Metallic Structures Based on Zinc Oxide Film for Enzyme Biorecognition
Abstract
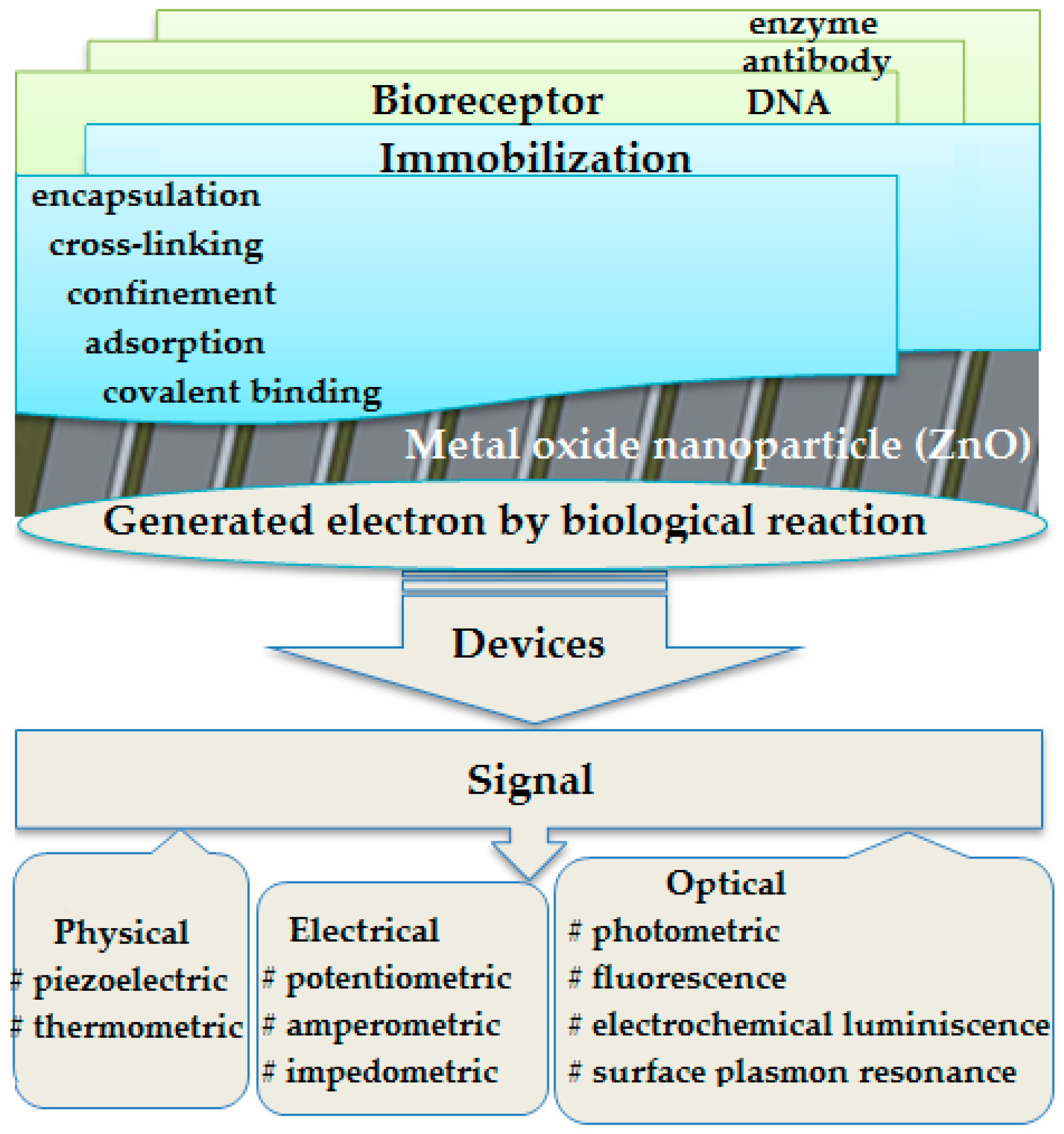
1. Introduction
2. Materials and Methods
2.1. Preparation of Ag/ZnO Structures
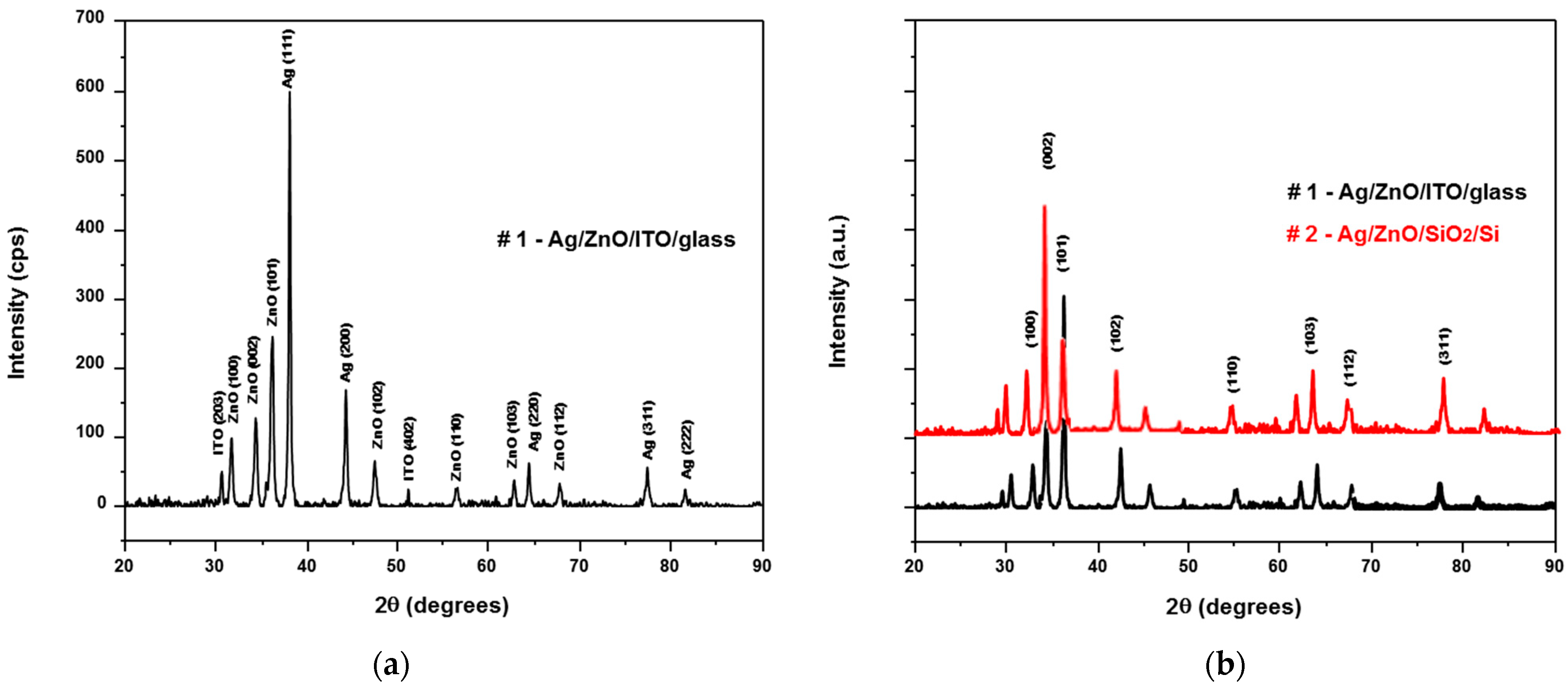
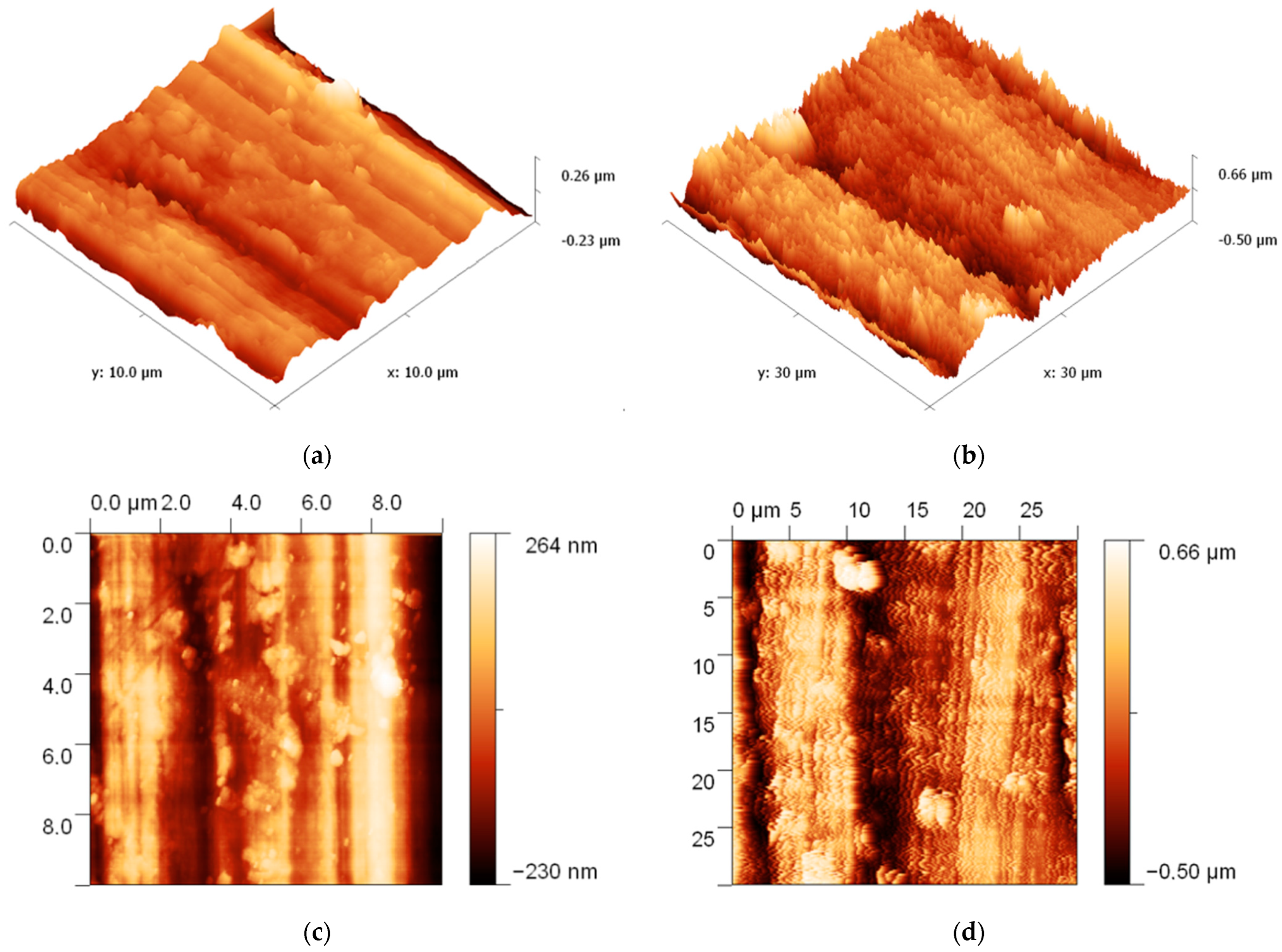
2.2. Characterization of Ag/ZnO Structures
3. Experimental Set-Up and Simulations
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Turner, A.P.F. Biosensors: Sense and sensibility. Chem. Soc. Rev. 2013, 42, 3184–3196. [Google Scholar] [CrossRef] [PubMed]
- Dzyadevych, S.V.; Arkhypova, V.N.; Soldatkin, A.P.; El’skaya, A.V.; Martelet, C.; Jaffrezic-Renault, N. Amperometric enzyme biosensors: Past, present and future. IRBM 2008, 29, 171–180. [Google Scholar] [CrossRef]
- Zhao, Z.; Lei, W.; Zhang, X.; Wang, B.; Jiang, H. ZnO-based amperometric enzyme biosensors. Sensors 2010, 10, 1216–1231. [Google Scholar] [CrossRef]
- Ariga, K.; Mori, Q.J.T.; Naito, M.; Yamauchi, Y.; Abe, H.; Hill, J.P. Enzyme nanoarchitectonics: Organization and device application. Chem. Soc. Rev. 2013, 42, 6322–6345. [Google Scholar] [CrossRef]
- Lee, S.H.; Sung, J.H.; Park, T.H. Nanomaterial-based biosensor as an emerging tool for biomedical applications. Ann. Biomed. Eng. 2012, 40, 1384–1397. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Wang, J.; Shi, J.; Deng, K.; Fu, W. rhEPO/EPO discrimination with ultrasensitive electrochemical biosensor based on sandwich-typenano-Au/ZnOsol–gel/nano-Au signal amplification. Biosensensors Bioelectron. 2013, 50, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Rambu, A.P.; Iftimie, N.; Nica, V. Effect of in incorporation on the structural, electrical, and gas sensing properties of ZnO films. J. Mater. Sci. 2012, 47, 6979–6985. [Google Scholar] [CrossRef]
