Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma
Abstract
1. Introduction
2. Sites of EOC Metastases
3. Mechanisms Regulating Transcoelomic Metastasis from EOC
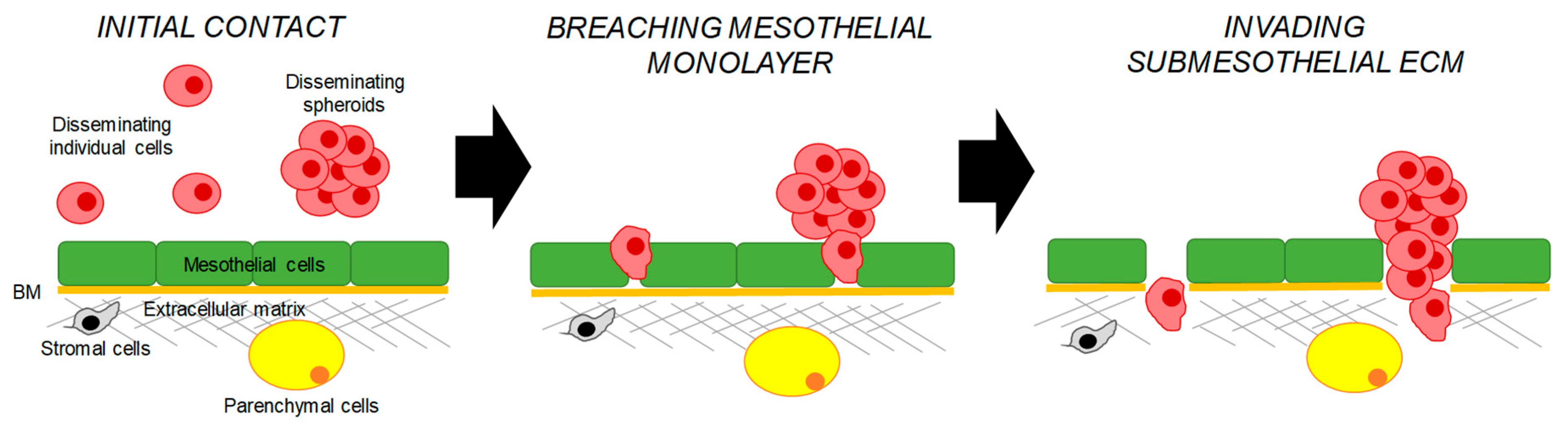
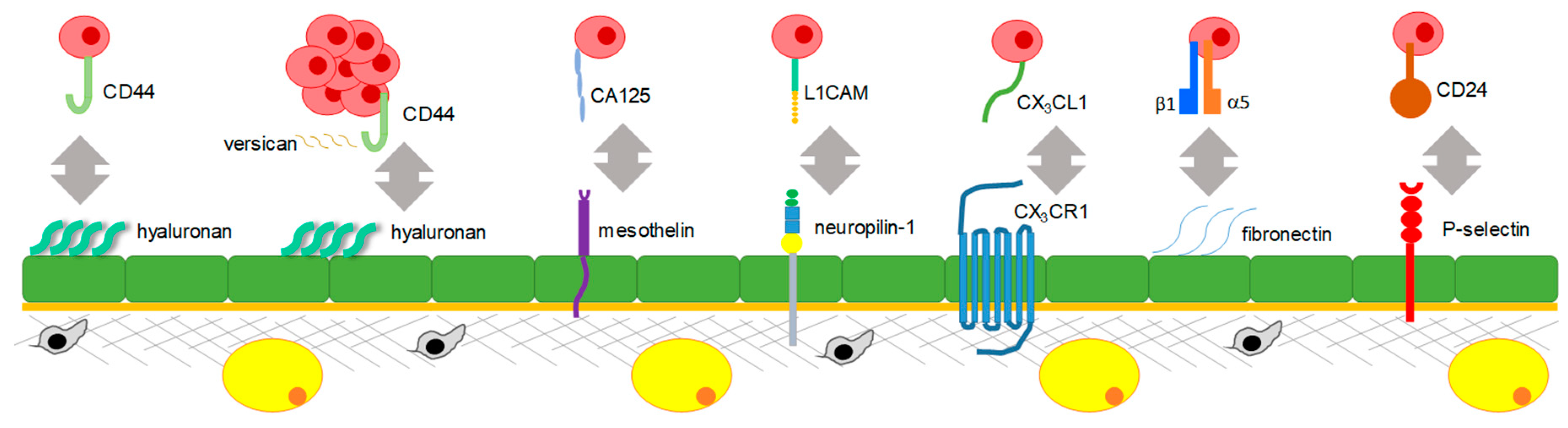
3.1. Mechanisms Regulating Peritoneal Adhesion
3.2. Mechanisms Regulating the Transcoelomic Omental Metastasis
3.3. Mechanisms Regulating Transcoelomic Metastasis to Other Intraperitoneal Organs and Tissues
4. Mechanisms Regulating Hematogenous Metastasis from EOC
5. Mechanisms Regulating Lymphatic Metastasis from EOC
6. Targeted Therapies in Ovarian Cancer
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Karnezis, A.N.; Cho, K.R. Of mice and women—Non-ovarian origins of “ovarian” cancer. Gynecol. Oncol. 2017, 144, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Karnezis, A.N.; Cho, K.R.; Gilks, C.B.; Pearce, C.L.; Huntsman, D.G. The disparate origins of ovarian cancers: Pathogenesis and prevention strategies. Nat. Rev. Cancer 2017, 17, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Kurman, R.J.; Ie, M.S. The Dualistic Model of Ovarian Carcinogenesis: Revisited, Revised, and Expanded. Am. J. Pathol. 2016, 186, 733–747. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.S.; Agarwal, R.; Kaye, S.B. Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 2006, 7, 925–934. [Google Scholar] [CrossRef]
- Whitney, C.W.; Spirtos, N. Gynecologic Oncology Group Surgical Procedures Manual; Gynecologic Oncology Group: Philadelphia, PA, USA, 2010. [Google Scholar]
- Shih, K.K.; Chi, D.S. Maximal cytoreductive effort in epithelial ovarian cancer surgery. J. Gynecol. Oncol. 2010, 21, 75–80. [Google Scholar] [CrossRef] [PubMed]
- du Bois, A.; Reuss, A.; Pujade-Lauraine, E.; Harter, P.; Ray-Coquard, I.; Pfisterer, J. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: A combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: By the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d’Investigateurs Nationaux Pour les Etudes des Cancers de l’Ovaire (GINECO). Cancer 2009, 115, 1234–1244. [Google Scholar] [PubMed]
- Wright, A.A.; Bohlke, K.; Armstrong, D.K.; Bookman, M.A.; Cliby, W.A.; Coleman, R.L.; Dizon, D.S.; Kash, J.J.; Meyer, L.A.; Moore, K.N.; et al. Neoadjuvant Chemotherapy for Newly Diagnosed, Advanced Ovarian Cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2016, 34, 3460–3473. [Google Scholar] [CrossRef] [PubMed]
- Vergote, I.; Trope, C.G.; Amant, F.; Kristensen, G.B.; Ehlen, T.; Johnson, N.; Verheijen, R.H.; van der Burg, M.E.; Lacave, A.J.; Panici, P.B.; et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 2010, 363, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Matulonis, U.A.; Sood, A.K.; Fallowfield, L.; Howitt, B.E.; Sehouli, J.; Karlan, B.Y. Ovarian cancer. Nat. Rev. Dis Primers 2016, 2, 16061. [Google Scholar] [CrossRef] [PubMed]
- Goldie, J.H.; Coldman, A.J. A mathematic model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat. Rep. 1979, 63, 1727–1733. [Google Scholar] [PubMed]
- Curley, M.D.; Garrett, L.A.; Schorge, J.O.; Foster, R.; Rueda, B.R. Evidence for cancer stem cells contributing to the pathogenesis of ovarian cancer. Front. Biosci. 2011, 16, 368–392. [Google Scholar] [CrossRef]
- Zhang, S.; Balch, C.; Chan, M.W.; Lai, H.C.; Matei, D.; Schilder, J.M.; Yan, P.S.; Huang, T.H.; Nephew, K.P. Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res. 2008, 68, 4311–4320. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.K. Relapsed ovarian cancer: Challenges and management strategies for a chronic disease. Oncologist 2002, 7 (Suppl. 5), 20–28. [Google Scholar] [CrossRef] [PubMed]
- Yim, G.W.; Eoh, K.J.; Kim, S.W.; Nam, E.J.; Kim, Y.T. Malnutrition Identified by the Nutritional Risk Index and Poor Prognosis in Advanced Epithelial Ovarian Carcinoma. Nutr. Cancer 2016, 68, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Balogun, N.; Forbes, A.; Widschwendter, M.; Lanceley, A. Noninvasive nutritional management of ovarian cancer patients: Beyond intestinal obstruction. Int. J. Gynecol. Cancer 2012, 22, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Gadducci, A.; Cosio, S.; Fanucchi, A.; Genazzani, A.R. Malnutrition and cachexia in ovarian cancer patients: Pathophysiology and management. Anticancer Res. 2001, 21, 2941–2947. [Google Scholar] [PubMed]
