Author Contributions
Conceptualization, C.-T.P., C.-H.L., J.S.C.J., H.-K.L., C.-N.K. and J.C.H.; data curation, C.-T.P., C.-H.L. and J.C.H.; methodology, Y.-K.H., J.S.C.J., H.-K.L., C.-N.K. and D.-Y.L.; project administration, C.-H.L.; resources, J.S.C.J., H.-K.L., C.-N.K. and D.-Y.L.; software, Y.-K.H.; supervision, J.C.H.; validation, D.-Y.L.; writing—original draft, Y.-K.H.; writing—review and editing, C.-T.P. All authors have read and agreed to the published version of the manuscript.
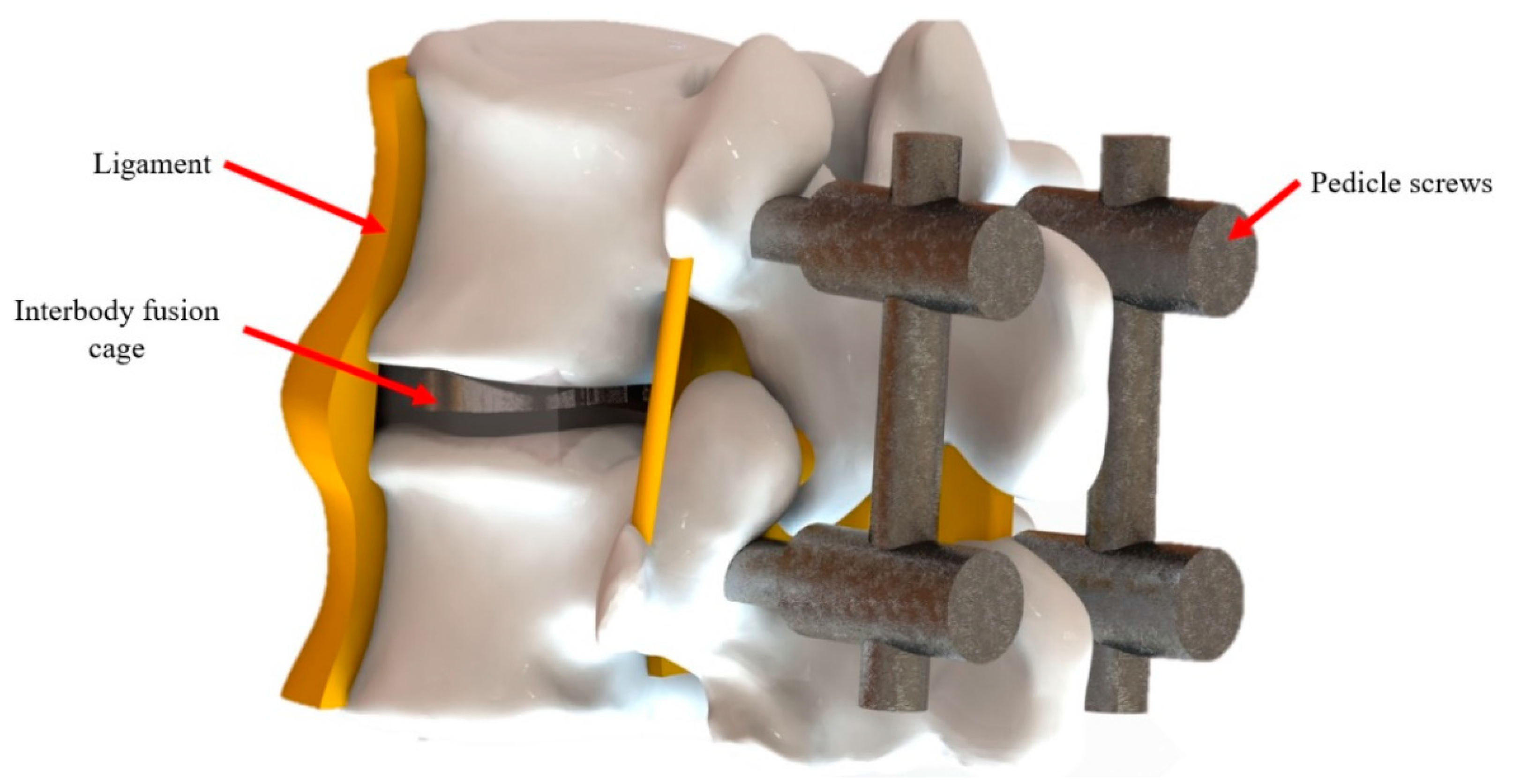
Figure 1.
