Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review
Abstract
1. Introduction
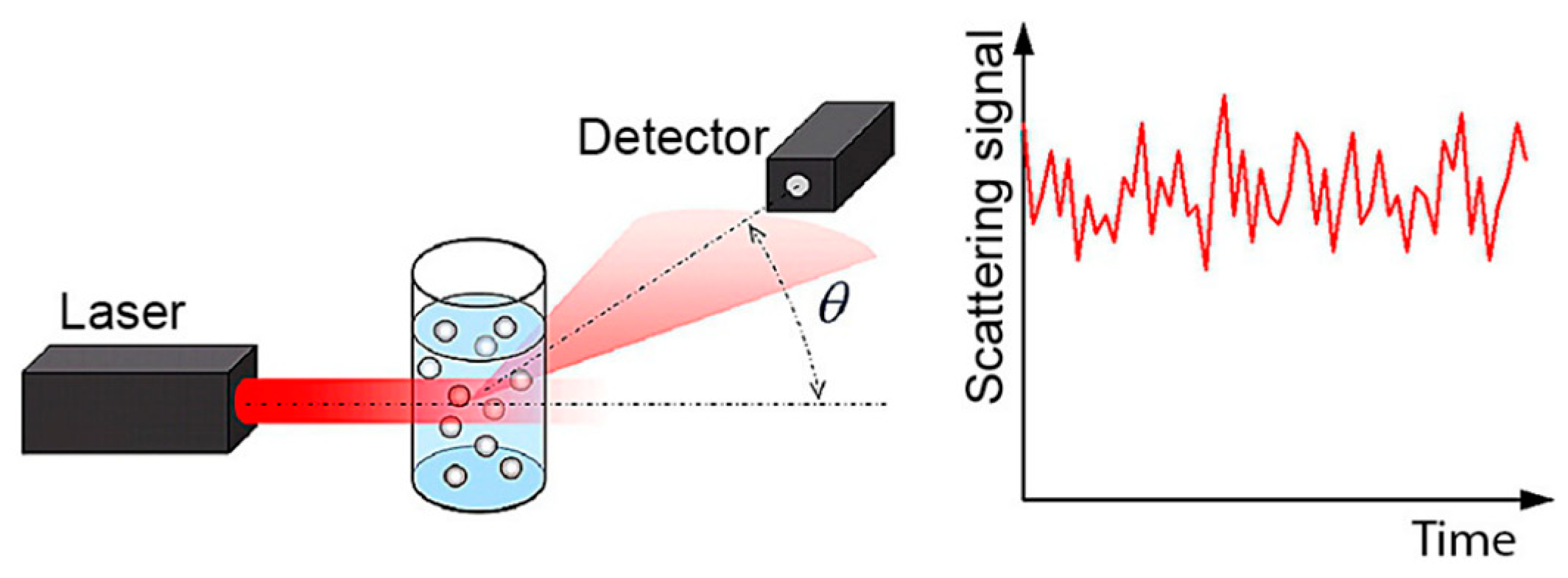
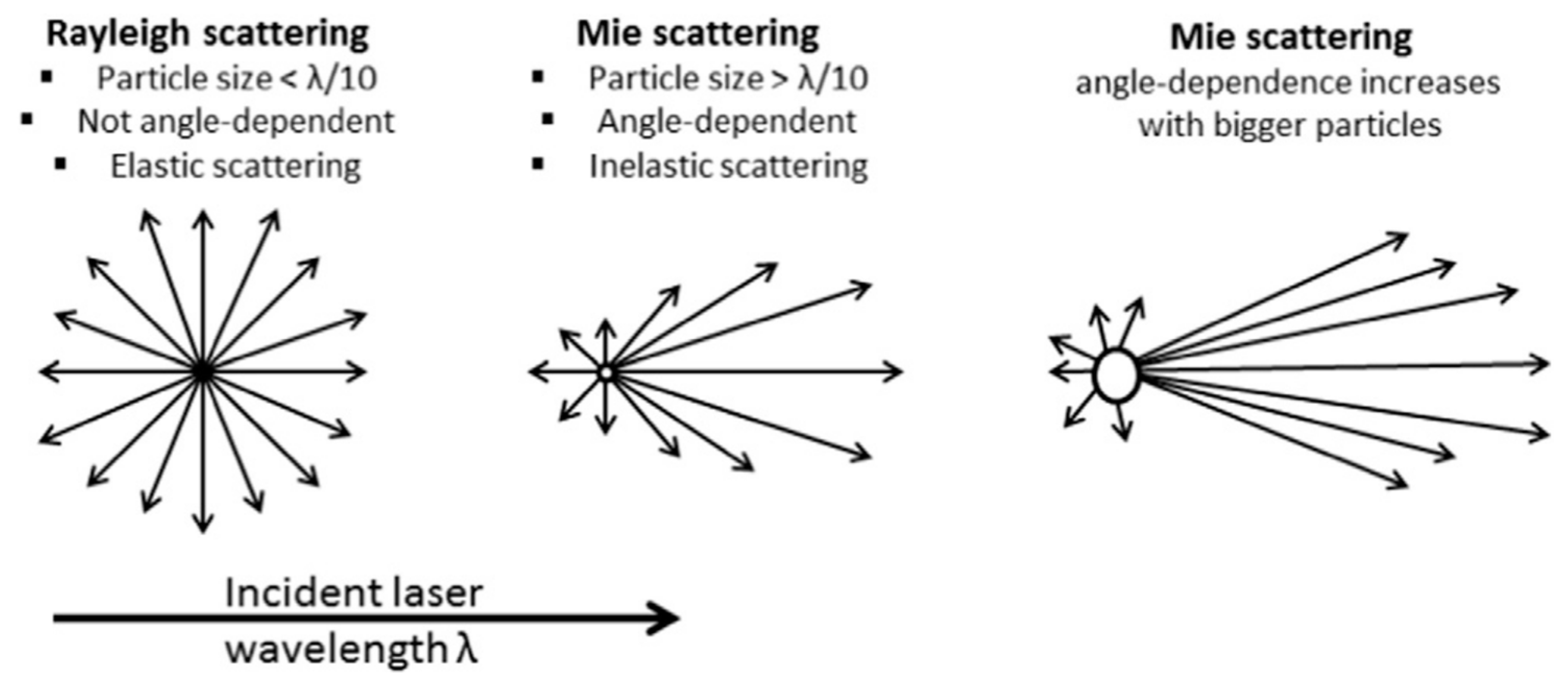
2. Dynamic Light Scattering: Particle Size, Zeta Potential, and Molecular Weight
2.1. Particle Size Theory
2.2. Molecular Weight Theory
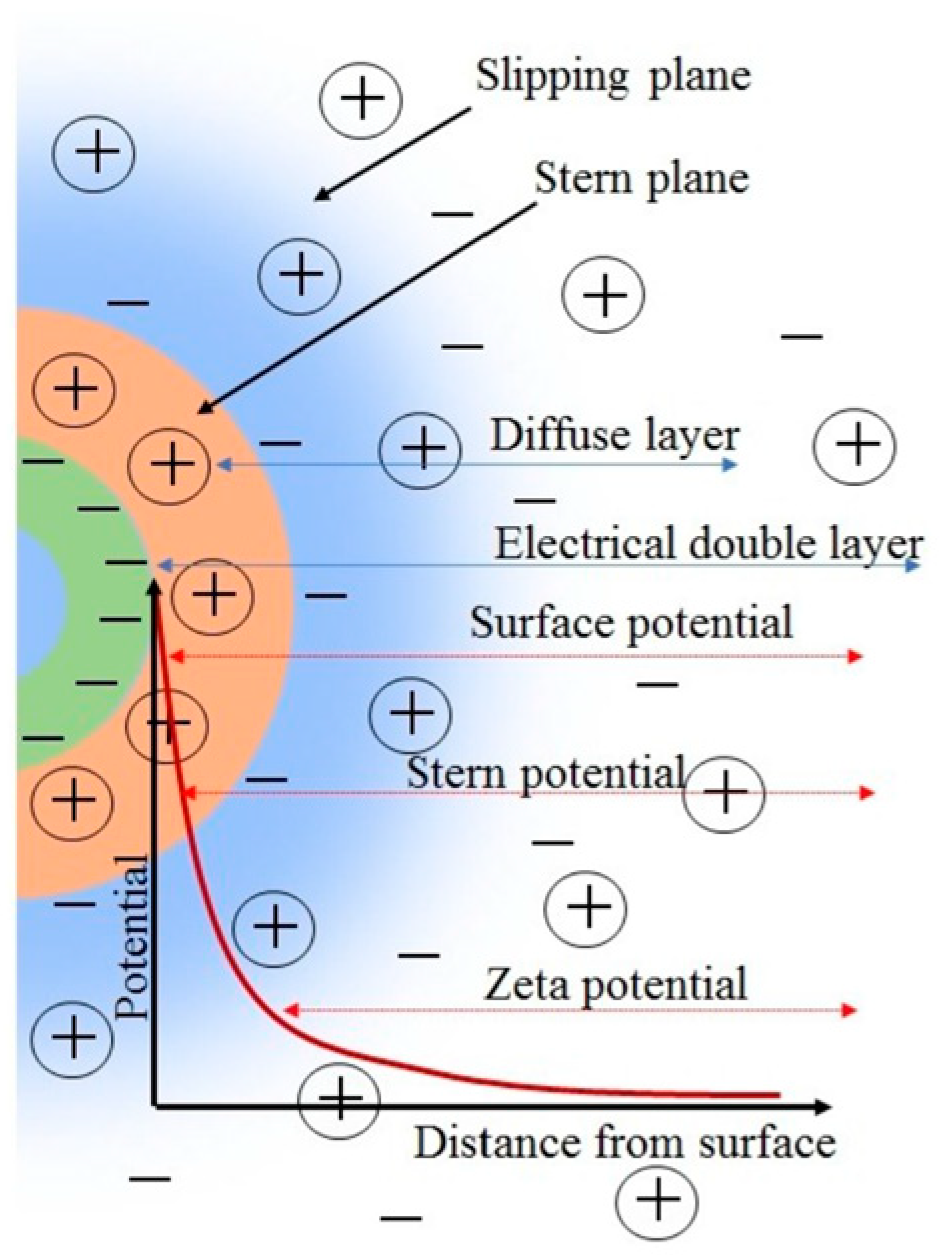
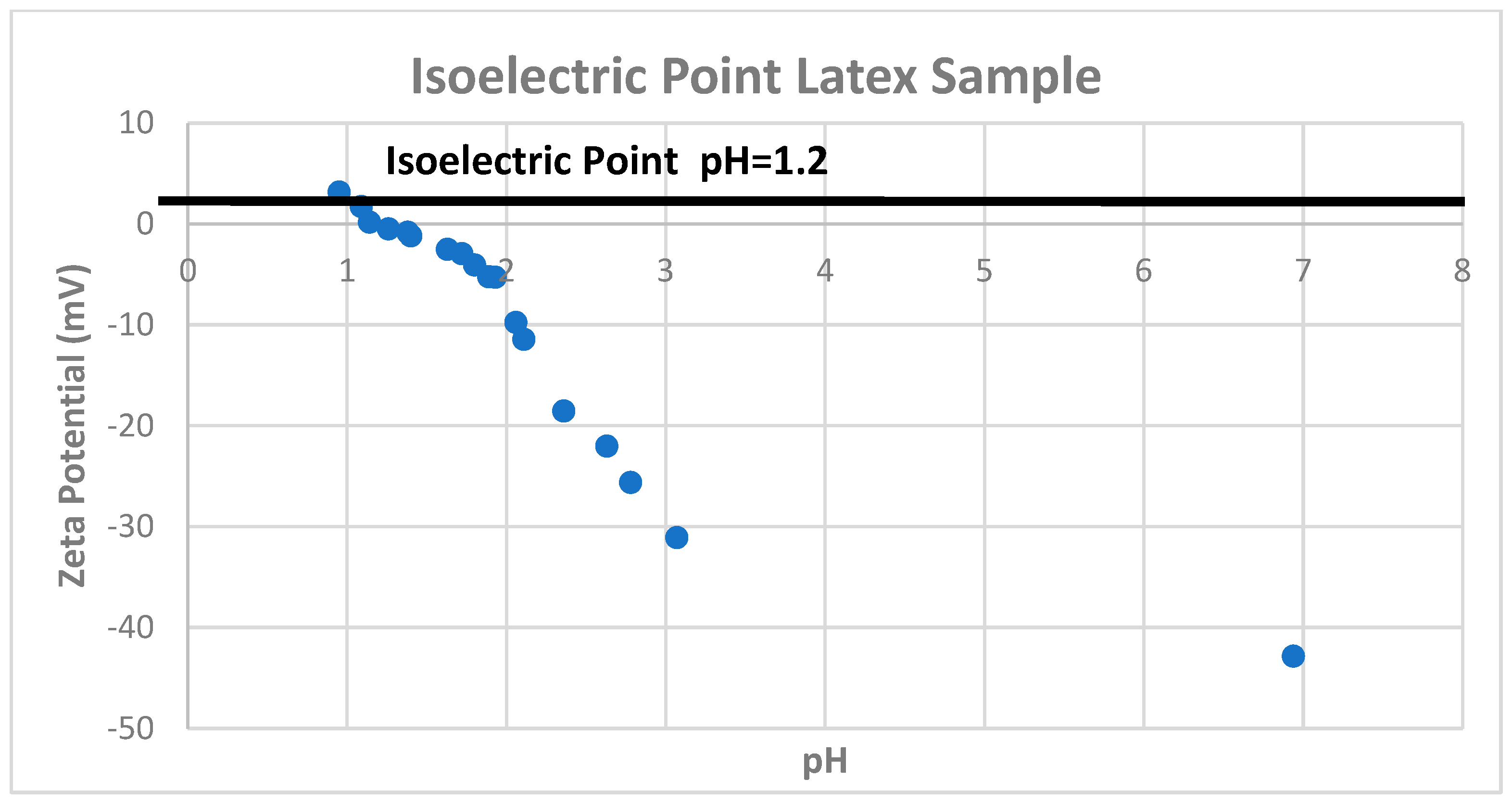
2.3. Zeta Potential Theory
3. DLS and Its Applications
3.1. Pharmaceutical Industry and Human Health
3.2. Material Science
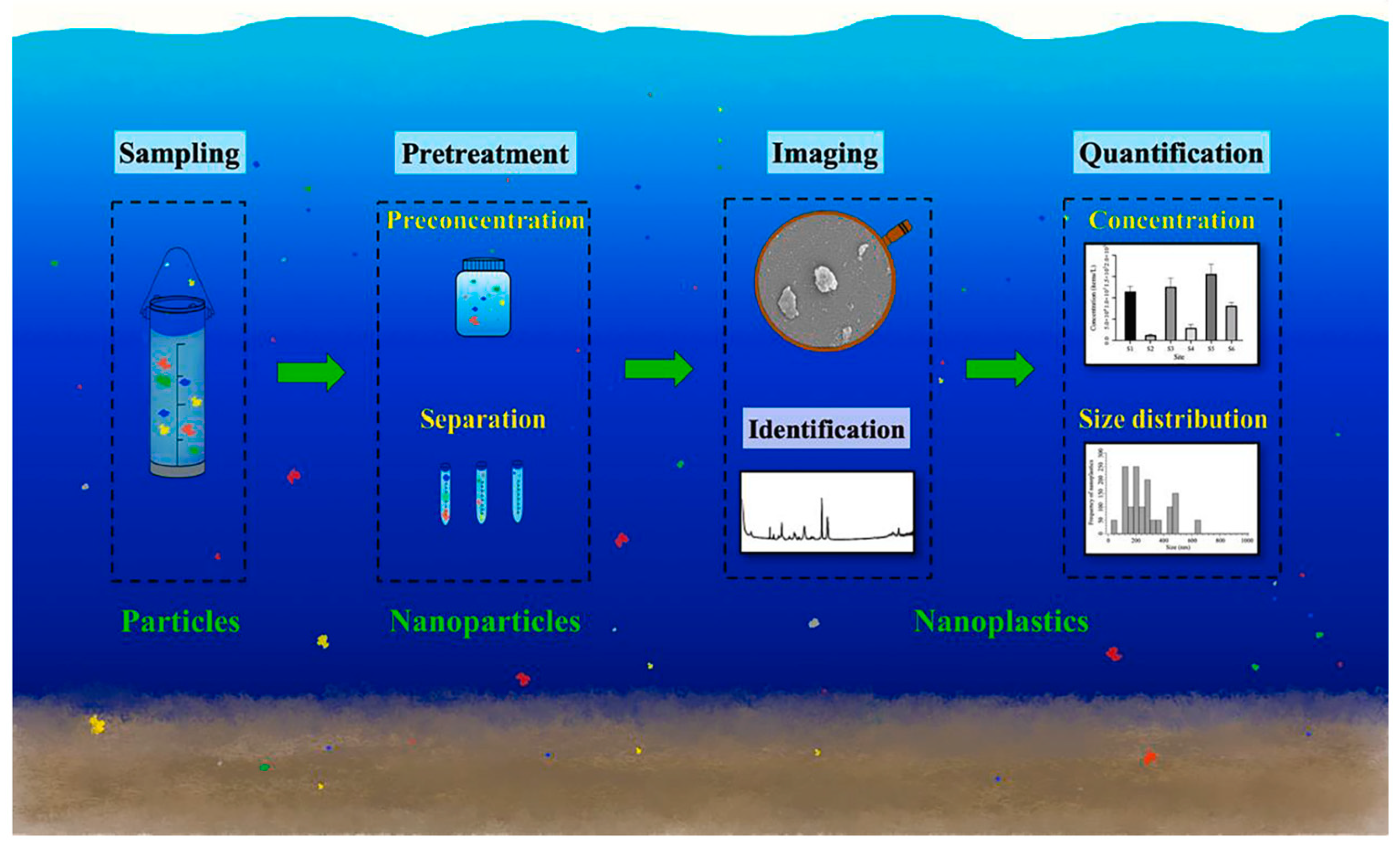
3.3. Environmental Protection, Remediation, and Toxicology
3.4. Food Science
3.5. Toners, Inks, and Pigments
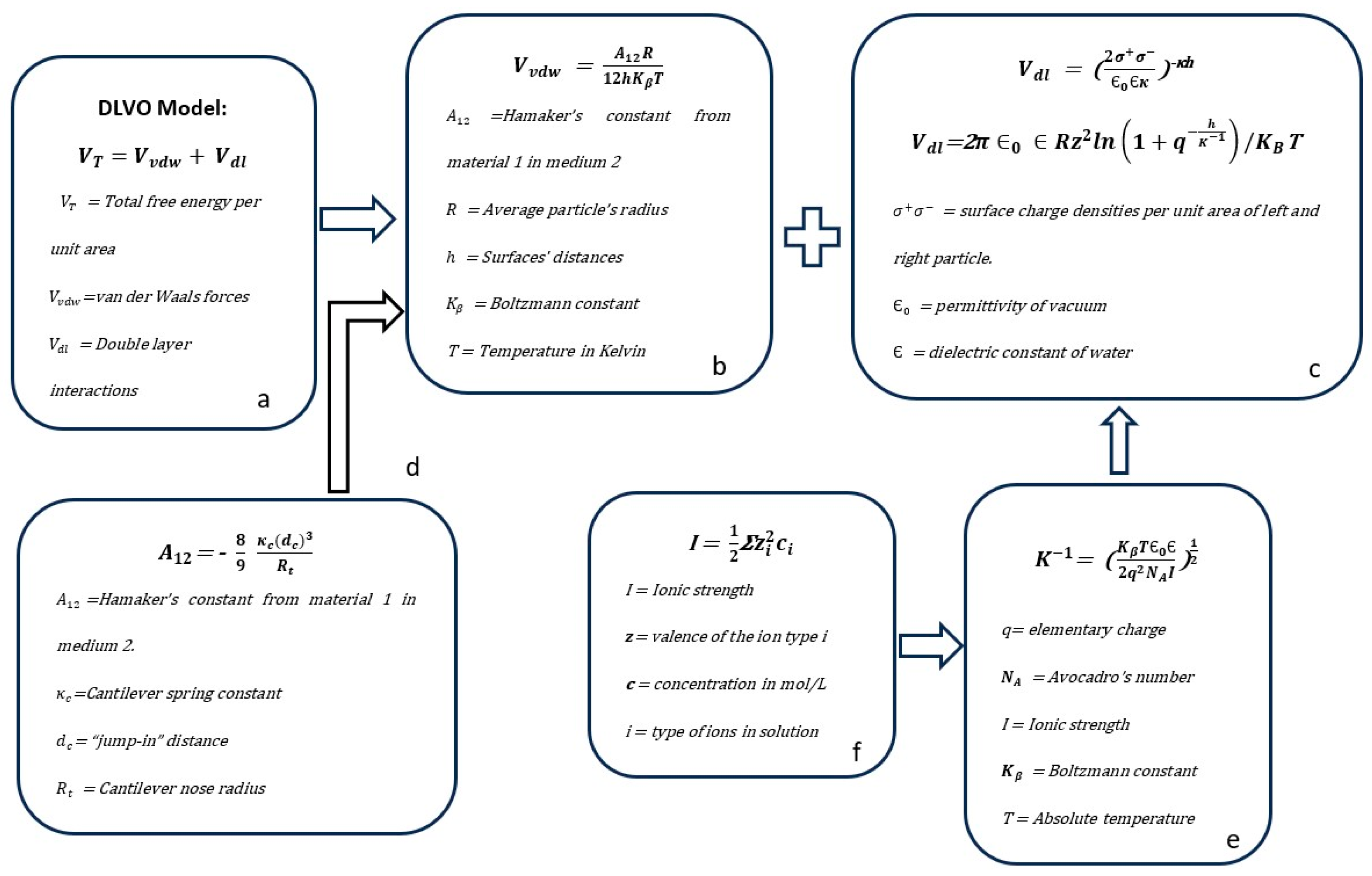
4. Particle Agglomeration Mechanisms and Their Control in Colloidal Systems
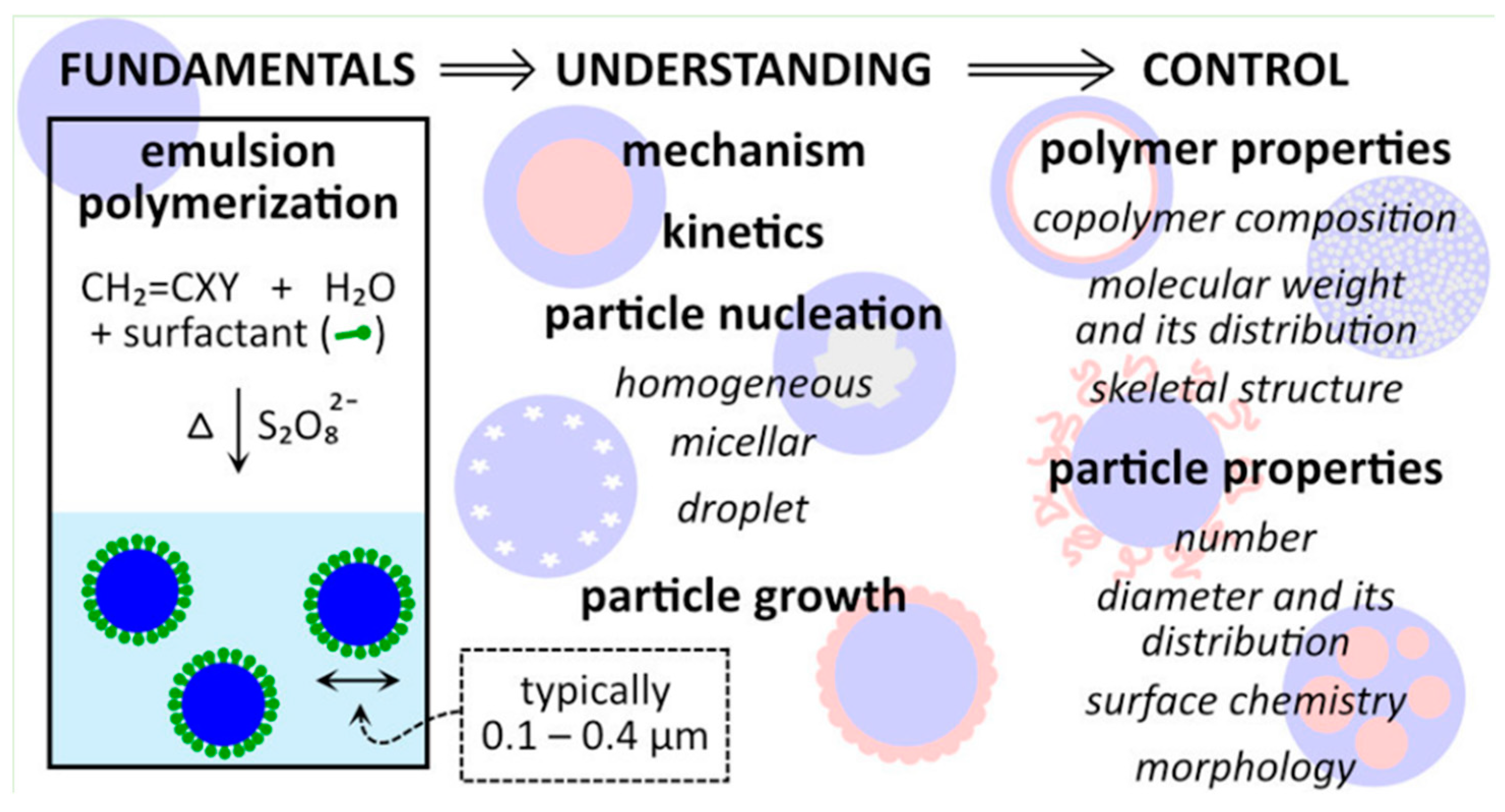
5. Particle Agglomeration Control in Chemically Processed Toner
- Ammonium persulfate and sodium metabisulfite. Ammonium persulfate is the oxidizing agent, while sodium metabisulfite is the reducing agent. The reaction between these two compounds generates free radicals that initiate polymerization.
- Cerium(IV) ammonium nitrate can be used in mini-emulsion polymerization. Cerium(IV) ammonium nitrate is the oxidizing agent and initiates polymerization at low temperatures [118].
- Hydrogen peroxide and ferrous sulfate can be used in mini-emulsion polymerization. Hydrogen peroxide acts as the reducing agent [118].
6. Conclusions and Future Remarks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dhont, J.K.G. An Introduction to Dynamics of Colloids; Elsevier Science: Amsterdam, The Netherlands, 1996; ISBN 0444820094. [Google Scholar]
- Linko, V.; Zhang, H.; Nonappa; Kostiainen, M.A.; Ikkala, O. From Precision Colloidal Hybrid Materials to Advanced Functional Assemblies. Acc. Chem. Res. 2022, 55, 1785–1795. [Google Scholar] [CrossRef]
- Aguirre, M.; Ballard, N.; Gonzalez, E.; Hamzehlou, S.; Sardon, H.; Calderon, M.; Paulis, M.; Tomovska, R.; Dupin, D.; Bean, R.H.; et al. Polymer Colloids: Current Challenges, Emerging Applications, and New Developments. Macromolecules 2023, 56, 2579–2607. [Google Scholar] [CrossRef]
- O’ Brien, C. A Mathematical Model for Colloidal Aggregation. Master’s Thesis, University of South Florida, Tampa, FL, USA, 2003. [Google Scholar]
- Daniels, E.S.; Sudol, E.D.; El-Aasser, M.S. Overview of Polymer Colloids: Preparation, Characterization, and Applications; The American Chemical Society: Washington, DC, USA, 2002. [Google Scholar]
- Garti, N.; Yuli-Amar, I. Delivery and Controlled Release of Bioactives in Foods and Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2008; ISBN 9781845691455. [Google Scholar]
- Abdullah; Liu, L.; Javed, H.U.; Xiao, J. Engineering Emulsion Gels as Functional Colloids Emphasizing Food Applications: A Review. Front. Nutr. 2022, 9, 890188. [Google Scholar] [CrossRef]
- McClements, D.J. The Future of Food Colloids: Next-Generation Nanoparticle Delivery Systems. Curr. Opin. Colloid. Interface Sci. 2017, 28, 7–14. [Google Scholar] [CrossRef]
- Patel, A.R. Functional and Engineered Colloids from Edible Materials for Emerging Applications in Designing the Food of the Future. Adv. Funct. Mater. 2020, 30, 1806809. [Google Scholar] [CrossRef]
