Determining the Dose–Response Curve of Exoelectrogens: A Microscale Microbial Fuel Cell Biosensor for Water Toxicity Monitoring
Abstract
:1. Introduction
2. Materials and Methods
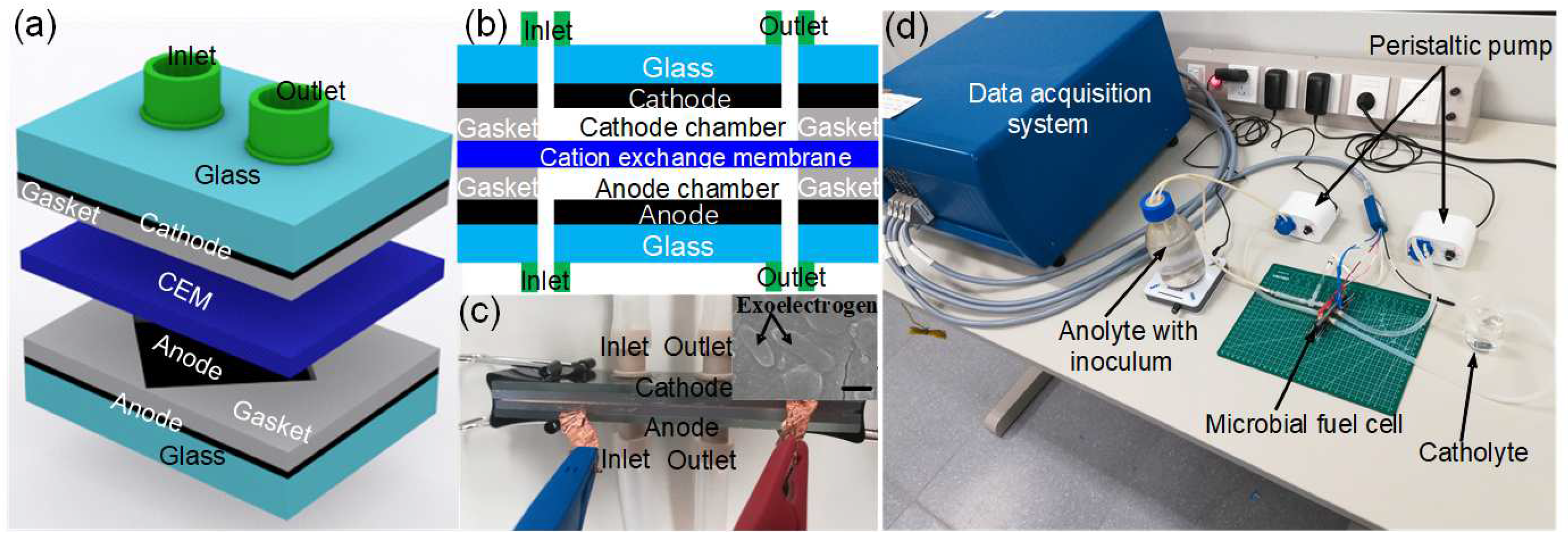
2.1. Operation Principle of the Microscale MFC Biosensor
2.2. Fabrication of the Microscale MFC Biosensor
2.3. Inoculum and Operation of the Microscale MFC Biosensor
2.4. Data Acquisition and Calculation
2.5. Formaldehyde Injection and Output Current Monitoring
2.6. Dose–Response Curve Fitting
2.7. Biofilm Fixation of the Microscale MFC Biosensor
3. Results and Discussion
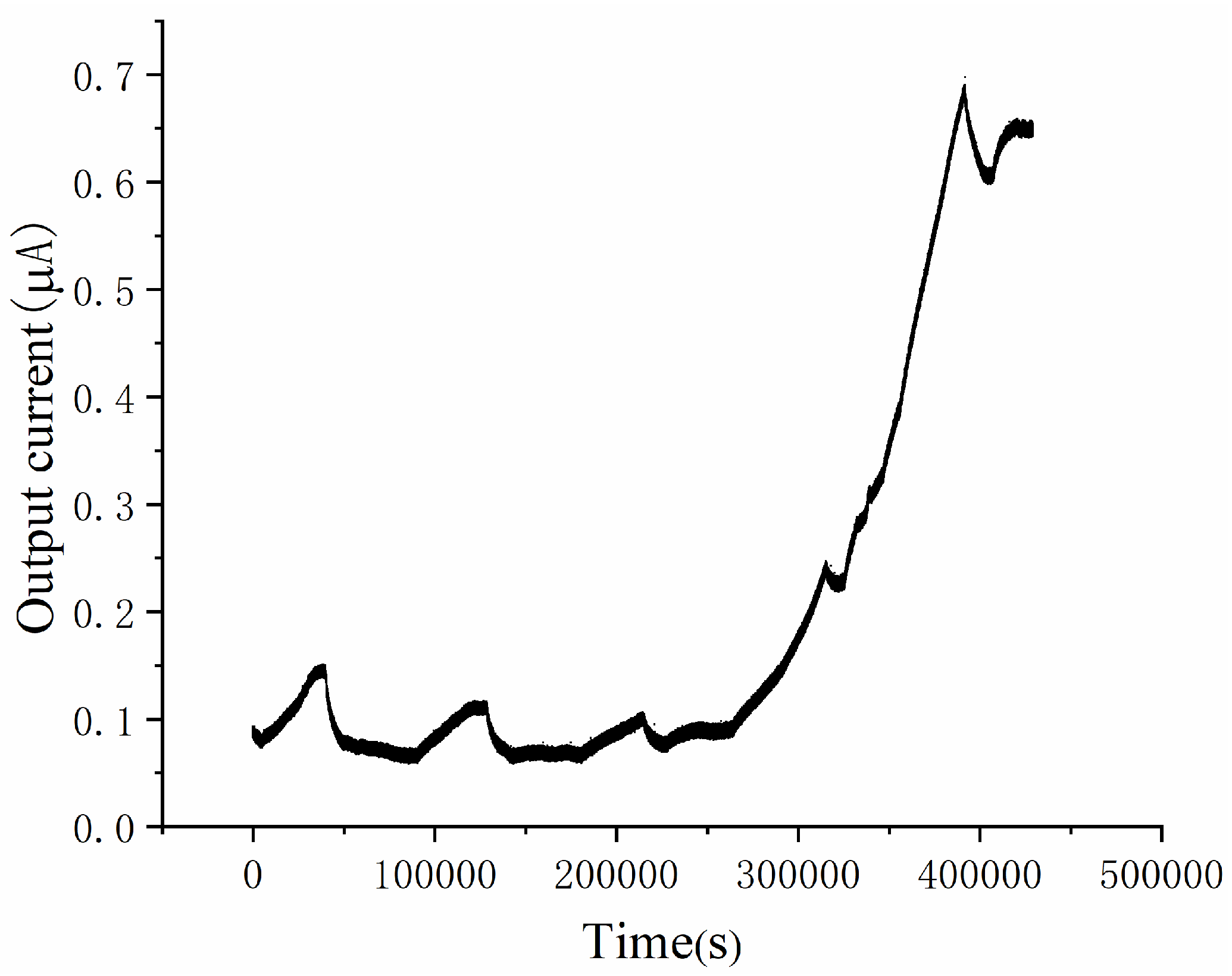
3.1. Start-Up Process of the Microscale MFC Biosensor
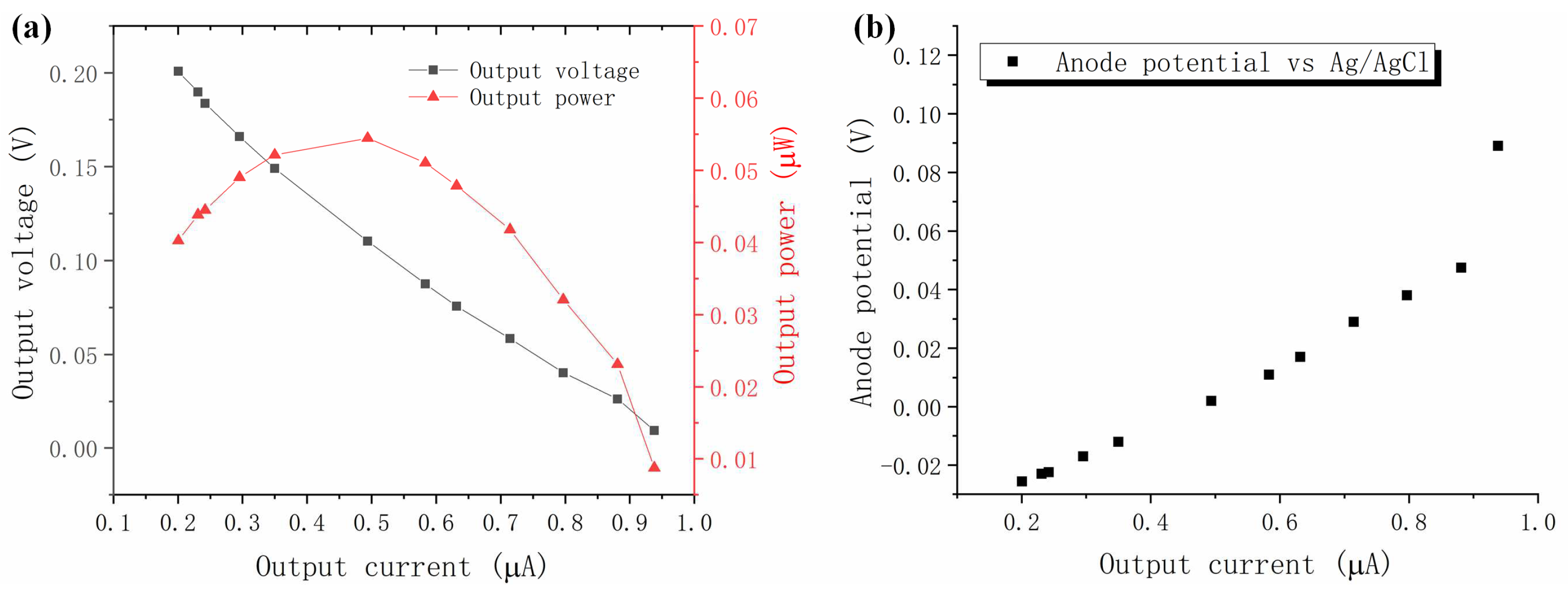
3.2. Polarization Curve of the Microscale MFC Biosensor
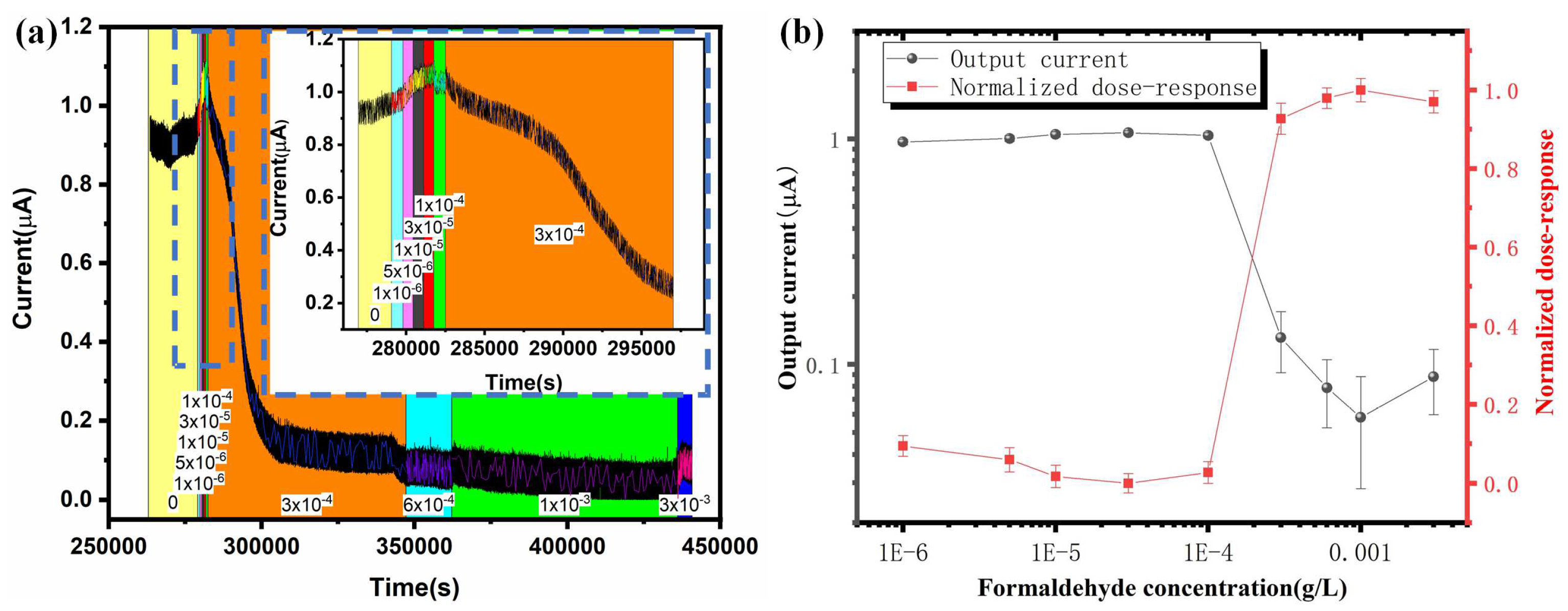
3.3. Microscale MFC Biosensor Water Toxicity Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mateo-Sagasta, J.; Raschid-Sally, L.; Thebo, A. Global wastewater and sludge production, treatment and use. In Wastewater; Springer: Berlin/Heidelberg, Germany, 2015; pp. 15–38. [Google Scholar]
- Mateo-Sagasta, J.; Zadeh, S.M.; Turral, H. More People, More Food, Worse Water? A Global Review of Water Pollution from Agriculture; FAO: Rome, Italy, 2018. [Google Scholar]
- Sato, T.; Qadir, M.; Yamamoto, S.; Endo, T.; Zahoor, A.J.A.W.M. Global, regional, and country level need for data on wastewater generation, treatment, and use. Agric. Water Manag. 2013, 130, 1–13. [Google Scholar] [CrossRef]
- Ryder, G. The United Nations World Water Development Report, 2017: Wastewater: The Untapped Resource; UNESCO: Paris, France, 2017. [Google Scholar]
