An Inexpensive Paper-Based Aluminum-Air Battery
Abstract
:1. Introduction
2. Materials and Methods
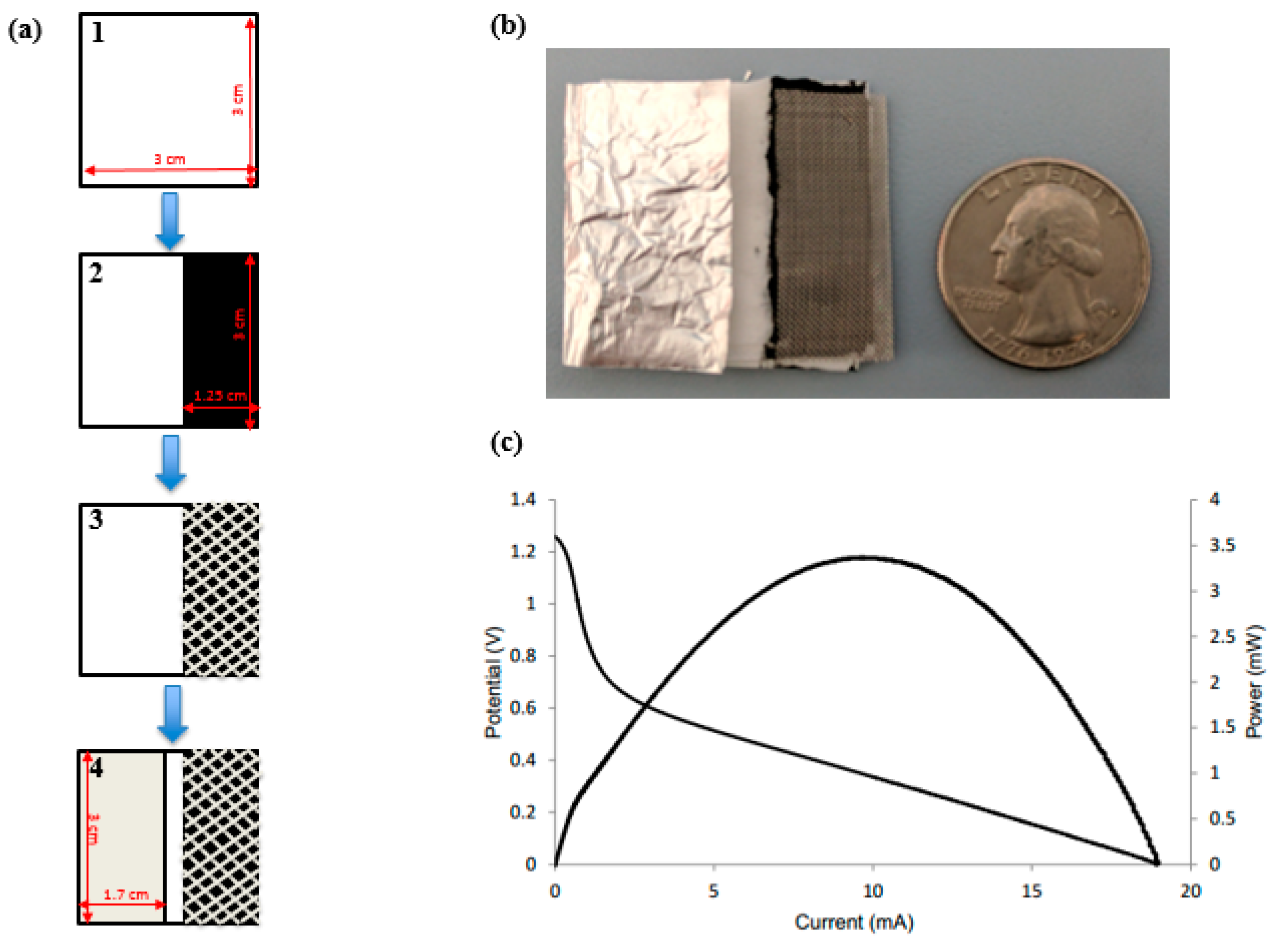
2.1. Device Fabrication
2.2. Instrumentation and Testing Procedures
3. Results
3.1. Working Principle
3.2. Cathode/Current Collector and Electrolyte
3.3. Dimensional Optimization
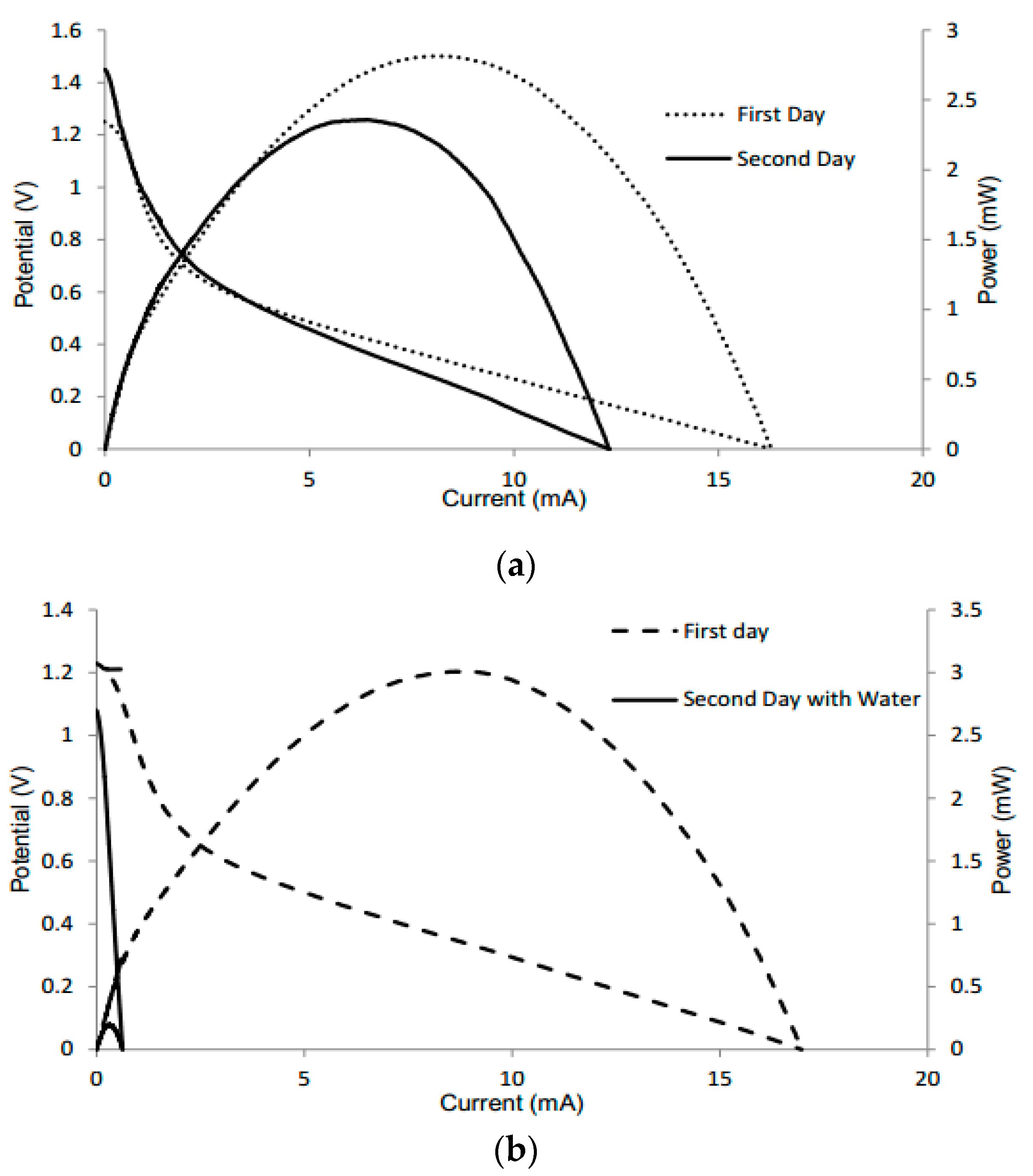
3.4. Electrolyte Optimization
3.5. Application
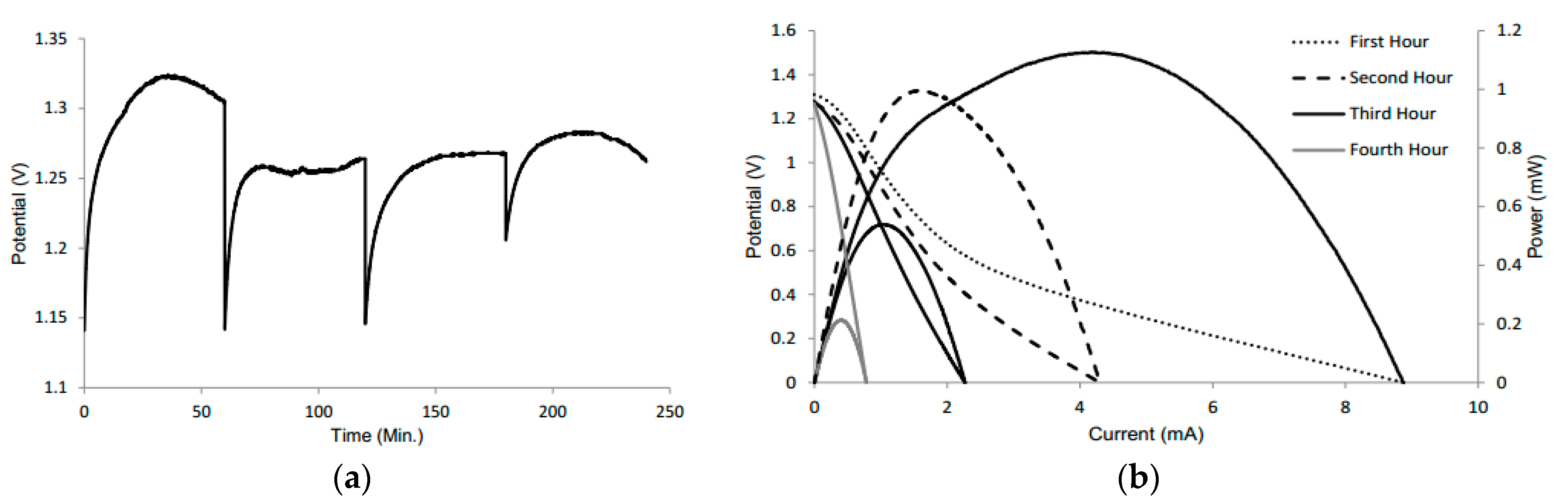
3.6. Exhaustion Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Winter, M.; Brodd, R.J. What are batteries, fuel cells, and supercapacitors? Chem. Rev. 2004, 10, 4245–4269. [Google Scholar] [CrossRef]
- Hulot, M. Comptes rendus hebdomadaires des séances de l’academie des sciences. Compt. Rend. 1855, 40, 148. [Google Scholar]
- Tommasi, D. Traite des Piles Electriques; George Carré: Paris, France, 1889; p. 131. [Google Scholar]
- Zaromb, S. The use and behavior of aluminum anodes in alkaline primary batteries. J. Electrochem. Soc. 1962, 109, 1125–1130. [Google Scholar] [CrossRef]
- Bockstie, D.; Trevethan, L.; Zaromb, S. Control of Al corrosion in caustic solutions. J. Electrochem. Soc. 1963, 110, 267–271. [Google Scholar] [CrossRef]