The schematic details of spinal fusion surgery.
Figure 1.
The schematic details of spinal fusion surgery.
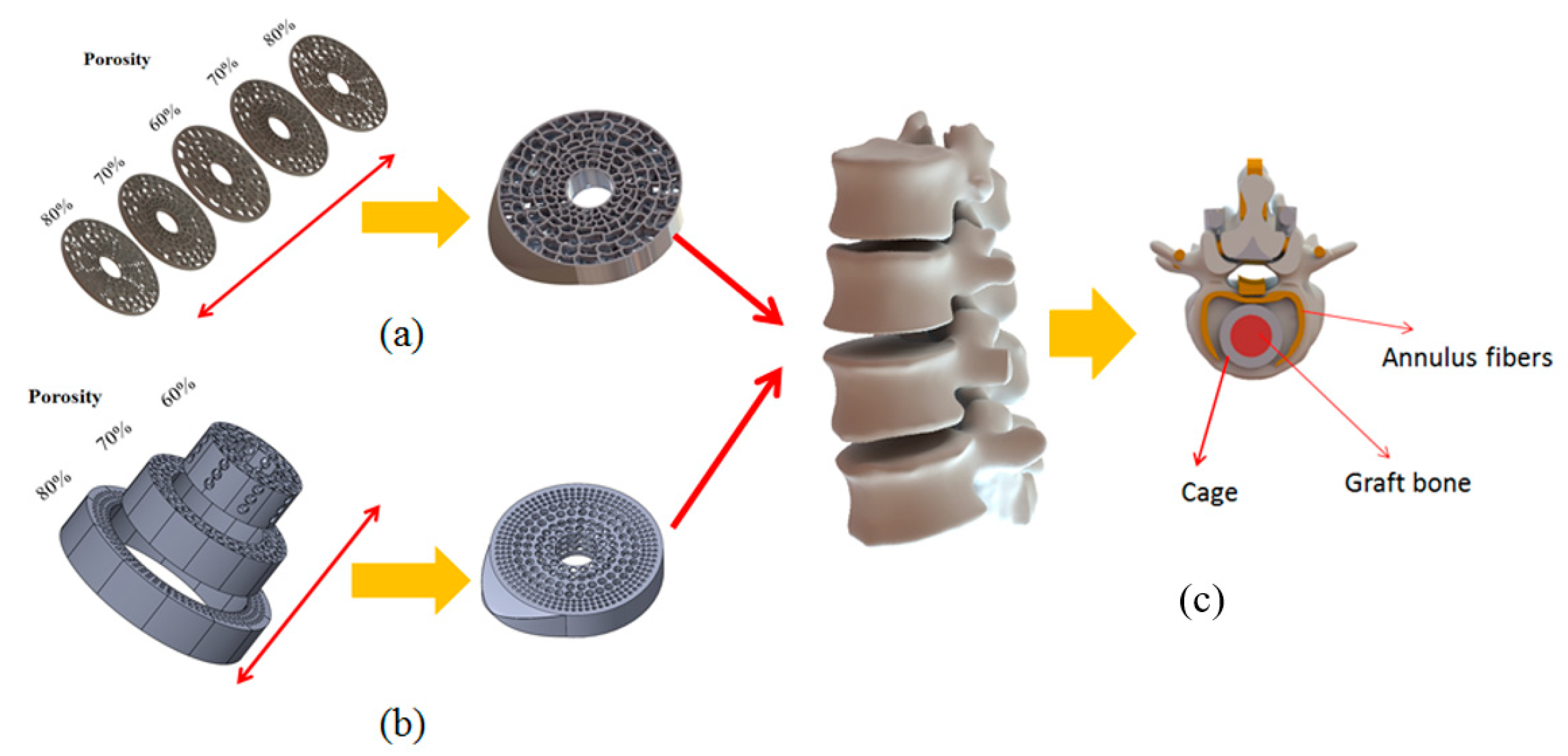
Figure 2.
Gradient porosity structure for cages. (a) Vertically gradient porosity. (b) Horizontally gradient porosity. (c) Cage placement in spinal fusion surgery.
Figure 2.
Gradient porosity structure for cages. (a) Vertically gradient porosity. (b) Horizontally gradient porosity. (c) Cage placement in spinal fusion surgery.
