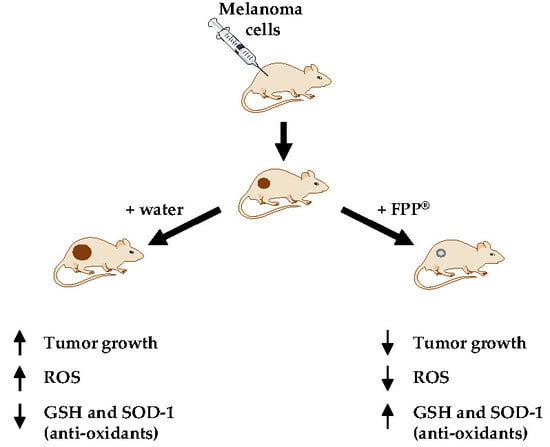
Oral Administration of Fermented Papaya (FPP®) Controls the Growth of a Murine Melanoma through the In Vivo Induction of a Natural Antioxidant Response
Abstract
1. Introduction
2. Results
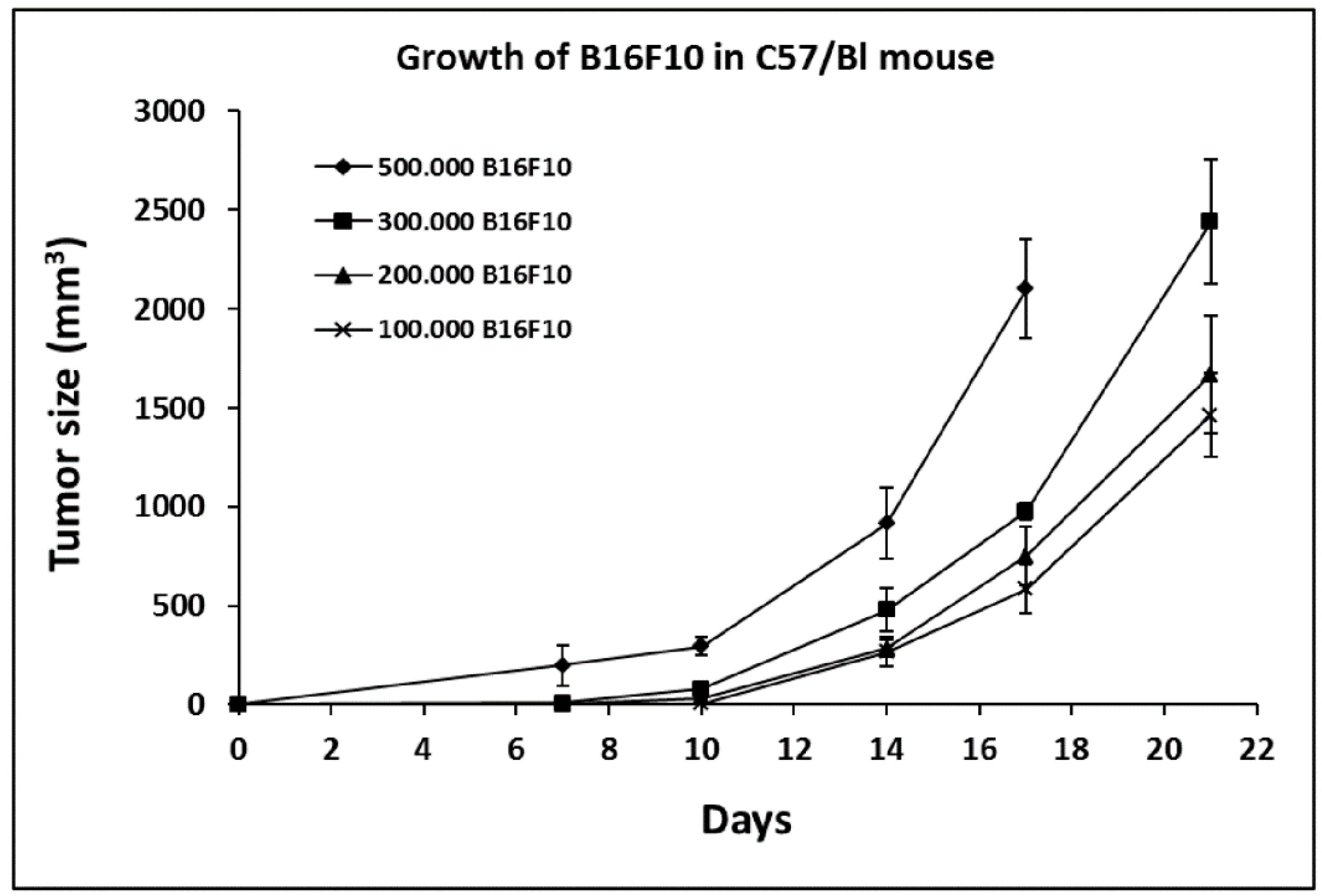
2.1. Growth Curve of B16F10 in C57/BL Mice
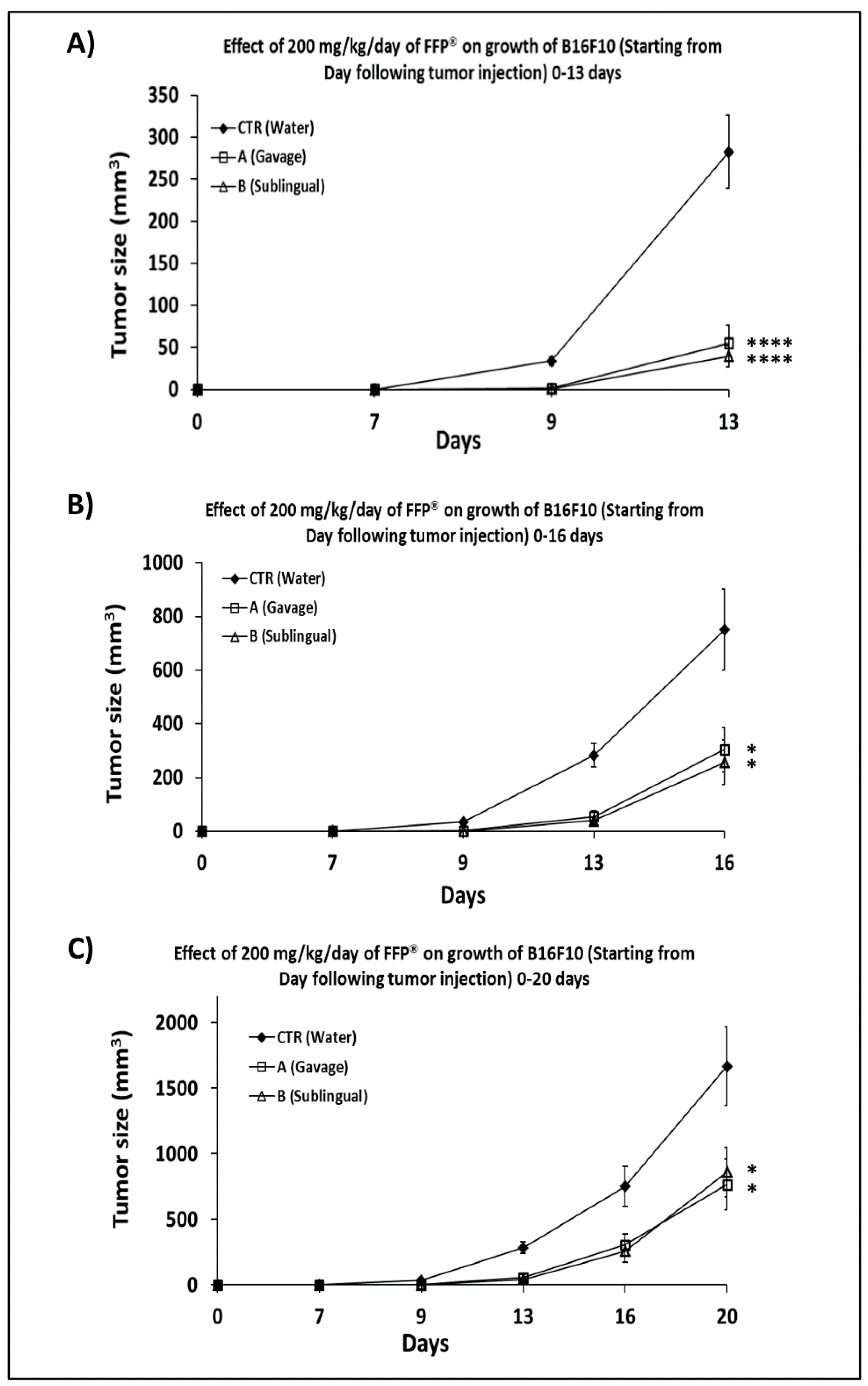
2.2. Dose-Response Curve in C57/Bl Mice Treated with FPP® by Oral Gavage
2.3. Comparison of the of FPP® Oral Gavage Versus Sublingual on the Growth of Melanoma in C57/BL Mice
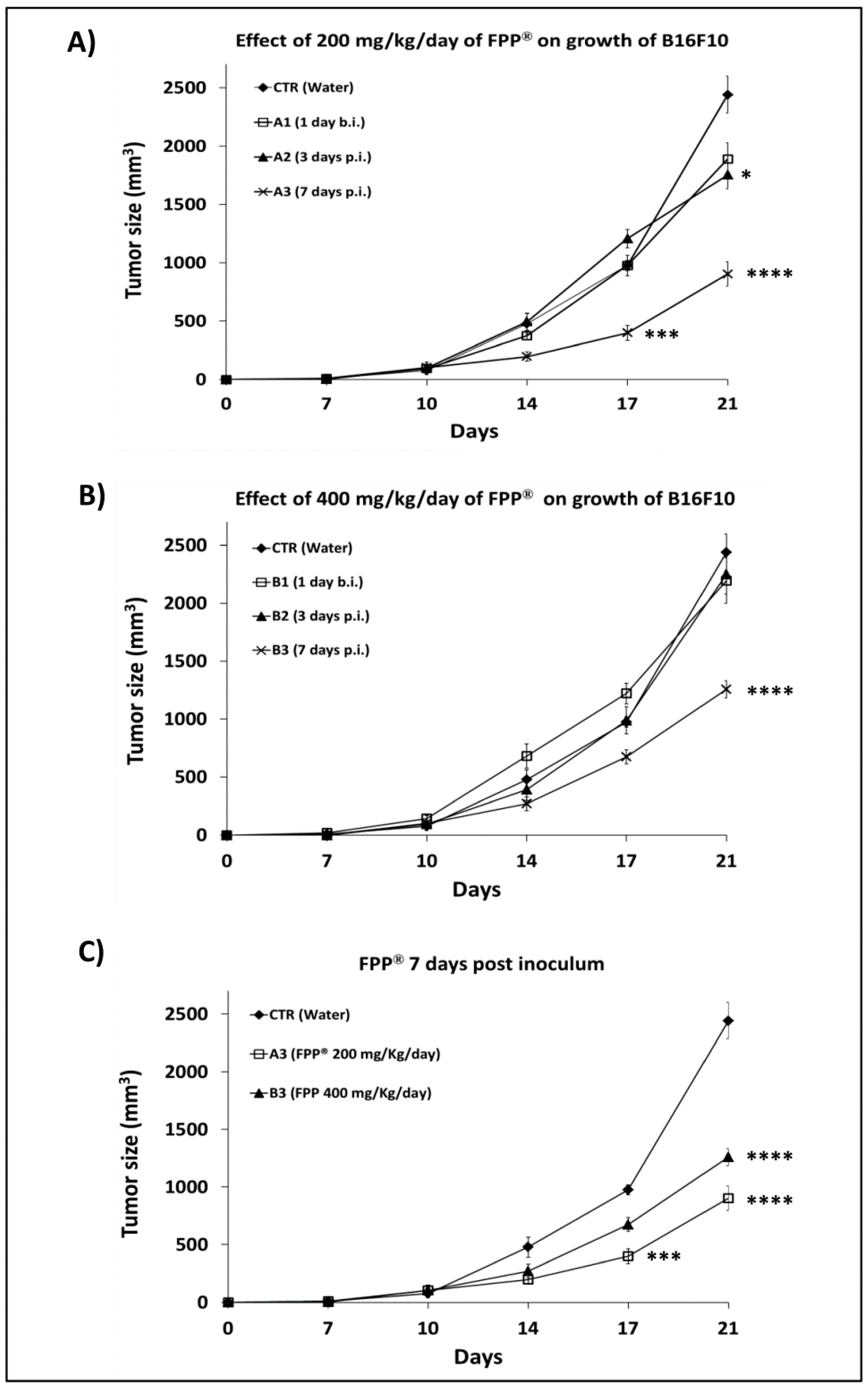
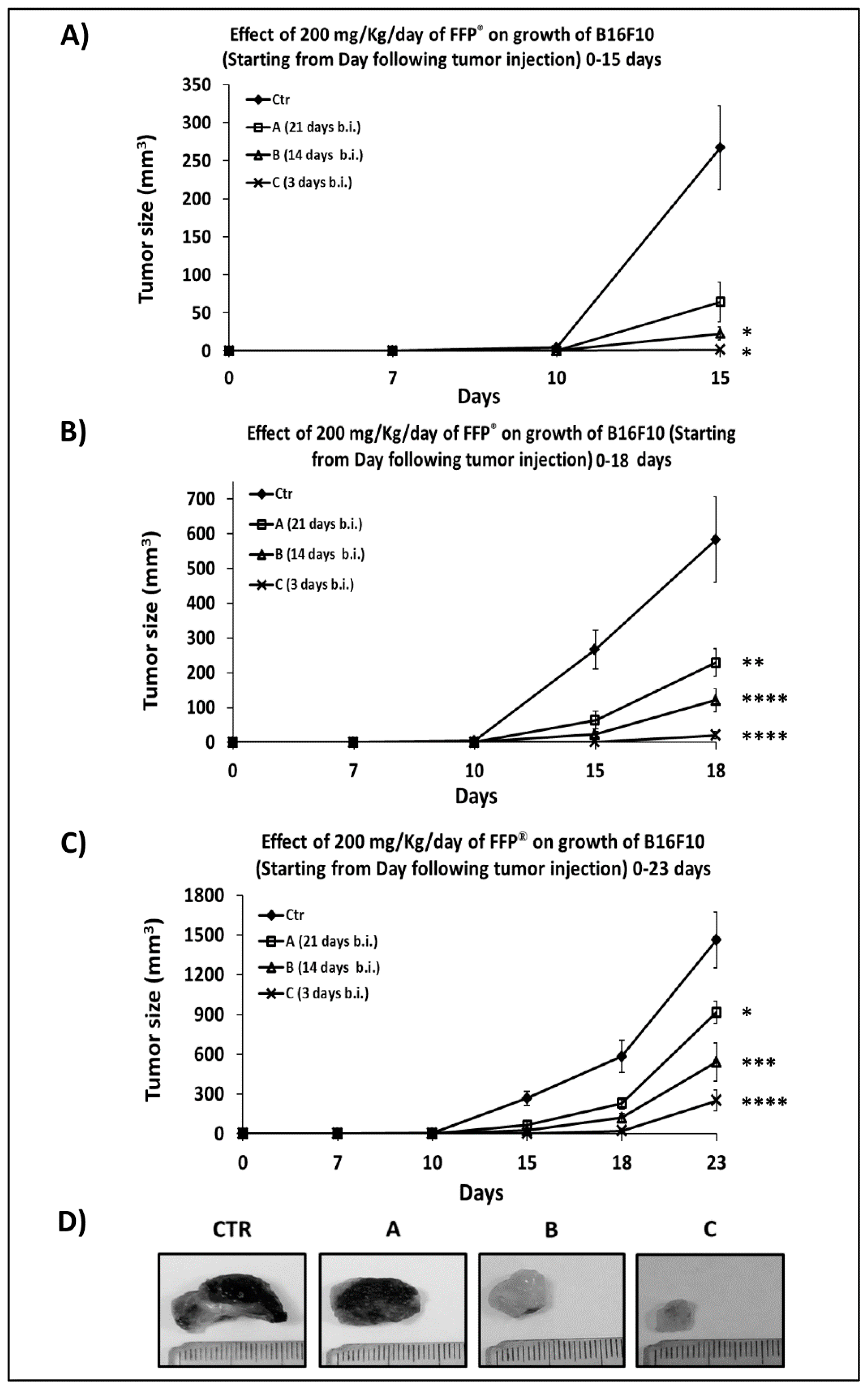
2.4. Combined Preventive and Therapeutic Effects of FPP® on Melanoma Growth
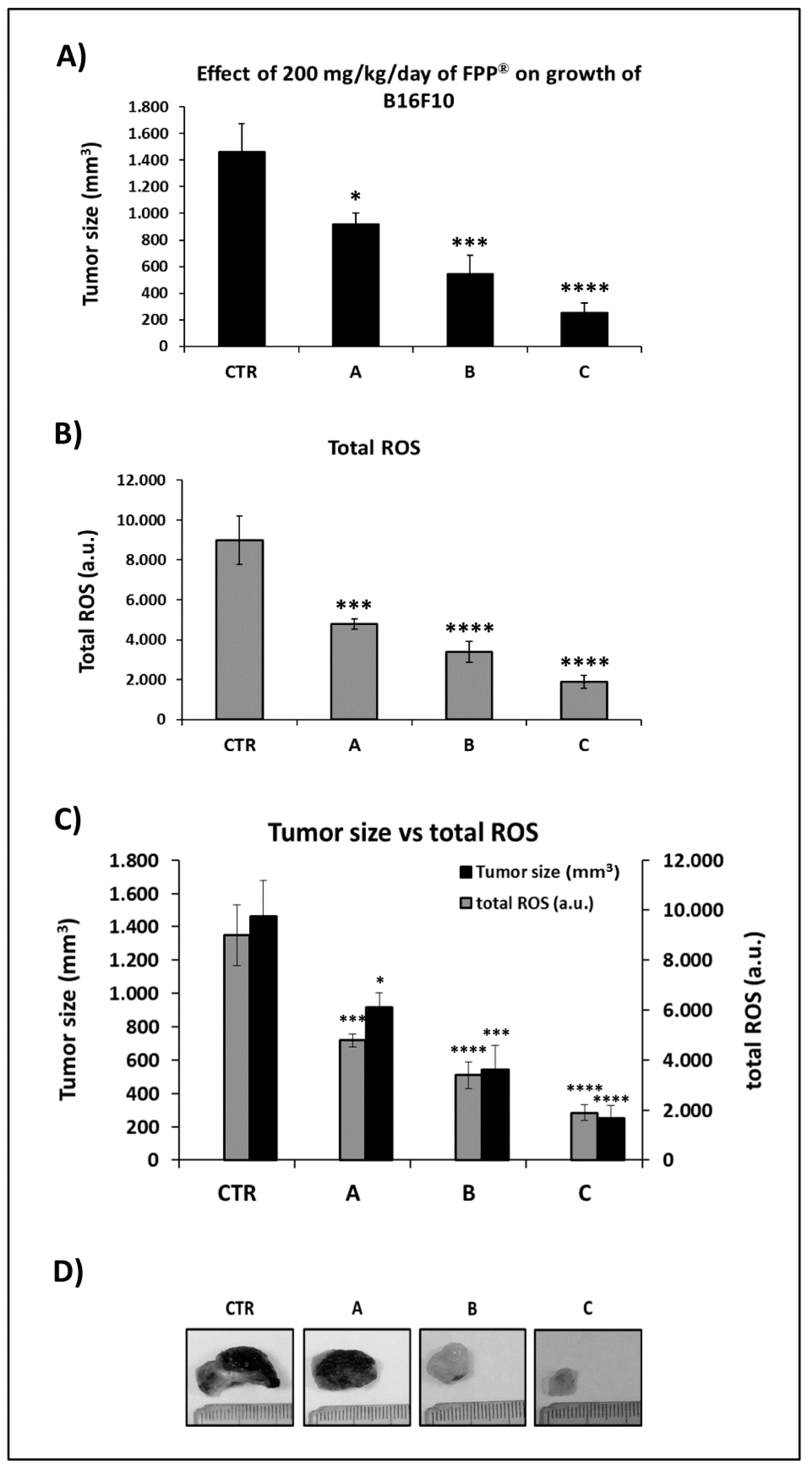
2.5. FPP® Treatment, Tumor Growth and Blood ROS Levels
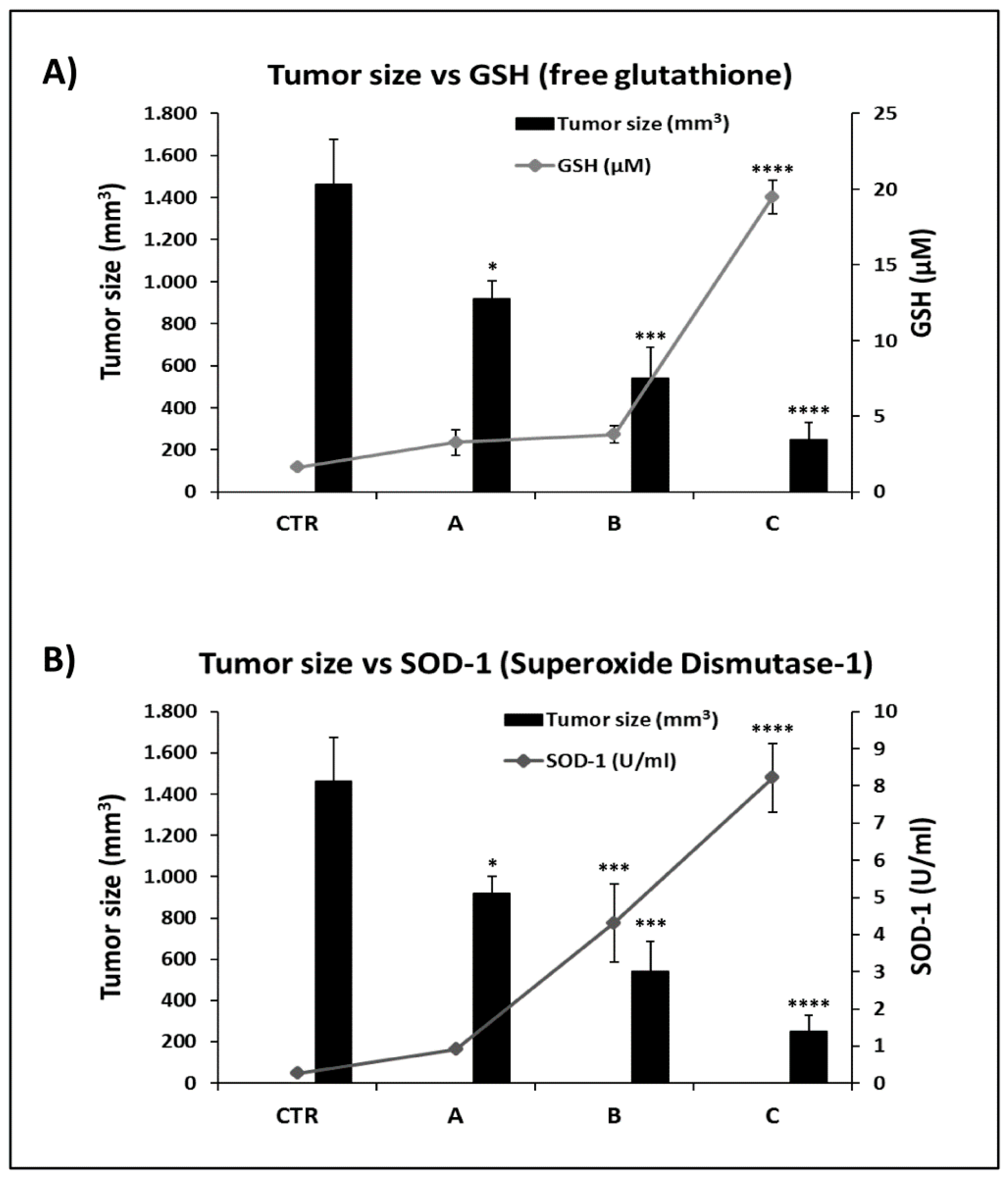
2.6. FPP® Treatment, Tumor Growth and Plasmatic Natural Antioxidant (GSH AND SOD-1) Levels
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Immun’Âge® - FPP® (Fermented Papaya Preparation)
4.3. In Vivo Studies
4.4. Total Ros Assay
4.5. SOD Activity Assay
4.6. GSH Activity Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Azzarito, T.; Lugini, L.; Spugnini, E.P.; Canese, R.; Gugliotta, A.; Fidanza, S.; Fais, S. Effect of Modified Alkaline Supplementation on Syngenic Melanoma Growth in CB57/BL Mice. PLoS ONE 2016, 11, e0159763. [Google Scholar] [CrossRef] [PubMed]
- Azzarito, T.; Venturi, G.; Cesolini, A.; Fais, S. Lansoprazole induces sensitivity to suboptimal doses of paclitaxel in human melanoma. Cancer Lett. 2015, 356, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- De Milito, A.; Canese, R.; Marino, M.L.; Borghi, M.; Iero, M.; Villa, A.; Venturi, G.; Lozupone, F.; Iessi, E.; Logozzi, M.; et al. pH-dependent antitumor activity of proton pump inhibitors against human melanoma is mediated by inhibition of tumor acidity. Int. J. Cancer 2010, 127, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Fais, S.; Venturi, G.; Gatenby, B. Microenvironmental acidosis in carcinogenesis and metastases: New strategies in prevention and therapy. Cancer Metastasis Rev. 2014, 33, 1095–1108. [Google Scholar] [CrossRef] [PubMed]
