Fabrication of Mesoscale Channel by Scanning Micro Electrochemical Flow Cell (SMEFC)
Abstract
:1. Introduction
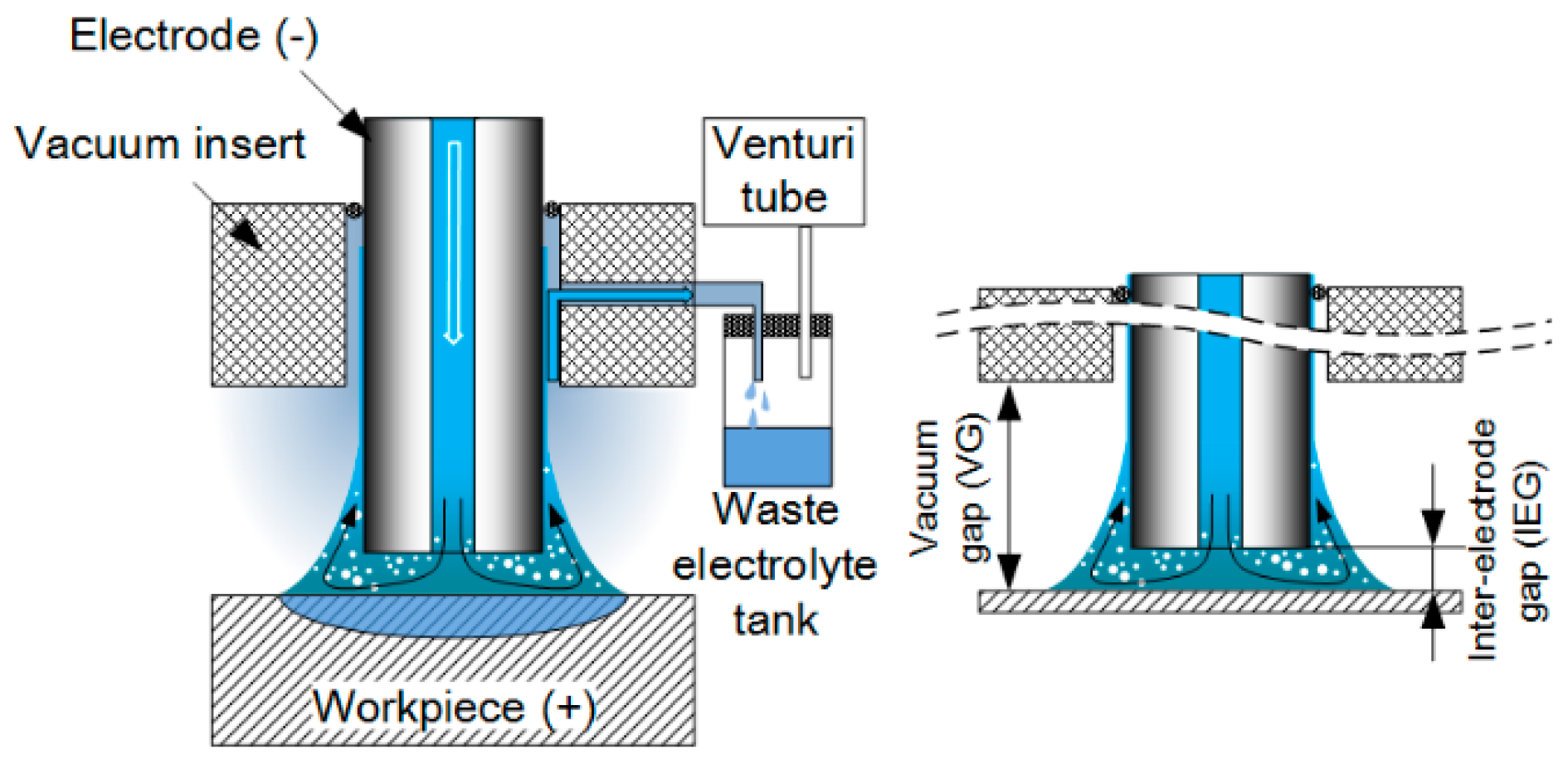
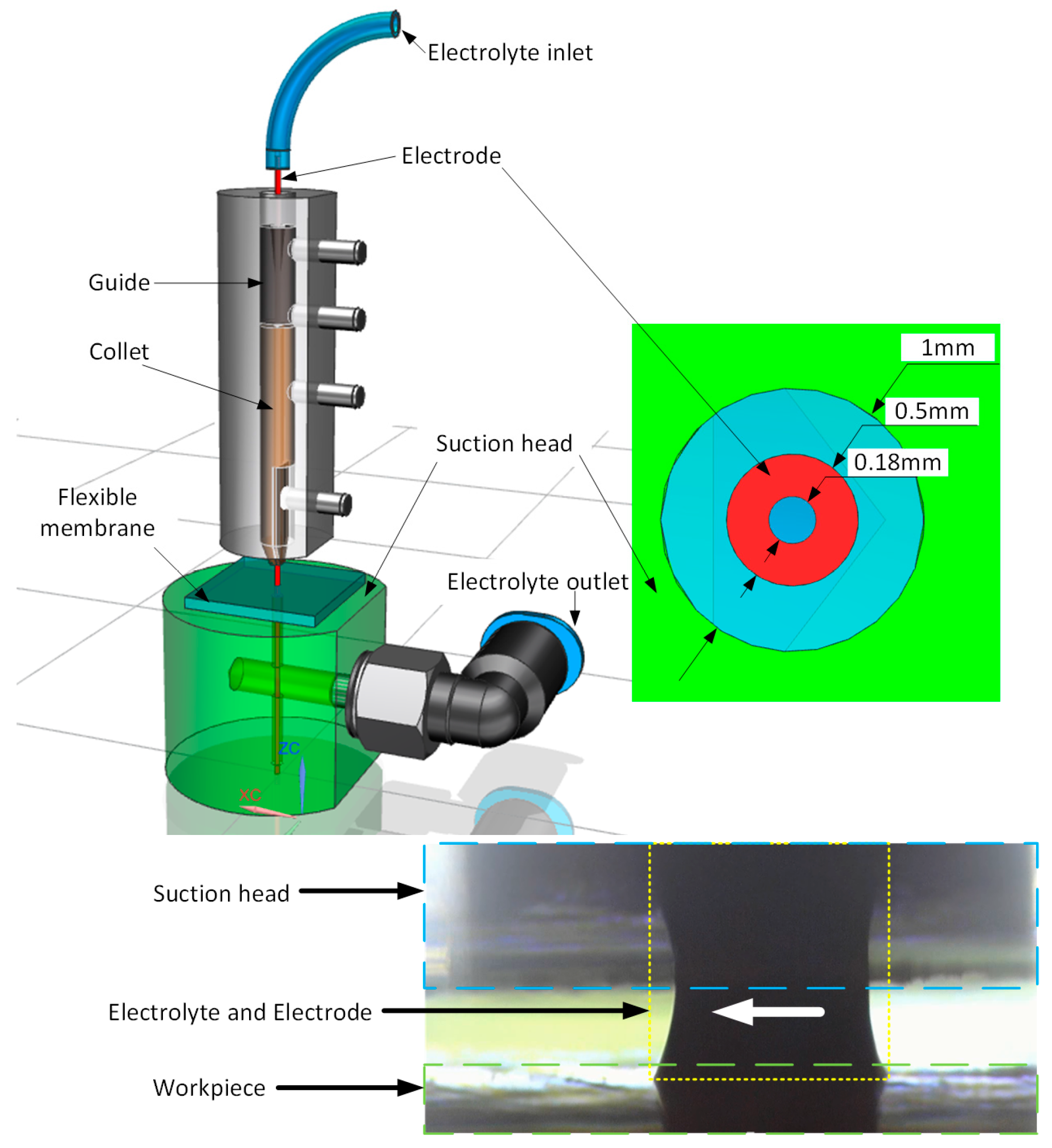
2. SMEFC Experimental System
3. SMEFC Machining Experiments and Discussion
3.1. Experimental Verification
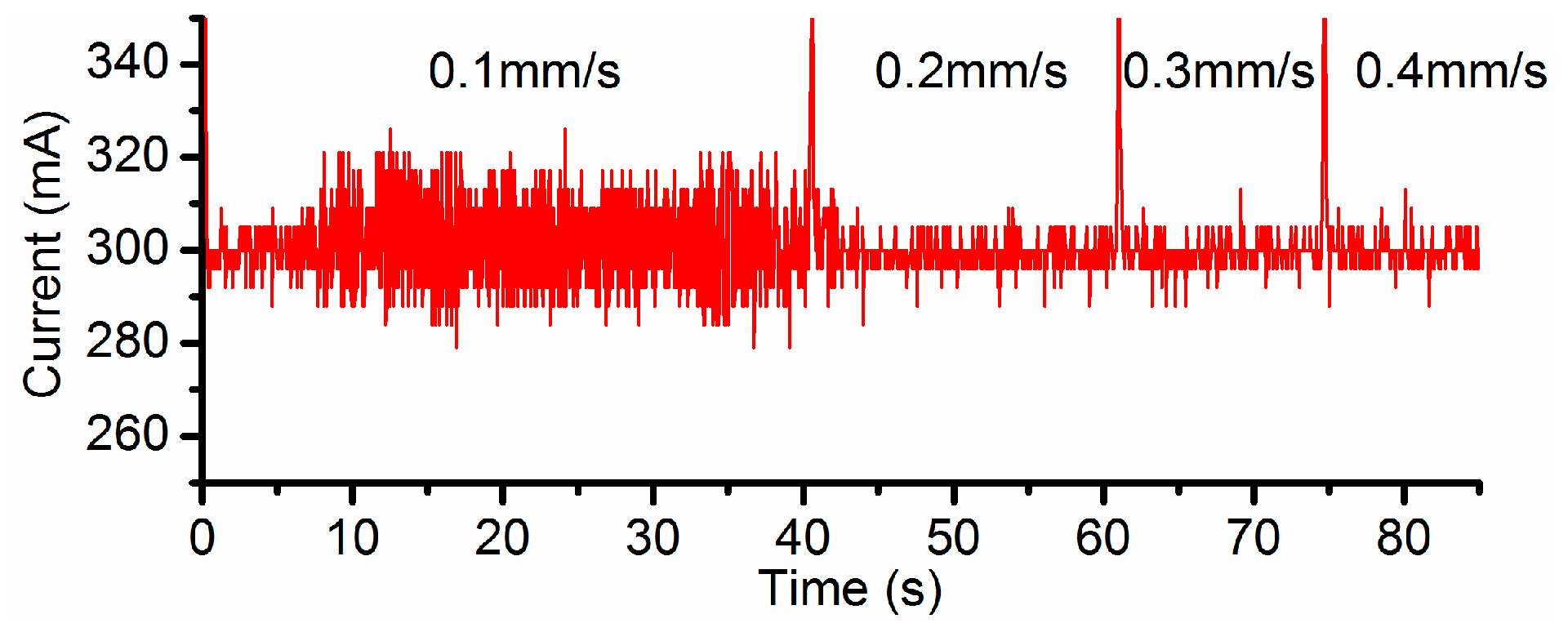
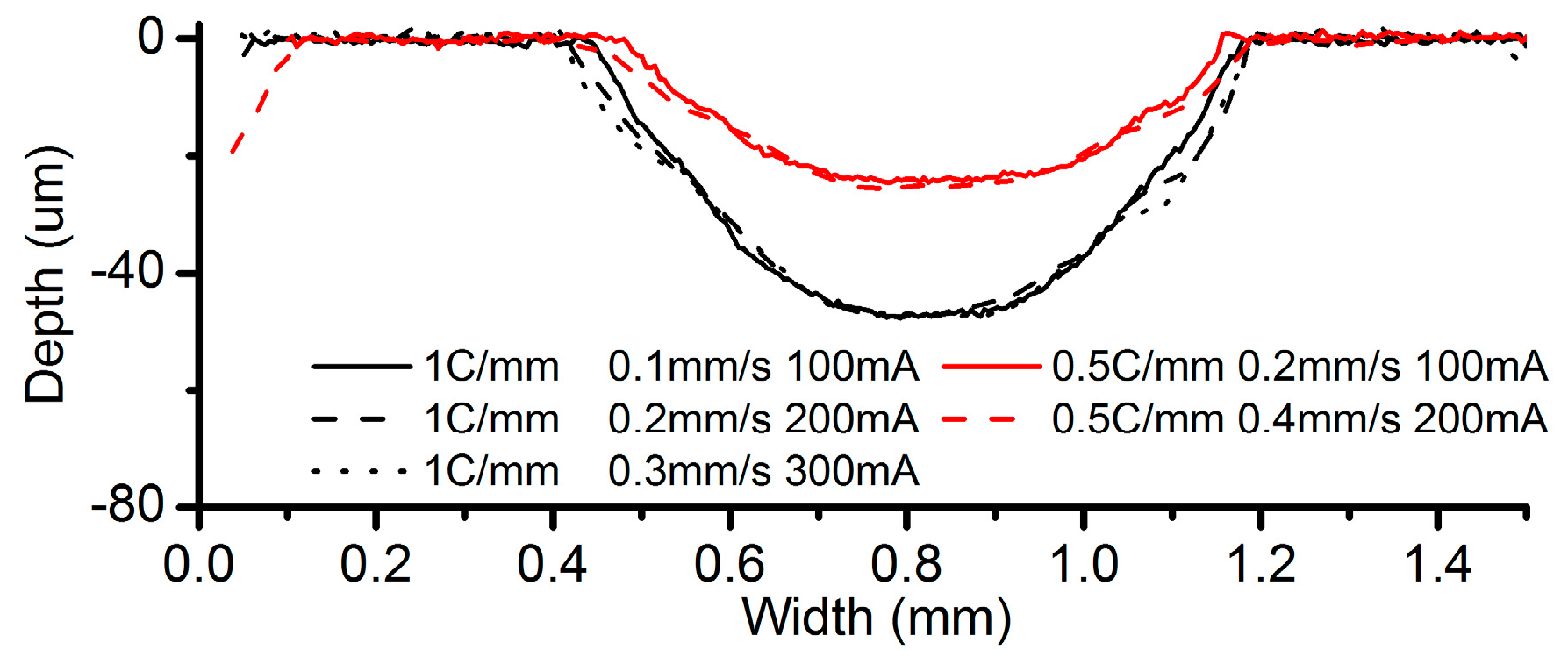
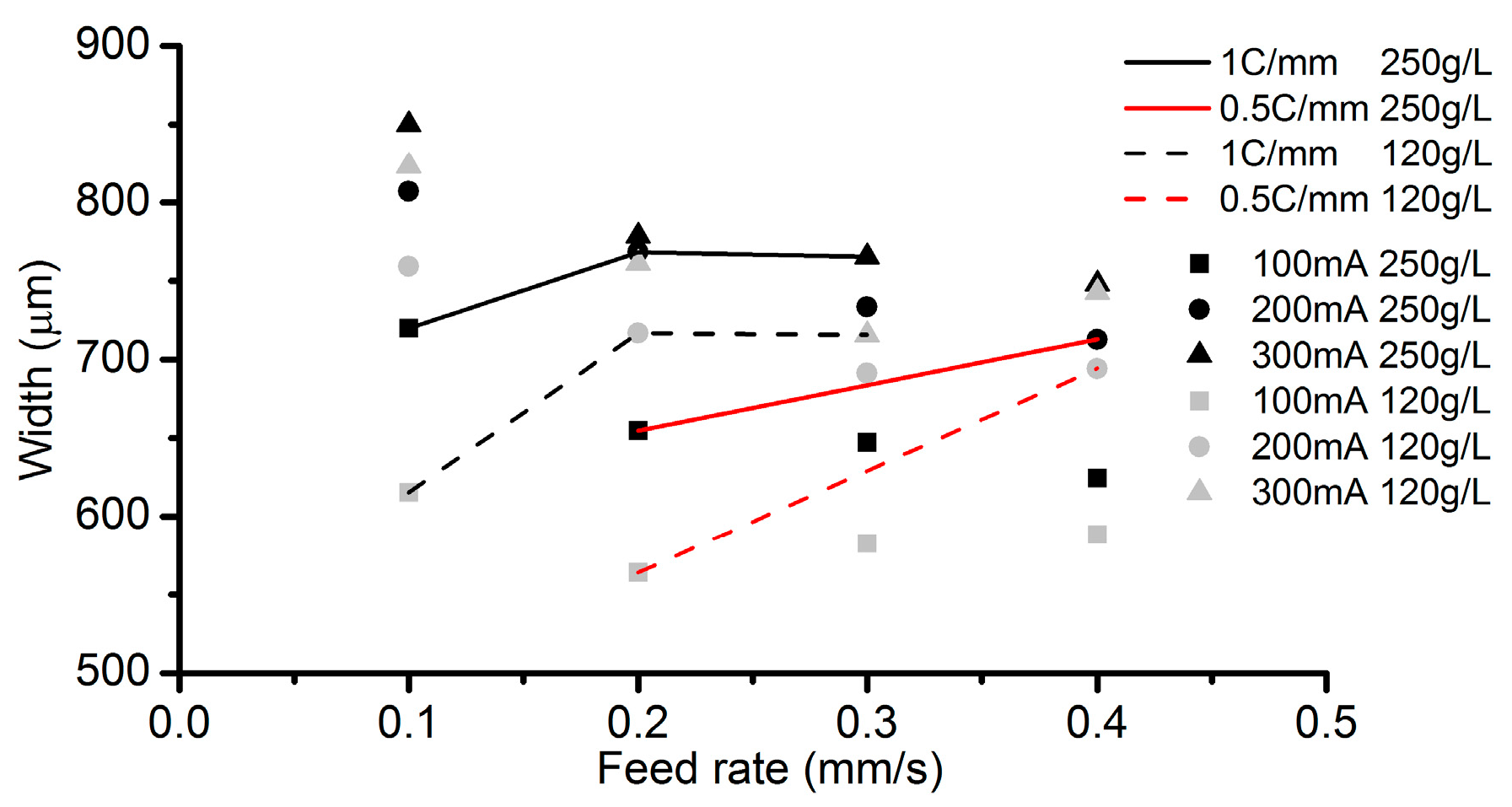