Figure 3.
The EOSINT M 280 selective laser melting (SLM) system.
Figure 3.
The EOSINT M 280 selective laser melting (SLM) system.
Figure 4.
The compression test of the universal testing machine.
Figure 4.
The compression test of the universal testing machine.
Figure 5.
Instron 5582 universal testing machine and SHIMADZU HMV-2 Vickers microhardness test machine.
Figure 5.
Instron 5582 universal testing machine and SHIMADZU HMV-2 Vickers microhardness test machine.
Figure 6.
Dynamic testing machine Bose 3510.
Figure 6.
Dynamic testing machine Bose 3510.
Figure 7.
Cage design was separated into three parts.
Figure 7.
Cage design was separated into three parts.
Figure 8.
Fusion cages fabricated with SLM. (a) Three parts of design, (b) Left, (c) middle, (d) right.
Figure 8.
Fusion cages fabricated with SLM. (a) Three parts of design, (b) Left, (c) middle, (d) right.
Figure 9.
The scanning electron microscope (SEM) photograph for specimens.
Figure 9.
The scanning electron microscope (SEM) photograph for specimens.
Figure 10.
The optical microscope (OM) photograph for specimens.
Figure 10.
The optical microscope (OM) photograph for specimens.
Figure 11.
Annealing parameters (a) 450 °C/120 min; (b) 490 °C/240 min; (c) 530 °C/80 min; (d) 570 °C/200 min; (e) 610 °C/40 min; (f) 650 °C/160 min.
Figure 11.
Annealing parameters (a) 450 °C/120 min; (b) 490 °C/240 min; (c) 530 °C/80 min; (d) 570 °C/200 min; (e) 610 °C/40 min; (f) 650 °C/160 min.
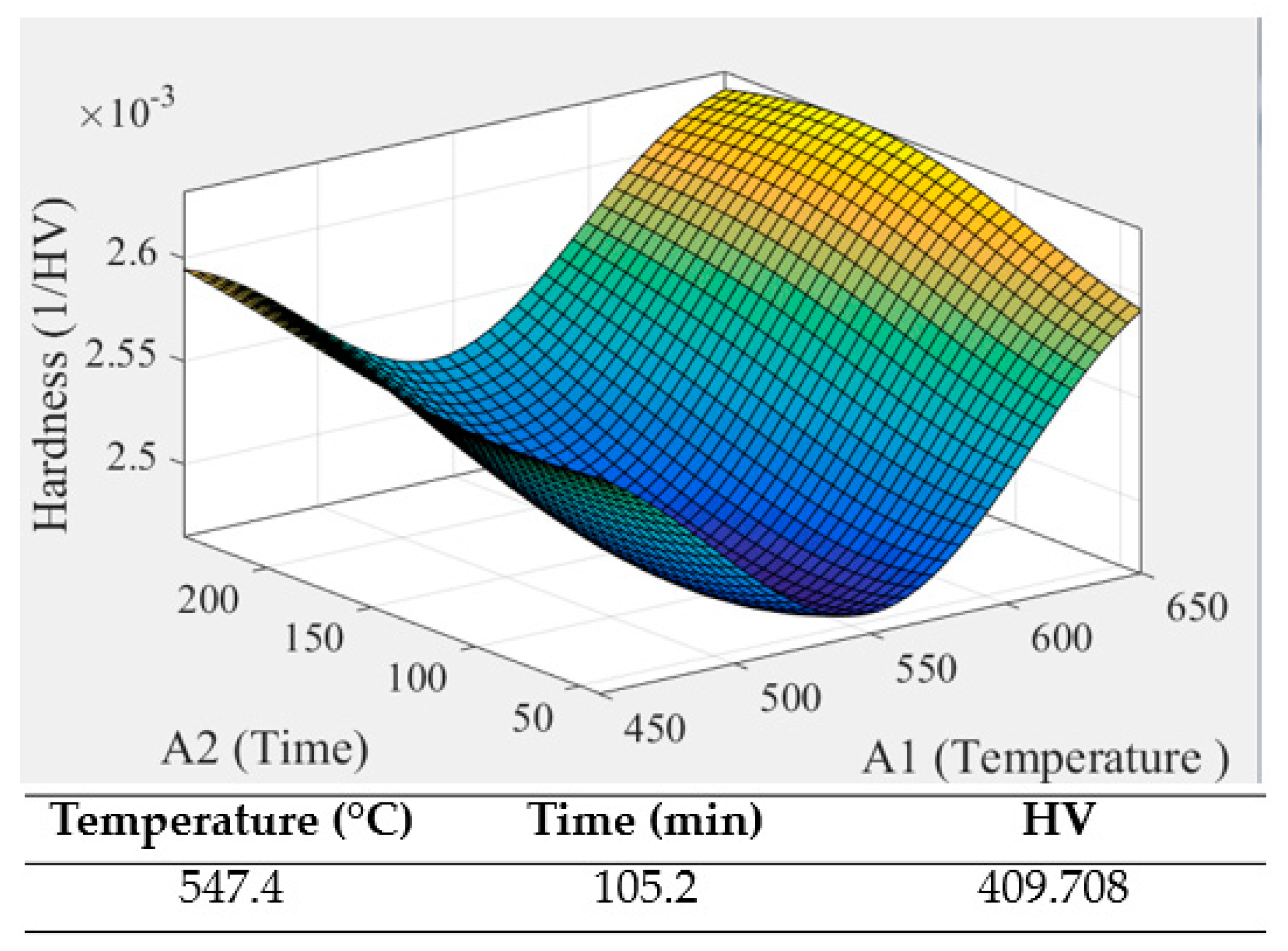
Figure 12.