- Vikesland, P.J. Nanosensors for water quality monitoring. Nat. Nanotechnol. 2018, 13, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Deng, A.; Himmelsbach, M.; Zhu, Q.-Z.; Frey, S.; Sengl, M.; Buchberger, W.; Niessner, R.; Knopp, D. Residue Analysis of the Pharmaceutical Diclofenac in Different Water Types Using ELISA and GC−MS. Environ. Sci. Technol. 2003, 37, 3422–3429. [Google Scholar] [CrossRef] [PubMed]
- Wylie, D.E.; Carlson, L.D.; Carlson, R.; Wagner, F.W.; Schuster, S.M. Detection of mercuric ions in water by ELISA with a mercury-specific antibody. Anal. Biochem. 1991, 194, 381–387. [Google Scholar] [CrossRef]
- Spoof, L.; Vesterkvist, P.; Lindholm, T.; Meriluoto, J. Screening for cyanobacterial hepatotoxins, microcystins and nodularin in environmental water samples by reversed-phase liquid chromatography–electrospray ionisation mass spectrometry. J. Chromatogr. A 2003, 1020, 105–119. [Google Scholar] [CrossRef]
- van de Merwe, J.P.; Leusch, F.D.L. A sensitive and high throughput bacterial luminescence assay for assessing aquatic toxicity—The BLT-Screen. Environ. Sci. Processes Impacts 2015, 17, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, W.; Ren, H.; Chae, J. Detection of copper ions in drinking water using the competitive adsorption of proteins. Biosens. Bioelectron. 2014, 57, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Kuklina, I.; Kouba, A.; Kozák, P. Real-time monitoring of water quality using fish and crayfish as bio-indicators: A review. Environ. Monit. Assess. 2013, 185, 5043–5053. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Lee, H.-S.; Chae, J. Miniaturizing microbial fuel cells for potential portable power sources: Promises and challenges. Microfluid. Nanofluid. 2012, 13, 353–381. [Google Scholar] [CrossRef]
- Ren, H.; Torres, C.I.; Parameswaran, P.; Rittmann, B.E.; Chae, J. Improved current and power density with a micro-scale microbial fuel cell due to a small characteristic length. Biosens. Bioelectron. 2014, 61, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Dziegielowski, J.; Metcalfe, B.; Di Lorenzo, M. Towards effective energy harvesting from stacks of soil microbial fuel cells. J. Power Sources 2021, 515, 230591. [Google Scholar] [CrossRef]
- Babanova, S.; Santoro, C.; Jones, J.; Phan, T.; Serov, A.; Atanassov, P.; Bretschger, O. Practical demonstration of applicability and efficiency of platinum group metal-free based catalysts in microbial fuel cells for wastewater treatment. J. Power Sources 2021, 491, 229582. [Google Scholar] [CrossRef]
- Liu, W.; Lin, L.; Meng, Y.; Zhao, Z.; Wang, Y.; Luan, F. Recovery and separation of uranium in a microbial fuel cell using a titanium dioxide nanotube array cathode. Environ. Sci. Nano 2021, 8, 2214–2222. [Google Scholar] [CrossRef]
- Reimers, C.E.; Wolf, M.; Alleau, Y.; Li, C. Benthic microbial fuel cell systems for marine applications. J. Power Sources 2022, 522, 231033. [Google Scholar] [CrossRef]
- Sonawane, J.M.; Ezugwu, C.I.; Ghosh, P.C. Microbial Fuel Cell-Based Biological Oxygen Demand Sensors for Monitoring Wastewater: State-of-the-Art and Practical Applications. ACS Sens. 2020, 5, 2297–2316. [Google Scholar] [CrossRef]