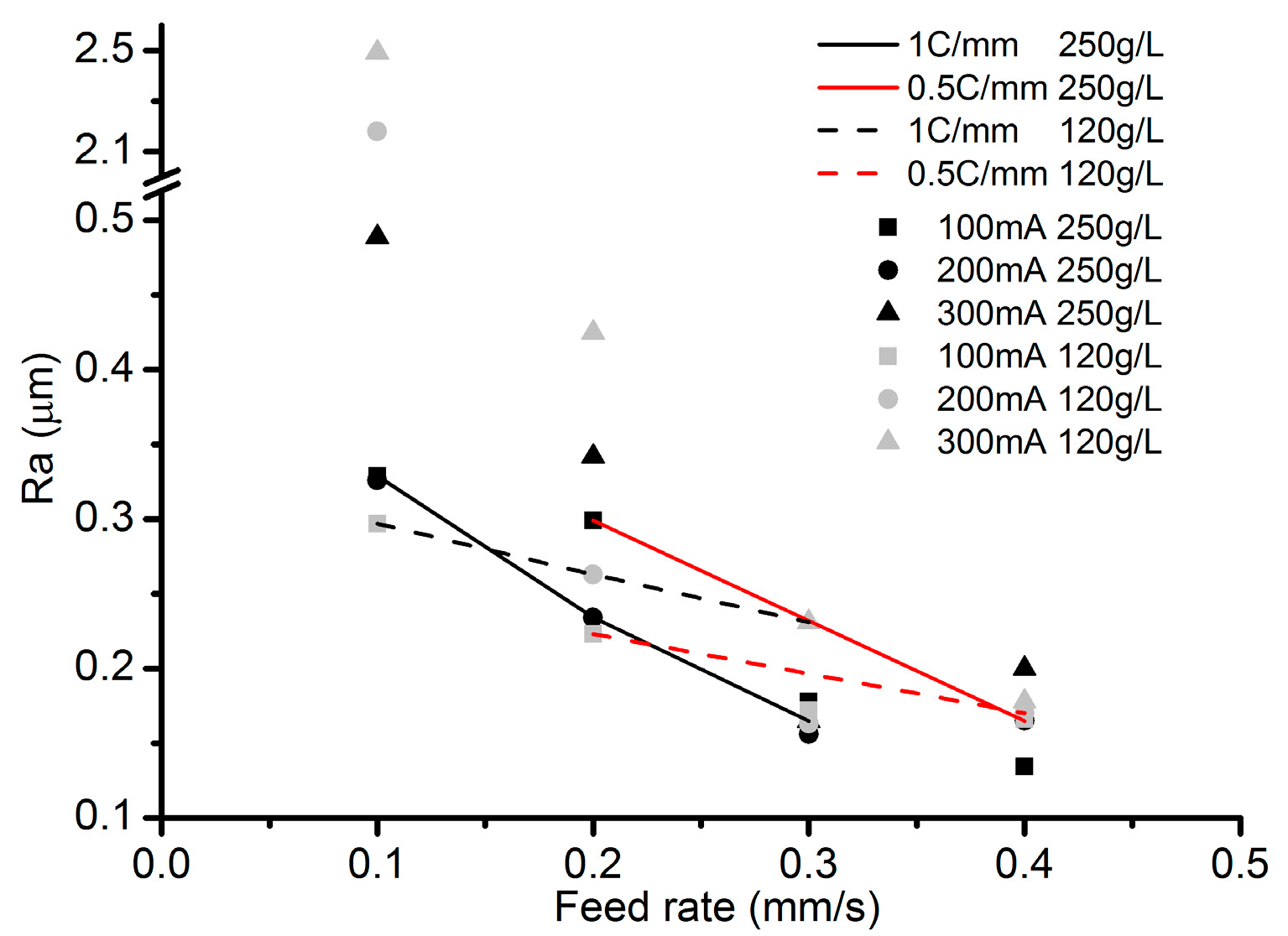
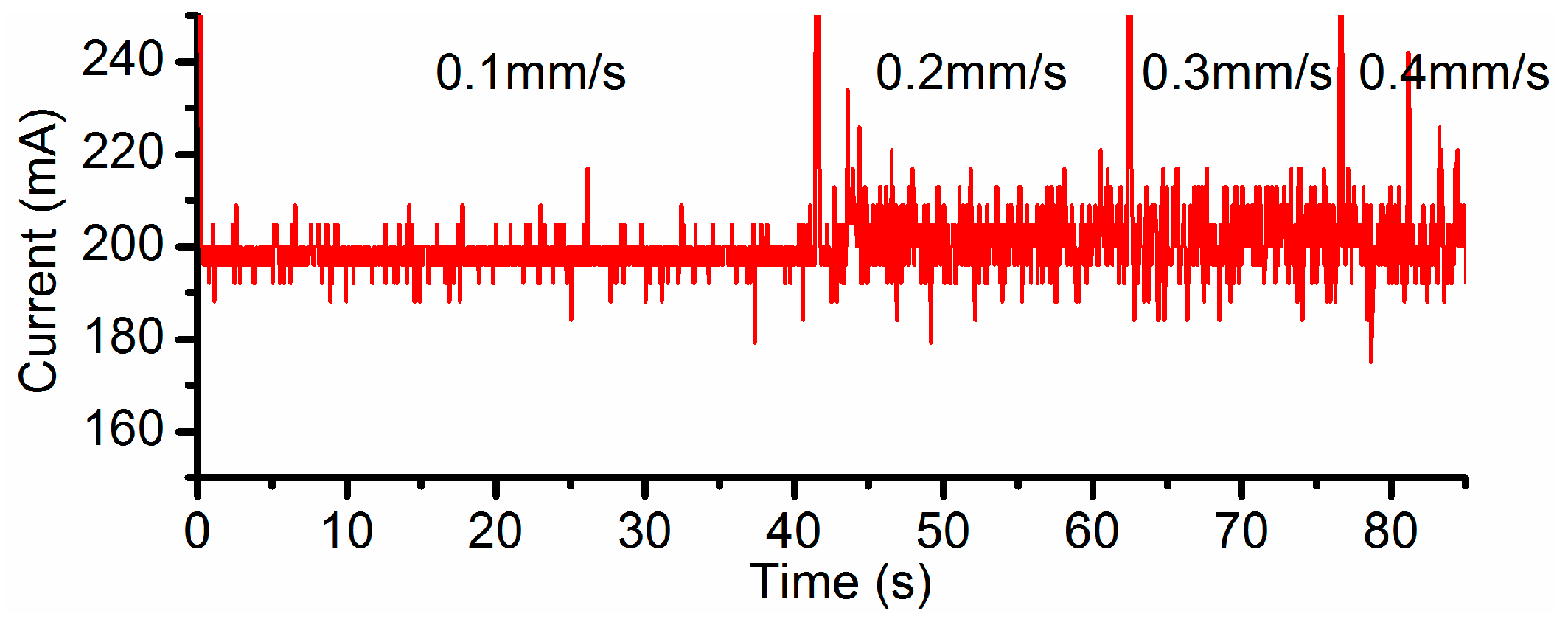
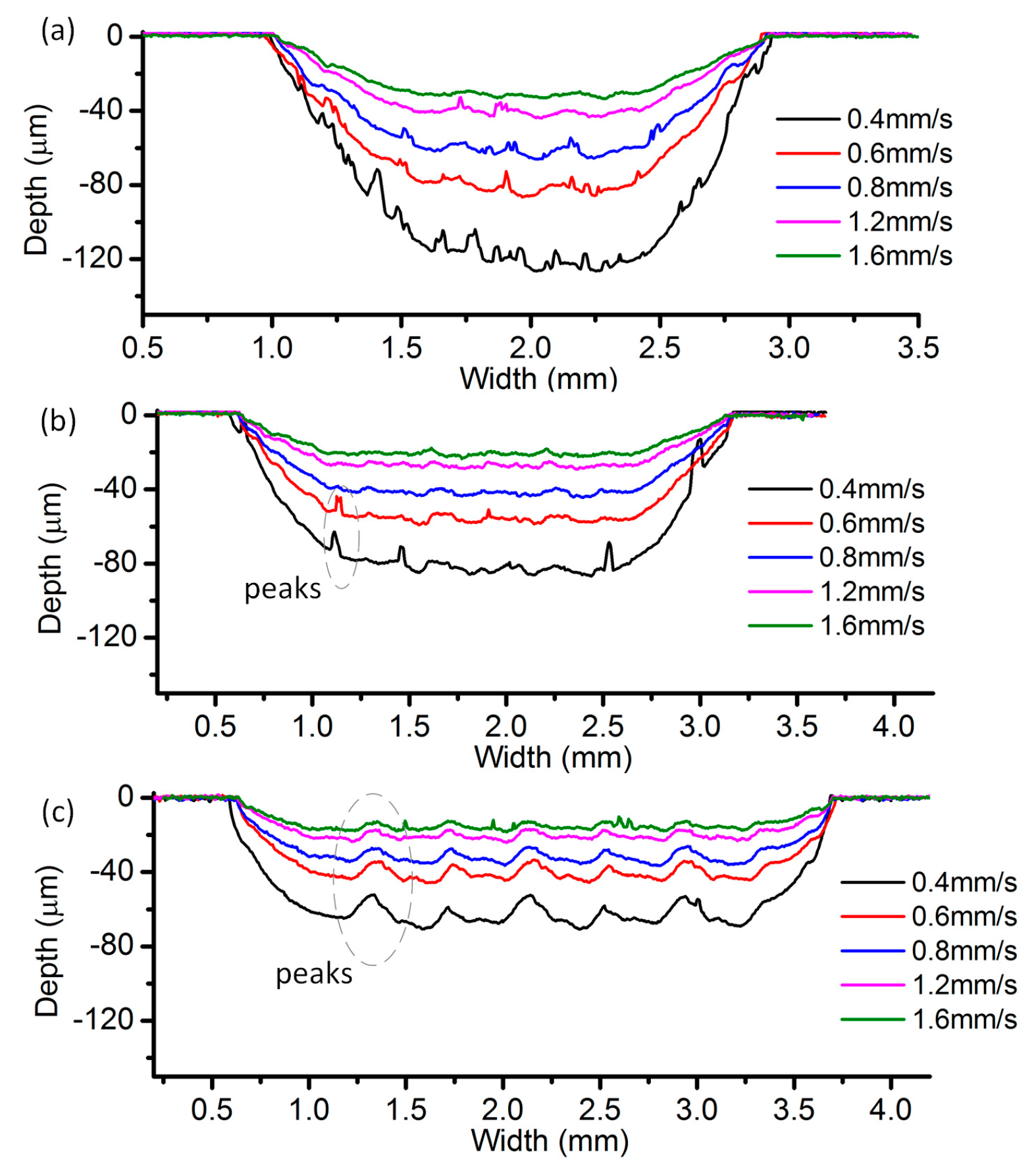
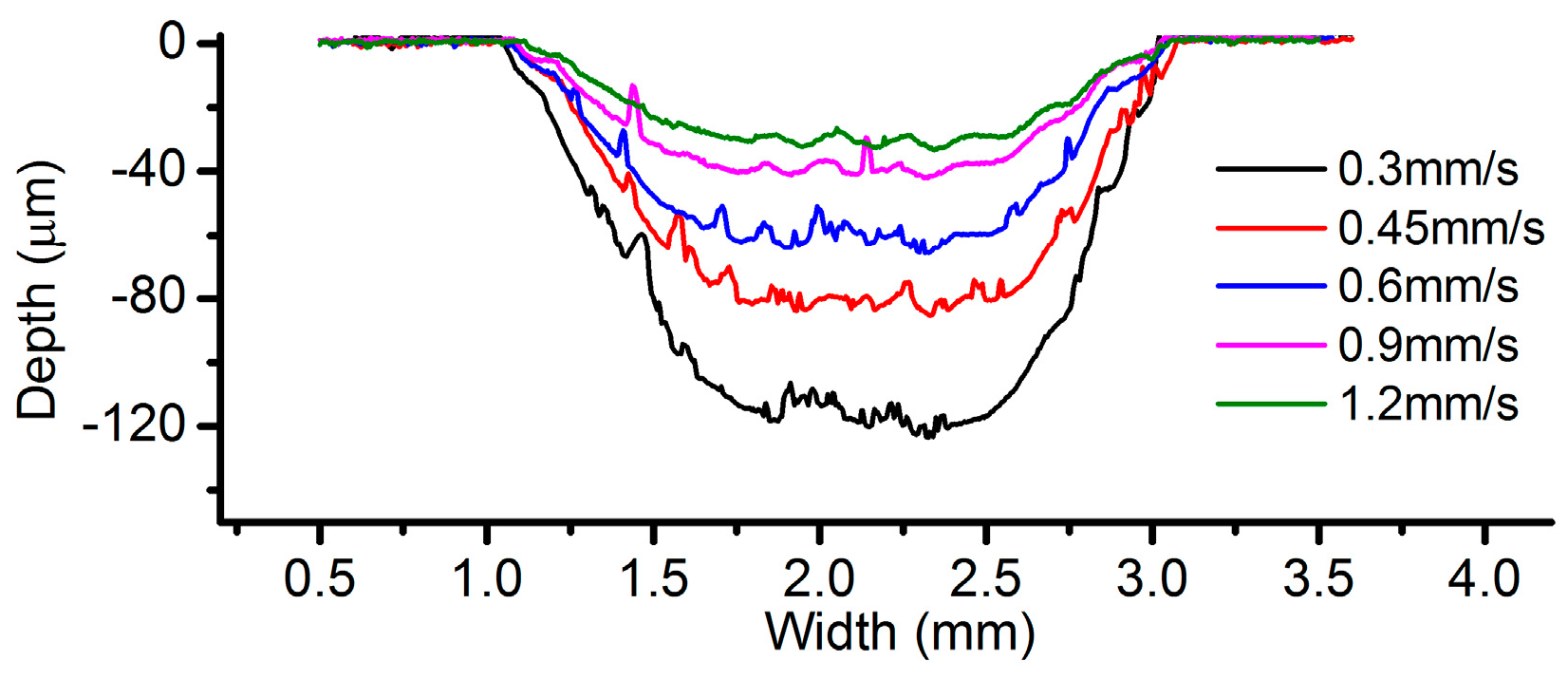
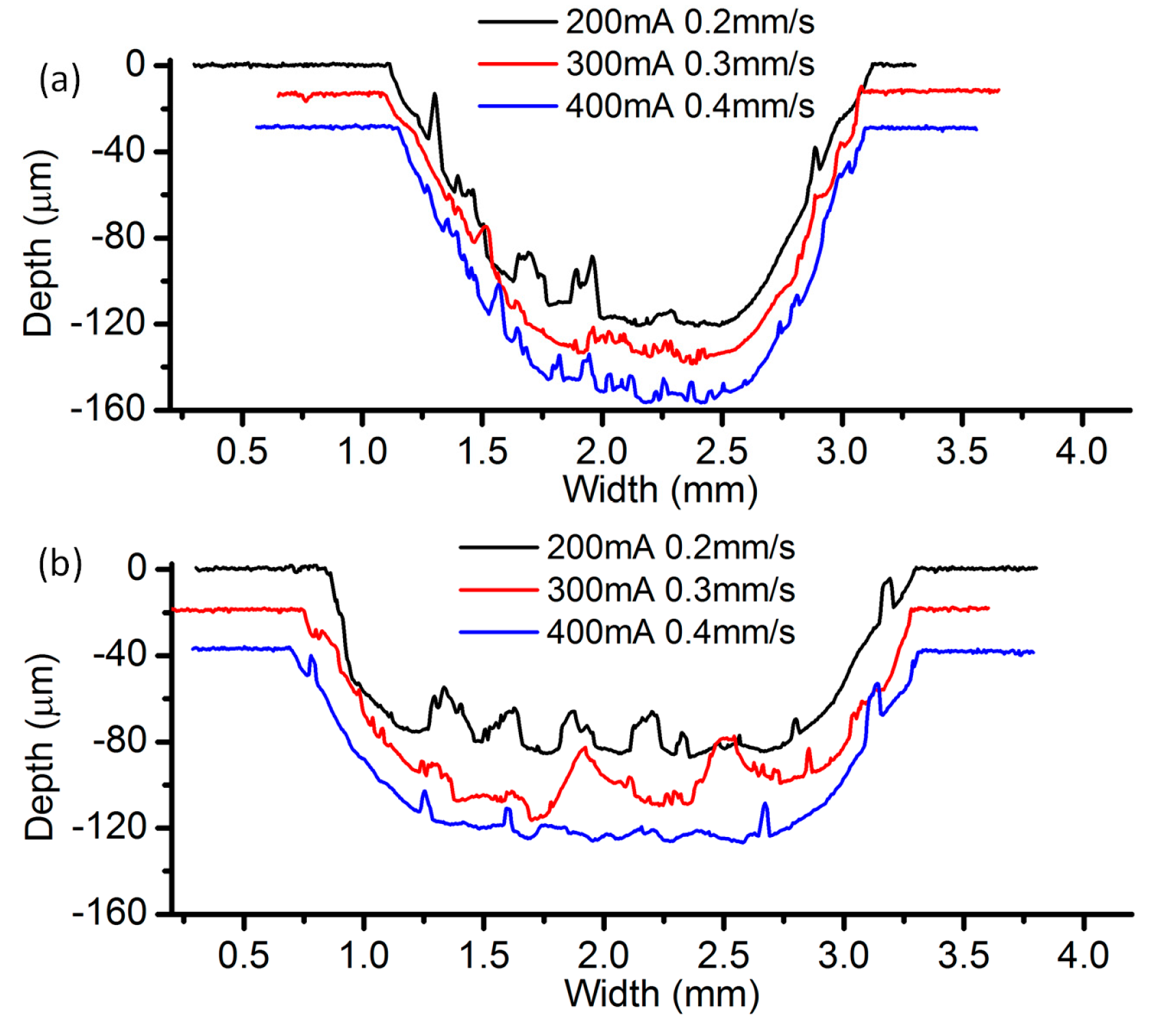
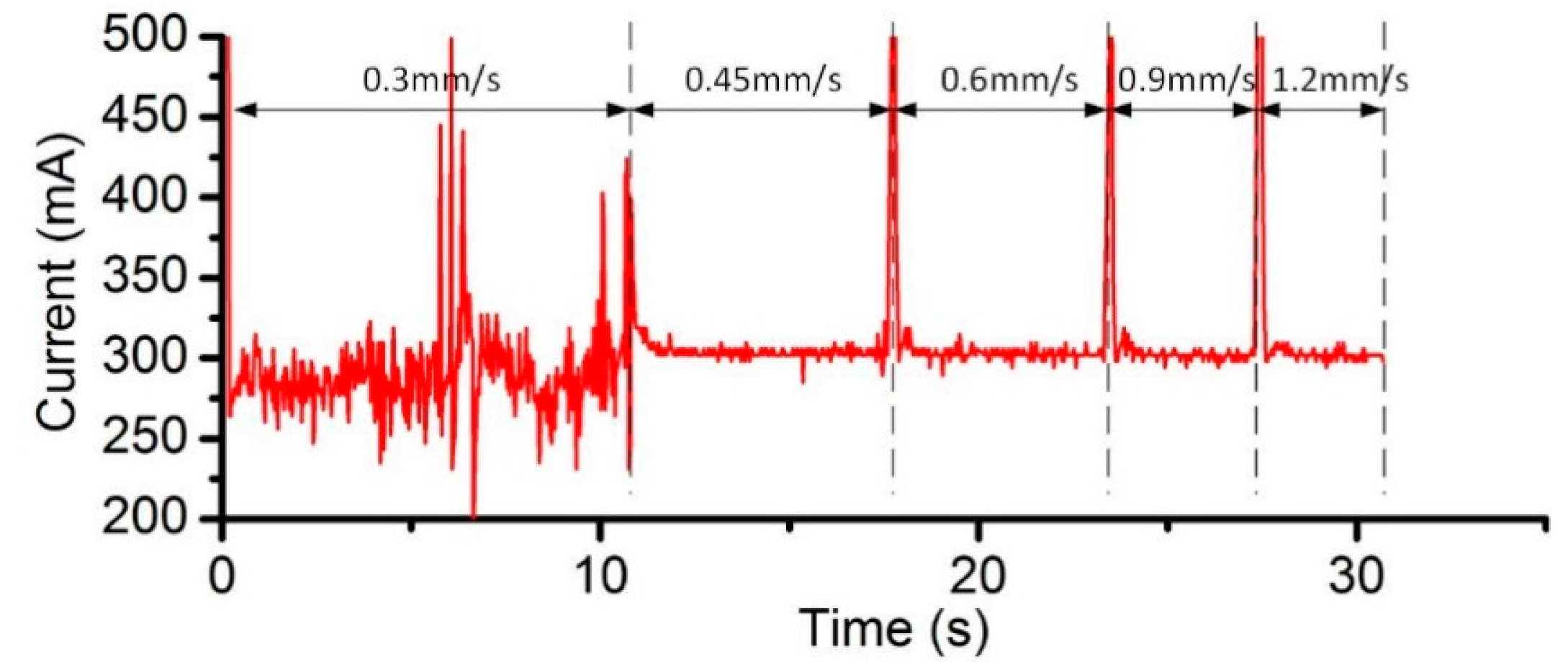
3.2. Effects of Current Density and Feed Rate
3.3. Effects of Vacuum Gap (VG)
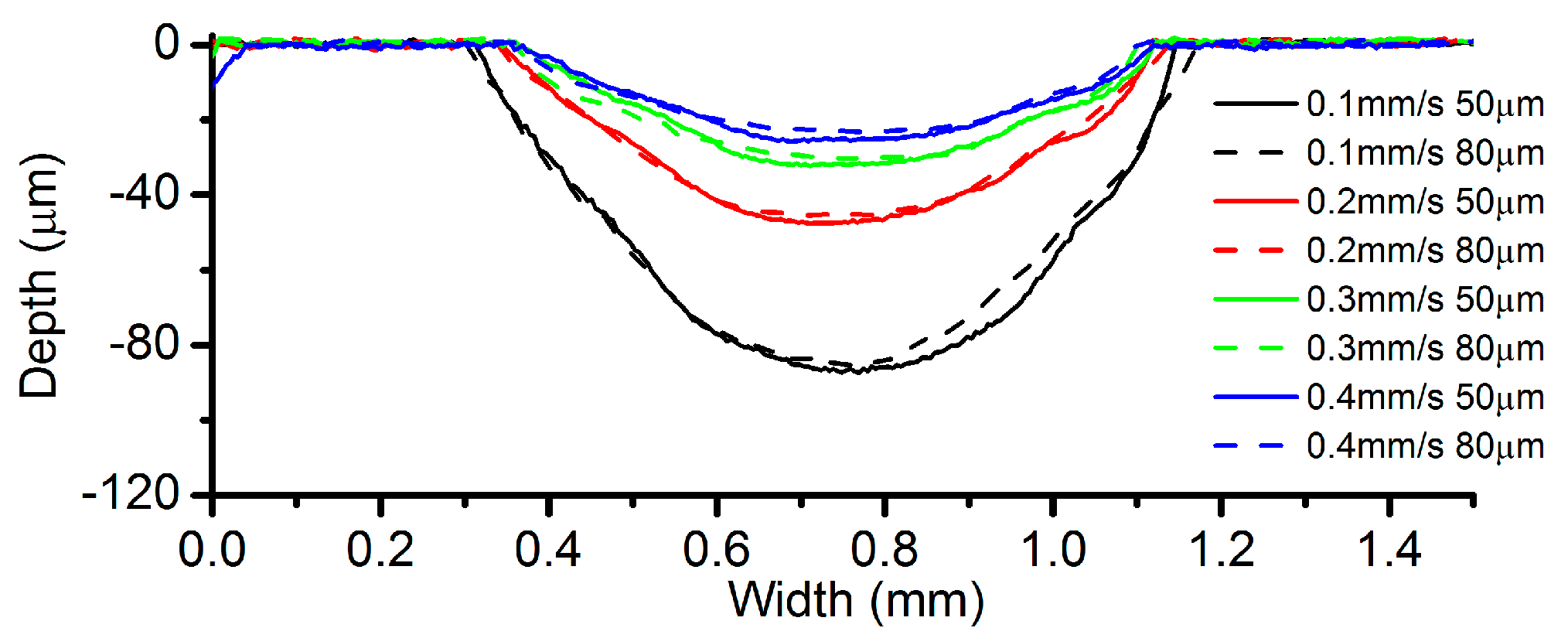
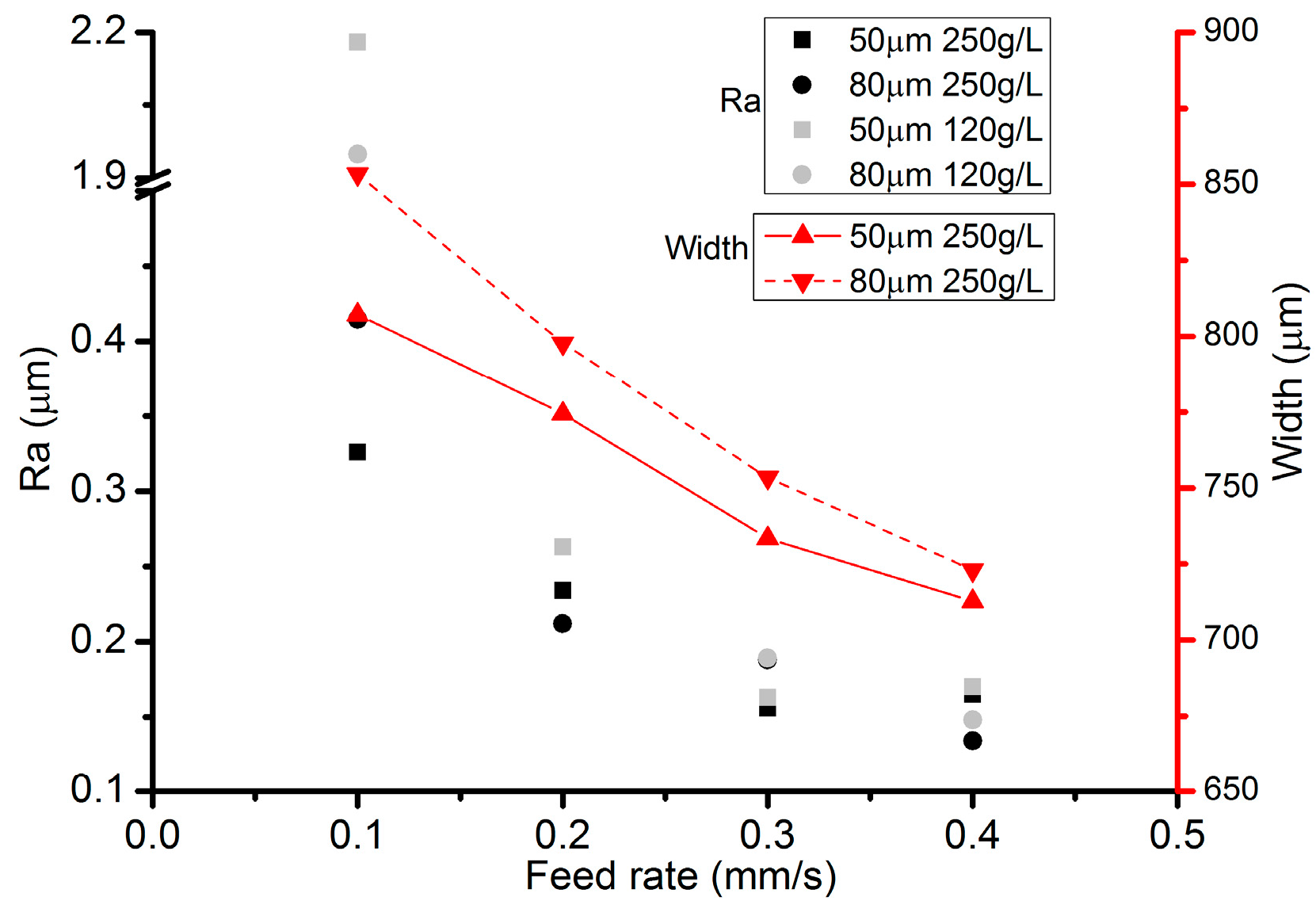
