Real-World Implications of Nonbiological Factors with Staging, Prognosis and Clinical Management in Colon Cancer
Abstract
1. Introduction
2. Patients and Methods
2.1. Study Design and Data Source
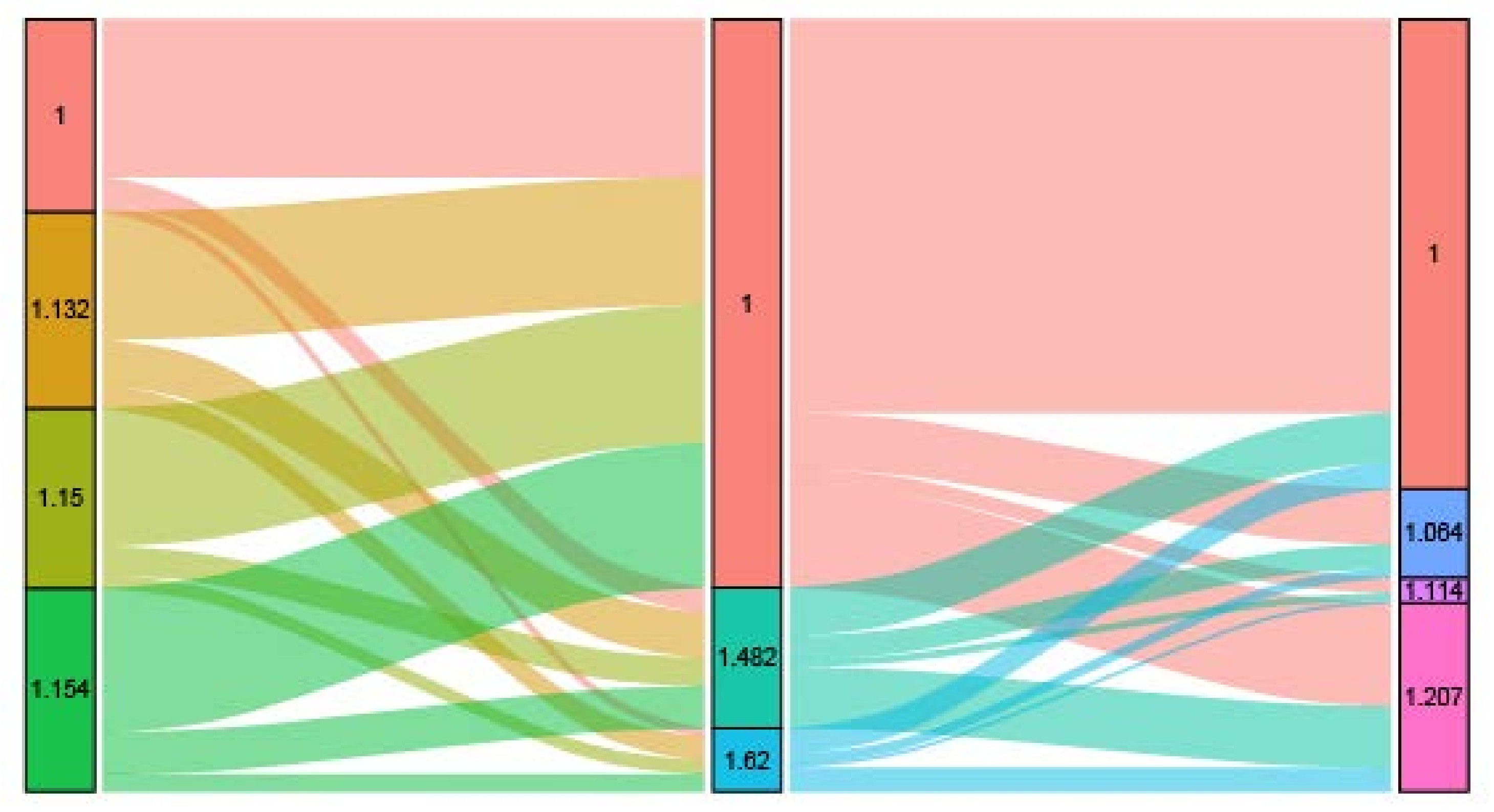
2.2. NBF Stage and Statistical Analysis
3. Results
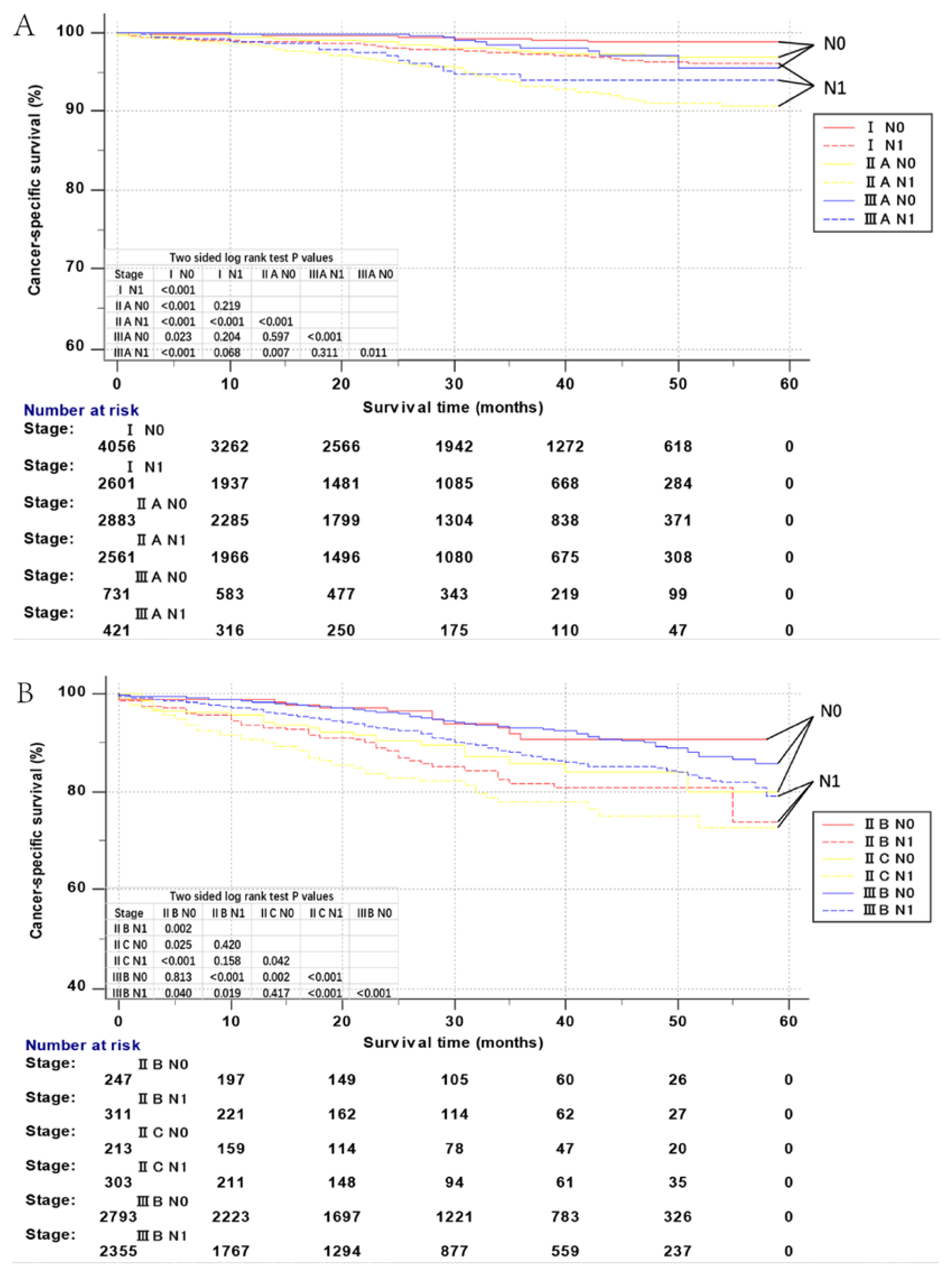
3.1. Three NBFs Were Strongly Associated with CSS of Colon Cancer
3.2. NBF Stage Was Strongly Associated with CSS in Colon Cancer
3.3. Prognostic Prediction of NBF-TNM Stage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A.; Ahnen, D.J.; Meester, R.G.S.; Barzi, A.; Jemal, A. Colorectal cancer statistics, 2017. CA A Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Arena, E.A.; Bilchik, A.J. What is the optimal means of staging colon cancer? Adv. Surg. 2013, 47, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Dienstmann, R.; Mason, M.J.; Sinicrope, F.A.; Phipps, A.I.; Tejpar, S.; Nesbakken, A.; Danielsen, S.A.; Sveen, A.; Buchanan, D.D.; Clendenning, M. Prediction of overall survival in stage II and III colon cancer beyond tnm system: A retrospective, pooled biomarker study. Ann. Oncol. 2017, 28, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Stintzing, S.; Tejpar, S.; Gibbs, P.; Thiebach, L.; Lenz, H.J. Understanding the role of primary tumour localisation in colorectal cancer treatment and outcomes. Eur. J. Cancer 2017, 84, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Lu, G.; Liang, L.; Li, X.; Cai, S. The influence of marital status on stage at diagnosis and survival of patients with colorectal cancer. Oncotarget 2015, 6, 985–988. [Google Scholar] [CrossRef] [PubMed]
- Aizer, A.A.; Chen, M.H.; Mccarthy, E.P.; Mendu, M.L.; Koo, S.; Wilhite, T.J.; Graham, P.L.; Choueiri, T.K.; Hoffman, K.E.; Martin, N.E. Marital status and survival in patients with cancer. J. Clin. Oncol. 2013, 31, 3869–3876. [Google Scholar] [CrossRef] [PubMed]
- Du, X.L.D.M.; Lin, C.C.; Johnson, N.J.; Altekruse, S.A.D. Effects of individual-level socioeconomic factors on racial disparities in cancer treatment and survival. Cancer 2011, 117, 3242–3251. [Google Scholar] [CrossRef] [PubMed]
- Koroukian, S.M.; Bakaki, P.M.; Raghavan, D. Survival disparities by medicaid status: An analysis of 8 cancers. Cancer 2012, 118, 4271–4279. [Google Scholar] [CrossRef] [PubMed]
