1. Introduction
Medical mattresses with advanced humidity sensors are receiving significant attention when it comes to treating patients with IAD [
1,
2,
3]. Designing the humidity transducer so that it can detect an amount of water vapour (
O) is one of the most important medical judgements and diagnoses. The recent development of humidity sensors for patients with IAD is gaining acceptance within medical devices [
4,
5,
6]. Some scholars have developed a humidity sensor with inflammatory cytokines [
7], skin temperature [
8], metal oxide nanomaterials [
9], transepidermal water [
10], and water contact angle [
11]. In the present designs, there has been a testing of mattresses which sense relative humidity (RH) in terms of resistance [
5,
12,
13,
14], film thickness [
15], and the refractive index (RI) [
16].
Humidity sensors for patients with IAD have been tested with moisture-associated dermatitis, liquid, and water vapour [
1,
8,
10]. The humidity-sensors system has acquired water vapour consisting of highly reactive dipolar molecules, temperature, gaseous form, moisture, and liquid [
1,
6,
9]. Previous studies have tested the humidity sensors’ design based on sensitivity, electro-active flow control devices, fast response, and low cost for sensing materials [
4,
17]. Many scholars have divided patients into three types when it comes to IAD-sensing devices: light, moderate, and heavy skin damage [
18,
19,
20]. Humidity sensors are mainly based on impedance and resistance, including metal oxides [
21], perovskites [
22], and organic polymers [
23].
Typically, the traditional design of humidity-sensing mattresses for patients with IAD has limitations, such as early IAD diagnosis [