- Li, Q.; Bjerrum, N.J. Aluminum as anode for energy storage and conversion: A review. J. Power Sources 2002, 110, 1–10. [Google Scholar] [CrossRef]
- Macdonald, D.D.; Real, S.; Smedley, S.I.; Macdonald, M.U. Evaluation of alloy anodes for aluminum-air batteries IV. Electrochemical impedance analysis of pure aluminum in 4M KOH at 25 °C. J. Electrochem. Soc. 1988, 135, 2410–2414. [Google Scholar] [CrossRef]
- Licht, S.; Tel-Vered, R.; Levitin, G.; Yarnitzky, C. Solution activators of aluminum electrochemistry in organic media. J. Electrochem. Soc. 2000, 147, 496–501. [Google Scholar] [CrossRef]
- Doche, M.L.; Novel-Cattin, F.; Durnad, R.; Rameau, J.J. Characterization of different grades of aluminum anodes for aluminum/air batteries. J. Power Sources 1997, 65, 197–205. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Fraiwan, A.; Choi, S. Paper-based batteries: A review. Biosens. Bioelectron. 2013, 54, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.W.; Phillips, S.T.; Butte, M.J.; Whitesides, G.M. Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew. Chem. 2007, 46, 1318–1320. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.B.J. Two-step activation of paper batteries for high power generation: Design and fabrication of biofluid- and water-activated paper batteries. Micromech. Microeng. 2006, 16, 2312–2317. [Google Scholar] [CrossRef]
- Hu, L.; Cui, Y. Energy and environmental nanotechnology in conductive paper and textiles. Energy Environ. Sci. 2012, 5, 6423–6435. [Google Scholar] [CrossRef]
- Ferreira, I.; Bras, B.; Correia, N.; Barquinha, P.; Fortunato, E.; Martins, R. Self-rechargeable paper thin-film batteries: Performance and applications. J. Disp. Technol. 2010, 6, 332–335. [Google Scholar] [CrossRef]
- Thom, N.K.; Lewis, G.G.; DiTucci, M.J.; Phillips, S.T. Two general designs for fluidic batteries in paper-based microfluidic devices that provide predictable and tunable sources of power for on-chip assays. RSC Adv. 2013, 3, 6888–6895. [Google Scholar] [CrossRef]
- Zhang, X.; Li, J.; Chen, C.; Lou, B.; Zhang, L.; Wang, E. A self-powered microfluidic origami electrochemiluminescence biosensing platform. Chem. Commun. 2013, 49, 3866–3868. [Google Scholar] [CrossRef] [PubMed]
- Bauxite and Alumina Statistics and Information. Available online: http://minerals.usgs.gov/minerals/pubs/commodity/bauxite (accessed on 18 March 2017).
