Abstract
Oncolytic virotherapy has been investigated for several decades and is emerging as a plausible biological therapy with several ongoing clinical trials and two viruses are now approved for cancer treatment in humans. The direct cytotoxicity and immune-stimulatory effects make oncolytic viruses an interesting strategy for cancer treatment. In this review, we summarize the results of in vitro and in vivo published studies of oncolytic viruses in different phases of evaluation in dogs, using PubMed and Google scholar as search platforms, without time restrictions (to date). Natural and genetically modified oncolytic viruses were evaluated with some encouraging results. The most studied viruses to date are the reovirus, myxoma virus, and vaccinia, tested mostly in solid tumors such as osteosarcomas, mammary gland tumors, soft tissue sarcomas, and mastocytomas. Although the results are promising, there are issues that need addressing such as ensuring tumor specificity, developing optimal dosing, circumventing preexisting antibodies from previous exposure or the development of antibodies during treatment, and assuring a reasonable safety profile, all of which are required in order to make this approach a successful therapy in dogs.
1. Introduction
The concept of treatment in cancer changed radically in recent years, with modulation of the immune response to tumor cells now shown to be an effective therapeutic strategy for achieving response in a growing number of cancer types [1]. This new stage in medicine was brought about by the first positive results in humans with solid tumors such as non-small-cell lung cancer, kidney cancer, and melanoma reporting prolonged survival, despite advanced disease, with the use of immune checkpoint inhibitors [2,3,4]. Oncolytic viruses, aside from displaying selective cytotoxicity toward cancer cells, may favor the restoration of the immune anti-cancer function [5,6]. This dual effect makes them an interesting strategy for the treatment of cancer, not only for humans but also for dogs.
Canine cancer is an increasing cause of death worldwide, principally in adult dogs, with an annual incidence rate of malignant tumors of about eight cases per 1000 dogs [7,8,9,10,11]. Among the most common types of cancers are lymphomas [8], hemangiosarcomas [12], mast cell tumors [13], melanomas [14], osteosarcomas [15], and mammary gland tumors [16]. After the introduction of chemotherapy for dogs in 1946 [17], different types of cancers became treatable with some prolonged clinical remissions using multidrug protocols [18]. However, although treatments evolved, complete long-lasting remissions in non-surgically curable tumors are still infrequent [19,20]. Importantly, the biological, genetic, phenotypic, and clinical similarities between dogs and humans allow comparative approaches favoring a faster clinical application of the research findings for both species [21,22,23].
2. Brief History of Oncolytic Viruses
Oncolytic viruses have a natural or acquired selectivity for cancer cells. Wild-type, attenuated, and genetically engineered viruses with varying anti-tumoral efficacies were studied [24,25,26]. From the beginning of the 20th century, case reports of tumor responses following a natural viral infection in people with cancer were published [27,28,29,30]. Preclinical studies in murine models showed tumor responses using viruses, and, by the 1950s, results from the first clinical trials in humans were reported with occasional responses, such as those using the Harries rabies vaccine with a response in 2/12 patients with melanoma, and the Egypt 10 virus in 4/34 patients with other solid tumors [31,32,33,34]. Clinical trials with oncolytic viruses are currently ongoing in humans and two viruses were approved for commercial use. The first, H101 or Oncorine®, is an adenoviral construct with an E1B deletion (in order to avoid replication in normal cells), approved in China in 2005 for the treatment of head-and-neck squamous cell carcinoma [35]. The second, OncoVexGM-CSF or ImlygicTM, is an engineered herpes simplex virus type I (HSV-1) that expresses the human granulocyte–monocyte colony-stimulating factor as an immune-stimulant, approved in 2015 by the United States of America (USA) Food and Drug Administration (FDA) for the local treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with recurrent melanoma after initial surgery [36]. In veterinary medicine, double- and single-stranded RNA and DNA viruses with natural oncolytic capacity, as well as genetically modified viruses, are being studied, still lagging behind human research. The initial results in dogs and humans are pushing forward the development of oncolytic viruses as exciting cancer treatment strategies.
3. Oncolytic Viruses for Canine Cancer Treatment
A total of 13 viral species were studied as oncolytics in dogs (Figure S1). The possible antitumor mechanisms are shown in Figure 1, and the findings of published data are described below with the information grouped by viral families. For information regarding each virus, we took data from the Fields Virology book into consideration, in addition to the references cited [37].
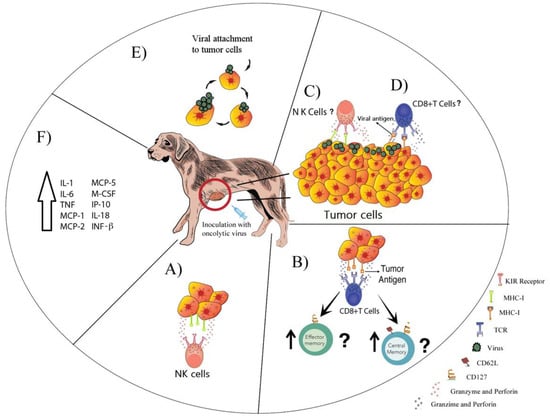
Figure 1.
Possible antitumor mechanisms of oncolytic viruses. The possible mechanisms exerted by oncolytic viruses include the recognition and elimination of infected tumor cells by (A) natural killer (NK) cells or (B) cluster of differentiation 8 positive (CD8+) T cells, which can generate central and effector memory CD8+ T cells. It remains unknown if (C) NK cells and (D) CD8+ T cells can also eliminate uninfected tumor cells. (E) The oncolytic virus can also infect tumor cells and induce direct cell lysis. In addition, (F) inoculation with oncolytic viruses can enhance the secretion of several pro-inflammatory cytokines. Several pathways may occur simultaneously.
3.1. Paramyxoviridae Family
These viruses enter cells via fusion or receptor-mediated endocytosis [38,39,40]. As oncolytics, paramyxoviruses (Table 1) promote immune-mediated tumor cell death. Importantly, many of the receptors used by paramyxoviruses are over-expressed in cancer cells. Moreover, due to tumor-associated genetic defects in the interferon (IFN) and apoptotic pathways, which may be seen in cancer cells, viral replication occurs naturally and preferentially in malignant cells [41].

Table 1.
Paramyxoviridae family and genera. Oncolytic paramyxoviruses are marked in bold.
3.1.1. Measles Virus (MV)
The oncolytic potential of MV was first observed in anecdotal reports describing the regression of hematopoietic neoplasms after accidental viral infection [42,43]. The first MV studied was the Edmonston-B vaccine strain, which is used worldwide for immunization against measles [44,45]. MV naturally infects lymphoid and respiratory epithelial cells. The infection of lymphoid cells is mediated by the binding of the H viral protein to CD150 (SLAM: signaling lymphocytic activation molecule) and to CD46 (membrane complement regulatory protein). In respiratory epithelial cells, MV binds through the nectin-4 receptor [46,47]. Nectin-4 is a receptor found to be overexpressed in some human cancers such as ovary, breast, and lung [48,49,50]. The MV genetic variant rMV-SLAMblind, which uses the related poliovirus 4 receptor (PVRL4/nectin-4) to enter cancer cells, was tested against human breast cancer with efficacy in xenografted mice and no relevant toxicity in primates [51]. Given the similarities between human breast cancer and canine mammary gland tumors (MGT), the virus was tested in canine MGT cells in vitro. The rMV-SLAMblind variant induced cell death in a manner dependent on the positive expression of canine nectin-4. Furthermore, immunosuppressed mice xenografted with canine nectin-4-positive MGT cells showed at least a 50% decrease in tumor volume compared to controls [52]. This therapy would make sense for dogs in the metastatic setting or in non-surgically resectable tumors, given that primary MGTs are usually well controlled with surgery [53]. Neurotoxicity still needs to be addressed and avoided [54]. In contrast to humans, the lack of pre-existing neutralizing antibodies against MV in dogs could favor in a systemic approach.
3.1.2. Canine Distemper Virus (CDV)
During a natural infection, CDV replicates in the epithelial cells of the respiratory tract [55]. The infection in dogs is clinically associated with respiratory and/or nervous system symptoms, in addition to severe long-term lymphoid depletion and immunosuppression [56,57]. Similar to measles, CDV employs CD150 and nectin-4 as cellular receptors [54,58]. In fact, central nervous system infection was associated with the expression of the neuronal nectin-4 receptor, present in dogs but not in humans [54].
In vitro, CDV was able to kill human cervical cancer cells [59] and acute T-cell lymphoblastic leukemia blasts [60]. In dogs, CDV was tested in histiocytic sarcoma and malignant lymphoid cells. The Onderstepoort strain of CDV infected and killed histiocytic sarcoma cells in vitro, inducing the expression of pro-inflammatory cytokines interleukin-1 (IL-1), (IL-6), and tumor necrosis factor (TNF) [61]. FXNO, YSA-TC, and MD-77 strains were also able to infect canine histiocytic sarcoma cells, with FXNO producing a more prominent and early cytopathic effect [62]. A recombinant strain, pCDVeGFPΔN, was able to infect canine malignant lymphoid cell lines, causing substantial cell death via apoptosis. An efficient infection with pCDVeGFPΔN strain was also demonstrated in primary canine malignant lymphocytes of B- and T-cell origin, although its oncolytic efficacy in those cells could not be demonstrated [60]. The possible inhibitory activity of morbilliviruses on cancer cells could be associated with the inhibition of metalloproteinases (MMPs), since persistent infection of the Onderstepoort strain in tumor cells derived from a malignant histiocytosis restored the activity of an MMP inhibitor decreasing the expression of MMPs [63]. These in vitro results are encouraging, because there is currently no effective established therapy for canine histiocytic sarcoma [64].
CDV was evaluated in seven dogs with lymphoma. The virus was injected intratumorally (IT) either in single or multiple doses of 1 × 106 plaque-forming units (PFU) showing a low-toxicity profile with an intense fibrotic reaction in the injected lymph nodes. The CDV antigen was variably positive determined using immunohistochemistry in the treated lymph nodes, and, using co-cultures, the virus could only be isolated from treated nodes, but neither from distant lymph nodes nor from peripheral blood mononuclear cells (PBMCs). A strong anti-CDV antibody response was documented, ruling out the possibility of repeated dosing [65]. Furthermore, CDV is part of routine vaccination in dogs; thus, pre-existing antibodies are also a limitation for oncolytic treatment [66].
3.1.3. Newcastle Disease Virus (NDV)
NDV naturally infects birds and, although it is considered a zoonosis, in humans, only self-limiting conjunctivitis was reported [67]. This virus showed natural oncolytic effects, principally in cells derived from human solid tumors [68,69,70]. A lentogenic strain was evaluated in canine cancer cells in vitro and in a dog with a spontaneous cancer [71,72]. In vitro, the NDV-MLS strain was preferentially toxic for canine malignant cells versus healthy PBMCs. The virus decreased the viability of primary canine B lymphoma cells by close to 40% [72]. In this same study, the early viral biodistribution was evaluated in a dog with a spontaneous T-cell lymphoma, 24 h after the intravenous (IV) administration of 1 × 1012 median tissue culture infectious dose (TCID50). Viral dissemination was detected by PCR only in healthy kidney, salivary gland, lung, and stomach, but not in tumor tissue, with no abnormalities upon histopathological assessment [72]. Interestingly, a complete long-term response in a dog with an aggressive chemotherapy-unresponsive lymphoma was reported [71].
It is probable that not all lymphomas are equally sensitive to NDV, and factors such as dose and route of administration influence the appropriate delivery of the virus to the tumor tissue. Furthermore, the historic safety profile of non-virulent strains [67] and immune-stimulatory potential [73,74,75] encourage the study of NDV for both human and canine cancers, with emphasis on its possible immune-stimulatory benefit.