- Pradeep, S.; Nabizadeh, M.; Jacob, A.R.; Jamali, S.; Hsiao, L.C. Jamming Distance Dictates Colloidal Shear Thickening. Phys. Rev. Lett. 2021, 127, 158002. [Google Scholar] [CrossRef]
- Jin, L.; Jarand, C.W.; Brader, M.L.; Reed, W.F. Angle-Dependent Effects in DLS Measurements of Polydisperse Particles. Meas. Sci. Technol. 2022, 33, 045202. [Google Scholar] [CrossRef]
- Matijevic, E. Medical Applications of Colloids; Springer: New York, NY, USA, 2008; ISBN 978-0-387-76920-2. [Google Scholar] [CrossRef]
- Zhou, C.; Qi, W.; Lewis, E.N.; Carpenter, J.F. Concomitant Raman Spectroscopy and Dynamic Light Scattering for Characterization of Therapeutic Proteins at High Concentrations. Anal. Biochem. 2015, 472, 7–20. [Google Scholar] [CrossRef]
- Dauer, K.; Pfeiffer-Marek, S.; Kamm, W.; Wagner, K.G. Microwell Plate-Based Dynamic Light Scattering as a High-Throughput Characterization Tool in Biopharmaceutical Development. Pharmaceutics 2021, 13, 172. [Google Scholar] [CrossRef]
- Jia, Z.; Li, J.; Gao, L.; Yang, D.; Kanaev, A. Dynamic Light Scattering: A Powerful Tool for In Situ Nanoparticle Sizing. Colloids Interfaces 2023, 7, 15. [Google Scholar] [CrossRef]
- Meyer, R.M.; Aleshkevich, S.; Berger, L.; Nerkamp, J.; Scheler, S.; Friess, W. Characterization of the Aggregation Propensity of Charge Variants of Recombinant Human Growth Hormone. Int. J. Pharm. 2022, 621, 121760. [Google Scholar] [CrossRef]
- Brunelli, A.; Foscari, A.; Basei, G.; Lusvardi, G.; Bettiol, C.; Semenzin, E.; Marcomini, A.; Badetti, E. Colloidal Stability Classification of TiO2 Nanoparticles in Artificial and in Natural Waters by Cluster Analysis and a Global Stability Index: Influence of Standard and Natural Colloidal Particles. Sci. Total Environ. 2022, 829, 154658. [Google Scholar] [CrossRef]
- Guo, Y.; Tang, N.; Guo, J.; Lu, L.; Li, N.; Hu, T.; Zhu, Z.; Gao, X.; Li, X.; Jiang, L.; et al. The Aggregation of Natural Inorganic Colloids in Aqueous Environment: A Review. Chemosphere 2023, 310, 136805. [Google Scholar] [CrossRef]
- Du, Q.; Zhou, L.; Lyu, F.; Liu, J.; Ding, Y. The Complex of Whey Protein and Pectin: Interactions, Functional Properties and Applications in Food Colloidal Systems–A Review. Colloids Surf. B Biointerfaces 2022, 210, 112253. [Google Scholar] [CrossRef]
- Lorenzo, T.; Marco, L. Brownian Dynamics Simulations of Shear-Induced Aggregation of Charged Colloidal Particles in the Presence of Hydrodynamic Interactions. J. Colloid Interface Sci. 2022, 624, 637–649. [Google Scholar] [CrossRef]
- Zeng, X.; Nyquist, Y.; Zhang, Q.; Butt, H.-J.; Wu, S. Light-Induced Assembly of Colloidal Nanoparticles Based on Photo-Controlled Metal–Ligand Coordination. Supramol. Mater. 2022, 1, 100004. [Google Scholar] [CrossRef]
- Israelachvili, J. Intermolecular and Surface Forces; Elsevier: Amsterdam, The Netherlands, 2011; ISBN 9780123751829. [Google Scholar]
- Hiemenz, P.C.R.R. Principles of Colloid and Surface Chemistry; Taylor & Francis Ltd.: London, UK, 1997; ISBN 0824793978. [Google Scholar]
