Neural Probes for Chronic Applications
Abstract
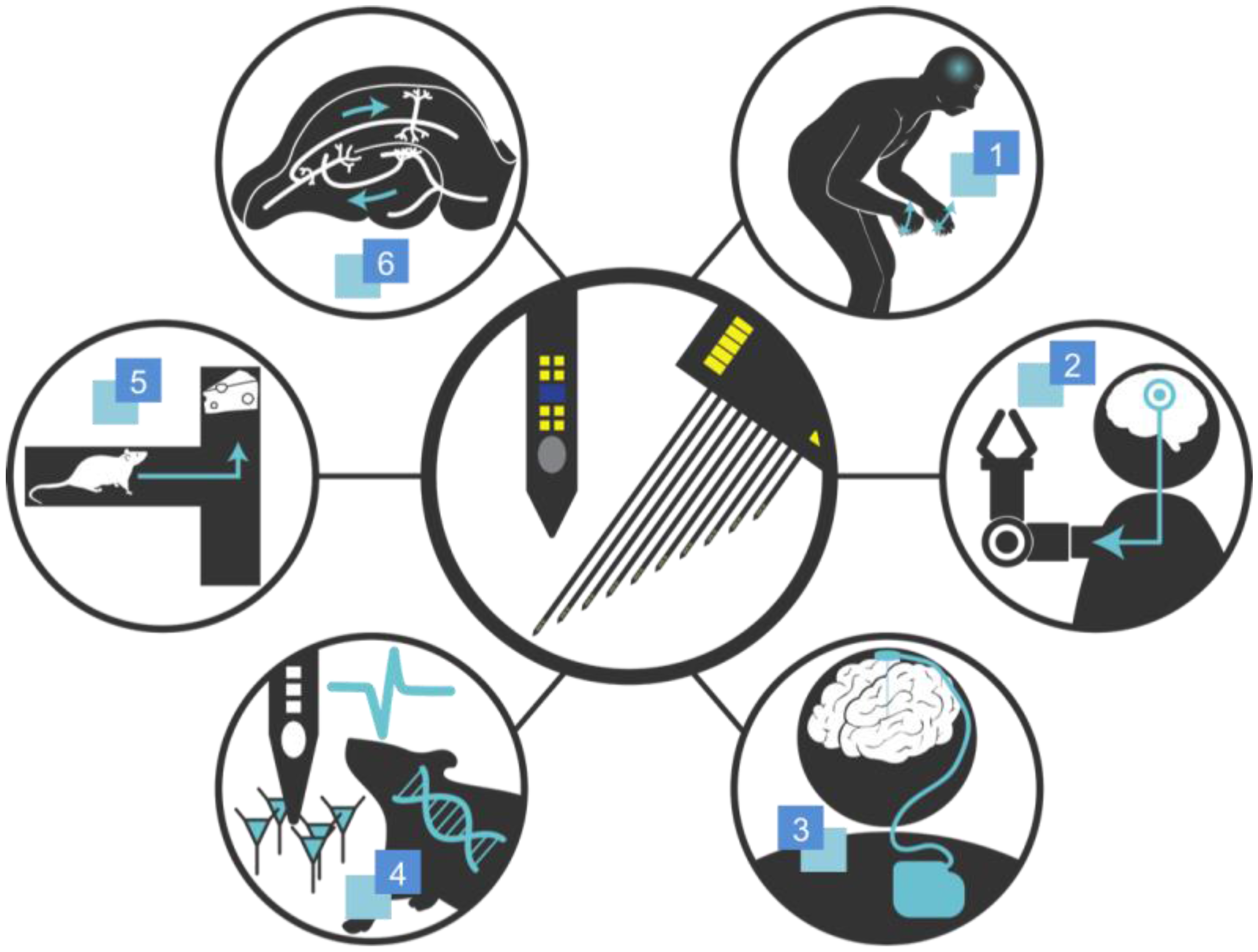
:1. Introduction
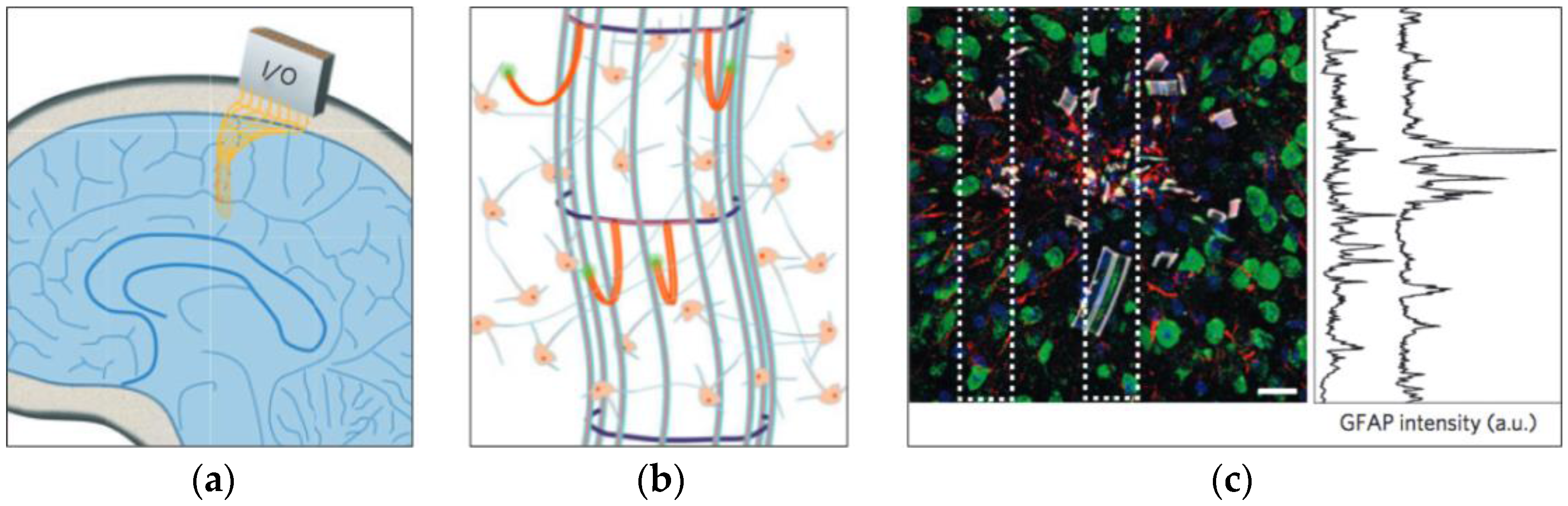
2. Potential Causes of Damage
3. Common Assessment Methods of Chronic Reliability
3.1. Biological Assays
3.2. Electrochemical Impedance Spectroscopy
3.3. Signal-to-Noise Ratio (SNR)
4. Strategies to Achieve Chronic Reliability
4.1. Implantation Procedure
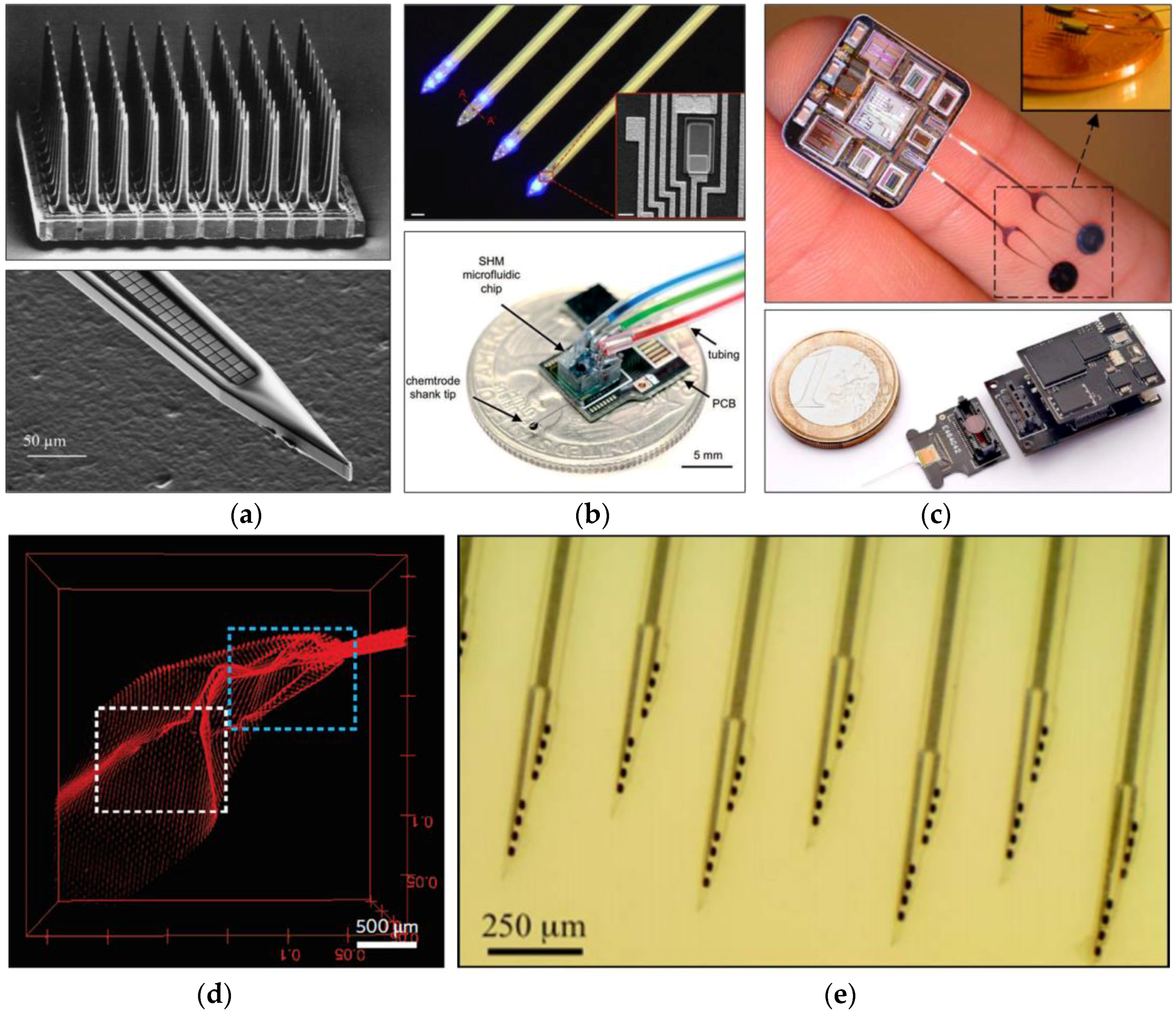
4.2. Design of Neural Probes
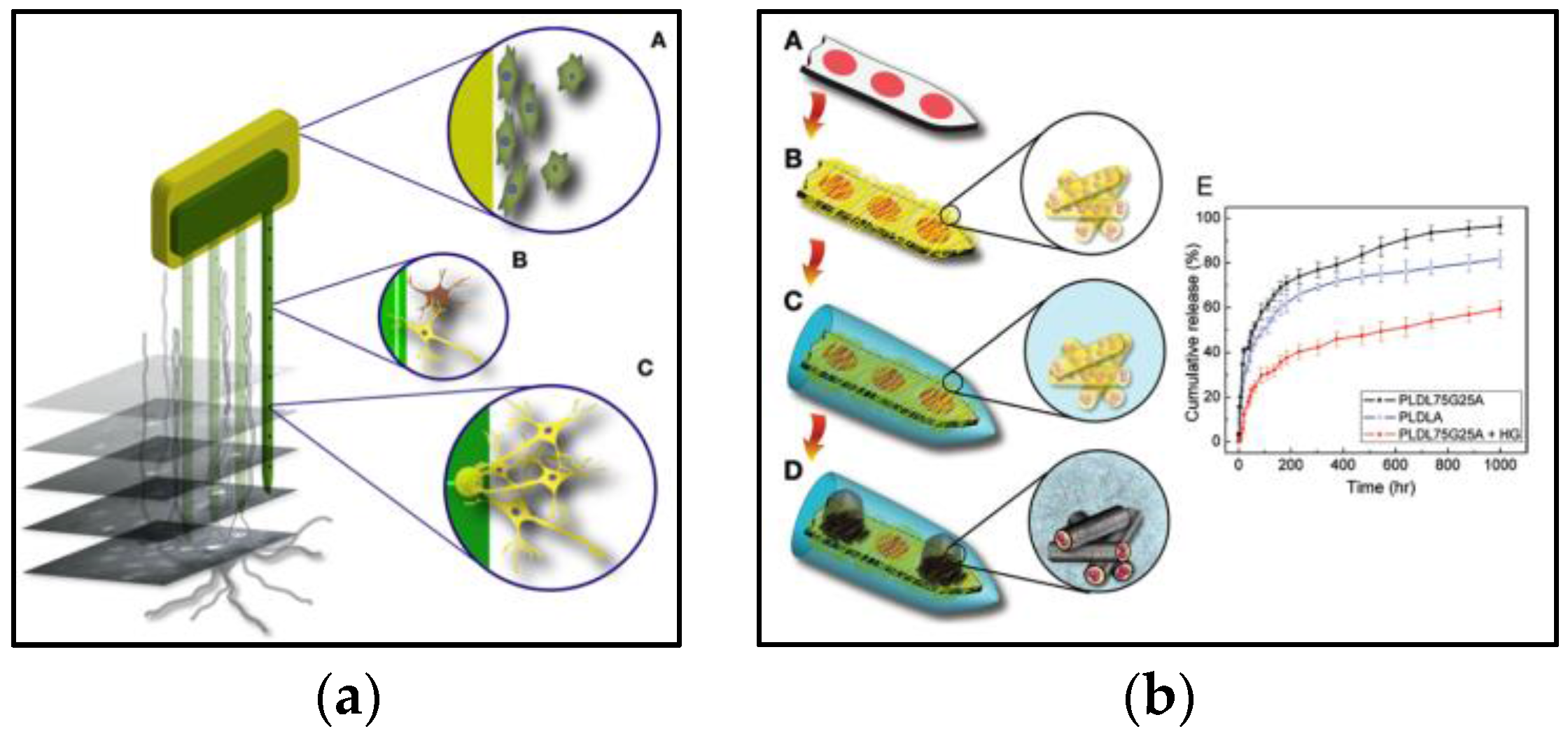
4.3. Biocompatibility
4.4. Flexible Substrate
4.5. Wireless Technology and Packaging
4.6. Other Novel Methods
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Calabresi, P.; Centonze, D.; Bernardi, G. Electrophysiology of dopamine in normal and denervated striatal neurons. Trends Neurosci. 2000, 23, S57–S63. [Google Scholar] [CrossRef]
- Altuna, A.; Bellistri, E.; Cid, E.; Aivar, P.; Gal, B.; Berganzo, J.; Gabriel, G.; Guimerà, A.; Villa, R.; Fernández, L.J. SU-8 based microprobes for simultaneous neural depth recording and drug delivery in the brain. Lab Chip 2013, 13, 1422–1430. [Google Scholar] [CrossRef] [PubMed]
- Alivisatos, A.P.; Chun, M.; Church, G.M.; Greenspan, R.J.; Roukes, M.L.; Yuste, R. The brain activity map project and the challenge of functional connectomics. Neuron 2012, 74, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Scanziani, M.; Häusser, M. Electrophysiology in the age of light. Nature 2009, 461, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, L.R.; Bacher, D.; Jarosiewicz, B.; Masse, N.Y.; Simeral, J.D.; Vogel, J.; Haddadin, S.; Liu, J.; Cash, S.S.; van der Smagt, P. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 2012, 485, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, L.R.; Serruya, M.D.; Friehs, G.M.; Mukand, J.A.; Saleh, M.; Caplan, A.H.; Branner, A.; Chen, D.; Penn, R.D.; Donoghue, J.P. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature 2006, 442, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Rajangam, S.; Tseng, P.-H.; Yin, A.; Lehew, G.; Schwarz, D.; Lebedev, M.A.; Nicolelis, M.A.L. Wireless cortical brain-machine interface for whole-body navigation in primates. Sci. Rep. 2016, 6, 22170. [Google Scholar] [CrossRef] [PubMed]
- Velliste, M.; Perel, S.; Spalding, M.C.; Whitford, A.S.; Schwartz, A.B. Cortical control of a prosthetic arm for self-feeding. Nature 2008, 453, 1098–1101. [Google Scholar] [CrossRef] [PubMed]