- Rambu, A.P.; Iftimie, N.; Rusu, G.I. Influence of the substrate nature on the properties of ZnO thin films. Mater. Sci. Eng. B 2012, 177, 157–163. [Google Scholar] [CrossRef]
- Rambu, A.P.; Sirbu, D.; Iftimie, N.; Rusu, G.I. Polycrystalline ZnO–In2O3 thin films as gas sensors. Thin Solid Film. 2011, 520, 1303–1307. [Google Scholar] [CrossRef]
- Steigmann, R.; Iftimie, N.; Savin, A. Characterization of the Thin Films Structures in Subwavelength Regime as Biosensing Materials. Appl. Mech. Mater. 2015, 772, 62–66. [Google Scholar] [CrossRef]
- Sassolas, A.; Blum, L.J.; Leca-Bouvier, B.D. Immobilization strategies to develop Enzymatic biosensors. Biotechnol. Adv. 2012, 30, 489–511. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.; Khranovskyy, V.; Söderlind, F.; Käll, P.O.; Yakimova, R.; Spetz, A.L. ZnO nanoparticles or ZnO films: A comparison of the gas sensing capabilities. Sens. Actuators B 2009, 137, 94–102. [Google Scholar] [CrossRef]
- Wang, J.; Sun, X.; Wei, A.; Lei, Y.; Cai, X.; Li, C.; Dong, Z. Zinc oxide nanocomb biosensor for glucose detection. Appl. Phys. Lett. 2006, 88, 233106. [Google Scholar] [CrossRef]
- Pendry, J.J.B.; Holden, A.J.; Stewart, W.J.; Youngs, I. Extremely Low Frequency Plasmons in Metallic Mesostructures. Phys. Rev. Lett. 1996, 76, 4773–4776. [Google Scholar] [CrossRef]
- Grimberg, R. Electromagnetic metamaterials. Mater. Sci. Eng. B 2013, 178, 1285–1295. [Google Scholar] [CrossRef]