- Gillies, R.J.; Raghunand, N.; Garcia-Martin, M.L.; Gatenby, R.A. pH imaging. A review of pH measurement methods and applications in cancers. IEEE Eng. Med. Biol. Mag. 2004, 23, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, S.; Hiraku, Y. Oxidative and nitrative DNA damage as biomarker for carcinogenesis with special reference to inflammation. Antioxid. Redox Signal. 2006, 8, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Thanan, R.; Ma, N.; Kawanishi, S. Role of Nitrative and Oxidative DNA Damage in Inflammation-Related Carcinogenesis. J. Biomed. Biotechnol. 2012, 2012, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed]
- Sosa, V.; Moliné, T.; Somoza, R.; Paciucci, R.; Kondoh, H.; LLeonart, M.E. Oxidative stress and cancer: An overview. Ageing Res. Rev. 2013, 12, 376–390. [Google Scholar] [CrossRef]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Gillies, R.J.; Brown, J.S.; Anderson, A.R.A.; Gatenby, R.A. Eco-evolutionary causes and consequences of temporal changes in intratumoural blood flow. Nat. Rev. Cancer 2018, 18, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Gillies, R.J.; Gatenby, R.A. Metabolism and its sequelae in cancer evolution and therapy. Cancer J. 2015, 21, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim-Hashim, A.; Robertson-Tessi, M.; Enriquez-Navas, P.M.; Damaghi, M.; Balagurunathan, Y.; Wojtkowiak, J.W.; Russell, S.; Yoonseok, K.; Lloyd, M.C.; Bui, M.M.; et al. Defining Cancer Subpopulations by Adaptive Strategies Rather Than Molecular Properties Provides Novel Insights into Intratumoral Evolution. Cancer Res. 2017, 77, 2242–2254. [Google Scholar] [CrossRef] [PubMed]
- Estrella, V.; Chen, T.; Lloyd, M.; Wojtkowiak, J.; Cornnell, H.H.; Ibrahim-Hashim, A.; Bailey, K.; Balagurunathan, Y.; Rothberg, J.M.; Sloane, B.F.; et al. Acidity generated by the tumor microenvironment drives local invasion. Cancer Res. 2013, 73, 1524–1535. [Google Scholar] [CrossRef] [PubMed]
- Calcinotto, A.; Filipazzi, P.; Grioni, M.; Iero, M.; De Milito, A.; Ricupito, A.; Cova, A.; Canese, R.; Jachetti, E.; Rossetti, M.; et al. Modulation of Microenvironment Acidity Reverses Anergy in Human and Murine Tumor-Infiltrating T Lymphocytes. Cancer Res. 2012, 72, 2746–2756. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.Q.; Pagel, M.D. Evaluating pH in the Extracellular Tumor Microenvironment Using CEST MRI and Other Imaging Methods. Adv. Radiol. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Luciani, F.; Spada, M.; De Milito, A.; Molinari, A.; Rivoltini, L.; Montinaro, A.; Marra, M.; Lugini, L.; Logozzi, M.; Lozupone, F.; et al. Effect of proton pump inhibitor pretreatment on resistance of solid tumors to cytotoxic drugs. J. Natl. Cancer Inst. 2004, 96, 1702–1713. [Google Scholar] [CrossRef]
