard, F.; Barkun, A.N.; Martel, M.; von Renteln, D. Systematic review of colorectal cancer screening guidelines for average-risk adults: Summarizing the current global recommendations. World J. Gastroenterol. 2018, 24, 124–138. [Google Scholar] [CrossRef]
- Schreuders, E.H.; Ruco, A.; Rabeneck, L.; Schoen, R.E.; Sung, J.J.; Young, G.P.; Kuipers, E.J. Colorectal cancer screening: A global overview of existing programmes. Gut 2015, 64, 1637–1649. [Google Scholar] [CrossRef]
- Zhu, M.M.; Xu, X.T.; Nie, F.; Tong, J.L.; Xiao, S.D.; Ran, Z.H. Comparison of immunochemical and guaiac-based fecal occult blood test in screening and surveillance for advanced colorectal neoplasms: A meta-analysis. J. Dig. Dis. 2010, 11, 148–160. [Google Scholar] [CrossRef]
- Park, D.I.; Ryu, S.; Kim, Y.H.; Lee, S.H.; Lee, C.K.; Eun, C.S.; Han, D.S. Comparison of guaiac-based and quantitative immunochemical fecal occult blood testing in a population at average risk undergoing colorectal cancer screening. Am. J. Gastroenterol. 2010, 105, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H.; Tao, S. Superior diagnostic performance of faecal immunochemical tests for haemoglobin in a head-to-head comparison with guaiac based faecal occult blood test among 2235 participants of screening colonoscopy. Eur. J. Cancer 2013, 49, 3049–3054. [Google Scholar] [CrossRef] [PubMed]
- Hol, L.; van Leerdam, M.E.; van Ballegooijen, M.; van Vuuren, A.J.; van Dekken, H.; Reijerink, J.C.; van der Togt, A.C.; Habbema, J.D.; Kuipers, E.J. Screening for colorectal cancer: Randomised trial comparing guaiac-based and immunochemical faecal occult blood testing and flexible sigmoidoscopy. Gut 2010, 59, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Vart, G.; Banzi, R.; Minozzi, S. Comparing participation rates between immunochemical and guaiac faecal occult blood tests: A systematic review and meta-analysis. Prev. Med. 2012, 55, 87–92. [Google Scholar] [CrossRef]
- Gies, A.; Bhardwaj, M.; Stock, C.; Schrotz-King, P.; Brenner, H. Quantitative fecal immunochemical tests for colorectal cancer screening. Int. J. Cancer 2018, 143, 234–244. [Google Scholar] [CrossRef]
- Oort, F.A.; van Turenhout, S.T.; Coupe, V.M.; van der Hulst, R.W.; Wesdorp, E.I.; Terhaar sive Droste, J.S.; Larbi, I.B.; Kanis, S.L.; van Hengel, E.; Bouman, A.A.; et al. Double sampling of a faecal immunochemical test is not superior to single sampling for detection of colorectal neoplasia: A colonoscopy controlled prospective cohort study. BMC Cancer 2011, 11, 434. [Google Scholar] [CrossRef]
- Hernandez, V.; Cubiella, J.; Gonzalez-Mao, M.C.; Iglesias, F.; Rivera, C.; Iglesias, M.B.; Cid, L.; Castro, I.; de Castro, L.; Vega, P.; et al. Fecal immunochemical test accuracy in average-risk colorectal cancer screening. World J. Gastroenterol. 2014, 20, 1038–1047. [Google Scholar] [CrossRef]