- Kipke, D.R.; Shain, W.; Buzsáki, G.; Fetz, E.; Henderson, J.M.; Hetke, J.F.; Schalk, G. Advanced neurotechnologies for chronic neural interfaces: New horizons and clinical opportunities. J. Neurosci. 2008, 28, 11830–11838. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Son, Y.; Kim, J.; Lee, C.J.; Yoon, E.-S.; Cho, I.-J. A multichannel neural probe with embedded microfluidic channels for simultaneous in vivo neural recording and drug delivery. Lab Chip 2015, 15, 1590–1597. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Lee, H.J.; Chae, U.; Kim, H.; Kim, J.; Choi, N.; Woo, J.; Cho, Y.; Lee, C.J.; Yoon, E.-S. Neural probes with multi-drug delivery capability. Lab Chip 2015, 15, 3730–3737. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.; Lee, H.J.; Kim, J.; Shin, H.; Choi, N.; Lee, C.J.; Yoon, E.-S.; Yoon, E.; Wise, K.D.; Kim, T.G. In vivo optical modulation of neural signals using monolithically integrated two-dimensional neural probe arrays. Sci. Rep. 2015, 5, 15466. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Stark, E.; Ku, P.-C.; Wise, K.D.; Buzsáki, G.; Yoon, E. Monolithically integrated μleds on silicon neural probes for high-resolution optogenetic studies in behaving animals. Neuron 2015, 88, 1136–1148. [Google Scholar] [CrossRef] [PubMed]
- Wise, K.D.; Angell, J.B.; Starr, A. An integrated-circuit approach to extracellular microelectrodes. IEEE Trans. Biomed. Eng. 1970, BME-17, 238–247. [Google Scholar] [CrossRef]
- Van Dijck, G.; Seidl, K.; Paul, O.; Ruther, P.; Van Hulle, M.M.; Maex, R. Enhancing the yield of high-density electrode arrays through automated electrode selection. Int. J. Neural Syst. 2012, 22, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Blanche, T.J.; Harrison, R.R.; Lester, H.A.; Masmanidis, S.C. Multiplexed, high density electrophysiology with nanofabricated neural probes. PLOS ONE 2011, 6, e26204. [Google Scholar] [CrossRef] [PubMed]
- Scholvin, J.; Kinney, J.P.; Bernstein, J.G.; Moore-Kochlacs, C.; Kopell, N.; Fonstad, C.G.; Boyden, E.S. Close-packed silicon microelectrodes for scalable spatially oversampled neural recording. IEEE Trans. Biomed. Eng. 2016, 63, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.M.; Andrei, A.; Mitra, S.; Welkenhuysen, M.; Eberle, W.; Bartic, C.; Puers, R.; Yazicioglu, R.F.; Gielen, G.G.E. An implantable 455-active-electrode 52-channel cmos neural probe. IEEE J. Solid-State Circuits 2014, 49, 248–261. [Google Scholar] [CrossRef]
- Sodagar, A.M.; Perlin, G.E.; Yao, Y.; Najafi, K.; Wise, K.D. An implantable 64-channel wireless microsystem for single-unit neural recording. IEEE J. Solid-State Circuits 2009, 44, 2591–2604. [Google Scholar] [CrossRef]