3.1.4. Sendai Virus (SV)
This virus, also known as hemagglutinating virus of Japan, is a restricted pathogen in rodents, producing transmissible respiratory tract infections. SV was evaluated in vitro and in vivo as an oncolytic for human cancers, mainly in colon carcinoma, melanoma, and glioblastoma [76,77,78]. In one study, SV was evaluated in vivo in six dogs as therapy for mastocytoma. Dogs were treated with 1 × 107 to 1 × 108.6 mean embryo infectious dose (EID50) given in multiple injections (intratumorally and intradermally surrounding the tumor). Two or more viral treatments at one- or two-week intervals were given alone or in combination with surgery. Side effects included slight tenderness and edema at the injection sites which were responsive to antihistamines with quick resolution. Four dogs with either primary or recurrent tumors achieved complete responses, three of them long-lasting. The remaining two achieved stable disease [79]. These results justify further clinical studies of SV for the treatment of canine cutaneous/subcutaneous mastocytoma. Small lesions showed a better response to SV therapy [79]; yet, in routine practice, these lesions are usually resected. [80]. It would be interesting to test the use of this virus in an adjuvant or neo-adjuvant setting for mastocytoma, in order to determine whether the virus is able to control residual disease or reduce tumor burden facilitating surgery. It also will be important to evaluate drug interactions, especially when combined with steroids, which could decrease its therapeutic efficacy [79]. Following a pathological exam, SV was shown to induce infiltration by dendritic cells, CD4 and CD8 T-cells, and natural killer (NK) cells [77,81].
3.2. Rabdoviridae Family
These viruses enter cells via clathrin-mediated endocytosis [82], and the only species studied thus far as an oncolytic is the vesicular stomatitis virus (VSV).
Vesicular Stomatitis Virus
VSV causes an important endemic disease in cattle, while humans can be infected, and little is known about the dog as a host [83,84]. Although VSV has a natural oncolytic ability, genetic variants were created to improve therapeutic safety. The VSV-IFNβ-NIS is a variant expressing the human IFN-β (interferon beta) and the sodium iodide symporter (NIS). IFN-β exerts a protective effect on healthy cells mediated by IFN, while the expression of NIS makes molecular imaging of viral spread with radioactive iodine possible [85]. VSV-IFNβ-NIS is cytotoxic for human leukemic cells in vitro and for human myeloma in murine models [86]. Regarding dogs, two studies were performed using this virus. In a safety study in healthy Beagles treated with escalating doses starting at 1 × 108 TCID50, no viral particles (v.p.) were detected in urine, saliva, or feces, and the intracranial and IV administration did not result in neurotoxicity [85]. Moreover, when 10 dogs with hematological and solid tumors were treated with 1 × 1010 TCID50/0.5 m2 IV, two short-lasting partial responses were observed in T-cell lymphoma with the longest response corresponding to a dog with sustained expression of IFN-β and higher number of viral copies in blood. Tumor infiltration by T cells was also documented in a case of anal adenocarcinoma. Regarding toxicity and environmental safety, all dogs developed mild short-lasting fever, as well as mild transient lymphopenia; two dogs developed transient hepatotoxicity, and there were no documented adverse events and no viral shedding in a dog with osteosarcoma (OSA) who received two viral doses [87]. Studies of VSV-IFNβ-NIS in dogs are a clear example of the benefit of dogs as preclinical models for humans. Regarding safety, neither humans nor dogs develop neurotoxicity [85,87], while, in rats and mice, neurotoxicity with seizures was seen within the first week post-treatment [88]. In fact, based on clinical responses to VSV-IFNβ-NIS in canine lymphoma patients, this VSV variant is currently being tested in a clinical trial for human patients with relapsed or refractory myeloid and lymphoid malignancies (clinicaltrials.gov identifier: MCT03017820).
Another VSV variant tested as an oncolytic is VSV-GP, in which the VSV glycoprotein G was substituted, inducing less neurotoxicity [89]. VSV-GP is able to lyse melanoma cells in vitro by 0.1 multiplicity of infection (MOI) of VSV-GP. VSV-GP was also shown to prolong survival in mouse melanoma models (human xenografts and syngeneic) when treated with two doses of 107 PFU, with a median survival of 45 vs. 22 days between treated and control mice, respectively. However, even though tumor growth was delayed, only few animals achieved long-term tumor remissions. [90]. The induction of type I interferon may have compromised the clinical efficacy due to the inhibition of viral replication. Interestingly, this may be overcome by inhibiting downstream signaling of the INF-receptor with the use of ruxolitinib, an inhibitor of Janus kinases 1/2 (JAK ½). These results suggest the possibility of using VSV-GP for canine melanoma and possibly for other tumors [90].
3.3. Togaviridae Family
This family includes the alphavirus genera. As oncolytic viruses, alphaviruses are non-virulent, remain non-pathogenic in healthy tissue, and are spontaneously eliminated by the immune system [91]. The majority of alphaviruses show neurotropism and are able to cross the blood–brain barrier [92]. The Semliki Forest virus is the only alphavirus that was tested as an oncolytic for canine cancer.
Semliki Forest Virus (SFV)
SFV can naturally infect humans and other animals causing neurological manifestations [93,94,95]. However, no natural infections were described in dogs, and viral injection into puppies did not cause neurological toxicity [96]. As an oncolytic, the VA7 attenuated strain was able to eradicate xenografts of human brain tumors and melanoma in immunocompromised mice [97,98]. The oncolytic capacity of attenuated SFV was shown to depend on the loss of IFN-I response in the tumor cells [99]. In the case of dogs, the VA7 strain expressing the enhanced green fluorescent protein (eGFP) caused progressive cytopathic effects and cell death in canine OSA cells in vitro. In addition, injection of approximately 2 × 105 PFU IV of SFV in two laboratory Beagles did not result in signs of infection or severe adverse events. The neutralizing antibody response indicated that both dogs mounted a humoral immune response in the absence of detectable viremia; there were no histopathological changes in injected tissues, and no viral antigen was detected through immunohistochemistry in any of the organs analyzed. Regarding environmental safety, the virus was not recovered from serum, urine, or feces [100]. Given the safety profile and the positive result obtained in human melanoma xenografts in mice [97], as well as the limited effective treatments available for canine melanoma [101], the study of SFV should be further pursued.
3.4. Reoviridae Family
Reoviruses have the ability to infect almost all known mammal species and cause mild physical illness in humans [102]. Anti-reovirus neutralizing antibodies are detected in almost all adult humans and in a high percentage of healthy dogs [103,104,105]. Despite this, several strains of reoviruses were evaluated as oncolytics in humans; however, only reovirus-3, naturally isolated from dogs with diarrhea, was tested in dogs [106].
Reovirus Serotype 3
Dearing is the most used oncolytic strain (Reolysin®, from OncolyticsTM Biotech Inc., Calgary, AB, Canada) to date. In humans, Reolysin® was tested in phase II and III clinical trials [106]. In dogs, Reolysin® showed potential in vitro for the treatment of mastocytoma (MCT), lymphoma, MGT, and melanoma cells. Canine visceral and cutaneous MCT cells were efficiently killed by Reolysin® via apoptosis. In addition, mice xenografted with visceral canine MCT experienced significant tumor regression after a single treatment (p < 0.05), and, when one of the tumors was intratumorally injected in a bilateral tumor model, both tumors showed significant regression with extensive necrotic lesions. This was associated with an adequate generation of progeny and hematogenous spread. However, Reolysin has low specificity for malignant mastocytes, with nearly 90% death of normal canine bone marrow mast cells in vitro [107]. Induction of apoptosis and a significant decrease in cell viability were also documented in both T- and B-cell lymphomas in vitro, and mice xenografted with canine T-cell lymphoma treated with 1 × 108 PFU IT showed significant suppression of tumor growth compared to those treated with ultraviolet (UV)-inactivated reovirus (p < 0.05) [25]. A similar in vitro effect was demonstrated in canine OSA, MGT, and melanoma cell lines, where Reolysin® reduced cell viability through caspase-3-mediated apoptosis. The production of viral progeny was detected in susceptible lines. Susceptibility of human cancer cells to reovirus infection was associated with the activation of the Ras signaling pathway [108]; yet, interestingly, oncolysis by reoviruses in canine cancer cells from MCT, lymphoma, OSA, MGT, and melanomas does not seem to depend on the state of Ras activation [25,107,109].
Importantly, the safety profile of Reolysin® was established in 19 dogs with advanced cancer. Most of the patients had MCT, lymphoma, oral melanoma, or soft tissue sarcoma (STS). The dogs received doses that varied from 1 × 108 to 5 × 109 TCID50 via either IT injection (10 dogs) or IV injection (nine dogs) daily for five consecutive days, for one or multiple treatment cycles. Live virus in serum was detected only in one dog during the first cycle of chemotherapy, but not in other cycles. All dogs experienced an increase in the anti-reovirus neutralizing antibody titer, and there was a reduction in the size of the lesions in five dogs; in six others, there was a benefit in clinical signs (improvement of urination, reduction of tumor pain, increased mobility, or improvement in appetite) [110]. Thus, there was a good safety profile, as well as tumor response, in a subset of dogs [110]. The significance of the presence of neutralizing antibodies prior to therapy and the effect on clinical response from increasing anti-reovirus neutralizing antibody titers with therapy need to be studied in dogs. Considering the experience observed in humans [111,112,113], a combined approach with chemotherapy, radiotherapy, or other therapies could be explored in dogs.
3.5. Parvoviridae Family
Parvoviruses are the smallest viruses in existence [114]. This family includes the mouse minute virus (MVM), the rat H-1PV virus, the LuIII virus, and canine parvovirus (CPV), which showed natural oncolytic potential for human cancers cells in vitro and/or in vivo [115,116,117,118,119]. CPV was genetically engineered for cancer therapy [120], and studies related to its life cycle were investigated and provided insights into its use as an oncolytic for dogs [119].
Canine Parvovirus Serotype 2 (CPV-2)
CPV-2 is an important pathogen in domestic and wild canids; it can cause acute hemorrhagic enteritis and myocarditis, and it is commonly fatal [121,122,123,124]. However, vaccination can provide effective protection [125,126,127]. During natural infections, CPV-2 binds to the transferrin receptor (TfR), followed by clathrin-mediated endocytosis [128]. In vitro, CPV-2 induces cell death of canine fibroma cells, characterized by apoptosis and subsequent necrosis [119].
Although benign, the infiltrative ability of fibrous proliferations, depending on the anatomical site, makes surgical procedures difficult and/or invasive [129,130,131,132,133]. CDV could represent an alternative option for these patients. The virus could be explored for canine cancer, and inflammation secondary to necrosis could be beneficial in oncolytic virotherapy, since inflammation associated with necrosis might favor the activation of the immune system [134].
3.6. Poxviridae Family
Poxviruses enter cells via fusion and replicate predominantly in the cytoplasm [135]. In dogs, three genera of poxviruses were studied as oncolytics (Table 2).

Table 2.
Poxviruses studied in canine tumors.
