Direct Synthesis of Mn3[Fe(CN)6]2·nH2O Nanosheets as Novel 2D Analog of Prussian Blue and Material for High-Performance Metal-Ion Batteries
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Mn3[Fe(CN)6]2 Nanosheets
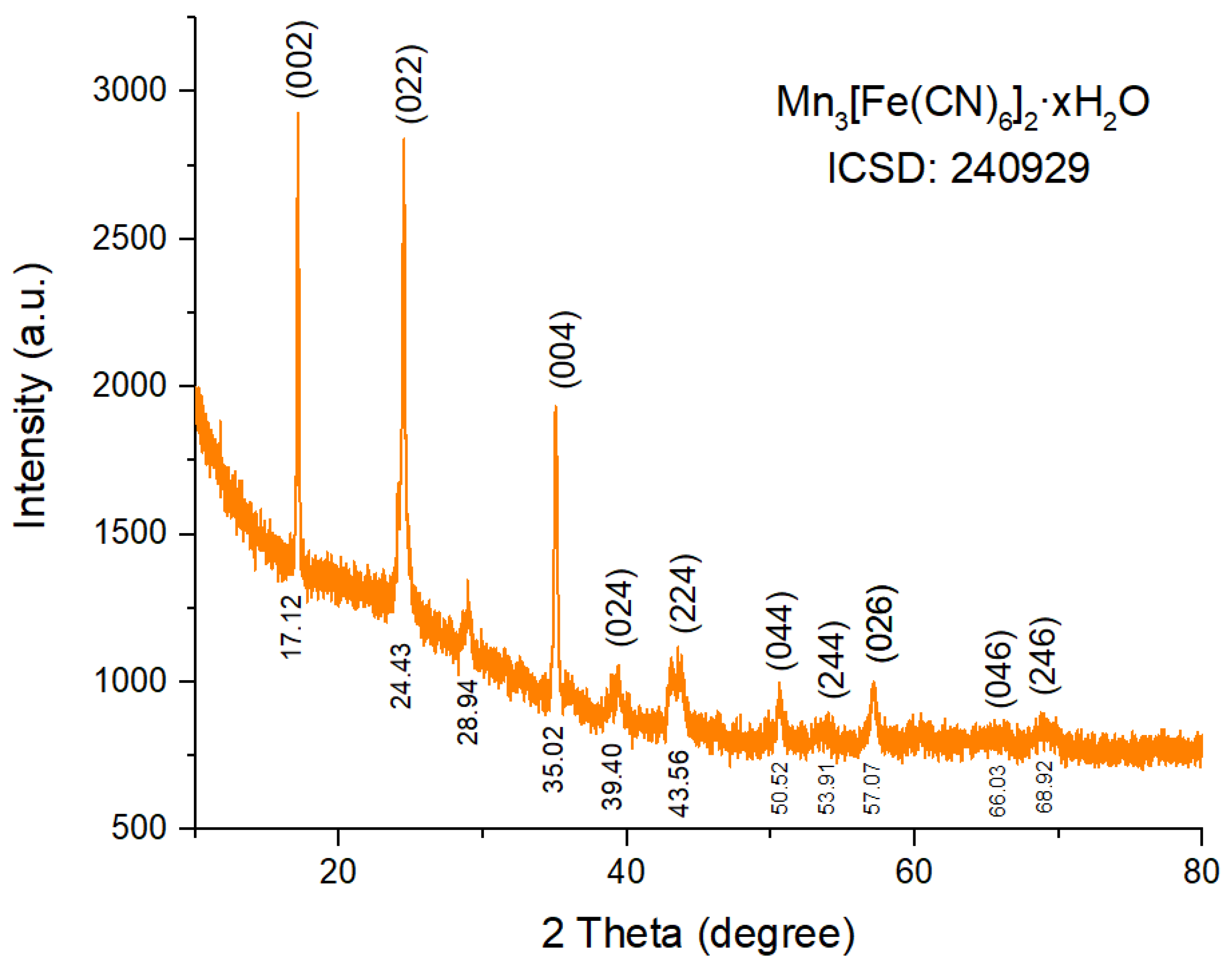
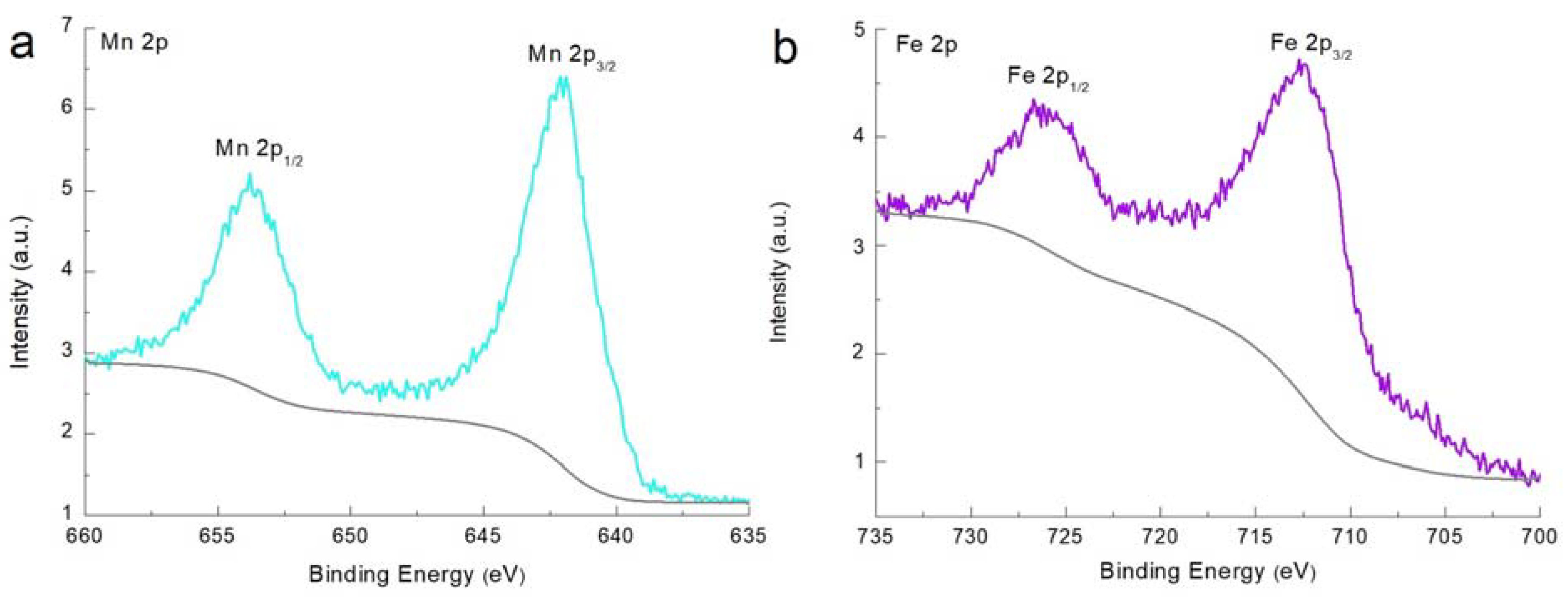
2.3. Material Characterization
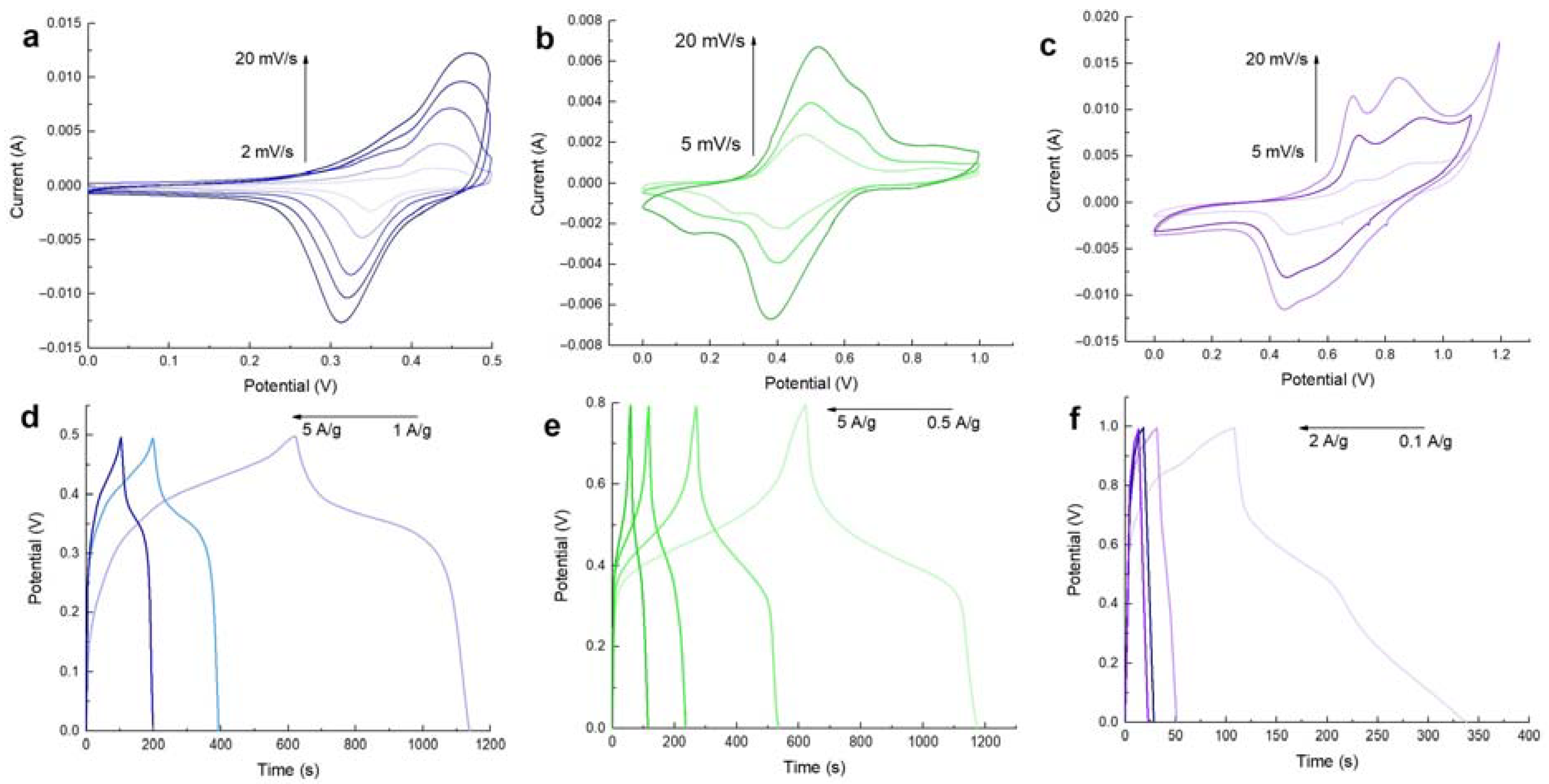
2.4. Electrochemical Characterization
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614. [Google Scholar] [PubMed]
- Wang, M.; Zhang, F.; Lee, C.-S.; Tang, Y. Low-Cost Metallic Anode Materials for High Performance Rechargeable Batteries. Adv. Energy Mater. 2017, 7, 1700536. [Google Scholar]
- Goodenough, J.B.; Park, K.S. The Li-ion rechargeable battery: A perspective. J. Am. Chem. Soc. 2013, 135, 1167–1176. [Google Scholar]
- Konarov, A.; Voronina, N.; Jo, J.H.; Bakenov, Z.; Sun, Y.-K.; Myung, S.-T. Present and Future Perspective on Electrode Materials for Rechargeable Zinc-Ion Batteries. ACS Energy Lett. 2018, 3, 2620–2640. [Google Scholar]
- Jin, J.; Geng, X.; Chen, Q.; Ren, T.L. A Better Zn-Ion Storage Device: Recent Progress for Zn-Ion Hybrid Supercapacitors. Nano-Micro Lett. 2022, 14, 64. [Google Scholar]
- Liu, P.; Lv, R.; He, Y.; Na, B.; Wang, B.; Liu, H. An integrated, flexible aqueous Zn-ion battery with high energy and power densities. J. Power Sources 2019, 410, 137–142. [Google Scholar]
- Wu, X.; Ru, Y.; Bai, Y.; Zhang, G.; Shi, Y.; Pang, H. PBA composites and their derivatives in energy and environmental applications. Coord. Chem. Rev. 2022, 451, 214260. [Google Scholar]
- Hurlbutt, K.; Wheeler, S.; Capone, I.; Pasta, M. Prussian Blue Analogs as Battery Materials. Joule 2018, 2, 1950–1960. [Google Scholar]