- Pasternak, G.; Greenman, J.; Ieropoulos, I. Self-powered, autonomous Biological Oxygen Demand biosensor for online water quality monitoring. Sens. Actuators B Chem. 2017, 244, 815–822. [Google Scholar] [CrossRef]
- Hou, H.; Li, L.; Cho, Y.; de Figueiredo, P.; Han, A. Microfabricated Microbial Fuel Cell Arrays Reveal Electrochemically Active Microbes. PLoS ONE 2009, 4, e6570. [Google Scholar] [CrossRef]
- Tahernia, M.; Mohammadifar, M.; Gao, Y.; Panmanee, W.; Hassett, D.J.; Choi, S. A 96-well high-throughput, rapid-screening platform of extracellular electron transfer in microbial fuel cells. Biosens. Bioelectron. 2020, 162, 112259. [Google Scholar] [CrossRef]
- Olias, L.G.; Di Lorenzo, M. Microbial fuel cells for in-field water quality monitoring. RSC Adv. 2021, 11, 16307–16317. [Google Scholar] [CrossRef]
- Choi, S. Microscale microbial fuel cells: Advances and challenges. Biosens. Bioelectron. 2015, 69, 8–25. [Google Scholar] [CrossRef]
- Xiao, N.; Wu, R.; Huang, J.J.; Selvaganapathy, P.R. Development of a xurographically fabricated miniaturized low-cost, high-performance microbial fuel cell and its application for sensing biological oxygen demand. Sens. Actuators B Chem. 2020, 304, 127432. [Google Scholar] [CrossRef]
- Wang, C.-T.; Ubando, A.T.; Katiyar, V.; Li, T.T.; Lin, Y.-A.; Culaba, A.B.; Jang, J.-H. Feasibility study on a mini autonomous biosensor based on microbial fuel cell for monitoring hexavalent chromium in wastewater. Int. J. Energy Res. 2021, 45, 6293–6302. [Google Scholar] [CrossRef]