3.6.1. Myxoma Virus (MYXV)
MYXV is only pathogenic for rabbits causing myxomatosis, a lethal disease [144]. As an oncolytic, MYXV is able to kill different types of human cancer cells in vitro (pancreas, ovary, glioblastoma, and hematological neoplasms) [145,146,147,148]. The greater susceptibility of these cells to MYXV was associated with the increased expression of phosphorylated protein kinase B (pAkt) [149]. MYXV and a MYXVΔserp1, which is a less pathogenic MYXV strain for rabbits [150], were evaluated in canine cancer cells. MYXVΔserp1 lost the expression of serp2, an anti-apoptotic virulence factor. At an MOI of 5, this virus was able to significantly kill hemangiosarcoma (HSA) and transitional cell carcinoma (TCC) cell lines, as well as primary HSA and STS cells. Compared to human cancer cells, MYXV was not as effective at killing canine cancer derived from dogs [151]. MYXVΔserp1 was studied in 10 dogs with STS, with either a single IT dose of 1 × 106 PFU or post-operatively (5–10 doses of 1 × 106 PFU) in incompletely excised tumors (positive surgical margins) in an attempt to control residual disease. Although the study demonstrated clinical and environmental safety parameters, the majority of patients treated intratumorally (4/5) only achieved stable disease and one progressed after a moth despite treatment. The report of outcome in post-surgically treated patients [136] and data on whether the virus can delay or decrease metastasis are still not available. Metastasis is another important complication in canine patients with high-grade STS [152]. Additionally, research on MYXV as an oncolytic for dogs should also consider the effectiveness related to pAkt expression, which would suggest that tumors such as canine melanoma and mastocytoma which expresses pAkt [153,154] may become possible targets.
3.6.2. Canarypox Virus
This is a virus of the genus Avipoxvirus. Avipoxviruses only infect some mammalian cells under laboratory conditions [155,156] and, although human healthy cells can be readily infected, the full replication cycle does not occur. However, a genetic variant (ALVAC) derived from the Kanapox strain of the canarypox virus was developed in order to address safety concerns. This variant is under investigation for its stimulatory properties on peripheral cytokine and chemokine response, and for possible use as therapy for human cancer [157]. When ALVAC-luciferase was administered IT in a dog with melanoma, the virus demonstrated limited viral dissemination; it was localized only along the needle path with no detectable virus in the periphery of the tumor [158]. This restricted dissemination could represent a safety advantage for dogs as was the case of ALVAC-derived recombinants that express the stimulatory cytokine IL-2, which were used successfully in the control of recurrent feline fibrosarcoma [158].
3.6.3. Vaccinia Virus (VACV)
VACV is widely used as a vaccine agent against smallpox [137], and diverse genetically engineered strains showed oncolytic potential [159,160]. Five VACVs derived from the Lister strain and one derived from the Copenhagen strain were evaluated as oncolytics for canine cancers. GLV-1h68 (GL-ONC1 from Genelux Corporation), a Lister-derived strain which encodes a light-emitting fusion gene, β-galactosidase, and β-glucuronidase, was first evaluated in a human breast xenograft mouse model, where a single dose was capable of causing complete tumor regression [161]. Furthermore, in in vitro studies, GLV-1h68 induced viral replication-dependent apoptosis of canine mammary adenoma cells [162], while, in canine mammary carcinoma, GLV-1h68 was able to efficiently infect, replicate, and lyse cells [138]. In murine models, GLV-1h68 induced significant tumor regression of canine adenoma xenografts [162], and a single IV dose of 5 × 106 PFU in nude mice xenografted with mammary carcinoma induced a strong inflammatory and oncolytic response that resulted in significant inhibition of tumor growth with confirmed infection and replication capability and tumor specificity [138]. GLV-1h68 also enhanced the expression of monocyte chemo-attractant protein (MCP)-1, -2, and -5, macrophage colony stimulating factor (M-CSF), interferon gamma-induced protein (IP)-10, and IL-18, which are known to increase innate immunity mediated by dendritic cells, neutrophils, and macrophages. This suggests that the activation of the innate immune system could act along with viral oncolysis to induce inhibition and tumor clearance in the murine model of canine mammary carcinoma [138].
Another VACV, LIVP1.1.1, was isolated from a wild-type stock of Lister strain. LIVP1.1.1, together with GLV-1h68, was evaluated in vitro and in vivo in a murine model of canine STS [139]. Both strains were shown to infect and destroy STS cells in vitro with better replication efficiency by LIVP1.1.1 [139]. Mice with canine xenografts of STS, treated IV with 1 × 107 PFU of LIVP1.1.1 or GLV-1h68, had reduced tumor growth [139]. The virus induced tumor necrosis and an immune infiltrate, as well as CD31 overexpression, which suggests that endothelial activation could be an additional reason for the increased accumulation of immune cells in the tumor site of the treated mice [139].
The GLV-1h109 strain is derived from GLV-1h68, but contains the GLAF-1 gene, which encodes for single-chain anti-vascular endothelial growth factor (anti-VEGF) antibody as a strategy to regulate tumor angiogenesis. Although anti-VEGF is directed against human and murine VEGF, it also recognizes canine VEGF [140]. In in vitro studies, GLV-1h109 infected, replicated, and lysed canine STS and prostatic carcinoma cells, decreasing viability by more than 70% [140]. In in vivo murine models, GLV-1h109 showed improved tumor-specific replication as compared to GLV-1h68. Treatment with GLV-1h109 inhibited the growth of murine xenografts of canine prostatic carcinoma and canine STS with acceptable toxicity. In these models, tumors infected with GLV-1h109 showed increased accumulation of neutrophils, myeloid-derived suppressor cells, and macrophages, as well as a reduction in blood vessel density in STS [140].
Another oncolytic strain is LIVP6.1.1, which was also isolated from a stock of wild-type Lister strain; it is less virulent as compared to other isolates. LIVP6.1.1 was evaluated in canine STS, prostate carcinoma, melanoma, and OSA [141]. In in vitro experiments, infection by LIVP6.1.1 efficiently killed the four cell types mentioned, causing at least 83% cytotoxicity [141]. In in vivo experiments in mice xenografted with canine STS or prostate carcinoma and treated with an IV dose of 5 × 106 PFU, there was a significant decrease in tumor growth (close to 50%), without signs of toxicity [141]. Interestingly, LIVP6.1.1 also showed preference for replication at the tumor site. In addition, an increase in immune cell infiltrate (granulocytes, monocytes, macrophages, and major histocompatibility complex class II positive (MHCII+) CD45+ cells) was documented in STS tumors [141]. In peripheral blood, no changes were found in cell populations, suggesting that response to virotherapy with LIVP6.1.1 in xenografts could be a combined result of direct oncolysis in addition to tumor-localized immune stimulation [141].
GLV-5b451 derived from the LIVP 6.1.1 strain contains the GLAF-1 gene encoding for a single chain of anti-canine VEGF antibody [142]. In dogs, VEGF overexpression was correlated with tumor malignancy and poor prognosis [143,163,164,165,166]. In in vitro studies, GLV-5b451 effectively infected, replicated, and killed canine cells from mammary carcinoma, mammary adenoma, prostatic carcinoma, and STS, inducing at least 60% cell death. These four cell types produce VEGF, especially prostatic carcinoma [142]. In a murine model of canine STS, a single dose of 1 × 107 PFU of GLV-5b541 or LIVP 6.1.1 was able to inhibit tumor growth without evidence of toxicity. GLV-5b541 showed greater efficacy and led to significant inhibition of tumor blood vessel development and increased expression of CD31 compared to LIVP 6.1.1. The upregulation of CD31 protein could mediate the trans-endothelial migration of immune cells to the site of infection. In this model, viral replication was preferentially seen in tumors compared to healthy tissues such as spleen, liver, and lungs [142].
Finally, NYVAC, derived from the Copenhagen strain, was first tested in healthy dogs. After parenteral (oral submucosa) administration, NYVAC-luciferase was recovered from the site of inoculation within 10 min, after which it was no longer detectable. Importantly, the virus was not detectable in saliva, feces, or blood at any time [158]. In addition, a dog with spontaneous melanoma treated IT with NYVAC showed localized dissemination only to the site of injection restricted to the needle path [158]. Viruses derived from the Lister strain of VACV seem to be promising agents for canine cancer immunotherapy. Studies of immune response in immune-competent animals are still warranted.
3.7. Adenoviridae Family
Adenoviruses naturally infect a great diversity of animals, including dogs and humans [167,168]; however, their tropism can be modified to direct the infection toward specific cell types [169]. Adenoviruses modified to selectively replicate in cancer cells and to induce cytolysis are termed “conditionally replicating oncolytic adenoviruses” (CRAds) [170]. CRAds based on human adenovirus 5 and canine adenovirus 2 were evaluated as oncolytics for canine cancer.
3.7.1. Canine Adenovirus Type 2 (CAV-2)
This virus causes a respiratory disease in dogs characterized mainly by bronchitis and bronchiolitis. It was used to immunize dogs against the related virus CAV-1, associated with highly contagious hepatitis in dogs [125].
The CAV-2-based vectors, OC-CAVE1 and ICOCAV17, were evaluated as oncolytics for canine cancer. OC-CAVE1 contains the osteocalcin (OC) promoter and, since tumors such as osteosarcoma (OSA) have high osteocalcin activity, this property allows targeting viral replication to these tumor cells. Treatment with OC-CAVE1 in a murine model xenografted with canine OSA showed a significant (p < 0.005) anti-tumor effect [171]. In healthy dogs treated IV with 2 × 1012 v.p., there were no clinical signs of infection and no macroscopic or microscopic changes upon pathological examination, although viral DNA was detected at high levels in the spleen and liver. Of note, there is a safety concern given that viral DNA was detected in blood for at least 48 h and for more than 96 h in urine and feces [172]. Another concern is that, since the presence of neutralizing antibodies develops as a consequence of previous infections or vaccination programs, those antibodies could interfere with viral anti-cancer response. To overcome this issue, the use of canine cancer cells as carriers of the virus was evaluated. Canine OSA cells as carriers of OC-CAVE1 were tested in canine xenografts in mice; they successfully reached the tumor site after IV administration, with reduction of viral hepatic uptake and with effective control of tumor growth despite the presence of anti-CAV neutralizing antibodies [173]. Additionally, when a polylysine was added to OC-CAVE1, its infectivity improved, as demonstrated in primary and immortalized canine OSA cells in vitro [174].
ICOCAV17 expresses hyaluronidase, an enzyme that dissociates the extracellular matrix and, therefore, enhances viral distribution after IT administration [175]. This virus showed functional activity in canine OSA and melanoma cells in vitro, while mice with canine melanoma or canine OSA xenografts treated with three weekly IT doses of 1 × 1010 v.p. showed increased survival without signs of toxicity [175]. In addition, six dogs with different spontaneous cancers (adenoma, mastocytoma, fibrosarcoma, metastatic neuroendocrine tumor, and osteosarcoma) were treated with 1 × 1012 v.p. and did not show adverse effects, even with viral titers in serum reaching 1 × 106 v.p./mL. Four of the six dogs responded to treatment, two with a partial and two with stable disease, and none of the six developed tumor lysis syndrome. Viral treatment was given concomitantly with the tyrosine kinase inhibitor, toceranib, in two of these patients. The virus caused tumor necrosis, and the viral genome could be detected in blood up to a week after treatment, with no viral shedding in urine, saliva, or feces at any time point [175]. Interestingly, this virus was used to infect canine mesenchymal stem cells, which were then infused intravenously as carriers for the virus [176]. There were no significant adverse effects in the 27 patients treated, with an overall response of 74% and 14.8% complete response rate, including remission of lung metastasis. Additionally, the virus induced inflammatory cell infiltration observed in tumor biopsies after four weeks of treatment. Apparently, the development of antibodies did not interfere with clinical benefit [176].
3.7.2. Human Adenovirus Type 5 (Ad5)
Ad5 causes infections of the respiratory tract in humans and was the platform most used for the development of oncolytic CRAds. In dogs, two vectors based on Ad5 were studied as oncolytics: Ad5CMVGFP and AdCD40L. Ad5CMVGFP expresses green fluorescent protein and was shown to efficiently infect primary canine OSA cells in vitro, although at a higher MOI than required in cells of human origin [169].
