On-Chip Method to Measure Mechanical Characteristics of a Single Cell by Using Moiré Fringe
Abstract
:1. Introduction
2. System Architecture for the Measurement of Cellular Mechanical Characteristics
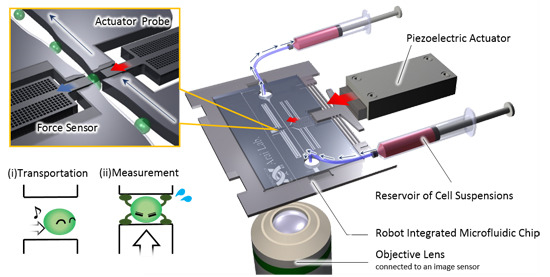
2.1. Overview of the On-Chip Cellular Measurement System
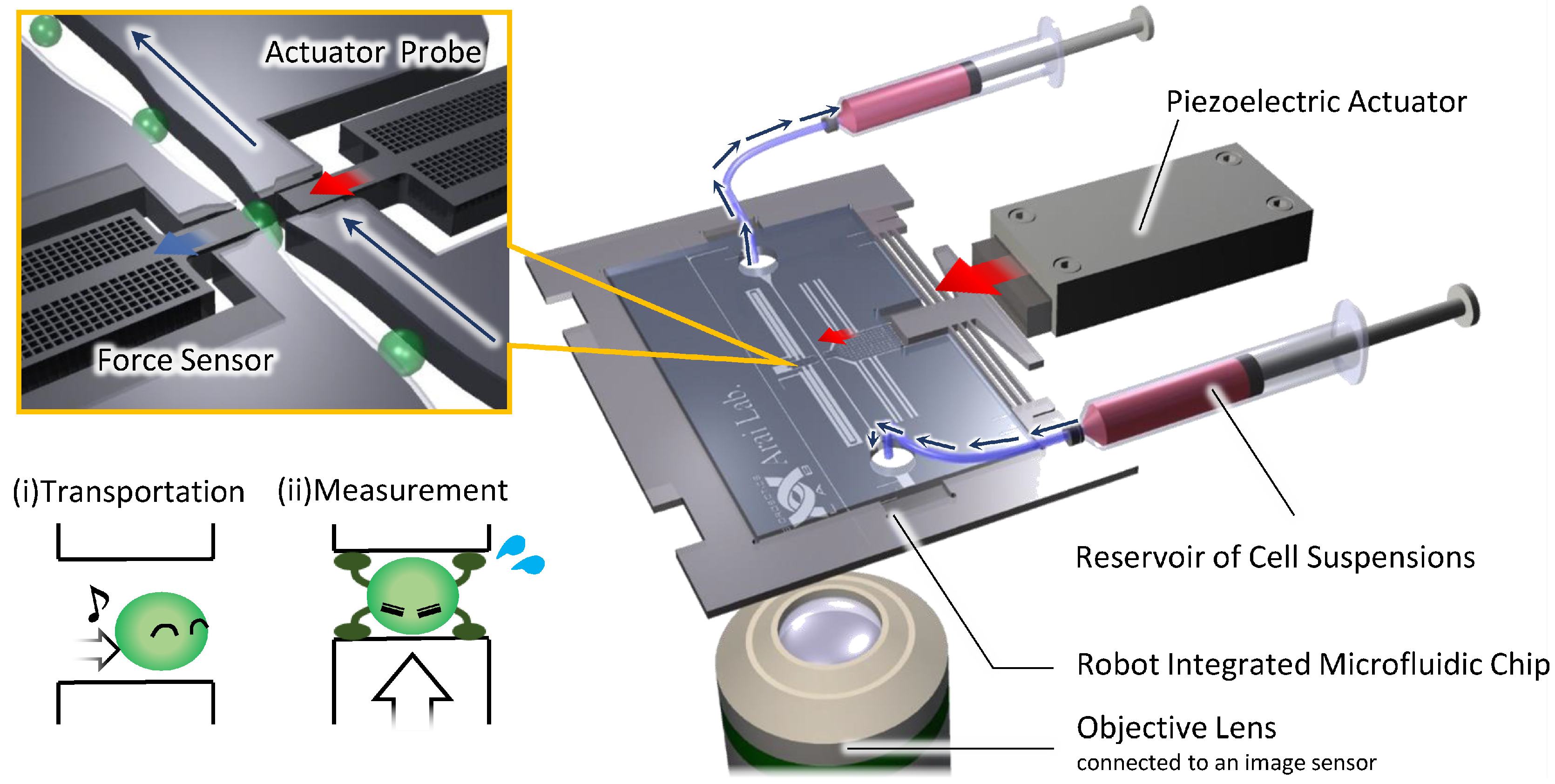
2.2. Fabrication Process of the Robot Integrated Microfluidic Chip
- (1)
- The clearance patterns are fabricated with PMER (Tokyo Ohka Co., Ltd., Tokyo, Japan).
