Bronchoalveolar Lavage Fluid from Chronic Obstructive Pulmonary Disease Patients Increases Neutrophil Chemotaxis Measured by a Microfluidic Platform
Abstract
1. Introduction
2. Methods
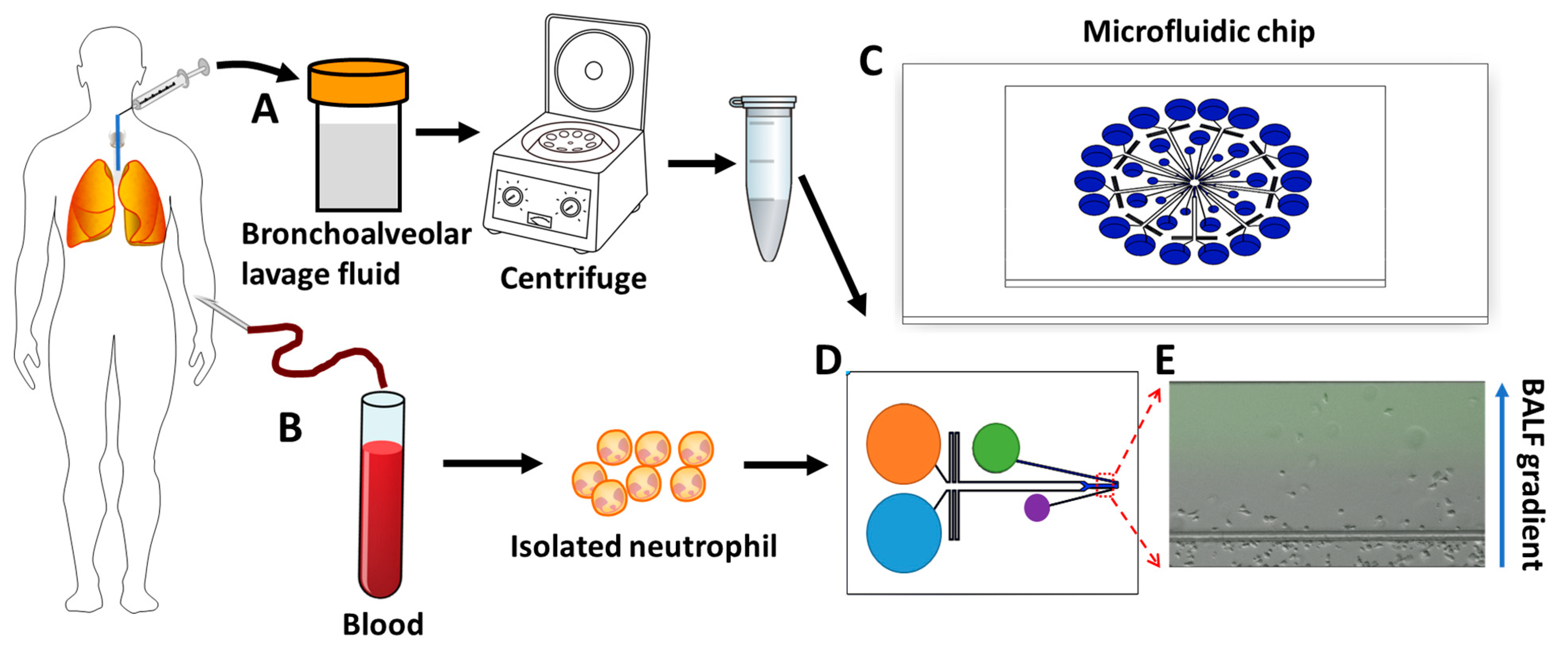
