Stereotactic Body Radiation Therapy for Patients with Pulmonary Interstitial Change: High Incidence of Fatal Radiation Pneumonitis in a Retrospective Multi-Institutional Study
Abstract
1. Introduction
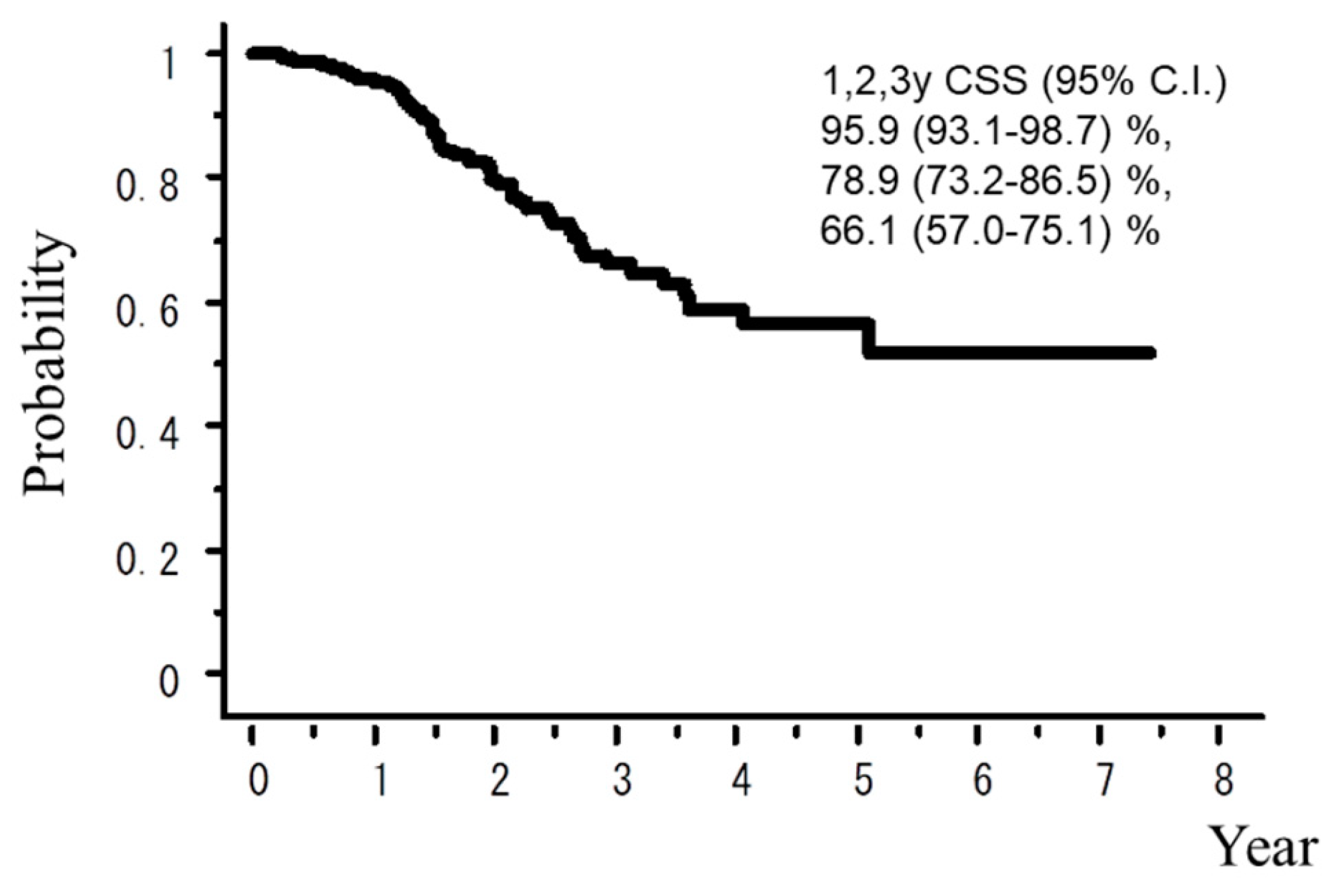
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Onishi, H.; Shirato, H.; Nagata, Y.; Hiraoka, M.; Fujino, M.; Gomi, K.; Niibe, Y.; Karasawa, K.; Hayakawa, K.; Takai, Y.; et al. Hypofractionated stereotactic radiotherapy (HypoFXSRT) for stage I non-small cell lung cancer: Updated results of 257 patients in a Japanese multi-institutional study. J. Thorac. Oncol. 2007, 2, S94–S100. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.Y.; Senan, S.; Paul, M.A.; Mehran, J.R.; Louie, A.V.; Balter, P.; Groen, H.J.M.; McRae, S.E.; Widder, J.; Feng, L.; et al. Stereotactic ablative radiotherapy versus lobectomy for operable stage I non-small-cell lung cancer: A pooled analysis of two randomised trials. Lancet Oncol. 2015, 16, 630–637. [Google Scholar] [CrossRef]
- Yamaguchi, S.; Ohguri, T.; Ide, S.; Aoki, T.; Imada, H.; Yahara, K.; Narisada, H.; Korogi, Y. Stereotactic body radiotherapy for lung tumors in patients with subclinical interstitial lung disease: The potential risk of extensive radiation pneumonitis. Lung Cancer 2013, 82, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Ueki, N.; Matsuo, Y.; Togashi, Y.; Kubo, T.; Shibuya, K.; Iizuka, Y.; Mizowaki, T.; Togashi, K.; Mishima, M.; Hiraoka, M. Impact of pretreatment interstitial lung disease on radiation pneumonitis and survival after stereotactic body radiation therapy for lung cancer. J. Thorac. Oncol. 