- Hoek, E.M.V.; Agarwal, G.K. Extended DLVO Interactions between Spherical Particles and Rough Surfaces. J. Colloid Interface Sci. 2006, 298, 50–58. [Google Scholar] [CrossRef]
- Bendersky, M.; Davis, J.M. DLVO Interaction of Colloidal Particles with Topographically and Chemically Heterogeneous Surfaces. J. Colloid Interface Sci. 2011, 353, 87–97. [Google Scholar] [CrossRef]
- Carroll, J.B.; Chase, J.; Truong, T.; Kyrlidis, A.; Bedford, E.; Corporation, C. Surface Modified Pigments for Improved Color in Chemically Prepared Toners. Direct 2009, 25, 39–42. [Google Scholar] [CrossRef]
- Trefalt, G.; Behrens, S.H.; Borkovec, M. Charge Regulation in the Electrical Double Layer: Ion Adsorption and Surface Interactions. Langmuir 2016, 32, 380–400. [Google Scholar] [CrossRef]
- Holmberg, J.P.; Ahlberg, E.; Bergenholtz, J.; Hassellöv, M.; Abbas, Z. Surface Charge and Interfacial Potential of Titanium Dioxide Nanoparticles: Experimental and Theoretical Investigations. J Colloid Interface Sci. 2013, 407, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Czarnecki, J. The Effects of Surface Inhomogeneities on the Interactions in Colloidal Systems and Colloid Stability. Adv. Colloid Interface Sci. 1985, 24, 283–319. [Google Scholar] [CrossRef]
- Montes Ruiz-Cabello, F.J.; Trefalt, G.; Maroni, P.; Borkovec, M. Electric Double-Layer Potentials and Surface Regulation Properties Measured by Colloidal-Probe Atomic Force Microscopy. Phys. Rev. E Stat. Nonlin Soft Matter Phys. 2014, 90, 012301. [Google Scholar] [CrossRef] [PubMed]
- Bazrafshan, Z.; Ataeefard, M.; Nourmohammadian, F. Modeling the Effect of Pigments and Processing Parameters in Polymeric Composite for Printing Ink Application Using the Response Surface Methodology. Prog. Org. Coat. 2015, 82, 68–73. [Google Scholar] [CrossRef]
- Senevirathne, I.C.; Kizilkaya, O.; Kurtz, R.L.; Sprunger, P.T. Nucleation, Growth and Morphology of Co Thin Films on Ag(110). Surf. Sci. 2009, 603, 2463–2468. [Google Scholar] [CrossRef]
- Butt, H.J.; Cappella, B.; Kappl, M. Force Measurements with the Atomic Force Microscope: Technique, Interpretation and Applications. Surf. Sci. Rep. 2005, 59, 1–152. [Google Scholar] [CrossRef]
- Trefalt, G.; Borkovec, M. Overview of DLVO Theory; Laboratory of Colloid and Surface Chemistry, University of Genebra: Geneva, Switzerland, 2014; Volume 304, pp. 1–10. [Google Scholar]
- Jang, M.H.; Kim, M.S.; Han, M.; Kwak, D.H. Experimental Application of a Zero-Point Charge Based on pH as a Simple Indicator of Microplastic Particle Aggregation. Chemosphere 2022, 299, 134388. [Google Scholar] [CrossRef]
- Schenkel, C.D.; Sorichetti, P.A.; Romano, S.D. ELECTRODOS INTERCAMBIABLES PARA MEDIR PROPIEDADES DIELÉCTRICAS EN LÍQUIDOS. ANALES AFA 2006, 17, 1. [Google Scholar]
- Aschauer, U.; Burgos-Montes, O.; Moreno, R.; Bowen, P. Hamaker 2: A Toolkit for the Calculation of Particle Interactions and Suspension Stability and Its Application to Mullite Synthesis by Colloidal Methods. J. Dispers. Sci. Technol. 2011, 32, 470–479. [Google Scholar] [CrossRef]
- Guaregua, J.A.; Squitieri, E.; Mujica, V. A General Algorithm Using Mathematica 2.0 for Calculting Dlvo Potential Interactions and Stability Ratios in Spherical Colloidal Particle Systems. J. Comput. Method. Sci. Eng. 2009, 9, 223–240. [Google Scholar] [CrossRef]