- Zeng, Y.X.; Lu, X.F.; Zhang, S.L.; Luan, D.Y.; Li, S.; Lou, X.W. Construction of Co-Mn Prussian Blue Analog Hollow Spheres for Efficient Aqueous Zn-ion Batteries. Angew. Chem. Int. Ed. 2021, 60, 22189–22194. [Google Scholar]
- Fan, L.; Guo, X.; Hang, X.; Pang, H. Synthesis of truncated octahedral zinc-doped manganese hexacyanoferrates and lowtemperature calcination activation for lithium-ion battery. J. Colloid Interface Sci. 2022, 607, 1898–1907. [Google Scholar]
- Lobinsky, A.; Popkov, V. Ultrathin 2D nanosheets of transition metal (hydro)oxides as prospective materials for energy storage devices: A short review. Electrochem. Mater. Technol. 2022, 1, 20221008. [Google Scholar] [CrossRef]
- Tolstoy, V.P. Successive ionic layer deposition. An application in nanotechnology. Russ. Chem. Rev. 2006, 75, 161. [Google Scholar] [CrossRef]
- Ratnayake, S.P.; Ren, J.; Colusso, E.; Guglielmi, M.; Martucci, A.; Della Gaspera, E. SILAR Deposition of Metal Oxide Nanostructured Films. Small 2021, 17, 2101666. [Google Scholar] [CrossRef] [PubMed]
- Soonmin, H. Recent advances in the growth and characterizations of SILAR-deposited thin films. Appl. Sci. 2022, 12, 8184. [Google Scholar] [CrossRef]
- Ghos, B.C.; Farhad, S.F.U.; Patwary, M.A.M.; Majumder, S.; Hossain, M.A.; Tanvir, N.I.; Rahman, M.A.; Tanaka, T.; Guo, Q. Influence of the substrate, process conditions, and postannealing temperature on the properties of ZnO thin films grown by the successive ionic layer adsorption and reaction method. ACS Omega 2021, 6, 2665–2674. [Google Scholar] [CrossRef]
- Farhad, S.F.U.; Majumder, S.; Hossain, M.A.; Tanvir, N.I.; Akter, R.; Patwary, M.A. Effect of solution pH and post-annealing temperatures on the optical bandgap of the copper oxide thin films grown by modified SILAR method. MRS Adv. 2019, 4, 937–944. [Google Scholar] [CrossRef]