- Rodday, A.M.; Parsons, S.K.; Snyder, F.; Simon, M.A.; Llanos, A.A.M.; Warren-Mears, V.; Dudley, D.; Lee, J.H.; Patierno, S.R.; Markossian, T.W. Impact of patient navigation in eliminating economic disparities in cancer care. Cancer 2015, 121, 4025–4034. [Google Scholar] [CrossRef] [PubMed]
- Weiser, M.R.; Gönen, M.; Chou, J.F.; Kattan, M.W.; Schrag, D. Predicting survival after curative colectomy for cancer: Individualizing colon cancer staging. J. Clin. Oncol. 2011, 29, 4796–4802. [Google Scholar] [CrossRef] [PubMed]
- Auclin, E.; Zaanan, A.; Vernerey, D.; Douard, R.; Gallois, C.; Laurentpuig, P.; Bonnetain, F.; Taieb, J. Subgroups and prognostication in stage iii colon cancer: Future perspectives for adjuvant therapy. Ann. Oncol. 2017, 28, mdx030. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Ma, Y.; Luo, D.; Cai, S.; Li, Q.; Li, X. Real-world study of a novel prognostic scoring system: For a more precise prognostication and better clinical treatment guidance in stages ii and iii colon cancer. Int. J. Colorectal Dis. 2018, 33, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Alwers, E.; Jia, M.; Kloor, M.; Bläker, H.; Brenner, H.; Hoffmeister, M. Associations between molecular classifications of colorectal cancer and patient survival: A systematic review. Clin. Gastroenterol. Hepatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Okugawa, Y.; Grady, W.M.; Goel, A. Epigenetic alterations in colorectal cancer: Emergingbiomarkers. Gastroenterology 2015, 149, 1204–1225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, Y.; Hu, H.; Huang, R.; Xie, L.; Liu, E.; Chen, Y.G.; Wang, G.; Wang, X. Impact of socioeconomic status on survival of colorectal cancer patients. Oncotarget 2017, 8, 106121–106131. [Google Scholar] [CrossRef] [PubMed]
- Gallo, L.C.; Troxel, W.M.; Matthews, K.A.; Kuller, L.H. Marital status and quality in middle-aged women: Associations with levels and trajectories of cardiovascular risk factors. Health Psychol. 2003, 22, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. The biology of vascular endothelial growth factor. Endocr. Rev. 1997, 18, 4–25. [Google Scholar] [CrossRef] [PubMed]