- Liles, E.G.; Perrin, N.; Rosales, A.G.; Smith, D.H.; Feldstein, A.C.; Mosen, D.M.; Levin, T.R. Performance of a quantitative fecal immunochemical test for detecting advanced colorectal neoplasia: A prospective cohort study. BMC Cancer 2018, 18, 509. [Google Scholar] [CrossRef]
- Guittet, L.; Guillaume, E.; Levillain, R.; Beley, P.; Tichet, J.; Lantieri, O.; Launoy, G. Analytical comparison of three quantitative immunochemical fecal occult blood tests for colorectal cancer screening. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1492–1501. [Google Scholar] [CrossRef]
- Lee, J.K.; Liles, E.G.; Bent, S.; Levin, T.R.; Corley, D.A. Accuracy of fecal immunochemical tests for colorectal cancer: Systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 171. [Google Scholar] [CrossRef]
- Daly, J.M.; Xu, Y.; Levy, B.T. Which fecal immunochemical test should i choose? J. Prim. Care Commun. Health 2017, 8, 264–277. [Google Scholar] [CrossRef] [PubMed]
- Gies, A.; Cuk, K.; Schrotz-King, P.; Brenner, H. Direct comparison of diagnostic performance of 9 quantitative fecal immunochemical tests for colorectal cancer screening. Gastroenterology 2018, 154, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Khalid-de Bakker, C.A.; Jonkers, D.M.; Sanduleanu, S.; de Bruine, A.P.; Meijer, G.A.; Janssen, J.B.; van Engeland, M.; Stockbrugger, R.W.; Masclee, A.A. Test performance of immunologic fecal occult blood testing and sigmoidoscopy compared with primary colonoscopy screening for colorectal advanced adenomas. Cancer Prev. Res. (Phila) 2011, 4, 1563–1571. [Google Scholar] [CrossRef] [PubMed]
- Stegeman, I.; de Wijkerslooth, T.R.; Stoop, E.M.; van Leerdam, M.; van Ballegooijen, M.; Kraaijenhagen, R.A.; Fockens, P.; Kuipers, E.J.; Dekker, E.; Bossuyt, P.M. Risk factors for false positive and for false negative test results in screening with fecal occult blood testing. Int. J. Cancer 2013, 133, 2408–2414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brenner, H.; Qian, J.; Werner, S. Variation of diagnostic performance of fecal immunochemical testing for hemoglobin by sex and age: Results from a large screening cohort. Clin. Epidemiol. 2018, 10, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H.; Haug, U.; Hundt, S. Sex differences in performance of fecal occult blood testing. Am. J. Gastroenterol. 2010, 105, 2457–2464. [Google Scholar] [CrossRef] [PubMed]