- Grimberg, R.; Savin, A.; Steigmann, R.; Serghiac, B.; Bruma, A. Electromagnetic non-destructive evaluation using metamaterials. Insight 2011, 53, 132–137. [Google Scholar] [CrossRef]
- Pendry, J.B.; Holden, A.J.; Robbins, D.J.; Stewart, W.J. Magnetism from conductors and enhanced nonlinear phenomena. IEEE Trans. Microw. Theory Tech. 1999, 47, 2075–2084. [Google Scholar] [CrossRef]
- Pendry, J.B. Negative refraction makes a perfect lens. Phys. Rev. Lett. 2000, 85, 3966. [Google Scholar] [CrossRef]
- Sievenpiper, D.; Zhang, L.; Broas, R.F.; Alexopolous, N.G.; Yablonovitch, E. High-impedance electromagnetic surfaces with a forbidden frequency band. IEEE Trans. Microw. Theory Tech. 1999, 47, 2059–2074. [Google Scholar] [CrossRef]
- Pendry, J.B.; Schurig, D.; Smith, D.R. Controlling electromagnetic fields. Science 2006, 312, 1780–1782. [Google Scholar] [CrossRef]
- Savin, A.; Steigmann, R.; Bruma, A. Metallic Strip Gratings in the Sub-Subwavelength Regime. Sensors 2014, 14, 11786–11804. [Google Scholar] [CrossRef] [PubMed]
- Kabashin, A.V.; Evans, P.; Pastkovsky, S.; Hendren, W.; Wurtz, G.A.; Atkinson, R.; Pollard, R.; Podolskiy, V.A.; Zayats, A.V. Plasmonic nanorod metamaterials for biosensing. Nat. Mater. 2009, 8, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Kakati, N.; Jee, S.H.; Kim, S.H.; Oh, J.Y.; Yoon, Y.S. Thickness dependency of sol-gel derived ZnO thin films on gas sensing behaviors. Thin Solid Film. 2010, 519, 494–498. [Google Scholar] [CrossRef]
- Dai, L.; Deng, H.; Chen, G. Ultraviolet emission properties of ZnO film with zinc deficiency by SS CVD. J. Appl. Surf. Sci. 2008, 254, 1599–1603. [Google Scholar] [CrossRef]