2.1. Bronchoalveolar Lavage Fluid (BALF) Sample Collection and Preparation
2.2. Neutrophil Preparation
2.3. Microfluidic Device Preparation
2.4. Microfluidic Chemotaxis Experiment
2.5. Drug Response Experiment
2.6. Data Analysis
3. Results
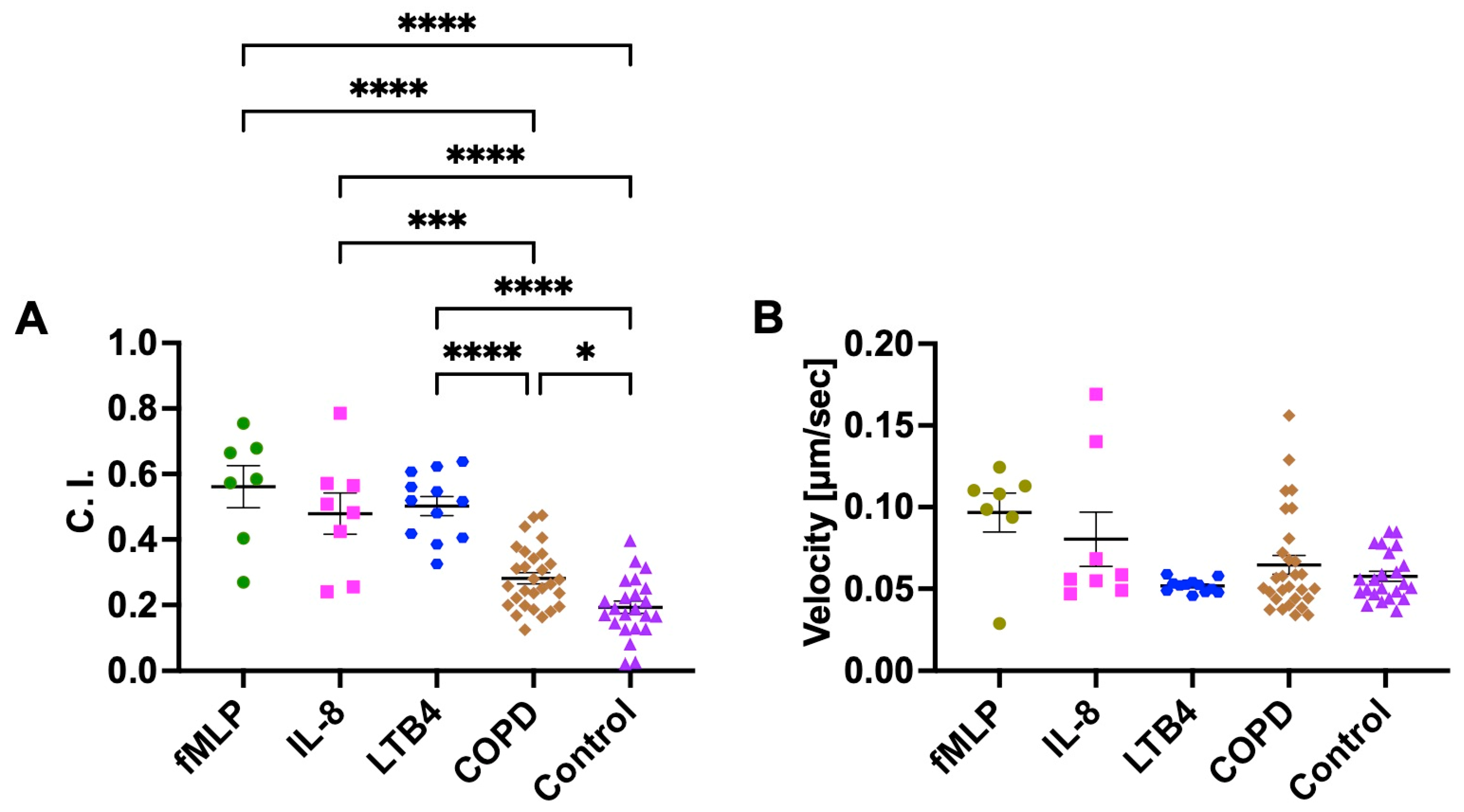