- Daniele, A.; Ferrero, A.; Fuso, L.; Mineccia, M.; Porcellana, V.; Vassallo, D.; Biglia, N.; Menato, G. Palliative care in patients with ovarian cancer and bowel obstruction. Support. Care Cancer 2015, 23, 3157–3163. [Google Scholar] [CrossRef] [PubMed]
- Aust, S.; Knogler, T.; Pils, D.; Obermayr, E.; Reinthaller, A.; Zahn, L.; Radlgruber, I.; Mayerhoefer, M.E.; Grimm, C.; Polterauer, S. Skeletal Muscle Depletion and Markers for Cancer Cachexia Are Strong Prognostic Factors in Epithelial Ovarian Cancer. PLoS ONE 2015, 10, e0140403. [Google Scholar] [CrossRef] [PubMed]
- Dell, D.D. Cachexia in patients with advanced cancer. Clin. J. Oncol. Nurs. 2002, 6, 235–238. [Google Scholar] [PubMed]
- Pradeep, S.; Kim, S.W.; Wu, S.Y.; Nishimura, M.; Chaluvally-Raghavan, P.; Miyake, T.; Pecot, C.V.; Kim, S.J.; Choi, H.J.; Bischoff, F.Z.; et al. Hematogenous metastasis of ovarian cancer: Rethinking mode of spread. Cancer Cell 2014, 26, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Coffman, L.G.; Burgos-Ojeda, D.; Wu, R.; Cho, K.; Bai, S.; Buckanovich, R.J. New models of hematogenous ovarian cancer metastasis demonstrate preferential spread to the ovary and a requirement for the ovary for abdominal dissemination. Transl. Res. 2016, 175, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Dvoretsky, P.M.; Richards, K.A.; Angel, C.; Rabinowitz, L.; Stoler, M.H.; Beecham, J.B.; Bonfiglio, T.A. Distribution of disease at autopsy in 100 women with ovarian cancer. Hum. Pathol. 1988, 19, 57–63. [Google Scholar] [CrossRef]
- Reed, E.; Zerbe, C.S.; Brawley, O.W.; Bicher, A.; Steinberg, S.M. Analysis of autopsy evaluations of ovarian cancer patients treated at the National Cancer Institute 1972–1988. Am. J. Clin. Oncol. 2000, 23, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.G.; Piver, M.S.; Tsukada, Y.; Lau, T.S. Metastatic patterns in histologic variants of ovarian cancer. An autopsy study. Cancer 1989, 64, 1508–1513. [Google Scholar] [CrossRef]
- Bachmann, C.; Bachmann, R.; Fend, F.; Wallwiener, D. Incidence and Impact of Lymph Node Metastases in Advanced Ovarian Cancer: Implications for Surgical Treatment. J. Cancer 2016, 7, 2241–2246. [Google Scholar] [CrossRef] [PubMed]
- Guth, U.; Arndt, V.; Stadlmann, S.; Huang, D.J.; Singer, G. Epidemiology in ovarian carcinoma: Lessons from autopsy. Gynecol. Oncol. 2015, 138, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Sahdev, A.; Hughes, J.H.; Barwick, T.; Rockall, A.G.; Gallagher, C.J.; Reznek, R.H. Computed tomography features of recurrent ovarian carcinoma according to time to relapse. Acta Radiol. 2007, 48, 1038–1044. [Google Scholar] [CrossRef] [PubMed]
- Hirose, S.; Tanabe, H.; Nagayoshi, Y.; Hirata, Y.; Narui, C.; Ochiai, K.; Isonishi, S.; Takano, H.; Okamoto, A. Retrospective analysis of sites of recurrence in stage I epithelial ovarian cancer. J. Gynecol. Oncol. 2018, 29, e37. [Google Scholar] [CrossRef] [PubMed]
- Guth, U.; Huang, D.; Bauer, G.; Stieger, M.; Wight, E.; Singer, G. Metastatic patterns at autopsy in patients with ovarian carcinoma. Cancer 2007, 110, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Cormio, G.; Rossi, C.; Cazzolla, A.; Resta, L.; Loverro, G.; Greco, P.; Selvaggi, L. Distant metastases in ovarian carcinoma. Int. J. Gynecol. Cancer 2003, 13, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Sood, A.K.; Sorosky, J.I.; Dolan, M.; Anderson, B.; Buller, R.E. Distant metastases in ovarian cancer: Association with p53 mutations. Clin. Cancer Res. 1999, 5, 2485–2490. [Google Scholar] [PubMed]