- Seymour, J.P.; Kipke, D.R. Neural probe design for reduced tissue encapsulation in CNS. Biomaterials 2007, 28, 3594–3607. [Google Scholar] [CrossRef] [PubMed]
- Lewitus, D.Y.; Smith, K.L.; Landers, J.; Neimark, A.V.; Kohn, J. Bioactive agarose carbon-nanotube composites are capable of manipulating brain–implant interface. J. Appl. Polym. Sci. 2014, 131, 40297. [Google Scholar] [CrossRef] [PubMed]
- Taub, A.H.; Hogri, R.; Magal, A.; Mintz, M.; Shacham-Diamand, Y. Bioactive anti-inflammatory coating for chronic neural electrodes. J. Biomed. Mater. Res. Part A 2012, 100, 1854–1858. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Gugel, Z.; Li, X.; Gilgunn, P.J.; Khilwani, R.; Ozdoganlar, O.B.; Fedder, G.K.; Weber, D.J.; Cui, X.T. Chronic tissue response to carboxymethyl cellulose based dissolvable insertion needle for ultra-small neural probes. Biomaterials 2014, 35, 9255–9268. [Google Scholar] [CrossRef] [PubMed]
- Normann, R.A.; Maynard, E.M.; Rousche, P.J.; Warren, D.J. A neural interface for a cortical vision prosthesis. Vis. Res. 1999, 39, 2577–2587. [Google Scholar] [CrossRef]
- Takeuchi, S.; Suzuki, T.; Mabuchi, K.; Fujita, H. 3D flexible multichannel neural probe array. J. Micromech. Microeng. 2003, 14, 104. [Google Scholar] [CrossRef]
- Chen, J.; Wise, K.D.; Hetke, J.F.; Bledsoe, S.C. A multichannel neural probe for selective chemical delivery at the cellular level. IEEE Trans. Biomed. Eng. 1997, 44, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Canales, A.; Jia, X.; Froriep, U.P.; Koppes, R.A.; Tringides, C.M.; Selvidge, J.; Lu, C.; Hou, C.; Wei, L.; Fink, Y. Multifunctional fibers for simultaneous optical, electrical and chemical interrogation of neural circuits in vivo. Nat. Biotechnol. 2015, 33, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-I.; McCall, J.G.; Jung, Y.H.; Huang, X.; Siuda, E.R.; Li, Y.; Song, J.; Song, Y.M.; Pao, H.A.; Kim, R.-H. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 2013, 340, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Segev, E.; Fowler, T.; Faraon, A.; Roukes, M.L. Visible array waveguide gratings for applications of optical neural probes. In Proceedings of the 2015 SPIE BiOS, San Francisco, CA, USA, 7–12 February 2015; International Society for Optics and Photonics: Bellingham, WA, USA, 2015. [Google Scholar]
- Chamanzar, M.; Denman, D.J.; Blanche, T.J.; Maharbiz, M.M. Ultracompact optoflex neural probes for high-resolution electrophysiology and optogenetic stimulation. In Proceedings of the 28th IEEE International Conference on Micro Electro Mechanical Systems (MEMS), Estoril, Portugal, 18–22 January 2015; pp. 682–685.