2,
7]. Indeed, patients with IAD range from 5.3–46.1% in residential care and 19–54.7% for critically ill patients [
24]. Previous scholars have tested the design of humidity sensors according to the clinical setting at 6% of moisture-associated skin damage in residential care [
25]. Matar et al. [
8] provided four types of sensing moisture-associated skin damage in IAD: erythema, localised swelling, vesicles, and crusting/scaling. The humidity sensor for IAD patients includes keeping the skin dry, maintaining pH balance, and offering moisturiser [
26]. Despite this, mattresses using a flexible humidity sensor constitute one of the most essential medical devices, with examples being polymer film-based sensors [
2], paper-based sensors [
27], and flexible fibre-optic humidity sensors [
28].
Thus, there still remains a lack of advanced humidity sensors which have been tested with patients exhibiting early signs of IAD. Indeed, the characteristics of sensing moisture-associated skin damage are limited, focusing on different types of sensing materials [
1]. Previous studies have set dimensions of 10 × 5 cm to cut a point on the sensitive fibre-optic mattress [
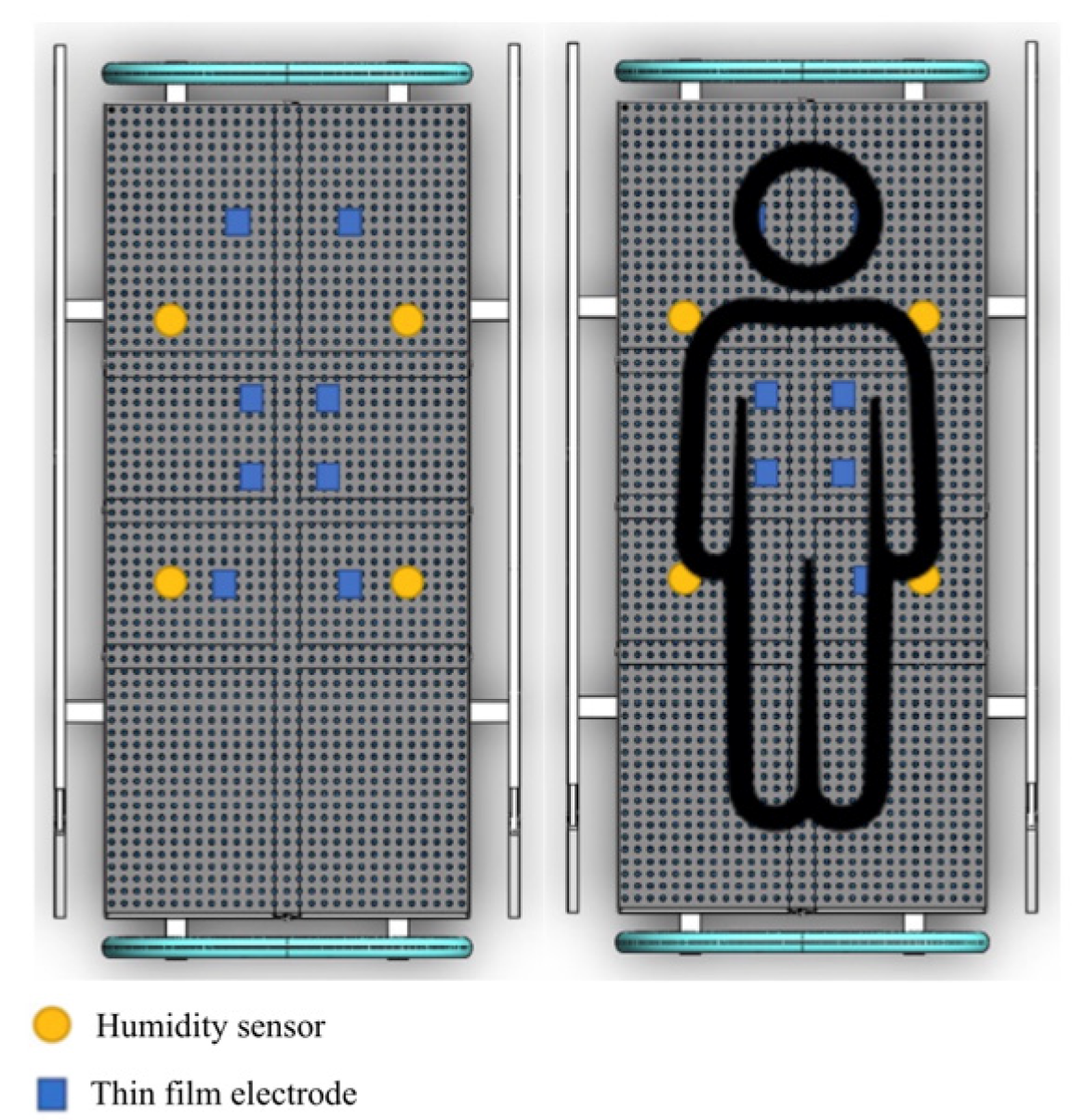
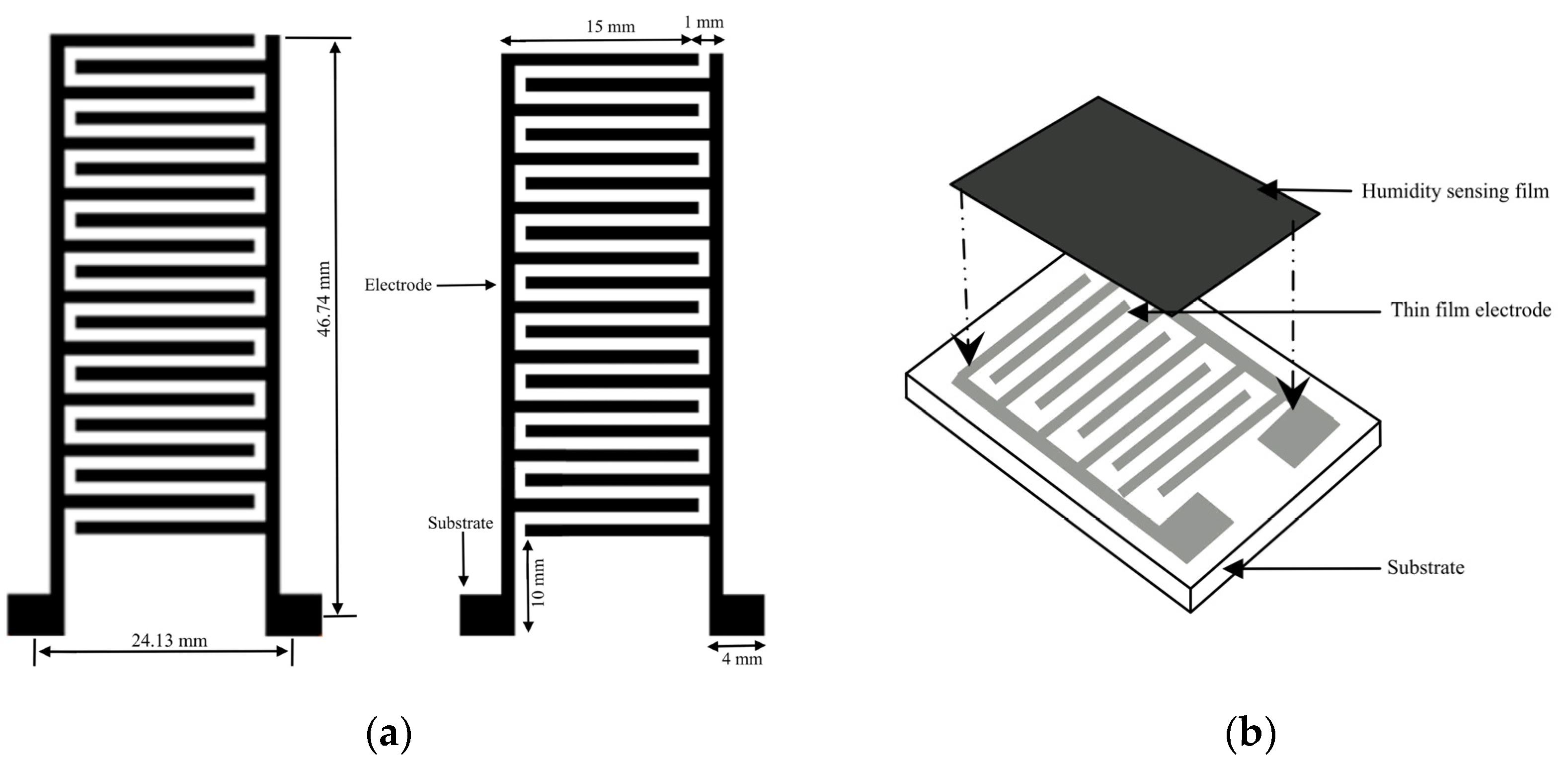
29]. A humidity sensor for IAD patients has the potential to increase the quality of devices, comfort, and sensitivity. The design includes using a thin firm electrode and glass substrate, which greatly tests the sensitivity of the humidity-sensing mattress for patients with IAD. Hence, the thin film electrode as an adhesion agent is reliable for mattress design but has not yet been applied to humidity sensors.
