Fabrication of All Glass Bifurcation Microfluidic Chip for Blood Plasma Separation
Abstract
:1. Introduction
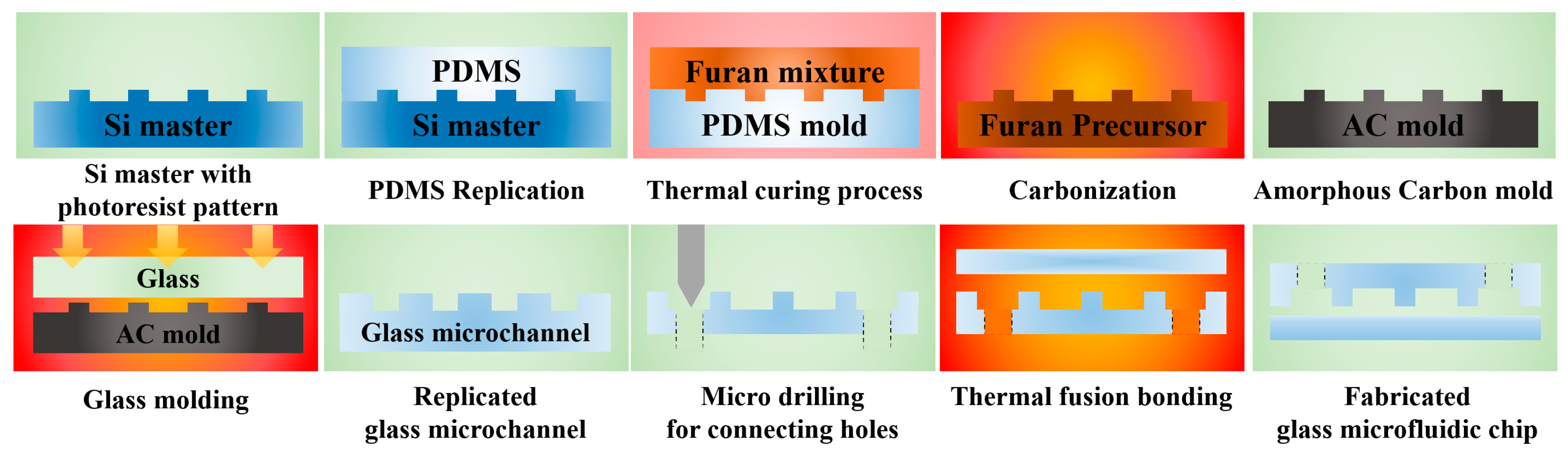
2. Fabrication of the All-Glass Microfluidic Chip Using a Glass Molded Microchannel
2.1. Design and Fabrication of the Silicon Master
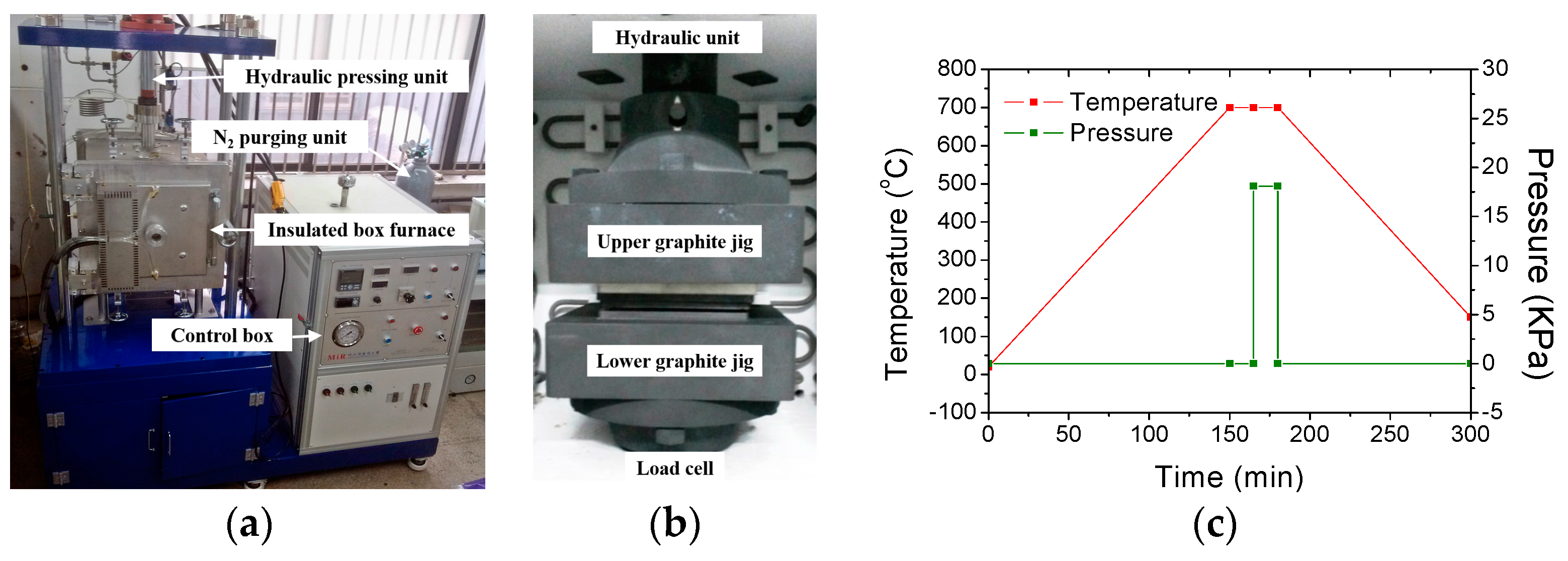
2.2. Fabrication of the AC Mold
2.3. Glass Molding of Microfluidic Channel Plate
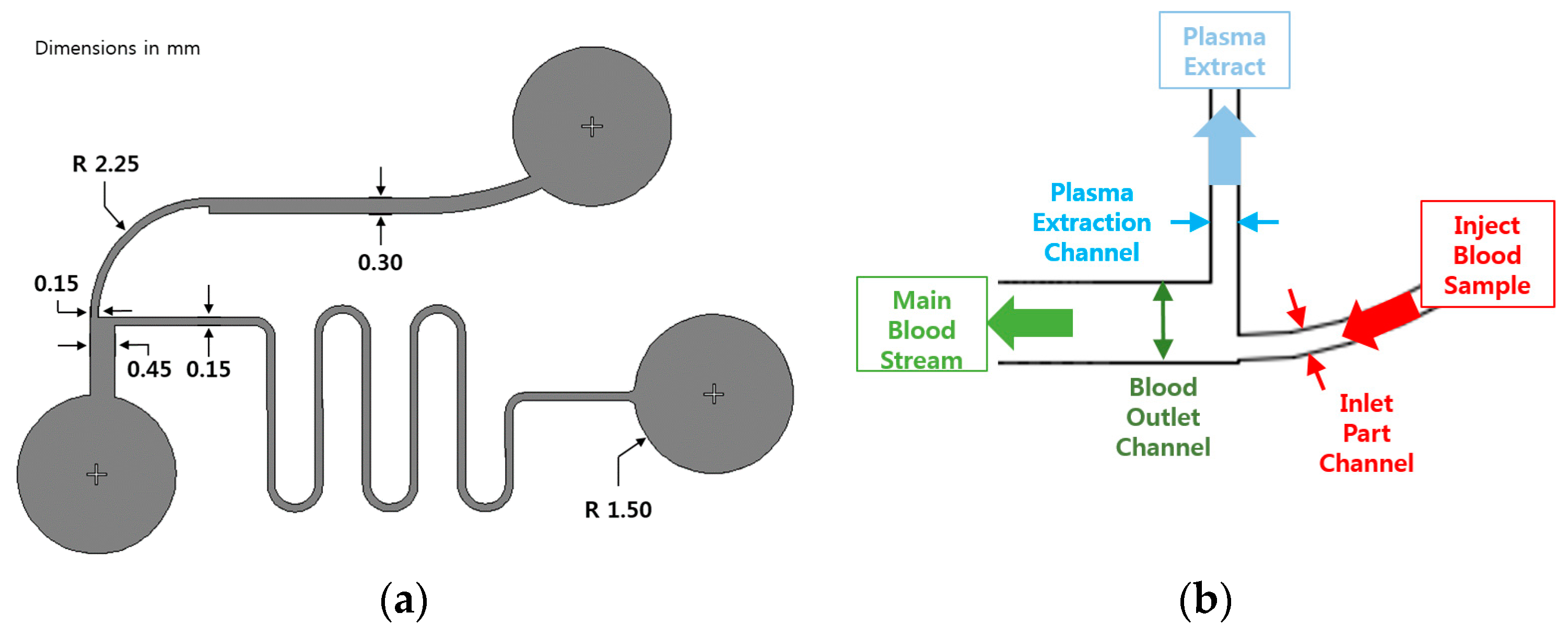
2.4. Preparation of the All-Glass Bifurcation Blood Plasma Separation Chip
3. Analysis of the Geometrical Properties in Each Fabrication Step
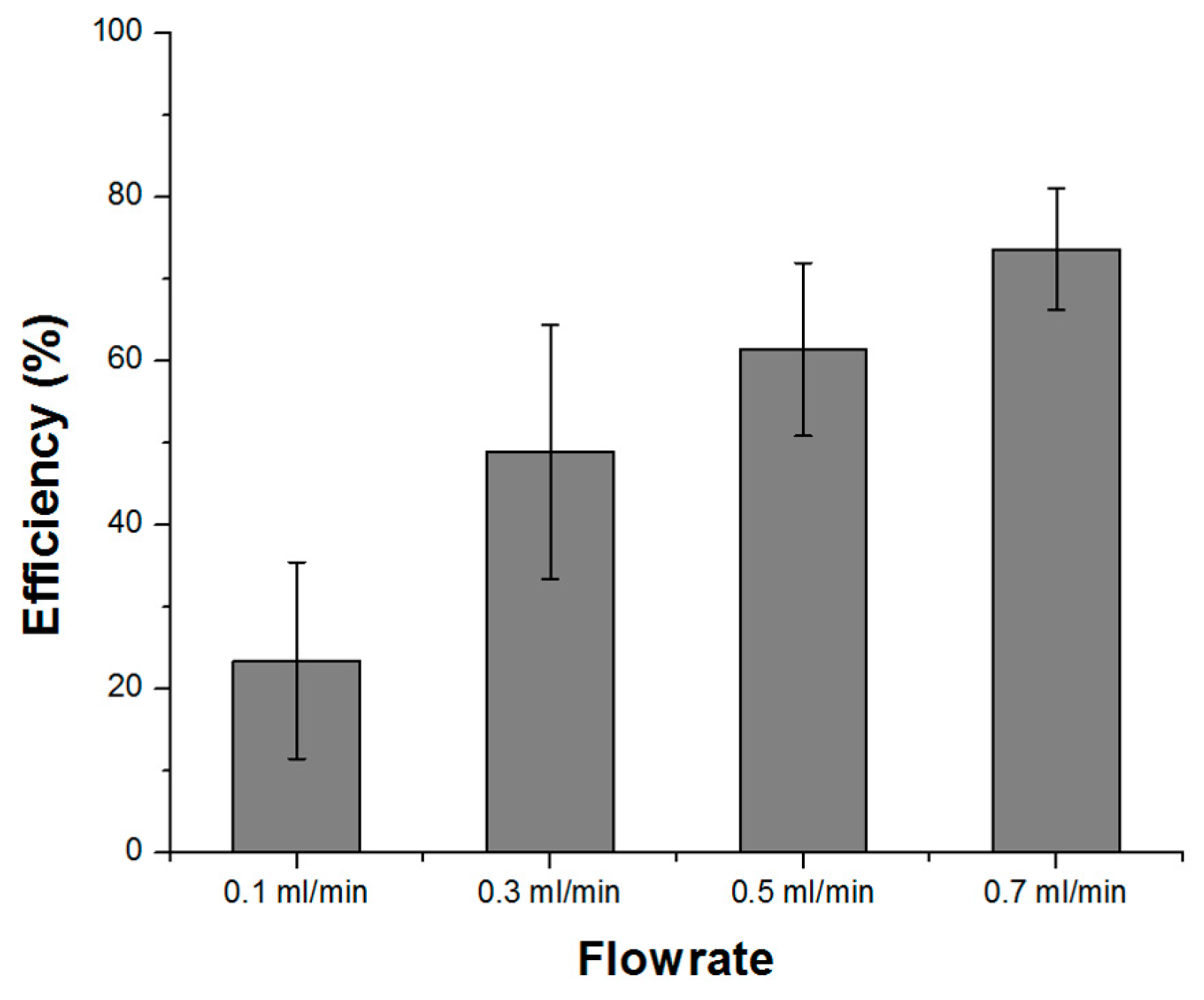
4. Feasibility Analysis of the All-Glass Bifurcation Chip for Blood Plasma Separation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chong, Z.Z.; Tor, S.B.; Loh, N.H.; Wong, T.N.; Gañán-Calvo, A.M.; Tan, S.H.; Nguyen, N.T. Acoustofluidic control of bubble size in microfluidic flow-focusing configuration. Lab Chip 2015, 15, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Nguyen, N.T.; Tan, S.H.; Asundi, A.K. A micro optofluidic lens with short focal length. J. Micromech. Microeng. 2009, 19, 085012. [Google Scholar] [CrossRef]
- Song, C.; Nguyen, N.T.; Tan, S.H.; Asundi, A.K. A tuneable micro-optofluidic biconvex lens with mathematically predictable focal length. Microfluid. Nanofluid. 2010, 9, 889–896. [Google Scholar] [CrossRef]
- Chong, Z.Z.; Tan, S.H.; Gañán-Calvo, A.M.; Tor, S.B.; Loh, N.H.; Nguyen, N.T. Active droplet generation in microfluidics. Lab Chip 2016, 16, 35–58. [Google Scholar] [CrossRef] [PubMed]