- Grand, L.; Wittner, L.; Herwik, S.; Göthelid, E.; Ruther, P.; Oscarsson, S.; Neves, H.; Dombovári, B.; Csercsa, R.; Karmos, G. Short and long term biocompatibility of neuroprobes silicon probes. J. Neurosci. Methods 2010, 189, 216–229. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, O.; Juliano, S.L. Transplants of NGF-secreting fibroblasts restore stimulus-evoked activity in barrel cortex of basal-forebrain-lesioned rats. J. Neurophysiol. 2001, 86, 2081–2096. [Google Scholar] [PubMed]
- Tien, L.W.; Wu, F.; Tang-Schomer, M.D.; Yoon, E.; Omenetto, F.G.; Kaplan, D.L. Silk as a multifunctional biomaterial substrate for reduced glial scarring around brain-penetrating electrodes. Adv. Funct. Mater. 2013, 23, 3185–3193. [Google Scholar] [CrossRef]
- Wu, F.; Tien, L.; Chen, F.; Kaplan, D.; Berke, J.; Yoon, E. A multi-shank silk-backed parylene neural probe for reliable chronic recording. In Proceedings of the 2013 Transducers & Eurosensors XXVII: The 17th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS & EUROSENSORS XXVII), Barcelona, Spain, 16–20 June 2013; pp. 888–891.
- Wu, F.; Tien, L.W.; Chen, F.; Berke, J.D.; Kaplan, D.L.; Yoon, E. Silk-backed structural optimization of high-density flexible intracortical neural probes. J. Microelectromech. Syst. 2015, 24, 62–69. [Google Scholar] [CrossRef]
- Winslow, B.D.; Christensen, M.B.; Yang, W.-K.; Solzbacher, F.; Tresco, P.A. A comparison of the tissue response to chronically implanted parylene-C-coated and uncoated planar silicon microelectrode arrays in rat cortex. Biomaterials 2010, 31, 9163–9172. [Google Scholar] [CrossRef] [PubMed]
- Mercanzini, A.; Reddy, S.T.; Velluto, D.; Colin, P.; Maillard, A.; Bensadoun, J.-C.; Hubbell, J.A.; Renaud, P. Controlled release nanoparticle-embedded coatings reduce the tissue reaction to neuroprostheses. J. Control. Release 2010, 145, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Jaquins-Gerstl, A.S.; Vazquez, A.L.; Michael, A.C.; Cui, X.T. Dexamethasone retrodialysis attenuates microglial response to implanted probes in vivo. Biomaterials 2016, 87, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Aregueta-Robles, U.A.; Woolley, A.J.; Poole-Warren, L.A.; Lovell, N.H.; Green, R.A. Organic electrode coatings for next-generation neural interfaces. Front. Neuroeng. 2015, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Lai, H.Y.; Kuo, L.W.; Liao, C.H.; Chang, P.H.; Liu, T.C.; Chen, S.Y.; Chen, Y.Y. Multifunctional 3D patternable drug-embedded nanocarrier-based interfaces to enhance signal recording and reduce neuron degeneration in neural implantation. Adv. Mater. 2015, 27, 4186–4193. [Google Scholar] [CrossRef] [PubMed]
- Metz, S.; Bertsch, A.; Bertrand, D.; Renaud, P. Flexible polyimide probes with microelectrodes and embedded microfluidic channels for simultaneous drug delivery and multi-channel monitoring of bioelectric activity. Biosens. Bioelectron. 2004, 19, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.-Y.; Liao, L.-D.; Lin, C.-T.; Hsu, J.-H.; He, X.; Chen, Y.-Y.; Chang, J.-Y.; Chen, H.-F.; Tsang, S.; Shih, Y.-Y.I. Design, simulation and experimental validation of a novel flexible neural probe for deep brain stimulation and multichannel recording. J. Neural Eng. 2012, 9, 036001. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Singh, A.; He, J.; Massia, S.; Kim, B.; Raupp, G. Polyimide based neural implants with stiffness improvement. Sens. Actuators B Chem. 2004, 102, 67–72. [Google Scholar] [CrossRef]
- Rousche, P.J.; Pellinen, D.S.; Pivin, D.P.; Williams, J.C.; Vetter, R.J.; Kipke, D.R. Flexible polyimide-based intracortical electrode arrays with bioactive capability. IEEE Trans. Biomed. Eng. 2001, 48, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Seymour, J.P.; Langhals, N.B.; Anderson, D.J.; Kipke, D.R. Novel multi-sided, microelectrode arrays for implantable neural applications. Biomed. Microdevices 2011, 13, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Sohal, H.S.; Vassilevski, K.; Jackson, A.; Baker, S.N.; O’Neill, A. Design and microfabrication considerations for reliable flexible intracortical implants. ArXiv E-Prints 2015. Available online: http://arxiv.org/abs/1506.04622 (accessed on 27 September 2016). [Google Scholar] [CrossRef]
- Castagnola, V.; Descamps, E.; Lecestre, A.; Dahan, L.; Remaud, J.; Nowak, L.G.; Bergaud, C. Parylene-based flexible neural probes with PEDOT coated surface for brain stimulation and recording. Biosens. Bioelectron. 2015, 67, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Kuo, J.T.W.; Hara, S.A.; Lee, C.D.; Yu, L.; Gutierrez, C.A.; Hoang, T.Q.; Pikov, V.; Meng, E. 3D parylene sheath neural probe for chronic recordings. J. Neural Eng. 2013, 10, 045002. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Roukes, M.L.; Masmanidis, S.C. Dual-side and three-dimensional microelectrode arrays fabricated from ultra-thin silicon substrates. J. Micromech. Microeng. 2009, 19, 075008. [Google Scholar] [CrossRef]
- Ong, X.C.; Forssell, M.; Fedder, G.K. Processing of platinum electrodes for parylene-c based neural probes. In Proceedings of the 2016 IEEE 29th International Conference on Micro Electro Mechanical Systems (MEMS), Shanghai, China, 24–28 January 2016; pp. 509–512.