- Messaâdi, A.; Dhouibi, N.; Hamda, H.; Bin, F.; Belgacem, M.; Adbelkader, Y.H.; Ouerfelli, N.; Hamzaoui, A.H. A New Equation Relating the Viscosity Arrhenius Temperature and the Activation Energy for Some Newtonian Classical Solvents. J. Chen. 2015, 2015, 163262. [Google Scholar] [CrossRef]
- Dembek, M.; Bocian, S.; Buszewski, B. Solvent Influence on Zeta Potential of Stationary Phase—Mobile Phase Interface. Molecules 2022, 27, 968. [Google Scholar] [CrossRef] [PubMed]
- Kramb, R.C. The Effects of Particle Shape, Size, and Interaction on Colloidal Glasses and Gels. Ph.D. Thesis, the Graduate College of the University of Illinois, Champaign, IL, USA, 2010. [Google Scholar]
- Kinsyo, T.; Nakanishi, H.; Hirai, K.; Noda, H.; Takikawa, T.; Yahiro, S. Development of Polyester Resin Particles for Toner with a Controlled Particle Size Distribution and Shape. Polym. J. 2017, 49, 593–600. [Google Scholar] [CrossRef]
- Pan, C.; Mahmoudabadbozchelou, M.; Duan, X.; Benneyan, J.C.; Jamali, S.; Erb, R.M. Increasing Efficiency and Accuracy of Magnetic Interaction Calculations in Colloidal Simulation through Machine Learning. J. Colloid Interface Sci. 2022, 611, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Takae, K.; Tanaka, H. Impact of Inverse Squeezing Flow on the Self-Assembly of Oppositely Charged Colloidal Particles under Electric Field. Phys. Rev. Lett. 2022, 129, 248001. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, B.Y.; Yon, K.-Y. Aggregation Behaviors of Colloidal Particles for Production of Polyester-Based Chemically Prepared Toner. J. Imaging Technol. 2013, 57, 352–355. [Google Scholar] [CrossRef]
- Nakouzi, E.; Kerisit, S.; Legg, B.A.; Yadav, S.; Li, D.; Stack, A.G.; Mundy, C.J.; Chun, J.; Schenter, G.K.; De Yoreo, J.J. Solution Structure and Hydration Forces between Mica and Hydrophilic Versus Hydrophobic Surfaces. The J. Phys. Chem. C 2023, 127, 2741–2752. [Google Scholar] [CrossRef]
- Shrestha, S.; Wang, B.; Dutta, P. Nanoparticle Processing: Understanding and Controlling Aggregation. Adv. Colloid Interface Sci. 2020, 279, 102162. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Lee, D. Making Waxy Salts in Water: Synthetic Control of Hydrophobicity for Anion-Induced and Aggregation-Enhanced Light Emission. Angew. Chem. Int. 2021, 60, 10858–10864. [Google Scholar] [CrossRef]
- Fukunaga, Y.; Miyagawa, A.; Harada, M.; Okada, T. Freeze Control of Nanoparticle Aggregation and Exploration as Surface Enhanced Raman Scattering (SERS) Platform. ChemNanoMat 2021, 7, 434–438. [Google Scholar] [CrossRef]
- Zhao, Q.; Shen, C.; Evans, C. Molecular Design of Precise Network Polymerized Ionic Liquids to Control Aggregation and Conductivity. APS March Meet. Abstr. 2019, 2019, R50-002. [Google Scholar]
- Mylon, S.E.; Rinciog, C.I.; Schmidt, N.; Gutierrez, L.; Wong, G.C.L.; Nguyen, T.H. Influence of Salts and Natural Organic Matter on the Stability of Bacteriophage MS2. Langmuir 2010, 26, 1035–1042. [Google Scholar] [CrossRef]