- Lobinsky, A.A.; Popkov, V.I. Direct SILD synthesis of efficient electroactive materials based on ultrathin nanosheets of amorphous CoCr-LDH. Mater. Lett. 2022, 322, 132472. [Google Scholar] [CrossRef]
- Kodintsev, I.A.; Martinson, K.D.; Lobinsky, A.A.; Popkov, V.I. Successive ionic layer deposition of co-doped Cu(OH)2 nanorods as electrode material for electrocatalytic reforming of ethanol. Nanosyst. Phys. Chem. Math. 2019, 10, 573–578. [Google Scholar] [CrossRef]
- Lobinsky, A.A.; Kaneva, M.V. Synthesis Ni-doped CuO nanorods via successive ionic layer deposition method and their capacitive performance. Nanosyst. Phys. Chem. Math. 2020, 11, 608–614. [Google Scholar] [CrossRef]
- Lobinsky, A.A.; Kodintsev, I.A.; Tenevich, M.I.; Popkov, V.I. A novel oxidation–reduction route for the morphology-controlled synthesis of manganese oxide Nanocoating as highly Effective Material for Pseudocapacitors. Coatings 2023, 13, 361. [Google Scholar] [CrossRef]
- Shameem, A.; Devendran, P.; Siva, V.; Raja, M.; Bahadur, S.A.; Manikandan, A. Preparation and characterization studies of nanostructured CdO thin films by SILAR method for photocatalytic applications. J. Inorg. Organomet. Polym. Mater. 2017, 27, 692–699. [Google Scholar] [CrossRef]
- Eswaran, M.; Swamiappan, S.; Chokkiah, B.; Dhanusuraman, R.; Bharathkumar, S.; Ponnusamy, V.K. A green and economical approach to derive nanostructured hydroxyapatite from Garra mullya fish scale waste for biocompatible energy storage applications. Mater. Lett. 2021, 302, 130341. [Google Scholar] [CrossRef]
- Deng, W.; Li, Z.; Ye, Y.; Zhou, Z.; Li, Y.; Zhang, M.; Yuan, X.; Hu, J.; Zhao, W.; Huang, Z.; et al. Zn2+ Induced Phase Transformation of K2MnFe(CN)6 Boosts Highly Stable Zinc-Ion Storage. Adv. Energy Mater. 2021, 11, 2003639. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.M.; Gerson, A.R.; Smart, R.S.C. Resolving surface chemical states in XPS analysis of first-row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717–2730. [Google Scholar] [CrossRef]