- Tanner, E.J.; Long, K.C.; Feffer, J.B.; Leitao, M.M., Jr.; Abu-Rustum, N.R.; Barakat, R.R.; Chi, D.S.; Gardner, G.J. Parenchymal splenic metastasis is an independent negative predictor of overall survival in advanced ovarian, fallopian tube, and primary peritoneal cancer. Gynecol. Oncol. 2013, 128, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Pakneshan, S.; Safarpour, D.; Tavassoli, F.; Jabbari, B. Brain metastasis from ovarian cancer: A systematic review. J. Neurooncol. 2014, 119, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pectasides, D.; Pectasides, M.; Economopoulos, T. Brain metastases from epithelial ovarian cancer: A review of the literature. Oncologist 2006, 11, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef] [PubMed]
- Al Habyan, S.; Kalos, C.; Szymborski, J.; McCaffrey, L. Multicellular detachment generates metastatic spheroids during intra-abdominal dissemination in epithelial ovarian cancer. Oncogene 2018, 37, 5127–5135. [Google Scholar] [CrossRef] [PubMed]
- Pease, J.C.; Brewer, M.; Tirnauer, J.S. Spontaneous spheroid budding from monolayers: A potential contribution to ovarian cancer dissemination. Biol. Open 2012, 1, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Moss, N.M.; Barbolina, M.V.; Liu, Y.; Sun, L.; Munshi, H.G.; Stack, M.S. Ovarian cancer cell detachment and multicellular aggregate formation are regulated by membrane type 1 matrix metalloproteinase: A potential role in I.p. metastatic dissemination. Cancer Res. 2009, 69, 7121–7129. [Google Scholar] [CrossRef] [PubMed]
- Steinkamp, M.P.; Winner, K.K.; Davies, S.; Muller, C.; Zhang, Y.; Hoffman, R.M.; Shirinifard, A.; Moses, M.; Jiang, Y.; Wilson, B.S. Ovarian tumor attachment, invasion, and vascularization reflect unique microenvironments in the peritoneum: Insights from xenograft and mathematical models. Front. Oncol. 2013, 3, 97. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Petignat, P. The bright side of ascites in ovarian cancer. Cell. Cycle 2014, 13, 2319. [Google Scholar] [CrossRef] [PubMed]
- Kipps, E.; Tan, D.S.; Kaye, S.B. Meeting the challenge of ascites in ovarian cancer: New avenues for therapy and research. Nat. Rev. Cancer 2013, 13, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Stenvers, K.L. Getting to know ovarian cancer ascites: Opportunities for targeted therapy-based translational research. Front. Oncol. 2013, 3, 256. [Google Scholar] [CrossRef] [PubMed]
- Sorbe, B.; Frankendal, B. Prognostic importance of ascites in ovarian carcinoma. Acta Obstet. Gynecol. Scand. 1983, 62, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Sigurdsson, K.; Alm, P.; Gullberg, B. Prognostic factors in malignant epithelial ovarian tumors. Gynecol. Oncol. 1983, 15, 370–380. [Google Scholar] [CrossRef]
- Ansell, S.M.; Rapoport, B.L.; Falkson, G.; Raats, J.I.; Moeken, C.M. Survival determinants in patients with advanced ovarian cancer. Gynecol. Oncol. 1993, 50, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Roszkowski, P.; Wronkowski, Z.; Szamborski, J.; Romejko, M. Evaluation of selected prognostic factors in ovarian cancer. Eur. J. Gynaecol. Oncol. 1993, 14, 140–145. [Google Scholar] [PubMed]
- Kim, S.; Kim, B.; Song, Y.S. Ascites modulates cancer cell behavior, contributing to tumor heterogeneity in ovarian cancer. Cancer Sci. 2016, 107, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Yung, S.; Li, F.K.; Chan, T.M. Peritoneal mesothelial cell culture and biology. Perit. Dial. Int. 2006, 26, 162–173. [Google Scholar] [PubMed]
- Jones, L.M.; Gardner, M.J.; Catterall, J.B.; Turner, G.A. Hyaluronic acid secreted by mesothelial cells: A natural barrier to ovarian cancer cell adhesion. Clin. Exp. Metastasis 1995, 13, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Yung, S.; Chan, T.M. Pathophysiology of the peritoneal membrane during peritoneal dialysis: The role of hyaluronan. J. Biomed. Biotechnol. 2011, 2011, 180594. [Google Scholar] [CrossRef] [PubMed]
- Howlader, N.; Krapcho, A.M.; Miller, D.; Bishop, K.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. (Eds.) SEER Cancer Statistics Review, 1975–2014; National Cancer Institute: Bethesda, MD, USA, 2017.