- Ferri, G.; Humbert, S.; Digne, M.; Schweitzer, J.-M.; Moreaud, M.; Schweitzer, J. Simulation of Large Aggregate Particles System with a New Morphological Model. Image Anal. Stereol. 2021, 40, 71–84. [Google Scholar] [CrossRef]
- Wang, F.; Kumar, R.N.; Prasannakumara, B.C.; Khan, U.; Zaib, A.; Abdel-Aty, A.H.; Yahia, I.S.; Alqahtani, M.S.; Galal, A.M. Aspects of Uniform Horizontal Magnetic Field and Nanoparticle Aggregation in the Flow of Nanofluid with Melting Heat Transfer. Nanomaterials 2022, 12, 1000. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, C.M.; Ilavsky, J.; Martel, A.; Hinrichs, S.; Meyer, A.; Pedersen, J.S.; Sokolova, A.V.; Svergun, D.I. Small-Angle X-ray and Neutron Scattering. Nat. Rev. Methods Primers 2021, 1, 70. [Google Scholar] [CrossRef]
- Li, Y.; Lubchenko, V.; Vekilov, P.G. The Use of Dynamic Light Scattering and Brownian Microscopy to Characterize Protein Aggregation. Rev.Sci. Instrum. 2011, 82, 053106. [Google Scholar] [CrossRef] [PubMed]
- Malm, A.V.; Corbett, J.C.W. Improved Dynamic Light Scattering Using an Adaptive and Statistically Driven Time Resolved Treatment of Correlation Data. Sci. Rep. 2019, 9, 13519. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, I.; Szabo, T.; Desert, A.; Trefalt, G.; Oncsik, T.; Borkovec, M. Particle Aggregation Mechanisms in Ionic Liquids. Phys. Chem. Chem. Phys. 2014, 16, 9515–9524. [Google Scholar] [CrossRef]
- Mihai, M.; Dragan, E.S.; Schwarz, S.; Janke, A. Dependency of Particle Sizes and Colloidal Stability of Polyelectrolyte Complex Dispersions on Polyanion Structure and Preparation Mode Investigated by Dynamic Light Scattering and Atomic Force Microscopy. J. Phys. Chem. B 2007, 111, 8668–8675. [Google Scholar] [CrossRef]
- Rottereau, M.; Gimel, J.C.; Nicolai, T.; Durand, D. Monte Carlo Simulation of Particle Aggregation and Gelation: I. Growth, Structure and Size Distribution of the Clusters. Eur. Phys. J. E 2004, 15, 133–140. [Google Scholar] [CrossRef]
- Pesmazoglou, I.; Kempf, A.M.; Navarro-Martinez, S. Stochastic Modelling of Particle Aggregation. Int. J. Multiph. Flow 2016, 80, 118–130. [Google Scholar] [CrossRef]
- Timokhin, I.V.; Matveev, S.A.; Siddharth, N.; Tyrtyshnikov, E.E.; Smirnov, A.P.; Brilliantov, N.V. Newton Method for Stationary and Quasi-Stationary Problems for Smoluchowski-Type Equations. J. Comput Phys. 2019, 382, 124–137. [Google Scholar] [CrossRef]
- Tandon, P.; Rosner, D.E. Monte Carlo Simulation of Particle Aggregation and Simultaneous Restructuring. J. Colloid Interface Sci. 1999, 213, 273–286. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, R.; Wang, Y.; Yu, Y.X. Molecular Dynamics Simulations of Stability at the Early Stages of Silica Materials Preparation. J. Mol. Struct. 2017, 1138, 198–207. [Google Scholar] [CrossRef]
- Hou, D.; Yu, J.; Wang, P. Molecular Dynamics Modeling of the Structure, Dynamics, Energetics and Mechanical Properties of Cement-Polymer Nanocomposite. Compos. B Eng. 2019, 162, 433–444. [Google Scholar] [CrossRef]
- Li, K.; Li, N.; Yan, N.; Wang, T.; Zhang, Y.; Song, Q.; Li, H. Adsorption of Small Hydrocarbons on Pristine, N-Doped and Vacancy Graphene by DFT Study. Appl. Surf. Sci. 2020, 515, 146028. [Google Scholar] [CrossRef]