- Da Silva, V.P.; Mesquita, C.B.; Nunes, J.S.; de Bem Prunes, B.; Rados, P.V.; Visioli, F. Effects of extracellular acidity on resistance to chemotherapy treatment: A systematic review. Med. Oncol. 2018, 35, 161. [Google Scholar] [CrossRef]
- De Milito, A.; Iessi, E.; Logozzi, M.; Lozupone, F.; Spada, M.; Marino, M.L.; Federici, C.; Perdicchio, M.; Matarrese, P.; Lugini, L.; et al. Proton pump inhibitors induce apoptosis of human B-cell tumors through a caspase-independent mechanism involving reactive oxygen species. Cancer Res. 2007, 67, 5408–5417. [Google Scholar] [CrossRef]
- Liou, G.-Y.; Storz, P. Reactive oxygen species in cancer. Free Radic. Res. 2010, 44, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Schumacker, P.T. Reactive oxygen species in cancer cells: Live by the sword, die by the sword. Cancer Cell 2006, 10, 175–176. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Cadenas, E. Oxidative stress: Damage to intact cells and organs. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1985, 311, 617–631. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Oxidative stress and cancer: Have we moved forward? Biochem. J. 2007, 401, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nunomura, A.; Castellani, R.J.; Zhu, X.; Moreira, P.I.; Perry, G.; Smith, M.A. Involvement of oxidative stress in Alzheimer disease. J. Neuropathol. Exp. Neurol. 2006, 65, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, L.; Pazhanisamy, S.K.; Li, H.; Meng, A.; Zhou, D. Total body irradiation causes residual bone marrow injury by induction of persistent oxidative stress in murine hematopoietic stem cells. Free Radic. Biol. Med. 2010, 48, 348–356. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality—A systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Basu, A.; Imrhan, V. Tomatoes versus lycopene in oxidative stress and carcinogenesis: Conclusions from clinical trials. Eur. J. Clin. Nutr. 2007, 61, 295–303. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, H.-Y. Cancer preventive mechanisms of the green tea polyphenol (-)-epigallocatechin-3-gallate. Molecules 2007, 12, 946–957. [Google Scholar] [CrossRef]