- Kersaudy-Kerhoas, M.; Sollier, E. Micro-scale blood plasma separation: From acoustophoresis to egg-beaters. Lab Chip 2013, 13, 3323–3346. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.J.; Wu, H.-Y.; George, S.; Weyhenmeyer, J.; Cunningham, B.T. Biochemical sensor tubing for point-of-care monitoring of intravenous drugs and metabolites. Lab Chip 2012, 12, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Gorkin, R.; Park, J.; Siegrist, J.; Amasia, M.; Lee, B.S.; Park, J.; Kim, J.; Kim, H.; Madou, M.; Cho, Y. Centrifugal microfluidics for biomedical applications. Lab Chip 2010, 10, 1758–1773. [Google Scholar] [CrossRef] [PubMed]
- Burger, R.; Reis, N.; Fonseca, J.G.D.; Ducree, J. Plasma extraction by centrifugo-pneumatically induced gating of flow. J. Micromech. Microeng. 2013, 23, 035035. [Google Scholar] [CrossRef]
- Petersson, F.; Åberg, L.; Swärd-Nilsson, A.-M.; Laurell, T. Free Flow Acoustophoresis: Microfluidic-Based Mode of Particle and Cell Separation. Anal. Chem. 2007, 79, 5117–5123. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, Y.; Hata, S.; Yasuda, T. Blood plasma separation and extraction from a minute amount of blood using dielectrophoretic and capillary forces. Sens. Actuators B 2010, 145, 561–569. [Google Scholar] [CrossRef]
- Jiang, H.; Weng, X.; Chon, C.H.; Wu, X.; Li, D. A microfluidic chip for blood plasma separation using electro-osmotic flow control. J. Micromech. Microeng. 2011, 21, 085019. [Google Scholar] [CrossRef]
- Furlani, E. Magnetophoretic separation of blood cells at the microscale. J. Phys. D Appl. Phys. 2007, 40, 1313. [Google Scholar] [CrossRef]
- Xiang, N.; Ni, Z. High-throughput blood cell focusing and plasma isolation using spiral inertial microfluidic devices. Biomed. Microdevices 2015, 17, 110. [Google Scholar] [CrossRef] [PubMed]
- Sollier, E.; Rostaing, H.; Pouteau, P.; Fouillet, Y.; Achard, J.-L. Passive microfluidic devices for plasma extraction from whole human blood. Sens. Actuators B 2009, 141, 617–624. [Google Scholar] [CrossRef]