- Mikula-Pietrasik, J.; Stryczynski, L.; Uruski, P.; Tykarski, A.; Ksiazek, K. Procancerogenic activity of senescent cells: A case of the peritoneal mesothelium. Ageing Res. Rev. 2018, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Loughran, E.A.; Leonard, A.K.; Hilliard, T.S.; Phan, R.C.; Yemc, M.G.; Harper, E.; Sheedy, E.; Klymenko, Y.; Asem, M.; Liu, Y.; et al. Aging Increases Susceptibility to Ovarian Cancer Metastasis in Murine Allograft Models and Alters Immune Composition of Peritoneal Adipose Tissue. Neoplasia 2018, 20, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Sawada, K.; Kinose, Y.; Yoshimura, A.; Toda, A.; Nakatsuka, E.; Hashimoto, K.; Mabuchi, S.; Morishige, K.I.; Kurachi, H.; et al. Exosomes Promote Ovarian Cancer Cell Invasion through Transfer of CD44 to Peritoneal Mesothelial Cells. Mol. Cancer Res. 2017, 15, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Xiao, Y.J.; Singh, L.S.; Zhao, X.; Zhao, Z.; Feng, L.; Rose, T.M.; Prestwich, G.D.; Xu, Y. Lysophosphatidic acid is constitutively produced by human peritoneal mesothelial cells and enhances adhesion, migration, and invasion of ovarian cancer cells. Cancer Res. 2006, 66, 3006–3014. [Google Scholar] [CrossRef] [PubMed]
- Cannistra, S.A.; Kansas, G.S.; Niloff, J.; DeFranzo, B.; Kim, Y.; Ottensmeier, C. Binding of ovarian cancer cells to peritoneal mesothelium in vitro is partly mediated by CD44H. Cancer Res. 1993, 53, 3830–3838. [Google Scholar] [PubMed]
- Gardner, M.J.; Catterall, J.B.; Jones, L.M.; Turner, G.A. Human ovarian tumour cells can bind hyaluronic acid via membrane CD44: A possible step in peritoneal metastasis. Clin. Exp. Metastasis 1996, 14, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Yakubov, B.; Chelladurai, B.; Schmitt, J.; Emerson, R.; Turchi, J.J.; Matei, D. Extracellular tissue transglutaminase activates noncanonical NF-kappaB signaling and promotes metastasis in ovarian cancer. Neoplasia 2013, 15, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, M.; Xie, J.; Gurler, H.; Muralidhar, G.G.; Sacks, J.D.; Burdette, J.E.; Barbolina, M.V. Versican regulates metastasis of epithelial ovarian carcinoma cells and spheroids. J. Ovarian Res. 2014, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Ween, M.P.; Hummitzsch, K.; Rodgers, R.J.; Oehler, M.K.; Ricciardelli, C. Versican induces a pro-metastatic ovarian cancer cell behavior which can be inhibited by small hyaluronan oligosaccharides. Clin. Exp. Metastasis 2011, 28, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Strobel, T.; Swanson, L.; Cannistra, S.A. In vivo inhibition of CD44 limits intra-abdominal spread of a human ovarian cancer xenograft in nude mice: A novel role for CD44 in the process of peritoneal implantation. Cancer Res. 1997, 57, 1228–1232. [Google Scholar] [PubMed]
- Rump, A.; Morikawa, Y.; Tanaka, M.; Minami, S.; Umesaki, N.; Takeuchi, M.; Miyajima, A. Binding of ovarian cancer antigen CA125/MUC16 to mesothelin mediates cell adhesion. J. Biol. Chem. 2004, 279, 9190–9198. [Google Scholar] [CrossRef] [PubMed]
- Gubbels, J.A.; Belisle, J.; Onda, M.; Rancourt, C.; Migneault, M.; Ho, M.; Bera, T.K.; Connor, J.; Sathyanarayana, B.K.; Lee, B.; et al. Mesothelin-MUC16 binding is a high affinity, N-glycan dependent interaction that facilitates peritoneal metastasis of ovarian tumors. Mol. Cancer 2006, 5, 50. [Google Scholar] [CrossRef] [PubMed]
- Stoeck, A.; Schlich, S.; Issa, Y.; Gschwend, V.; Wenger, T.; Herr, I.; Marme, A.; Bourbie, S.; Altevogt, P.; Gutwein, P. L1 on ovarian carcinoma cells is a binding partner for Neuropilin-1 on mesothelial cells. Cancer Lett. 2006, 239, 212–226. [Google Scholar] [CrossRef] [PubMed]