2015, 10, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Yoshitake, T.; Shioyama, Y.; Asai, K.; Nakamura, K.; Sasaki, T.; Ohga, S.; Kamitani, T.; Yamaguchi, T.; Ohshima, K.; Matsumoto, K.; et al. Impact of Interstitial Changes on Radiation Pneumonitis after Stereotactic Body Radiation Therapy for Lung Cancer. Anticancer Res. 2015, 35, 4909–4913. [Google Scholar] [PubMed]
- Matsuo, Y.; Shibuya, K.; Nakamura, M.; Narabayashi, M.; Sakanaka, K.; Ueki, N.; Miyagi, K.; Norihisa, Y.; Mizowaki, T.; Nagata, Y.; et al. Dose—Volume metrics associated with radiation pneumonitis after stereotactic body radiation therapy for lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, e545–e549. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Hiraoka, M.; Shibata, T.; Narabayashi, M.; Sakanaka, K.; Ueki, N.; Miyagi, K.; Norihisa, Y.; Mizowaki, T.; Nagata, Y.; et al. Prospective Trial of Stereotactic Body Radiation Therapy for Both Operable and Inoperable T1N0M0 Non-Small Cell Lung Cancer: Japan Clinical Oncology Group Study JCOG0403. Int. J. Radiat. Oncol. Biol. Phys. 2015, 93, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, H.; Nakagawa, K.; Nakamura, N.; Koyanagi, H.; Tago, M.; Igaki, H.; Shiraishi, K.; Sasano, N.; Ohtomo, K. Exceptionally high incidence of symptomatic grade 2-5 radiation pneumonitis after stereotactic radiation therapy for lung tumors. Radiat. Oncol. 