3.1. Neutrophil Chemotaxis in COPD BALF, Control BALF, and Known Recombinant Chemoattractants
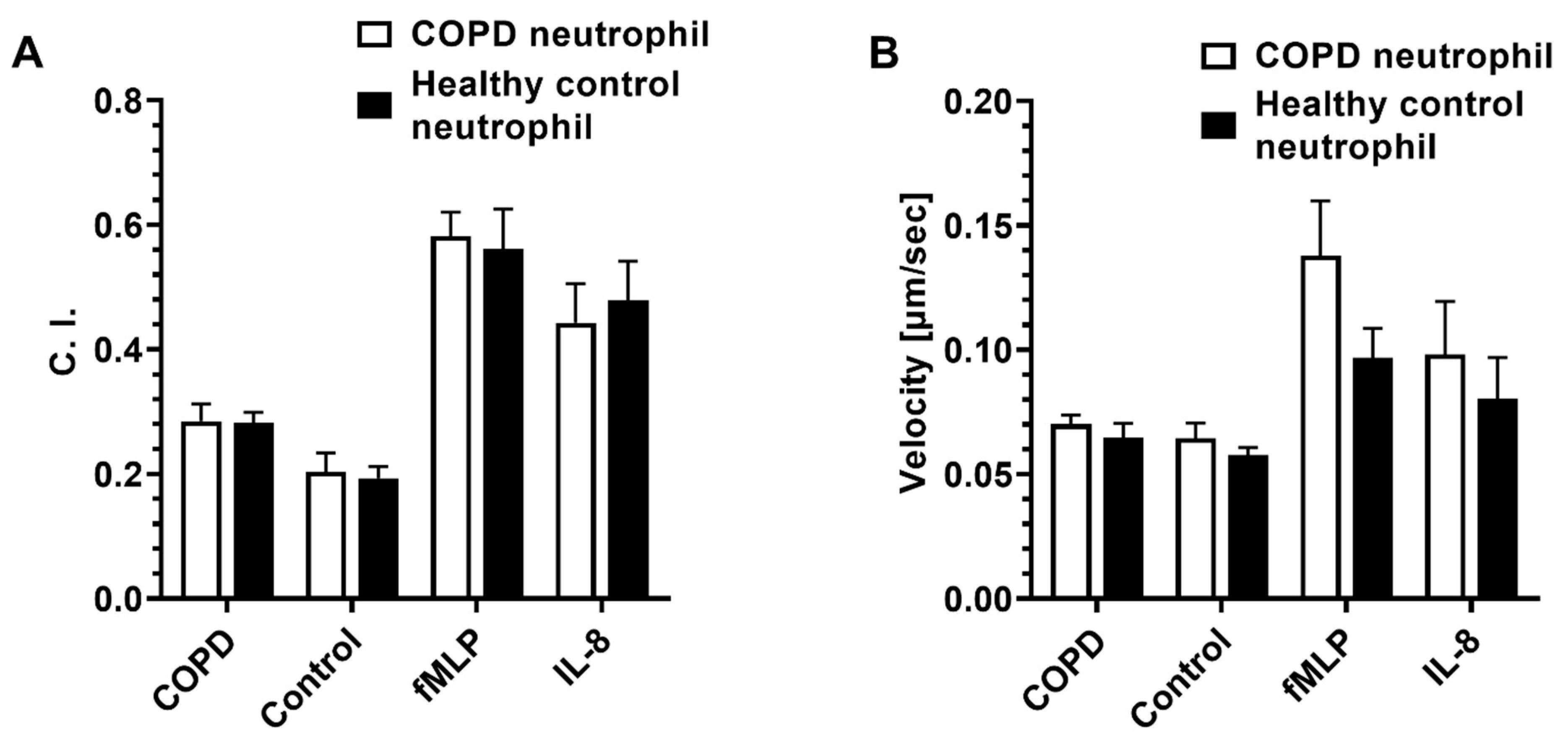
3.2. Effect of Dexamethasone on Neutrophil Chemotaxis
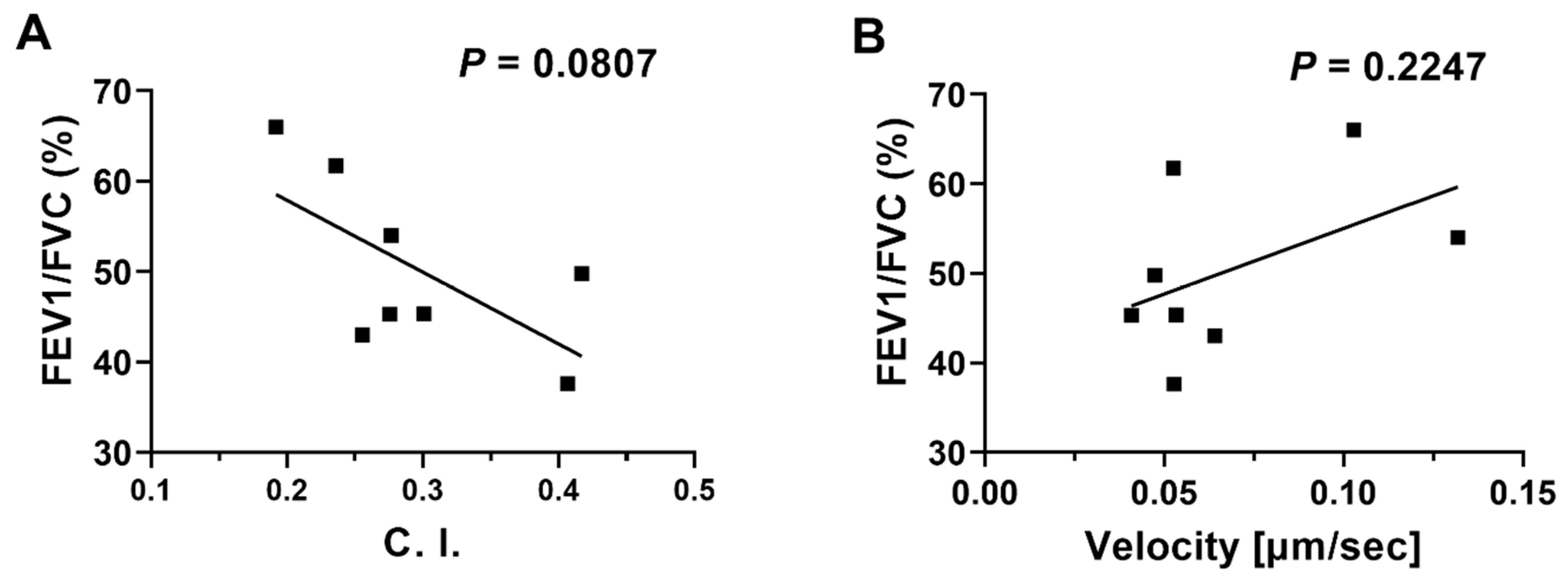
3.3. Linear Regression Analyses of Neutrophil migration in COPD BALF against Patients’ Clinical Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Chronic Obstructive Pulmonary Disease (COPD). 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd) (accessed on 22 December 2022).