The optimal parameter for annealing.
Figure 12.
The optimal parameter for annealing.
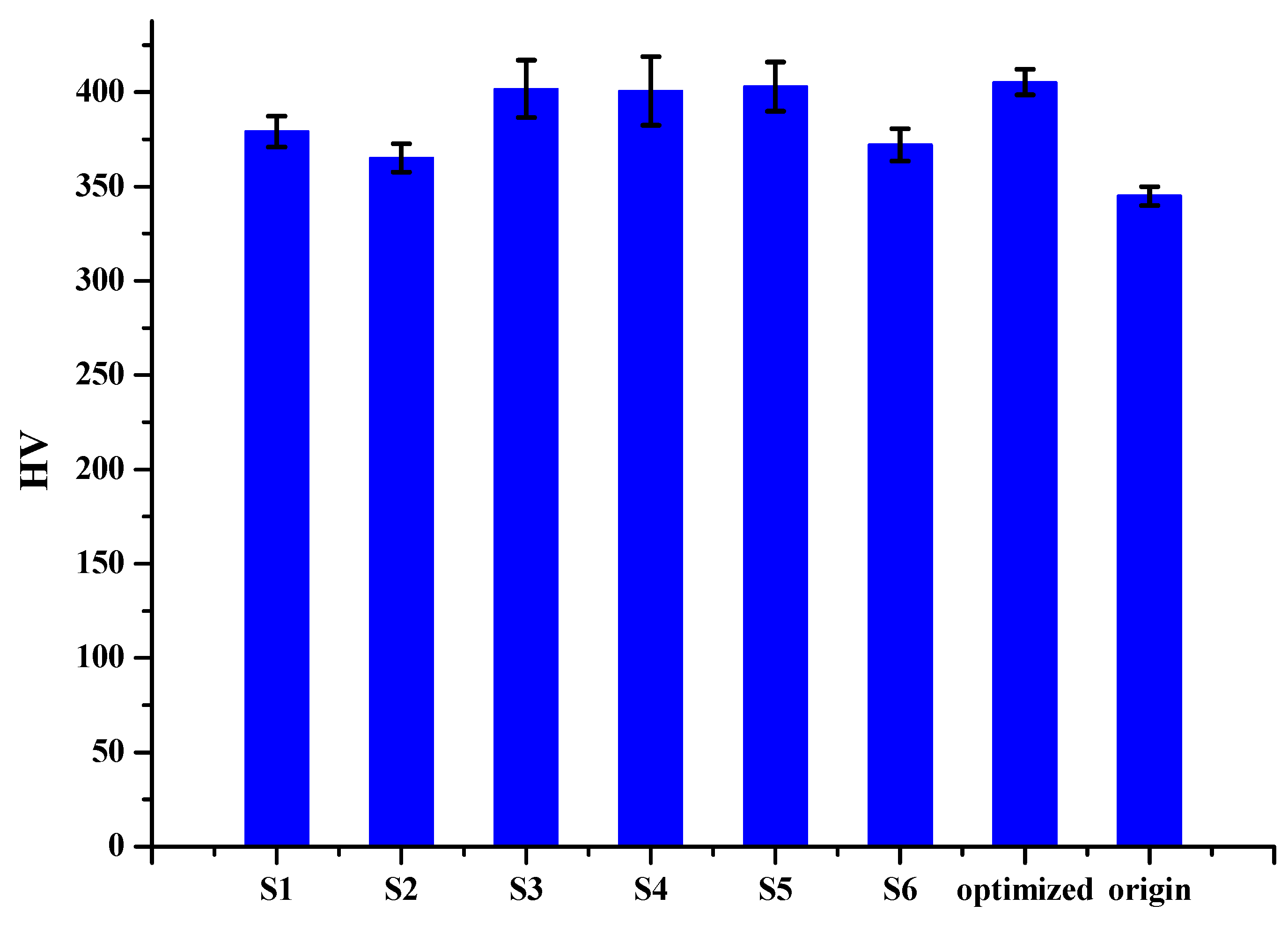
Figure 13.