2007, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, R.; McGarry, R.; Yiannoutsos, C.; Papiez, L.; Tudor, K.; DeLuca, J.; Ewing, M.; Abdulrahman, R.; DesRosiers, C.; Williams, M.; et al. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J. Clin. Oncol. 2006, 24, 4833–4839. [Google Scholar] [CrossRef] [PubMed]
- Hope, A.J.; Lindsay, P.E.; El Naqa, I.; Alaly, J.R.; Vicic, M.; Bradley, J.D.; Deasy, J.O. Modeling radiation pneumonitis risk with clinical, dosimetric, and spatial parameters. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Ohashi, T.; Kunieda, E.; Enomoto, T.; Sanuki, N.; Takeda, T.; Shigematsu, N. Early graphical appearance of radiation pneumonitis correlates with the severity of radiation pneumonitis after stereotactic body radiotherapy (SBRT) in patients with lung tumors. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Collard, H.R.; Moore, B.B.; Flaherty, K.R.; Brown, K.K.; Kaner, R.J.; King, T.E., Jr.; Lasky, J.A.; Loyd, J.E.; Noth, I.; Olman, M.A.; et al. Acute exacerbations of idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2007, 176, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Enomoto, T.; Sanuki, N.; Nakajima, T.; Takeda, T.; Sayama, K.; Kunieda, E. Acute exacerbation of subclinical idiopathic pulmonary fibrosis triggered by hypofractionated stereotactic body radiotherapy in a patient with primary lung cancer and slightly focal honeycombing. Radiat. Med. 2008, 26, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.M.; Ao, X.; Wang, L.; Lawrence, T.S. The