- La Vecchia, C.; Altieri, A.; Tavani, A. Vegetables, fruit, antioxidants and cancer: A review of Italian studies. Eur. J. Nutr. 2001, 40, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Eat Wholegrains, Vegetables, Fruit & Beans. Available online: https://www.wcrf.org/dietandcancer/recommendations/wholegrains-veg-fruit-beans (accessed on 8 October 2018).
- WHO|Increasing Fruit and Vegetable Consumption to Reduce the Risk of Noncommunicable Diseases. Available online: http://www.who.int/elena/titles/fruit_vegetables_ncds/en/ (accessed on 8 October 2018).
- Aruoma, O.I.; Colognato, R.; Fontana, I.; Gartlon, J.; Migliore, L.; Koike, K.; Coecke, S.; Lamy, E.; Mersch-Sundermann, V.; Laurenza, I.; et al. Molecular effects of fermented papaya preparation on oxidative damage, MAP Kinase activation and modulation of the benzo[a]pyrene mediated genotoxicity. Biofactors 2006, 26, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Aruoma, O.I.; Hayashi, Y.; Marotta, F.; Mantello, P.; Rachmilewitz, E.; Montagnier, L. Applications and bioefficacy of the functional food supplement fermented papaya preparation. Toxicology 2010, 278, 6–16. [Google Scholar] [CrossRef]
- Aruoma, O.I.; Somanah, J.; Bourdon, E.; Rondeau, P.; Bahorun, T. Diabetes as a risk factor to cancer: Functional role of fermented papaya preparation as phytonutraceutical adjunct in the treatment of diabetes and cancer. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2014, 768, 60–68. [Google Scholar] [CrossRef]
- Dickerson, R.; Banerjee, J.; Rauckhorst, A.; Pfeiffer, D.R.; Gordillo, G.M.; Khanna, S.; Osei, K.; Roy, S. Does oral supplementation of a fermented papaya preparation correct respiratory burst function of innate immune cells in type 2 diabetes mellitus patients? Antioxid. Redox Signal. 2015, 22, 339–345. [Google Scholar] [CrossRef]
- Marotta, F.; Koike, K.; Lorenzetti, A.; Jain, S.; Signorelli, P.; Metugriachuk, Y.; Mantello, P.; Locorotondo, N. Regulating Redox Balance Gene Expression in Healthy Individuals by Nutraceuticals: A Pilot Study. Rejuvenation Res. 2010, 13, 175–178. [Google Scholar] [CrossRef]
- Osato, J.A.; Santiago, L.A.; Remo, G.M.; Cuadra, M.S.; Mori, A. Antimicrobial and antioxidant activities of unripe papaya. Life Sci. 1993, 53, 1383–1389. [Google Scholar] [CrossRef]