- Yang, T.; Gao, J.; Zhou, L.; He, Q.; Chen, S.; He, P.; Zhang, H. Tailored manganese hexacyanoferrate/graphene oxide nanocomposites: One-pot facile synthesis and favorable capacitance behavior for supercapacitors. J. Mater. Sci. Mater. Electron. 2020, 31, 2720–2728. [Google Scholar] [CrossRef]
- Wang, J.-G.; Ren, L.; Hou, Z.; Shao, M. Flexible reduced graphene oxide/prussian blue films for hybrid supercapacitors. Chem. Eng. J. 2020, 397, 125521. [Google Scholar] [CrossRef]
- Yin, X.; Li, H.; Wang, H.; Zhang, Z.; Yuan, R.; Lu, J.; Fu, Q. Self-Templating Synthesis of Cobalt Hexacyanoferrate Hollow Structures with Superior Performance for Na-Ion Hybrid Supercapacitors. ACS Appl. Mater. Interfaces 2018, 10, 29496–29504. [Google Scholar] [CrossRef]
- Zhao, F.; Wang, Y.; Xu, X.; Liu, Y.; Song, R.; Lu, G.; Li, Y. Cobalt Hexacyanoferrate Nanoparticles as a High-Rate and Ultra-Stable Supercapacitor Electrode Material. ACS Appl. Mater. Interfaces 2014, 6, 11007–11012. [Google Scholar] [CrossRef]
- Sun, X.; Li, S.; Liu, R.; Sun, X.; Liu, X.; Li, A.; Li, W. A three-dimensional heterogeneous ZnCo-PBA@α-Co(OH)2 nanostructure for high-performance supercapacitors. J. Nanoparticle Res. 2020, 22, 1–12. [Google Scholar] [CrossRef]
- Ren, J.; Meng, Q.; Xu, Z.; Zhang, X.; Chen, J. CoS2 hollow nanocubes derived from Co-Co Prussian blue analogue: High-performance electrode materials for supercapacitors. J. Electroanal. Chem. 2019, 836, 30–37. [Google Scholar] [CrossRef]
- Xu, P.; Wang, G.; Wang, H.; Li, Y.; Miao, C.; Qu, J.; Zhang, X. K2.25Ni0.55Co0.37Fe(CN)6 nanoparticle connected by cross-linked carbon nanotubes conductive skeletons for high-performance energy storage. Chem. Eng. J. 2017, 328, 834–843. [Google Scholar] [CrossRef]