use of blood biomarkers to predict radiation lung toxicity: A potential strategy to individualize thoracic radiation therapy. Cancer Control 2008, 15, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Liao, Z.; Liu, Z.; Wang, L.E.; Tucker, S.L.; Mao, L.; Wang, X.S.; Martel, M.; Komaki, R.; Cox, J.D.; et al. Single nucleotide polymorphism at rs1982073: T869C of the TGFbeta 1 gene is associated with the risk of radiation pneumonitis in patients with non-small-cell lung cancer treated with definitive radiotherapy. J. Clin. Oncol. 2009, 27, 3370–3378. [Google Scholar] [CrossRef] [PubMed]
- Stromberg, J.S.; Sharpe, M.B.; Kim, L.H.; Kini, V.R.; Jaffray, D.A.; Martinez, A.A.; Wong, J.W. Active breathing control (ABC) for Hodgkin’s disease: Reduction in normal tissue irradiation with deep inspiration and implications for treatment. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 797–806. [Google Scholar] [CrossRef]
- Ebara, T.; Shimada, H.; Kawamura, H.; Shirai, K.; Saito, J.; Kawashima, M.; Tashiro, M.; Ohno, T.; Kanai, T.; Nakano, T. Dosimetric analysis between carbon ion radiotherapy and stereotactic body radiotherapy in stage I lung cancer. Anticancer Res. 2014, 34, 5099–5104. [Google Scholar] [PubMed]
- Tsurugai, Y.; Kozuka, T.; Ishizuka, N.; Oguchi, M. Relationship between the consolidation to maximum tumor diameter ratio and outcomes following stereotactic body radiotherapy for stage I non-small-cell lung cancer. Lung Cancer 