- (2)
- The clearance is formed using reactive ion etching (RIE).
- (3)
- The sacrifice layer for the bonding process is deposited by chrome sputtering.
- (4)
- The shape of the glass layers is patterned with NCM-250 (Nichigo Morton Co., Ltd., Gifu, Japan).
- (5)
- The shape of the glass layers is formed by sandblasting.
- (1)
- The patterns of the device layer are fabricated with SU-8 (Nihon Kayaku Sangyo Co. Ltd, Gumma, Japan).
- (2)
- The shapes of the device are formed using deep-RIE. In this process, we fabricated grating structures onto the probe. Figure 2b is the scanning electron microscope (SEM) image of the fabricated grating structure. This is key to accurate measurement, as stated in the following section.
- (3)
- The bottom cover glass is bonded with the device layer using anodic bonding.
- (4)
- The patterns of the handling layer are fabricated with SU-8.
- (5)
- The connecting point to the actuator and frame of the chip are formed by using deep-RIE.
- (6)
- The exposed insulator layer is etched with HF solution.
- (7)
- The top cover glass is bonded by using anodic bonding. In this step, the device layer is connected to the ground via some holes on the bottom glass layer. The holes are buried with silver paste for the wiring.
- (8)
- The sacrificial chrome layers are removed by chrome etchant.

3. Improvement of the Sensing Precision and Accuracy
3.1. Technological Challenges for Improving Sensing Accuracy
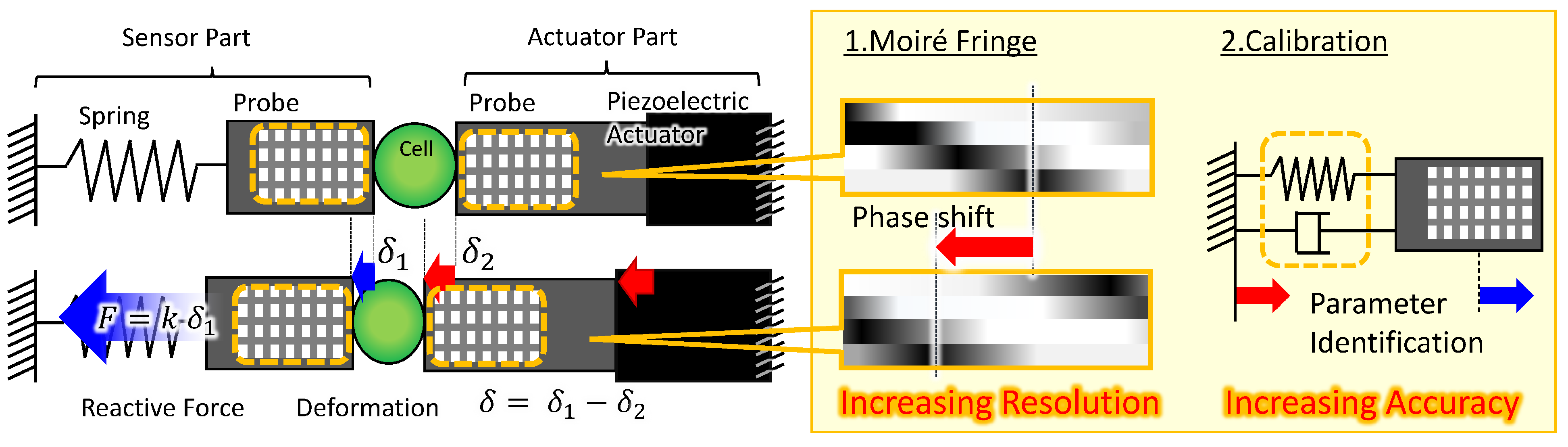
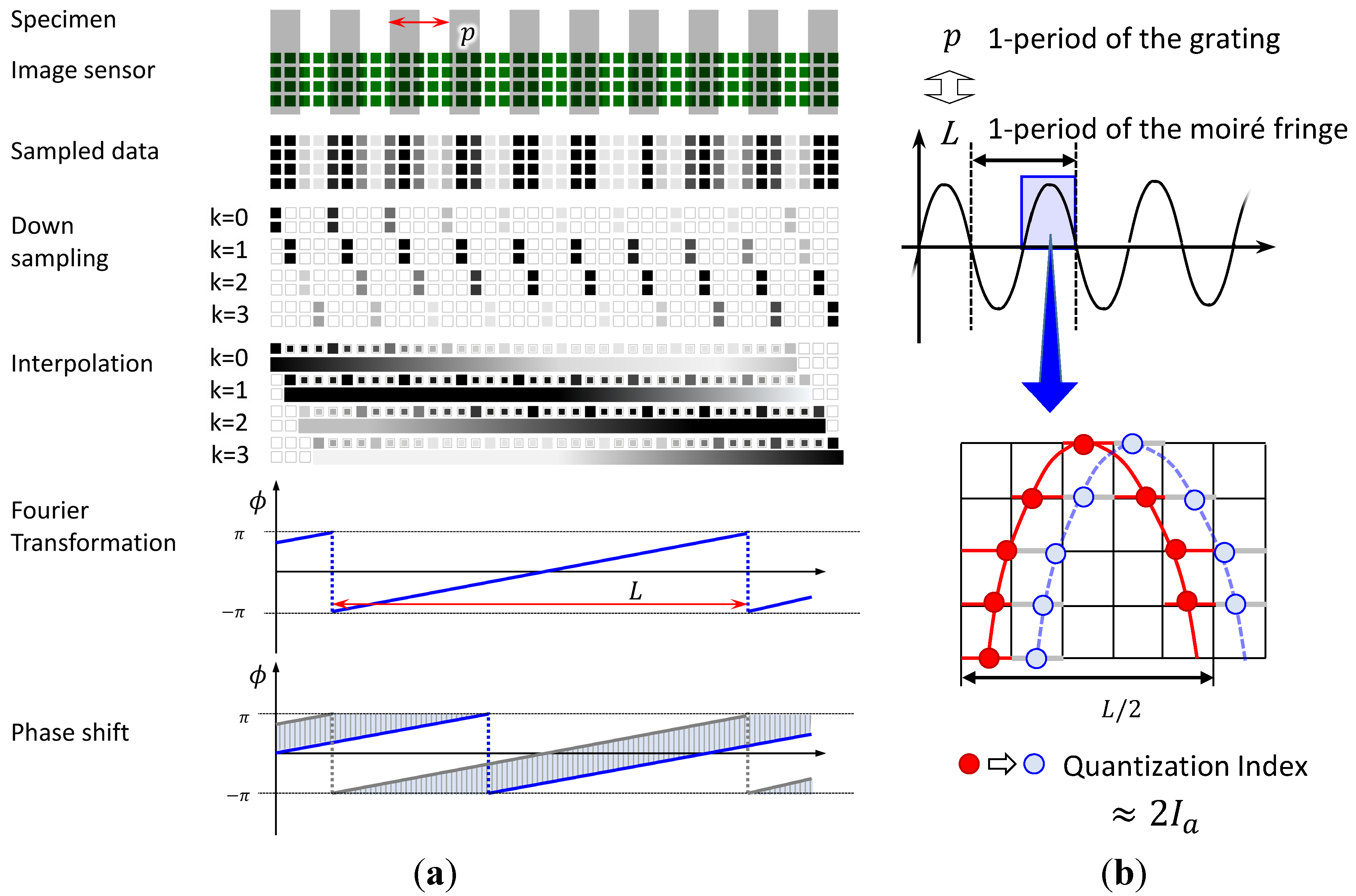
3.2. High-Resolution Sensing Method Using Phase Detection with Moiré Fringe
3.3. Calibration of the Force Sensor

4. Experiment and Result
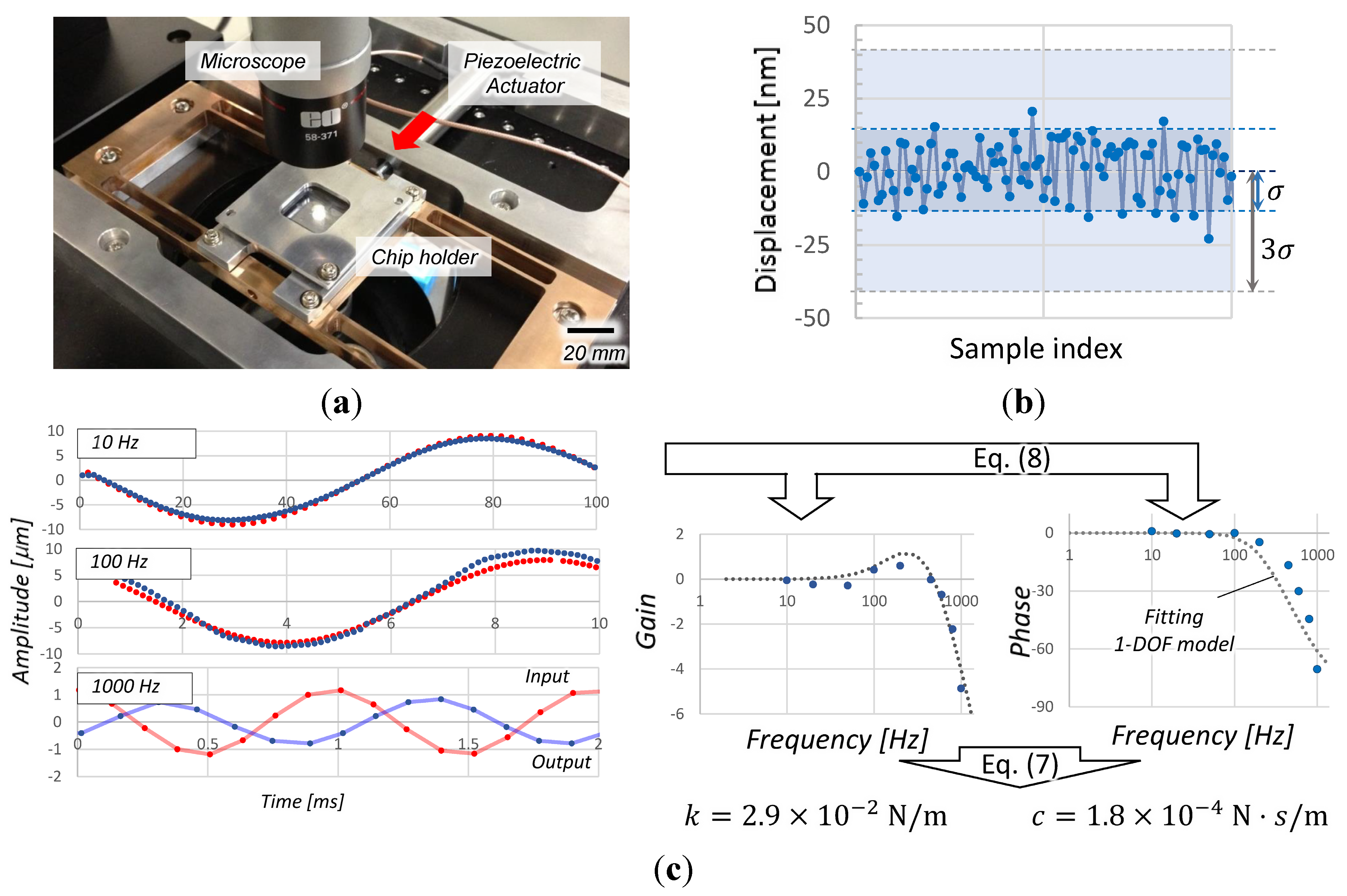
4.1. Performance of the Method
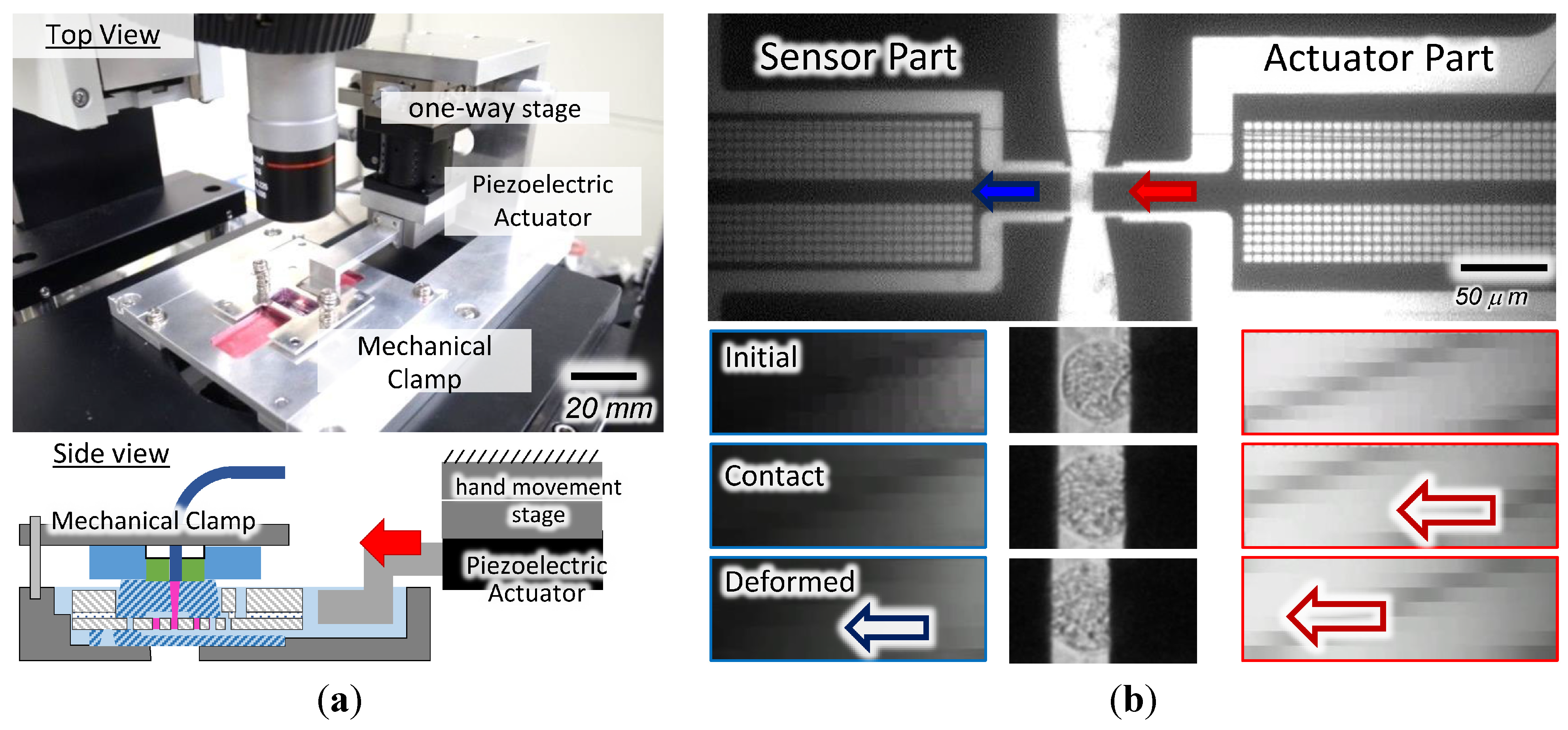