- Flickyngerova, S.; Shtereva, K.; Stenova, V.; Hasko, D.; Novotny, I.; Tvarozek, V.; Sutta, P.; Vavrinsky, E. Structural and optical properties of sputtered ZnO thin films. Appl. Surf. Sci. 2007, 254, 3643–3647. [Google Scholar] [CrossRef]
- Iftimie, N.; Tascu, S.; Salaoru, I.; Steigmann, R.; Savin, A.; Irimia, M.; Iacomi, F. The evanescent waves in metallic strip gratings and complex structures in subwavelength regime. Mater. Today Proc. 2015, 2, 3486–3852. [Google Scholar] [CrossRef]
- Alvarado, J.A.; Maldonado, A.; Juarez, H.; Pacio, M.; Perez, R. Characterization of nanostructured ZnO thin films deposited through vacuum evaporation. Beilstein J. Nanotechnol. 2015, 6, 971–975. [Google Scholar] [CrossRef]
- Bensmaine, S.; Benyoucef, B. Experimental Characterization of ZnO Thins Films and Identification of Frequency Peaks in ZnO/SiO2/Si SAW Devices. Am. J. Mater. Sci. 2013, 3, 100–103. [Google Scholar]
- Bensmaine, S.; Benyoucef, B. Effect of the temperature on ZnO thin films deposited by r.f. magnetron. Phys. Procedia 2014, 55, 144–149. [Google Scholar] [CrossRef][Green Version]
- Sainz, R.; del Pozo, M.; Vazquez, L.; Vilas-Varela, M.; Castro-Esteban, J.; Blanco, E.; Petit-Domínguez, M.D.; Quintana, C.; Casero, E. Lactate biosensing based on covalent immobilization of lactate oxidase onto chevron-like5etamate nanoribbons via diazotization-coupling reaction. Anal. Chim. Acta 2022, 1208, 339851. [Google Scholar] [CrossRef]
- Paliwal, A.; Tomar, M.; Gupta, V. Table top surface plasmon resonance measurement system for efficient urea biosensing using ZnO thin film matrix. J. Biomed. Opt. 2016, 21, 087006. [Google Scholar] [CrossRef] [PubMed]