3.4. Effects of Inter-Electrode Gap (IEG)
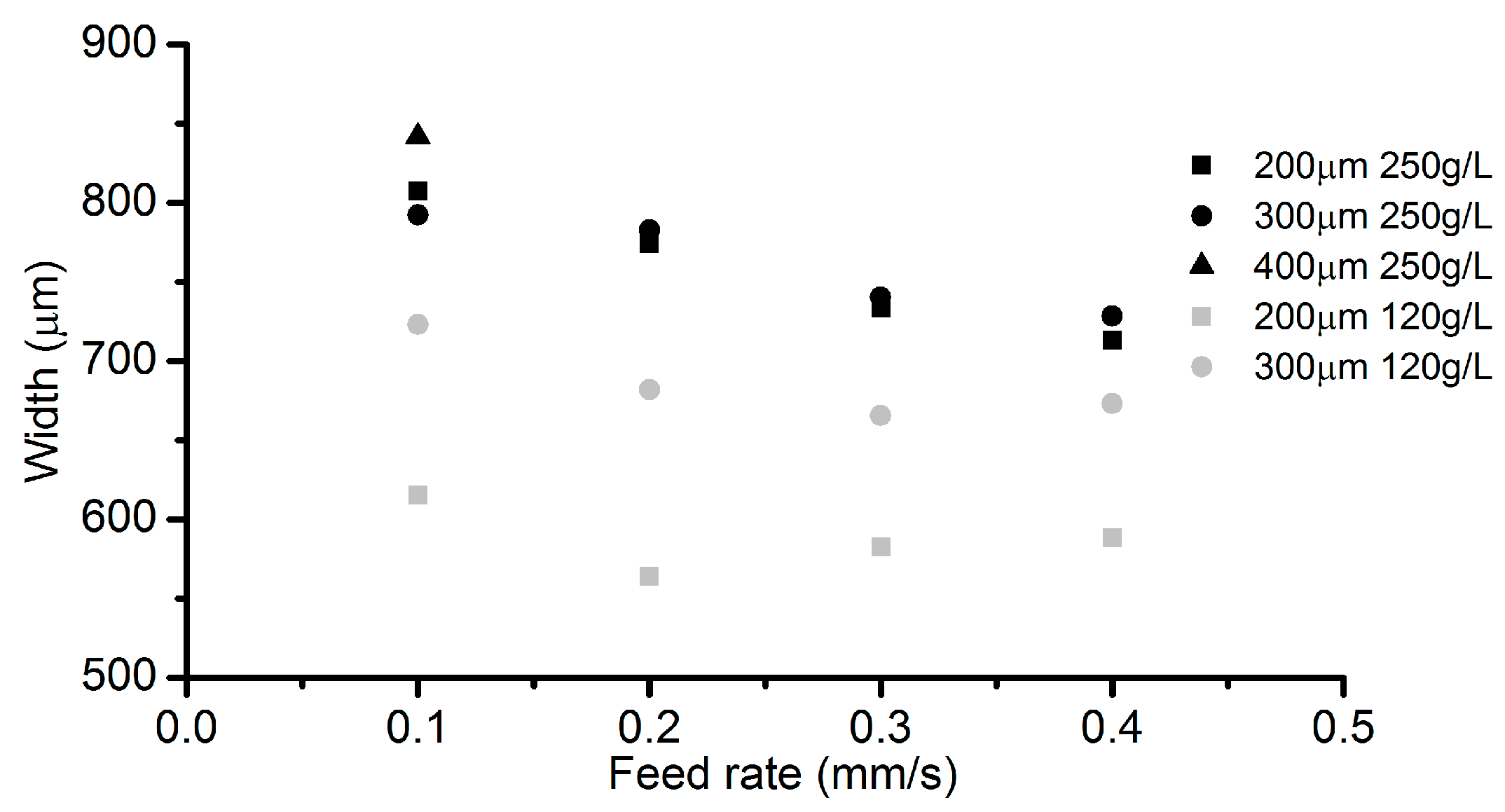
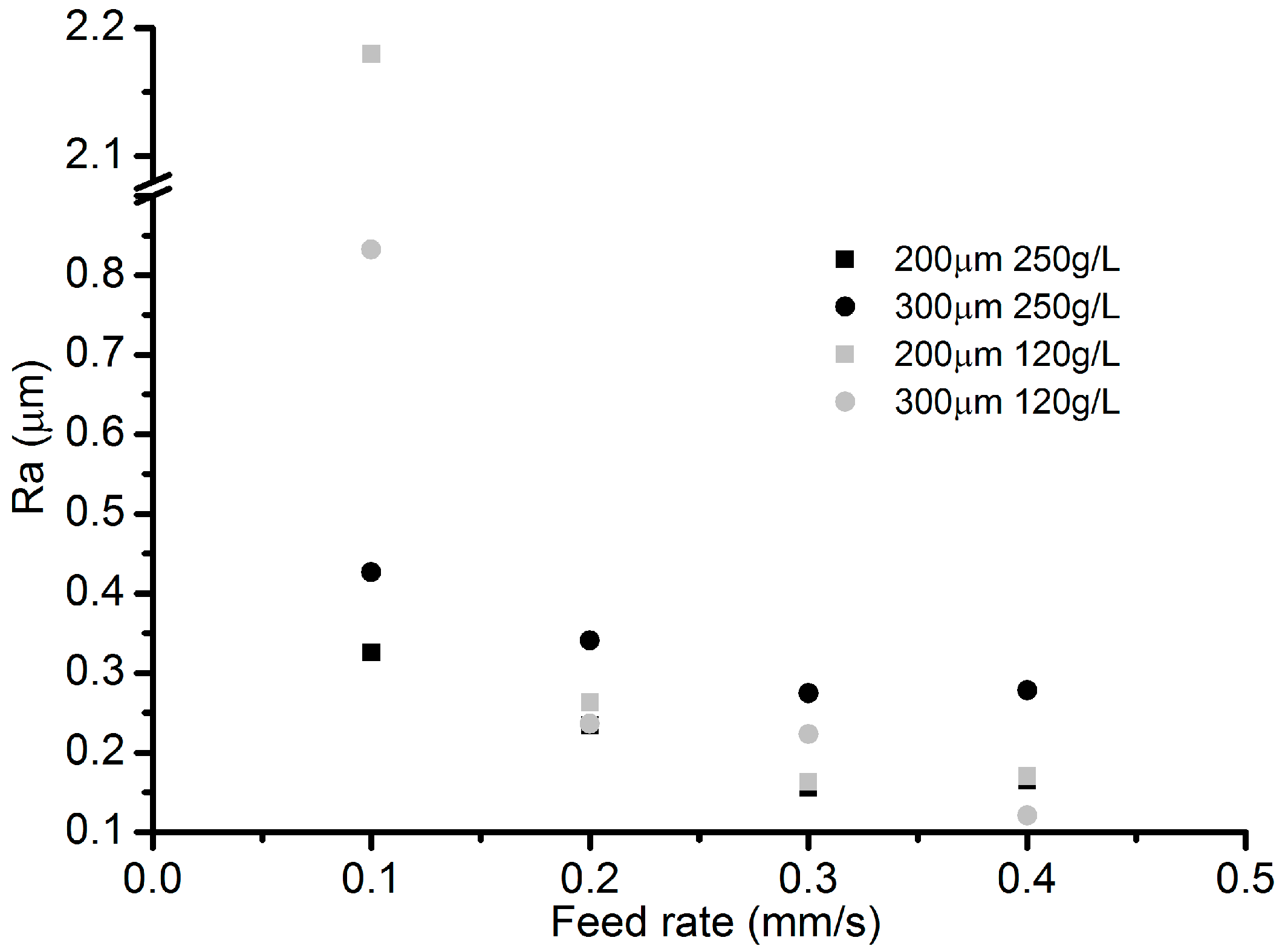
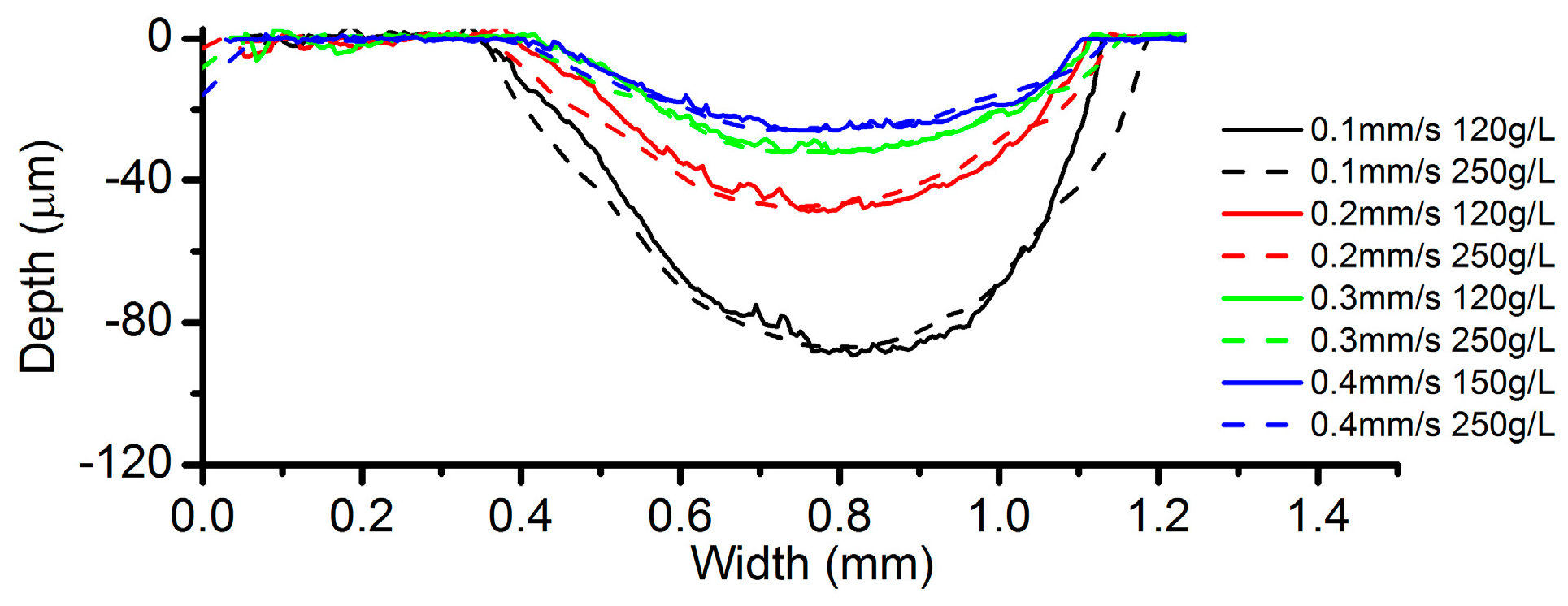
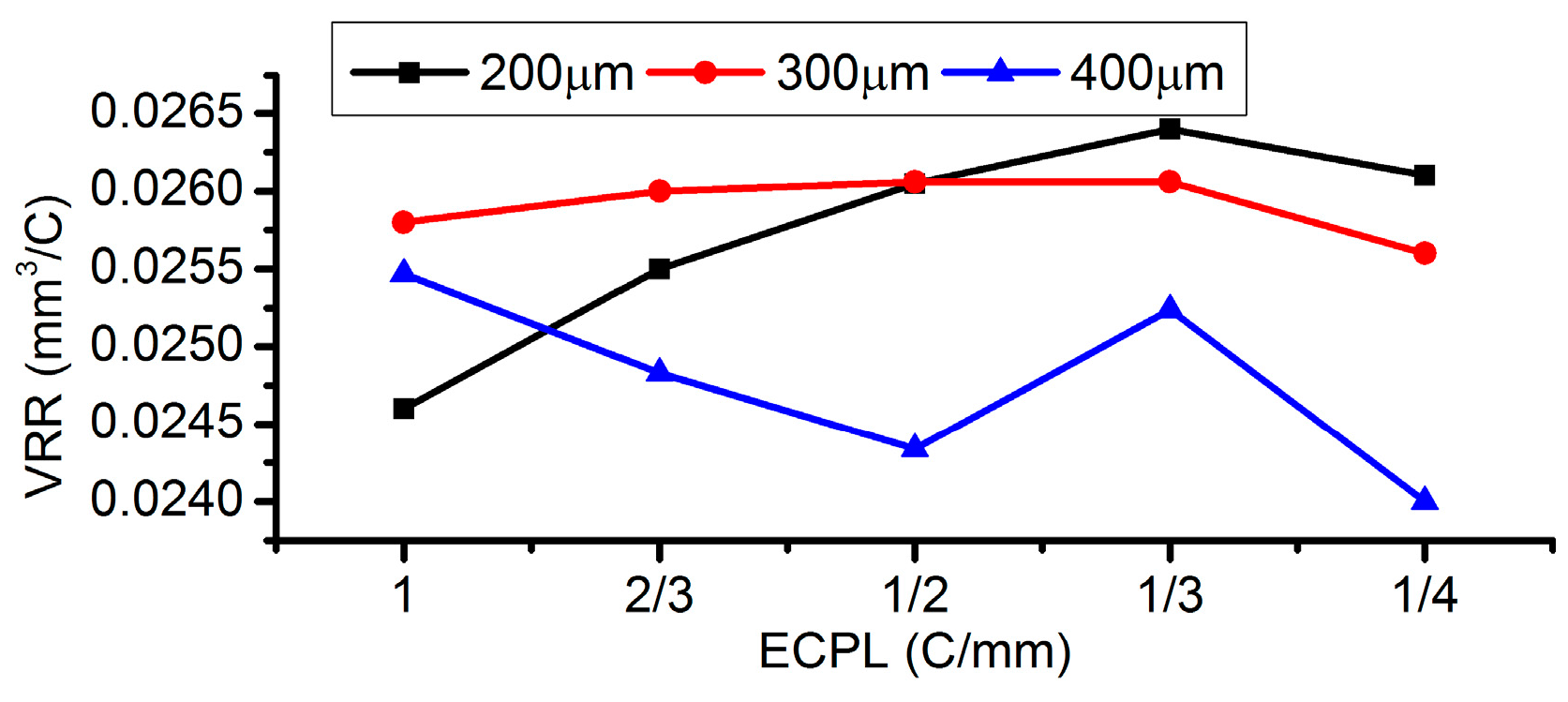
3.5. Effects of Electrolyte Concentration
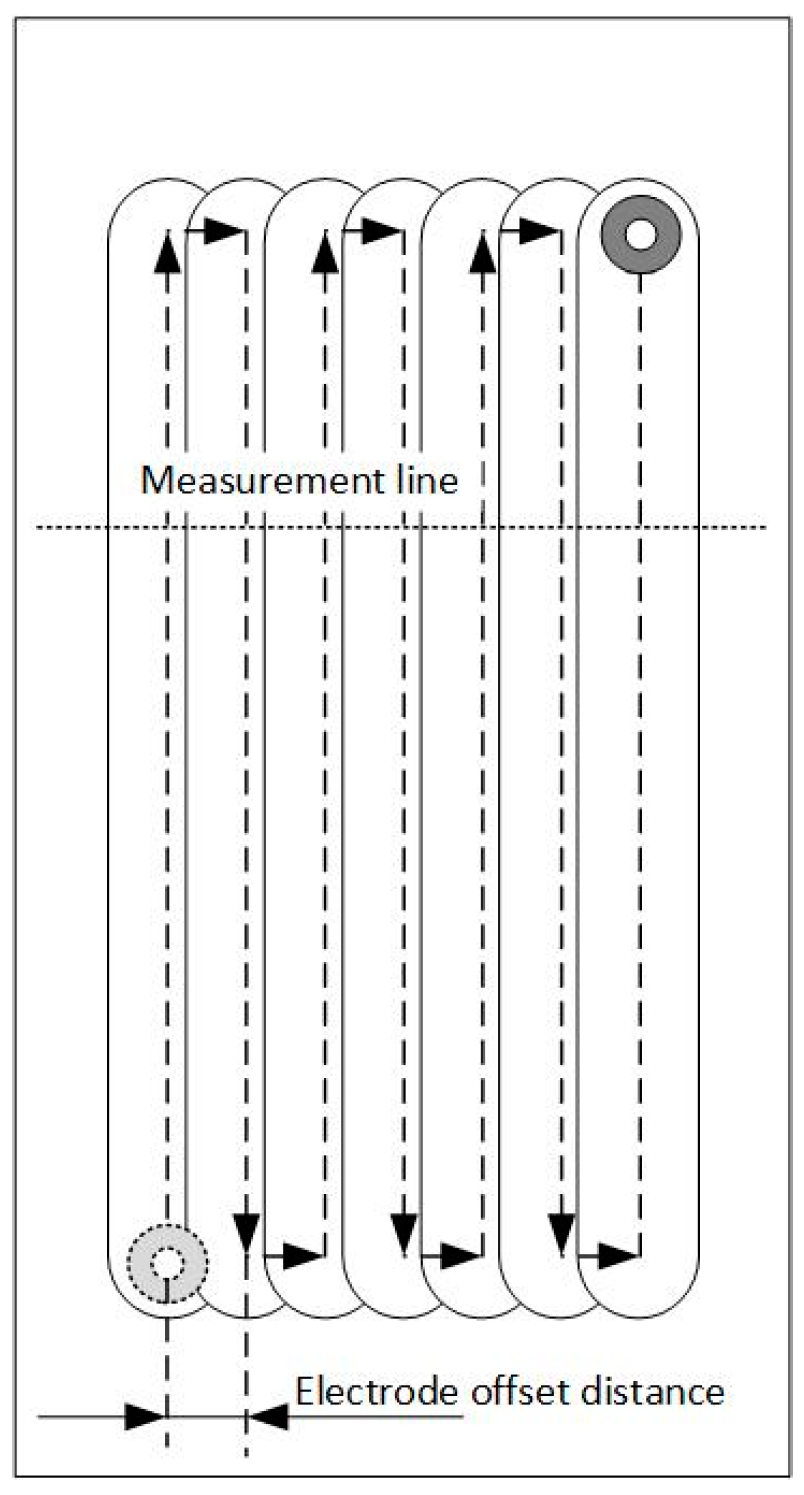
3.6. Multiple Processing
4. Conclusions
- An integrable scanning micro electrochemical flow cell (SMEFC) unit has