4.2. Cellular Characterization

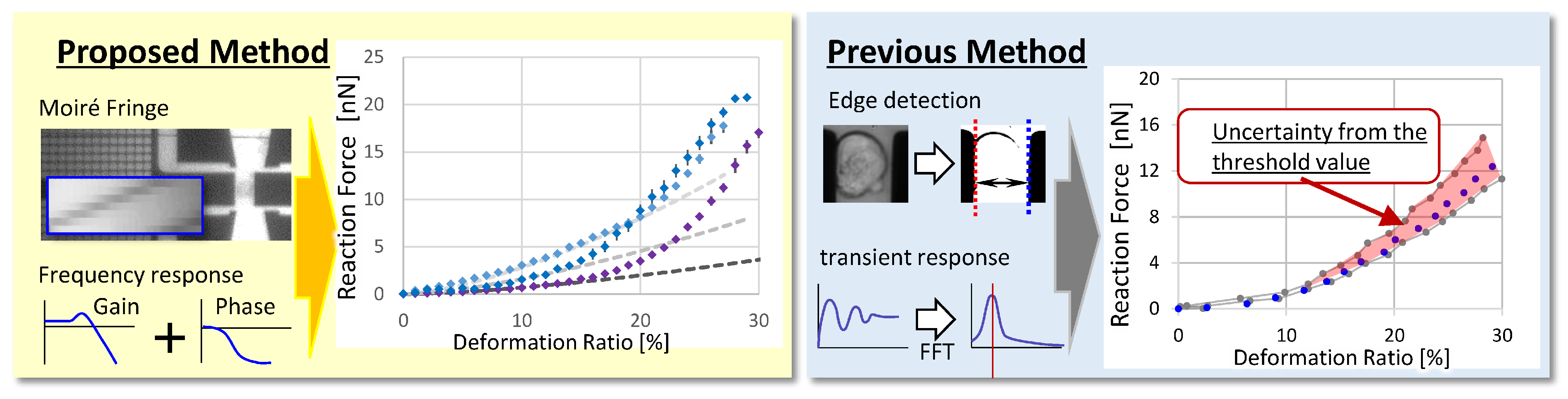
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Supplementary Materials
Conflicts of Interest
References
- Zheng, X.Y.; Zhang, X. Microsystems for cellular force measurement: A review. J. Micromech. Microeng. 2011, 21, 1–13. [Google Scholar] [CrossRef]
- Janmey, P.A.; McCulloch, C.A. Cell mechanics: Integrating cell responses to mechanical stimuli. Annu. Biomed. Eng. 2007, 9, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Miyanaga, N.; Akaza, H.; Hamakawa, M.; Shina, T. Tissue elasticity imaging for diagnosis of prostate cancer: A preliminary report. J. Urol. Res. 2004, 13, 1514–1518. [Google Scholar] [CrossRef] [PubMed]
- Beil, M.; Micouletm, A.; Whichert, O.; Seuferlien, T. Sphingosylphosphorylcholine regulates keratin network architecture and visco-elastic properties of human cancer cells. Nat. Cell