- Mohamed Sadek, K. Antioxidant and immunostimulant effect of Carica papaya Linn. Aqueous extract in acrylamide intoxicated rats. Acta Inform. Med. 2012, 20, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Imao, M.; Sato, E.F.; Inoue, M.; Mori, A. Effect of fermented papaya preparation on dermal and intestinal mucosal immunity and allergic inflammations. J. Sci. Food Agric. 2008, 88, 1151–1157. [Google Scholar] [CrossRef]
- Collard, E.; Roy, S. Improved function of diabetic wound-site macrophages and accelerated wound closure in response to oral supplementation of a fermented papaya preparation. Antioxid. Redox Signal. 2010, 13, 599–606. [Google Scholar] [CrossRef]
- Das, A.; Dickerson, R.; Ghatak, P.D.; Gordillo, G.M.; Chaffee, S.; Saha, A.; Khanna, S.; Roy, S. May Dietary Supplementation Augment Respiratory Burst in Wound-Site Inflammatory Cells? Antioxid. Redox Signal. 2018, 28, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Nayak, B.S.; Ramdeen, R.; Adogwa, A.; Ramsubhag, A.; Marshall, J.R. Wound-healing potential of an ethanol extract of Carica papaya (Caricaceae) seeds. Int. Wound J. 2012, 9, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Ching, L.S.; Mohamed, S. Alpha-tocopherol content in 62 edible tropical plants. J. Agric. Food Chem. 2001, 49, 3101–3105. [Google Scholar] [CrossRef] [PubMed]
- Miean, K.H.; Mohamed, S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J. Agric. Food Chem. 2001, 49, 3106–3112. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Haldar, S. Dietary isothiocyanate mediated apoptosis of human cancer cells is associated with Bcl-xL phosphorylation. Int. J. Oncol. 2008, 33, 657–663. [Google Scholar] [PubMed]
- Nakamura, Y.; Yoshimoto, M.; Murata, Y.; Shimoishi, Y.; Asai, Y.; Park, E.Y.; Sato, K.; Nakamura, Y. Papaya seed represents a rich source of biologically active isothiocyanate. J. Agric. Food Chem. 2007, 55, 4407–4413. [Google Scholar] [CrossRef] [PubMed]
- Van Breemen, R.B.; Pajkovic, N. Multitargeted therapy of cancer by lycopene. Cancer Lett. 2008, 269, 339–351. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Shaw, P.N.; Parat, M.-O.; Hewavitharana, A.K. Anticancer activity of Carica papaya: A review. Mol. Nutr. Food Res. 2013, 57, 153–164. [Google Scholar] [CrossRef]
- Otsuki, N.; Dang, N.H.; Kumagai, E.; Kondo, A.; Iwata, S.; Morimoto, C. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J. Ethnopharmacol. 2010, 127, 760–767. [Google Scholar] [CrossRef]