- Galvan, V.; Domalaon, K.; Tang, C.; Sotez, S.; Mendez, A.; Jalali-Heravi, M.; Purohit, K.; Pham, L.; Haan, J.; Gomez, F.A. An improved alkaline direct formate paper microfluidic fuel cell. Electrophoresis 2016, 37, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Appel, M.; Appleby, A.J. A ring-disk electrode study of the reduction of oxygen on active carbon in alkaline solution. Electrochim. Acta 1978, 23, 1243–1246. [Google Scholar] [CrossRef]
- Appleby, A.J.; Marie, J. Kinetics of oxygen reduction on carbon materials in alkaline solution. Electrochim. Acta 1979, 24, 195–202. [Google Scholar] [CrossRef]
- Yeager, E. Dioxygen electrocatalysis: Mechanisms in relation to catalyst structure. J. Mol. Catal. 1986, 38, 5–25. [Google Scholar] [CrossRef]
- Song, C.; Zhang, J. Electrocatalytic oxygen reduction reaction. In PEM Fuel Cell Electrocatalysts and Catalyst Layers; Springer: London, UK, 2008; pp. 89–134. [Google Scholar]
- Chen, L.D.; Norskov, J.K.; Luntz, A.C. Al-air batteries: Fundamental thermodynamic limitations from first-principles theory. J. Phys. Chem. Lett. 2015, 6, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Egan, D.R.; Ponce de Leon, C.; Wood, R.J.K.; Jones, R.L.; Stokes, K.R.; Walsh, F.C. Developments in electrode materials and electrolytes for aluminum-air batteries. J. Power Sources 2013, 236, 293–310. [Google Scholar] [CrossRef]
- Petrocelli, J.V. The electrochemical behavior of aluminum. J. Electrochem. Soc. 1951, 98, 183–186. [Google Scholar] [CrossRef]
- Wang, L.; Liu, F.; Wang, W.; Yang, G.; Zheng, D.; Wu, Z.; Leung, M.K.H. A high-capacity dual-electrolyte aluminum/air electrochemical cell. RSC Adv. 2014, 4, 30857–30863. [Google Scholar] [CrossRef]
- Patnaik, R.S.M.; Ganesh, S.; Ashok, G.; Ganesan, M.; Kapali, V. Heat management in aluminum/air batteries. J. Power Sources 1994, 50, 331–342. [Google Scholar] [CrossRef]
- Chasteen, S.V.; Chasteen, N.D.; Doherty, P. The salty science of the aluminum-air battery. Phys. Teach. 2008, 46, 544–547. [Google Scholar] [CrossRef]
- Morris, R.K.; Ottewill, G.A.; Barker, B.D. The aluminum-air cell: A hands-on approach to the teaching of electrochemical technology. Int. J. Eng. Educ. 2002, 18, 379–388. [Google Scholar]
- Qu, D. Investigation of oxygen reduction on activated carbon electrodes in alkaline solution. Carbon 2007, 45, 1296–1301. [Google Scholar] [CrossRef]
- Rondero-Daniel, E. Aluminum and stainless steel corrosion in ethanol and KOH solutions. Revista Mexicana Física 2009, 55, 72–75. [Google Scholar]
- Gregory, D.P. Metal-Air Batteries; Mills and Boon: London, UK, 1972. [Google Scholar]

| 1 M NaOH | Anode | Cathode | Average Voltage | Average Current | Average Power | RSD of Current |
| Aluminum | Silver epoxy with steel mesh and carbon | 1.38 V | 0.83 mA | 0.35 mW | 14.8% | |
| Aluminum | Carbon with copper | 1.21 V | 2.58 mA | 0.83 mW | 19.7% | |
| Aluminum | Carbon with steel mesh | 1.27 V | 2.92 mA | 0.55 mW | 3.7% | |
| Aluminum | Carbon | 1.28 V | 2.95 mA | 0.57 mW | 5.3% | |