- Kapidzic, A.; van der Meulen, M.P.; Hol, L.; van Roon, A.H.; Looman, C.W.; Lansdorp-Vogelaar, I.; van Ballegooijen, M.; van Vuuren, A.J.; Reijerink, J.C.; van Leerdam, M.E.; et al. Gender differences in fecal immunochemical test performance for early detection of colorectal neoplasia. Clin. Gastroenterol. Hepatol. 2015, 13, 1464–1471. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Kato, J.; Yamaji, Y.; Wada, R.; Mitsushima, T.; Shiratori, Y. A comparison of the immunochemical fecal occult blood test and total colonoscopy in the asymptomatic population. Gastroenterology 2005, 129, 422–428. [Google Scholar] [CrossRef]
- Tao, S.; Seiler, C.M.; Ronellenfitsch, U.; Brenner, H. Comparative evaluation of nine faecal immunochemical tests for the detection of colorectal cancer. Acta Oncol. 2013, 52, 1667–1675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imperiale, T.F.; Ransohoff, D.F.; Itzkowitz, S.H.; Levin, T.R.; Lavin, P.; Lidgard, G.P.; Ahlquist, D.A.; Berger, B.M. Multitarget stool DNA testing for colorectal-cancer screening. N. Engl. J. Med. 2014, 370, 1287–1297. [Google Scholar] [CrossRef]
- Gies, A.; Cuk, K.; Schrotz-King, P.; Brenner, H. Direct comparison of ten quantitative fecal immunochemical tests for hemoglobin stability in colorectal cancer screening. Clin. Transl. Gastroenterol. 2018, 9, 168. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, R.E.; Kochwa, S.; Kaczera, Z.; Maimon, J. Nonuniform distribution of occult blood in feces. Am. J. Clin. Pathol. 1979, 71, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Werner, S.; Brenner, H. Fresh vs frozen samples and ambient temperature have little effect on detection of colorectal cancer or adenomas by a fecal immunochemical test in a colorectal cancer screening cohort in Germany. Clin. Gastroenterol. Hepatol. 2017, 15, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Bossuyt, P.M.; Reitsma, J.B.; Bruns, D.E.; Gatsonis, C.A.; Glasziou, P.P.; Irwig, L.; Lijmer, J.G.; Moher, D.; Rennie, D.; de Vet, H.C.; et al. STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ 2015, 351, h5527. [Google Scholar] [CrossRef] [PubMed]
- Fraser, C.G.; Allison, J.E.; Young, G.P.; Halloran, S.P.; Seaman, H.E. Improving the reporting of evaluations of faecal immunochemical tests for haemoglobin: The FITTER standard and checklist. Eur. J. Cancer Prev. 2015, 24, 24–26. [Google Scholar] [CrossRef]
- Fraser, C.G.; Allison, J.E.; Halloran, S.P.; Young, G.P.; on behalf of the Expert Working Group on Fecal Immunochemical Tests for Hemoglobin, Colorectal Cancer Screening Committee, World Endoscopy Organization. A proposal to standardize reporting units for fecal immunochemical tests for hemoglobin. J. Natl. Cancer Inst. 2012, 104, 810–814. [Google Scholar] [CrossRef]
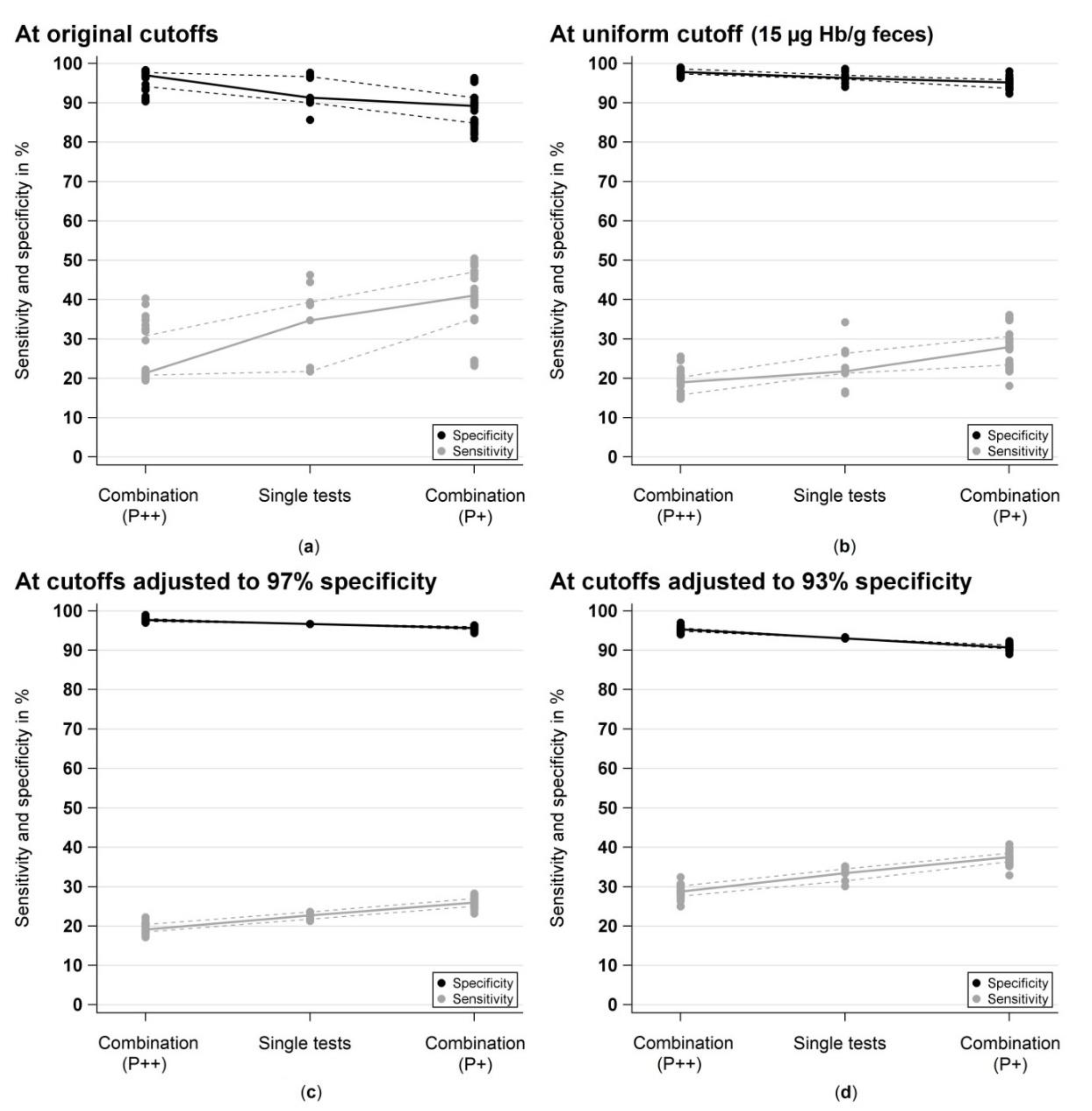
| Cutoff | Test(s) | Metric | Participants of Screening Colonoscopy (n = 516) | Clinical CRC Cases | |||