- Pelalak, R.; Soltani, R.; Heidari, Z.; Malekshah, R.E.; Aallaei, M.; Marjani, A.; Rezakazemi, M.; Kurniawan, T.A.; Shirazian, S. Molecular Dynamics Simulation of Novel Diamino-Functionalized Hollow Mesosilica Spheres for Adsorption of Dyes from Synthetic Wastewater. J. Mol. Liq. 2021, 322, 114812. [Google Scholar] [CrossRef]
- Yaphary, Y.L.; Yu, Z.; Lam, R.H.W.; Hui, D.; Lau, D. Molecular Dynamics Simulations on Adhesion of Epoxy-Silica Interface in Salt Environment. Compos. B Eng. 2017, 131, 165–172. [Google Scholar] [CrossRef]
- O’Kane, M.E.; Smith, J.A.; Kilbride, R.C.; Spooner, E.L.K.; Duif, C.P.; Catley, T.E.; Washington, A.L.; King, S.M.; Parnell, S.R.; Parnell, A.J. Exploring Nanoscale Structure in Perovskite Precursor Solutions Using Neutron and Light Scattering. Chem. Mater. 2022, 34, 7232–7241. [Google Scholar] [CrossRef]
- Gommes, C.J.; Jaksch, S.; Frielinghaus, H. Small-Angle Scattering for Beginners. J. App. Crystallogr. 2021, 54, 1832–1843. [Google Scholar] [CrossRef]
- King, S.M.; Jarvie, H.P. Exploring How Organic Matter Controls Structural Transformations in Natural Aquatic Nanocolloidal Dispersions. Environ. Sci. Technol. 2012, 46, 6959–6967. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frungieri, G.; Vanni, M. Aggregation and Breakup of Colloidal Particle Aggregates in Shear Flow: A Combined Monte Carlo-Stokesian Dynamics Approach. Powder Technol. 2021, 388, 357–370. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Babick, F. Dynamic Light Scattering (DLS). In Characterization of Nanoparticles: Measurement Processes for Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2019; pp. 137–172. ISBN 9780128141830. [Google Scholar] [CrossRef]
- Michaelides, E.E. Brownian Movement and Thermophoresis of Nanoparticles in Liquids. Int. J. Heat. Mass. Transf. 2015, 81, 179–187. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and Zeta Potential—What They Are and What They Are Not? J. Control. Rel. 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Kamble, S.; Agrawal, S.; Cherumukkil, S.; Sharma, V.; Jasra, R.V.; Munshi, P. Revisiting Zeta Potential, the Key Feature of Interfacial Phenomena, with Applications and Recent Advancements. ChemistrySelect 2022, 7. [Google Scholar] [CrossRef]
- Pereira, A.S.; Tavares, P.; Limão-Vieira, P. Radiation in Bioanalysis: Spectroscopic Techniques and Theoretical Methods; Springer International Publishing: Cham, Switzerland, 2019; Volume 8, ISBN 978-3-030-28247-9. [Google Scholar]
- Russo, P.S.; Streletzky, K.A.; Huberty, W.; Zhang, X.; Edwin, N. Characterization of Polymers by Static Light Scattering; Elsevier: Amsterdam, The Netherlands, 2021; ISBN 9780128197684. [Google Scholar]
- López-Maldonado, E.A.; Oropeza-Guzmán, M.T.; Ochoa-Terán, A. Improving the Efficiency of a Coagulation-Flocculation Wastewater Treatment of the Semiconductor Industry through Zeta Potential Measurements. J. Chem. 2014, 2014, 969720. [Google Scholar] [CrossRef]
- Bodycomb, J. (HORIBA) Horiba Scientific: Introduction to Zeta Potential Analysis. Available online: https://www.horiba.com/gbr/particlewebinars/meeting-green-goals-with-zeta-potential-and-the-sz-100/ (accessed on 25 October 2023).