AdCD40L codifies for human CD40L, a molecule activating dendritic cells and T helper 1 (Th1) lymphocytes. Human and canine CD40L have high homology (85%), which led to the study of AdCD40L in dogs. Initially, two dogs with malignant melanoma were treated with peritumoral and IT injections of 9.6 × 108 v.p. One dog with a progressive conjunctival melanoma received six weekly treatments and, after initial inflammation, the tumor rapidly decreased in size; at four-month follow-up, there were no signs of progression or metastasis [177]. The other dog had an oral stage III melanoma and received two treatments with a four-day interval, achieving a long-term complete response. Interestingly, after viral treatment, the tumor tissue was infiltrated by both T and B lymphocytes. In both cases, the tumor lost pigmentation and there were no relevant signs of systemic autoimmunity [177]. It is important to mention that dogs with stage III melanoma treated only with surgery rarely survive more than 22 weeks [178]. AdCD40L was further evaluated in another 19 dogs with melanoma (14 oral, four cutaneous, and one conjunctival). The dogs received between one and six weekly IT doses with 37 µL of concentrated viral stock solution at 7 × 1010 PFU/mL at weekly intervals, followed by cytoreductive surgery in nine cases. Although there were mild adverse effects in 8/19 dogs, these did not recur with subsequent treatments. Additionally, there were five complete and eight partial documented responses, with four dogs being stable and two having progressive disease. Again, in 8/11 dogs, pathology showed tumor infiltration by B and T lymphocytes, suggesting local immune stimulation [179]. Oncolytic adenovirus-based vectors may represent an alternative option for the treatment of canine cancer. Mechanisms to circumvent the effect of pre-existing antibodies that could compromise effectiveness should be studied.
4. Discussion
There is growing interest in the study of oncolytic viruses as an approach for the treatment of canine cancer. Table 3 summarizes the different studies performed to date in vitro and in vivo related to dogs. Most of the studies were performed in vitro, mainly with the use of reovirus, myxoma, and vaccinia, with encouraging results in tumor cells derived from osteosarcomas, mammary gland tumors, soft tissue sarcomas, and mastocytomas. There are very few studies addressing hematological malignancies; yet, some reports show promising oncolytic responses in canine lymphoma cells with the use of paramyxoviruses. Regarding in vivo studies in preclinical models (canine xenografts in mice), the control of tumor growth was consistently accompanied by virus-induced tumor infiltration by immune cells, as well as a decrease in tumor blood vessel density, and induction and over-expression of molecules involved in innate immunity, as seen with the vaccinia virus. It is important to note that dogs may better reflect the clinical efficacy and safety parameters associated with oncolytic virotherapy than mouse models, as seen with VSV. The published results strongly encourage continuing the study of oncolytic viruses using a comparative approach between dogs and humans.

Table 3.
Oncolytic viral strains studied in dogs.
5. Conclusions
Oncolytic viruses are gaining ground as an alternative approach for treating cancer in dogs and humans. Although most of the current information was obtained in vitro, there are encouraging in vivo results and growing interest in performing clinical studies. The tolerance and safety for viruses such as vesicular stomatitis, Semliki Forest, canarypox, and vaccinia were established in healthy dogs. Favorable clinical responses are being reported both with natural and genetically modified viruses in tumors such as lymphomas, melanomas, adenomas, and cutaneous mastocytomas. In order to improve the usefulness of oncolytic viruses for the treatment of tumors in dogs, several issues must be addressed. These include a better characterization of tumors, which still lags far behind that of studies in humans, as well as the search for canine cell-specific therapeutic targets, viral modifications to enhance tumor specificity, safety studies regarding viral shedding, the establishment of therapeutic regimens (doses and routes of administration), and the evaluation of combined therapies for improving tumor responses. These studies should lead, in the not too far future, to the use of oncolytic viruses as part of cancer treatment.
Supplementary Materials
The following are available online at http://www.mdpi.com/2072-6694/10/11/404/s1, Figure S1: Oncolytic viruses studied in dogs.
Author Contributions
D.S., G.C.-M., and M.L. wrote the paper and made the tables; A.A.-M. contributed to reviewing the manuscript and made figures.
Funding
This research received no external funding.
Acknowledgments
We thank the DGAPA UNAM for support to D.S., and support from the Instituto Nacional de Cancerología, México (018/026/IBI) (CEI/1245/18).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Couzin-Frankel, J. Breakthrough of the year 2013. Cancer immunotherapy. Science (New York, N.Y.) 2013, 342, 1432–1433. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, activity, and immune correlates of anti-pd-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; O‘Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Kluger, H.; Callahan, M.K.; Postow, M.A.; Rizvi, N.A.; Lesokhin, A.M.; Segal, N.H.; Ariyan, C.E.; Gordon, R.A.; Reed, K.; et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 2013, 369, 122–133. [Google Scholar] [CrossRef] [PubMed]
- White, C.L.; Twigger, K.R.; Vidal, L.; De Bono, J.S.; Coffey, M.; Heinemann, L.; Morgan, R.; Merrick, A.; Errington, F.; Vile, R.G.; et al. Characterization of the adaptive and innate immune response to intravenous oncolytic reovirus (dearing type 3) during a phase i clinical trial. Gene Ther. 2008, 15, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.D.; Meza-Perez, S.; Bevis, K.S.; Randall, T.D.; Gillespie, G.Y.; Langford, C.; Alvarez, R.D. Il-12 expressing oncolytic herpes simplex virus promotes anti-tumor activity and immunologic control of metastatic ovarian cancer in mice. J. Ovarian Res. 2016, 9, 70. [Google Scholar] [CrossRef] [PubMed]
- Khanna, C.; Lindblad-Toh, K.; Vail, D.; London, C.; Bergman, P.; Barber, L.; Breen, M.; Kitchell, B.; McNeil, E.; Modiano, J.F.; et al. The dog as a cancer model. Nat. Biotechnol. 2006, 24, 1065–1066. [Google Scholar] [CrossRef] [PubMed]
- Baioni, E.; Scanziani, E.; Vincenti, M.C.; Leschiera, M.; Bozzetta, E.; Pezzolato, M.; Desiato, R.; Bertolini, S.; Maurella, C.; Ru, G. Estimating canine cancer incidence: Findings from a population-based tumour registry in northwestern italy. BMC Vet. Res. 2017, 13, 203. [Google Scholar] [CrossRef] [PubMed]
- Kidd, C. The many challenges of veterinary oncology. Can. Vet. J. 2008, 49, 1132–1135. [Google Scholar] [PubMed]
- Bronson, R.T. Variation in age at death of dogs of different sexes and breeds. Am. J. Vet. Res. 1982, 43, 2057–2059. [Google Scholar] [PubMed]
- Fleming, J.M.; Creevy, K.E.; Promislow, D.E. Mortality in north american dogs from 1984 to 2004: An investigation into age-, size-, and breed-related causes of death. J. Vet. Intern. Med. 2011, 25, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Spangler, W.L.; Culbertson, M.R. Prevalence, type, and importance of splenic diseases in dogs: 1,480 cases (1985–1989). J. Am. Vet. Med. Assoc. 1992, 200, 829–834. [Google Scholar] [PubMed]
- Grabarevic, Z.; Spoljar, J.B.; Kurilj, A.G.; Sostaric-Zuckermann, I.C.; Artukovic, B.; Hohsteter, M.; Beck, A.; Dzaja, P.; Strmecki, N.M. Mast cell tumor in dogs--incidence and histopathological characterization. Coll. Antropol. 2009, 33, 253–258. [Google Scholar] [PubMed]
- Ramos-Vara, J.A.; Beissenherz, M.E.; Miller, M.A.; Johnson, G.C.; Pace, L.W.; Fard, A.; Kottler, S.J. Retrospective study of 338 canine oral melanomas with clinical, histologic, and immunohistochemical review of 129 cases. Vet. Pathol. 2000, 37, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Egenvall, A.; Nodtvedt, A.; von Euler, H. Bone tumors in a population of 400 000 insured swedish dogs up to 10 y of age: Incidence and survival. Can. J. Vet. Res. 2007, 71, 292–299. [Google Scholar] [PubMed]
- Salas, Y.; Marquez, A.; Diaz, D.; Romero, L. Epidemiological study of mammary tumors in female dogs diagnosed during the period 2002-2012: A growing animal health problem. PLoS ONE 2015, 10, e0127381. [Google Scholar] [CrossRef] [PubMed]
- Innes, J.R.; Parry, H.B.; Berger, J. Leukaemia in dogs; including a record of a case treated by urethane. Br. Vet. J. 1946, 102, 383–393. [Google Scholar] [PubMed]
- Brick, J.O.; Roenigk, W.J.; Wilson, G.P. Chemotherapy of malignant lymphoma in dogs and cats. J. Am. Vet. Med. Assoc. 1968, 153, 47–52. [Google Scholar] [PubMed]
- Wang, S.L.; Lee, J.J.; Liao, A.T. Comparison of efficacy and toxicity of doxorubicin and mitoxantrone in combination chemotherapy for canine lymphoma. Can. Vet. J. 2016, 57, 271–276. [Google Scholar] [PubMed]
- Kiupel, M.; Webster, J.D.; Bailey, K.L.; Best, S.; DeLay, J.; Detrisac, C.J.; Fitzgerald, S.D.; Gamble, D.; Ginn, P.E.; Goldschmidt, M.H.; et al. Proposal of a 2-tier histologic grading system for canine cutaneous mast cell tumors to more accurately predict biological behavior. Vet. Pathol. 2011, 48, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Shearin, A.L.; Hedan, B.; Cadieu, E.; Erich, S.A.; Schmidt, E.V.; Faden, D.L.; Cullen, J.; Abadie, J.; Kwon, E.M.; Grone, A.; et al. The mtap-cdkn2a locus confers susceptibility to a naturally occurring canine cancer. Cancer Epidemiol. Biomarkers Prev. 2012, 21, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Im, K.S.; Kim, N.H.; Lim, H.Y.; Kim, H.W.; Shin, J.I.; Sur, J.H. Analysis of a new histological and molecular-based classification of canine mammary neoplasia. Vet. Pathol. 2014, 51, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Bushell, K.R.; Kim, Y.; Chan, F.C.; Ben-Neriah, S.; Jenks, A.; Alcaide, M.; Fornika, D.; Grande, B.M.; Arthur, S.; Gascoyne, R.D.; et al. Genetic inactivation of traf3 in canine and human b-cell lymphoma. Blood 2015, 125, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Yurchenko, K.S.; Zhou, P.; Kovner, A.V.; Zavjalov, E.L.; Shestopalova, L.V.; Shestopalov, A.M. Oncolytic effect of wild-type newcastle disease virus isolates in cancer cell lines in vitro and in vivo on xenograft model. PLoS ONE 2018, 13, e0195425. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.C.; Umeki, S.; Igase, M.; Coffey, M.; Noguchi, S.; Okuda, M.; Mizuno, T. The effects of oncolytic reovirus in canine lymphoma cell lines. Vet. Comp. Oncol. 2016, 14 (Suppl. 1), 61–73. [Google Scholar] [CrossRef]