2016, 92, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Teramukai, S.; Kondo, H.; Watanabe, A.; Ebina, M.; Kishi, K.; Fujii, Y.; Mitsudomi, T.; Yoshimura, M.; Maniwa, T.; et al. Impact and predictors of acute exacerbation of interstitial lung diseases after pulmonary resection for lung cancer. J. Thorac. Cardiovasc. Surg. 2014, 147, 1604–1611. [Google Scholar] [CrossRef] [PubMed]

| Total patients number | 242 |
| Gender | Male: 214, Female: 28 |
| Age | 55–92 (median: 77) y.o |
| Performance status (ECOG) | PS 0, 1, 2, 3, 4, NA * = 99, 114, 20, 2, 0, 7 |
| Histology | squamous cell cancer: 92 |
| adenocarcinoma: 77 | |
| unspecified non-small cell lung cancer: 18 | |
| unproven: 55 | |
| Tumor size | 3–50 mm (median: 27 mm) |
| T stage (7th UICC) | T1, T2 = 160, 82 |
| Medical operability | operable:54, inoperable:175, NA *: 13 |
| Smoking history | (+): 153, (−): 25, NA *: 64 |
| Pulmonary emphysema | (+): 87, (−): 118, NA *: 37 |
| Steroid administration before SBRT | (+): 20, (−): 174, NA *: 50 |
| % vital capacity (% VC) | 45.6–150.6% (mean: 91.9%) |
| FEV1.0% ** | 37.1–97.1% (mean: 67.9%) |
| SBRT dose | 40–70 Gy in 4–10 fractions |
| V20 *** | 1.1–21.7% (mean: 6.6%) |
| Variables (If Significant, Left Was Better) | Univariate | Multivariate |
|---|---|---|
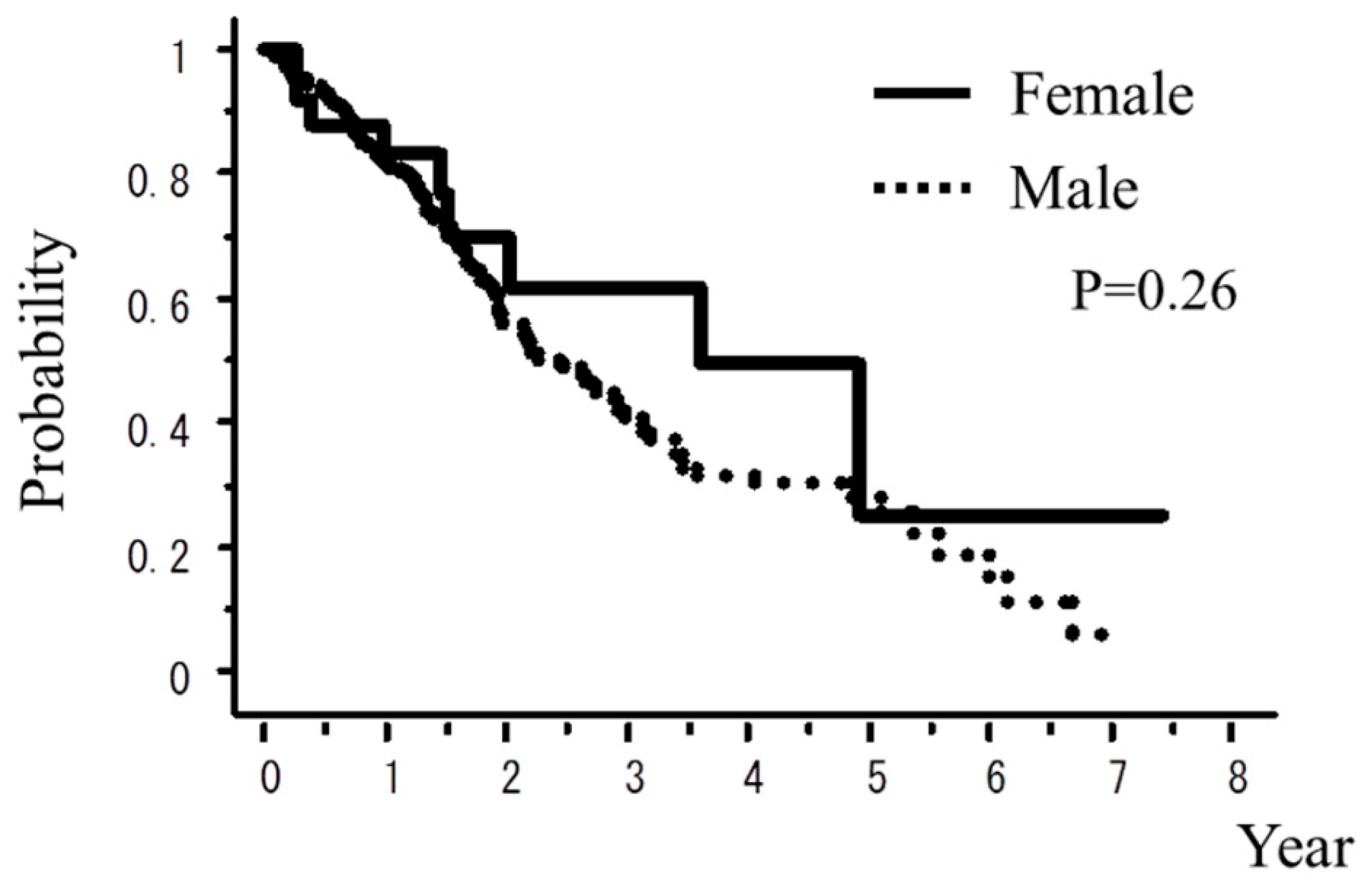
| Female vs. Male | 0.26 | 0.03 |
| Age <80 years vs. ≥80 years | 0.69 | 0.88 |
| Performance status 0,1 vs. 2,3,4 | 0.50 | 0.03 |
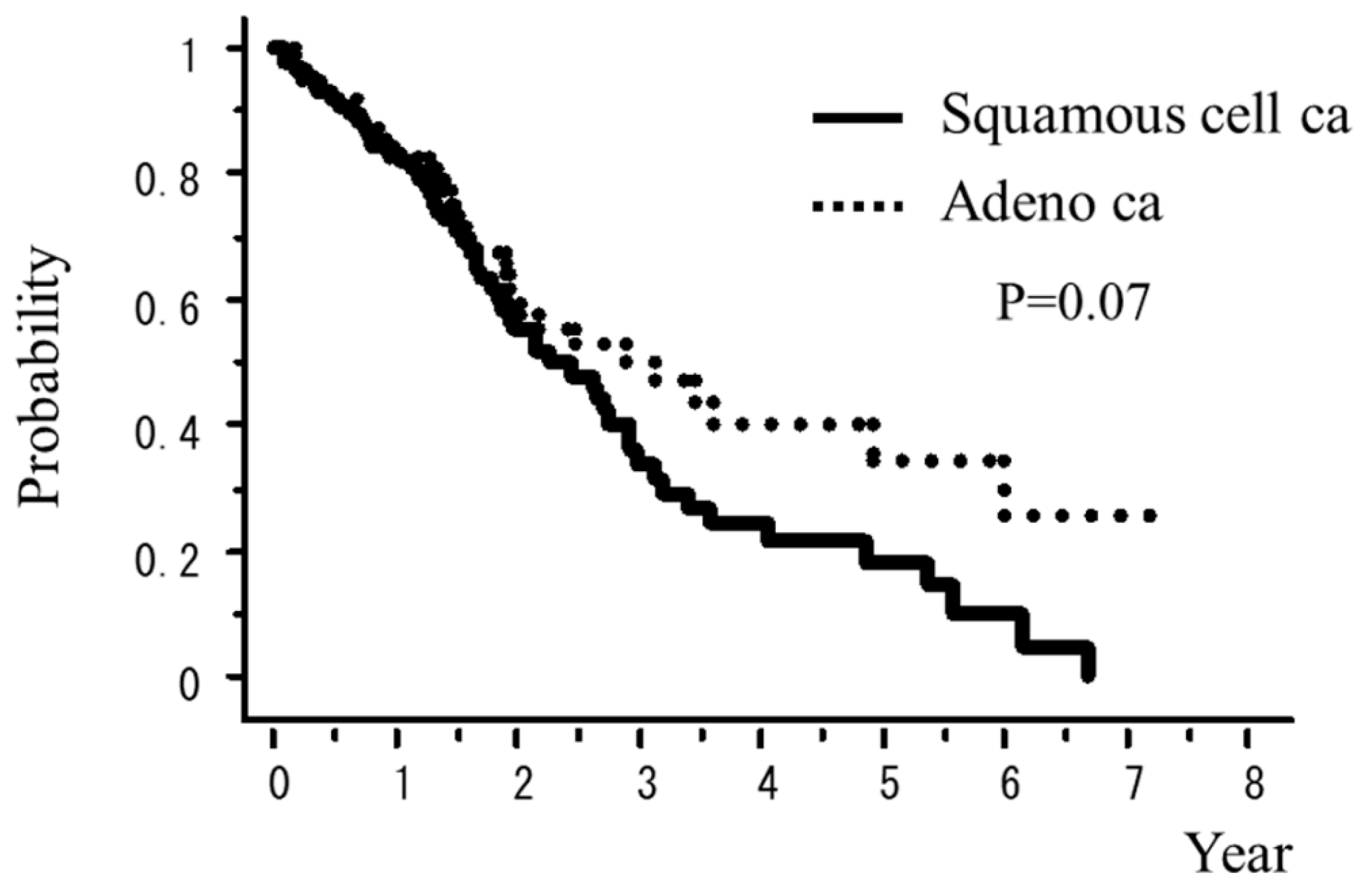