- Xie, Z. Heat Transfer of Power-Law Fluids with Slip-Dependent Zeta Potential. Colloids Surf. A Physicochem. Eng. Asp. 2022, 651, 129710. [Google Scholar] [CrossRef]
- Gittings, M.R.; Saville, D.A. The Determination of Hydrodynamic Size and Zeta Potential Fromelectrophoretic Mobility and Light Scattering Measurements. Colloids Surf. 1997, 141, 111–117. [Google Scholar] [CrossRef]
- Yukselen-Aksoy, Y.; Kaya, A. A Study of Factors Affecting on the Zeta Potential of Kaolinite and Quartz Powder. Environ. Earth Sci. 2011, 62, 697–705. [Google Scholar] [CrossRef]
- Sadeghian, F.; Jahandari, S.; Haddad, A.; Rasekh, H.; Li, J. Effects of Variations of Voltage and pH Value on the Shear Strength of Soil and Durability of Different Electrodes and Piles during Electrokinetic Phenomenon. J. Mech. Geotech. Eng. 2022, 14, 625–636. [Google Scholar] [CrossRef]
- Wu, H.; Chen, X.; Tan, R.; Luo, Y.; Hu, Y.; Li, Y.; Hou, J.; Kang, W. Controllable Regulation of Emulsion Stability by a pH-Responsive Zwitterionic/Anionic Surfactant System. Fuel 2022, 312, 122921. [Google Scholar] [CrossRef]
- Carvalho, P.M.; Felício, M.R.; Santos, N.C.; Gonçalves, S.; Domingues, M.M. Application of Light Scattering Techniques to Nanoparticle Characterization and Development. Front. Chem. 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Goldhaber, S.Z.; Grasso-Correnti, N. Treatment of Blood Clots. Circulation 2002, 106, 138–140. [Google Scholar] [CrossRef] [PubMed]
- Barshtein, G.; Pajic-Lijakovic, I.; Gural, A. Deformability of Stored Red Blood Cells. Front. Physiol. 2021, 12, 722896. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Pierre-Pierre, N.; Huo, Q. Dynamic Light Scattering for Gold Nanorod Size Characterization and Study of Nanorod-Protein Interactions. Gold. Bull. 2012, 45, 187–195. [Google Scholar] [CrossRef]
- Li, B.L.; Zhang, J.; Jin, W.; Chen, X.Y.; Yang, J.M.; Chi, S.M.; Ruan, Q.; Zhao, Y. Oral Administration of pH-Responsive Polyamine Modified Cyclodextrin Nanoparticles for Controlled Release of Anti-Tumor Drugs. React. Funct. Polym. 2022, 172, 105175. [Google Scholar] [CrossRef]
- Reshetenko, T.; Odgaard, M.; Randolf, G.; Ohtaki, K.K.; Bradley, J.P.; Zulevi, B.; Lyu, X.; Cullen, D.A.; Jafta, C.J.; Serov, A.; et al. Design of PGM-Free Cathodic Catalyst Layers for Advanced PEM Fuel Cells. Appl. Catal. B 2022, 312, 121424. [Google Scholar] [CrossRef]
- Ramirez, L.M.F.; Rihouey, C.; Chaubet, F.; Le Cerf, D.; Picton, L. Characterization of Dextran Particle Size: How Frit-Inlet Asymmetrical Flow Field-Flow Fractionation (FI-AF4) Coupled Online with Dynamic Light Scattering (DLS) Leads to Enhanced Size Distribution. J. Chromatogr. A 2021, 1653, 462404. [Google Scholar] [CrossRef]
- Hassett, K.J.; Higgins, J.; Woods, A.; Levy, B.; Xia, Y.; Hsiao, C.J.; Acosta, E.; Almarsson, Ö.; Moore, M.J.; Brito, L.A. Impact of Lipid Nanoparticle Size on mRNA Vaccine Immunogenicity. J. Control. Release 2021, 335, 237–246. [Google Scholar] [CrossRef]
- Patel, N.; Davis, Z.; Hofmann, C.; Vlasak, J.; Loughney, J.W.; DePhillips, P.; Mukherjee, M. Development and Characterization of an In Vitro Cell-Based Assay to Predict Potency of mRNA–LNP-Based Vaccines. Vaccines 2023, 11, 1224. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Liu, Y.; Wagner, A.M.; Chen, M.; Zhao, Y.; Smith, K.J.; Some, D.; Abend, A.M.; Pennington, J. Enabling Online Determination of the Size-Dependent RNA Content of Lipid Nanoparticle-Based RNA Formulations. J. Chromatogr. B Analy.t Technol. Biomed. Life Sci. 2021, 1186, 123015. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Yeap, S.P.; Che, H.X.; Low, S.C. Characterization of Magnetic Nanoparticle by Dynamic Light Scattering. Nanoscale Res. Lett. 2013, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Hassan, P.A.; Rana, S.; Verma, G. Making Sense of Brownian Motion: Colloid Characterization by Dynamic Light Scattering. Langmuir 2015, 31, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Besra, L.; Liu, M. A Review on Fundamentals and Applications of Electrophoretic Deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–61. [Google Scholar] [CrossRef]
- Cai, H.; Xu, E.G.; Du, F.; Li, R.; Liu, J.; Shi, H. Analysis of Environmental Nanoplastics: Progress and Challenges. Chem. Eng. J. 2021, 410, 128208. [Google Scholar] [CrossRef]
- Jiang, C.; Liu, S.; Zhang, T.; Liu, Q.; Alvarez, P.J.J.; Chen, W. Current Methods and Prospects for Analysis and Characterization of Nanomaterials in the Environment. Environ. Sci. Technol. 2022, 56, 7426–7447. [Google Scholar] [CrossRef]
- Tareq, S.M.; Boutchuen, A.; Roy, S.; Zimmerman, D.; Jur, G.; Bathi, J.R.; Palchoudhury, S. A Dynamic Light Scattering Approach for Detection of Nanomaterials in Tennessee River. Water Resour. Res. 2021, 57, e2020WR028687. [Google Scholar] [CrossRef]
- Baalousha, M.; Lead, J.R. Rationalizing Nanomaterial Sizes Measured by Atomic Force Microscopy, Flow Field-Flow Fractionation, and Dynamic Light Scattering: Sample Preparation, Polydispersity, and Particle Structure. Environ. Sci. Technol. 2012, 46, 6134–6142. [Google Scholar] [CrossRef]
- Domingos, R.F.; Baalousha, M.A.; Ju-Nam, Y.; Reid, M.M.; Tufenkji, N.; Lead, J.R.; Leppard, G.G.; Wilkinson, K.J. Characterizing Manufactured Nanoparticles in the Environment: Multimethod Determination of Particle Sizes. Environ. Sci. Technol. 2009, 43, 7277–7284. [Google Scholar] [CrossRef]
- Hernandez, L.M.; Yousefi, N.; Tufenkji, N. Are There Nanoplastics in Your Personal Care Products? Environ. Sci. Technol. Lett. 2017, 4, 280–285. [Google Scholar] [CrossRef]
- Shahid, M.; Farooqi, Z.H.; Begum, R.; Arif, M.; Irfan, A.; Azam, M. Extraction of Cobalt Ions from Aqueous Solution by Microgels for In-Situ Fabrication of Cobalt Nanoparticles to Degrade Toxic Dyes: A Two-Fold-Environmental Application. Chem. Phys. Lett. 2020, 754, 137645. [Google Scholar] [CrossRef]
- Kebe, M.; Traore, A.; Manousakas, M.I.; Vasilatou, V.; Ndao, A.S.; Wague, A.; Eleftheriadis, K. Source Apportionment and Assessment of Air Quality Index of PM2.5–10 and PM2.5 in at Two Different Sites in Urban Background Area in Senegal. Atmosphere 2021, 12, 182. [Google Scholar] [CrossRef]