- Mostafa, A.A.; Meyers, D.E.; Thirukkumaran, C.M.; Liu, P.J.; Gratton, K.; Spurrell, J.; Shi, Q.; Thakur, S.; Morris, D.G. Oncolytic reovirus and immune checkpoint inhibition as a novel immunotherapeutic strategy for breast cancer. Cancers 2018, 10, E205. [Google Scholar] [CrossRef] [PubMed]
- Bierman, H.R.; Crile, D.M.; Dod, K.S.; Kelly, K.H.; Petrakis, N.L.; White, L.P.; Shimkin, M.B. Remissions in leukemia of childhood following acute infectious disease: Staphylococcus and streptococcus, varicella, and feline panleukopenia. Cancer 1953, 6, 591–605. [Google Scholar] [CrossRef]
- Pelner, L.; Fowler, G.A.; Nauts, H.C. Effects of concurrent infections and their toxins on the course of leukemia. Acta Med. Scand. 1958, 338, 1–47. [Google Scholar] [CrossRef]
- Bluming, A.Z.; Ziegler, J.L. Regression of burkitt‘s lymphoma in association with measles infection. Lancet 1971, 2, 105–106. [Google Scholar] [CrossRef]
- Gross, S. Measles and leukaemia. Lancet 1971, 1, 397–398. [Google Scholar] [CrossRef]
- Huebner, R.J.; Rowe, W.P.; Schatten, W.E.; Smith, R.R.; Thomas, L.B. Studies on the use of viruses in the treatment of carcinoma of the cervix. Cancer 1956, 9, 1211–1218. [Google Scholar] [PubMed]
- Pack, G.T. Note on the experimental use of rabies vaccine for melanomatosis. AMA Arch. Derm. Syphilol. 1950, 62, 694–695. [Google Scholar] [CrossRef] [PubMed]
- Southam, C.M.; Moore, A.E. Clinical studies of viruses as antineoplastic agents with particular reference to egypt 101 virus. Cancer 1952, 5, 1025–1034. [Google Scholar] [CrossRef]
- Lerner, B.H. Sins of omission--cancer research without informed consent. N. Engl. J. Med. 2004, 351, 628–630. [Google Scholar] [CrossRef] [PubMed]
- Garber, K. China approves worlds first oncolytic virus therapy for cancer treatment. J. Natl. Cancer Inst. 2006, 98, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Dolgin, E. Oncolytic viruses get a boost with first fda-approval recommendation. Nat. Rev. Drug Discov. 2015, 14, 369–371. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.N.; Knipe, D.M.; Howley, P.M. Fields Virology; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Farzan, S.F.; Palermo, L.M.; Yokoyama, C.C.; Orefice, G.; Fornabaio, M.; Sarkar, A.; Kellogg, G.E.; Greengard, O.; Porotto, M.; Moscona, A. Premature activation of the paramyxovirus fusion protein before target cell attachment with corruption of the viral fusion machinery. J. Biol. Chem. 2011, 286, 37945–37954. [Google Scholar] [CrossRef] [PubMed]
- Cantin, C.; Holguera, J.; Ferreira, L.; Villar, E.; Munoz-Barroso, I. Newcastle disease virus may enter cells by caveolae-mediated endocytosis. J. Gen. Virol. 2007, 88, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Goncalves-Carneiro, D.; McKeating, J.A.; Bailey, D. The measles virus receptor slamf1 can mediate particle endocytosis. J. Virol. 2017, 91, e02255-16. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.; Palese, P.; Zamarin, D. Oncolytic specificity of newcastle disease virus is mediated by selectivity for apoptosis-resistant cells. J. Virol. 2011, 85, 6015–6023. [Google Scholar] [CrossRef] [PubMed]
- Zygiert, Z. Hodgkin‘s disease: Remissions after measles. Lancet 1971, 1, 593. [Google Scholar] [CrossRef]
- Taqi, A.M.; Abdurrahman, M.B.; Yakubu, A.M.; Fleming, A.F. Regression of hodgkin‘s disease after measles. Lancet 1981, 1, 1112. [Google Scholar] [CrossRef]
- Beck, M.; Smerdel, S.; Dedic, I.; Delimar, N.; Rajninger-Miholic, M.; Juzbasic, M.; Manhalter, T.; Vlatkovic, R.; Borcic, B.; Mihajic, Z. Immune response to edmonston-zagreb measles virus strain in monovalent and combined mmr vaccine. Dev. Biol. Stand. 1986, 65, 95–100. [Google Scholar] [PubMed]
- Grote, D.; Russell, S.J.; Cornu, T.I.; Cattaneo, R.; Vile, R.; Poland, G.A.; Fielding, A.K. Live attenuated measles virus induces regression of human lymphoma xenografts in immunodeficient mice. Blood 2001, 97, 3746–3754. [Google Scholar] [CrossRef] [PubMed]
- Noyce, R.S.; Richardson, C.D. Nectin 4 is the epithelial cell receptor for measles virus. Trends Microbiol. 2012, 20, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Muhlebach, M.D.; Mateo, M.; Sinn, P.L.; Prufer, S.; Uhlig, K.M.; Leonard, V.H.; Navaratnarajah, C.K.; Frenzke, M.; Wong, X.X.; Sawatsky, B.; et al. Adherens junction protein nectin-4 is the epithelial receptor for measles virus. Nature 2011, 480, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Derycke, M.S.; Pambuccian, S.E.; Gilks, C.B.; Kalloger, S.E.; Ghidouche, A.; Lopez, M.; Bliss, R.L.; Geller, M.A.; Argenta, P.A.; Harrington, K.M.; et al. Nectin 4 overexpression in ovarian cancer tissues and serum: Potential role as a serum biomarker. Am. J. Clin. Pathol. 2010, 134, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Fabre-Lafay, S.; Monville, F.; Garrido-Urbani, S.; Berruyer-Pouyet, C.; Ginestier, C.; Reymond, N.; Finetti, P.; Sauvan, R.; Adelaide, J.; Geneix, J.; et al. Nectin-4 is a new histological and serological tumor associated marker for breast cancer. BMC Cancer 2007, 7, 73. [Google Scholar] [CrossRef] [PubMed]
- Takano, A.; Ishikawa, N.; Nishino, R.; Masuda, K.; Yasui, W.; Inai, K.; Nishimura, H.; Ito, H.; Nakayama, H.; Miyagi, Y.; et al. Identification of nectin-4 oncoprotein as a diagnostic and therapeutic target for lung cancer. Cancer Res. 2009, 69, 6694–6703. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Yoneda, M.; Kuraishi, T.; Hattori, S.; Inoue, Y.; Sato, H.; Kai, C. Measles virus selectively blind to signaling lymphocyte activation molecule as a novel oncolytic virus for breast cancer treatment. Gene Ther. 2013, 20, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Shoji, K.; Yoneda, M.; Fujiyuki, T.; Amagai, Y.; Tanaka, A.; Matsuda, A.; Ogihara, K.; Naya, Y.; Ikeda, F.; Matsuda, H.; et al. Development of new therapy for canine mammary cancer with recombinant measles virus. Mol. Ther. Oncolytics 2016, 3, 15022. [Google Scholar] [CrossRef] [PubMed]
- Betz, D.; Schoenrock, D.; Mischke, R.; Baumgartner, W.; Nolte, I. Postoperative treatment outcome in canine mammary tumors. Multivariate analysis of the prognostic value of pre- and postoperatively available information. Tierarztl. Prax. Ausg. K Kleintiere Heimtiere 2012, 40, 235–242. [Google Scholar] [PubMed]
- Pratakpiriya, W.; Seki, F.; Otsuki, N.; Sakai, K.; Fukuhara, H.; Katamoto, H.; Hirai, T.; Maenaka, K.; Techangamsuwan, S.; Lan, N.T.; et al. Nectin4 is an epithelial cell receptor for canine distemper virus and involved in neurovirulence. J. Virol. 2012, 86, 10207–10210. [Google Scholar] [CrossRef] [PubMed]
- Sawatsky, B.; Wong, X.X.; Hinkelmann, S.; Cattaneo, R.; von Messling, V. Canine distemper virus epithelial cell infection is required for clinical disease but not for immunosuppression. J. Virol. 2012, 86, 3658–3666. [Google Scholar] [CrossRef] [PubMed]
- Qeska, V.; Barthel, Y.; Herder, V.; Stein, V.M.; Tipold, A.; Urhausen, C.; Gunzel-Apel, A.R.; Rohn, K.; Baumgartner, W.; Beineke, A. Canine distemper virus infection leads to an inhibitory phenotype of monocyte-derived dendritic cells in vitro with reduced expression of co-stimulatory molecules and increased interleukin-10 transcription. PLoS ONE 2014, 9, e96121. [Google Scholar] [CrossRef] [PubMed]
- Schobesberger, M.; Summerfield, A.; Doherr, M.G.; Zurbriggen, A.; Griot, C. Canine distemper virus-induced depletion of uninfected lymphocytes is associated with apoptosis. Vet. Immunol. Immunopathol. 2005, 104, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, M.; Bringolf, F.; Röthlisberger, S.; Bieringer, M.; Schneider-Schaulies, J.; Zurbriggen, A.; Origgi, F.; Plattet, P. Canine distemper virus fusion activation: Critical role of residue e123 of cd150/slam. J. Virol. 2016, 90, 1622–1637. [Google Scholar] [CrossRef] [PubMed]
- Del Puerto, H.L.; Martins, A.S.; Milsted, A.; Souza-Fagundes, E.M.; Braz, G.F.; Hissa, B.; Andrade, L.O.; Alves, F.; Rajao, D.S.; Leite, R.C.; et al. Canine distemper virus induces apoptosis in cervical tumor derived cell lines. Virol. J. 2011, 8, 334. [Google Scholar] [CrossRef] [PubMed]
- Suter, S.E.; Chein, M.B.; von Messling, V.; Yip, B.; Cattaneo, R.; Vernau, W.; Madewell, B.R.; London, C.A. In vitro canine distemper virus infection of canine lymphoid cells: A prelude to oncolytic therapy for lymphoma. Clin. Cancer Res. 2005, 11, 1579–1587. [Google Scholar] [CrossRef] [PubMed]
- Grone, A.; Fonfara, S.; Baumgartner, W. Cell type-dependent cytokine expression after canine distemper virus infection. Viral Immunol. 2002, 15, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, R.; Kojimoto, A.; Sakai, H.; Uchida, K.; Sugano, S.; Tateyama, S. Growth characteristics of canine distemper virus in a new cell line cct cells originated from canine malignant histiocytosis. J. Vet. Med. Sci. 2005, 67, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Puff, C.; Krudewig, C.; Imbschweiler, I.; Baumgartner, W.; Alldinger, S. Influence of persistent canine distemper virus infection on expression of reck, matrix-metalloproteinases and their inhibitors in a canine macrophage/monocytic tumour cell line (dh82). Vet. J. 2009, 182, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Dervisis, N.G.; Kiupel, M.; Qin, Q.; Cesario, L. Clinical prognostic factors in canine histiocytic sarcoma. Vet. Comp. Oncol. 2017, 15, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Henson, M.S.; Suter, S.E.; von Messling, V.A.; Cattaneo, R.; Fielding, A.K. 803. The effects of intratumoral injection of a replicating morbillivirus in a canine model of naturally occurring lymphoma. Mol. Ther. 2005, 11, 312. [Google Scholar]