- Tomar, B.M.; Gupta, V. Al:ZnO thin film: An efficient matrix for cholesterol detection. J. Appl. Phys. 2012, 112, 114701. [Google Scholar]
- Malhotra, B.D.; Ali, M.A. Nanomaterials in Biosensors: Fundamentals and Applications. Nanomater. Biosens. 2018, 1–74. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ahammad, A.J.S.; Jin, J.H.; Ahn, S.J.; Lee, J.J. A Comprehensive Review of Glucose Biosensors Based on Nanostructured Metal Oxides-Review. Sensors 2010, 10, 4855–4886. [Google Scholar] [CrossRef]
- Sarma, A.K.; Vatsyayan, P.; Goswami, P.; Minteerc, S.D. Recent advances in material science for developing enzyme electrodes-Review. Biosens. Bioelectron. 2009, 24, 2313–2322. [Google Scholar] [CrossRef] [PubMed]
- Savin, A.; Steigmann, R.; Bruma, A.; Sturm, R. An electromagnetic sensors with a metamaterial lens for nondestructive evaluation of composite materials. Sensors 2015, 15, 15903–15920. [Google Scholar] [CrossRef]
- Grimberg, R.; Tian, G.Y. High Frequency Electromagnetic Non-destructive Evaluation for High Spatial Resolution using Metamaterial. Proc. R. Soc. A 2012, 468, 3080–3099. [Google Scholar] [CrossRef]
- Savin, A.; Steigmann, R. Electromagnetic Transducer for Assessing Integrity of Braids of Single-Layer Printed Wiring on Flexible Support and of Layered Meso-Structures. Patent RO129801-A0, 2014. [Google Scholar]
- Born, M.; Wolf, E. Principle of Optics, 5th ed.; Pergamon Press: Oxford, UK, 1975. [Google Scholar]
- REMCOM. Available online: https://www.remcom.com/electromagnetic-applications (accessed on 13 November 2022).