- Dávila, D.; Esquivel, J.P.; Sabaté, N.; Mas, J. Silicon-based microfabricated microbial fuel cell toxicity sensor. Biosens. Bioelectron. 2011, 26, 2426–2430. [Google Scholar] [CrossRef] [PubMed]
- Chouler, J.; Cruz-Izquierdo, Á.; Rengaraj, S.; Scott, J.L.; Di Lorenzo, M. A screen-printed paper microbial fuel cell biosensor for detection of toxic compounds in water. Biosens. Bioelectron. 2018, 102, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.H.; Gao, Y.; Ryu, J.; Choi, S. Portable, Disposable, Paper-Based Microbial Fuel Cell Sensor Utilizing Freeze-Dried Bacteria for In Situ Water Quality Monitoring. ACS Omega 2020, 5, 13940–13947. [Google Scholar] [CrossRef] [PubMed]
- Welp, G.; Brümmer, G.W. Toxicity of Increased Amounts of Chemicals and the Dose–Response Curves for Heterogeneous Microbial Populations in Soil. Ecotoxicol. Environ. Saf. 1997, 37, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Dalzell, D.J.B.; Alte, S.; Aspichueta, E.; de la Sota, A.; Etxebarria, J.; Gutierrez, M.; Hoffmann, C.C.; Sales, D.; Obst, U.; Christofi, N. A comparison of five rapid direct toxicity assessment methods to determine toxicity of pollutants to activated sludge. Chemosphere 2002, 47, 535–545. [Google Scholar] [CrossRef]
- Yu, D.; Yong, Y.-C.; Liu, C.; Fang, Y.; Bai, L.; Dong, S. New applications of genetically modified Pseudomonas aeruginosa for toxicity detection in water. Chemosphere 2017, 184, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Li, R.; Sun, X.; Zhang, H.; Yu, H.; Dong, S. Colorimetric and Electrochemical Dual-Signal Method for Water Toxicity Detection Based on Escherichia coli and p-Benzoquinone. ACS Sens. 2021, 6, 2674–2681. [Google Scholar] [CrossRef] [PubMed]