been developed and utilized to fabricate mesoscale channels. The SMEFC can confine the electrolyte droplet just in the area of around 0.5 mm2 without leakage to the other non-processing region.
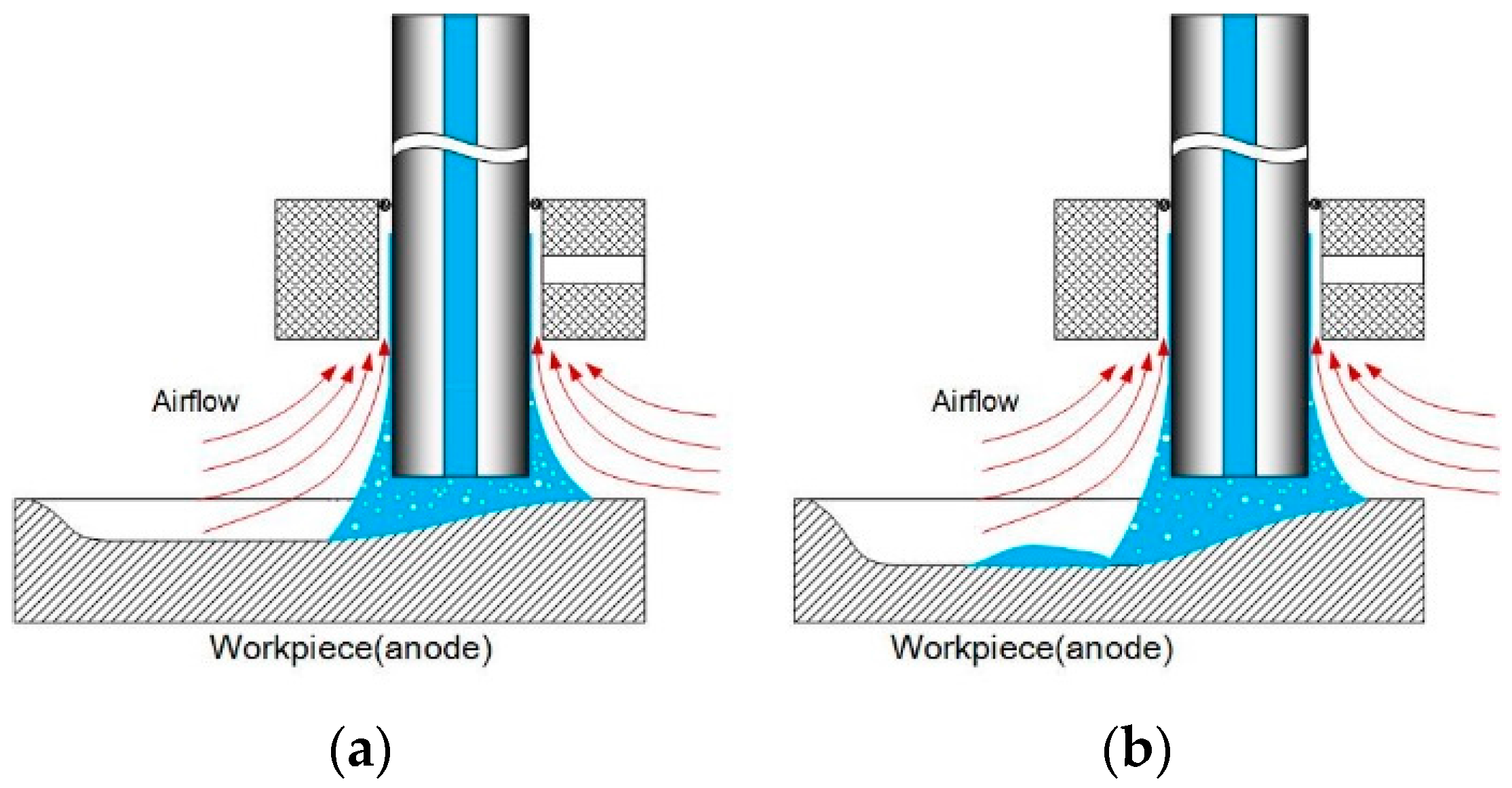
- The roughness of the original workpiece surface affects the shape of the moving electrolyte droplet. Smooth surfaces tend to induce longer trials of the electrolyte droplet above the top of the workpiece surface.
- Among all the geometrical parameters of the SMEFC configuration, the vacuum gap significantly affects the shape of the moving electrolyte droplet. A smaller vacuum gap tends to provide a better control of the electrolyte droplet, which contributes to the consistency of the surface microstructures. At the same level of the electric consumption per unit length, the channel depth maintain the same on the whole, but suitable combinations of the current densities and feed rates can generate better surface quality and roughness. The concentration of the electrolyte influences the formation of the supersaturated layer and further affects the roughness. A higher current density, a smaller inter-electrode gap and a higher electrolyte concentration improve the current efficiency.