- Cowan, R.A.; Tseng, J.; Ali, N.; Dearie, H.; Murthy, V.; Gennarelli, R.L.; Iasonos, A.; Aburustum, N.R.; Chi, D.S.; Long, K.R. Exploring the impact of income and race on survival for women with advanced ovarian cancer undergoing primary debulking surgery at a high-volume center. Gynecol. Oncol. 2018, 149, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Yarbro, J.W.; Page, D.L.; Fielding, L.P.; Partridge, E.E.; Murphy, G.P. American joint committee on cancer prognostic factors consensus conference. Cancer 2000, 88, 1739–1757. [Google Scholar] [CrossRef]
- Weiser, M.R.; Landmann, R.G.; Kattan, M.W.; Gonen, M.; Shia, J.; Chou, J.; Paty, P.B.; Guillem, J.G.; Temple, L.K.; Schrag, D. Individualized prediction of colon cancer recurrence using a nomogram. J. Clin. Oncol. 2008, 26, 380–385. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, J.B.; Maggard, M.A.; Ko, C.Y. Colon cancer survival rates with the new american joint committee on cancer sixth edition staging. J. Natl. Cancer Inst. 2004, 96, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Thirunavukarasu, P.; Sukumar, S.; Sathaiah, M.; Mahan, M.; Pragatheeshwar, K.D.; Pingpank, J.F.; Zeh, H.; Bartels, C.J.; Lee, K.K.W.; Bartlett, D.L. C-stage in colon cancer: Implications of carcinoembryonic antigen biomarker in staging, prognosis, and management. J. Nat. Cancer Inst. 2011, 103, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Rd, B.A.; Schrag, D.; Somerfield, M.R.; Cohen, A.M.; Figueredo, A.T.; Flynn, P.J.; Krzyzanowska, M.K.; Maroun, J.; Mcallister, P.; Van, C.E. American society of clinical oncology recommendations on adjuvant chemotherapy for stage ii colon cancer. J. Clin. Oncol. 2016, 22, 3408–3419. [Google Scholar]
- Ganapathi, A.M.; Speicher, P.J.; Englum, B.R.; Castleberry, A.W.; Migaly, J.; Hsu, D.S.; Mantyh, C.R. Adjuvant chemotherapy for t1 node-positive colon cancers provides significant survival benefit. Dis. Colon Rectum 2014, 57, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Casadaban, L.; Rauscher, G.; Aklilu, M.; Villenes, D.; Freels, S.; Maker, A.V. Adjuvant chemotherapy is associated with improved survival in patients with stage II colon cancer. Cancer 2016, 122, 3277–3287. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, E.S.; Greenblatt, D.Y.; Loconte, N.K.; Gangnon, R.E.; Liou, J.I.; Heise, C.P.; Smith, M.A. Adjuvant chemotherapy for stage II colon cancer with poor prognostic features. J. Clin. Oncol. 2011, 29, 3381–3388. [Google Scholar] [CrossRef] [PubMed]