- Gurler Main, H.; Xie, J.; Muralidhar, G.G.; Elfituri, O.; Xu, H.; Kajdacsy-Balla, A.A.; Barbolina, M.V. Emergent role of the fractalkine axis in dissemination of peritoneal metastasis from epithelial ovarian carcinoma. Oncogene 2017, 36, 3025–3036. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Rooper, L.; Xie, J.; Kajdacsy-Balla, A.A.; Barbolina, M.V. Fractalkine receptor CX(3)CR1 is expressed in epithelial ovarian carcinoma cells and required for motility and adhesion to peritoneal mesothelial cells. Mol. Cancer Res. 2012, 10, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Strobel, T.; Cannistra, S.A. Beta1-integrins partly mediate binding of ovarian cancer cells to peritoneal mesothelium in vitro. Gynecol. Oncol. 1999, 73, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Kenny, H.A.; Chiang, C.Y.; White, E.A.; Schryver, E.M.; Habis, M.; Romero, I.L.; Ladanyi, A.; Penicka, C.V.; George, J.; Matlin, K.; et al. Mesothelial cells promote early ovarian cancer metastasis through fibronectin secretion. J. Clin. Investig. 2014, 124, 4614–4628. [Google Scholar] [CrossRef] [PubMed]
- Carroll, M.J.; Fogg, K.C.; Patel, H.A.; Krause, H.B.; Mancha, A.S.; Patankar, M.S.; Weisman, P.S.; Barroilhet, L.; Kreeger, P.K. Alternatively-Activated Macrophages Upregulate Mesothelial Expression of P-Selectin to Enhance Adhesion of Ovarian Cancer Cells. Cancer Res. 2018, 78, 3560–3573. [Google Scholar] [CrossRef] [PubMed]
- Gharpure, K.M.; Lara, O.D.; Wen, Y.; Pradeep, S.; LaFargue, C.; Ivan, C.; Rupaimoole, R.; Hu, W.; Mangala, L.S.; Wu, S.Y.; et al. ADH1B promotes mesothelial clearance and ovarian cancer infiltration. Oncotarget 2018, 9, 25115–25126. [Google Scholar] [CrossRef] [PubMed]
- Iwanicki, M.P.; Davidowitz, R.A.; Ng, M.R.; Besser, A.; Muranen, T.; Merritt, M.; Danuser, G.; Ince, T.A.; Brugge, J.S. Ovarian cancer spheroids use myosin-generated force to clear the mesothelium. Cancer Discov. 2011, 1, 144–157. [Google Scholar] [CrossRef] [PubMed]
- Roy, L.; Bobbs, A.; Sattler, R.; Kurkewich, J.L.; Dausinas, P.B.; Nallathamby, P.; Cowden Dahl, K.D. CD133 Promotes Adhesion to the Ovarian Cancer Metastatic Niche. Cancer Growth Metastasis 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Klymenko, Y.; Kim, O.; Loughran, E.; Yang, J.; Lombard, R.; Alber, M.; Stack, M.S. Cadherin composition and multicellular aggregate invasion in organotypic models of epithelial ovarian cancer intraperitoneal metastasis. Oncogene 2017, 36, 5840–5851. [Google Scholar] [CrossRef] [PubMed]
- Ellerbroek, S.M.; Wu, Y.I.; Overall, C.M.; Stack, M.S. Functional interplay between type I collagen and cell surface matrix metalloproteinase activity. J. Biol. Chem. 2001, 276, 24833–24842. [Google Scholar] [CrossRef] [PubMed]
- Moser, T.L.; Pizzo, S.V.; Bafetti, L.M.; Fishman, D.A.; Stack, M.S. Evidence for preferential adhesion of ovarian epithelial carcinoma cells to type I collagen mediated by the alpha2beta1 integrin. Int. J. Cancer 1996, 67, 695–701. [Google Scholar] [CrossRef]
- Sodek, K.L.; Ringuette, M.J.; Brown, T.J. MT1-MMP is the critical determinant of matrix degradation and invasion by ovarian cancer cells. Br. J. Cancer 2007, 97, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Barbolina, M.V.; Adley, B.P.; Ariztia, E.V.; Liu, Y.; Stack, M.S. Microenvironmental Regulation of Membrane Type 1 Matrix Metalloproteinase Activity in Ovarian Carcinoma Cells via Collagen-induced EGR1 Expression. J. Biol. Chem. 2007, 282, 4924–4931. [Google Scholar] [CrossRef] [PubMed]
- Barbolina, M.V.; Liu, Y.; Gurler, H.; Kim, M.; Kajdacsy-Balla, A.A.; Rooper, L.; Shepard, J.; Weiss, M.; Shea, L.D.; Penzes, P.; et al. Matrix Rigidity Activates Wnt Signaling through Down-regulation of Dickkopf-1 Protein. J. Biol. Chem. 2013, 288, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Moss, N.M.; Liu, Y.; Johnson, J.J.; Debiase, P.; Jones, J.; Hudson, L.G.; Munshi, H.G.; Stack, M.S. Epidermal growth factor receptor-mediated membrane type 1 matrix metalloproteinase endocytosis regulates the transition between invasive versus expansive growth of ovarian carcinoma cells in three-dimensional collagen. Mol. Cancer Res. 2009, 7, 809–820. [Google Scholar] [CrossRef] [PubMed]
- Yoo, E.; Kim, J.H.; Kim, M.J.; Yu, J.S.; Chung, J.J.; Yoo, H.S.; Kim, K.W. Greater and lesser omenta: Normal anatomy and pathologic processes. Radiographics 2007, 27, 707–720. [Google Scholar] [CrossRef] [PubMed]
- Meza-Perez, S.; Randall, T.D. Immunological Functions of the Omentum. Trends Immunol. 2017, 38, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Platell, C.; Cooper, D.; Papadimitriou, J.M.; Hall, J.C. The omentum. World J. Gastroenterol. 2000, 6, 169–176. [Google Scholar] [PubMed]