Overall hardness for each sample.
Figure 13.
Overall hardness for each sample.
Figure 14.
The compressive stress-strain curves of the gradient porosity structure.
Figure 14.
The compressive stress-strain curves of the gradient porosity structure.
Figure 15.
Scraps after the dynamic test.
Figure 15.
Scraps after the dynamic test.
Figure 16.
Specimens observed under the OM. (a) Before test; (b) after test.
Figure 16.
Specimens observed under the OM. (a) Before test; (b) after test.
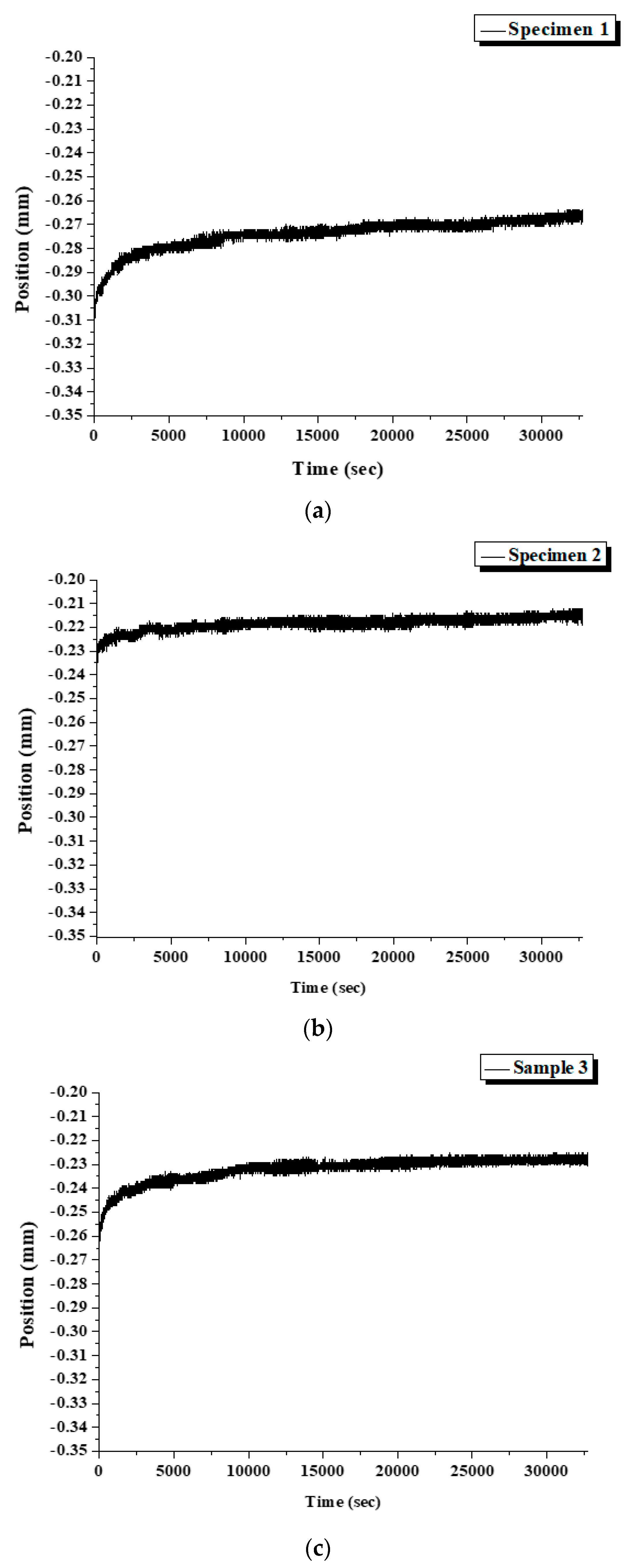
Figure 17.
