Assessment of Lagrangian Modeling of Particle Motion in a Spiral Microchannel for Inertial Microfluidics
Abstract
:1. Introduction
Present Study
2. Computational Model
2.1. Generalized Model for Particle Tracking
2.2. Drag Force
2.3. Inertial Lift Model
2.4. Formulation for Particle Tracking
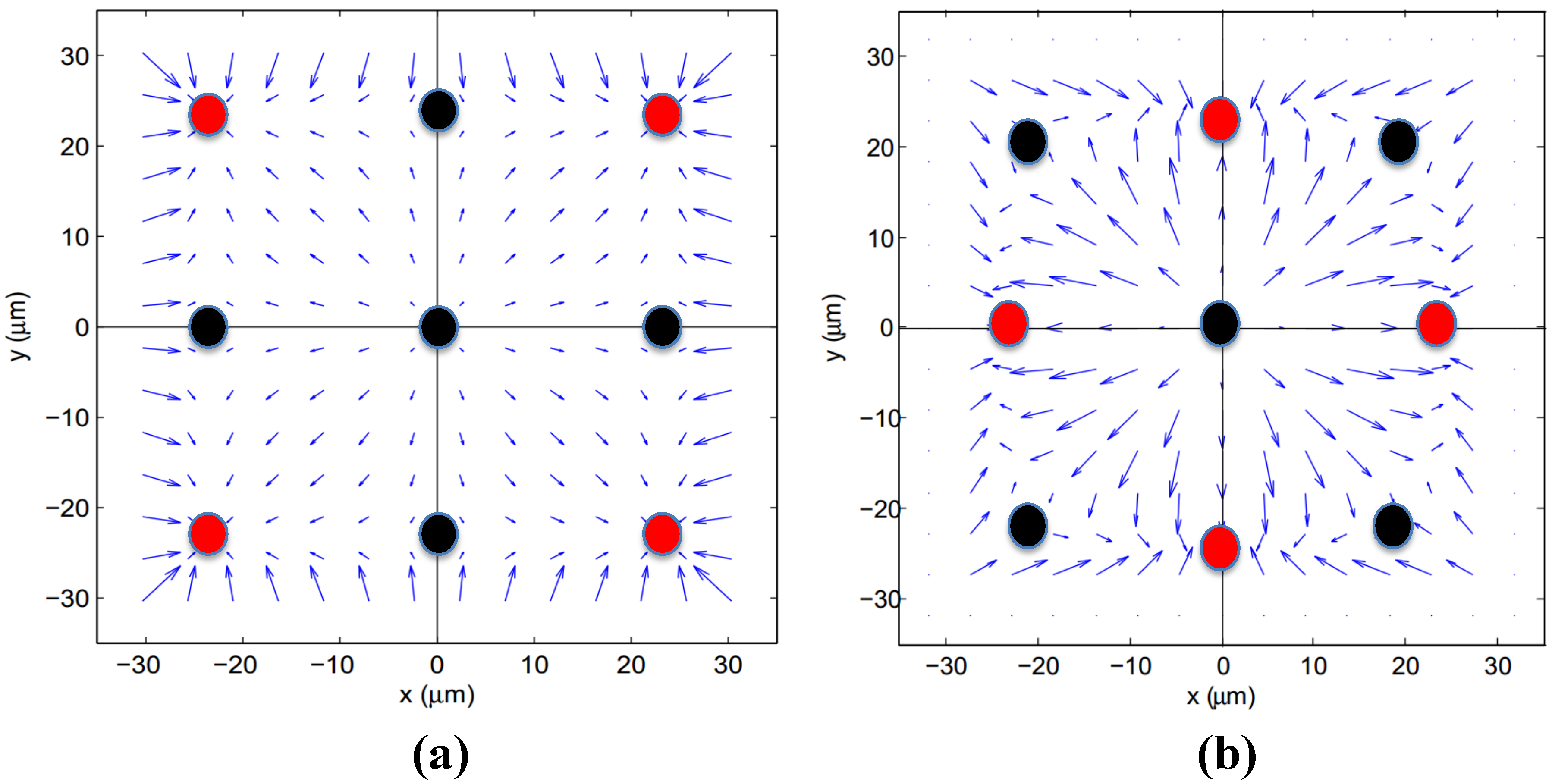
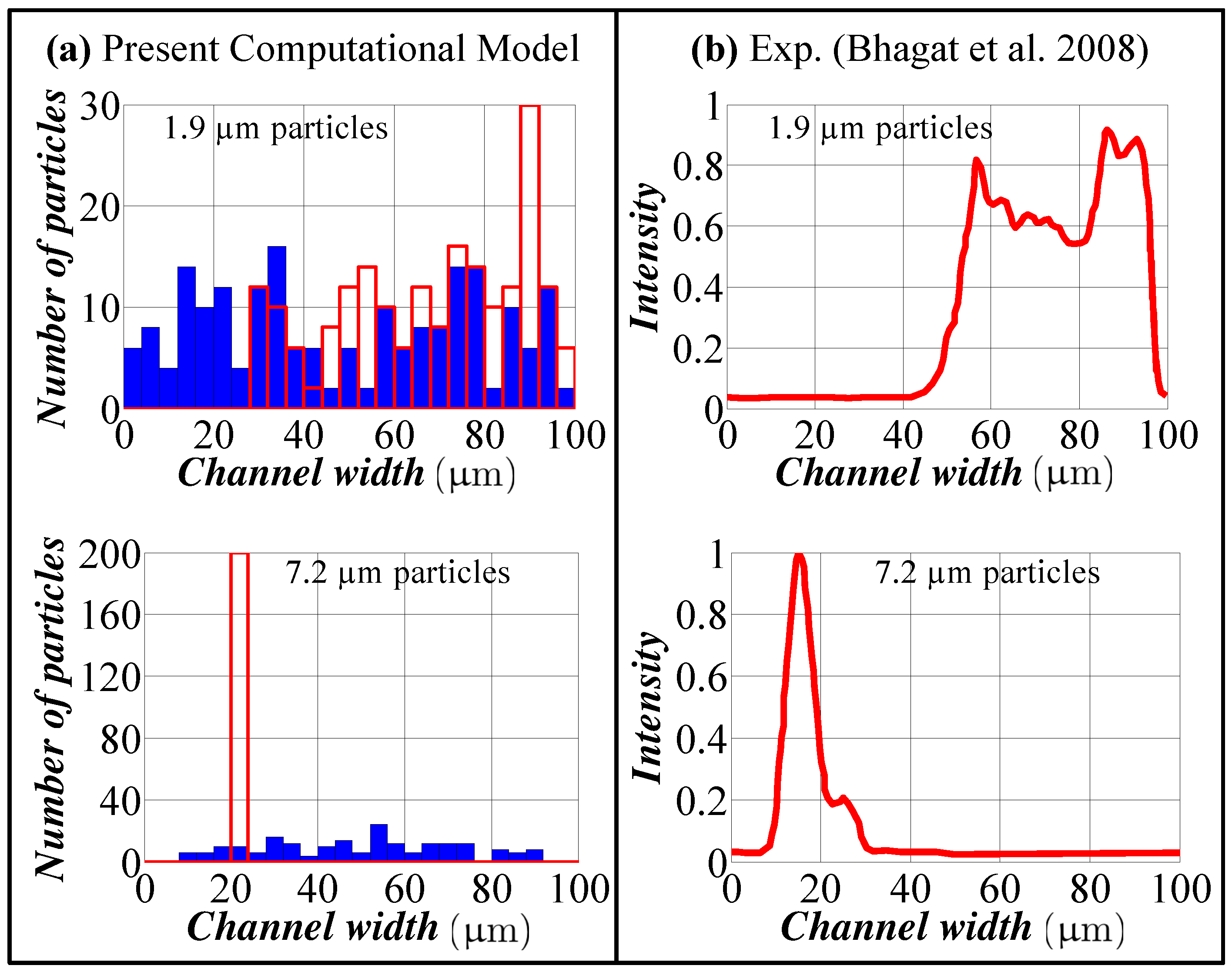
3. Model Verification
4. Computational Results
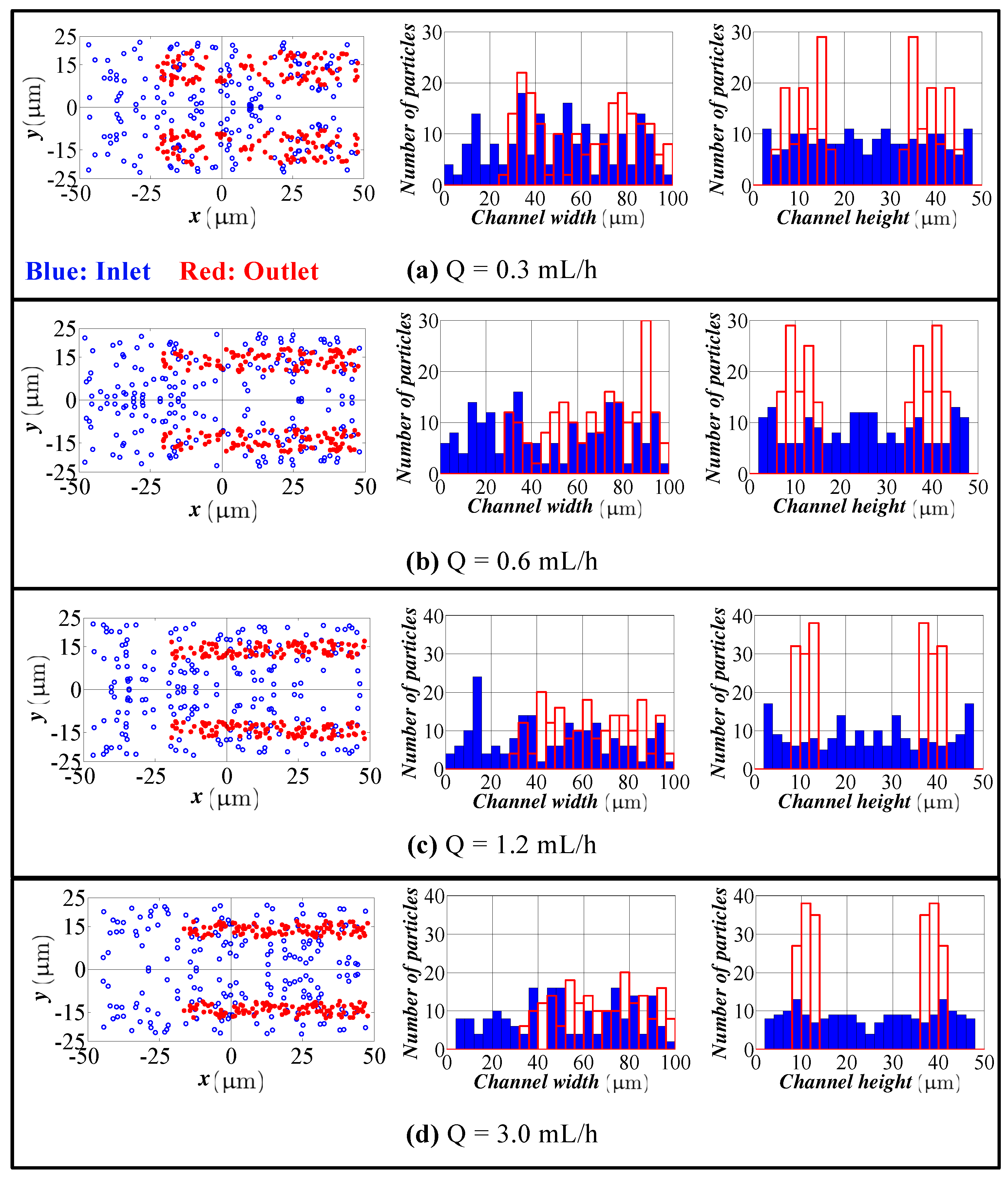
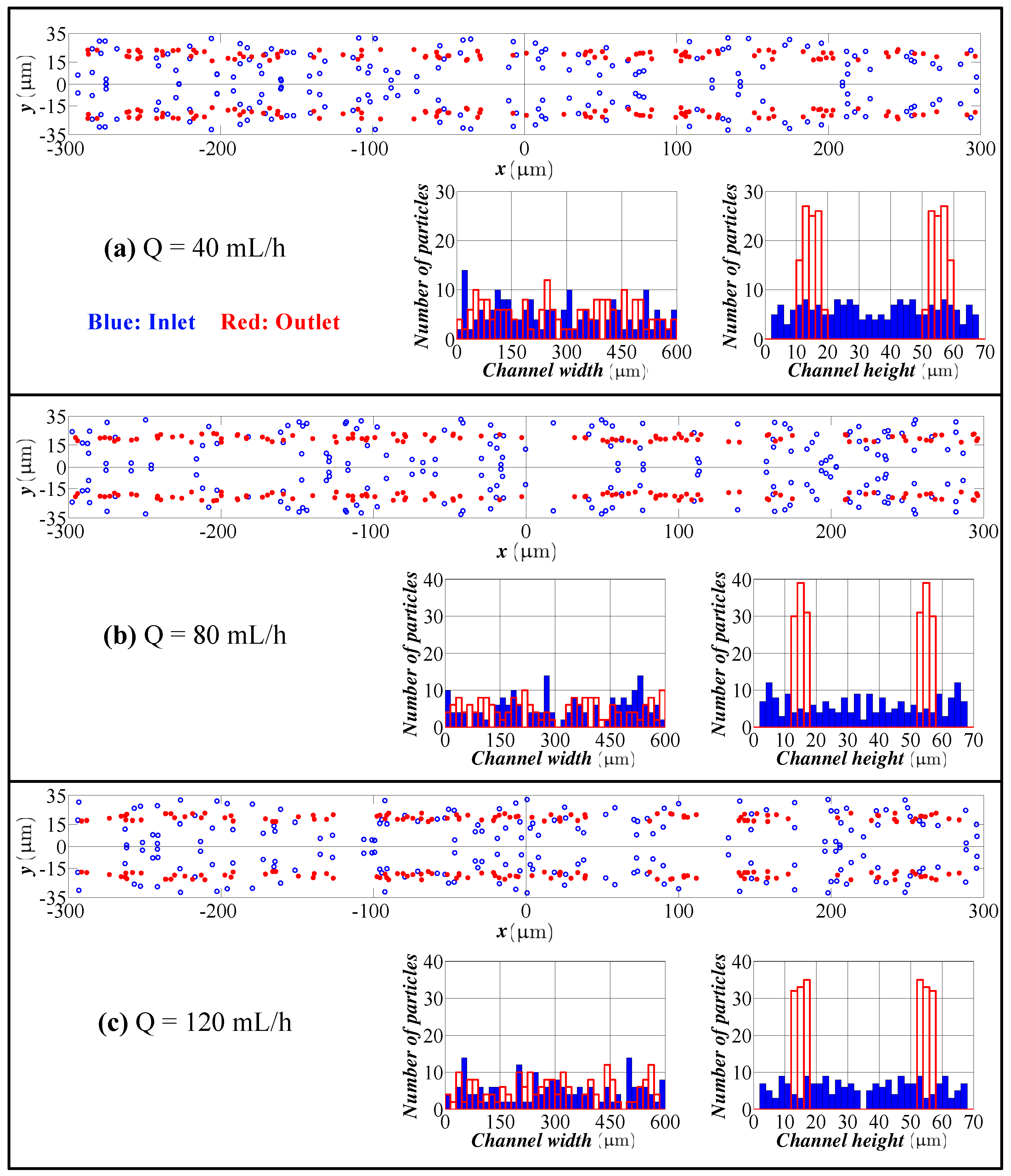
4.1. Spiral Channel with an AR 2.0
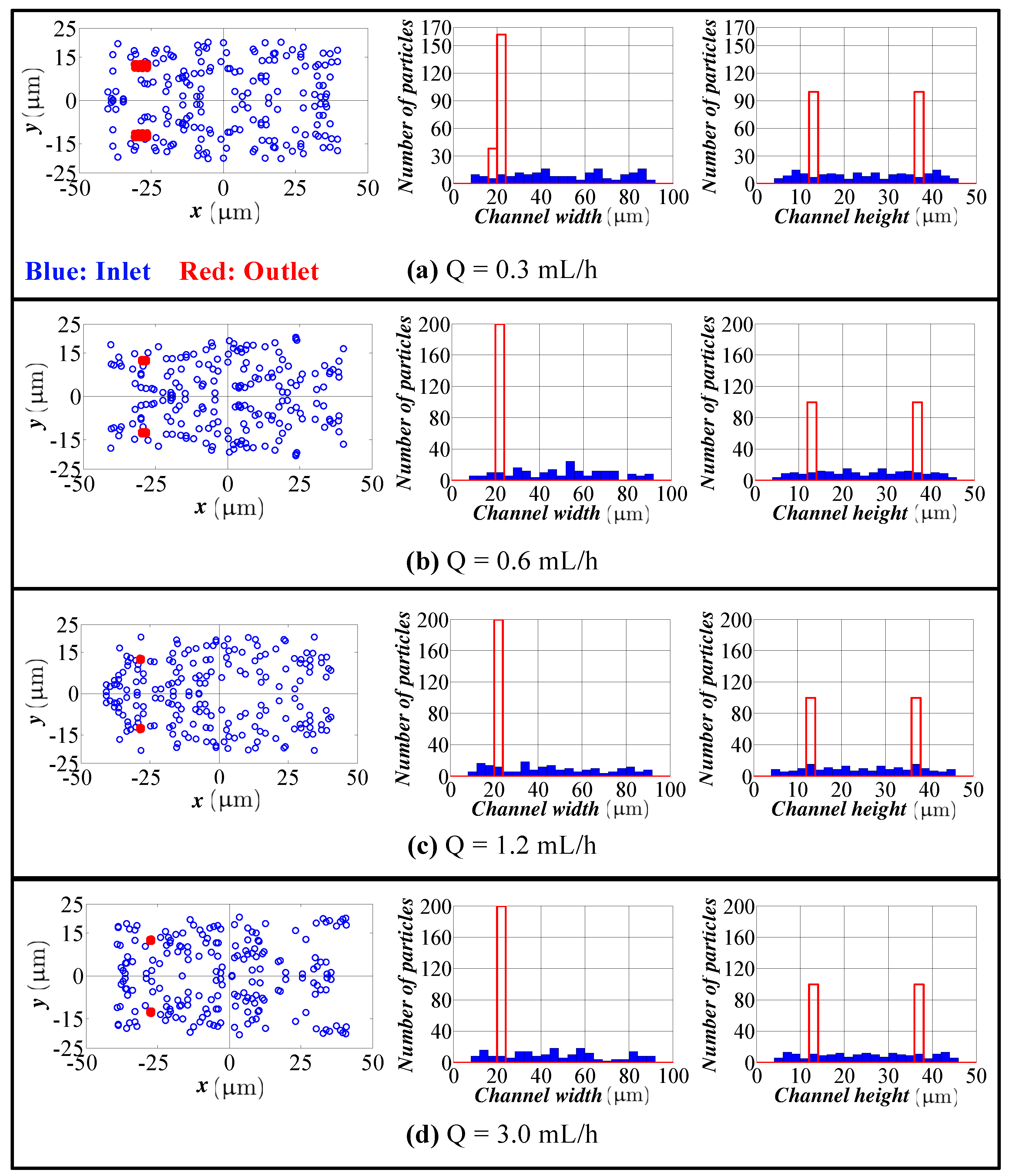
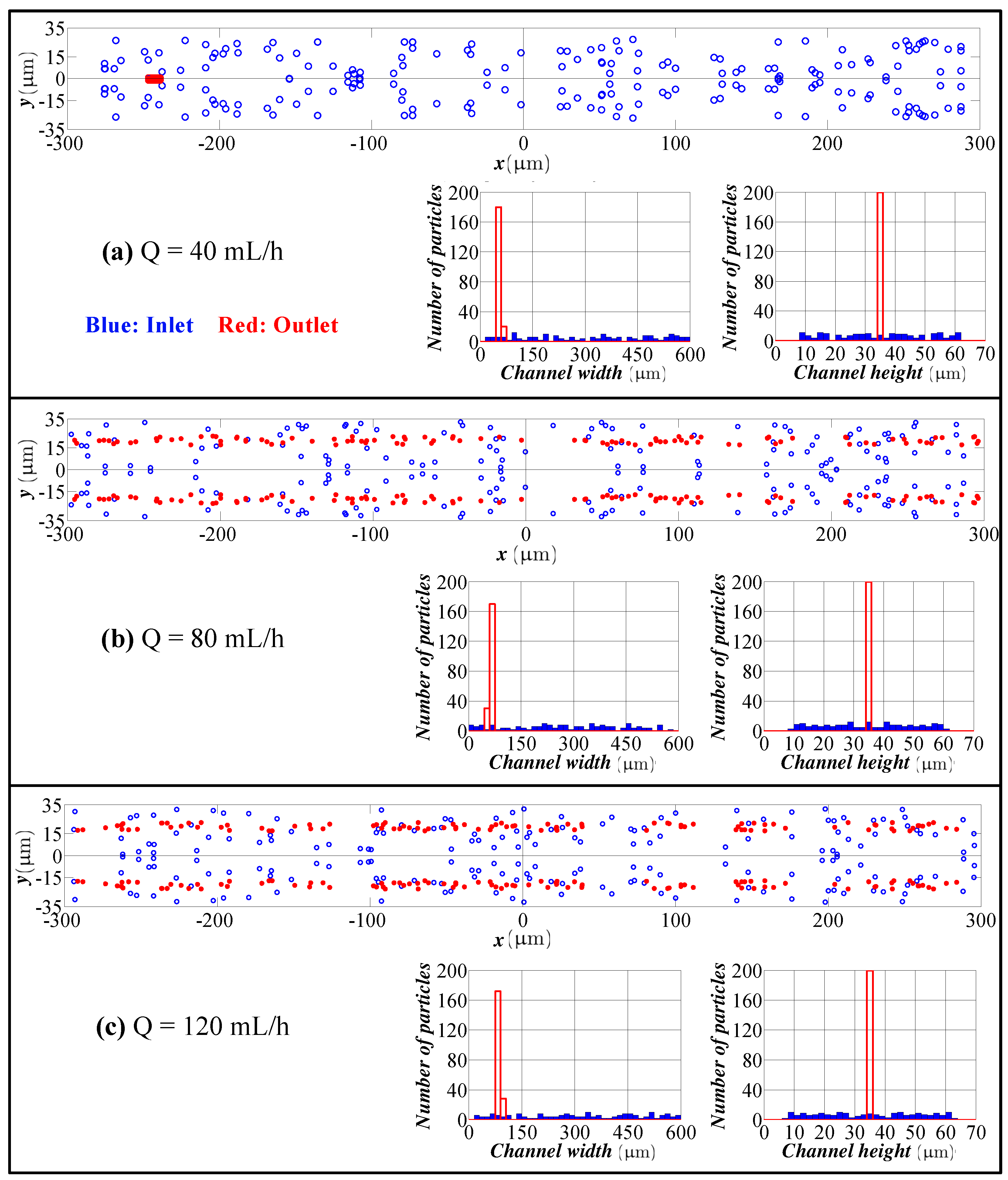
4.2. Spiral Channel with an AR 9.0
5. Experimentation