- Ford, R.B.; Larson, L.J.; McClure, K.D.; Schultz, R.D.; Welborn, L.V. 2017 aaha canine vaccination guidelines. J. Am. Anim. Hosp. Assoc. 2017, 53, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Nelson, C.B.; Pomeroy, B.S.; Schrall, K.; Park, W.E.; Lindeman, R.J. An outbreak of conjunctivitis due to newcastle disease virus (ndv) occurring in poultry workers. Am. J. Public Health Nations Health 1952, 42, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Zulkifli, M.M.; Ibrahim, R.; Ali, A.M.; Aini, I.; Jaafar, H.; Hilda, S.S.; Alitheen, N.B.; Abdullah, J.M. Newcastle diseases virus strain v4upm displayed oncolytic ability against experimental human malignant glioma. Neurol. Res. 2009, 31, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Bian, J.; Wang, K.; Kong, X.; Liu, H.; Chen, F.; Hu, M.; Zhang, X.; Jiao, X.; Ge, B.; Wu, Y.; et al. Caspase- and p38-mapk-dependent induction of apoptosis in a549 lung cancer cells by newcastle disease virus. Arch. Virol. 2011, 156, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Buijs, P.R.; van Eijck, C.H.; Hofland, L.J.; Fouchier, R.A.; van den Hoogen, B.G. Different responses of human pancreatic adenocarcinoma cell lines to oncolytic newcastle disease virus infection. Cancer Gene Ther. 2014, 21, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, D.; Pelayo, R.; Sarmiento, R.E.; Medina, L.A.; Cesarman-Maus, G.N.; Nuñez, L.; Carrillo, N.; de Jesus Paredes, J.; Corona, H.; Vadillo, E. In Vitro and in Vivo oncolytic Activity of Lasota Strain of Newcastle Disease Virus on a Lymphoma B-Cell Line and a Canine Cutaneous T-Cell Lymphoma. Blood 2014, 124, 5504. [Google Scholar]
- Sanchez, D.; Pelayo, R.; Medina, L.A.; Vadillo, E.; Sanchez, R.; Nunez, L.; Cesarman-Maus, G.; Sarmiento-Silva, R.E. Newcastle disease virus: Potential therapeutic application for human and canine lymphoma. Viruses 2016, 8, E3. [Google Scholar] [CrossRef] [PubMed]
- Jarahian, M.; Watzl, C.; Fournier, P.; Arnold, A.; Djandji, D.; Zahedi, S.; Cerwenka, A.; Paschen, A.; Schirrmacher, V.; Momburg, F. Activation of natural killer cells by newcastle disease virus hemagglutinin-neuraminidase. J. Virol. 2009, 83, 8108–8121. [Google Scholar] [CrossRef] [PubMed]
- Koks, C.A.; Garg, A.D.; Ehrhardt, M.; Riva, M.; Vandenberk, L.; Boon, L.; De Vleeschouwer, S.; Agostinis, P.; Graf, N.; Van Gool, S.W. Newcastle disease virotherapy induces long-term survival and tumor-specific immune memory in orthotopic glioma through the induction of immunogenic cell death. Int. J. Cancer 2015, 136, E313–E325. [Google Scholar] [CrossRef] [PubMed]
- Termeer, C.C.; Schirrmacher, V.; Brocker, E.B.; Becker, J.C. Newcastle disease virus infection induces b7-1/b7-2-independent t-cell costimulatory activity in human melanoma cells. Cancer Gene Ther. 2000, 7, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Tai, J.A.; Li, S.; Nishikawa, T.; Kaneda, Y. Virus-stimulated neutrophils in the tumor microenvironment enhance t cell-mediated anti-tumor immunity. Oncotarget 2016, 7, 42195–42207. [Google Scholar] [CrossRef] [PubMed]
- Kurooka, M.; Kaneda, Y. Inactivated sendai virus particles eradicate tumors by inducing immune responses through blocking regulatory t cells. Cancer Res. 2007, 67, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Shimbo, T.; Kikuchi, Y.; Matsuda, M.; Kaneda, Y. Sterile alpha motif containing domain 9 is involved in death signaling of malignant glioma treated with inactivated sendai virus particle (hvj-e) or type i interferon. Int. J. Cancer 2010, 126, 1982–1991. [Google Scholar] [CrossRef] [PubMed]
- Ilyinskaya, G.V.; Mukhina, E.V.; Soboleva, A.V.; Matveeva, O.V.; Chumakov, P.M. Oncolytic sendai virus therapy of canine mast cell tumors (a pilot study). Front. Vet. Sci. 2018, 5, 116. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, L.; Mullin, C.; Balko, J.; Goldschmidt, M.; Krick, E.; Hume, C.; Brown, D.C.; Sorenmo, K. Evaluation of histological grade and histologically tumour-free margins as predictors of local recurrence in completely excised canine mast cell tumours. Vet. Comp. Oncol. 2015, 13, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Fujihara, A.; Kurooka, M.; Miki, T.; Kaneda, Y. Intratumoral injection of inactivated sendai virus particles elicits strong antitumor activity by enhancing local cxcl10 expression and systemic nk cell activation. Cancer Immunol. Immunother. 2008, 57, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Piccinotti, S.; Kirchhausen, T.; Whelan, S.P. Uptake of rabies virus into epithelial cells by clathrin-mediated endocytosis depends upon actin. J. Virol. 2013, 87, 11637–11647. [Google Scholar] [CrossRef] [PubMed]
- Scherer, C.F.; O‘Donnell, V.; Golde, W.T.; Gregg, D.; Estes, D.M.; Rodriguez, L.L. Vesicular stomatitis new jersey virus (vsnjv) infects keratinocytes and is restricted to lesion sites and local lymph nodes in the bovine, a natural host. Vet. Res. 2007, 38, 375–390. [Google Scholar] [CrossRef] [PubMed]
- Quiroz, E.; Moreno, N.; Peralta, P.H.; Tesh, R.B. A human case of encephalitis associated with vesicular stomatitis virus (indiana serotype) infection. Am. J. Trop. Med. Hyg. 1988, 39, 312–314. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, A.K.; Naik, S.; Galyon, G.D.; Jenks, N.; Steele, M.; Peng, K.W.; Federspiel, M.J.; Donnell, R.; Russell, S.J. Safety studies on intravenous administration of oncolytic recombinant vesicular stomatitis virus in purpose-bred beagle dogs. Hum. Gene Ther. Clin. Dev. 2013, 24, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Patnaik, M.M.; Ruiz, A.; Russell, S.J.; Peng, K.W. Immunovirotherapy with vesicular stomatitis virus and pd-l1 blockade enhances therapeutic outcome in murine acute myeloid leukemia. Blood 2016, 127, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Naik, S.; Galyon, G.D.; Jenks, N.J.; Steele, M.B.; Miller, A.C.; Allstadt, S.D.; Suksanpaisan, L.; Peng, K.W.; Federspiel, M.J.; Russell, S.J.; et al. Comparative oncology evaluation of intravenous recombinant oncolytic vesicular stomatitis virus therapy in spontaneous canine cancer. Mol. Cancer Ther. 2018, 17, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Jenks, N.; Myers, R.; Greiner, S.M.; Thompson, J.; Mader, E.K.; Greenslade, A.; Griesmann, G.E.; Federspiel, M.J.; Rakela, J.; Borad, M.J.; et al. Safety studies on intrahepatic or intratumoral injection of oncolytic vesicular stomatitis virus expressing interferon-β in rodents and nonhuman primates. Hum. Gene Ther. 2010, 21, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Muik, A.; Kneiske, I.; Werbizki, M.; Wilflingseder, D.; Giroglou, T.; Ebert, O.; Kraft, A.; Dietrich, U.; Zimmer, G.; Momma, S.; et al. Pseudotyping vesicular stomatitis virus with lymphocytic choriomeningitis virus glycoproteins enhances infectivity for glioma cells and minimizes neurotropism. J. Virol. 2011, 85, 5679–5684. [Google Scholar] [CrossRef] [PubMed]
- Kimpel, J.; Urbiola, C.; Koske, I.; Tober, R.; Banki, Z.; Wollmann, G.; von Laer, D. The oncolytic virus vsv-gp is effective against malignant melanoma. Viruses 2018, 10, E108. [Google Scholar] [CrossRef] [PubMed]
- Vaha-Koskela, M.J.; Tuittila, M.T.; Nygardas, P.T.; Nyman, J.K.; Ehrengruber, M.U.; Renggli, M.; Hinkkanen, A.E. A novel neurotropic expression vector based on the avirulent a7(74) strain of semliki forest virus. J. Neurovirol. 2003, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fros, J.J.; Pijlman, G.P. Alphavirus infection: Host cell shut-off and inhibition of antiviral responses. Viruses 2016, 8, E166. [Google Scholar] [CrossRef] [PubMed]
- Mathiot, C.C.; Grimaud, G.; Garry, P.; Bouquety, J.C.; Mada, A.; Daguisy, A.M.; Georges, A.J. An outbreak of human semliki forest virus infections in central african republic. Am. J. Trop. Med. Hyg. 1990, 42, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Willems, W.R.; Kaluza, G.; Boschek, C.B.; Bauer, H.; Hager, H.; Schutz, H.J.; Feistner, H. Semliki forest virus: Cause of a fatal case of human encephalitis. Science 1979, 203, 1127–1129. [Google Scholar] [CrossRef] [PubMed]
- Bradish, C.J.; Allner, K.; Maber, H.B. The virulence of original and derived strains of semliki forest virus for mice, guinea-pigs and rabbits. J. Gen. Virol. 1971, 12, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Reagan, R.L.; Strand, N.; Brueckner, A.L. Bwamba fever virus and semliki forest virus in young dogs. Proc. Soc. Exp. Biol. Med. 1953, 82, 642–643. [Google Scholar] [CrossRef] [PubMed]
- Vaha-Koskela, M.J.; Kallio, J.P.; Jansson, L.C.; Heikkila, J.E.; Zakhartchenko, V.A.; Kallajoki, M.A.; Kahari, V.M.; Hinkkanen, A.E. Oncolytic capacity of attenuated replicative semliki forest virus in human melanoma xenografts in severe combined immunodeficient mice. Cancer Res. 2006, 66, 7185–7194. [Google Scholar] [CrossRef] [PubMed]
- Heikkila, J.E.; Vaha-Koskela, M.J.; Ruotsalainen, J.J.; Martikainen, M.W.; Stanford, M.M.; McCart, J.A.; Bell, J.C.; Hinkkanen, A.E. Intravenously administered alphavirus vector va7 eradicates orthotopic human glioma xenografts in nude mice. PLoS ONE 2010, 5, e8603. [Google Scholar] [CrossRef] [PubMed]
- Ruotsalainen, J.J.; Kaikkonen, M.U.; Niittykoski, M.; Martikainen, M.W.; Lemay, C.G.; Cox, J.; De Silva, N.S.; Kus, A.; Falls, T.J.; Diallo, J.S.; et al. Clonal variation in interferon response determines the outcome of oncolytic virotherapy in mouse ct26 colon carcinoma model. Gene Ther. 2015, 22, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Autio, K.P.; Ruotsalainen, J.J.; Anttila, M.O.; Niittykoski, M.; Waris, M.; Hemminki, A.; Vaha-Koskela, M.J.; Hinkkanen, A.E. Attenuated semliki forest virus for cancer treatment in dogs: Safety assessment in two laboratory beagles. BMC Vet. Res. 2015, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Nishiya, A.T.; Massoco, C.O.; Felizzola, C.R.; Perlmann, E.; Batschinski, K.; Tedardi, M.V.; Garcia, J.S.; Mendonça, P.P.; Teixeira, T.F.; Zaidan Dagli, M.L. Comparative aspects of canine melanoma. Vet. Sci. 2016, 3, E7. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.J.; Bishop, R.F.; Veenstra, A.A.; Barnes, G.L.; Holmes, I.H.; Ruck, B.J. Pattern of shedding of two noncultivable viruses in stools of newborn babies. J. Med. Virol. 1978, 2, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.C.; Mochizuki, M.; Maeda, K.; Okuda, M.; Mizuno, T. Seroepidemiology of reovirus in healthy dogs in six prefectures in japan. J. Vet. Med. Sci. 2014, 76, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Selb, B.; Weber, B. A study of human reovirus igg and iga antibodies by elisa and western blot. J. Virol. Methods 1994, 47, 15–25. [Google Scholar] [CrossRef]