- Quah, H.M.; Chou, J.F.; Gonen, M.; Shia, J.; Schrag, D.; Landmann, R.G.; Guillem, J.G.; Paty, P.B.; Temple, L.K.; Wong, W.D. Identification of patients with high-risk stage II colon cancer for adjuvant therapy. Dis. Colon Rectum 2008, 51, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.; Loprinzi, C.L.; Sargent, D.J.; Thomé, S.D.; Alberts, S.R.; Haller, D.G.; Benedetti, J.; Francini, G.; Shepherd, L.E.; Francois, S.J. Pooled analysis of fluorouracil-based adjuvant therapy for stage ii and iii colon cancer: Who benefits and by how much? J. Clin. Oncol. 2004, 22, 1797–1806. [Google Scholar] [CrossRef] [PubMed]
- Sargent, D.J.; Goldberg, R.M.; Jacobson, S.D.; Macdonald, J.S.; Labianca, R.; Haller, D.G.; Shepherd, L.E.; Seitz, J.F.; Francini, G. A pooled analysis of adjuvant chemotherapy for resected colon cancer in elderly patients. N. Engl. J. Med. 2001, 345, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Verhoeff, S.R.; Van Erning, F.N.; Lemmens, V.E.P.P.; De Wilt, J.H.W.; Pruijt, J.F.M. Adjuvant chemotherapy is not associated with improved survival for all high-risk factors in stage ii colon cancer. Int. J. Cancer J. Int. Du Cancer 2016, 139, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Roth, A.D.; Delorenzi, M.; Tejpar, S.; Yan, P.; Klingbiel, D.; Fiocca, R.; D’Ario, G.; Cisar, L.; Labianca, R.; Cunningham, D. Integrated analysis of molecular and clinical prognostic factors in stage ii/iii colon cancer. J. Nat. Cancer Inst. 2012, 104, 1635–1646. [Google Scholar] [CrossRef] [PubMed]
- Wolmark, N.; Fisher, B.; Wieand, H.S.; Henry, R.S.; Lerner, H.; Legault-Poisson, S.; Deckers, P.J.; Dimitrov, N.; Gordon, P.H.; Jochimsen, P. The prognostic significance of preoperative carcinoembryonic antigen levels in colorectal cancer. Results from nsabp (national surgical adjuvant breast and bowel project) clinical trials. Ann. Surg. 1984, 199, 375–382. [Google Scholar] [CrossRef] [PubMed]



| Characteristic | No. (%) | |
|---|---|---|
| Race | White | 21,179 (73.5) |
| Black | 4706 (16.3) | |
| Other | 2933 (10.2) | |
| Gender | Male | 15,402 (53.4) |
| Female | 13,416 (46.6) | |
| Tumor location | Appendix | 359 (1.2) |