5.1. Experimental Setup and Procedure
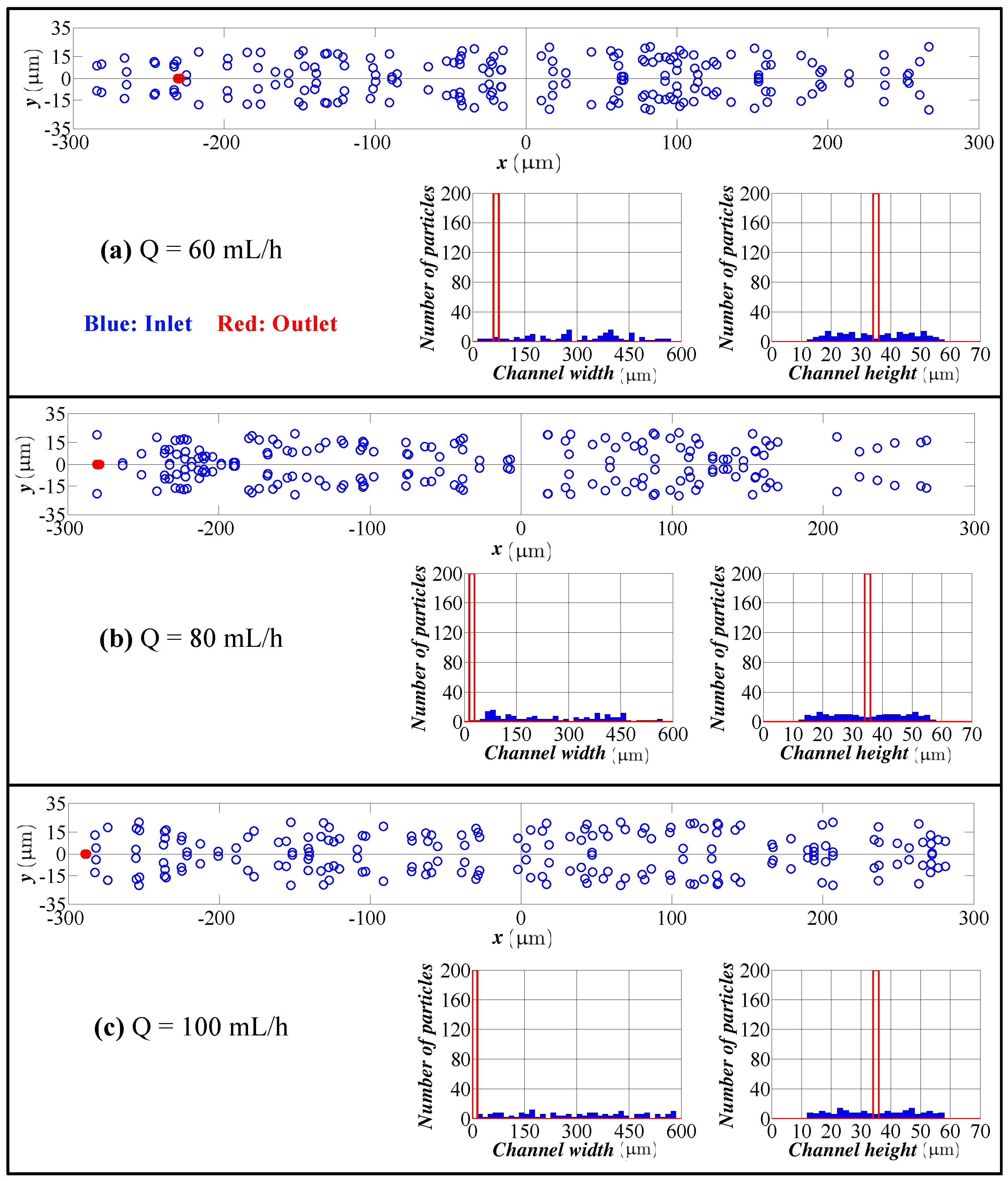
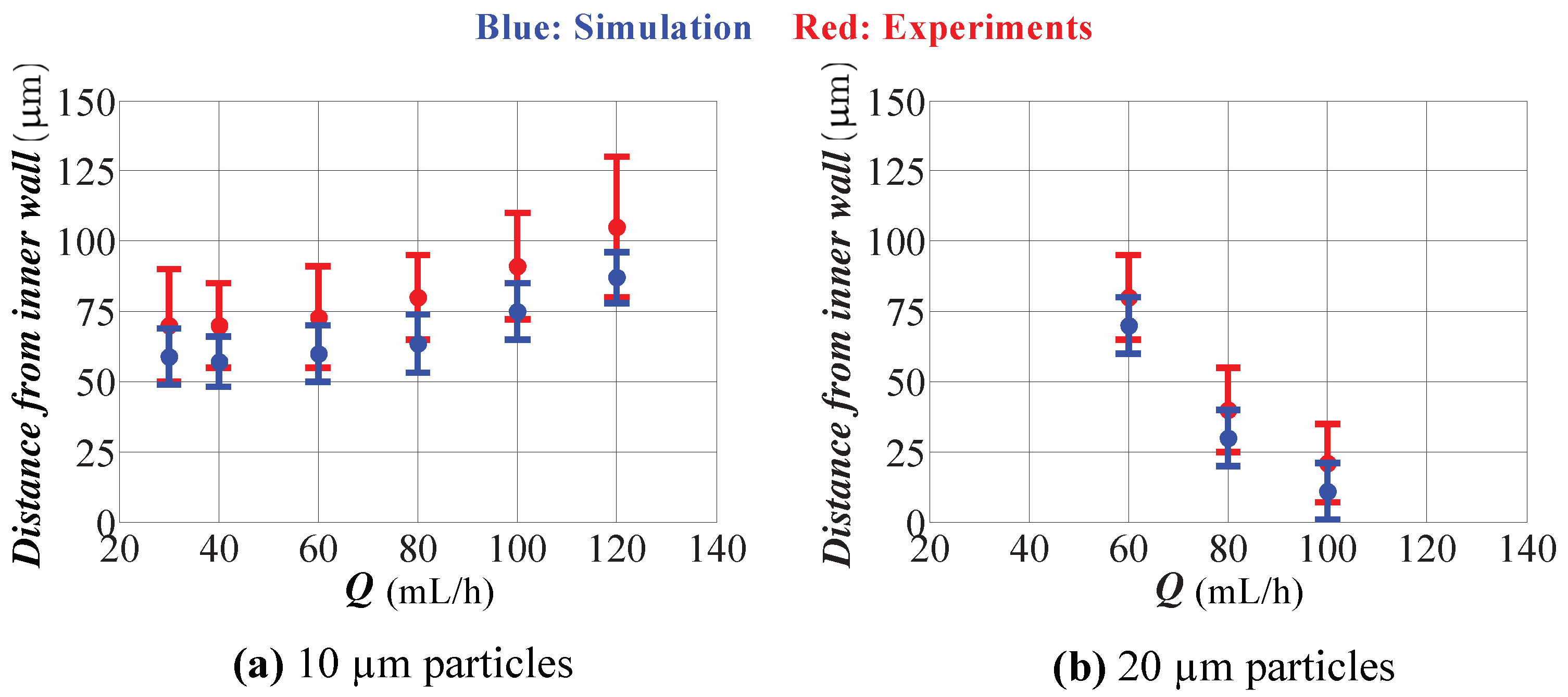
5.2. Experimental Results
6. Concluding Remarks and Outlook
- Since the model is based on Lagrangian modeling, once the flow field is obtained and correlation and/or numerical data are available for the drag and lift forces, simulations of many particles can be performed quite quickly and easily, which makes Monte Carlo-type simulations possible. With Monte Carlo-type simulations, a distribution of particles is obtained that gives realistic predictions for the actual performance.