- Tai, J.H.; Williams, J.V.; Edwards, K.M.; Wright, P.F.; Crowe, J.E., Jr.; Dermody, T.S. Prevalence of reovirus-specific antibodies in young children in nashville, tennessee. J. Infect. Dis. 2005, 191, 1221–1224. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Sachdev, E.; Mita, A.C.; Mita, M.M. Clinical development of reovirus for cancer therapy: An oncolytic virus with immune-mediated antitumor activity. World J. Methodol. 2016, 6, 25–42. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.C.; Umeki, S.; Kubo, M.; Hayashi, T.; Shimoda, H.; Mochizuki, M.; Maeda, K.; Baba, K.; Hiraoka, H.; Coffey, M.; et al. Oncolytic reovirus in canine mast cell tumor. PLoS ONE 2013, 8, e73555. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.L.; Hirasawa, K.; Yang, A.D.; Shields, M.A.; Lee, P.W. Reovirus oncolysis: The ras/ralgef/p38 pathway dictates host cell permissiveness to reovirus infection. Proc. Natl. Acad. Sci. USA. 2004, 101, 11099–11104. [Google Scholar] [CrossRef] [PubMed]
- Igase, M.; Hwang, C.C.; Coffey, M.; Okuda, M.; Noguchi, S.; Mizuno, T. The oncolytic effects of reovirus in canine solid tumor cell lines. J. Vet. Med. Sci. 2015, 77, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.C.; Igase, M.; Sakurai, M.; Haraguchi, T.; Tani, K.; Itamoto, K.; Shimokawa, T.; Nakaichi, M.; Nemoto, Y.; Noguchi, S.; et al. Oncolytic reovirus therapy: Pilot study in dogs with spontaneously occurring tumours. Vet. Comp. Oncol. 2017, 16, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Mahalingam, D.; Fountzilas, C.; Moseley, J.; Noronha, N.; Tran, H.; Chakrabarty, R.; Selvaggi, G.; Coffey, M.; Thompson, B.; Sarantopoulos, J. A phase ii study of reolysin((r)) (pelareorep) in combination with carboplatin and paclitaxel for patients with advanced malignant melanoma. Cancer Chemother. Pharmacol. 2017, 79, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Saunders, M.; Anthoney, A.; Coffey, M.; Mettinger, K.; Thompson, B.; Melcher, A.; Nutting, C.M.; Harrington, K. Results of a phase ii study to evaluate the biological effects of intratumoral (itu) reolysin in combination with low dose radiotherapy (rt) in patients (pts) with advanced cancers. J. Clin. Oncol. 2009, 27, e14514. [Google Scholar]
- Jaime-Ramirez, A.C.; Yu, J.G.; Caserta, E.; Yoo, J.Y.; Zhang, J.; Lee, T.J.; Hofmeister, C.; Lee, J.H.; Kumar, B.; Pan, Q.; et al. Reolysin and histone deacetylase inhibition in the treatment of head and neck squamous cell carcinoma. Mol. Ther. Oncolytics 2017, 5, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Simmonds, P.; Lipkin, W.I. Discovery and characterization of mammalian endogenous parvoviruses. J. Virol. 2010, 84, 12628–12635. [Google Scholar] [CrossRef] [PubMed]
- Malerba, M.; Daeffler, L.; Rommelaere, J.; Iggo, R.D. Replicating parvoviruses that target colon cancer cells. J. Virol. 2003, 77, 6683–6691. [Google Scholar] [CrossRef] [PubMed]
- Grekova, S.P.; Raykov, Z.; Zawatzky, R.; Rommelaere, J.; Koch, U. Activation of a glioma-specific immune response by oncolytic parvovirus minute virus of mice infection. Cancer Gene Ther. 2012, 19, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Angelova, A.L.; Barf, M.; Geletneky, K.; Unterberg, A.; Rommelaere, J. Immunotherapeutic potential of oncolytic h-1 parvovirus: Hints of glioblastoma microenvironment conversion towards immunogenicity. Viruses 2017, 9, 382. [Google Scholar] [CrossRef] [PubMed]
- Paglino, J.C.; Ozduman, K.; van den Pol, A.N. Luiii parvovirus selectively and efficiently targets, replicates in, and kills human glioma cells. J. Virol. 2012, 86, 7280–7291. [Google Scholar] [CrossRef] [PubMed]
- Nykky, J.; Tuusa, J.E.; Kirjavainen, S.; Vuento, M.; Gilbert, L. Mechanisms of cell death in canine parvovirus-infected cells provide intuitive insights to developing nanotools for medicine. Int. J. Nanomed. 2010, 5, 417–428. [Google Scholar]
- Singh, P.; Destito, G.; Schneemann, A.; Manchester, M. Canine parvovirus-like particles, a novel nanomaterial for tumor targeting. J. Nanobiotechnol. 2006, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Cooper, B.J.; Carmichael, L.E.; Appel, M.J.; Greisen, H. Canine viral enteritis. Ii. Morphologic lesions in naturally occurring parvovirus infection. Cornell Vet. 1979, 69, 134–144. [Google Scholar] [PubMed]
- Martin, V.; Najbar, W.; Gueguen, S.; Grousson, D.; Eun, H.M.; Lebreux, B.; Aubert, A. Treatment of canine parvoviral enteritis with interferon-omega in a placebo-controlled challenge trial. Vet. Microbiol. 2002, 89, 115–127. [Google Scholar] [CrossRef]
- Ford, J.; McEndaffer, L.; Renshaw, R.; Molesan, A.; Kelly, K. Parvovirus infection is associated with myocarditis and myocardial fibrosis in young dogs. Vet. Pathol. 2017, 54, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Mech, L.D.; Goyal, S.M.; Paul, W.J.; Newton, W.E. Demographic effects of canine parvovirus on a free-ranging wolf population over 30 years. J. Wildl. Dis. 2008, 44, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Abdelmagid, O.Y.; Larson, L.; Payne, L.; Tubbs, A.; Wasmoen, T.; Schultz, R. Evaluation of the efficacy and duration of immunity of a canine combination vaccine against virulent parvovirus, infectious canine hepatitis virus, and distemper virus experimental challenges. Vet. Ther. 2004, 5, 173–186. [Google Scholar] [PubMed]
- Gore, T.C.; Lakshmanan, N.; Duncan, K.L.; Coyne, M.J.; Lum, M.A.; Sterner, F.J. Three-year duration of immunity in dogs following vaccination against canine adenovirus type-1, canine parvovirus, and canine distemper virus. Vet. Ther. 2005, 6, 5–14. [Google Scholar] [PubMed]
- Bergman, J.G.; Muniz, M.; Sutton, D.; Fensome, R.; Ling, F.; Paul, G. Comparative trial of the canine parvovirus, canine distemper virus and canine adenovirus type 2 fractions of two commercially available modified live vaccines. Vet. Rec. 2006, 159, 733–736. [Google Scholar] [CrossRef] [PubMed]
- Palermo, L.M.; Hueffer, K.; Parrish, C.R. Residues in the apical domain of the feline and canine transferrin receptors control host-specific binding and cell infection of canine and feline parvoviruses. J. Virol. 2003, 77, 8915–8923. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Towle, H.A.; Heng, H.G.; Greenberg, C.B.; Pool, R.R. Mandibular ossifying fibroma in a dog. Vet. Pathol. 2008, 45, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.R.; Bell, C.M.; Pinkerton, M.E.; Soukup, J.W. Maxillary ameloblastic fibroma in a dog. Vet. Pathol. 2011, 48, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Soltero-Rivera, M.; Engiles, J.B.; Reiter, A.M.; Reetz, J.; Lewis, J.R.; Sanchez, M.D. Benign and malignant proliferative fibro-osseous and osseous lesions of the oral cavity of dogs. Vet. Pathol. 2015, 52, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Phattarataratip, E.; Pholjaroen, C.; Tiranon, P. A clinicopathologic analysis of 207 cases of benign fibro-osseous lesions of the jaws. Int. J. Surg. Pathol. 2014, 22, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Fox, L.E.; Geoghegan, S.L.; Davis, L.H.; Hartzel, J.S.; Kubilis, P.; Gruber, L.A. Owner satisfaction with partial mandibulectomy or maxillectomy for treatment of oral tumors in 27 dogs. J. Am. Anim. Hosp. Assoc. 1997, 33, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.J. Cell death and inflammation: The case for il-1 family cytokines as the canonical damps of the immune system. FEBS J. 2016, 283, 2599–2615. [Google Scholar] [CrossRef] [PubMed]
- Laliberte, J.P.; Weisberg, A.S.; Moss, B. The membrane fusion step of vaccinia virus entry is cooperatively mediated by multiple viral proteins and host cell components. PLoS Pathog. 2011, 7, e1002446. [Google Scholar] [CrossRef] [PubMed]
- MacNeill, A.L.; Weishaar, K.M.; Seguin, B.; Powers, B.E. Safety of an oncolytic myxoma virus in dogs with soft tissue sarcoma. Viruses 2018, 10, 398. [Google Scholar] [CrossRef] [PubMed]
- Frey, S.E.; Newman, F.K.; Cruz, J.; Shelton, W.B.; Tennant, J.M.; Polach, T.; Rothman, A.L.; Kennedy, J.S.; Wolff, M.; Belshe, R.B.; et al. Dose-related effects of smallpox vaccine. N. Engl. J. Med. 2002, 346, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Gentschev, I.; Ehrig, K.; Donat, U.; Hess, M.; Rudolph, S.; Chen, N.; Yu, Y.A.; Zhang, Q.; Bullerdiek, J.; Nolte, I.; et al. Significant growth inhibition of canine mammary carcinoma xenografts following treatment with oncolytic vaccinia virus glv-1h68. J. Oncol. 2010, 2010, 736907. [Google Scholar] [CrossRef] [PubMed]
- Gentschev, I.; Adelfinger, M.; Josupeit, R.; Rudolph, S.; Ehrig, K.; Donat, U.; Weibel, S.; Chen, N.G.; Yu, Y.A.; Zhang, Q.; et al. Preclinical evaluation of oncolytic vaccinia virus for therapy of canine soft tissue sarcoma. PLoS ONE 2012, 7, e37239. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.S.; Gentschev, I.; Adelfinger, M.; Donat, U.; Hess, M.; Weibel, S.; Nolte, I.; Frentzen, A.; Szalay, A.A. Virotherapy of canine tumors with oncolytic vaccinia virus glv-1h109 expressing an anti-vegf single-chain antibody. PLoS ONE 2012, 7, e47472. [Google Scholar] [CrossRef] [PubMed]
- Gentschev, I.; Patil, S.S.; Adelfinger, M.; Weibel, S.; Geissinger, U.; Frentzen, A.; Chen, N.G.; Yu, Y.A.; Zhang, Q.; Ogilvie, G.; et al. Characterization and evaluation of a new oncolytic vaccinia virus strain livp6.1.1 for canine cancer therapy. Bioengineered 2013, 4, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Adelfinger, M.; Bessler, S.; Frentzen, A.; Cecil, A.; Langbein-Laugwitz, J.; Gentschev, I.; Szalay, A.A. Preclinical testing oncolytic vaccinia virus strain glv-5b451 expressing an anti-vegf single-chain antibody for canine cancer therapy. Viruses 2015, 7, 4075–4092. [Google Scholar] [CrossRef] [PubMed]