- Shlager, K.L.; Schneider, J.B. A selective survey of the finite-difference time-domain literature. IEEE Antennas Propagat. Mag. 1995, 37, 39–56. [Google Scholar] [CrossRef]
- Lagendijk, A.; Van Tiggelen, B.; Wiersma, D.S. Fifty years of Anderson localization. Physics Today 1991, 62, 24–29. [Google Scholar] [CrossRef]
- Grimberg, R.; Savin, A. Electromagnetic Transducer for Evaluation of Structure and Integrity of the Composite Materials with Polymer Matrix Reinforced with Carbon Fibers. Patent RO126245-A0, 2011. [Google Scholar]
- Peterson, A.F.; Ray, S.L.; Mittra, R. Computational Methods for Electromagnetics; Institute of Electrical and Electronics Engineers 2: IEEE Press: New York, NY, USA, 1998; ISBN 978-0-780-31122-0. [Google Scholar]
- Smith, D.R.; Pendry, J.B.; Wiltshire, M.C. Metamaterials and Negative Refractive Index. Science 2004, 305, 788–792. [Google Scholar] [CrossRef]
- Saha, S.; Gupta, V. Influence of surface defects in ZnO thin films on its biosensing response characteristic. J. Appl. Phys. 2011, 110, 064904. [Google Scholar] [CrossRef]