- Nieman, K.M.; Kenny, H.A.; Penicka, C.V.; Ladanyi, A.; Buell-Gutbrod, R.; Zillhardt, M.R.; Romero, I.L.; Carey, M.S.; Mills, G.B.; Hotamisligil, G.S.; et al. Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nat. Med. 2011, 17, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.M.; Funk, H.M.; Thiolloy, S.; Lotan, T.L.; Hickson, J.; Prins, G.S.; Drew, A.F.; Rinker-Schaeffer, C.W. In vitro metastatic colonization of human ovarian cancer cells to the omentum. Clin. Exp. Metastasis 2010, 27, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Arie, A.B.; McNally, L.; Kapp, D.S.; Teng, N.N. The omentum and omentectomy in epithelial ovarian cancer: A reappraisal: Part II--The role of omentectomy in the staging and treatment of apparent early stage epithelial ovarian cancer. Gynecol. Oncol. 2013, 131, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Kawana, K.; Adachi, K.; Fujimoto, A.; Yoshida, M.; Nakamura, H.; Nishida, H.; Inoue, T.; Taguchi, A.; Ogishima, J.; et al. Detachment from the primary site and suspension in ascites as the initial step in metabolic reprogramming and metastasis to the omentum in ovarian cancer. Oncol. Lett. 2018, 15, 1357–1361. [Google Scholar] [CrossRef] [PubMed]
- Yung, M.M.; Tang, H.W.; Cai, P.C.; Leung, T.H.; Ngu, S.F.; Chan, K.K.; Xu, D.; Yang, H.; Ngan, H.Y.; Chan, D.W. GRO-alpha and IL-8 enhance ovarian cancer metastatic potential via the CXCR2-mediated TAK1/NFkappaB signaling cascade. Theranostics 2018, 8, 1270–1285. [Google Scholar] [CrossRef] [PubMed]
- Shimotsuma, M.; Shields, J.W.; Simpson-Morgan, M.W.; Sakuyama, A.; Shirasu, M.; Hagiwara, A.; Takahashi, T. Morpho-physiological function and role of omental milky spots as omentum-associated lymphoid tissue (OALT) in the peritoneal cavity. Lymphology 1993, 26, 90–101. [Google Scholar] [PubMed]
- Clark, R.; Krishnan, V.; Schoof, M.; Rodriguez, I.; Theriault, B.; Chekmareva, M.; Rinker-Schaeffer, C. Milky spots promote ovarian cancer metastatic colonization of peritoneal adipose in experimental models. Am. J. Pathol. 2013, 183, 576–591. [Google Scholar] [CrossRef] [PubMed]
- Ladanyi, A.; Mukherjee, A.; Kenny, H.A.; Johnson, A.; Mitra, A.K.; Sundaresan, S.; Nieman, K.M.; Pascual, G.; Benitah, S.A.; Montag, A.; et al. Adipocyte-induced CD36 expression drives ovarian cancer progression and metastasis. Oncogene 2018, 37, 2285–2301. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.K.; Chiang, C.Y.; Tiwari, P.; Tomar, S.; Watters, K.M.; Peter, M.E.; Lengyel, E. Microenvironment-induced downregulation of miR-193b drives ovarian cancer metastasis. Oncogene 2015, 34, 5923–5932. [Google Scholar] [CrossRef] [PubMed]
- Lau, T.S.; Chan, L.K.; Wong, E.C.; Hui, C.W.; Sneddon, K.; Cheung, T.H.; Yim, S.F.; Lee, J.H.; Yeung, C.S.; Chung, T.K.; et al. A loop of cancer-stroma-cancer interaction promotes peritoneal metastasis of ovarian cancer via TNFalpha-TGFalpha-EGFR. Oncogene 2017, 36, 3576–3587. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F. Cancer and the chemokine network. Nat. Rev. Cancer 2004, 4, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Proudfoot, A.E. Chemokine receptors: Multifaceted therapeutic targets. Nat. Rev. Immunol. 2002, 2, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Scotton, C.J.; Wilson, J.L.; Scott, K.; Stamp, G.; Wilbanks, G.D.; Fricker, S.; Bridger, G.; Balkwill, F.R. Multiple actions of the chemokine CXCL12 on epithelial tumor cells in human ovarian cancer. Cancer Res. 2002, 62, 5930–5938. [Google Scholar] [PubMed]