- In this study, only the inlet location of the particles is varied. If there exists a significant size variation of the particles like in the case of cells, the size variation of the particles can be also included. For a given variation, the computational model can predict a variation of the particles’ location at the exit. Either for focusing or separation applications, the location of the particles at the exit of the microchannel is the important parameter for the design of the outlet section of a microfluidic device.
- The model predicts the position of the particles in the width direction, as well as in the height direction. Although visual inspection in the width direction is straight forward in the experimentation, the data for the height direction is not straight forward and requires a microscope with automated scanning in the height direction with an appropriate image processing software. Therefore, the data for the height direction through a numerical simulation are quite valuable.
- Predictions of the model for the location of the focused particles do not perfectly match with the experiments. This discrepancy is quite expected since our model uses Stokes’ law for the drag calculation, which is valid for a spherical particle in an infinite medium, and Hood’s results, which were derived for a spherical particle in a 3D Poiseuille flow in a straight channel. The model consistently underestimates the width of the focusing region and the location of the focusing region to the inner wall of the spiral channel for different flow rates. Keeping in mind this issue, one can extrapolate the numerical data for a better prediction of the actual operation.
- One important limitation of our computational model is the neglecting of particle-particle interactions. In microfluidics, typically, the number concentration of particles is low to avoid any blockage of the microchannel. Since flow rates are high for inertial microfluidics, blockage is not an issue; therefore the number concentration can be pushed to higher values which may violate the low concentration assumption in our model. The inclusion of particle-particle interactions requires simulations with the presence of the particles, which is a very challenging problem even with today’s computers.
- The current model can be extended for particles with different shapes other than spheres and/or channels with different cross-sectional geometries (one important geometry for inertial microfluidics is a tapered geometry in the literature). However, drag and inertial force correlations are required to employ LPDM for different particles and/or channel geometries. At this point, implementation of direct numerical simulation may be a viable option for the calculation of drag and lift force as proposed by [37].