| Cecum | 5276 (18.3) | |
| Ascending colon | 4703 (16.3) | |
| Hepatic flexure | 1165 (4.0) | |
| Transverse colon | 2538 (8.8) | |
| Splenic flexure | 1062 (3.7) | |
| Descending colon | 2350 (8.2) | |
| Sigmoid Colon | 11,365 (39.4) | |
| Tumor grade | Grade I | 2360 (8.2) |
| Grade II | 19,568 (67.9) | |
| Grade III | 4103 (14.2) | |
| Grade IV | 723 (2.5) | |
| Unknown | 2064 (7.2) | |
| AJCC stage | I | 6926 (24.0) |
| IIA | 5645 (19.6) | |
| IIB | 589 (2.0) | |
| IIC | 546 (1.9) | |
| IIIA | 1182 (4.1) | |
| IIIB | 5323 (18.5) | |
| IIIC | 1938 (6.7) | |
| IVA | 3461 (12.0) | |
| IVB | 3208 (11.1) | |
| Surgery | Surgery performed | 26,296(91.2) |
| Surgery not performed | 2522(8.8) | |
| County % with bachelor degree | 5.95–20.77% | 7070 (24.5) |
| 20.78–29.91% | 5826 (20.2) | |
| 29.92–35.57% | 8689 (30.2) | |
| 35.58–64.01% | 7233 (25.1) | |
| County-level median household income # | 19.15–48.57 K | 7180 (24.9) |
| 48.58–55.87 K | 6719 (23.3) | |
| 55.88–67.29 K | 7587 (26.3) | |
| 67.30–106.52 K | 7332 (25.4) | |
| County % were unemployed | 1.92–8.66% | 7180 (24.9) |
| 8.67–9.60% | 6719 (23.3) | |
| 9.61–11.27% | 7587 (26.3) | |
| 11.28–21.21% | 7332 (25.4) | |
| Year of diagnosis | 2010 | 5757 (20.0) |
| 2011 | 5634 (19.6) | |
| 2012 | 5596 (19.4) | |
| 2013 | 5773 (20.0) | |
| 2014 | 6058 (21.0) | |
| Tumor size | ≤5 cm | 16,409 (56.9) |
| >5 cm | 8409 (29.2) | |
| Unknown | 4000 (13.9) | |
| Age at diagnosis (years) | ≤50 | 8245 (28.6) |
| 51–55 | 6313 (21.9) | |
| 56–60 | 7335 (25.5) | |
| ≥61 | 6925 (24.0) | |
| Insurance status | Insured | 21,198 (73.6) |
| Medicaid | 5258 (18.2) | |
| Uninsured | 2362 (8.2) | |
| Marital status | Married | 17,515 (60.8) |
| Single | 7040 (24.4) | |
| Divorced | 3321 (11.5) | |
| Widowed | 942 (3.3) | |
| Variable | Reference | Characteristic | Cancer-Specific Survival | ||
|---|---|---|---|---|---|
| HR (95%) | SE | p Value | |||
| Race | White | Black | 1.179 (1.089–1.275) | 0.040 | <0.001 |
| Other | 1.046 (0.941–1.162) | 0.054 | 0.405 | ||
| Gender | Male | Female | 0.851 (0.801–0.904) | 0.031 | <0.001 |
| Tumor location | Appendix | Cecum | 0.871 (0.682–1.113) | 0.125 | 0.270 |
| Ascending colon | 0.864 (0.675–1.107) | 0.126 | 0.249 | ||
| Hepatic flexure | 0.859 (0.652–1.132) | 0.141 | 0.281 | ||