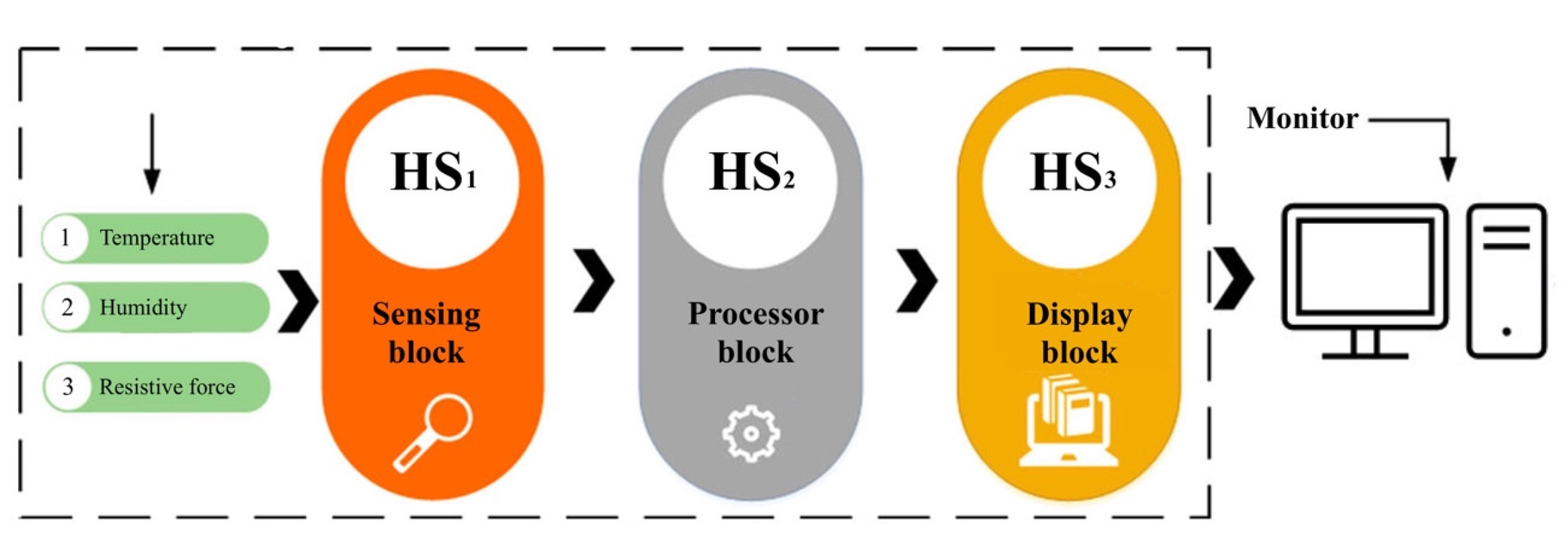
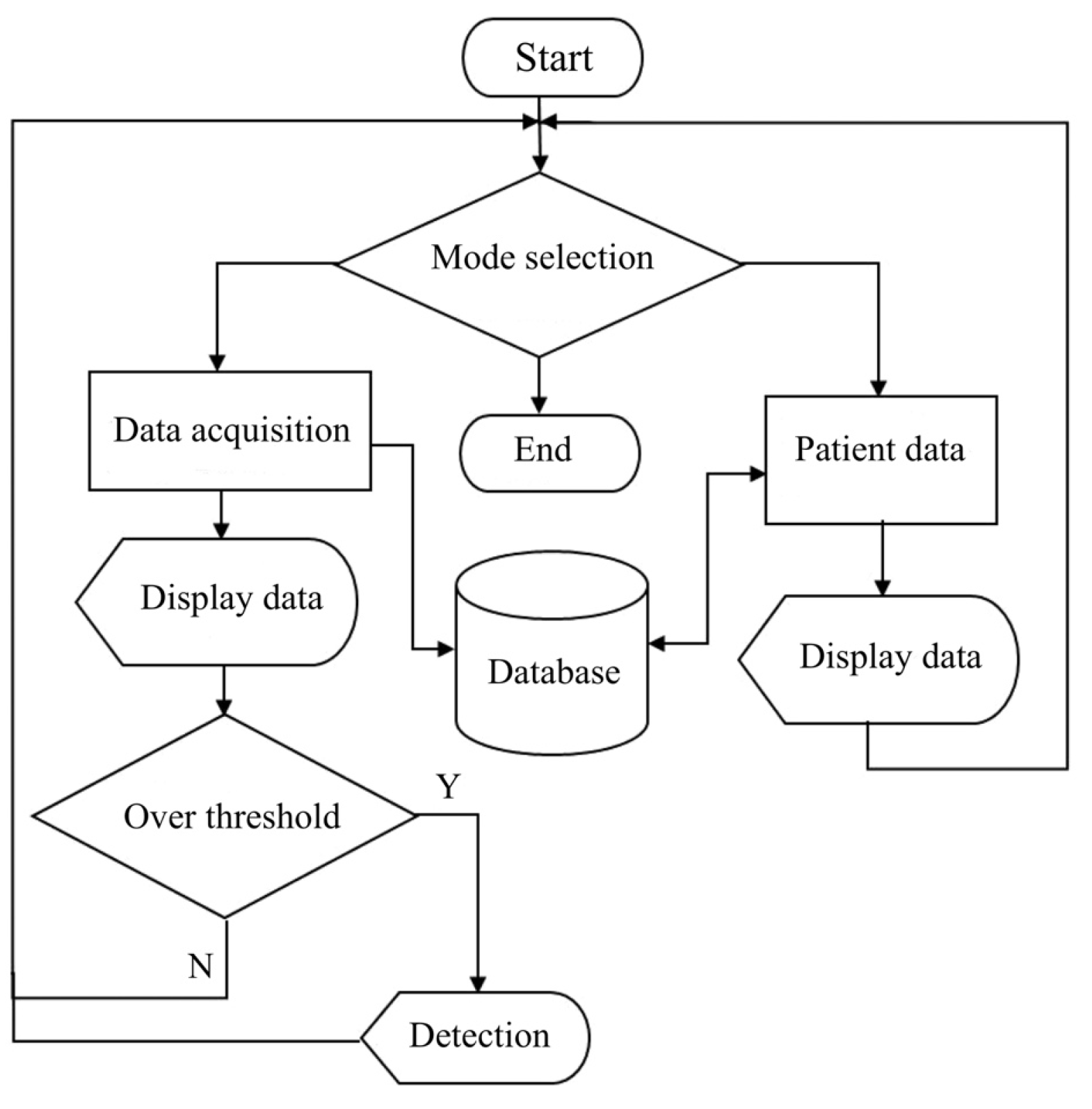
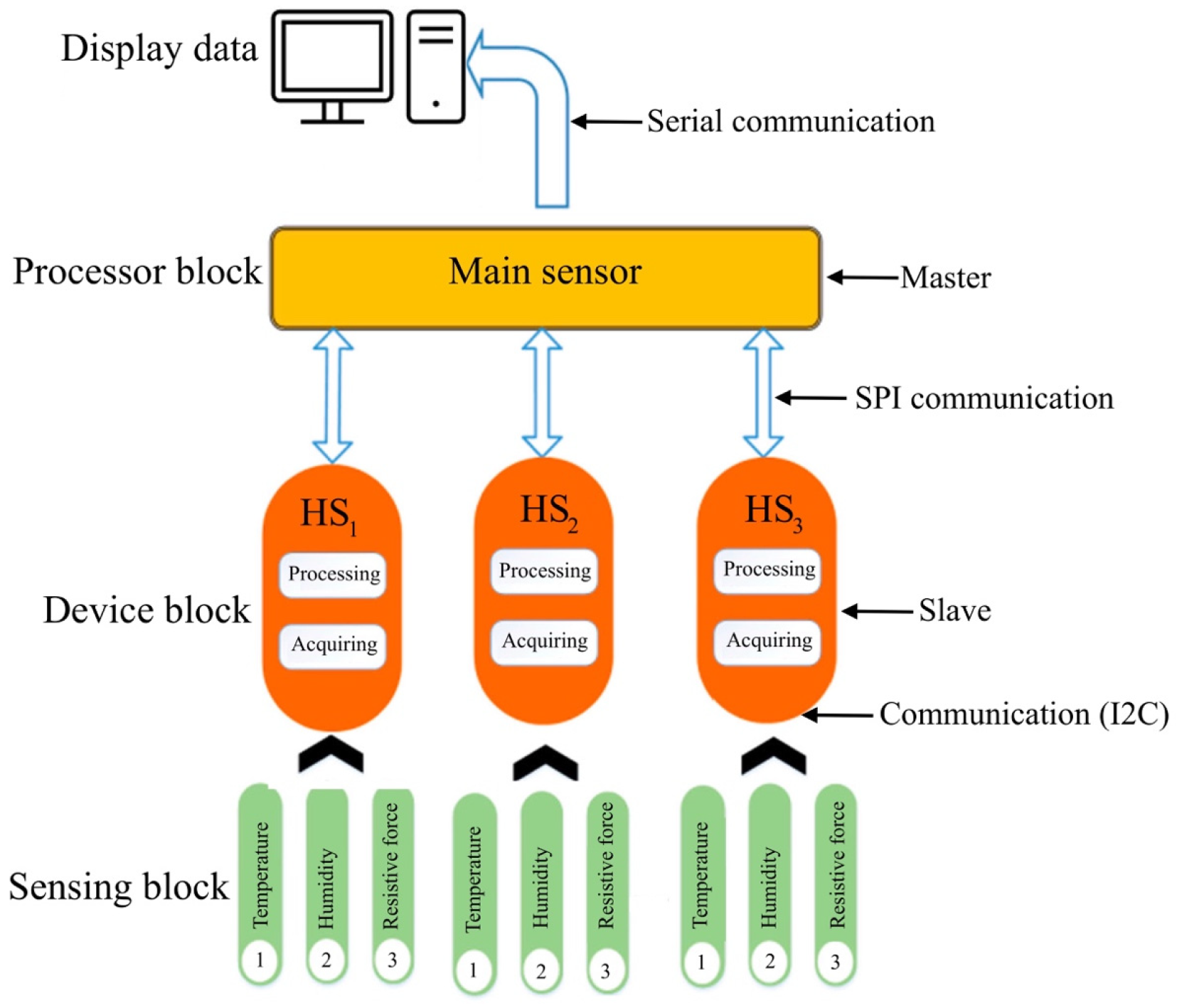
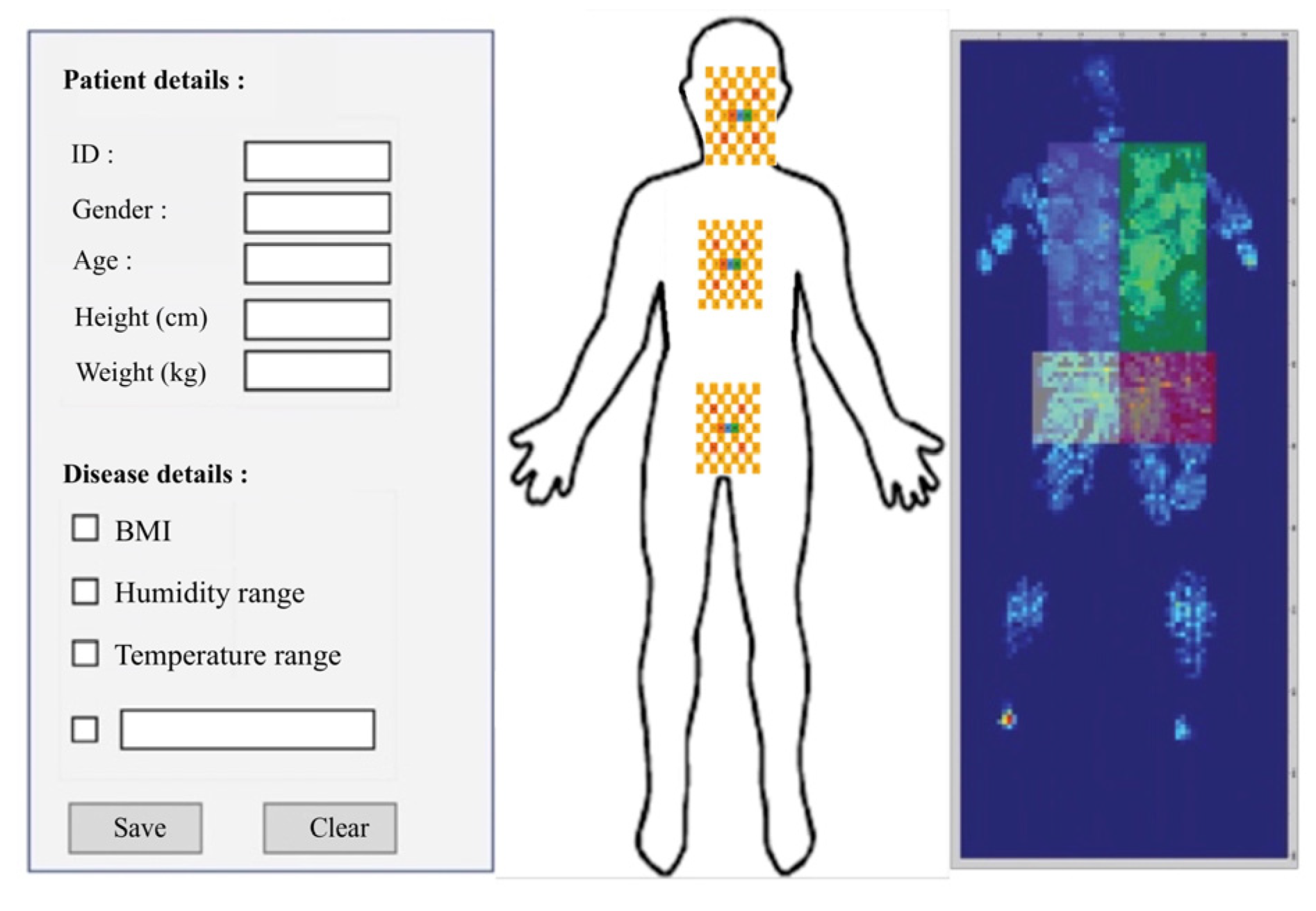
Accordingly, our study has designed a humidity-sensing mattress for patients with IAD following exposure to experimental testing in clinical settings. Firstly, the design of the mattress consists of a humidity-sensing film, a thin film electrode, and a glass substrate. Secondly, the design is also focused on the stability of the humidity sensors (RH, AB, and D/F PT), ambient temperature, and adsorption–desorption dynamic cycles. Finally, when testing the response time of 10–120 s with five volts, the magnitude ranged from 1–1 , and the pore ranged from 0–10 . The results of this study attest not only to humidity sensor designs for IAD patients but also the development of sensitive mattresses which can be developed in clinical settings.