- Tripathi, S.; Kumar, Y.B.V.; Prabhakar, A.; Joshi, S.S.; Agrawal, A. Performance study of microfluidic devices for blood plasma separation—A designer’s perspective. J. Micromech. Microeng. 2015, 25, 084004. [Google Scholar] [CrossRef]
- Maria, M.S.; Kumar, B.; Chandra, T.; Sen, A. Development of a microfluidic device for cell concentration and blood cell-plasma separation. Biomed. Microdevices 2015, 17, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Liu, C.; Xu, Z.; Li, J. Extraction of plasma from whole blood using a deposited microbead plug (DMBP) in a capillary-driven microfluidic device. Biomed. Microdevices 2012, 14, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, C.C.; Li, H. Microfluidic chip for blood cell separation and collection based on crossflow filtration. Sens. Actuators B 2008, 130, 216–221. [Google Scholar] [CrossRef]
- Kim, Y.C.; Kim, S.-H.; Kim, D.; Park, S.-J.; Park, J.-K. Plasma extraction in a capillary-driven microfluidic device using surfactant-added poly(dimethylsiloxane). Sens. Actuators B 2010, 145, 861–868. [Google Scholar] [CrossRef]
- Yang, S.; Undar, A.; Zahn, J.D. A microfluidic device for continuous, real time blood plasma separation. Lab Chip 2006, 6, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, A.; Kumar, Y.V.B.V.; Tripathi, S.; Agrawal, A. A novel, compact and efficient microchannel arrangement with multiple hydrodynamic effects for blood plasma separation. Microfluid. Nanofluid. 2015, 18, 995–1006. [Google Scholar] [CrossRef]