| Transverse colon | 0.811 (0.626–1.050) | 0.132 | 0.112 | ||
| Splenic flexure | 0.773 (0.585–1.023) | 0.143 | 0.072 | ||
| Descending colon | 0.670 (0.516–0.869) | 0.133 | 0.003 | ||
| Sigmoid Colon | 0.620 (0.487–0.791) | 0.124 | <0.001 | ||
| Tumor grade | Grade I | Grade II | 1.042 (0.892–1.217) | 0.079 | 0.601 |
| Grade III | 1.720 (1.461–2.025) | 0.083 | <0.001 | ||
| Grade IV | 2.111 (1.716–2.596) | 0.106 | <0.001 | ||
| Unknown | 1.164 (0.980–1.382) | 0.088 | 0.083 | ||
| AJCC stage | I | IIA | 2.266 (1.738–2.955) | 0.135 | <0.001 |
| IIB | 6.547 (4.614–9.291) | 0.179 | <0.001 | ||
| IIC | 9.819 (7.124–13.535) | 0.164 | <0.001 | ||
| IIIA | 1.864 (1.199–2.899) | 0.225 | 0.006 | ||
| IIIB | 5.600 (4.399–7.129) | 0.123 | <0.001 | ||
| IIIC | 16.630 (13.051–21.192) | 0.124 | <0.001 | ||
| IVA | 39.280 (31.324–49.257) | 0.115 | <0.001 | ||
| IVB | 54.228 (43.145–68.159) | 0.117 | <0.001 | ||
| Surgery | Surgery performed | Surgery not performed | 2.649 (2.417–2.903) | 0.047 | <0.001 |
| Insurance status | Insured | Medicaid | 1.482 (1.376–1.597) | 0.038 | <0.001 |
| Uninsured | 1.620 (1.476–1.778) | 0.048 | <0.001 | ||
| Marital status | Married | Single | 1.207 (1.123–1.297) | 0.037 | <0.001 |
| Divorced | 1.064 (0.969–1.169) | 0.048 | 0.195 | ||
| Widowed | 1.114 (0.945–1.314) | 0.084 | 0.199 | ||
| Tumor size | ≤5 cm | >5 cm | 1.157 (1.080–1.240) | 0.035 | <0.001 |
| Unknown | 1.187 (1.082–1.302) | 0.047 | <0.001 | ||
| Age at diagnosis (years) | ≤50 | 51–55 | 1.078 (0.988–1.175) | 0.044 | 0.092 |
| 56–60 | 1.188 (1.096–1.288) | 0.041 | <0.001 | ||
| ≥61 | 1.338 (1.232–1.452) | 0.042 | <0.001 | ||
| County % with bachelor degree | 35.58–64.01% | 29.92–35.57% | 1.017 (0.918–1.127) | 0.052 | 0.745 |
| 20.78–29.91% | 1.035 (0.926–1.157) | 0.057 | 0.547 | ||
| 5.95–20.77% | 1.138 (1.005–1.289) | 0.063 | 0.041 | ||
| County-level median household income | 67.30–106.52 K | 55.88–67.29 K | 1.150 (1.037–1.276) | 0.053 | 0.008 |
| 48.58–55.87 K | 1.154 (1.027–1.297) | 0.060 | 0.016 | ||
| 19.15–48.57 K | 1.132 (1.001–1.281) | 0.063 | 0.048 | ||
| County % were unemployed | 1.92–8.66% | 8.67–9.60% | 1.074 (0.984–1.171) | 0.044 | 0.110 |
| 9.61–11.27% | 1.014 (0.922–1.115) | 0.049 | 0.771 | ||
| 11.28–21.21% | 1.062 (0.968–1.265) | 0.047 | 0.201 | ||