|---|---|---|---|---|---|---|---|
| Sensitivity (%) | Specificity (%) | Sensitivity (%) | |||||
| CRC (n = 16) | AA (n = 200) | AN (n = 216) | No AN (n = 300) | CRC (n = 50) | |||
| Original cutoff (range: 2–17 µg/g) | Single tests (n = 9) | Median | 81.3 | 31.0 | 34.7 | 91.3 | 74.0 |
| Range | 62.5 to 81.3 | 18.0 to 43.5 | 21.8 to 46.3 | 85.7 to 97.7 | 64.0 to 84.0 | ||
| Combinations (n = 36) | Median | 68.8 | 18.0 | 21.4 | 97.0 | 68.0 | |
| Range | 62.5 to 81.3 | 16.0 to 37.5 | 19.4 to 40.3 | 90.3 to 98.3 | 62.0 to 80.0 | ||
| Difference (n = 36) † | Median | −12.5 | −14.0 | −13.7 | 0.7 | −8.0 | |
| Range | −18.8 to 0.0 | −26.5 to −1.5 | −25.5 to −1.4 | 0.0 to 4.7 | −22.0 to −2.0 | ||
| Uniform Cutoff ( = 15 µg/g) | Single tests (n = 9) | Median | 68.8 | 18.5 | 21.8 | 96.3 | 70.0 |
| Range | 56.3 to 81.3 | 13.0 to 30.5 | 16.2 to 34.3 | 94.0 to 98.7 | 56.0 to 76.0 | ||
| Combinations (n = 36) | Median | 62.5 | 15.0 | 19.0 | 97.8 | 68.0 | |
| Range | 56.3 to 75.0 | 11.5 to 21.6 | 14.8 to 25.6 | 96.3 to 99.0 | 52.0 to 72.0 | ||
| Difference (n = 36) † | Median | −9.4 | −7.0 | −7.0 | 0.7 | −6.0 | |
| Range | −25.0 to 0.0 | −18.0 to −1.5 | −18.5 to −1.9 | 0.0 to 1.7 | −20.0 to −0.0 | ||
| Cutoffs adjusted to 96.7% specificity (range: 6–30 µg/g) | Single tests (n = 9) | Median | 68.8 | 19.0 | 22.7 | 96.7 | 68.0 |
| Range | 62.5 to 75.0 | 17.5 to 20.1 | 21.3 to 23.6 | 96.7 to 96.7 | 64.0 to 74.0 | ||
| Combinations (n = 36) | Median | 62.5 | 15.5 | 19.1 | 97.7 | 66.0 | |
| Range | 62.5 to 68.8 | 13.5 to 18.5 | 17.1 to 22.2 | 97.0 to 99.0 | 62.0 to 70.0 | ||
| Difference (n = 36) † | Median | −6.3 | -4.0 | −4.2 | 1.0 | −4.0 | |
| Range | −12.5 to 0.0 | −6.0 to −1.5 | −5.6 to −1.4 | 0.3 to 2.3 | −12.0 to 0.0 | ||
| Cutoffs adjusted to 93.0% specificity (range: 2–13 µg/g) ‡ | Single tests (n = 8) | Median | 78.1 | 29.6 | 33.4 | 93.0 | 74.0 |
| Range | 68.8 to 81.3 | 26.5 to 31.5 | 30.1 to 35.2 | 93.0 to 93.3 § | 72.0 to 80.0 | ||
| Combinations (n = 28) | Median | 75.0 | 25.3 | 28.8 | 95.3 | 71.0 | |
| Range | 68.8 to 81.3 | 21.0 to 28.5 | 25.0 to 32.4 | 94.0 to 97.0 | 70.0 to 74.0 | ||
| Difference (n = 28) † | Median | −6.3 | −5.0 | −5.3 | 2.3 | −4.0 | |
| Range | −12.5 to 0.0 | −9.5 to −3.0 | −9.3 to −2.8 | 0.7 to 4.0 | −10.0 to 0.0 | ||
| Cutoff | Test(s) | Metric | Participants of Screening Colonscopy (n = 516) | Clinical CRC Cases | |||
|---|---|---|---|---|---|---|---|
| Sensitivity (%) | Specificity (%) | Sensitivity (%) | |||||
| CRC (n = 16) | AA (n = 200) | AN (n = 216) | No AN (n = 300) | CRC (n = 50) | |||
| Original cutoff (range: 2–17 µg/g) | Single tests (n = 9) | Median | 81.3 | 31.0 | 34.7 | 91.3 | 74.0 |
| Range | 62.5 to 81.3 | 18.0 to 43.5 | 21.8 to 46.3 | 85.7 to 97.7 | 64.0 to 84.0 | ||