The displacement of specimens during the test. (a) Specimen 1, (b) Specimen 2, (c) Specimen 3.
Figure 17.
The displacement of specimens during the test. (a) Specimen 1, (b) Specimen 2, (c) Specimen 3.
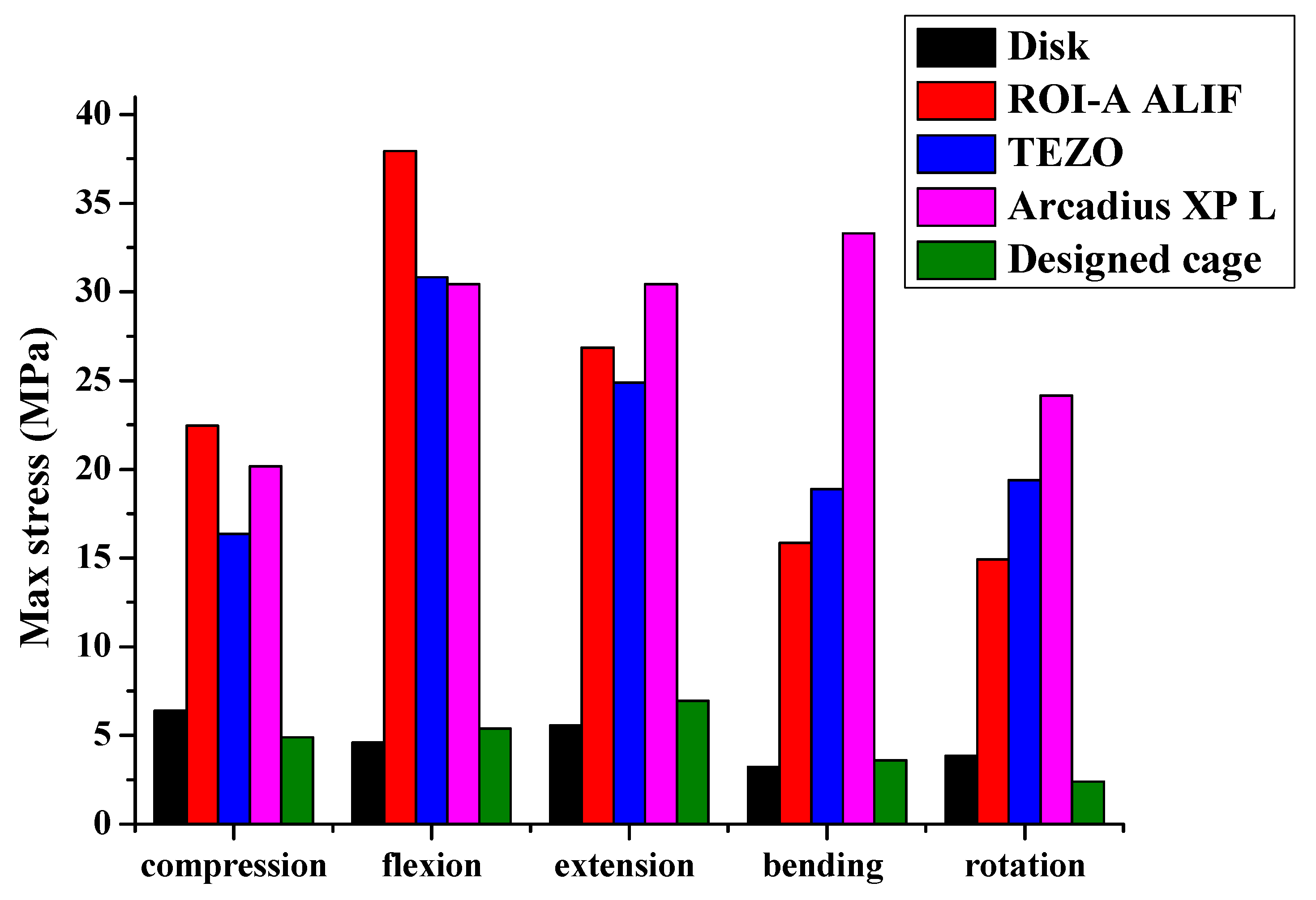
Figure 18.