- Liu, C.C. Electrochemical based biosensors. Biosensors 2012, 2, 269–272. [Google Scholar] [CrossRef] [PubMed]
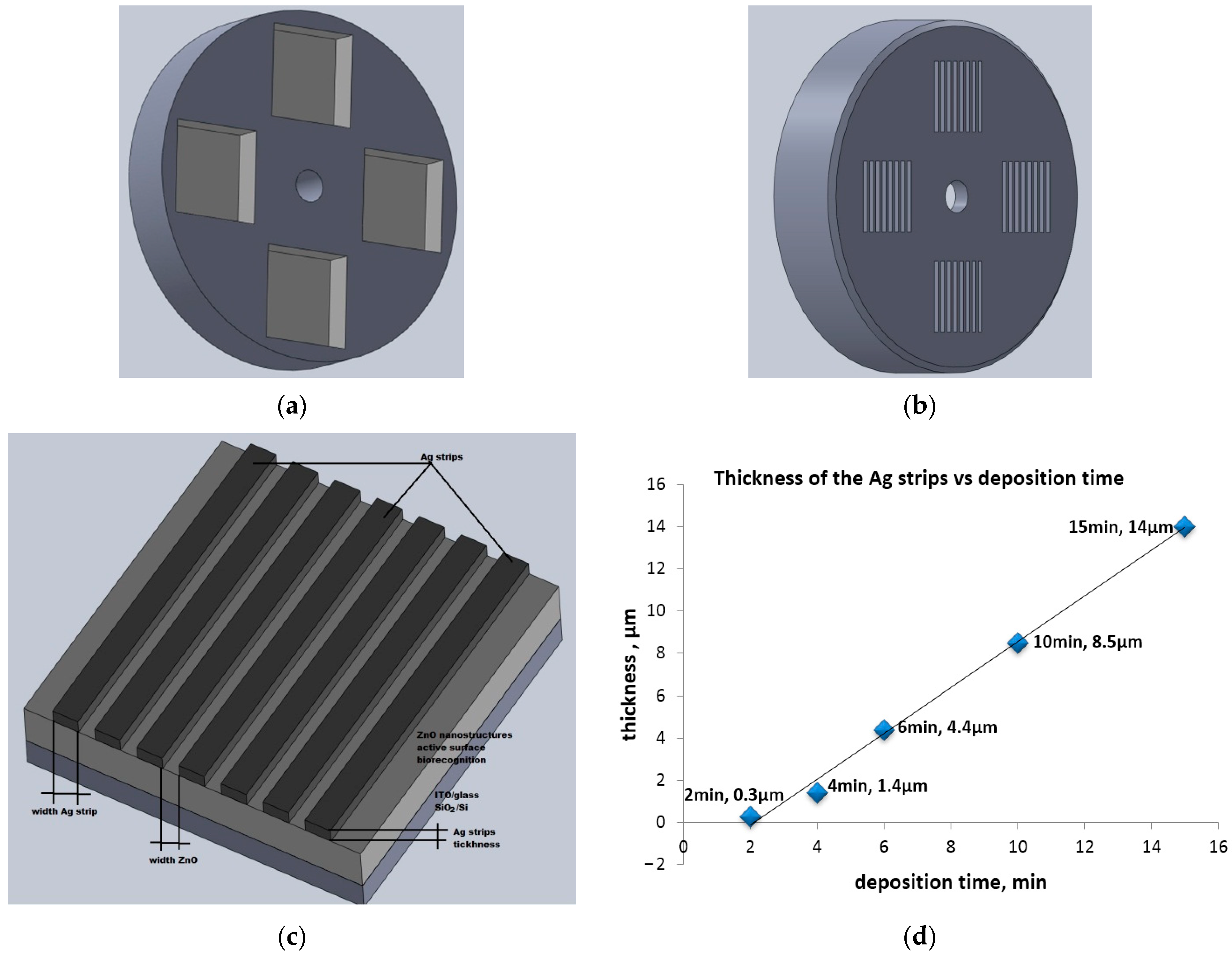


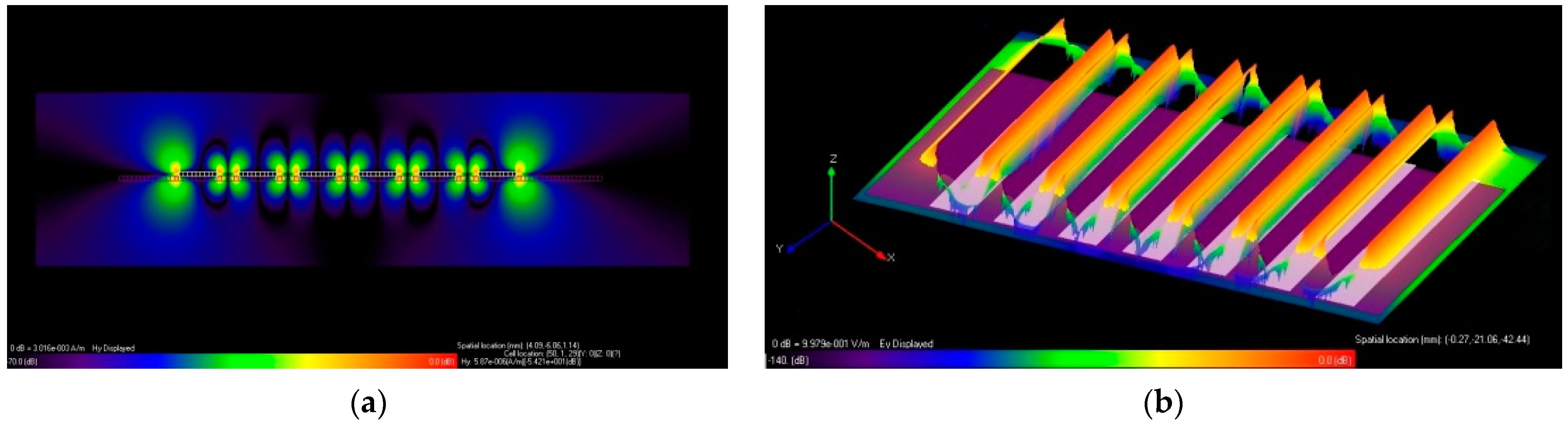

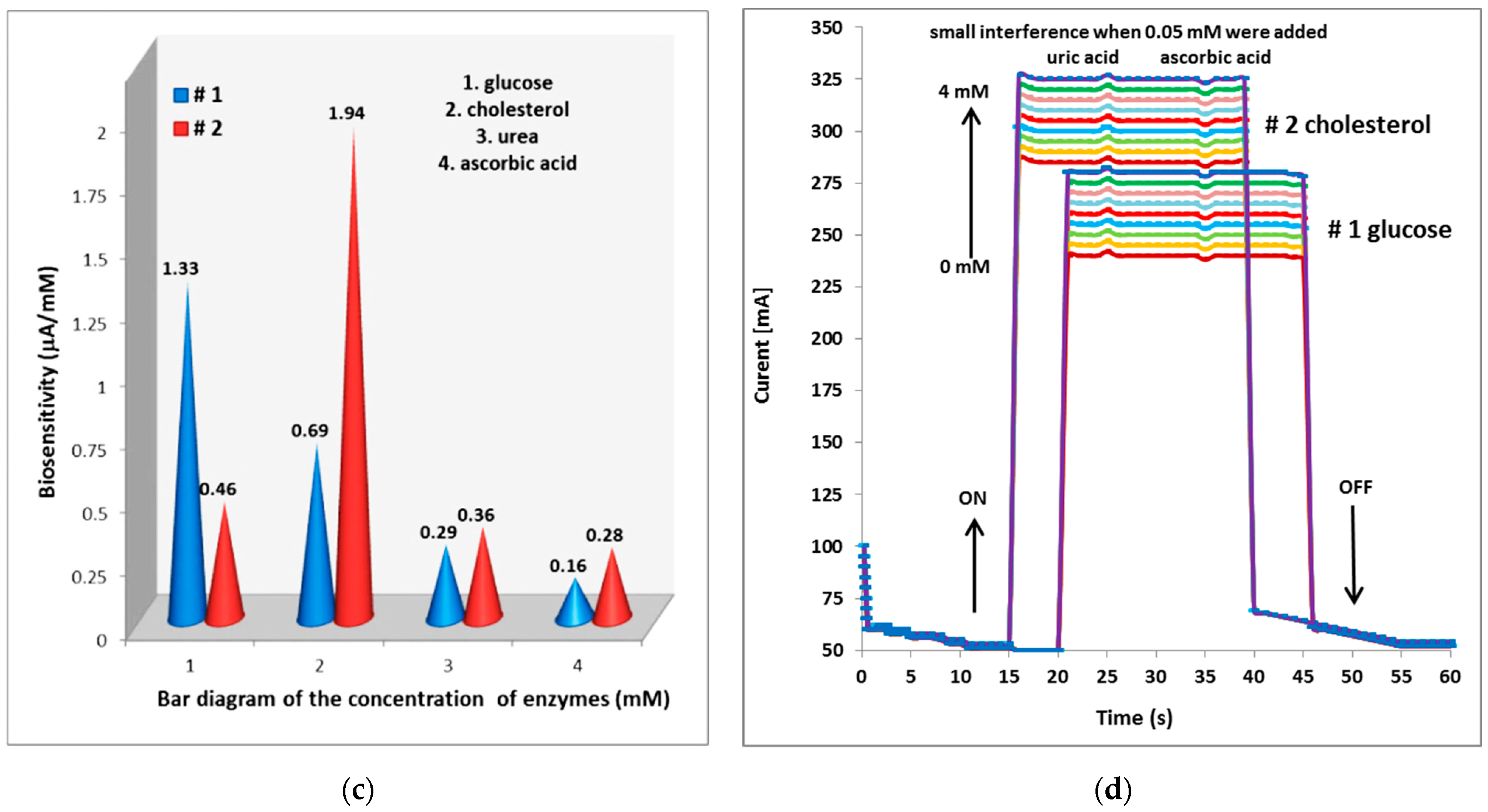
| No. Sets | Metallic Strip Grating Structures (MSG) | Deposited Time [min] | Width (Ag) [mm] | Width (ZnO) [mm] | Thickness Ag Strip [μm] |
|---|---|---|---|---|---|
| set I | #1 [Ag/ZnO/ITO/glass] | 2 | [0.9–1.2] | [0.6–0.8] | 0.9 |
| #2 [Ag/ZnO/SiO2/Si] | |||||
| #3 [Ag/ZnO/glass] | |||||
| #4 [Ag/ZnO/nSi[100]] | |||||
| set 2 | #1 [Ag/ZnO/ITO/glass] | 4 | [0.9–1.2] | [0.6–0.8] | 1.4 |
| #2 [Ag/ZnO/SiO2/Si] | |||||
| #3 [Ag/ZnO/glass] | |||||
| #4 [Ag/ZnO/nSi[100]] | |||||
| set 3 | #1 [Ag/ZnO/ITO/glass] | 6 | [0.9–1.2] | [0.6–0.8] | 4.4 |
| #2 [Ag/ZnO/SiO2/Si] | |||||
| #3 [Ag/ZnO/glass] | |||||
| #4 [Ag/ZnO/nSi[100]] | |||||
| set 4 | #1 [Ag/ZnO/ITO/glass] | 10 | [0.9–1.2] | [0.6–0.8] | 8.5 |
| #2 [Ag/ZnO/SiO2/Si] | |||||
| #3 [Ag/ZnO/glass] | |||||
| #4 [Ag/ZnO/nSi[100]] | |||||