| Combinations (n = 36) | Median | 81.3 | 37.8 | 41.1 | 89.2 | 78.0 | |
| Range | 62.5 to 87.5 | 19.5 to 47.5 | 23.2 to 50.5 | 81.0 to 96.3 | 68.0 to 88.0 | ||
| Difference (n = 36) † | Median | 0.0 | 1.0 | 1.2 | −6.7 | 0.0 | |
| Range | 0.0 to 6.3 | 0.0 to 5.0 | 0.0 to 5.1 | −12.7 to −1.3 | 0.0 to 4.0 | ||
| Uniform Cutoff (= 15 µg/g) | Single tests (n = 9) | Median | 68.8 | 18.5 | 21.8 | 96.3 | 70.0 |
| Range | 56.3 to 81.3 | 13.0 to 30.5 | 16.2 to 34.3 | 94.0 to 98.7 | 56.0 to 76.0 | ||
| Combinations (n = 36) | Median | 75.0 | 24.1 | 27.9 | 95.2 | 72.0 | |
| Range | 56.3 to 81.3 | 15.0 to 32.5 | 18.1 to 36.1 | 92.3 to 98.0 | 68.0 to 78.0 | ||
| Difference (n = 36) † | Median | 0.0 | 1.5 | 1.4 | −2.3 | 0.0 | |
| Range | 0.0 to 6.3 | 0.0 to 4.0 | 0.0 to 4.2 | −4.7 to −0.7 | 0.0 to 4.0 | ||
| Cutoffs adjusted to 96.7% specificity (range: 6–30 µg/g) | Single tests (n = 9) | Median | 68.8 | 19.0 | 22.7 | 96.7 | 68.0 |
| Range | 62.5 to 75.0 | 17.5 to 20.1 | 21.3 to 23.6 | 96.7 to 96.7 | 64.0 to 74.0 | ||
| Combinations (n = 36) | Median | 68.8 | 22.5 | 25.9 | 95.7 | 70.0 | |
| Range | 62.5 to 75.0 | 19.5 to 24.6 | 23.2 to 28.2 | 94.3 to 96.3 | 68.0 to 76.0 | ||
| Difference (n = 36) † | Median | 0.0 | 3.0 | 2.8 | −1.0 | 0.0 | |
| Range | 0.0 to 0.0 | 1.0 to 5.0 | 0.9 to 4.7 | −2.3 to −0.3 | 0.0 to 2.0 | ||
| Cutoffs adjusted to 93.0% specificity (range: 2–13 µg/g) ‡ | Single tests (n = 8) | Median | 78.1 | 29.6 | 33.4 | 93.0 | 74.0 |
| Range | 68.8 to 81.3 | 26.5 to 31.5 | 30.1 to 35.2 | 93.0 to 93.3 § | 72.0 to 80.0 | ||
| Combinations (n = 28) | Median | 81.3 | 34.0 | 37.5 | 90.7 | 76.0 | |
| Range | 75.0 to 81.3 | 29.5 to 37.5 | 32.9 to 40.7 | 89.0 to 92.3 | 74.0 to 84.0 | ||
| Difference (n = 28) † | Median | 0.0 | 3.2 | 3.0 | −2.3 | 2.0 | |
| Range | 0.0 to 6.3 | 1.0 to 7.0 | 1.4 to 6.5 | −4.0 to −1.0 | 0.0 to 4.0 | ||
| FIT Brand | Ridascreen Hb | QuantOn Hem | immoCARE‑C † | CAREprime Hb | Eurolyser FOB test | OC Sensor | QuikRead go iFOBT | SENTiFIT-FOB Gold |
|---|---|---|---|---|---|---|---|---|
| IDK Hb ELISA | CRC 0.90 | CRC 0.89 | CRC 0.84 | CRC 0.83 | CRC 0.91 | CRC 0.85 | CRC 0.92 | CRC 0.83 |
| AA 0.95 | AA 0.89 | AA 0.87 | AA 0.80 | AA 0.74 | AA 0.81 | AA 0.62 | AA 0.75 | |
| No AN 0.81 | No AN 0.62 | No AN 0.50 | No AN 0.48 | No AN 0.26 | No AN 0.44 | No AN 0.28 | No AN 0.31 | |
| Total 0.90 | Total 0.78 | Total 0.74 | Total 0.68 | Total 0.58 | Total 0.66 | Total 0.51 | Total 0.59 | |
| Ridascreen Hb | CRC 0.89 | CRC 0.90 | CRC 0.90 | CRC 0.90 | CRC 0.90 | CRC 0.88 | CRC 0.89 | |
| AA 0.85 | AA 0.87 | AA 0.83 | AA 0.77 | AA 0.81 | AA 0.61 | AA 0.76 | ||
| No AN 0.62 | No AN 0.57 | No AN 0.52 | No AN 0.32 | No AN 0.50 | No AN 0.34 | No AN 0.40 | ||