4. Discussion
This is the first study to develop a new sensing mattress for patients with IAD to test the humidity-sensor designs. The design is expected to serve as a sensing device in clinical settings, providing an objective database through which to validate humidity-sensor designs. The volunteer tests showed resistance-sensing humidity at 35 °C, a slope of 1.13 V/fF, and = 1 MHz with 20–90% RH. The sensors showed a response time of 20 s at 2 m, and 300 mm. Our repeatable sensitivity increased to −0.22% RH/°C at 100 °C, decreasing to −0.07% RH/°C at 0% RH. The characterisation of the humidity sensor designs showed a short response of 10 s, magnitude at 1–1 , 1 mol%, and , F, respectively. We found that resistance values of the different parameters were close to 37 °C (i.e., ±1 °C), with values in the range of 20–80% RH, and air velocity of 0.06–0.3 m/s.
The stability of the humidity values is also an important factor affecting the sensitivity of three main sensors: the occipital muscle, rhomboid major muscles, and spine endpoint (see
Table 4 and
Table 5). The highest frequency changes up to
= 1 MHz were observed for three sensors of magnesium ferrite (MgF
) on ratios of lithium (M
F
) at 0.2 ≤ x ≤ 0.6. It is clear that increasing to a 90% RH concentration results in a more viscous solution, which can serve as an effective humidity sensor (see
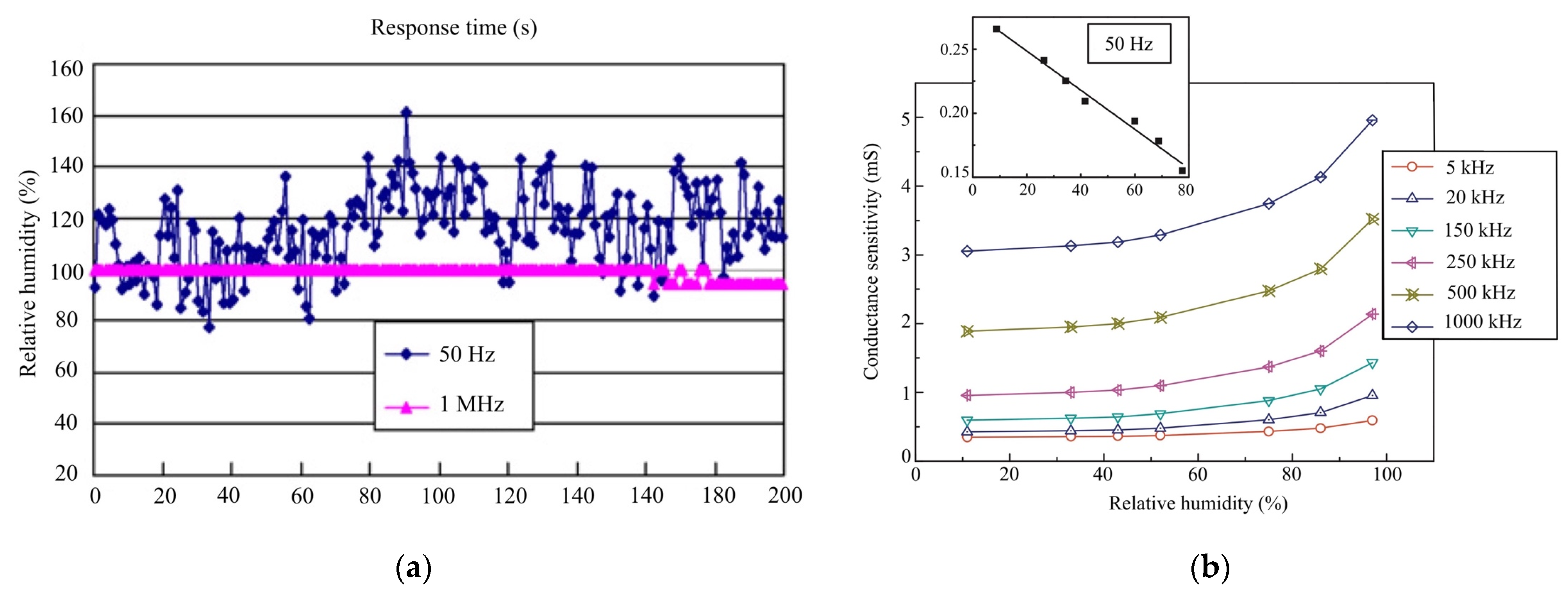
Figure 8a,b). At the same time, we compared the humidity-sensing characteristics of three sensors at 100 kHz with a response time of 10 s, which has proven to be the best frequency from 25% RH–95% RH (see
Figure 10a,b). As the result shows, a response time of 10 s with a five-volt voltage has a small amount of recovery time (the humidity difference at the same impedance value), which can effectively reduce the error of humidity sensors.