- Hoenderdos, K.; Condliffe, A. The neutrophil in chronic obstructive pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2013, 48, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Angelis, N.; Porpodis, K.; Zarogoulidis, P.; Spyratos, D.; Kioumis, I.; Papaiwannou, A.; Pitsiou, G.; Tsakiridis, K.; Mpakas, A.; Arikas, S.; et al. Airway inflammation in chronic obstructive pulmonary disease. J. Thorac. Dis. 2014, 6 (Suppl. S1), S167–S172. [Google Scholar]
- Fricker, M.; Deane, A.; Hansbro, P.M. Animal models of chronic obstructive pulmonary disease. Expert Opin. Drug Discov. 2014, 9, 629–645. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.L.; Cosio, M.; Churg, A. Animal models of chronic obstructive pulmonary disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, L1–L15. [Google Scholar] [CrossRef]
- Sutherland, E.R.; Martin, R.J. Airway inflammation in chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2003, 112, 819–827. [Google Scholar] [CrossRef]
- Lowell, F.C.; Franklin, W.; Michelson, A.L.; Schiller, I.W. Chronic obstructive pulmonary emphysema: A disease of smokers. Ann. Intern. Med. 1956, 45, 268–274. [Google Scholar]
- Halbert, R.J.; Natoli, J.L.; Gano, A.; Badamgarav, E.; Buist, A.S.; Mannino, D.M. Global burden of COPD: Systematic review and meta-analysis. Eur. Respir. J. 2006, 28, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Harber, P.; Tashkin, D.P.; Simmons, M.; Crawford, L.; Hnizdo, E.; Connett, J.; Lung Health Study, G. Effect of occupational exposures on decline of lung function in early chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2007, 176, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Inflammatory mechanisms in patients with chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2016, 138, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Cellular and molecular mechanisms of chronic obstructive pulmonary disease. Clin. Chest Med. 2014, 35, 71–86. [Google Scholar] [CrossRef]
- Rosales, C. Neutrophils at the crossroads of innate and adaptive immunity. J. Leukoc. Biol. 2020, 108, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Afonso, P.V.; Janka-Junttila, M.; Lee, Y.J.; McCann, C.P.; Oliver, C.M.; Aamer, K.A.; Losert, W.; Cicerone, M.T.; Parent, C.A. LTB4 is a signal-relay molecule during neutrophil chemotaxis. Dev. Cell. 2012, 22, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Oved, J.H.; Paris, A.J.; Gollomp, K.; Dai, N.; Rubey, K.; Wang, P.; Spruce, L.A.; Seeholzer, S.H.; Poncz, M.; Worthen, G.S. Neutrophils promote clearance of nuclear debris following acid-induced lung injury. Blood 2021, 137, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Ham, J.; Kim, J.; Ko, Y.G.; Kim, H.Y. The Dynamic Contribution of Neutrophils in the Chronic Respiratory Diseases. Allergy Asthma Immunol. Res. 2022, 14, 361–378. [Google Scholar] [CrossRef] [PubMed]
- Hogg, J.C.; Chu, F.; Utokaparch, S. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 2004, 350, 2645–2653. [Google Scholar] [CrossRef] [PubMed]
- Pesci, A.; Majori, M.; Cuomo, A.; Borciani, N.; Bertacco, S.; Cacciani, G.; Gabrielli, M. Neutrophils infiltrating bronchial epithelium in chronic obstructive pulmonary disease. Respir. Med. 1998, 92, 863–870. [Google Scholar] [CrossRef]
- Pilette, C.; Colinet, B.; Kiss, R.; Andre, S.; Kaltner, H.; Gabius, H.J.; Delos, M.; Vaerman, J.P.; Decramer, M.; Sibille, Y. Increased galectin-3 expression and intra-epithelial neutrophils in small airways in severe COPD. Eur. Respir. J. 2007, 29, 914–922. [Google Scholar] [CrossRef]