- Fibach, E.; Rachmilewitz, E.A. The Effect of Fermented Papaya Preparation on Radioactive Exposure. Radiat. Res. 2015, 184, 304–313. [Google Scholar] [CrossRef]
- Somanah, J.; Bourdon, E.; Rondeau, P.; Bahorun, T.; Aruoma, O.I. Relationship between fermented papaya preparation supplementation, erythrocyte integrity and antioxidant status in pre-diabetics. Food Chem. Toxicol. 2014, 65, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Marotta, F.; Yoshida, C.; Barreto, R.; Naito, Y.; Packer, L. Oxidative-inflammatory damage in cirrhosis: Effect of vitamin E and a fermented papaya preparation. J. Gastroenterol. Hepatol. 2007, 22, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, R.; Deshpande, B.; Gnyawali, U.; Lynch, D.; Gordillo, G.M.; Schuster, D.; Osei, K.; Roy, S. Correction of aberrant NADPH oxidase activity in blood-derived mononuclear cells from type II diabetes mellitus patients by a naturally fermented papaya preparation. Antioxid. Redox Signal. 2012, 17, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Marotta, F.; Yadav, H.; Kumari, A.; Catanzaro, R.; Jain, S.; Polimeni, A.; Lorenzetti, A.; Soresi, V. Cardioprotective effect of a biofermented nutraceutical on endothelial function in healthy middle-aged subjects. Rejuvenation Res. 2012, 15, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Rimbach, G.; Guo, Q.; Akiyama, T.; Matsugo, S.; Moini, H.; Virgili, F.; Packer, L. Ferric nitrilotriacetate induced DNA and protein damage: Inhibitory effect of a fermented papaya preparation. Anticancer Res. 2000, 20, 2907–2914. [Google Scholar] [PubMed]
- Prus, E.; Fibach, E. The antioxidant effect of fermented papaya preparation involves iron chelation. J. Biol. Regul. Homeost. Agents 2012, 26, 203–210. [Google Scholar]
- Rimbach, G.; Park, Y.C.; Guo, Q.; Moini, H.; Qureshi, N.; Saliou, C.; Takayama, K.; Virgili, F.; Packer, L. Nitric oxide synthesis and TNF-alpha secretion in RAW 264.7 macrophages: Mode of action of a fermented papaya preparation. Life Sci. 2000, 67, 679–694. [Google Scholar] [CrossRef]
- Bertuccelli, G.; Zerbinati, N.; Marcellino, M.; Nanda Kumar, N.S.; He, F.; Tsepakolenko, V.; Cervi, J.; Lorenzetti, A.; Marotta, F. Effect of a quality-controlled fermented nutraceutical on skin aging markers: An antioxidant-control, double-blind study. Exp. Ther. Med. 2016, 11, 909–916. [Google Scholar] [CrossRef]