The sensing fitness of the resistant response of
= 1 MHz is presented in
Table 7 and
Figure 10. Our experimental testing explained the humidity-sensing mechanism, as suggested in previous studies [
1,
4,
5,
11,
13,
26,
32]. At higher frequencies, there was a change in humidity sensors because adsorbed water molecules cannot be polarised with a rapid change in clinical settings [
16,
33]. The repeatability test with increasing RH proves the suitability of patients with IAD in clinical settings. Therefore, we validated 100 kHz as the operating frequency in the following testing, related to previous studies [
1,
4,
5,
31,
34]. With a variation of 20–90% RH, the change in five-volt voltage exhibited a relatively small pore size of 0.1
This means that, if a mattress is subjected to a high humidity change, it can be inconvenient and the sensing material of the humidity sensor is not good for patients with IAD in clinical settings [
1,
2,
4,
8,
28].
Humidification was performed, with a value of 0.43%/HR% at 30 °C, a grain size of 20 nm, and a frequency between 100 Hz and 100 kHz. It was tested to be 0.5% when compared with the photon crystal fibre sensor of graphene oxide [
35]. At low RH, a layer of the sensing pad of water molecules was adsorbed to form hydroxyl and sensing performance. Subsequently, as the humidity sensor increased, a layer of water molecules was adsorbed with double hydroxyl bonding. In bulk liquid,
þ released
to the adjacent water molecules and then cycled down to 0.5% RH, with a response time increase of 40 s and a recovery time of approximately 230 s. The low hysteresis in the sensor response was due to the highly hydrophobic structure of the thick film [
36].