| Total 0.78 | Total 0.77 | Total 0.72 | Total 0.64 | Total 0.70 | Total 0.55 | Total 0.65 | ||
| QuantOn Hem | CRC 0.91 | CRC 0.91 | CRC 0.89 | CRC 0.89 | CRC 0.93 | CRC 0.92 | ||
| AA 0.77 | AA 0.72 | AA 0.68 | AA 0.71 | AA 0.60 | AA 0.69 | |||
| No AN 0.38 | No AN 0.34 | No AN 0.24 | No AN 0.22 | No AN 0.27 | No AN 0.28 | |||
| Total 0.63 | Total 0.58 | Total 0.56 | Total 0.53 | Total 0.52 | Total 0.57 | |||
| immoCARE‑C † | CRC 0.92 | CRC 0.89 | CRC 0.83 | CRC 0.88 | CRC 0.89 | |||
| AA 0.80 | AA 0.79 | AA 0.77 | AA 0.63 | AA 0.76 | ||||
| No AN 0.47 | No AN 0.41 | No AN 0.51 | No AN 0.32 | No AN 0.45 | ||||
| Total 0.68 | Total 0.67 | Total 0.68 | Total 0.54 | Total 0.65 | ||||
| CAREprime Hb | CRC 0.81 | CRC 0.96 | CRC 0.89 | CRC 0.98 | ||||
| AA 0.79 | AA 0.82 | AA 0.66 | AA 0.77 | |||||
| No AN 0.40 | No AN 0.54 | No AN 0.31 | No AN 0.43 | |||||
| Total 0.65 | Total 0.71 | Total 0.53 | Total 0.65 | |||||
| Eurolyser FOB test | CRC 0.83 | CRC 0.85 | CRC 0.82 | |||||
| AA 0.81 | AA 0.80 | AA 0.92 | ||||||
| No AN 0.45 | No AN 0.54 | No AN 0.79 | ||||||
| Total 0.69 | Total 0.75 | Total 0.89 | ||||||
| OC Sensor | CRC 0.87 | CRC 0.96 | ||||||
| AA 0.67 | AA 0.80 | |||||||
| No AN 0.35 | No AN 0.48 | |||||||
| Total 0.57 | Total 0.69 | |||||||
| QuikRead go iFOBT | CRC 0.92 | |||||||
| AA 0.80 | ||||||||
| No AN 0.57 | ||||||||
| Total 0.77 |
| FIT Brand | IDK Hb ELISA | Ridascreen Hb | QuantOn Hem | immoCARE‑C † | CAREprime Hb | Eurolyser FOB Test | OC Sensor | QuikRead go iFOBT | SENTiFIT-FOB Gold |
|---|---|---|---|---|---|---|---|---|---|
| IDK Hb ELISA | CRC 1.00 | CRC 0.59 | CRC 1.00 | CRC 1.00 | CRC 0.56 | CRC 0.67 | CRC 0.56 | CRC 0.67 | |
| AA 0.82 | AA 0.80 | AA 0.71 | AA 0.63 | AA 0.44 | AA 0.42 | AA 0.41 | AA 0.40 | ||
| No AN 0.76 | No AN 0.62 | No AN 0.71 | No AN 0.53 | No AN 0.19 | No AN 0.25 | No AN 0.30 | No AN 0.29 | ||
| Total 0.83 | Total 0.76 | Total 0.76 | Total 0.66 | Total 0.44 | Total 0.46 | Total 0.45 | Total 0.45 | ||
| Ridascreen Hb | CRC 0.86 | CRC 0.59 | CRC 1.00 | CRC 1.00 | CRC 0.56 | CRC 0.67 | CRC 0.56 | CRC 0.67 | |
| AA 0.82 | AA 0.70 | AA 0.81 | AA 0.75 | AA 0.53 | AA 0.54 | AA 0.48 | AA 0.51 | ||
| No AN 0.90 | No AN 0.51 | No AN 0.85 | No AN 0.72 | No AN 0.29 | No AN 0.38 | No AN 0.45 | No AN 0.43 | ||
| Total 0.86 | Total 0.68 | Total 0.86 | Total 0.79 | Total 0.54 | Total 0.57 | Total 0.54 | Total 0.57 | ||
| QuantOn Hem | CRC 0.85 | CRC 0.71 | CRC 0.59 | CRC 0.59 | CRC 0.56 | CRC 0.67 | CRC 0.56 | CRC 0.67 | |
| AA 0.76 | AA 0.79 | AA 0.66 | AA 0.56 | AA 0.42 | AA 0.43 | AA 0.44 | AA 0.43 | ||
| No AN 0.69 | No AN 0.59 | No AN 0.46 | No AN 0.40 | No AN 0.15 | No AN 0.25 | No AN 0.26 | No AN 0.25 | ||