- Kolarcik, C.L.; Luebben, S.D.; Sapp, S.A.; Hanner, J.; Snyder, N.; Kozai, T.D.Y.; Chang, E.; Nabity, J.A.; Nabity, S.T.; Lagenaur, C.F. Elastomeric and soft conducting microwires for implantable neural interfaces. Soft Matter 2015, 11, 4847–4861. [Google Scholar] [CrossRef] [PubMed]
- Rubehn, B.; Wolff, S.B.E.; Tovote, P.; Lüthi, A.; Stieglitz, T. A polymer-based neural microimplant for optogenetic applications: Design and first in vivo study. Lab Chip 2013, 13, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Yoon, I.; Hamaguchi, K.; Borzenets, I.V.; Finkelstein, G.; Mooney, R.; Donald, B.R. Intracellular neural recording with pure carbon nanotube probes. PLOS ONE 2013, 8, e65715. [Google Scholar] [CrossRef] [PubMed]
- Hess, A.E.; Capadona, J.R.; Shanmuganathan, K.; Hsu, L.; Rowan, S.J.; Weder, C.; Tyler, D.J.; Zorman, C.A. Development of a stimuli-responsive polymer nanocomposite toward biologically optimized, mems-based neural probes. J. Micromech. Microeng. 2011, 21, 054009. [Google Scholar] [CrossRef]
- Williams, D.F. There is no such thing as a biocompatible material. Biomaterials 2014, 35, 10009–10014. [Google Scholar] [CrossRef] [PubMed]
- Lo, M.-C.; Wang, S.; Singh, S.; Damodaran, V.B.; Kaplan, H.M.; Kohn, J.; Shreiber, D.I.; Zahn, J.D. Coating flexible probes with an ultra fast degrading polymer to aid in tissue insertion. Biomed. Microdevices 2015, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.D.; Hara, S.A.; Yu, L.; Kuo, J.T.W.; Kim, B.J.; Hoang, T.; Pikov, V.; Meng, E. Matrigel coatings for parylene sheath neural probes. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.P.; Rajdev, P.; Ellison, C.; Irazoqui, P.P. Toward a comparison of microelectrodes for acute and chronic recordings. Brain Res. 2009, 1282, 183–200. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.-W.; Shin, G.; Park, S.I.; Yu, K.J.; Xu, L.; Rogers, J.A. Soft materials in neuroengineering for hard problems in neuroscience. Neuron 2015, 86, 175–186. [Google Scholar] [CrossRef] [PubMed]
- HajjHassan, M.; Chodavarapu, V.; Musallam, S. Neuromems: Neural probe microtechnologies. Sensors 2008, 8, 6704–6726. [Google Scholar] [CrossRef]
- Patil, A.C.; Thakor, N.V. Implantable neurotechnologies: A review of micro-and nanoelectrodes for neural recording. Med. Biol. Eng. Comput. 2016, 54, 23–44. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fu, T.-M.; Cheng, Z.; Hong, G.; Zhou, T.; Jin, L.; Duvvuri, M.; Jiang, Z.; Kruskal, P.; Xie, C. Syringe-injectable electronics. Nat. Nanotechnol. 2015, 10, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.C.; Renaud, P.; Tanila, H.; Djupsund, K. Flexible polyimide microelectrode array for in vivo recordings and current source density analysis. Biosens. Bioelectron. 2007, 22, 1783–1790. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, K.A.; Uram, J.D.; Yang, J.; Martin, D.C.; Kipke, D.R. Chronic neural recordings using silicon microelectrode arrays electrochemically deposited with a poly (3, 4-ethylenedioxythiophene)(PEDOT) film. J. Neural Eng. 2006, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Langhals, N.B.; Patel, P.R.; Deng, X.; Zhang, H.; Smith, K.L.; Lahann, J.; Kotov, N.A.; Kipke, D.R. Ultrasmall implantable composite microelectrodes with bioactive surfaces for chronic neural interfaces. Nat. Mater. 2012, 11, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Vetter, R.J.; Williams, J.C.; Hetke, J.F.; Nunamaker, E.A.; Kipke, D.R. Chronic neural recording using silicon-substrate microelectrode arrays implanted in cerebral cortex. IEEE Trans. Biomed. Eng. 2004, 51, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, M.A.; Nicolelis, M.A.L. Brain–machine interfaces: Past, present and future. Trends Neurosci. 2006, 29, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Xue, Q.-S.; Sankar, V.; Nishida, T.; Shaw, G.; Streit, W.J.; Sanchez, J.C. Comprehensive characterization and failure modes of tungsten microwire arrays in chronic neural implants. J. Neural Eng. 2012, 9, 056015. [Google Scholar] [CrossRef] [PubMed]
- Karumbaiah, L.; Saxena, T.; Carlson, D.; Patil, K.; Patkar, R.; Gaupp, E.A.; Betancur, M.; Stanley, G.B.; Carin, L.; Bellamkonda, R.V. Relationship between intracortical electrode design and chronic recording function. Biomaterials 2013, 34, 8061–8074. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Jaquins-Gerstl, A.S.; Vazquez, A.L.; Michael, A.C.; Cui, X.T. Brain tissue responses to neural implants impact signal sensitivity and intervention strategies. ACS Chem. Neurosci. 2015, 6, 48–67. [Google Scholar] [CrossRef] [PubMed]
- Gunasekera, B.; Saxena, T.; Bellamkonda, R.; Karumbaiah, L. Intracortical recording interfaces: Current challenges to chronic recording function. ACS Chem. Neurosci. 2015, 6, 68–83. [Google Scholar] [CrossRef] [PubMed]
- Prodanov, D.; Delbeke, J. Mechanical and biological interactions of implants with the brain and their impact on implant design. Front. Neurosci. 2016, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Jorfi, M.; Skousen, J.L.; Weder, C.; Capadona, J.R. Progress towards biocompatible intracortical microelectrodes for neural interfacing applications. J. Neural Eng. 2014, 12, 011001. [Google Scholar] [CrossRef] [PubMed]
- Sommakia, S.; Lee, H.C.; Gaire, J.; Otto, K.J. Materials approaches for modulating neural tissue responses to implanted microelectrodes through mechanical and biochemical means. Curr. Opin. Solid State Mater. Sci. 2014, 18, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Szarowski, D.H.; Andersen, M.D.; Retterer, S.; Spence, A.J.; Isaacson, M.; Craighead, H.G.; Turner, J.N.; Shain, W. Brain responses to micro-machined silicon devices. Brain Res. 2003, 983, 23–35. [Google Scholar] [CrossRef]
- Barker, D.J.; Root, D.H.; Coffey, K.R.; Ma, S.; West, M.O. A procedure for implanting organized arrays of microwires for single-unit recordings in awake, behaving animals. J. Vis. Exp. 2014, 84, 51004. [Google Scholar] [CrossRef] [PubMed]
- Gage, G.J.; Stoetzner, C.R.; Richner, T.; Brodnick, S.K.; Williams, J.C.; Kipke, D.R. Surgical implantation of chronic neural electrodes for recording single unit activity and electrocorticographic signals. J. Vis. Exp. 2012, 60, 3565. [Google Scholar] [CrossRef] [PubMed]