- Barbagallo, M.; Marotta, F.; Dominguez, L.J. Oxidative stress in patients with Alzheimer’s disease: Effect of extracts of fermented papaya powder. Mediators Inflamm. 2015, 2015, 624801. [Google Scholar] [CrossRef]
- Bolner, A.; Micciolo, R.; Bosello, O.; Nordera, G. Effect of papaya supplementation on oxidative stress markers in Parkinsons disease. Oxid. Antioxid. Med. Sci. 2016, 5, 49. [Google Scholar] [CrossRef]
- Somanah, J.; Ramsaha, S.; Verma, S.; Kumar, A.; Sharma, P.; Singh, R.K.; Aruoma, O.I.; Bourdon, E.; Bahorun, T. Fermented papaya preparation modulates the progression of N-methyl-N-nitrosourea induced hepatocellular carcinoma in Balb/c mice. Life Sci. 2016, 151, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Germann, P.G.; Ockert, D. Granulomatous inflammation of the oropharyngeal cavity as a possible cause for unexpected high mortality in a Fischer 344 rat carcinogenicity study. Lab. Anim. Sci. 1994, 44, 338–343. [Google Scholar] [PubMed]
- Murphy, S.J.; Smith, P.; Shaivitz, A.B.; Rossberg, M.I.; Hurn, P.D. The effect of brief halothane anesthesia during daily gavage on complications and body weight in rats. Contemp. Top. Lab. Anim. Sci. 2001, 40, 9–12. [Google Scholar]
- Brown, A.P.; Dinger, N.; Levine, B.S. Stress produced by gavage administration in the rat. Contemp. Top. Lab. Anim. Sci. 2000, 39, 17–21. [Google Scholar] [PubMed]
- Pilon-Thomas, S.; Kodumudi, K.N.; El-Kenawi, A.E.; Russell, S.; Weber, A.M.; Luddy, K.; Damaghi, M.; Wojtkowiak, J.W.; Mulé, J.J.; Ibrahim-Hashim, A.; et al. Neutralization of Tumor Acidity Improves Antitumor Responses to Immunotherapy. Cancer Res. 2016, 76, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Pilot, C.; Mahipal, A.; Gillies, R.J. Buffer Therapy—Buffer Diet. J. Nutr. Food Sci. 2018, 8. [Google Scholar] [CrossRef]
- Mazzocca, A.; Ferraro, G.; Misciagna, G.; Fais, S. Moving the systemic evolutionary approach to cancer forward: Therapeutic implications. Med. Hypotheses 2018, 121, 80–87. [Google Scholar] [CrossRef]
- Korn, E.L.; Liu, P.-Y.; Lee, S.J.; Chapman, J.-A.W.; Niedzwiecki, D.; Suman, V.J.; Moon, J.; Sondak, V.K.; Atkins, M.B.; Eisenhauer, E.A.; et al. Meta-analysis of phase II cooperative group trials in metastatic stage IV melanoma to determine progression-free and overall survival benchmarks for future phase II trials. J. Clin. Oncol. 2008, 26, 527–534. [Google Scholar] [CrossRef]