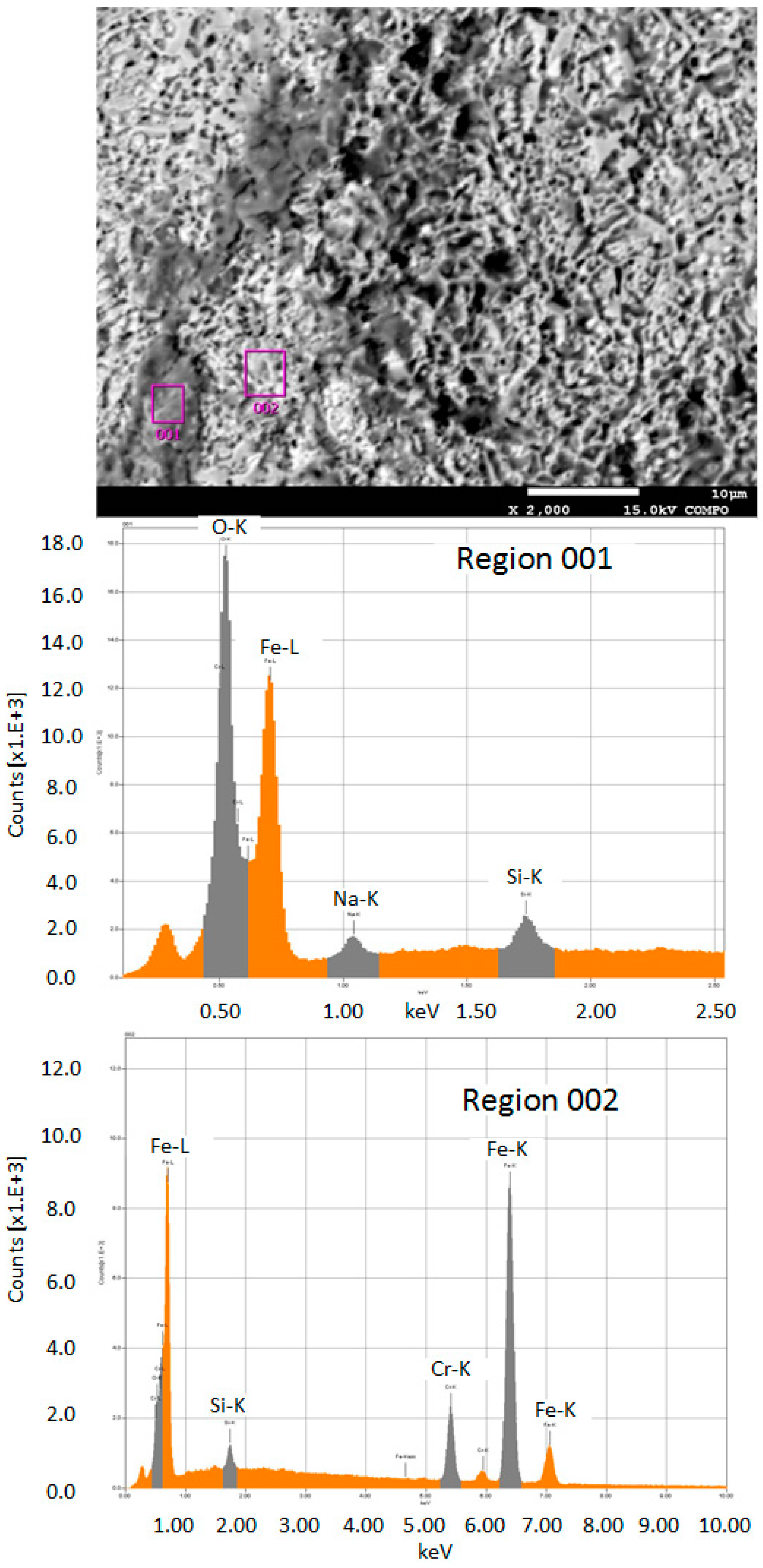
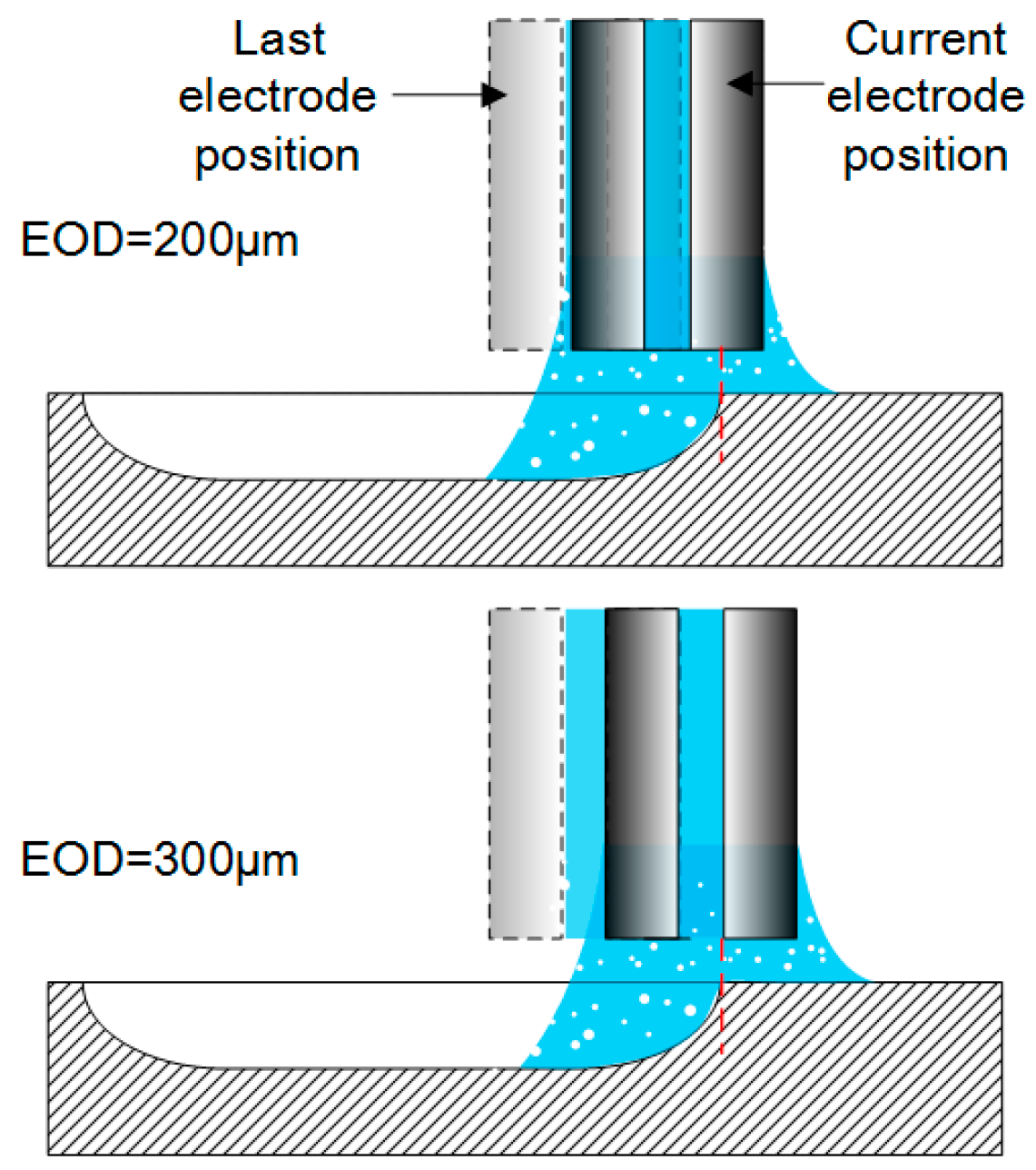
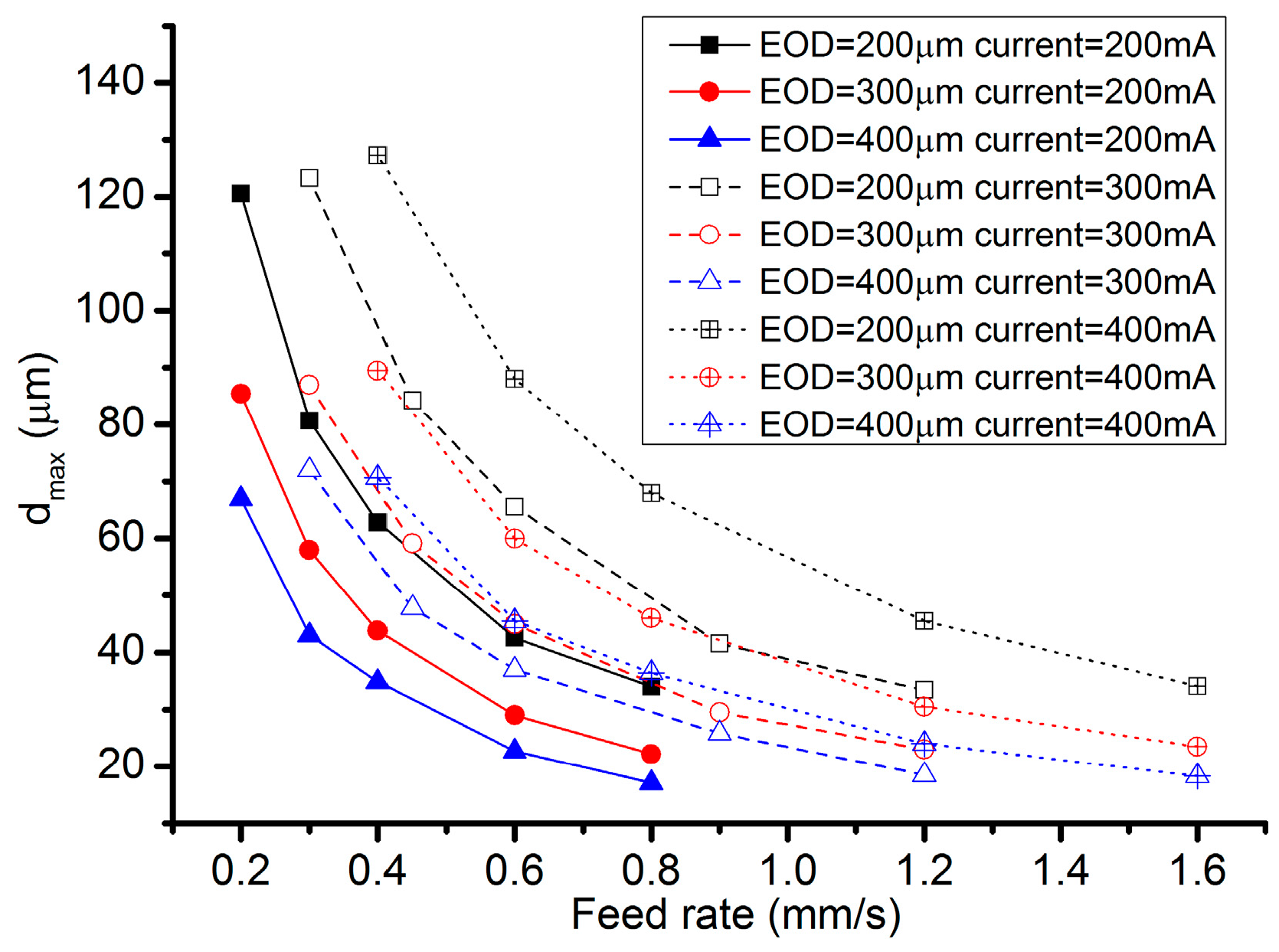
- As for the electrochemical milling by the SMEFC, the cavities machined with different electrode offset distances and different electric consumption per unit length have been compared. The electrode offset distance plays a significant role on the milling performance. Through the comparison of the cross-sectional profiles and SEM pictures, the electrode offset distance of 300 µm together with the current of 400 mA is a better combination for obtaining a relatively smooth bottom surface and a stable material removal rate. The removal depth can be adjusted by tuning the feed rate. A larger electrode offset distance and a higher current contribute to decrease of the residues of the electrolyte crystallization.