| Variable | Reference | Characteristic | Cancer-Specific Survival | ||
|---|---|---|---|---|---|
| HR (95%) | SE | p Value | |||
| Race | White | Black | 1.210 (1.121–1.307) | 0.039 | <0.001 |
| Other | 1.038 (0.936–1.152) | 0.053 | 0.477 | ||
| Gender | Male | Female | 0.842 (0.793–0.894) | 0.031 | <0.001 |
| Tumor location | Appendix | Cecum | 0.897 (0.702–1.146) | 0.125 | 0.386 |
| Ascending colon | 1.881 (0.688–1.129) | 0.126 | 0.317 | ||
| Hepatic flexure | 0.878 (0.667–1.156) | 0.140 | 0.353 | ||
| Transverse colon | 0.824 (0.637–1.067) | 0.132 | 0.143 | ||
| Splenic flexure | 0.785 (0.594–1.038) | 0.143 | 0.09 | ||
| Descending colon | 0.680 (0.524–0.882) | 0.133 | 0.004 | ||
| Sigmoid Colon | 0.637 (0.500–0.811) | 0.124 | <0.001 | ||
| Tumor grade | Grade I; | Grade II | 1.027 (0.879–1.199) | 0.079 | 0.737 |
| Grade III | 1.698 (1.443–1.999) | 0.083 | <0.001 | ||
| Grade IV | 2.076 (1.688–2.554) | 0.106 | <0.001 | ||
| Unknown | 1.152 (0.970–1.368) | 0.088 | 0.106 | ||
| Surgery | Surgery performed | Surgery not performed | 2.632 (2.402–2.884) | 0.047 | <0.001 |
| Tumor size | ≤5 cm | >5 cm | 1.169 (1.090–1.252) | 0.035 | <0.001 |
| Unknown | 1.186 (1.081–1.300) | 0.047 | <0.001 | ||
| Age at diagnosis (years) | ≤50 | 51–55 | 1.086 (0.995–1.184) | 0.044 | 0.063 |
| 56–60 | 1.189 (1.097–1.289) | 0.041 | <0.001 | ||
| ≥61 | 1.340 (1.236–1.454) | 0.041 | <0.001 | ||
| County % with bachelor degree | 5.95–20.77% | 20.78–29.91% | 1.072 (0.978–1.176) | 0.047 | 0.136 |
| 29.92–35.57% | 1.119 (1.021–1.228) | 0.047 | 0.017 | ||
| 35.58–64.01% | 1.254 (1.139–1.382) | 0.049 | <0.001 | ||
| County % were unemployed | 1.92–8.66% | 8.67–9.60% | 1.090 (1.000–1.188) | 0.044 | 0.050 |
| 9.61–11.27% | 1.050 (0.959–1.150) | 0.047 | 0.293 | ||
| 11.28–21.21% | 1.075 (0.981–1.178) | 0.047 | 0.121 | ||
| AJCC stage | I | IIA | 2.296 (1.761–2.994) | 0.135 | <0.001 |
| IIB | 6.704 (4.725–9.512) | 0.178 | <0.001 | ||
| IIC | 9.979 (7.240–13.753) | 0.164 | <0.001 | ||
| IIIA | 1.859 (1.195–2.890) | 0.225 | 0.006 | ||
| IIIB | 5.651 (4.439–7.194) | 0.123 | <0.001 | ||
| IIIC | 16.921 (13.279–21.562) | 0.124 | <0.001 | ||
| IVA | 40.051 (31.941–50.220) | 0.115 | <0.001 | ||
| IVB | 55.404 (44.081–69.636) | 0.117 | <0.001 | ||