Stress as a function of fusion cages for five different motion modes.
Figure 18.
Stress as a function of fusion cages for five different motion modes.
Table 1.
The material properties for simulation.
Table 1.
The material properties for simulation.
| Material | E (GPa) | ν | Density (g/cm3) | Yield Strength (GPa) |
|---|
| Cortical bone | 12 | 0.3 | 1.7 | - |
| Cancellous bone | 0.1 | 0.2 | 1.1 | - |
| Annulus fibers | 0.55 | 0.3 | 1 | - |
| Nucleus pulposus | 0.001 | 0.4999 | 1.02 | - |
| Anterior longitudinal | 0.015 | 0.3 | 1 | - |
| Posterior longitudinal | 0.02 | 0.3 | 1 | - |
| Ligamentum flavum | 0.019 | 0.3 | 1 | - |
| Intertransverse | 0.059 | 0.3 | 1 | - |
| Capsular | 0.033 | 0.3 | 1 | - |
| Interspinous | 0.012 | 0.3 | 1 | - |
| Supraspinous | 0.015 | 0.3 | 1 | - |
| Ti64ELI | 110 | 0.31 | 4.43 | 0.99 |
| Ti64ELI 60% porous | 24.4 | 0.3 | 1.76 | 0.23 |
| Ti64ELI 70% porous | 9.7 | 0.3 | 1.27 | 0.06 |
| Ti64ELI 80% porous | 2.42 | 0.3 | 0.66 | 0.02 |
Table 2.
Dimensions of the left part of the cage.
Table 2.
Dimensions of the left part of the cage.
| (a) Left (mm) | Avg. | σ | Origin |
|---|
| Width | 9.44 | 0.25 | 9.5 |
| Height | 5.99 | 0.25 | 6 |
| Depth | 15.93 | 0.33 | 16 |
Table 3.
Dimensions of the middle part of the cage.
Table 3.
Dimensions of the middle part of the cage.
| (b) Middle (mm) | Avg. | σ | Origin |
|---|
| Width | 11.70 | 0.20 | 11.8 |
| Height | 6.06 | 0.03 | 6 |
| Depth | 18.18 | 0.43 | 18.4 |
Table 4.
Dimensions of the right part of the cage.
Table 4.
Dimensions of the right part of the cage.
| (c) Right (mm) | Avg. | σ | Origin |
|---|
| Width | 11.20 | 0.43 | 11.3 |
| Height | 6.08 | 0.25 | 6 |
| Depth | 16.42 | 0.33 | 16.5 |
Table 5.
Beam dimensions measured by the scanning electron microscope (SEM) from specimens 1 to 6.
Table 5.
Beam dimensions measured by the scanning electron microscope (SEM) from specimens 1 to 6.
| Middle (mm) | Avg. | σ | Origin |
|---|
| A | 0.277 | 0.015 | 0.25 |
| B | 0.229 | 0.029 | 0.20 |
| C | 0.655 | 0.059 | 0.64 |
Table 6.
Hole dimensions measured by the SEM form specimens 1 to 6.
Table 6.
Hole dimensions measured by the SEM form specimens 1 to 6.
| Middle (mm) | Avg. | σ | Origin |
|---|
| A | 1.063 | 0.018 | 1.22 |
| B | 3.373 | 0.113 | 3.54 |
| C | 2.188 | 0.173 | 2.34 |
Table 7.
Dimensions measured by the optical microscope (OM) from specimens 1 to 6.
Table 7.
Dimensions measured by the optical microscope (OM) from specimens 1 to 6.
| Middle (mm) | Avg. | σ | Origin |
|---|
| I | 0.2317 | 0.0280 | 0.12 |
| II | 0.2328 | 0.0169 | 0.21 |
| III | 0.4926 | 0.0458 | 0.59 |
Table 8.
The uniform design experimentation (UDE) method for annealing parameters.
Table 8.
The uniform design experimentation (UDE) method for annealing parameters.
| Sample | Temperature (°C) | Time (min) |
|---|
| 1 | 450 | 120 |
| 2 | 490 | 240 |
| 3 | 530 | 80 |
| 4 | 570 | 200 |
| 5 | 610 | 40 |
| 6 | 650 | 160 |
Table 9.
Overall hardness for each specimen.
Table 9.