| set 5 | #1 [Ag/ZnO/ITO/glass] | 15 | [0.9–1.2] | [0.6–0.8] | 14 |
| #2 [Ag/ZnO/SiO2/Si] | |||||
| #3 [Ag/ZnO/glass] | |||||
| #4 [Ag/ZnO/nSi[100]] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iftimie, N.; Steigmann, R.; Faktorova, D.; Savin, A. Metallic Structures Based on Zinc Oxide Film for Enzyme Biorecognition. Micromachines 2022, 13, 1997. https://doi.org/10.3390/mi13111997
Iftimie N, Steigmann R, Faktorova D, Savin A. Metallic Structures Based on Zinc Oxide Film for Enzyme Biorecognition. Micromachines. 2022; 13(11):1997. https://doi.org/10.3390/mi13111997
Chicago/Turabian StyleIftimie, Nicoleta, Rozina Steigmann, Dagmar Faktorova, and Adriana Savin. 2022. "Metallic Structures Based on Zinc Oxide Film for Enzyme Biorecognition" Micromachines 13, no. 11: 1997. https://doi.org/10.3390/mi13111997
APA StyleIftimie, N., Steigmann, R., Faktorova, D., & Savin, A. (2022). Metallic Structures Based on Zinc Oxide Film for Enzyme Biorecognition. Micromachines, 13(11), 1997. https://doi.org/10.3390/mi13111997