- Barbolina, M.V.; Kim, M.; Liu, Y.; Shepard, J.; Belmadani, A.; Miller, R.J.; Shea, L.D.; Stack, M.S. Microenvironmental regulation of chemokine (C-X-C-motif) receptor 4 in ovarian carcinoma. Mol. Cancer Res. 2010, 8, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Righi, E.; Kashiwagi, S.; Yuan, J.; Santosuosso, M.; Leblanc, P.; Ingraham, R.; Forbes, B.; Edelblute, B.; Collette, B.; Xing, D.; et al. CXCL12/CXCR4 Blockade Induces Multimodal Antitumor Effects that Prolong Survival in an Immunocompetent Mouse Model of Ovarian Cancer. Cancer Res. 2011, 71, 5522–5534. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Rooper, L.; Xie, J.; Rayahin, J.; Burdette, J.E.; Kajdacsy-Balla, A.; Barbolina, M.V. The Lymphotactin Receptor Is Expressed in Epithelial Ovarian Carcinoma and Contributes to Cell Migration and Proliferation. Mol. Cancer Res. 2012. [Google Scholar] [CrossRef] [PubMed]
- Holcroft, J.; Kressel, H.Y.; Prager, R.; Trunkey, D.; Jacobs, R.P. An experience with a LeVeen ascites shunt. Arch. Surg. 1976, 111, 302–303. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, G.F.; Stanley, M.M. Peritoneovenous shunting for ascites. Surg. Gynecol. Obstet. 1977, 145, 419–424. [Google Scholar] [PubMed]
- White, M.A.; Agle, S.C.; Padia, R.K.; Zervos, E.E. Denver peritoneovenous shunts for the management of malignant ascites: A review of the literature in the post LeVeen Era. Am. Surg. 2011, 77, 1070–1075. [Google Scholar] [PubMed]
- Souter, R.G.; Wells, C.; Tarin, D.; Kettlewell, M.G. Surgical and pathologic complications associated with peritoneovenous shunts in management of malignant ascites. Cancer 1985, 55, 1973–1978. [Google Scholar] [CrossRef]
- Tarin, D.; Price, J.E.; Kettlewell, M.G.; Souter, R.G.; Vass, A.C.; Crossley, B. Clinicopathological observations on metastasis in man studied in patients treated with peritoneovenous shunts. Br. Med. J. (Clin. Res. Ed.) 1984, 288, 749–751. [Google Scholar] [CrossRef]
- Tarin, D.; Price, J.E.; Kettlewell, M.G.W.; Souter, R.G.; Vass, A.C.R.; Crossley, B. Mechanisms of Human-Tumor Metastasis Studied in Patients with Peritoneovenous Shunts. Cancer Res. 1984, 44, 3584–3592. [Google Scholar] [PubMed]
- Tarin, D.; Vass, A.C.; Kettlewell, M.G.; Price, J.E. Absence of metastatic sequelae during long-term treatment of malignant ascites by peritoneo-venous shunting. A clinico-pathological report. Invasion Metastasis 1984, 4, 1–12. [Google Scholar] [PubMed]
- Matsuo, K.; Carter, C.M.; Ahn, E.H.; Prather, C.P.; Eno, M.L.; Im, D.D.; Rosenshein, N.B. Inferior vena cava filter placement and risk of hematogenous distant metastasis in ovarian cancer. Am. J. Clin. Oncol. 2013, 36, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Kamran, P.; Sereti, K.I.; Zhao, P.; Ali, S.R.; Weissman, I.L.; Ardehali, R. Parabiosis in mice: A detailed protocol. J. Vis. Exp. 2013, 80, e50556. [Google Scholar] [CrossRef] [PubMed]
- Figueras, A.; Alsina-Sanchis, E.; Lahiguera, A.; Abreu, M.; Muinelo-Romay, L.; Moreno-Bueno, G.; Casanovas, O.; Graupera, M.; Matias-Guiu, X.; Vidal, A.; et al. A Role for CXCR4 in Peritoneal and Hematogenous Ovarian Cancer Dissemination. Mol. Cancer Ther. 2018, 17, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Berek, J.S.; Kehoe, S.T.; Kumar, L.; Friedlander, M. Cancer of the ovary, fallopian tube, and peritoneum. Int J. Gynaecol. Obstet. 2018, 143 (Suppl. 2), 59–78. [Google Scholar] [CrossRef] [PubMed]
- Tammela, T.; Alitalo, K. Lymphangiogenesis: Molecular mechanisms and future promise. Cell 2010, 140, 460–476. [Google Scholar] [CrossRef] [PubMed]
- Kuerti, S.; Oliveira-Ferrer, L.; Milde-Langosch, K.; Schmalfeldt, B.; Legler, K.; Woelber, L.; Prieske, K.; Mahner, S.; Trillsch, F. VEGF-C expression attributes the risk for lymphatic metastases to ovarian cancer patients. Oncotarget 2017, 8, 43218–43227. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Yu, N. Ubiquitin-specific protease 7 expression is a prognostic factor in epithelial ovarian cancer and correlates with lymph node metastasis. OncoTargets Ther. 2016, 9, 1559–1569. [Google Scholar]