- Otchy, T.M.; Ӧlveczky, B.P. Design and assembly of an ultra-light motorized microdrive for chronic neural recordings in small animals. J. Vis. Exp. 2012, 69, e4314. [Google Scholar] [CrossRef] [PubMed]
- Polikov, V.S.; Tresco, P.A.; Reichert, W.M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 2005, 148, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.; Horch, K.; Normann, R. Biocompatibility of silicon-based electrode arrays implanted in feline cortical tissue. J. Biomed. Mater. Res. 1993, 27, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Marin, C.; Fernández, E. Biocompatibility of intracortical microelectrodes: Current status and future prospects. Front. Neuroeng. 2010, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Biran, R.; Martin, D.C.; Tresco, P.A. Neuronal cell loss accompanies the brain tissue response to chronically implanted silicon microelectrode arrays. Exp. Neurol. 2005, 195, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Wiler, J.; Dzaman, M.; Altschuler, R.A.; Martin, D.C. In vivo studies of polypyrrole/peptide coated neural probes. Biomaterials 2003, 24, 777–787. [Google Scholar] [CrossRef]
- Potter, K.A.; Buck, A.C.; Self, W.K.; Callanan, M.E.; Sunil, S.; Capadona, J.R. The effect of resveratrol on neurodegeneration and blood brain barrier stability surrounding intracortical microelectrodes. Biomaterials 2013, 34, 7001–7015. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Liu, J.; Fu, T.-M.; Dai, X.; Zhou, W.; Lieber, C.M. Three-dimensional macroporous nanoelectronic networks as minimally invasive brain probes. Nat. Mater. 2015, 14, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Yuen, T.G.H.; Agnew, W.F. Histological evaluation of polyesterimide-insulated gold wires in brain. Biomaterials 1995, 16, 951–956. [Google Scholar] [CrossRef]
- Liu, X.; McCreery, D.B.; Carter, R.R.; Bullara, L.A.; Yuen, T.G.H.; Agnew, W.F. Stability of the interface between neural tissue and chronically implanted intracortical microelectrodes. IEEE Trans. Rehabil. Eng. 1999, 7, 315–326. [Google Scholar] [PubMed]
- Harris, J.P.; Hess, A.E.; Rowan, S.J.; Weder, C.; Zorman, C.A.; Tyler, D.J.; Capadona, J.R. In vivo deployment of mechanically adaptive nanocomposites for intracortical microelectrodes. J. Neural Eng. 2011, 8, 046010. [Google Scholar] [CrossRef] [PubMed]
- Nicolelis, M.A.L.; Dimitrov, D.; Carmena, J.M.; Crist, R.; Lehew, G.; Kralik, J.D.; Wise, S.P. Chronic, multisite, multielectrode recordings in macaque monkeys. Proc. Natl. Acad. Sci. USA 2003, 100, 11041–11046. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.S.; Michael, A.C. Invasive consequences of using micro-electrodes and microdialysis probes in the brain. TrAC Trends Anal. Chem. 2003, 22, 503–508. [Google Scholar] [CrossRef]
- Kristensen, B.W.; Noraberg, J.; Thiébaud, P.; Koudelka-Hep, M.; Zimmer, J. Biocompatibility of silicon-based arrays of electrodes coupled to organotypic hippocampal brain slice cultures. Brain Res. 2001, 896, 1–17. [Google Scholar] [CrossRef]
- Kozai, T.D.Y.; Li, X.; Bodily, L.M.; Caparosa, E.M.; Zenonos, G.A.; Carlisle, D.L.; Friedlander, R.M.; Cui, X.T. Effects of caspase-1 knockout on chronic neural recording quality and longevity: Insight into cellular and molecular mechanisms of the reactive tissue response. Biomaterials 2014, 35, 9620–9634. [Google Scholar] [CrossRef] [PubMed]
- Potter, K.A.; Buck, A.C.; Self, W.K.; Capadona, J.R. Stab injury and device implantation within the brain results in inversely multiphasic neuroinflammatory and neurodegenerative responses. J. Neural Eng. 2012, 9, 046020. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, T. Single neuron studies of the human brain: Probing cognition. Neuropathol. Exp. Neurol. 2015, 74, 1039. [Google Scholar] [CrossRef]
- Malaga, K.A.; Schroeder, K.E.; Patel, P.R.; Irwin, Z.T.; Thompson, D.E.; Bentley, J.N.; Lempka, S.F.; Chestek, C.A.; Patil, P.G. Data-driven model comparing the effects of glial scarring and interface interactions on chronic neural recordings in non-human primates. J. Neural Eng. 2015, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Purcell, E.K.; Thompson, D.E.; Ludwig, K.A.; Kipke, D.R. Flavopiridol reduces the impedance of neural prostheses in vivo without affecting recording quality. J. Neurosci. Methods 2009, 183, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Czanner, G.; Sarma, S.V.; Ba, D.; Eden, U.T.; Wu, W.; Eskandar, E.; Lim, H.H.; Temereanca, S.; Suzuki, W.A.; Brown, E.N. Measuring the signal-to-noise ratio of a neuron. Proc. Natl. Acad. Sci. USA 2015, 112, 7141–7146. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Du, Z.; Gugel, Z.V.; Smith, M.A.; Chase, S.M.; Bodily, L.M.; Caparosa, E.M.; Friedlander, R.M.; Cui, X.T. Comprehensive chronic laminar single-unit, multi-unit, and local field potential recording performance with planar single shank electrode arrays. J. Neurosci. Methods 2015, 242, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.K.; Jones, K.E.; Huber, R.J.; Horch, K.W.; Normann, R.A. A silicon-based, three-dimensional neural interface: Manufacturing processes for an intracortical electrode array. IEEE Trans. Biomed. Eng. 1991, 38, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Maynard, E.M.; Fernandez, E.; Normann, R.A. A technique to prevent dural adhesions to chronically implanted microelectrode arrays. J. Neurosci. Methods 2000, 97, 93–101. [Google Scholar] [CrossRef]
- Rousche, P.J.; Normann, R.A. A method for pneumatically inserting an array of penetrating electrodes into cortical tissue. Ann. Biomed. Eng. 1992, 20, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Jackson, N.; Sridharan, A.; Anand, S.; Baker, M.; Okandan, M.; Muthuswamy, J. Long-term neural recordings using mems based moveable microelectrodes in the brain. Front. Neuroeng. 2010, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Im, M.; Yoon, E. A flexible fish-bone-shaped neural probe strengthened by biodegradable silk coating for enhanced biocompatibility. In Proceedings of the 16th International Solid-State Sensors, Actuators and Microsystems Conference, Beijing, China, 5–9 June 2011; pp. 966–969.