- Qiu, Y.; Mu, Z.; Wang, N.; Wang, X.; Xu, M.; Li, H. The Aggregation and Sedimentation of Two Different Sized Copper Oxide Nanoparticles in Soil Solutions: Dependence on pH and Dissolved Organic Matter. Sci. Total Environ. 2020, 731, 139215. [Google Scholar] [CrossRef]
- Tosi, M.M.; Ramos, A.P.; Esposto, B.S.; Jafari, S.M. Dynamic Light Scattering (DLS) of Nanoencapsulated Food Ingredients. Charact. Nanoencapsulated Food Ingred. 2020, 4, 191–211. [Google Scholar] [CrossRef]
- Sats, A.; Yurchenko, S.; Kaart, T.; Tatar, V.; Lutter, L.; Jõudu, I. Bovine Colostrum: Postpartum Changes in Fat Globule Size Distribution and Fatty Acid Profile. J. Dairy. Sci. 2022, 105, 3846–3860. [Google Scholar] [CrossRef]
- Rao, P.; Yu, Z.; Han, H.; Xu, Y.; Ke, L. Dynamic Light Scattering for Food Quality Evaluation. In Evaluation Technologies for Food Quality; Elsevier: Amsterdam, The Netherlands, 2019; pp. 535–557. ISBN 9780128142189. [Google Scholar] [CrossRef]
- Fatehi, P.; Baba, A.S.; Eh suk, V.R.; Misran, M. Preparation and Characterization of Palm Oil in Water Microemulsion for Application in the Food Industry. Br. Food J. 2020, 122, 3077–3088. [Google Scholar] [CrossRef]
- Esposto, B.S.; Pinho, S.G.B.; Thomazini, M.; Ramos, A.P.; Tapia-Blacido, D.R.; Martelli-Tosi, M. TPP-Chitosomes as Potential Encapsulation System to Protect Carotenoid-Rich Extract Obtained from Carrot by-Product: A Comparison with Liposomes and Chitosomes. Food Chem. 2022, 397, 133857. [Google Scholar] [CrossRef]
- Gaida, R.; Dziubek, K.; Król, K.; Ostaszewska, M.; Wit, M. Influence of Different Dispersants on the Quality and Stability of Aqueous Quinacridone Magenta Pigment Dispersion for Ink-Jet Applications. Color. Technol. 2023, 139, 637–653. [Google Scholar] [CrossRef]
- Pandian, B.; Ramalingam, S.; Sreeram, K.J.; Rao, J.R. Natural Pigment: Preparation of Brown Pigment from Lignin Biomass for Coloring Application. Dye. Pigment. 2021, 195, 109704. [Google Scholar] [CrossRef]
- Vincent, B. Early (Pre-DLVO) Studies of Particle Aggregation. Adv. Colloid Interface Sci. 2012, 170, 56–67. [Google Scholar] [CrossRef]
- Ataeefard, M. Production of Carbon Black Acrylic Composite as an Electrophotographic Toner Using Emulsion Aggregation Method: Investigation the Effect of Agitation Rate. Compos. B Eng. 2014, 64, 78–83. [Google Scholar] [CrossRef]
- Ataeefard, M.; Shadman, A.; Saeb, M.R.; Mohammadi, Y. A Hybrid Mathematical Model for Controlling Particle Size, Particle Size Distribution, and Color Properties of Toner Particles. Appl. Phys. A 2016, 122, 726. [Google Scholar] [CrossRef]
- Andami, F.; Ataeefard, M.; Najafi, F.; Saeb, M.R. From Suspension toward Emulsion and Mini-Emulsion Polymerisation to Control Particle Size, Particle Size Distribution, and Sphereness of Printing Toner. Pigment. Resin. Technol. 2016, 45, 363–370. [Google Scholar] [CrossRef]
- Fisher, M. In Situ Manufacturing of Polymer Nanocomposites for Energetic Applications–DSIAC. Available online: https://dsiac.org/articles/in-situ-manufacturing-of-polymer-nanocomposites-for-energetic-applications/ (accessed on 6 April 2023).
- Bhanu, V.A.; Rangarajan, P.; Wiles, K.; Bortner, M.; Sankarpandian, M.; Godshall, D.; Glass, T.E.; Banthia, A.K.; Yang, J.; Wilkes, G.; et al. Synthesis and Characterization of Acrylonitrile Methyl Acrylate Statistical Copolymers as Melt Processable Carbon Fiber Precursors. Polymer 2002, 43, 4841–4850. [Google Scholar] [CrossRef]
- Lovell, P.A.; Schork, F.J. Fundamentals of Emulsion Polymerization. Biomacromolecules 2020, 21, 4396–4441. [Google Scholar] [CrossRef]
- Zhang, G.-J.; Chen, Z.-H.; Zeng, X.-R.; Yu, F.; Wang, J. Preparation and Characterization of Poly(MMA–BA)/Nano-ATO Hybrid Latex via Miniemulsion Polymerization. J. Coat. Technol. Res. 2011, 8, 505–511. [Google Scholar] [CrossRef]
- Ataeefard, M. Production of Black Toner through Emulsion Aggregation of Magnetite, Carbon Black, and Styrene-Acrylic Co-Polymer: Investigation on the Effect of Variation in Components. J. Compos. Mater. 2015, 49, 1553–1561. [Google Scholar] [CrossRef]
- Asua, J.M. Miniemulsion Polymerization. Prog. Polym. Sci. 2002, 27, 1283–1346. [Google Scholar] [CrossRef]
- Niaounakis, M. Emulsions/Dispersions/Solutions/Gels. In Biopolymers: Processing and Products; Elsevier Science: Amsterdam, The Netherlands, 2015; pp. 187–214. ISBN 0-323-27938-4. [Google Scholar]

| Assessment of Stability | Zeta Potential (mV) |
|---|---|
| Flocculation of coagulation | 0 to +/−5 |
| Incipient instability | +/−10 to +/−30 |
| Moderate stability | +/−30 to +/−40 |
| Good stability | +/−40 to +/−60 |
| Excellent stability | Greater than +/−60 |
| Force | Influence |
|---|---|
| van der Waals | Short-range electromagnetic force between NPs, attractive. |
| Electrical Double Layer | Electrical interaction between NPs due to the overlap of electric double layer, typically repulsive. |
| Hydration Force | Interaction between water molecules on hydrophilic NPs, repulsive nature. |
| Hydrophobic Force | Attractive interaction between hydrophobic NPs in water. |
| Steric, Electronic, and Electrostatic Forces | Surface coatings: inorganic, surfactants, polymers, and polyelectrolyte on NP surfaces. Polymers can form bridges leading to osmotic forces for interpenetrate chains. Surface coatings can have attractive or repulsive effects. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez-Loya, J.; Lerma, M.; Gardea-Torresdey, J.L. Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review. Micromachines 2024, 15, 24. https://doi.org/10.3390/mi15010024
Rodriguez-Loya J, Lerma M, Gardea-Torresdey JL. Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review. Micromachines. 2024; 15(1):24. https://doi.org/10.3390/mi15010024
Chicago/Turabian StyleRodriguez-Loya, Jesus, Maricarmen Lerma, and Jorge L. Gardea-Torresdey. 2024. "Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review" Micromachines 15, no. 1: 24. https://doi.org/10.3390/mi15010024
APA StyleRodriguez-Loya, J., Lerma, M., & Gardea-Torresdey, J. L. (2024). Dynamic Light Scattering and Its Application to Control Nanoparticle Aggregation in Colloidal Systems: A Review. Micromachines, 15(1), 24. https://doi.org/10.3390/mi15010024