| Group | Start of Treatments | Number of Animals | Dosage FPP®/Mouse (mg/Kg/day) | Route of Administration |
|---|---|---|---|---|
| Control (CTR) | / | 5 | sterile water | oral gavage |
| A1 | 1 day before inoculum | 5 | 200 | oral gavage |
| A2 | 3 days post inoculum | 5 | 200 | oral gavage |
| A3 | 7 days post inoculum | 5 | 200 | oral gavage |
| B1 | 1 day before inoculum | 5 | 400 | oral gavage |
| B2 | 3 days post inoculum | 5 | 400 | oral gavage |
| B3 | 7 days post inoculum | 5 | 400 | oral gavage |
| Group | Start of Treatments | Number of Animals | Dosage FPP®/Mouse (mg/Kg/day) | Route of Administration |
|---|---|---|---|---|
| Control (CTR) | / | 6 | sterile water | oral gavage |
| A | 7 days post inoculum | 6 | 200 | oral gavage |
| B | 7 days post inoculum | 6 | 200 | sublingual |
| Group | Start of Treatments | Number of Animals | Dosage FPP®/Mouse (mg/Kg/day) | Route of Administration |
|---|---|---|---|---|
| Control (CTR) | / | 10 | sterile water | sublingual |
| A | 21 day before inoculum | 10 | 200 | sublingual |
| B | 14 days post inoculum | 10 | 200 | sublingual |
| C | 3 days post inoculum | 10 | 200 | sublingual |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Logozzi, M.; Mizzoni, D.; Di Raimo, R.; Macchia, D.; Spada, M.; Fais, S. Oral Administration of Fermented Papaya (FPP®) Controls the Growth of a Murine Melanoma through the In Vivo Induction of a Natural Antioxidant Response. Cancers 2019, 11, 118. https://doi.org/10.3390/cancers11010118
Logozzi M, Mizzoni D, Di Raimo R, Macchia D, Spada M, Fais S. Oral Administration of Fermented Papaya (FPP®) Controls the Growth of a Murine Melanoma through the In Vivo Induction of a Natural Antioxidant Response. Cancers. 2019; 11(1):118. https://doi.org/10.3390/cancers11010118
Chicago/Turabian StyleLogozzi, Mariantonia, Davide Mizzoni, Rossella Di Raimo, Daniele Macchia, Massimo Spada, and Stefano Fais. 2019. "Oral Administration of Fermented Papaya (FPP®) Controls the Growth of a Murine Melanoma through the In Vivo Induction of a Natural Antioxidant Response" Cancers 11, no. 1: 118. https://doi.org/10.3390/cancers11010118
APA StyleLogozzi, M., Mizzoni, D., Di Raimo, R., Macchia, D., Spada, M., & Fais, S. (2019). Oral Administration of Fermented Papaya (FPP®) Controls the Growth of a Murine Melanoma through the In Vivo Induction of a Natural Antioxidant Response. Cancers, 11(1), 118. https://doi.org/10.3390/cancers11010118