| Adeno ca vs. Squamous cell ca | 0.07 | <0.01 |
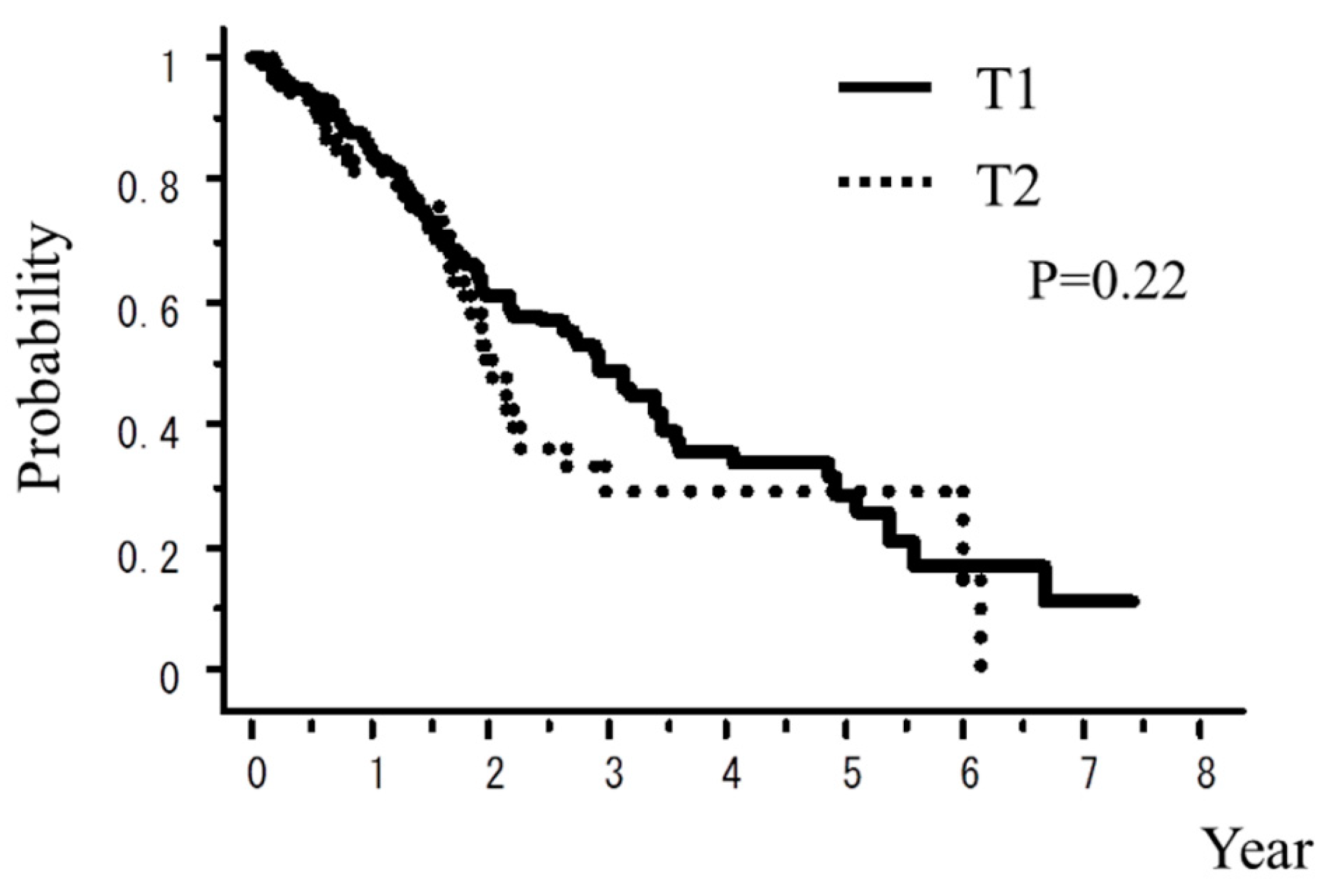
| T1 vs. T2 | 0.22 | 0.01 |
| Medical operable vs inoperable | 0.44 | 0.92 |
| Smoking history (−) vs. (+) | 0.33 | 0.10 |
| Pulmonary emphysema (−) vs. (+) | 0.24 | 0.13 |
| Steroid administration before SBRT (−) vs. (+) | 0.49 | <0.01 |
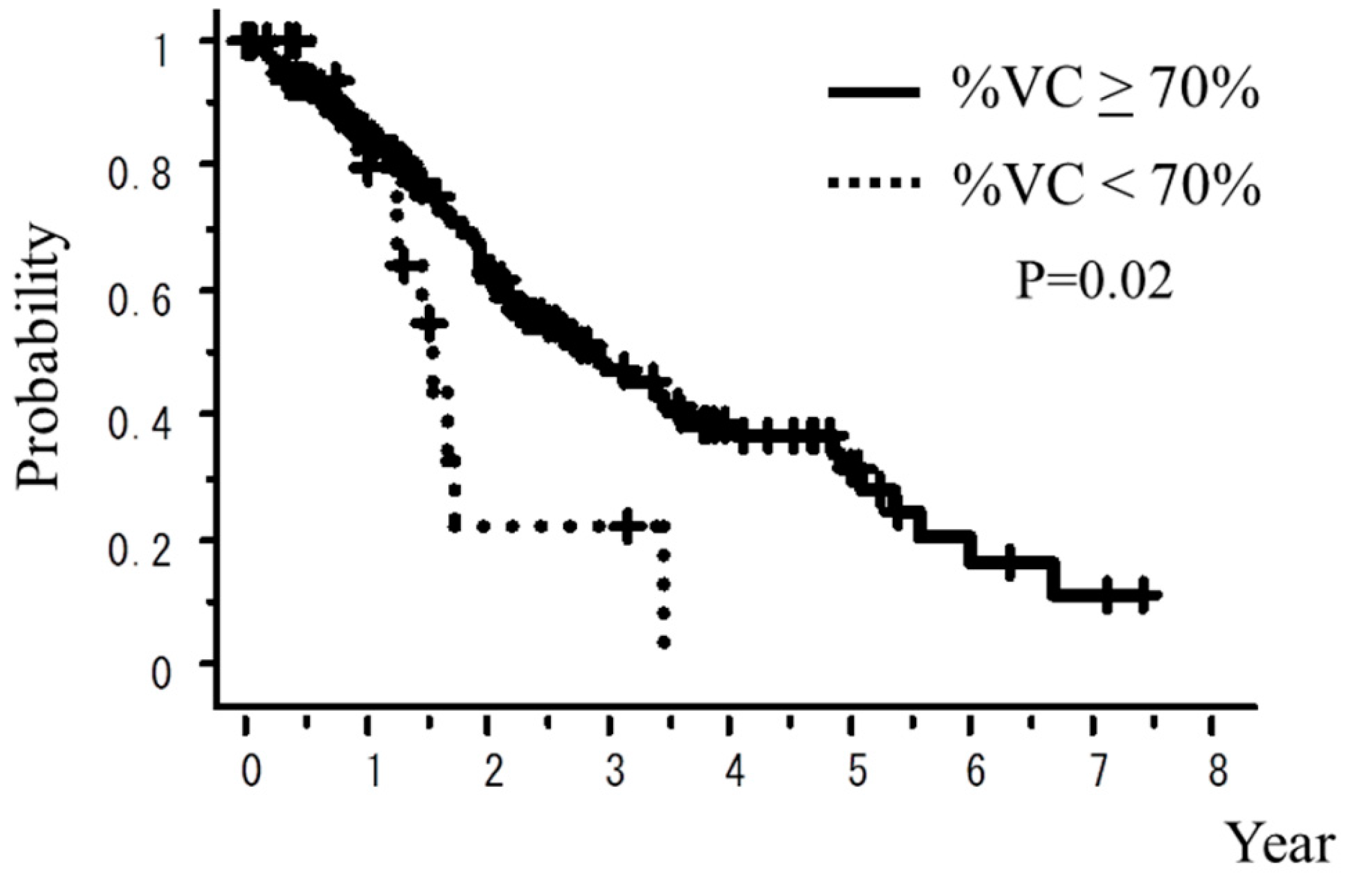
| % vital capacity (% VC) ≥70% vs. <70% | 0.02 | 0.60 |
| FEV1.0% *** ≥70% vs. <70% | 0.98 | <0.01 |
| Biological effective dose ≥100 Gy vs. <100 Gy | 0.13 | 0.90 |
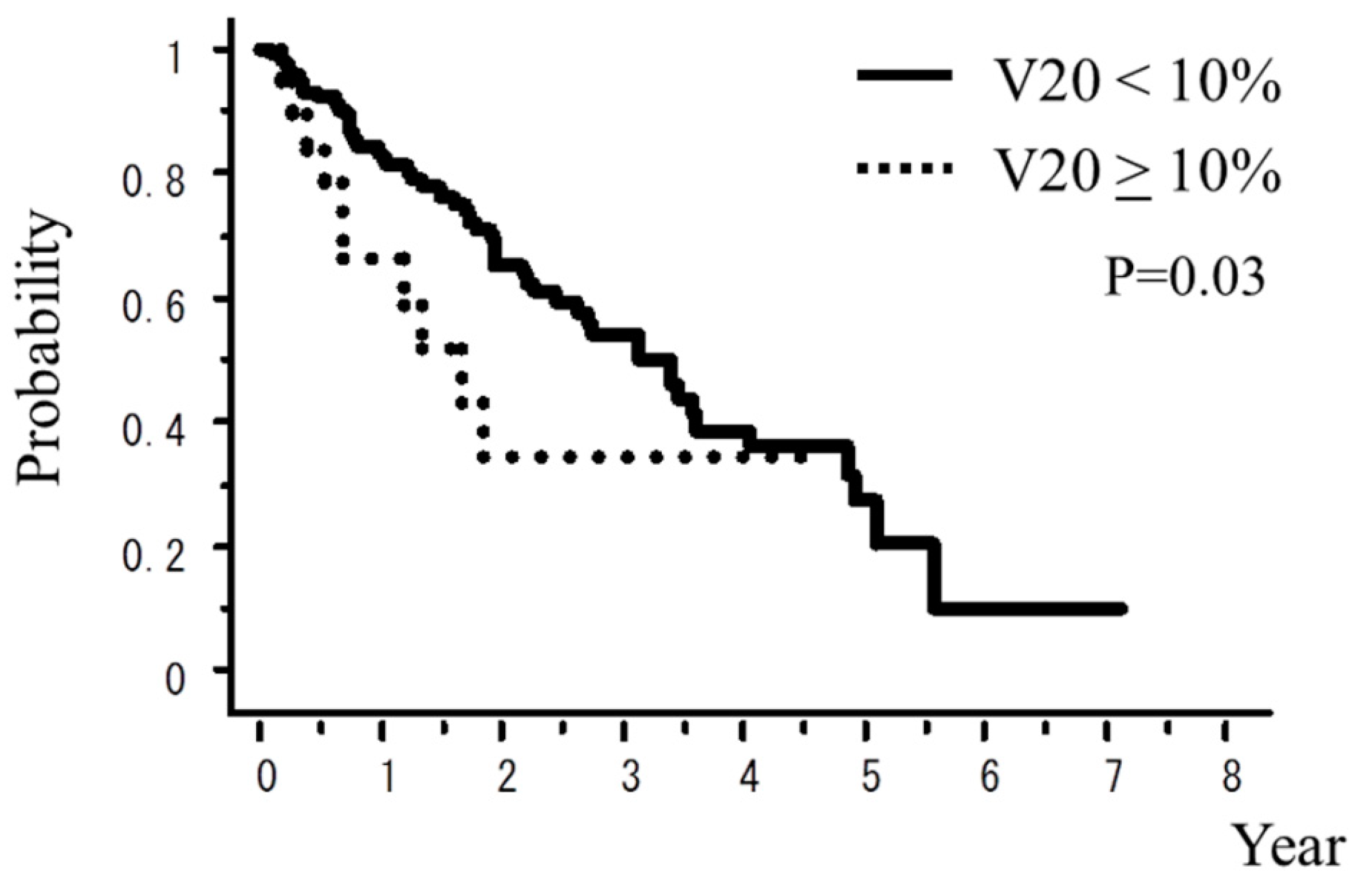
| V20 **** <10% vs. ≥10% | 0.03 | 0.13 |
| Patient Factors | Grade 3–5 | Grade 5 |
|---|---|---|
| Female vs. Male | 12.1% vs. 15.4% | 11.5% vs. 6.3% |
| Age <80 years vs. ≥80 years | 13.2% vs. 11.2% | 6.9% vs. 6.7% |
| Performance status 0,1 vs. 2,3,4 | 10.7% vs. 25.0% | 6.8% vs. 5.0% |
| Adeno ca vs. Squamous cell ca | 14.9% vs. 11.0% | 9.5% vs. 5.5% |
| T1 vs. T2 | 11.6% vs. 10.7% | 6.2% vs. 5.3% |
| Medical operable vs. inoperable | 5.7% vs. 15.5% | 5.7% vs. 7.7% |
| Smoking history (−) vs. (+) | 13.0% vs. 12.8% | 8.7% vs. 6.1% |
| Pulmonary emphysema (−) vs. (+) | 11.5% vs. 16.7% | 7.1% vs. 9.5% |
| Steroid administration before SBRT (−) vs. (+) | 12.6% vs. 21.1% | 7.8% vs. 5.3% |
| % vital capacity (%VC) ≥70% vs. <70% | 5.3% vs. 12.0% | 5.3% vs. 5.3% |
| FEV1.0% * ≥70% vs. <70% | 9.9% vs. 13.4% | 4.2% vs. 6.0% |
| Biological effective dose ≥100 Gy vs. <100 Gy | 11.8% vs. 16.7% | 6.9% vs. 6.7% |