| Electrode Material | Electrolyte | Current Density (A/g) | Specific Capacitance (F/g) | Refs. |
|---|---|---|---|---|
| MnHCF/GO | 1.0 M Na2SO4 | 0.3 | 279.3 | [25] |
| PB/rGO films | 1.0 M Na2SO4 | 0.3 | 286.0 | [26] |
| CoHCF | 0.5 M Na2SO4 | 1.0 | 284.0 | [27] |
| CoHCF | 0.5 M Na2SO4 | 1.0 | 250.0 | [28] |
| ZnCo-PBA@α-Co(OH)2 | 1.0 M KOH | 1.0 | 423.9 | [29] |
| CoS2-derived Co-Co PBA | 2.0 M KOH | 1.0 | 936.0 | [30] |
| K2.25Ni0.55Co0.37Fe(CN)6/CNTs | 4.0 M KCl | 0.2 | 600.0 | [31] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lobinsky, A.A.; Kaneva, M.V.; Tenevich, M.I.; Popkov, V.I. Direct Synthesis of Mn3[Fe(CN)6]2·nH2O Nanosheets as Novel 2D Analog of Prussian Blue and Material for High-Performance Metal-Ion Batteries. Micromachines 2023, 14, 1083. https://doi.org/10.3390/mi14051083
Lobinsky AA, Kaneva MV, Tenevich MI, Popkov VI. Direct Synthesis of Mn3[Fe(CN)6]2·nH2O Nanosheets as Novel 2D Analog of Prussian Blue and Material for High-Performance Metal-Ion Batteries. Micromachines. 2023; 14(5):1083. https://doi.org/10.3390/mi14051083
Chicago/Turabian StyleLobinsky, Artem A., Maria V. Kaneva, Maxim I. Tenevich, and Vadim I. Popkov. 2023. "Direct Synthesis of Mn3[Fe(CN)6]2·nH2O Nanosheets as Novel 2D Analog of Prussian Blue and Material for High-Performance Metal-Ion Batteries" Micromachines 14, no. 5: 1083. https://doi.org/10.3390/mi14051083
APA StyleLobinsky, A. A., Kaneva, M. V., Tenevich, M. I., & Popkov, V. I. (2023). Direct Synthesis of Mn3[Fe(CN)6]2·nH2O Nanosheets as Novel 2D Analog of Prussian Blue and Material for High-Performance Metal-Ion Batteries. Micromachines, 14(5), 1083. https://doi.org/10.3390/mi14051083