Bio. 2003, 5, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Erin, B.; Jing, L.; Bonnecaze, Y.; Roger, T.; Muhammad, H. Cancer cell stiffness: Integrated roles of three-dimensional matrix stiffness and transforming potential. J Biophys. 2005, 99, 3689–3698. [Google Scholar]
- Manfred, R. Measuring the elastic properties of biological samples with the AFM. IEEE J. Transl. Eng. Health Med. Mag. 1997, 16, 47–57. [Google Scholar]
- Guilak, F.; Erickson, G.; Ting-Beall, H. The effects of osmotic stress on the viscoelastic and physical roperties of articular chondrocytes. J Biophys. 2002, 82, 720–729. [Google Scholar] [CrossRef]
- Takahashia, R.; Ichikawaa, S.; Subagyoa, A.; Sueokaa, K.; Okajima, T. Atomic force microscopy measurements of mechanical properties of single cells patterned by microcontact printing. J. Adv. Robot. 2014, 28, 449–455. [Google Scholar] [CrossRef]
- Zhang, Z.; Frerenczi, M.; Lush, A.; Thomas, C. A novel micromanipulation technique for measuring the bursting strength of single mammalian cells. Appl. Microbiol. Biotech. 1991, 36, 208–210. [Google Scholar] [CrossRef]
- Tran-Son-Tay, R.; Kirk, T.; Zhelev, D.; Hochmuth, R. Numerical simulation of the flow of highly viscous drops down a tapered tube. J. Biomech. Eng. 1994, 116, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, S.; Arai, F. Cellular force measurement using a nanometric-probe-integrated microfluidic chip with a displacement reduction mechanism. J. Mech. Robot. 2013, 25, 277–284. [Google Scholar]
- Kakio, T.; Bilal, T.; Sakuma, S.; Kaneko, M.; Arai, F. On-chip cellular force measurement using direct outer drive mechanism. In Proceedings of 2013 International Symposium on Micro-Nano Mechatronics and Human Science, Nagoya, Japan, 10–13 November 2013; pp. 261–262.