| V20 ** < 10% vs. ≥10% | 11.1% vs. 29.4% | 6.0% vs. 28.6% |
| Pulmonary Interstitial Change | Author [Ref.] | Study Design | Patient Number | Dose/Fraction | Grade of Radiation Pneumonitis | Frequency | Risk Factor |
|---|---|---|---|---|---|---|---|
| No pulmonary interstitial change | Yamaguchi [3] | Retrospective | 86 | 48 Gy/4 fr | 4–5 | 0.00% | |
| Ueki N [4] | Retrospective | 137 | 48–60 Gy/4–8 fr | 3–5 | 1.40% | ||
| Yoshitake [5] | Retrospective | 242 | 48 Gy/4 fr | 3 | 1.20% | ||
| 4–5 | 0.00% | ||||||
| Matsuo Y [6] | Retrospective | 74 | 48 Gy/4 fr | 3 | 10.60% | V25 | |
| 4 | 1.90% | ||||||
| Nagata Y [7] | Prospective | 104 (inoperable) | 48 Gy/4 fr | 3 | 6.20% | ||
| 65 (operable) | 48 Gy/4 fr | 3 | 3.60% | ||||
| With pulmonary interstitial change | Yamaguchi [3] | Retrospective | 16 | 48 Gy/4 fr | 3 | 6.30% | V5–25, MLD |
| 4 | 6.30% | ||||||
| 5 | 6.30% | ||||||
| Ueki N [4] | Retrospective | 20 | 40–60 Gy/4–8 fr | 3–5 | 10.00% | ||
| Yoshitake [5] | Retrospective | 18 | 48 Gy/4 fr | 3 | 16.70% | KL-6, V5, V10, MLD | |
| 4–5 | 22.30% | ||||||
| This study | Retrospective | 242 | Various (mainly 48 Gy/4 fr) | 3–5 | 12.40% | % VC, FEV1.0 (%), Squamous cell ca., V20, PS, T stage, steroid before SBRT | |
| 5 | 6.90% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onishi, H.; Yamashita, H.; Shioyama, Y.; Matsumoto, Y.; Takayama, K.; Matsuo, Y.; Miyakawa, A.; Matsushita, H.; Aoki, M.; Nihei, K.; et al. Stereotactic Body Radiation Therapy for Patients with Pulmonary Interstitial Change: High Incidence of Fatal Radiation Pneumonitis in a Retrospective Multi-Institutional Study. Cancers 2018, 10, 257. https://doi.org/10.3390/cancers10080257