- Olias, L.G.; Otero, A.R.; Cameron, P.J.; Di Lorenzo, M. A soil microbial fuel cell-based biosensor for dissolved oxygen monitoring in water. Electrochim. Acta 2020, 362, 137108. [Google Scholar] [CrossRef]
- Shen, Y.J.; Lefebvre, O.; Tan, Z.; Ng, H.Y. Microbial fuel-cell-based toxicity sensor for fast monitoring of acidic toxicity. Water Sci. Technol. 2012, 65, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Abrevaya, X.C.; Sacco, N.J.; Bonetto, M.C.; Hilding-Ohlsson, A.; Cortón, E. Analytical applications of microbial fuel cells. Part II: Toxicity, microbial activity and quantification, single analyte detection and other uses. Biosens. Bioelectron. 2015, 63, 591–601. [Google Scholar] [CrossRef]
- Jangir, Y.; French, S.; Momper, L.M.; Moser, D.P.; Amend, J.P.; El-Naggar, M.Y. Isolation and Characterization of Electrochemically Active Subsurface Delftia and Azonexus Species. Front. Microbiol. 2016, 7, 756. [Google Scholar] [CrossRef] [PubMed]
- Ritz, C. Toward a unified approach to dose–response modeling in ecotoxicology. Environ. Toxicol. Chem. 2010, 29, 220–229. [Google Scholar] [CrossRef]
- Wu, M.; Wang, X.; Jia, Z.; De Schamphelaere, K.; Ji, D.; Li, X.; Chen, X. Modeling acute toxicity of metal mixtures to wheat (Triticum aestivum L.) using the biotic ligand model-based toxic units method. Sci. Rep. 2017, 7, 9443. [Google Scholar] [CrossRef]
- Schaffner, D.W.; Ross, W.H.; Montville, T.J. Analysis of the influence of environmental parameters on Clostridium botulinum time-to-toxicity by using three modeling approaches. Appl. Environ. Microbiol. 1998, 64, 4416–4422. [Google Scholar] [CrossRef] [PubMed]