| Total 0.79 | Total 0.78 | Total 0.64 | Total 0.57 | Total 0.41 | Total 0.46 | Total 0.46 | Total 0.45 | ||
| immoCARE‑C † | CRC 0.86 | CRC 1.00 | CRC 0.71 | CRC 1.00 | CRC 0.56 | CRC 0.67 | CRC 0.56 | CRC 0.67 | |
| AA 0.80 | AA 0.71 | AA 0.69 | AA 0.80 | AA 0.57 | AA 0.58 | AA 0.54 | AA 0.55 | ||
| No AN 0.79 | No AN 0.69 | No AN 0.48 | No AN 0.69 | No AN 0.33 | No AN 0.35 | No AN 0.42 | No AN 0.41 | ||
| Total 0.83 | Total 0.77 | Total 0.71 | Total 0.80 | Total 0.57 | Total 0.59 | Total 0.57 | Total 0.58 | ||
| CAREprime Hb | CRC 1.00 | CRC 0.86 | CRC 0.85 | CRC 0.86 | CRC 0.56 | CRC 0.67 | CRC 0.56 | CRC 0.67 | |
| AA 0.85 | AA 0.78 | AA 0.73 | AA 0.78 | AA 0.67 | AA 0.66 | AA 0.65 | AA 0.66 | ||
| No AN 0.79 | No AN 0.79 | No AN 0.48 | No AN 0.69 | No AN 0.49 | No AN 0.40 | No AN 0.53 | No AN 0.57 | ||
| Total 0.87 | Total 0.82 | Total 0.74 | Total 0.80 | Total 0.67 | Total 0.65 | Total 0.66 | Total 0.68 | ||
| Eurolyser FOB test | CRC 1.00 | CRC 0.86 | CRC 0.85 | CRC 0.86 | CRC 1.00 | CRC 0.86 | CRC 1.00 | CRC 0.86 | |
| AA 0.79 | AA 0.75 | AA 0.66 | AA 0.72 | AA 0.91 | AA 0.89 | AA 0.84 | AA 0.85 | ||
| No AN 0.59 | No AN 0.59 | No AN 0.28 | No AN 0.69 | No AN 0.69 | No AN 0.61 | No AN 0.51 | No AN 0.69 | ||
| Total 0.80 | Total 0.76 | Total 0.66 | Total 0.77 | Total 0.89 | Total 0.86 | Total 0.81 | Total 0.84 | ||
| OC Sensor | CRC 1.00 | CRC 0.86 | CRC 0.85 | CRC 0.86 | CRC 1.00 | CRC 1.00 | CRC 0.86 | CRC 1.00 | |
| AA 0.82 | AA 0.78 | AA 0.69 | AA 0.72 | AA 0.88 | AA 0.81 | AA 0.85 | AA 0.86 | ||
| No AN 0.79 | No AN 0.90 | No AN 0.48 | No AN 0.59 | No AN 0.90 | No AN 0.59 | No AN 0.58 | No AN 0.66 | ||
| Total 0.86 | Total 0.84 | Total 0.72 | Total 0.75 | Total 0.91 | Total 0.81 | Total 0.83 | Total 0.86 | ||
| QuikRead go iFOBT | CRC 0.86 | CRC 1.00 | CRC 0.71 | CRC 1.00 | CRC 0.86 | CRC 0.86 | CRC 0.86 | CRC 0.86 | |
| AA 0.80 | AA 0.72 | AA 0.67 | AA 0.69 | AA 0.86 | AA 0.86 | AA 0.79 | AA 0.92 | ||
| No AN 0.69 | No AN 0.59 | No AN 0.59 | No AN 0.59 | No AN 0.79 | No AN 0.48 | No AN 0.69 | No AN 0.65 | ||
| Total 0.81 | Total 0.76 | Total 0.71 | Total 0.74 | Total 0.87 | Total 0.81 | Total 0.81 | Total 0.87 | ||
| SENTiFIT- FOB Gold | CRC 1.00 | CRC 0.86 | CRC 0.85 | CRC 0.86 | CRC 1.00 | CRC 1.00 | CRC 1.00 | CRC 0.86 | |
| AA 0.81 | AA 0.73 | AA 0.68 | AA 0.67 | AA 0.87 | AA 0.87 | AA 0.84 | AA 0.92 | ||
| No AN 0.69 | No AN 0.69 | No AN 0.38 | No AN 0.69 | No AN 0.90 | No AN 0.79 | No AN 0.79 | No AN 0.69 | ||
| Total 0.83 | Total 0.77 | Total 0.69 | Total 0.74 | Total 0.90 | Total 0.89 | Total 0.87 | Total 0.88 |
| FIT Brand | Manufacturer, City, Country | Fecal Sampling Tube (Fecal Mass/Buffer Volume) | Analytical Instrument | Analytical Reading Technique | Analytical Working Range (µg Hb/g feces) | Original Cutoff (µg Hb/g feces) |