- Rodriguez-Villarreal, A.I.; Arundell, M.; Carmona, M.; Samitier, J. High flow rate microfluidic device for blood plasma separation using a range of temperatures. Lab Chip 2010, 10, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Feketea, Z.; Nagya, P.; Huszkac, B.; Tolnerb, F.; Pongrácza, A.; Fürjesa, P. Performance characterization of micromachined particle separation system based on Zweifach–Fung effect. Sens. Actuators B 2012, 162, 89–94. [Google Scholar] [CrossRef]
- Blattert, C.; Jurischka, R.; Tahhan, I.; Schoth, A.; Kerth, P.; Menz, W. Microfluidic blood/plasma separation unit based on microchannel bend structures. In Proceedings of the 3rd IEEE/EMBS Special Topic Conference on Microtechnology in Medicine and Biology, Oahu, HI, USA, 12–15 May 2005; IEEE: New York, NY, USA, 2005; pp. 38–41. [Google Scholar]
- Lopes, R.; Rodrigues, R.O.; Pinho, D.; Garcia, V.; Schütte, H.; Lima, R.; Gassmann, S. Low cost microfluidic device for partial cell separation: Micromilling approach, Industrial Technology (ICIT). In Proceedings of the 2015 IEEE International Conference on Intelligent Transportation Systems, Canary Islands, Spain, 15–18 September 2015; IEEE: New York, NY, USA, 2015; pp. 3347–3350. [Google Scholar]
- Nieto, D.; Delgado, T.; Flores-Arias, M.T. Fabrication of microchannels on soda-lime glass substrates with a Nd:YVO4 laser. Opt. Lasers Eng. 2014, 63, 11–18. [Google Scholar] [CrossRef]
- Bahadorimehr, A.; Majlis, B.Y. Fabrication of Glass-based Microfluidic Devices with Photoresist as Mask. Elektron. Elektrotech. 2011, 116, 45–48. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Chen, Q.; Maccioni, G. Fabrication of microfluidics structures on different glasses by simplified imprinting technique. Curr. Appl. Phys. 2013, 13, 256–261. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Kuo, C.-H.; Hsiao, W.-T.; Huang, K.-C.; Tseng, S.-F.; Chou, C.-P. Glass biochip fabrication by laser micromachining and glass-molding process. J. Mater. Process. Technol. 2012, 212, 633–639. [Google Scholar] [CrossRef]