- Although our model is implemented for a spiral channel, the current model is applicable for the assessment of a general curvilinear channel (e.g., serpentine microchannel).
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Cetin, B.; Ozer, M.B.; Solmaz, M.E. Microfluidic bio-particle manipulation for biotechnology. Biochem. Eng. J. 2014, 92, 63–82. [Google Scholar] [CrossRef] [Green Version]
- Segre, G. Radial particle displacements in Poiseuille flow of suspensions. Nature 1961, 189, 209–210. [Google Scholar] [CrossRef]
- Segre, G.; Silberberg, A. Behaviour of macroscopic rigid spheres in Poiseuille flow Part 2. Experimental results and interpretation. J. Fluid Mech. 1962, 14, 136–157. [Google Scholar] [CrossRef]
- Amini, H.; Lee, W.; Di Carlo, D. Inertial microfluidic physics. Lab Chip 2014, 14, 2739–2761. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Seo, K.W.; Lee, S.J. Lateral and cross-lateral focusing of spherical particles in a square microchannel. Lab Chip 2011, 11, 460–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Papautsky, I. Fundamentals of inertial focusing in microchannels. Lab Chip 2013, 13, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.J.; Gossett, D.R.; Carlo, D.D. Three dimensional, sheathless, and high-throughput microparticle inertial focusing through geometry-induced secondary flows. Small 2013, 9, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Choi, S.; Park, J. Inertial separation in a contraction–expansion array microchannel. J. Chromatogr. A 2011, 1218, 4138–4143. [Google Scholar] [CrossRef] [PubMed]
- Bhagat, A.A.S.; Kuntaegowdanahalli, S.S.; Papautsky, I. Continuous particle separation in spiral microchannels using dean flows and differential migration. Lab Chip 2008, 8, 1906–1914. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.A.; Okagbare, P.I.; Feng, J.; Hupert, M.L.; Patterson, D.; Gottert, J.; McCarley, R.L.; Nikitopoulos, D.; Murphy, M.C.; Soper, S.A. Highly Efficient Circulating Tumor Cell Isolation from Whole Blood and Label-Free Enumeration Using Polymer-Based Microfluidics with an Integrated Conductivity Sensor. J. Am. Chem. Soc. 2008, 130, 8633–8641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, B.; Ladd, A.J.C. Inertial migration of neutrally buoyant particles in a square duct: An investigation of multiple equilibrium positions. Phys. Fluids 2006, 18, 031704. [Google Scholar] [CrossRef]
- Carlo, D.D.; Edd, J.F.; Humphry, K.J.; Stoneand, H.A.; Toner, M. Particle segregation and dynamics in confined flows. Phys. Rev. Lett. 2009, 102, 094503. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Amini, H.; Stone, H.; Carlo, D.D. Dynamic self-assembly and control of microfluidic particle crystals. Proc. Natl. Acad. Sci. USA 2010, 107, 22413–22418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhagat, A.A.S.; Kuntaegowdanahalli, S.S.; Kaval, N.; Seliskar, C.J.; Papautsky, I. Inertial microfluidics for sheath-less high-throughput flow cytometry. Biomed. Microdevices 2010, 12, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Mach, A.J.; Carlo, D.D. Continuous scalable blood filtration device using inertial microfluidics. Biotechnol. Bioeng. 2010, 107, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Hur, S.C.; Tse, H.T.K.; Carlo, D.D. Sheathless inertial cell ordering for extreme throughput flow cytometry. Lab Chip 2010, 10, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Amini, H.; Sollier, E.; Weaver, W.M.; Carlo, D.D. Intrinsic particle-induced lateral transport in microchannels. Proc. Natl. Acad. Sci. USA 2012, 109, 11593–11598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hur, S.C.; Brinckerhoff, T.Z.; Walthers, C.M.; Dunn, J.C.Y.; Carlo, D.D. Label-free enrichment of adrenal cortical progenitor cells using inertial microfluidics. PLoS ONE 2012, 7, e46550. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Li, M.; Liu, C.; Zhang, Y.; Liu, D.; Liu, W.; Huand, G.; Jiang, X. Double spiral microchannel for label-free tumor cell separation and enrichment. Lab Chip 2012, 12, 3952–3960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Liu, C.; Li, M.; Wang, J.; Xianyu, Y.; Hu, G.; Jiang, X. Size-based hydrodynamic rare tumor cell separation in curved microfluidic channels. Biomicrofluidics 2013, 7, 011802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, H.W.; Warkiani, M.E.; Khoo, B.L.; Li, Z.R.; Soo, R.A.; Tan, D.S.W.; Lim, W.T.; Han, J.; Bhagat, A.A.S.; Lim, C.T. Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci. Rep. 2013, 3, 1259. [Google Scholar] [CrossRef] [PubMed]