| NBF stage | Stage 0 | Stage 1 | 1.504 (1.415–1.600) | 0.031 | <0.001 |
| Stage | No. of Patients | Cancer-Specific Survival | ||
|---|---|---|---|---|
| HR (95% CI) | SE | p Value | ||
| I NBF0 | 4190 | 1.000 (Referent) | ||
| I NBF1 | 2736 | 3.172 (2.009–5.008) | 0.233 | p < 0.001 |
| IIA NBF0 | 2975 | 2.391 (1.503–3.801) | 0.237 | p < 0.001 |
| IIA NBF1 | 2670 | 6.570 (4.348–9.928) | 0.211 | p < 0.001 |
| IIB NBF0 | 258 | 7.130 (3.689–13.781) | 0.336 | p < 0.001 |
| IIB NBF1 | 331 | 18.142 (11.143–29.537) | 0.249 | p < 0.001 |
| IIC NBF0 | 223 | 14.397 (8.217–25.224) | 0.286 | p < 0.001 |
| IIC NBF1 | 323 | 23.679 (14.859–37.736) | 0.238 | p < 0.001 |
| IIIA NBF0 | 748 | 2.153 (1.072–4.326) | 0.356 | p < 0.001 |
| IIIA NBF1 | 434 | 5.595 (2.987–10.478) | 0.320 | p < 0.001 |
| IIIB NBF0 | 2867 | 8.364 (5.594–12.506) | 0.205 | p < 0.001 |
| IIIB NBF1 | 2456 | 13.382 (9.005–19.886) | 0.202 | p < 0.001 |
| IIIC NBF0 | 949 | 26.248 (17.553–39.249) | 0.205 | p < 0.001 |
| IIIC NBF1 | 989 | 38.634 (25.996–57.416) | 0.202 | p < 0.001 |
| IVA NBF0 | 1681 | 61.161 (41.193–89.505) | 0.194 | p < 0.001 |
| IVA NBF1 | 1780 | 92.326 (63.182–134.911) | 0.194 | p < 0.001 |
| IVB NBF0 | 1435 | 91.451 (62.434–133.954) | 0.195 | p < 0.001 |
| IVB NBF1 | 1773 | 121.179 (82.852–177.236) | 0.194 | p < 0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Q.; Luo, D.; Cai, S.; Li, Q.; Li, X. Real-World Implications of Nonbiological Factors with Staging, Prognosis and Clinical Management in Colon Cancer. Cancers 2018, 10, 263. https://doi.org/10.3390/cancers10080263
Liu Q, Luo D, Cai S, Li Q, Li X. Real-World Implications of Nonbiological Factors with Staging, Prognosis and Clinical Management in Colon Cancer. Cancers. 2018; 10(8):263. https://doi.org/10.3390/cancers10080263
Chicago/Turabian StyleLiu, Qi, Dakui Luo, Sanjun Cai, Qingguo Li, and Xinxiang Li. 2018. "Real-World Implications of Nonbiological Factors with Staging, Prognosis and Clinical Management in Colon Cancer" Cancers 10, no. 8: 263. https://doi.org/10.3390/cancers10080263
APA StyleLiu, Q., Luo, D., Cai, S., Li, Q., & Li, X. (2018). Real-World Implications of Nonbiological Factors with Staging, Prognosis and Clinical Management in Colon Cancer. Cancers, 10(8), 263. https://doi.org/10.3390/cancers10080263