- Lind, G.; Linsmeier, C.E.; Schouenborg, J. The density difference between tissue and neural probes is a key factor for glial scarring. Sci. Rep. 2013, 3, 2942. [Google Scholar] [CrossRef] [PubMed]
- Edell, D.J.; Toi, V.V.; McNeil, V.M.; Clark, L.D. Factors influencing the biocompatibility of insertable silicon microshafts in cerebral cortex. IEEE Trans. Biomed. Eng. 1992, 39, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.; Catt, K.; Li, X.; Gugel, Z.V.; Olafsson, V.T.; Vazquez, A.L.; Cui, X.T. Mechanical failure modes of chronically implanted planar silicon-based neural probes for laminar recording. Biomaterials 2014, 37, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Subbaroyan, J.; Martin, D.C.; Kipke, D.R. A finite-element model of the mechanical effects of implantable microelectrodes in the cerebral cortex. J. Neural Eng. 2005, 2, 103. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Zhou, H.; Li, T.; Li, C.; Duan, Y.Y. Polyethylene glycol-containing polyurethane hydrogel coatings for improving the biocompatibility of neural electrodes. Acta Biomater. 2012, 8, 2233–2242. [Google Scholar] [CrossRef] [PubMed]
- He, W.; McConnell, G.C.; Bellamkonda, R.V. Nanoscale laminin coating modulates cortical scarring response around implanted silicon microelectrode arrays. J. Neural Eng. 2006, 3, 316. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Richardson-Burns, S.M.; Hendricks, J.L.; Sequera, C.; Martin, D.C. Effect of immobilized nerve growth factor on conductive polymers: Electrical properties and cellular response. Adv. Funct. Mater. 2007, 17, 79–86. [Google Scholar] [CrossRef]
- Cui, X.; Martin, D.C. Electrochemical deposition and characterization of poly (3, 4-ethylenedioxythiophene) on neural microelectrode arrays. Sens. Actuators B Chem. 2003, 89, 92–102. [Google Scholar] [CrossRef]
- Kolarcik, C.L.; Bourbeau, D.; Azemi, E.; Rost, E.; Zhang, L.; Lagenaur, C.F.; Weber, D.J.; Cui, X.T. In vivo effects of L1 coating on inflammation and neuronal health at the electrode-issue interface in rat spinal cord and dorsal root ganglion. Acta Biomater. 2012, 8, 3561–3575. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Saito, I.; Hoshino, T.; Suzuki, T.; Mabuchi, K. Preliminary study of multichannel flexible neural probes coated with hybrid biodegradable polymer. In Proceedings of the 28th IEEE Annual International Conference on Engineering in Medicine and Biology Society (EMBS’06), New York, NY, USA, 29–30 August 2006; pp. 660–663.
- Ignatius, M.J.; Sawhney, N.; Gupta, A.; Thibadeau, B.M.; Monteiro, O.R.; Brown, I.G. Bioactive surface coatings for nanoscale instruments: Effects on cns neurons. J. Biomed. Mater. Res. 1998, 40, 264–274. [Google Scholar] [CrossRef]
- Singh, A.; Ehteshami, G.; Massia, S.; He, J.; Storer, R.G.; Raupp, G. Glial cell and fibroblast cytotoxicity study on plasma-deposited diamond-like carbon coatings. Biomaterials 2003, 24, 5083–5089. [Google Scholar] [CrossRef]
- Abidian, M.R.; Martin, D.C. Multifunctional nanobiomaterials for neural interfaces. Adv. Funct. Mater. 2009, 19, 573–585. [Google Scholar] [CrossRef]
- Kam, L.; Shain, W.; Turner, J.N.; Bizios, R. Selective adhesion of astrocytes to surfaces modified with immobilized peptides. Biomaterials 2002, 23, 511–515. [Google Scholar] [CrossRef]
- Hickey, T.; Kreutzer, D.; Burgess, D.J.; Moussy, F. In vivo evaluation of a dexamethasone/plga microsphere system designed to suppress the inflammatory tissue response to implantable medical devices. J. Biomed. Mater. Res. 2002, 61, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Bellamkonda, R.V. Dexamethasone-coated neural probes elicit attenuated inflammatory response and neuronal loss compared to uncoated neural probes. Brain Res. 2007, 1148, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Potter, K.A.; Jorfi, M.; Householder, K.T.; Foster, E.J.; Weder, C.; Capadona, J.R. Curcumin-releasing mechanically adaptive intracortical implants improve the proximal neuronal density and blood–brain barrier stability. Acta Biomater. 2014, 10, 2209–2222. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.K.; Park, D.J.; Skousen, J.L.; Hess-Dunning, A.E.; Tyler, D.J.; Rowan, S.J.; Weder, C.; Capadona, J.R. Mechanically-compliant intracortical implants reduce the neuroinflammatory response. J. Neural Eng. 2014, 11, 056014. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.C.; Rennaker, R.L.; Kipke, D.R. Stability of chronic multichannel neural recordings: Implications for a long-term neural interface. Neurocomputing 1999, 26, 1069–1076. [Google Scholar] [CrossRef]
- Takeuchi, S.; Ziegler, D.; Yoshida, Y.; Mabuchi, K.; Suzuki, T. Parylene flexible neural probes integrated with microfluidic channels. Lab Chip 2005, 5, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; He, J.; Clement, R.; Massia, S.; Kim, B. Biocompatible benzocyclobutene (BCB)-based neural implants with micro-fluidic channel. Biosens. Bioelectron. 2004, 20, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Rousche, P.J.; Normann, R.A. Chronic recording capability of the utah intracortical electrode array in cat sensory cortex. J. Neurosci. Methods 1998, 82, 1–15. [Google Scholar] [CrossRef]
- Suner, S.; Fellows, M.R.; Vargas-Irwin, C.; Nakata, G.K.; Donoghue, J.P. Reliability of signals from a chronically implanted, silicon-based electrode array in non-human primate primary motor cortex. IEEE Trans. Neural Syst. Rehabil. Eng. 2005, 13, 524–541. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Catt, K.; Du, Z.; Na, K.; Srivannavit, O.; Razi-ul, M.H.; Seymour, J.; Wise, K.D.; Yoon, E.; Cui, X.T. Chronic in vivo evaluation of PEDOT/CNT for stable neural recordings. IEEE Trans. Biomed. Eng. 2016, 63, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Myllymaa, S.; Myllymaa, K.; Korhonen, H.; Töyräs, J.; Jääskeläinen, J.E.; Djupsund, K.; Tanila, H.; Lappalainen, R. Fabrication and testing of polyimide-based microelectrode arrays for cortical mapping of evoked potentials. Biosens. Bioelectron. 2009, 24, 3067–3072. [Google Scholar] [CrossRef] [PubMed]