- Donaldson, G.C.; Seemungal, T.A.; Patel, I.S.; Bhowmik, A.; Wilkinson, T.M.; Hurst, J.R.; Maccallum, P.K.; Wedzicha, J.A. Airway and systemic inflammation and decline in lung function in patients with COPD. Respir. Med. COPD Update 2006, 1, 101–102. [Google Scholar] [CrossRef][Green Version]
- Sapey, E.; Stockley, J.A.; Greenwood, H.; Ahmad, A.; Bayley, D.; Lord, J.M.; Insall, R.H.; Stockley, R.A. Behavioral and structural differences in migrating peripheral neutrophils from patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2011, 183, 1176–1186. [Google Scholar] [CrossRef] [PubMed]
- Stănescu, D.; Sanna, A.; Veriter, C.; Kostianev, S.; Calcagni, P.G.; Fabbri, L.M.; Maestrelli, P. Airways obstruction, chronic expectoration, and rapid decline of FEV1 in smokers are associated with increased levels of sputum neutrophils. Thorax 1996, 51, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, M.L.; Di Franco, A.; Vagaggini, B.; Bacci, E.; Cianchetti, S.; Dente, F.L.; Tonelli, M.; Paggiaro, P.L. Biological markers in induced sputum of patients with different phenotypes of chronic airway obstruction. Respiration 2009, 77, 265–272. [Google Scholar] [CrossRef]
- Lacoste, J.Y.; Bousquet, J.; Chanez, P.; Van Vyve, T.; Simony-Lafontaine, J.; Lequeu, N.; Vic, P.; Enander, I.; Godard, P.; Michel, F.B. Eosinophilic and neutrophilic inflammation in asthma, chronic bronchitis, and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 1993, 92, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Rytila, P.; Plataki, M.; Bucchieri, F.; Uddin, M.; Nong, G.; Kinnula, V.L.; Djukanovic, R. Airway neutrophilia in COPD is not associated with increased neutrophil survival. Eur. Respir. J. 2006, 28, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Edwards, L.; Tal-Singer, R.; Rennard, S. Sputum neutrophils as a biomarker in COPD: Findings from the ECLIPSE study. Respir. Res. 2010, 11, 77. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Hillier, C.; Komenda, P.; Lobato de Faria, R.; Levin, D.; Zhang, M.; Lin, F. A Microfluidic Platform for Evaluating Neutrophil Chemotaxis Induced by Sputum from COPD Patients. PLoS ONE 2015, 10, e0126523. [Google Scholar] [CrossRef]
- Lonergan, M.; Dicker, A.J.; Crichton, M.L.; Keir, H.R.; Van Dyke, M.K.; Mullerova, H.; Miller, B.E.; Tal-Singer, R.; Chalmers, J.D. Blood neutrophil counts are associated with exacerbation frequency and mortality in COPD. Respir. Res. 2020, 21, 166. [Google Scholar] [CrossRef]
- Franz, C.M.; Jones, G.E.; Ridley, A.J. Cell Migration in Development and Disease. Dev. Cell 2002, 2, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Luster, A.D.; Alon, R.; von Andrian, U.H. Immune cell migration in inflammation: Present and future therapeutic targets. Nat. Immunol. 2005, 6, 1182–1190. [Google Scholar] [CrossRef]
- Woolhouse, I.; Bayley, D.; Stockley, R. Sputum chemotactic activity in chronic obstructive pulmonary disease: Effect of α1–antitrypsin deficiency and the role of leukotriene B4 and interleukin 8. Thorax 2002, 57, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Beeh, K.M.; Kornmann, O.; Buhl, R.; Culpitt, S.V.; Giembycz, M.A.; Barnes, P.J. Neutrophil chemotactic activity of sputum from patients with COPD: Role of interleukin 8 and leukotriene B4. Chest 2003, 123, 1240–1247. [Google Scholar] [CrossRef]