- Tseng, S.-F.; Chen, M.-F.; Hsiao, W.-T.; Huang, C.-Y.; Yang, C.-H.; Chen, Y.-S. Laser micromilling of convex microfluidic channels onto glassy carbon for glass molding dies. Opt. Lasers Eng. 2014, 57, 58–63. [Google Scholar] [CrossRef]
- Ju, J.; Lim, S.; Seok, J.; Kim, S.M. A method to fabricate low-cost and large area vitreous carbon mold for glass molded microstructures. Int. J. Precis. Eng. Manuf. 2015, 16, 287–291. [Google Scholar] [CrossRef]
- Faustino, V.; Catarino, S.O.; Lima, R.; Minas, G. Biomedical microfluidic devices by using low-cost fabrication techniques: A review. J. Biomech. 2016, 49, 2280–2292. [Google Scholar] [CrossRef] [PubMed]
- Pinto, V.C.; Sousa, P.J.; Cardoso, V.F.; Minas, G. Optimized SU-8 processing for low-cost microstructures fabrication without cleanroom facilities. Micromachines 2014, 5, 738–755. [Google Scholar] [CrossRef]
- Pinto, E.; Faustino, V.; Rodrigues, R.O.; Pinho, D.; Garcia, V.; Miranda, J.M.; Lima, R. A rapid and low-cost nonlithographic method to fabricate biomedical microdevices for blood flow analysis. Micromachines 2014, 6, 121–135. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, W.; Hou, Z.; Wu, Z. A rapid prototyping technique for microfluidics with high robustness and flexibility. Micromachines 2016, 7. [Google Scholar] [CrossRef]
- Tripathi, S.; Prabhakar, A.; Kumar, N.; Singh, S.G.; Agrawal, A. Blood plasma separation in elevated dimension T-shaped microchannel. Biomed. Microdevices 2013, 15, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Biggs, R.; Macmillan, R.L. The Error of the Red Cell Count. J. Clin. Pathol. 1948, 1, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-H.; Lee, Y.-M.; Shin, K.-H.; Heo, Y.-M. A study on the aspheric glass tens forming analysis in the progressive GMP process. J. Opt. Soc. Korea 2007, 11, 85–92. [Google Scholar] [CrossRef]