- Dudley, R.A.; Edwards, P.; Ekins, R.P.; Finney, D.J.; McKenzie, I.G.; Raab, G.M.; Rodbard, D.; Rodgers, R.P. Guidelines for immunoassay data processing. Clin. Chem. 1985, 31, 1264–1271. [Google Scholar] [CrossRef]
- Ren, H.; Tian, H.; Gardner, C.L.; Ren, T.-L.; Chae, J. A miniaturized microbial fuel cell with three-dimensional graphene macroporous scaffold anode demonstrating a record power density of over 10,000 W m−3. Nanoscale 2016, 8, 3539–3547. [Google Scholar] [CrossRef]
- Ren, H.; Lee, H.-S.; Zhang, J.; Gardner, C.L.; Chae, J. A quantitative extracellular electron transfer (EET) kinetics study of Geobacter sulfurreducens enriched microbial community reveals the transition of EET limiting step during biofilm growth. Int. J. Hydrogen Energy 2021, 46, 3123–3134. [Google Scholar] [CrossRef]
- Ren, H.; Rangaswami, S.; Lee, H.-S.; Chae, J. Enhanced current and power density of micro-scale microbial fuel cells with ultramicroelectrode anodes. J. Micromech. Microeng. 2016, 26, 095016. [Google Scholar] [CrossRef]
- Ringeisen, B.R.; Henderson, E.; Wu, P.K.; Pietron, J.; Ray, R.; Little, B.; Biffinger, J.C.; Jones-Meehan, J.M. High power density from a miniature microbial fuel cell using Shewanella oneidensis DSP10. Environ. Sci. Technol. 2006, 40, 2629–2634. [Google Scholar] [CrossRef] [PubMed]
- Spicher, G.; Peters, J. Microbial resistance to formaldehyde. I. Comparative quantitative studies in some selected species of vegetative bacteria, bacterial spores, fungi, bacteriophages and viruses. Zentralbl. Bakteriol. Orig. B 1976, 163, 486–508. [Google Scholar] [PubMed]