|---|---|---|---|---|---|---|
| IDK Hb ELISA | Immundiagnostik, Bensheim, Germany | IDK Extract (15 mg/1.5 mL) | DSX by Dynex Technologies | Enzyme-linked immunosorbent assay (ELISA) | 0.086 to 50 | 2.00 |
| RIDASCREEN Hb | R-Biopharm, Darmstadt, Germany | RIDA TUBE Hb (10 mg/2.5 mL) | DSX by Dynex Technologies | Enzyme-linked immunosorbent assay (ELISA) | 0.65 to 50 | 8.00 |
| QuantOn Hem | Immundiagnostik, Bensheim, Germany | QuantOn Hem TUBE (15 mg/1.5 mL) | QuantOn Hem test cassette & Smartphone † | Immunoaffinity chromatography & Photometry | 0.3 to 100 | 3.70 |
| immoCARE-C | CARE diagnostica, Möllersdorf, Austria | Sample Collection Tube (20 mg/2.5 mL) | immoCARE‑C test cassette & CAREcube | Immunoaffinity chromatography & Photometry | 3.75 to 250 | 6.25 |
| CAREprime Hb | Alfresa Pharma, Osaka, Japan | Specimen Collection Container A (9.5 mg/1.9 mL) | CAREprime | Immunoturbidimetry | 1.6 to 240 | 6.30 |
| Eurolyser FOB test | Eurolyser Diagnostica, Salzburg, Austria | Eurolyser FOB Sample Collector (19.9 mg/1.6 mL) | Eurolyser CUBE | Immunoturbidimetry | 2.01 to 80.4 | 8.04 |
| OC Sensor | Eiken Chemical, Tokyo, Japan | OC Auto-Sampling Bottle 3 (10 mg/2.0 mL) | OC Sensor io | Immunoturbidimetry | 10 to 200 | 10.0 |
| QuikRead go iFOBT | Orion Diagnostica, Espoo, Finland | QuikRead go iFOBT Sampling Set (10 mg/2.0 mL) | QuikRead go | Immunoturbidimetry | 15 to 200 | 15.0 |
| SENTiFIT‑FOB Gold | Sentinel Diagnostics, Milan, Italy | SENTiFIT pierceTube (10 mg/1.7 mL) | SENTiFIT 270 analyzer | Immunoturbidimetry | 1.7 to 129.88 | 17.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gies, A.; Cuk, K.; Schrotz-King, P.; Brenner, H. Combination of Different Fecal Immunochemical Tests in Colorectal Cancer Screening: Any Gain in Diagnostic Performance? Cancers 2019, 11, 120. https://doi.org/10.3390/cancers11010120
Gies A, Cuk K, Schrotz-King P, Brenner H. Combination of Different Fecal Immunochemical Tests in Colorectal Cancer Screening: Any Gain in Diagnostic Performance? Cancers. 2019; 11(1):120. https://doi.org/10.3390/cancers11010120
Chicago/Turabian StyleGies, Anton, Katarina Cuk, Petra Schrotz-King, and Hermann Brenner. 2019. "Combination of Different Fecal Immunochemical Tests in Colorectal Cancer Screening: Any Gain in Diagnostic Performance?" Cancers 11, no. 1: 120. https://doi.org/10.3390/cancers11010120
APA StyleGies, A., Cuk, K., Schrotz-King, P., & Brenner, H. (2019). Combination of Different Fecal Immunochemical Tests in Colorectal Cancer Screening: Any Gain in Diagnostic Performance? Cancers, 11(1), 120. https://doi.org/10.3390/cancers11010120