- Zizzo, N.; Patruno, R.; Zito, F.A.; Di Summa, A.; Tinelli, A.; Troilo, S.; Misino, A.; Ruggieri, E.; Goffredo, V.; Gadaleta, C.D.; et al. Vascular endothelial growth factor concentrations from platelets correlate with tumor angiogenesis and grading in a spontaneous canine non-hodgkin lymphoma model. Leuk. Lymphoma 2010, 51, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Kerr, P.J.; Ghedin, E.; DePasse, J.V.; Fitch, A.; Cattadori, I.M.; Hudson, P.J.; Tscharke, D.C.; Read, A.F.; Holmes, E.C. Evolutionary history and attenuation of myxoma virus on two continents. PLoS Pathog. 2012, 8, e1002950. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.; Kelly, K.J.; Stanford, M.M.; Galanis, C.; Chun, Y.S.; Fong, Y.; McFadden, G. Myxoma virus is oncolytic for human pancreatic adenocarcinoma cells. Ann. Surg. Oncol. 2008, 15, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Correa, R.J.; Komar, M.; Tong, J.G.; Sivapragasam, M.; Rahman, M.M.; McFadden, G.; Dimattia, G.E.; Shepherd, T.G. Myxoma virus-mediated oncolysis of ascites-derived human ovarian cancer cells and spheroids is impacted by differential akt activity. Gynecol. Oncol. 2012, 125, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Lun, X.; Yang, W.; Alain, T.; Shi, Z.Q.; Muzik, H.; Barrett, J.W.; McFadden, G.; Bell, J.; Hamilton, M.G.; Senger, D.L.; et al. Myxoma virus is a novel oncolytic virus with significant antitumor activity against experimental human gliomas. Cancer Res. 2005, 65, 9982–9990. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Madlambayan, G.J.; Rahman, M.M.; Smallwood, S.E.; Meacham, A.M.; Hosaka, K.; Scott, E.W.; Cogle, C.R.; McFadden, G. Myxoma virus targets primary human leukemic stem and progenitor cells while sparing normal hematopoietic stem and progenitor cells. Leukemia 2009, 23, 2313–2317. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Barrett, J.W.; Stanford, M.; Werden, S.J.; Johnston, J.B.; Gao, X.; Sun, M.; Cheng, J.Q.; McFadden, G. Infection of human cancer cells with myxoma virus requires akt activation via interaction with a viral ankyrin-repeat host range factor. Proc. Natl. Acad. Sci. USA. 2006, 103, 4640–4645. [Google Scholar] [CrossRef] [PubMed]
- Nathaniel, R.; MacNeill, A.L.; Wang, Y.X.; Turner, P.C.; Moyer, R.W. Cowpox virus crma, myxoma virus serp2 and baculovirus p35 are not functionally interchangeable caspase inhibitors in poxvirus infections. J. Gen. Virol. 2004, 85, 1267–1278. [Google Scholar] [CrossRef] [PubMed]
- Urbasic, A.S.; Hynes, S.; Somrak, A.; Contakos, S.; Rahman, M.M.; Liu, J.; MacNeill, A.L. Oncolysis of canine tumor cells by myxoma virus lacking the serp2 gene. Am. J. Vet. Res. 2012, 73, 1252–1261. [Google Scholar] [CrossRef] [PubMed]
- Dennis, M.M.; McSporran, K.D.; Bacon, N.J.; Schulman, F.Y.; Foster, R.A.; Powers, B.E. Prognostic factors for cutaneous and subcutaneous soft tissue sarcomas in dogs. Vet. Pathol. 2011, 48, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Kent, M.S.; Collins, C.J.; Ye, F. Activation of the akt and mammalian target of rapamycin pathways and the inhibitory effects of rapamycin on those pathways in canine malignant melanoma cell lines. Am. J. Vet. Res. 2009, 70, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.; Fadlalla, K.; Graham, T.; Tameru, B.; Fermin, C.D.; Samuel, T. Immunohistochemical evaluation of akt protein activation in canine mast cell tumours. J. Comp. Pathol. 2012, 147, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Costers, S.; Lefebvre, D.J.; Delputte, P.L.; Nauwynck, H.J. Porcine reproductive and respiratory syndrome virus modulates apoptosis during replication in alveolar macrophages. Arch. Virol. 2008, 153, 1453–1465. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.P.; Wu, J.L.; Su, Y.C.; Hong, J.R. Anti-bcl-2 family members, zfbcl-x(l) and zfmcl-1a, prevent cytochrome c release from cells undergoing betanodavirus-induced secondary necrotic cell death. Apoptosis 2007, 12, 1043–1060. [Google Scholar] [CrossRef] [PubMed]
- Teigler, J.E.; Phogat, S.; Franchini, G.; Hirsch, V.M.; Michael, N.L.; Barouch, D.H. The canarypox virus vector alvac induces distinct cytokine responses compared to the vaccinia virus-based vectors mva and nyvac in rhesus monkeys. J. Virol. 2014, 88, 1809–1814. [Google Scholar] [CrossRef] [PubMed]
- Jourdier, T.M.; Moste, C.; Bonnet, M.C.; Delisle, F.; Tafani, J.P.; Devauchelle, P.; Tartaglia, J.; Moingeon, P. Local immunotherapy of spontaneous feline fibrosarcomas using recombinant poxviruses expressing interleukin 2 (il2). Gene Ther. 2003, 10, 2126–2132. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.S.; Naik, A.; O‘Malley, M.E.; Popovic, P.; Demarco, R.; Hu, Y.; Yin, X.; Yang, S.; Zeh, H.J.; Moss, B.; et al. The enhanced tumor selectivity of an oncolytic vaccinia lacking the host range and antiapoptosis genes spi-1 and spi-2. Cancer Res. 2005, 65, 9991–9998. [Google Scholar] [CrossRef] [PubMed]
- Greiner, S.; Humrich, J.Y.; Thuman, P.; Sauter, B.; Schuler, G.; Jenne, L. The highly attenuated vaccinia virus strain modified virus ankara induces apoptosis in melanoma cells and allows bystander dendritic cells to generate a potent anti-tumoral immunity. Clin. Exp. Immunol. 2006, 146, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yu, Y.A.; Wang, E.; Chen, N.; Danner, R.L.; Munson, P.J.; Marincola, F.M.; Szalay, A.A. Eradication of solid human breast tumors in nude mice with an intravenously injected light-emitting oncolytic vaccinia virus. Cancer Res. 2007, 67, 10038–10046. [Google Scholar] [CrossRef] [PubMed]
- Gentschev, I.; Stritzker, J.; Hofmann, E.; Weibel, S.; Yu, Y.A.; Chen, N.; Zhang, Q.; Bullerdiek, J.; Nolte, I.; Szalay, A.A. Use of an oncolytic vaccinia virus for the treatment of canine breast cancer in nude mice: Preclinical development of a therapeutic agent. Cancer Gene Ther. 2009, 16, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Patruno, R.; Arpaia, N.; Gadaleta, C.D.; Passantino, L.; Zizzo, N.; Misino, A.; Lucarelli, N.M.; Catino, A.; Valerio, P.; Ribatti, D.; et al. Vegf concentration from plasma-activated platelets rich correlates with microvascular density and grading in canine mast cell tumour spontaneous model. J. Cell. Mol. Med. 2009, 13, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, J.H.; Duncan, R.B.; Huckle, W.R.; Troy, G.C. Expression of vascular endothelial growth factor in tumors and plasma from dogs with primary intracranial neoplasms. Am. J. Vet. Res. 2007, 68, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Restucci, B.; Papparella, S.; Maiolino, P.; De Vico, G. Expression of vascular endothelial growth factor in canine mammary tumors. Vet. Pathol. 2002, 39, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Sobczynska-Rak, A.; Polkowska, I.; Silmanowicz, P. Elevated vascular endothelial growth factor (vegf) levels in the blood serum of dogs with malignant neoplasms of the oral cavity. Acta Vet. Hung. 2014, 62, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Balboni, A.; Mollace, C.; Giunti, M.; Dondi, F.; Prosperi, S.; Battilani, M. Investigation of the presence of canine adenovirus (cadv) in owned dogs in northern italy. Res. Vet. Sci. 2014, 97, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Roy, S. Isolation and characterization of adenoviruses persistently shed from the gastrointestinal tract of non-human primates. PLoS Pathog. 2009, 5, e1000503. [Google Scholar] [CrossRef] [PubMed]
- Ternovoi, V.V.; Le, L.P.; Belousova, N.; Smith, B.F.; Siegal, G.P.; Curiel, D.T. Productive replication of human adenovirus type 5 in canine cells. J. Virol. 2005, 79, 1308–1311. [Google Scholar] [CrossRef] [PubMed]
- Abou El Hassan, M.A.I.; van der Meulen-Muileman, I.; Abbas, S.; Kruyt, F.A.E. Conditionally replicating adenoviruses kill tumor cells via a basic apoptotic machinery-independent mechanism that resembles necrosis-like programmed cell death. J. Virol. 2004, 78, 12243–12251. [Google Scholar] [CrossRef] [PubMed]
- Hemminki, A.; Kanerva, A.; Kremer, E.J.; Bauerschmitz, G.J.; Smith, B.F.; Liu, B.; Wang, M.; Desmond, R.A.; Keriel, A.; Barnett, B.; et al. A canine conditionally replicating adenovirus for evaluating oncolytic virotherapy in a syngeneic animal model. Mol. Ther. 2003, 7, 163–173. [Google Scholar] [CrossRef]
- Smith, B.F.; Curiel, D.T.; Ternovoi, V.V.; Borovjagin, A.V.; Baker, H.J.; Cox, N.; Siegal, G.P. Administration of a conditionally replicative oncolytic canine adenovirus in normal dogs. Cancer Biother. Radiopharm. 2006, 21, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Alcayaga-Miranda, F.; Cascallo, M.; Rojas, J.J.; Pastor, J.; Alemany, R. Osteosarcoma cells as carriers to allow antitumor activity of canine oncolytic adenovirus in the presence of neutralizing antibodies. Cancer Gene Ther. 2010, 17, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Le, L.P.; Rivera, A.A.; Glasgow, J.N.; Ternovoi, V.V.; Wu, H.; Wang, M.; Smith, B.F.; Siegal, G.P.; Curiel, D.T. Infectivity enhancement for adenoviral transduction of canine osteosarcoma cells. Gene Ther. 2006, 13, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Laborda, E.; Puig-Saus, C.; Rodriguez-Garcia, A.; Moreno, R.; Cascallo, M.; Pastor, J.; Alemany, R. A prb-responsive, rgd-modified, and hyaluronidase-armed canine oncolytic adenovirus for application in veterinary oncology. Mol. Ther. 2014, 22, 986–998. [Google Scholar] [CrossRef] [PubMed]
- Cejalvo, T.; Perise-Barrios, A.J.; Del Portillo, I.; Laborda, E.; Rodriguez-Milla, M.A.; Cubillo, I.; Vazquez, F.; Sardon, D.; Ramirez, M.; Alemany, R.; et al. Remission of spontaneous canine tumors after systemic cellular viroimmunotherapy. Cancer Res. 2018, 78, 4891–4901. [Google Scholar] [CrossRef] [PubMed]
- von Euler, H.; Sadeghi, A.; Carlsson, B.; Rivera, P.; Loskog, A.; Segall, T.; Korsgren, O.; Totterman, T.H. Efficient adenovector cd40 ligand immunotherapy of canine malignant melanoma. J. Immunother. 2008, 31, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Smedley, R.C.; Spangler, W.L.; Esplin, D.G.; Kitchell, B.E.; Bergman, P.J.; Ho, H.Y.; Bergin, I.L.; Kiupel, M. Prognostic markers for canine melanocytic neoplasms: A comparative review of the literature and goals for future investigation. Vet. Pathol. 2011, 48, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Westberg, S.; Sadeghi, A.; Svensson, E.; Segall, T.; Dimopoulou, M.; Korsgren, O.; Hemminki, A.; Loskog, A.S.; Totterman, T.H.; von Euler, H. Treatment efficacy and immune stimulation by adcd40l gene therapy of spontaneous canine malignant melanoma. J. Immunother. 2013, 36, 350–358. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).