- Boyden, S. The Chemotactic Effect of Mixtures of Antibody and Antigen on Polymorphonuclear Leucocytes. J. Exp. Med. 1962, 115, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Zicha, D.; Dunn, G.A.; Brown, A.F. A new direct-viewing chemotaxis chamber. J. Cell Sci. 1991, 99 Pt 4, 769–775. [Google Scholar] [CrossRef]
- Ren, J.; Wang, N.; Guo, P.; Fan, Y.; Lin, F.; Wu, J. Recent advances in microfluidics-based cell migration research. Lab Chip 2022, 22, 3361–3376. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wu, X.; Lin, F. Recent developments in microfluidics-based chemotaxis studies. Lab Chip 2013, 13, 2484–2499. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Kumar-Kanojia, A.; Hombach-Klonisch, S.; Klonisch, T.; Lin, F. A radial microfluidic platform for higher throughput chemotaxis studies with individual gradient control. Lab Chip 2018, 18, 3855–3864. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.M.; Balhoff, J.B.; Landwehr, G.M.; Rahman, S.M.; Vaithiyanathan, M.; Melvin, A.T. Microfluidic and Paper-Based Devices for Disease Detection and Diagnostic Research. Int. J. Mol. Sci. 2018, 19, 2731. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Butcher, E.C. T cell chemotaxis in a simple microfluidic device. Lab Chip 2006, 5, 1462–1469. [Google Scholar] [CrossRef]
- Sackmann, E.K.; Berthier, E.; Schwantes, E.A.; Fichtinger, P.S.; Evans, M.D.; Dziadzio, L.L.; Huttenlocher, A.; Mathur, S.K.; Beebe, D.J. Characterizing asthma from a drop of blood using neutrophil chemotaxis. Proc. Natl. Acad. Sci. USA 2014, 111, 5813–5818. [Google Scholar] [CrossRef]
- Stockley, J.A.; Walton, G.M.; Lord, J.M.; Sapey, E. Aberrant neutrophil functions in stable chronic obstructive pulmonary disease: The neutrophil as an immunotherapeutic target. Int. Immunopharmacol. 2013, 17, 1211–1217. [Google Scholar] [CrossRef]
- Liu, Y.; Ren, X.; Wu, J.; Wilkins, J.A.; Lin, F. T Cells Chemotaxis Migration Studies with a Multi-Channel Microfluidic Device. Micromachines 2022, 13, 1567. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, H.; Zhang, H.; Ma, W.; Wang, F.; Liu, C.; He, S. Increased interleukin (IL)-8 and decreased IL-17 production in chronic obstructive pulmonary disease (COPD) provoked by cigarette smoke. Cytokine 2011, 56, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, J.; Meng, Y.; Adcock, I.M.; Yao, X. Role of inflammatory cells in airway remodeling in COPD. Int. J. Chron. Obstruct. Pulm. Dis. 2018, 13, 3341–3348. [Google Scholar] [CrossRef]
- Finney-Hayward, T.K.; Bahra, P.; Li, S.; Poll, C.T.; Nicholson, A.G.; Russell, R.E.; Ford, P.A.; Westwick, J.; Fenwick, P.S.; Barnes, P.J.; et al. Leukotriene B4 release by human lung macrophages via receptor- not voltage-operated Ca2+ channels. Eur. Respir. J. 2009, 33, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.T.; Bayley, D.; Stockley, R.A. The interrelationship of sputum inflammatory markers in patients with chronic bronchitis. Am. J. Respir. Crit. Care Med. 1999, 160, 893–898. [Google Scholar] [CrossRef]
- Barnes, P. Chronic obstructive pulmonary disease • 12: New treatments for COPD. Thorax 2003, 58, 803–808. [Google Scholar] [CrossRef]
- Ding, Y.L.; Yao, W.Z.; Zheng, J.; Zhu, Y.L.; Liu, Z. Changes of leukotriene B4 in chronic obstructive pulmonary disease and effects of theophylline on leukotriene B4. Beijing Da Xue Xue Bao Yi Xue Ban 2005, 37, 393–397. [Google Scholar] [PubMed]
- Wang, C.; Zhou, J.; Wang, J.; Li, S.; Fukunaga, A.; Yodoi, J.; Tian, H. Progress in the mechanism and targeted drug therapy for COPD. Signal Transduct. Target. Ther. 2020, 5, 248. [Google Scholar] [CrossRef]