Onishi H, Yamashita H, Shioyama Y, Matsumoto Y, Takayama K, Matsuo Y, Miyakawa A, Matsushita H, Aoki M, Nihei K, et al. Stereotactic Body Radiation Therapy for Patients with Pulmonary Interstitial Change: High Incidence of Fatal Radiation Pneumonitis in a Retrospective Multi-Institutional Study. Cancers. 2018; 10(8):257. https://doi.org/10.3390/cancers10080257
Chicago/Turabian StyleOnishi, Hiroshi, Hideomi Yamashita, Yoshiyuki Shioyama, Yasuo Matsumoto, Kenji Takayama, Yukinori Matsuo, Akifumi Miyakawa, Haruo Matsushita, Masahiko Aoki, Keiji Nihei, and et al. 2018. "Stereotactic Body Radiation Therapy for Patients with Pulmonary Interstitial Change: High Incidence of Fatal Radiation Pneumonitis in a Retrospective Multi-Institutional Study" Cancers 10, no. 8: 257. https://doi.org/10.3390/cancers10080257
APA StyleOnishi, H., Yamashita, H., Shioyama, Y., Matsumoto, Y., Takayama, K., Matsuo, Y., Miyakawa, A., Matsushita, H., Aoki, M., Nihei, K., Kimura, T., Ishiyama, H., Murakami, N., Nakata, K., Takeda, A., Uno, T., Nomiya, T., Takanaka, T., Seo, Y., ... Kuriyama, K. (2018). Stereotactic Body Radiation Therapy for Patients with Pulmonary Interstitial Change: High Incidence of Fatal Radiation Pneumonitis in a Retrospective Multi-Institutional Study. Cancers, 10(8), 257. https://doi.org/10.3390/cancers10080257