- Kipke, D.R.; Vetter, R.J.; Williams, J.C.; Hetke, J.F. Silicon-substrate intracortical microelectrode arrays for long-term recording of neuronal spike activity in cerebral cortex. IEEE Trans. Neural Syst. Rehabil. Eng. 2003, 11, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Nurmikko, A.V.; Donoghue, J.P.; Hochberg, L.R.; Patterson, W.R.; Song, Y.-K.; Bull, C.W.; Borton, D.A.; Laiwalla, F.; Park, S.; Ming, Y. Listening to brain microcircuits for interfacing with external world—Progress in wireless implantable microelectronic neuroengineering devices. Proc. IEEE 2010, 98, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Wise, K.D.; Anderson, D.J.; Hetke, J.F.; Kipke, D.R.; Najafi, K. Wireless implantable microsystems: High-density electronic interfaces to the nervous system. Proc. IEEE 2004, 92, 76–97. [Google Scholar] [CrossRef]
| Stage | Before Implantation | During Implantation | Post-Implant (<1 Week) | Post-Implant (>1 Week) |
|---|---|---|---|---|
| In vivo procedure | Acute |  | Chronic | |
 |  |  |  | |
| Schematic view | 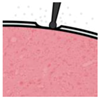 | 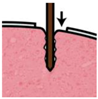 | 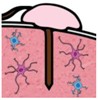 | 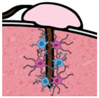 |
| Potential causes of damage |
|
|
|
|
| Induced effects |
|
|
|
|
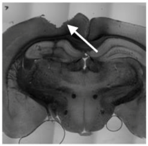 | 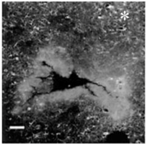 | 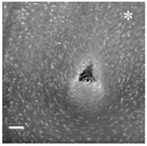 | 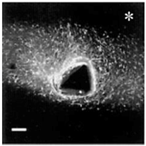 | |
| Results |
|
|
|
|
| Proposed solutions |
|
|
|
|
| Specificity | Name of Stain/Antibody | Purpose |
|---|---|---|
| Histology (Not cell-type-specific) | Hemotoxylin and Eosin (H & E) | Neurons (axons) |
| Cresyl violet (Nissl staining) | Neurons (somata) | |
| Luxol Fast Blue | Myelin (fatty acid sheath surrounding axons) | |
| Immunohistochemistry (Cell-type-specific) | Anti-NeuN a, Anti-MAP-2 b | Neurons |
| Anti-GFAP c | Astrocytes | |
| Anti-CD68 d, Anti-ED1 e, Anti-Iba1 f, Anti-OX42 g | Microglia/macrophages | |
| Anti-Neurofilament 200 | Neurofilaments | |
| Anti-Vimentin | Vimentin (both present in astrocytes and epithelial cells of meningeal fibroblasts) | |
| Anti-fibronectin | Extracellular matrix (ECM) protein (up-regulated in central nervous system (CNS) scar tissue) | |
| Anti-IgG h | BBB bleach |
| Surface Modification | Author (Year) | Materials | Animal | Experiments |
|---|---|---|---|---|
| Biocompatible Coating | Cui [111] (2003) | PEDOT a/DCDPGYIGSR | Guinea pig |
|
| Ludwig [64] (2006) | PEDOT | Rat |
| |
| Rao [108] (2012) | PEG b-PU c hydrogel | Rat |
| |
| Bioactive Coating | He [109] (2006) | Laminin | Rat |
|
| Kim [110] (2007) | PPy d/PSS e/NGF f PEDOT/PBS g/NGF | in vitro |
| |
| Kolarcik [112] (2012) | L1 | in vitro Rat |
| |
| Drug Release | Kato [113] (2006) | NGF (in microsphere) | Rat |
|
| Zhong [119] (2007) | DEX h (in nitrocellulose coating) | Rat |
| |
| Mercanzini [37] (2010) | DEX (in nanoparticles) | Rat |
| |
| Tien [33] (2013) | chABC i (in silk film) | in vitro |
| |
| Potter [120] (2014) | Curcumin (in poly(vinyl alcohol)) | Rats |
| |
| Huang [40] (2015) | PMSC j-OPC k (in nanogels) | Rat |
|
| Author (Year) | Neural Probe | Animal | Recording | |||||
|---|---|---|---|---|---|---|---|---|
| Sub., Elec. | w (μm), t (μm) | Type | No. | Implant Location | Weeks | Assessment Methods | Reason for Termination | |
| Maynard [100] (2000) | Si, n/a | n/a | Cat | 8 | Visual cortex | 35 | IHC, SNR | F |
| Vetter [66] (2004) | Si, Ir | 55, 15 | Rat | 10 | Auditory & motor cortex | 16 | IHC, SNR | S |
| Suner [126] (2005) | Si, Pt | 40, 2.6 | Monkey | 3 | Motor cortex | 137 | EIS, SNR | P |
| Ludwig [64] (2006) | Si, PEDOT | 90, 15 | Rat | 8 | Motor cortex | 6 | EIS, SNR | S |
| Kozai [127] (2016) | Si, PEDOT/CNT | 55, n/a | Mouse | 5 | Visual cortex | 22 | EIS, SNR | S |
| Jackson [102] (2010) | PolySi, PolySi | 50, 4 | Rat | 12 | Cortex | 20 | SNR | A |
| Kim [48] (2013) | Parylene, Pt | 350, 50 | Rat | 13 | Motor cortex | 4 | IHC, EIS, SNR | S |
| Wu [34] (2015) | Parylene, Au | 25, 65 | Rat | 1 | Motor cortex | 5 | SNR | S |
| Lee [57] (2015) | Parylene (Matrigel), Pt | 50–300, 11 | Rat | 2 | Motor cortex | 14 | IHC, EIS, SNR | n/a |
| Cheung [63] (2007) | PI, Pt | 75, 15 | Rat | 1 | Cortex & Hippocampus | 8 | IHC, SNR | F |
| Myllymaa [128] (2009) | PI, Au | 16, 5 | Rat | 5 | Thalamus | 7 | EIS, SNR | n/a |
| Huang [40] (2015) | PI, PLGA | 220, 50 | Rat | 10 | Thalamic VPM/VPL | 4 | IHC, EIS, SNR | n/a |
| Canales [27] (2014) | PC, CPE | 200, 200 | Mouse | 1 | Cortex | 8 | IHC, EIS, SNR | n/a |
| Kozai [65] (2012) | PEG, PEDOT | 8, 8 | Rat | 7 | Motor cortex | 5 | IHC, SNR | S |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kook, G.; Lee, S.W.; Lee, H.C.; Cho, I.-J.; Lee, H.J. Neural Probes for Chronic Applications. Micromachines 2016, 7, 179. https://doi.org/10.3390/mi7100179
Kook G, Lee SW, Lee HC, Cho I-J, Lee HJ. Neural Probes for Chronic Applications. Micromachines. 2016; 7(10):179. https://doi.org/10.3390/mi7100179
Chicago/Turabian StyleKook, Geon, Sung Woo Lee, Hee Chul Lee, Il-Joo Cho, and Hyunjoo Jenny Lee. 2016. "Neural Probes for Chronic Applications" Micromachines 7, no. 10: 179. https://doi.org/10.3390/mi7100179
APA StyleKook, G., Lee, S. W., Lee, H. C., Cho, I.-J., & Lee, H. J. (2016). Neural Probes for Chronic Applications. Micromachines, 7(10), 179. https://doi.org/10.3390/mi7100179