- Warkiani, M.E.; Guan, G.; Luan, K.B.; Lee, W.C.; Bhagat, A.A.S.; Chaudhuri, P.K.; Tan, D.S.W.; Lim, W.T.; Lee, S.C.; Chen, P.C.Y.; et al. Slanted spiral microfluidics for the ultra-fast, label-free isolation of circulating tumor cells. Lab Chip 2014, 14, 128–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhar, M.; Wong, J.; Karimi, A.; Che, J.; Renier, C.; Matsumoto, M.; Triboulet, M.; Garon, E.B.; Goldman, J.W.; Rettig, M.B.; et al. High efficiency vortex trapping of circulating tumor cells. Biomicrofluidics 2015, 9, 064116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Hu, G.; Jiang, X.; Sun, J. Inertial focusing of spherical particles in rectangular microchannels over a wide range of Reynolds numbers. Lab Chip 2015, 15, 1168–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warkiani, M.E.; Khoo, B.L.; Wu, L.; Tay, A.K.P.; Bhagat, A.A.S.; Han, J.; Lim, C.T. Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics. Nat. Protocols 2016, 11, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Gascoyne, P.R.C.; Sangjo, S.; Jamileh, N.; Katherine, S.H. Correlations between the dielectric properties and exterior morphology of cells revealed by dielectrophoretic field-flow fractionation. Electrophoresis 2012, 34, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Coumans, F.A.W.; van Dalum, G.; Beck, M.; Terstappen, L.W.M.M. Filter Characteristics Influencing Circulating Tumor Cell Enrichment from Whole Blood. PLoS ONE 2013, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Karakaya, Z.; Baranoglu, B.; Cetin, B.; Yazici, A. A parallel boundary element formulation for tracking multiple particle trajectories in Stoke’s flow for microfluidic applications. Comput. Model. Eng. Sci. 2015, 104, 227–249. [Google Scholar]
- House, D.L.; Luo, H. Electrophoretic mobility of a colloidal cylinder between two parallel walls. Eng. Anal. Bound. Elem. 2010, 34, 471–476. [Google Scholar] [CrossRef]
- Solmaz, M.E.; Cetin, B.; Baranoglu, B.; Serhathoglu, M.; Biyikli, N. Boundary element method for optical force calibration in microfluidic dual-beam optical trap. Proc. SPIE 2015, 9548. [Google Scholar] [CrossRef]
- Cetin, B.; Oner, S.D.; Baranoglu, B. Modeling of dielectrophoretic particle motion: Point particle vs finite-sized particle. Electrophoresis 2017, in press. [Google Scholar]
- Sun, D.K.; Wang, Y.; Dong, A.P.; Sun, B.D. A three-dimensional quantitative study on the hydrodynamic focusing of particles with the immersed boundary Lattice Boltzmann method. Int. J. Heat Mass Transfer 2016, 94, 306–315. [Google Scholar] [CrossRef]
- Asmolov, E.S. The inertial lift on a spherical particle in a plane Poiseuille flow at large channel Reynolds number. J. Fluid Mech. 1999, 381, 63–87. [Google Scholar] [CrossRef]
- Hood, K.; Lee, S.; Roper, M. Inertial migration of a rigid sphere in three-dimensional Poiseuille flow. J. Fluid Mech. 2015, 765, 452–479. [Google Scholar] [CrossRef] [Green Version]
- Hood, K.; Kahkeshani, S.; Carlo, D.D.; Roper, M. Direct measurement of particle inertial migration in rectangular microchannels. Lab Chip 2016, 16, 2840–2850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakagawa, N.; Yabu, T.; Otomo, R.; Kase, A.; Makino, M.; Itano, T.; Sugihara-Seki, M. Inertial migration of a spherical particle in laminar square channel flows from low to high Reynolds numbers. J. Fluid Mech. 2015, 779, 776. [Google Scholar] [CrossRef]
- Lium, C.; Xue, C.; Sun, J.; Hu, G. A generalized formula for inertial lift on a sphere in microchannels. Lab Chip 2016, 16, 884–892. [Google Scholar] [Green Version]
- Buyukkocak, S.; Ozer, M.B.; Cetin, B. Numerical modeling of ultrasonic particle manipulation for microfluidic applications. Microfluid. Nanofluid. 2014, 17, 1025–1037. [Google Scholar] [CrossRef] [Green Version]
- Cetin, B.; Ozer, M.B.; Cagatay, E.; Buyukkocak, S. An integrated acoustic and dielectrophoretic particle manipulation in a microfluidic device for particle wash and separation fabricated by mechanical machining. Biomicrofluidics 2016, 10, 014112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winzen, A.; Oishi, M.; Oshima, M. A numerical model-assisted experimental design study of inertia-based particle focusing in stepped microchannels. Microfluid. Nanofluid. 2018, 22, 28. [Google Scholar] [CrossRef]
- Shamloo, A.; Mashhadian, A. Inertial particle focusing in serpentine channels on a centrifugal platform. Phys. Fluids 2018, 30, 012002. [Google Scholar] [CrossRef]
- Morsi, S.A.; Alexander, A.J. An investigation of particle trajectories in two-phase flow systems. J. Fluid Mech. 1972, 55, 193–208. [Google Scholar] [CrossRef]
- Rasooli, R. Modeling of Inertial Particle Flow and Entry Gas Flow in Micro-Channels. Master’s Thesis, Bilkent University, Ankara, Turkey, 2017. [Google Scholar]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasooli, R.; Çetin, B. Assessment of Lagrangian Modeling of Particle Motion in a Spiral Microchannel for Inertial Microfluidics. Micromachines 2018, 9, 433. https://doi.org/10.3390/mi9090433
Rasooli R, Çetin B. Assessment of Lagrangian Modeling of Particle Motion in a Spiral Microchannel for Inertial Microfluidics. Micromachines. 2018; 9(9):433. https://doi.org/10.3390/mi9090433
Chicago/Turabian StyleRasooli, Reza, and Barbaros Çetin. 2018. "Assessment of Lagrangian Modeling of Particle Motion in a Spiral Microchannel for Inertial Microfluidics" Micromachines 9, no. 9: 433. https://doi.org/10.3390/mi9090433
APA StyleRasooli, R., & Çetin, B. (2018). Assessment of Lagrangian Modeling of Particle Motion in a Spiral Microchannel for Inertial Microfluidics. Micromachines, 9(9), 433. https://doi.org/10.3390/mi9090433