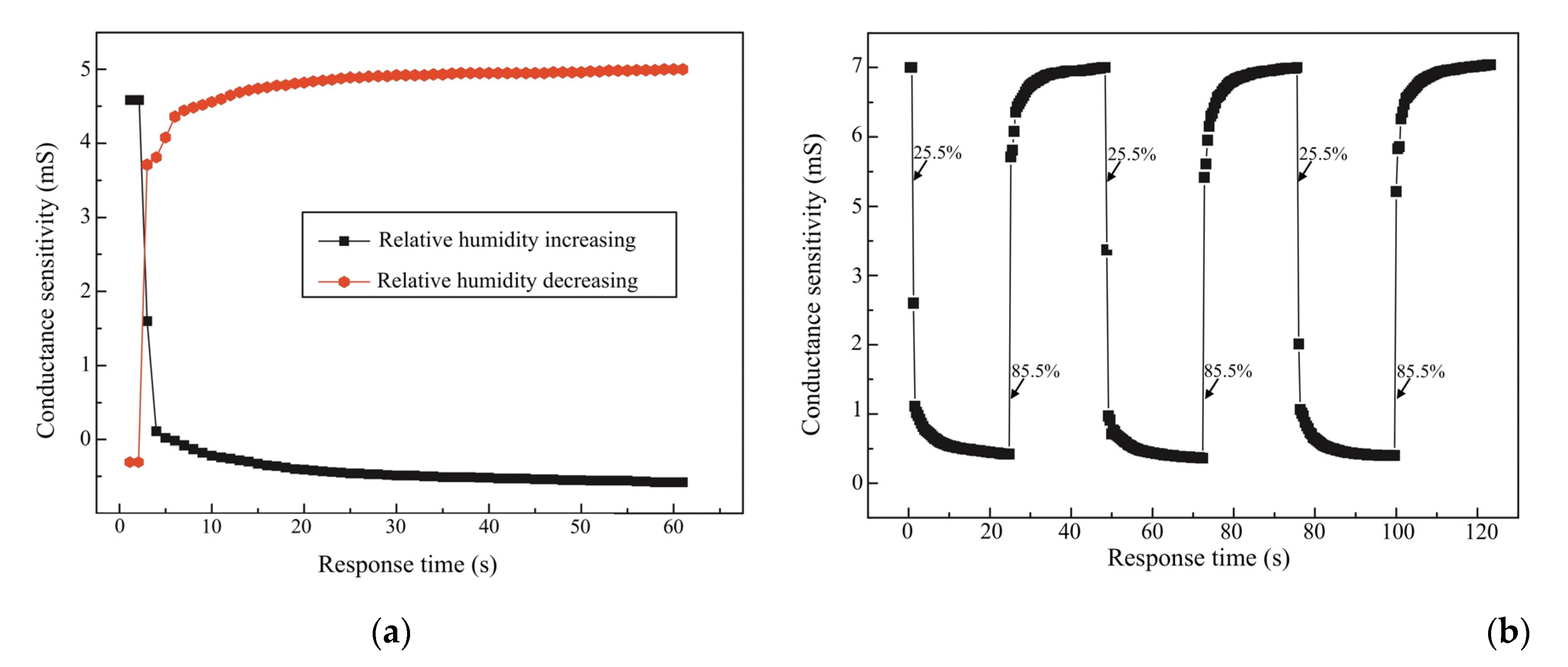
Response and recovery times constitute one of the significant features of the humidity-sensing mattress. The response time was tested by quickly moving the humidity sensor in a relatively dry environment (35% RH), which achieved 90% of the total impedance change. The sensitivity exhibited a fast response and a recovery time of between 10 s and 30 s, which is the best reported time for a resistive type of humidity-sensing mattress for IAD in clinical settings [
24,
32,
37]. A sputtered metal oxide film (1.2
) acted as both an RH and a piezoelectric actuation layer for the electrode. Yang et al. [
38], Bian et al. [
39] and Chu et al. [
40] fabricated humidity sensors using response times of 0.701 nm/RH% and 2.728 nm/RH%, ranging between 30 °C and 110 °C for
= −1
m. The repeatable sensing mattress is based on humidity, perhaps showing a slight response of 50 nm, but there was a noticeably slower recovery (>600 ms) at 35 °C and a rate of 0.8% HR per minute.
5. Conclusions
In conclusion, the design of the humidity-sensing mattress has been tested with regard to the water vapour with RH, AB, PPM, and D/F PT. The testing showed resistance-humidity characteristics of the sensor at 35 °C ( = 30 V, = 350 mV), the slope at 1.13 V/fF, = 1 MHz with 20–90% RH, a response time of 20 s at 2 m, and 300 mm. Repeatable sensitivity demonstrated that the humidity sensor increased to 0.22% RH/°C at 100 °C, and decreased to −0.07% RH/°C at 0.5% RH. The conductivity range was 30% RH with 10 s, a magnitude of 1–1 , 1 mol%, and and F, respectively. Further, the feasible humidity sensor integrated into the water vapour was tested by embedding that sensor in the thin-film electrode. The repeatable humidity sensor has many advantages, with a response time of less than 10 s, whilst the addition of 2 mol % of can greatly improve the sensitivity over the whole range of RH.
The development of the humidity-sensing mattress is a crucial step in the production of a novel piece of medical technology that provides a faster response, high sensitivity, and recovery time; indeed, it may also be a potential design for practical application. Notably, the design of the humidity-sensing mattress compounded with the thin-film electrode as the RH increased to 90%, showing excellent sensitivity. This property enables the sensor to be applied to medical diagnosis for patients with IAD. When the design has been applied, and the enhanced clinical acceptance of sensing material is a reality, it will be possible to provide and develop a low-cost mattress for IAD in clinical homecare. Once available, this humidity-sensing mattress has broad application prospects in the field of flexible sensors, wearable medical diagnostic devices, health detection, etc.