- Jones, P.W.; Harding, G.; Berry, P.; Wiklund, I.; Chen, W.H.; Kline Leidy, N. Development and first validation of the COPD Assessment Test. Eur. Respir. J. 2009, 34, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Ardestani, M.E.; Kalantary, E.; Samaiy, V.; Taherian, K. Methyl prednisolone vs Dexamethasone in Management of COPD Exacerbation; a Randomized Clinical Trial. Emergency 2017, 5, e35. [Google Scholar]
- Moslem, S.; Hossein, D.; Afrasiabifar, A.; Ahad, H. Evaluation of Effectiveness of Dexamethasone versus Hydrocortisone in Reducing Hospital Length, Peak Expiratory Flow, Oxidative Stress Factors and Inflammatory Mediators in Chronic Obstructive Pulmonary Disease (COPD) Exacerbation Patients: A Randomized Controlled Trial. Res. Sq. 2022, 1–10. [Google Scholar] [CrossRef]
- Grundy, S.; Plumb, J.; Kaur, M.; Ray, D.; Singh, D. Additive anti-inflammatory effects of corticosteroids and phosphodiesterase-4 inhibitors in COPD CD8 cells. Respir. Res. 2016, 17, 9. [Google Scholar] [CrossRef] [PubMed]


| No. | Gender | Age | FEV1 (L) | FEV1 (%) | FVC (L) | FVC (%) | FEV1/FVC (%) | |
|---|---|---|---|---|---|---|---|---|
| COPD | 1 | M | 61 | 0.82 | 29.60 | 2.18 | 60.30 | 37.63 |
| 2 | M | 72 | 1.53 | 52.41 | 3.37 | 87.59 | 45.35 | |
| 3 | M | 59 | 1.76 | 54.92 | 2.85 | 70.45 | 61.74 | |
| 4 | M | 71 | 0.89 | 35.11 | 2.08 | 60.87 | 43.01 | |
| 5 | M | 68 | 0.79 | 40.51 | 1.75 | 70.23 | 45.32 | |
| 6 | M | 65 | 1.38 | 45.80 | 2.78 | 71.70 | 49.80 | |
| 7 | M | 67 | 1.55 | 59.07 | 2.87 | 85.13 | 54.01 | |
| 8 | M | 63 | 1.98 | 69.00 | 2.99 | 79.19 | 66.00 | |
| Control | 1 | F | 53 | 2.13 | 91.80 | 2.8 | 102.20 | 76.16 |
| 2 | M | 46 | 3.37 | 97.78 | 4.69 | 110.52 | 71.88 | |
| 3 | M | 40 | 3.72 | 97.63 | 4.5 | 97.37 | 82.80 | |
| 4 | M | 46 | 3.74 | 116.49 | 4.46 | 114.40 | 83.84 | |
| 5 | M | 36 | 3.69 | 99.14 | 4.45 | 99.79 | 82.96 | |
| 6 | F | 57 | 2.48 | 111.48 | 2.88 | 105.65 | 86.14 | |
| 7 | M | 38 | 3.65 | 95.65 | 4.49 | 97.87 | 81.16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, J.; Chen, W.; Zhong, Z.; Wang, N.; Chen, X.; Yang, H.; Li, J.; Tang, P.; Fan, Y.; Lin, F.; et al. Bronchoalveolar Lavage Fluid from Chronic Obstructive Pulmonary Disease Patients Increases Neutrophil Chemotaxis Measured by a Microfluidic Platform. Micromachines 2023, 14, 1740. https://doi.org/10.3390/mi14091740
Ren J, Chen W, Zhong Z, Wang N, Chen X, Yang H, Li J, Tang P, Fan Y, Lin F, et al. Bronchoalveolar Lavage Fluid from Chronic Obstructive Pulmonary Disease Patients Increases Neutrophil Chemotaxis Measured by a Microfluidic Platform. Micromachines. 2023; 14(9):1740. https://doi.org/10.3390/mi14091740
Chicago/Turabian StyleRen, Jiaqi, Wenfang Chen, Zhicheng Zhong, Ning Wang, Xi Chen, Hui Yang, Jing Li, Ping Tang, Yanping Fan, Francis Lin, and et al. 2023. "Bronchoalveolar Lavage Fluid from Chronic Obstructive Pulmonary Disease Patients Increases Neutrophil Chemotaxis Measured by a Microfluidic Platform" Micromachines 14, no. 9: 1740. https://doi.org/10.3390/mi14091740
APA StyleRen, J., Chen, W., Zhong, Z., Wang, N., Chen, X., Yang, H., Li, J., Tang, P., Fan, Y., Lin, F., Bai, C., & Wu, J. (2023). Bronchoalveolar Lavage Fluid from Chronic Obstructive Pulmonary Disease Patients Increases Neutrophil Chemotaxis Measured by a Microfluidic Platform. Micromachines, 14(9), 1740. https://doi.org/10.3390/mi14091740