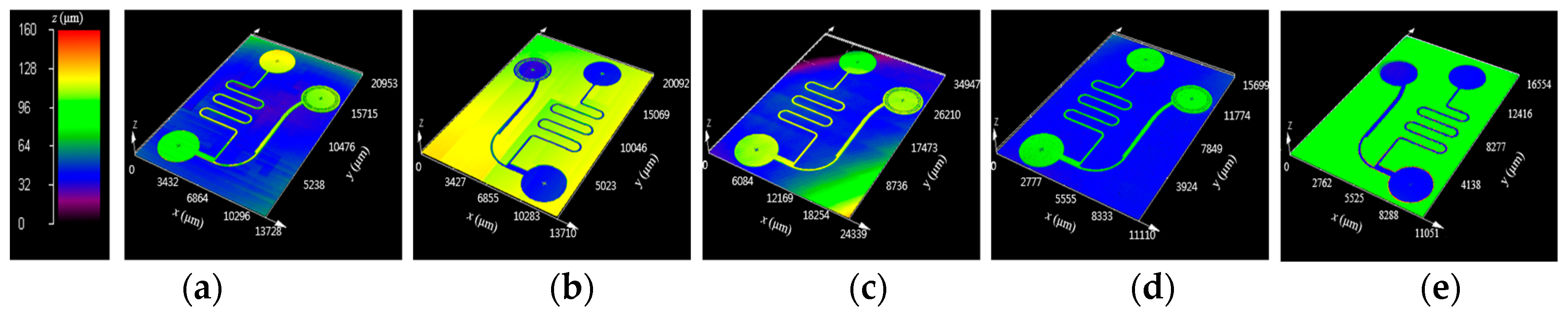


| Samples | Inlet Channel | Outlet Channel | Extraction Channel | |||
|---|---|---|---|---|---|---|
| Width (μm) | Height (μm) | Width (μm) | Height (μm) | Width (μm) | Height (μm) | |
| Silicon master | 204.5 | 71.4 | 600.4 | 71.9 | 200.3 | 71.6 |
| polydimethylsiloxane (PDMS) mold | 202.5 | 71.0 | 584.9 | 71.7 | 190.6 | 71.1 |
| Furan precursor | 196.4 | 67.8 | 585.9 | 68.2 | 190.8 | 68.9 |
| amorphous carbon (AC) mold | 153.6 | 51.6 | 462.7 | 53.7 | 151.0 | 53.4 |
| Glass replica | 154.9 | 52.9 | 464.6 | 54.4 | 155.4 | 53.9 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, H.; Haq, M.R.; Ju, J.; Kim, Y.; Kim, S.-m.; Lim, J. Fabrication of All Glass Bifurcation Microfluidic Chip for Blood Plasma Separation. Micromachines 2017, 8, 67. https://doi.org/10.3390/mi8030067
Jang H, Haq MR, Ju J, Kim Y, Kim S-m, Lim J. Fabrication of All Glass Bifurcation Microfluidic Chip for Blood Plasma Separation. Micromachines. 2017; 8(3):67. https://doi.org/10.3390/mi8030067
Chicago/Turabian StyleJang, Hyungjun, Muhammad Refatul Haq, Jonghyun Ju, Youngkyu Kim, Seok-min Kim, and Jiseok Lim. 2017. "Fabrication of All Glass Bifurcation Microfluidic Chip for Blood Plasma Separation" Micromachines 8, no. 3: 67. https://doi.org/10.3390/mi8030067
APA StyleJang, H., Haq, M. R., Ju, J., Kim, Y., Kim, S.-m., & Lim, J. (2017). Fabrication of All Glass Bifurcation Microfluidic Chip for Blood Plasma Separation. Micromachines, 8(3), 67. https://doi.org/10.3390/mi8030067