Overall hardness for each specimen.
| Sample | σ | HV |
|---|
| 1 | 2.183 | 379.111 |
| 2 | 2.055 | 365.111 |
| 3 | 3.794 | 401.778 |
| 4 | 4.546 | 400.667 |
| 5 | 3.247 | 402.889 |
| 6 | 2.331 | 372.111 |
| Origin | | 340~350 |
Table 10.
The actual value of the optimal parameter.
Table 10.
The actual value of the optimal parameter.
| | 1 | 2 | 3 | σ | HV |
|---|
| Top | 404 | 406 | 404 | 0.943 | 404.667 |
| Middle | 405 | 405 | 406 | 0.471 | 405.333 |
| Bottom | 401 | 407 | 410 | 3.742 | 406 |
Table 11.
Mechanical properties of different porosity for Ti64ELI [
43].
Table 11.
Mechanical properties of different porosity for Ti64ELI [
43].
Designed Porosity
(%) | Real Porosity
(%) | Young’s Modulus
(GPa) | Yield Stress
(MPa) |
|---|
| 40 | 43 ± 0.4 | 55.0 ± 2.4 | 564.7 ± 3.1 |
| 50 | 49 ± 0.9 | 44.4 ± 1.3 | 465.4 ± 2.1 |
| 60 | 60 ± 0.4 | 24.4 ± 1.0 | 233.9 ± 3.4 |
| 70 | 67 ± 0.4 | 15.3 ± 1.4 | 128.7 ± 5.6 |
| 80 | 71 ± 0.1 | 9.7 ± 1.9 | 62.0 ± 7.9 |
Table 12.
Height of each observation point for specimens 1 to 3.
Table 12.
Height of each observation point for specimens 1 to 3.
| No. | T (mm) | T′ (mm) | R (mm) | R′ (mm) | L (mm) | L′ (mm) |
|---|
| 1 | 4 | 4 | 4 | 4 | 3.5 | 3.5 |
| 2 | 4.5 | 4.5 | 4 | 4 | 3 | 3 |
| 3 | 4.5 | 4.5 | 4 | 4 | 3.5 | 3.5 |
Table 13.
The max stress and strain of the horizontally porous gradient structure.
Table 13.
The max stress and strain of the horizontally porous gradient structure.
| Horizontally Porous | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 10.727 | 18.25 | 9.847 | 10.904 | 13.01 |
| Max Strain (%) | 0.045 | 0.737 | 0.410 | 0.454 | 1.408 |
Table 14.
The max stress and strain of the vertically porous gradient structure.
Table 14.
The max stress and strain of the vertically porous gradient structure.
| Vertically Porous | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 6.324 | 8.982 | 7.581 | 7.614 | 9.193 |
| Max Strain (%) | 0.256 | 0.372 | 0.308 | 0.325 | 0.988 |
Table 15.
The max stress and strain of the ROI-A ALIF model.
Table 15.
The max stress and strain of the ROI-A ALIF model.
| ROI-A ALIF | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 22.462 | 37.951 | 26.875 | 15.841 | 14.943 |
| Max Strain (%) | 0.0028 | 0.0051 | 0.0038 | 0.0019 | 0.0015 |
Table 16.
The max stress and strain of the TEZO model.
Table 16.
The max stress and strain of the TEZO model.
| TEZO | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 16.349 | 30.813 | 24.876 | 18.899 | 19.383 |
| Max Strain (%) | 0.0016 | 0.0029 | 0.0023 | 0.0018 | 0.0019 |
Table 17.
The max stress and strain of the Arcadius XP L model.
Table 17.
The max stress and strain of the Arcadius XP L model.
| Arcadius XP L | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 20.183 | 30.441 | 30.429 | 33.284 | 24.15 |
| Max Strain (%) | 0.0022 | 0.0031 | 0.0032 | 0.0038 | 0.0023 |
Table 18.
The max stress and strain of the designed cage.
Table 18.
The max stress and strain of the designed cage.
| Designed Cage | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 4.888 | 5.383 | 6.942 | 3.601 | 2.4 |
| Max Strain (%) | 0.202 | 0.240 | 0.287 | 0.149 | 0.12 |
Table 19.
The max stress and strain of the disk.
Table 19.
The max stress and strain of the disk.
| Disk | Vertical Compression | Flexion | Extension | Bending | Rotation |
|---|
| Max Stress (MPa) | 6.398 | 4.61 | 5.568 | 3.246 | 3.864 |
| Max Strain (%) | 0.143 | 0.405 | 0.254 | 0.38 | 0.07 |