- Kang, Y.; Pu, T.; Cai, Q.; Hong, S.; Zhang, M.; Li, G.; Zhu, Z.; Xu, C. Identification of lymphatic metastasis-associated genes in a metastatic ovarian cancer cell line. Mol. Med. Rep. 2015, 12, 2741–2748. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hermanson, D.L.; Moriarity, B.S.; Kaufman, D.S. Human iPSC-Derived Natural Killer Cells Engineered with Chimeric Antigen Receptors Enhance Anti-tumor Activity. Cell Stem Cell 2018, 23, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Golfier, S.; Kopitz, C.; Kahnert, A.; Heisler, I.; Schatz, C.A.; Stelte-Ludwig, B.; Mayer-Bartschmid, A.; Unterschemmann, K.; Bruder, S.; Linden, L.; et al. Anetumab ravtansine: A novel mesothelin-targeting antibody-drug conjugate cures tumors with heterogeneous target expression favored by bystander effect. Mol. Cancer Ther. 2014, 13, 1537–1548. [Google Scholar] [CrossRef] [PubMed]
- Bell-McGuinn, K.M.; Matthews, C.M.; Ho, S.N.; Barve, M.; Gilbert, L.; Penson, R.T.; Lengyel, E.; Palaparthy, R.; Gilder, K.; Vassos, A.; et al. A phase II, single-arm study of the anti-alpha5beta1 integrin antibody volociximab as monotherapy in patients with platinum-resistant advanced epithelial ovarian or primary peritoneal cancer. Gynecol. Oncol. 2011, 121, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Delmonte, A.; Del Conte, G.; Sessa, C.; Perotti, A.; Fasolo, A.; Williams, E.; Smith, K.; Dall’O’, E.; Colombo, N.; Gianni, L. Results from a phase 1/2 study of volociximab in combination with liposomal doxorubicin in relapsed advanced epithelial ovarian and primary peritoneal carcinoma. J. Clin. Oncol. 2008, 26, 16527. [Google Scholar] [CrossRef]
- Reyes, H.D.; Thiel, K.W.; Carlson, M.J.; Meng, X.; Yang, S.; Stephan, J.M.; Leslie, K.K. Comprehensive profiling of EGFR/HER receptors for personalized treatment of gynecologic cancers. Mol. Diagn. Ther. 2014, 18, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Citri, A.; Yarden, Y. EGF-ERBB signalling: Towards the systems level. Nat. Rev. Mol. Cell Biol. 2006, 7, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Killock, D. Lung cancer: A new generation of EGFR inhibition. Nat. Rev. Clin. Oncol. 2015, 12, 373. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Attri, B.K.; Gill, R.K.; Bariwal, J. Review on EGFR Inhibitors: Critical Updates. Mini Rev. Med. Chem. 2016, 16, 1134–1166. [Google Scholar] [CrossRef] [PubMed]
- Morrison, J.; Thoma, C.; Goodall, R.J.; Lyons, T.J.; Gaitskell, K.; Wiggans, A.J.; Bryant, A. Epidermal growth factor receptor blockers for the treatment of ovarian cancer. Cochrane Database Syst. Rev. 2018, 10, CD007927. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.J.; Minion, L.E.; Coleman, R.L. Anti-angiogenic agents in ovarian cancer: Past, present, and future. Ann. Oncol. 2016, 27 (Suppl. 1), i33–i39. [Google Scholar] [CrossRef] [PubMed]
- Coward, J.I.; Middleton, K.; Murphy, F. New perspectives on targeted therapy in ovarian cancer. Int. J. Womens Health 2015, 7, 189–203. [Google Scholar] [CrossRef] [PubMed]
| Patient Population, Institution | Number of Cases | Method of Study | Number of Cases with Peritoneal Metastasis (Percentage of Total) | Time of Assessment | Reference |
|---|---|---|---|---|---|
| USA, Roswell Park Institute | 381* | Autopsy | 316 (83%) | At autopsy | [27] |
| USA, University of Rochester Medical School | 100 | Autopsy | 73 (73%) | At autopsy | [25] |
| USA, National Cancer Institute | 73 | Autopsy | 39 (53%) | At autopsy | [26] |
| Switzerland, University of Basel | 166 ** | Autopsy | 164 (99%) | At autopsy | [32] |
| UK, St. Bartholomew’s Hospital | 67 *** | Computed tomography | 59 (88%) | At relapse | [30] |
| Japan, The Jikei University School of Medicine | 70 **** | Imaging, cytometry, CA125 level | 49 (70%) | At relapse | [31] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbolina, M.V. Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma. Cancers 2018, 10, 444. https://doi.org/10.3390/cancers10110444
Barbolina MV. Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma. Cancers. 2018; 10(11):444. https://doi.org/10.3390/cancers10110444
Chicago/Turabian StyleBarbolina, Maria V. 2018. "Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma" Cancers 10, no. 11: 444. https://doi.org/10.3390/cancers10110444
APA StyleBarbolina, M. V. (2018). Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma. Cancers, 10(11), 444. https://doi.org/10.3390/cancers10110444