- In future research, a linear power supply can replace the switching power supply for a better current holding capability. The electrode with the outer diameter of 0.3 mm will also be utilized to obtain smaller features. Multiple vision units will be installed at different angles to monitor and analyze the moving electrolyte droplet.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lee, S.-J.; Lee, C.-Y.; Yang, K.-T.; Kuan, F.-H.; Lai, P.-H. Simulation and fabrication of micro-scaled flow channels for metallic bipolar plates by the electrochemical micro-machining process. J. Power Sources 2008, 185, 1115–1121. [Google Scholar] [CrossRef]
- Hung, J.-C.; Chang, C.-H.; Chiu, K.-C.; Lee, S.-J. Simulation-based fabrication of micro-helical grooves in a hydrodynamic thrust bearing by using ecmm. Int. J. Adv. Manuf. Technol. 2013, 64, 813–820. [Google Scholar] [CrossRef]
- Liu, G.X.; Zhang, Y.J.; Jiang, S.Z.; Liu, J.W.; Gyimah, G.K.; Luo, H.P. Investigation of pulse electrochemical sawing machining of micro-inner annular groove on metallic tube. Int. J. Mach. Tools Manuf. 2016, 102, 22–34. [Google Scholar] [CrossRef]
- Ryu, S.H. Eco-friendly ecm in citric acid electrolyte with microwire and microfoil electrodes. Int. J. Precis. Eng. Manuf. 2015, 16, 233–239. [Google Scholar] [CrossRef]
- Jia, L.; Xiaochen, J.; Di, Z. Electrochemical machining of multiple slots with low-frequency tool vibrations. Procedia CIRP 2016, 42, 799–803. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, Y.; Deng, Y.; Wei, H.; Zhou, C.; Liu, J.; Luo, H. The tool design and experiments on pulse electrochemical machining of micro channel arrays on metallic bipolar plate using multifunctional cathode. Int. J. Adv. Manuf. Technol. 2017, 89, 407–416. [Google Scholar] [CrossRef]
- Natsu, W.; Ikeda, T.; Kunieda, M. Generating complicated surface with electrolyte jet machining. Precis. Eng. 2007, 31, 33–39. [Google Scholar] [CrossRef]
- Hackert, M.; Meichsner, G.; Schubert, A. Generating micro geometries with air assisted jet electrochemical machining. In Proceedings of the Euspen 10th Anniversary International Conference, Zurich, Switzerland, 18–22 May 2008; pp. 420–424. [Google Scholar]
- Kai, S.; Sai, H.; Kunieda, M.; Izumi, H. Study on electrolyte jet cutting. Procedia CIRP 2012, 1, 627–632. [Google Scholar] [CrossRef]
- Kunieda, M.; Mizugai, K.; Watanabe, S.; Shibuya, N.; Iwamoto, N. Electrochemical micromachining using flat electrolyte jet. CIRP Ann. Manuf. Technol. 2011, 60, 251–254. [Google Scholar] [CrossRef]
- Hackert-Oschätzchen, M.; Meichsner, G.; Zinecker, M.; Martin, A.; Schubert, A. Micro machining with continuous electrolytic free jet. Precis. Eng. 2012, 36, 612–619. [Google Scholar] [CrossRef]
- Ghoshal, B.; Bhattacharyya, B. Investigation on profile of microchannel generated by electrochemical micromachining. J. Mater. Process. Technol. 2015, 222, 410–421. [Google Scholar] [CrossRef]
- Ghoshal, B.; Bhattacharyya, B. Electrochemical micromachining of microchannel using optimum scan feed rate. J. Manuf. Process. 2016, 23, 258–268. [Google Scholar] [CrossRef]
- Kim, B.H.; Ryu, S.H.; Choi, D.K.; Chu, C.N. Micro electrochemical milling. J. Micromech. Microeng. 2005, 15, 124. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, D.; Qu, N.; Wang, M. Theoretical and experimental investigation on electrochemical micromachining. Microsyst. Technol. 2007, 13, 607–612. [Google Scholar] [CrossRef]
- Shin, H.S.; Kim, B.H.; Chu, C.N. Analysis of the side gap resulting from micro electrochemical machining with a tungsten wire and ultrashort voltage pulses. J. Micromech. Microeng. 2008, 18, 075009. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, D.; Zeng, Y.; Liu, Y. Micro wire electrode electrochemical cutting with low frequency and small amplitude tool vibration. Int. J. Adv. Manuf. Technol. 2011, 53, 535–544. [Google Scholar] [CrossRef]
- Liu, Z.; Nouraei, H.; Spelt, J.K.; Papini, M. Electrochemical slurry jet micro-machining of tungsten carbide with a sodium chloride solution. Precis. Eng. 2015, 40, 189–198. [Google Scholar] [CrossRef]
- Yuan, L.; Xu, J.; Zhao, J.; Zhang, H. Research on hybrid process of laser drilling with jet electrochemical machining. J. Manuf. Sci. Eng. 2012, 134, 064502. [Google Scholar] [CrossRef]
- Zhang, Z.; Cai, M.; Feng, Q.; Zeng, Y. Comparison of different laser-assisted electrochemical methods based on surface morphology characteristics. Int. J. Adv. Manuf. Technol. 2014, 71, 565–571. [Google Scholar] [CrossRef]
- Sakairi, M.; Sato, F.; Gotou, Y.; Fushimi, K.; Kikuchi, T.; Takahashi, H. Development of a novel microstructure fabrication method with co-axial dual capillary solution flow type droplet cells and electrochemical deposition. Electrochim. Acta 2008, 54, 616–622. [Google Scholar] [CrossRef]
- Drensler, S.; Milenkovic, S.; Hassel, A.W. Microvials with tungsten nanowire arrays. J. Solid State Electrochem. 2014, 18, 2955–2961. [Google Scholar] [CrossRef]
- Hu, J.-F.; Kuo, C.-L. Study on Micro Electrochemical Machining Using Coaxial for Gushing and Sucking Method. Master’s Thesis, National Yunlin University of Science & Technology, Yunlin County, Taiwan, 2007. [Google Scholar]
- Kuo, K.-Y.; Wu, K.-L.; Yang, C.-K.; Yan, B.-H. Wire electrochemical discharge machining (WECDM) of quartz glass with titrated electrolyte flow. Int. J. Mach. Tools Manuf. 2013, 72, 50–57. [Google Scholar] [CrossRef]
- Leroy, P.; Lassin, A.; Azaroual, M.; André, L. Predicting the surface tension of aqueous 1:1 electrolyte solutions at high salinity. Geochim. Cosmochim. Acta 2010, 74, 5427–5442. [Google Scholar] [CrossRef]
- Deconinck, D.; Hoogsteen, W.; Deconinck, J. A temperature dependent multi-ion model for time accurate numerical simulation of the electrochemical machining process. Part III: Experimental validation. Electrochim. Acta 2013, 103, 161–173. [Google Scholar] [CrossRef]
- Rosenkranz, C.; Lohrengel, M.; Schultze, J. The surface structure during pulsed ecm of iron in NaNO3. Electrochim. Acta 2005, 50, 2009–2016. [Google Scholar] [CrossRef]
- Schubert, A.; Hackert-Oschätzchen, M.; Martin, A.; Winkler, S.; Kuhn, D.; Meichsner, G.; Zeidler, H.; Edelmann, J. Generation of complex surfaces by superimposed multi-dimensional motion in electrochemical machining. Procedia CIRP 2016, 42, 384–389. [Google Scholar] [CrossRef]
- Martin, A.; Eckart, C.; Lehnert, N.; Hackert-Oschätzchen, M.; Schubert, A. Generation of defined surface waviness on tungsten carbide by jet electrochemical machining with pulsed current. Procedia CIRP 2016, 45, 231–234. [Google Scholar] [CrossRef]









| Current (mA) | Feed Rate (mm/s) | ECPL (C/mm) | Sexp (mm2) | Sthe (mm2) | η1 | η2 |
|---|---|---|---|---|---|---|
| 100 | 0.1 | 1 | 0.0234 | 0.02327 | 100.6% | 82.6% |
| 200 | 0.2 | 1 | 0.0246 | 0.02327 | 105.7% | 86.8% |
| 300 | 0.3 | 1 | 0.0255 | 0.02327 | 109.6% | 90.0% |
| 100 | 0.2 | 0.5 | 0.0110 | 0.01165 | 94.4% | 77.5% |
| 200 | 0.4 | 0.5 | 0.0127 | 0.01165 | 109.0% | 89.5% |
| VG (µm) | Feed Rate (mm/s) | ECPL (C/mm) | Sexp (mm2) | Sthe (mm2) | η1 | η2 |
|---|---|---|---|---|---|---|
| 200 | 0.1 | 2 | 0.0498 | 0.0466 | 106.9% | 87.8% |
| 200 | 0.2 | 1 | 0.0246 | 0.0233 | 105.6% | 86.7% |
| 200 | 0.3 | 0.667 | 0.0164 | 0.0155 | 105.8% | 86.9% |
| 200 | 0.4 | 0.5 | 0.0127 | 0.01165 | 109.0% | 89.5% |
| 300 | 0.1 | 2 | 0.0499 | 0.0466 | 107.1% | 87.9% |
| 300 | 0.2 | 1 | 0.0246 | 0.0233 | 105.6% | 86.7% |
| 300 | 0.3 | 0.667 | 0.0169 | 0.0155 | 109.0% | 89.5% |
| 300 | 0.4 | 0.5 | 0.0122 | 0.01165 | 104.7% | 85.9% |
| 400 | 0.1 | 2 | 0.0499 | 0.0466 | 107.1% | 87.9% |
| IEG (µm) | Feed Rate (mm/s) | ECPL (C/mm) | Sexp (mm2) | Sthe (mm2) | η1 | η2 |
|---|---|---|---|---|---|---|
| 50 | 0.1 | 2 | 0.0498 | 0.0466 | 106.9% | 87.8% |
| 80 | 0.1 | 2 | 0.0477 | 0.0466 | 102.4% | 84.1% |
| 50 | 0.2 | 1 | 0.0246 | 0.0233 | 105.6% | 86.7% |
| 80 | 0.2 | 1 | 0.0245 | 0.0233 | 105.6% | 86.7% |
| 50 | 0.3 | 0.667 | 0.0164 | 0.0155 | 105.8% | 86.9% |
| 80 | 0.3 | 0.667 | 0.0161 | 0.0155 | 103.9% | 85.3% |
| 50 | 0.4 | 0.5 | 0.0127 | 0.01165 | 109.0% | 89.5% |
| 80 | 0.4 | 0.5 | 0.0120 | 0.01165 | 103.0% | 84.6% |
| Concentration (g/L) | Feed Rate (mm/s) | ECPL (C/mm) | Sexp (mm2) | Sthe (mm2) | η1 | η2 |
|---|---|---|---|---|---|---|
| 120 | 0.1 | 2 | 0.0473 | 0.0466 | 101.5% | 83.3% |
| 250 | 0.1 | 2 | 0.0498 | 0.0466 | 106.9% | 87.8% |
| 120 | 0.2 | 1 | 0.0242 | 0.0233 | 103.9% | 85.3% |
| 250 | 0.2 | 1 | 0.0246 | 0.0233 | 105.6% | 86.7% |
| 120 | 0.3 | 0.667 | 0.0152 | 0.0155 | 98.1% | 80.5% |
| 250 | 0.3 | 0.667 | 0.0164 | 0.0155 | 105.8% | 86.7% |
| 120 | 0.4 | 0.5 | 0.0116 | 0.01165 | 99.6% | 81.8% |
| 250 | 0.4 | 0.5 | 0.0127 | 0.01165 | 109.0% | 89.5% |
| ECPL (C/mm) | 1 | 2/3 | 1/2 | 1/3 | 1/4 | |
|---|---|---|---|---|---|---|
| Current (mA) | ||||||
| 200 | 0.2 | 0.3 | 0.4 | 0.6 | 0.8 | |
| 300 | 0.3 | 0.45 | 0.6 | 0.9 | 1.2 | |
| 400 | 0.4 | 0.6 | 0.8 | 1.2 | 1.6 | |
| EOD (µm) | 200 | 300 | 400 | |
|---|---|---|---|---|
| Current (mA) | ||||
| 200 | ||||
| 300 | ||||
| 400 | ||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, C.; Qian, J.; Reynaerts, D. Fabrication of Mesoscale Channel by Scanning Micro Electrochemical Flow Cell (SMEFC). Micromachines 2017, 8, 143. https://doi.org/10.3390/mi8050143
Guo C, Qian J, Reynaerts D. Fabrication of Mesoscale Channel by Scanning Micro Electrochemical Flow Cell (SMEFC). Micromachines. 2017; 8(5):143. https://doi.org/10.3390/mi8050143
Chicago/Turabian StyleGuo, Cheng, Jun Qian, and Dominiek Reynaerts. 2017. "Fabrication of Mesoscale Channel by Scanning Micro Electrochemical Flow Cell (SMEFC)" Micromachines 8, no. 5: 143. https://doi.org/10.3390/mi8050143
APA StyleGuo, C., Qian, J., & Reynaerts, D. (2017). Fabrication of Mesoscale Channel by Scanning Micro Electrochemical Flow Cell (SMEFC). Micromachines, 8(5), 143. https://doi.org/10.3390/mi8050143