| Aluminum | Silver epoxy and steel mesh | 1.39 V | 1.36 mA | 0.27 mW | 23.9% | |
| 1 M KOH | Anode | Cathode | Average Voltage | Average Current | Average Power | RSD of Current |
| Aluminum | Silver epoxy with steel mesh and Carbon | 1.65 V | 1.43 mA | 0.54 mW | 28.6% | |
| Aluminum | Carbon with copper | 1.23 V | 2.23 mA | 0.78 mW | 3.8% | |
| Aluminum | Carbon with steel mesh | 1.32 V | 2.67 mA | 0.53 mW | 3.5% | |
| Aluminum | Carbon | 1.23 V | 3.26 mA | 0.65 mW | 4.1% | |
| Aluminum | Silver epoxy and steel mesh | 1.43 V | 2.13 mA | 0.46 mW | 54.4% |
| Platform Size | Average Voltage | Average Current | Average Power | RSD of Current |
|---|---|---|---|---|
| 1 cm × 1 cm | 1.30 V | 2.69 mA | 0.54 mW | 10.0% |
| 1.5 cm × 1.5 cm | 1.32 V | 2.67 mA | 0.53 mW | 3.5% |
| 2 cm × 2 cm | 1.33 V | 2.32 mA | 0.49 mW | 7.1% |
| 3 cm × 3 cm | 1.33 V | 2.70 mA | 0.50 mW | 7.3% |
| Platform Size | Electrode Size | Average Voltage | Average Current | Average Power | RSD of Current |
|---|---|---|---|---|---|
| 1 cm × 1 cm | 0.67 cm × 0.33 cm | 1.27 V | 2.68 mA | 0.50 mW | 15.2% |
| 1.5 cm × 1.5 cm | 1.0 cm × 0.5 cm | 1.32 V | 2.67 mA | 0.53 mW | 3.5% |
| 2 cm × 2 cm | 1.3 cm × 0.67 cm | 1.32 V | 3.07 mA | 0.67 mW | 4.8% |
| 3 cm × 3 cm | 2 cm × 1 cm | 1.30 V | 6.00 mA | 1.12 mW | 3.4% |
| Anode Size | Average Voltage | Average Current | Average Power | RSD of Current |
|---|---|---|---|---|
| 1.5 cm × 0.5 cm | 1.33 V | 3.22 mA | 0.62 mW | 6.0% |
| 2 cm × 1 cm | 1.30 V | 6.00 mA | 1.12 mW | 3.4% |
| 2.5 cm × 1.5 cm | 1.33 V | 8.85 mA | 1.62 mW | 10.0% |
| 3 cm × 2 cm | 1.34 V | 11.10 mA | 2.07 mW | 11.7% |
| Cathode Size | Average Voltage | Average Current | Average Power | RSD of Current |
|---|---|---|---|---|
| 1.5 cm × 0.5 cm | 1.34 V | 2.56 mA | 0.54 mW | 5.1% |
| 2 cm × 1 cm | 1.30 V | 6.00 mA | 1.12 mW | 3.4% |
| 2.5 cm × 1.5 cm | 1.33 V | 10.41 mA | 1.97 mW | 5.4% |
| 3 cm × 2 cm | 1.38 V | 11.08 mA | 2.16 mW | 20.2% |
| KOH Concentration | Average Voltage | Average Current | Average Power | RSD of Current |
|---|---|---|---|---|
| 0.5 M | 1.35 V | 10.12 mA | 1.96 mW | 12.1% |
| 1 M | 1.31 V | 14.04 mA | 1.81 mW | 1.8% |
| 1.5 M | 1.27 V | 17.43 mA | 3.05 mW | 8.6% |
| 2 M | 1.24 V | 16.81 mA | 2.80 mW | 5.2% |
| 2.5 M | 1.24 V | 16.86 mA | 2.85 mW | 12.4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avoundjian, A.; Galvan, V.; Gomez, F.A. An Inexpensive Paper-Based Aluminum-Air Battery. Micromachines 2017, 8, 222. https://doi.org/10.3390/mi8070222
Avoundjian A, Galvan V, Gomez FA. An Inexpensive Paper-Based Aluminum-Air Battery. Micromachines. 2017; 8(7):222. https://doi.org/10.3390/mi8070222
Chicago/Turabian StyleAvoundjian, Ani, Vicente Galvan, and Frank A. Gomez. 2017. "An Inexpensive Paper-Based Aluminum-Air Battery" Micromachines 8, no. 7: 222. https://doi.org/10.3390/mi8070222
APA StyleAvoundjian, A., Galvan, V., & Gomez, F. A. (2017). An Inexpensive Paper-Based Aluminum-Air Battery. Micromachines, 8(7), 222. https://doi.org/10.3390/mi8070222