- Ziaie, B.; Baldi, A.; Lei, M.; Gu, Y.; Siegel, R.A. Hard and soft micromachining for BioMEMS: Review of techniques and examples of applications in microfluidics and drug delivery. Biosens. Drug Deliv. Microscale 2004, 56, 145–172. [Google Scholar] [CrossRef]
- Guelpa, V.; Laurent, G.; Sandoz, P.; Zea, J.; Clévy, C. Subpixelic measurement of large 1D displacements: Principle, processing algorithms, performances and software. Sensors 2014, 14, 5056–5057. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Wyant, J. Phase shifter calibration in phase-shifting interferometry. Appl. Opt. 1985, 24, 3049–3052. [Google Scholar] [CrossRef] [PubMed]
- Ri, S.; Fujigaki, M.; Morimoto, Y. Sampling moiré method for accurate small deformation distribution measurement. Exp. Mech. 2010, 50, 501–508. [Google Scholar] [CrossRef]
- Ai, C.; Wyant, J. Effect of piezoelectric transducer nonlinearity on phase shifted interferometry. Appl. Opt. 1987, 26, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Saarela, V.; Franssila, S.; Tuomikoski, S.; Marttila, S.; Osman, P.; Sikanen, T.; Koriaho, T.; Kostiainen, R. Re-usable multi-inlet, PDMS fluidic connector. Sens. Actuators B Chem. 2006, 114, 552–557. [Google Scholar] [CrossRef]
- Kogut, L.; Etsion, I. A Semi-Analytical Solution for the Sliding Inception of a Spherical Contact. J. Tribol. 2003, 125, 499–506. [Google Scholar] [CrossRef]
- Steletenkmap, S.; Rommelm, C.; Wegener, J.; Jansoff, A. Membrane stiffness of animal cells challenged by osmotic stress. Small 2006, 2, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.T.; Zhoua, E.H.; Quekb, S.T. Mechanical models for living cells. J. Biomech. 2006, 39, 195–216. [Google Scholar] [CrossRef] [PubMed]
- Ladjal, H.; Hanus, L.; Ferreria, A. Micro-to-nano biomechanical modeling for assisted biological cell injection. IEEE Trans. Biomed. Eng. 2013, 60, 2461–2471. [Google Scholar] [CrossRef] [PubMed]
- Tran-Son-Tay, R.; Kan, H.; Udaykumar, H.; Damay, E.; Shyy, W. Rheological modelling of leukocytes. Med. Biol. Eng. Comput. 1998, 36, 246–250. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugiura, H.; Sakuma, S.; Kaneko, M.; Arai, F. On-Chip Method to Measure Mechanical Characteristics of a Single Cell by Using Moiré Fringe. Micromachines 2015, 6, 660-673. https://doi.org/10.3390/mi6060660
Sugiura H, Sakuma S, Kaneko M, Arai F. On-Chip Method to Measure Mechanical Characteristics of a Single Cell by Using Moiré Fringe. Micromachines. 2015; 6(6):660-673. https://doi.org/10.3390/mi6060660
Chicago/Turabian StyleSugiura, Hirotaka, Shinya Sakuma, Makoto Kaneko, and Fumihito Arai. 2015. "On-Chip Method to Measure Mechanical Characteristics of a Single Cell by Using Moiré Fringe" Micromachines 6, no. 6: 660-673. https://doi.org/10.3390/mi6060660
APA StyleSugiura, H., Sakuma, S., Kaneko, M., & Arai, F. (2015). On-Chip Method to Measure Mechanical Characteristics of a Single Cell by Using Moiré Fringe. Micromachines, 6(6), 660-673. https://doi.org/10.3390/mi6060660