- Piegorsch, W.W.; Bailer, A.J. Quantitative risk assessment with stimulus-response data. Analyzing Environmental Data; Wiley: Chichester, UK, 2005; pp. 171–214. ISBN 978-0-470-84836-4. [Google Scholar]
- Nordberg, G.F.; Fowler, B.A. Chapter 22—Risk Assessment. In Handbook on the Toxicology of Metals, 4th ed.; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 461–486. [Google Scholar]
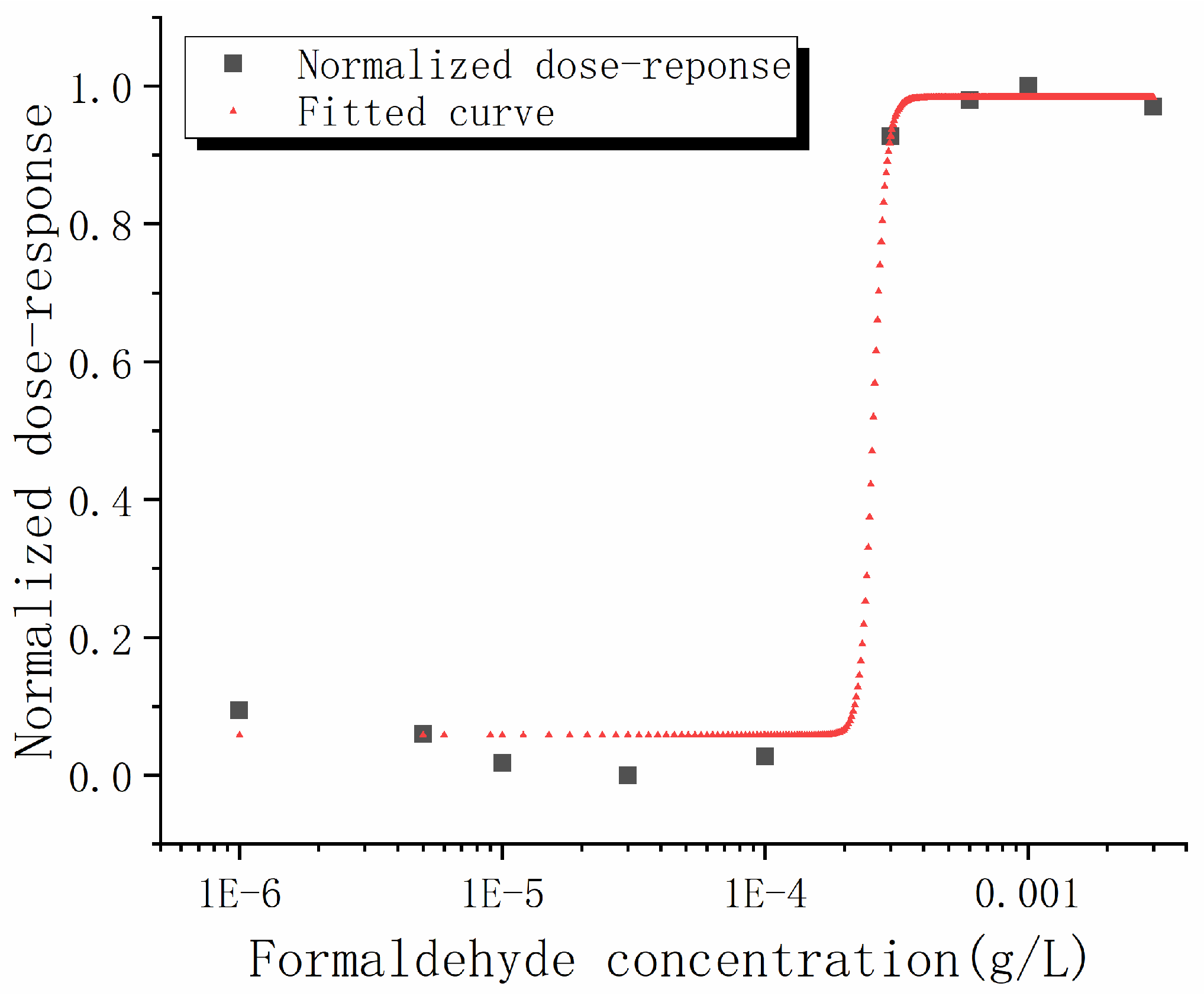
| Fitting Model Parameters | Fitted Values |
|---|---|
| A1 | 0.983 |
| A2 | 0.058 |
| A3 | 18.31 |
| A4 | 2.583 × 10−4 |
| R squared | 0.992 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fei, S.; Ren, H. Determining the Dose–Response Curve of Exoelectrogens: A Microscale Microbial Fuel Cell Biosensor for Water Toxicity Monitoring. Micromachines 2022, 13, 1560. https://doi.org/10.3390/mi13101560
Fei S, Ren H. Determining the Dose–Response Curve of Exoelectrogens: A Microscale Microbial Fuel Cell Biosensor for Water Toxicity Monitoring. Micromachines. 2022; 13(10):1560. https://doi.org/10.3390/mi13101560
Chicago/Turabian StyleFei, Sitao, and Hao Ren. 2022. "Determining the Dose–Response Curve of Exoelectrogens: A Microscale Microbial Fuel Cell Biosensor for Water Toxicity Monitoring" Micromachines 13, no. 10: 1560. https://doi.org/10.3390/mi13101560
APA StyleFei, S., & Ren, H. (2022). Determining the Dose–Response Curve of Exoelectrogens: A Microscale Microbial Fuel Cell Biosensor for Water Toxicity Monitoring. Micromachines, 13(10), 1560. https://doi.org/10.3390/mi13101560





