Monolayer Quantum-Dot Based Light-Sensor by a Photo-Electrochemical Mechanism
Abstract
1. Introduction
2. Experimental Section
2.1. Synthesis of Quantum Dots
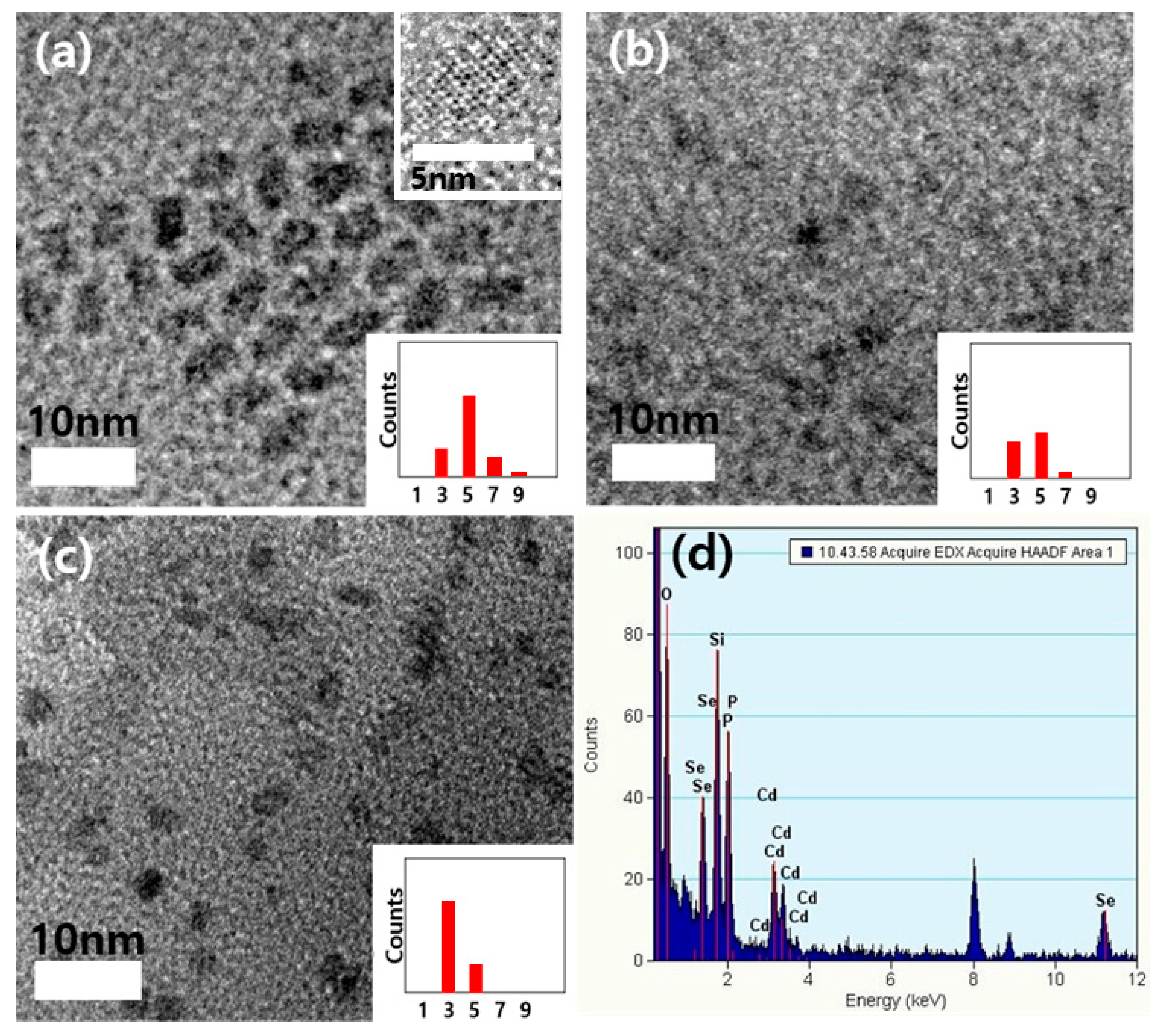
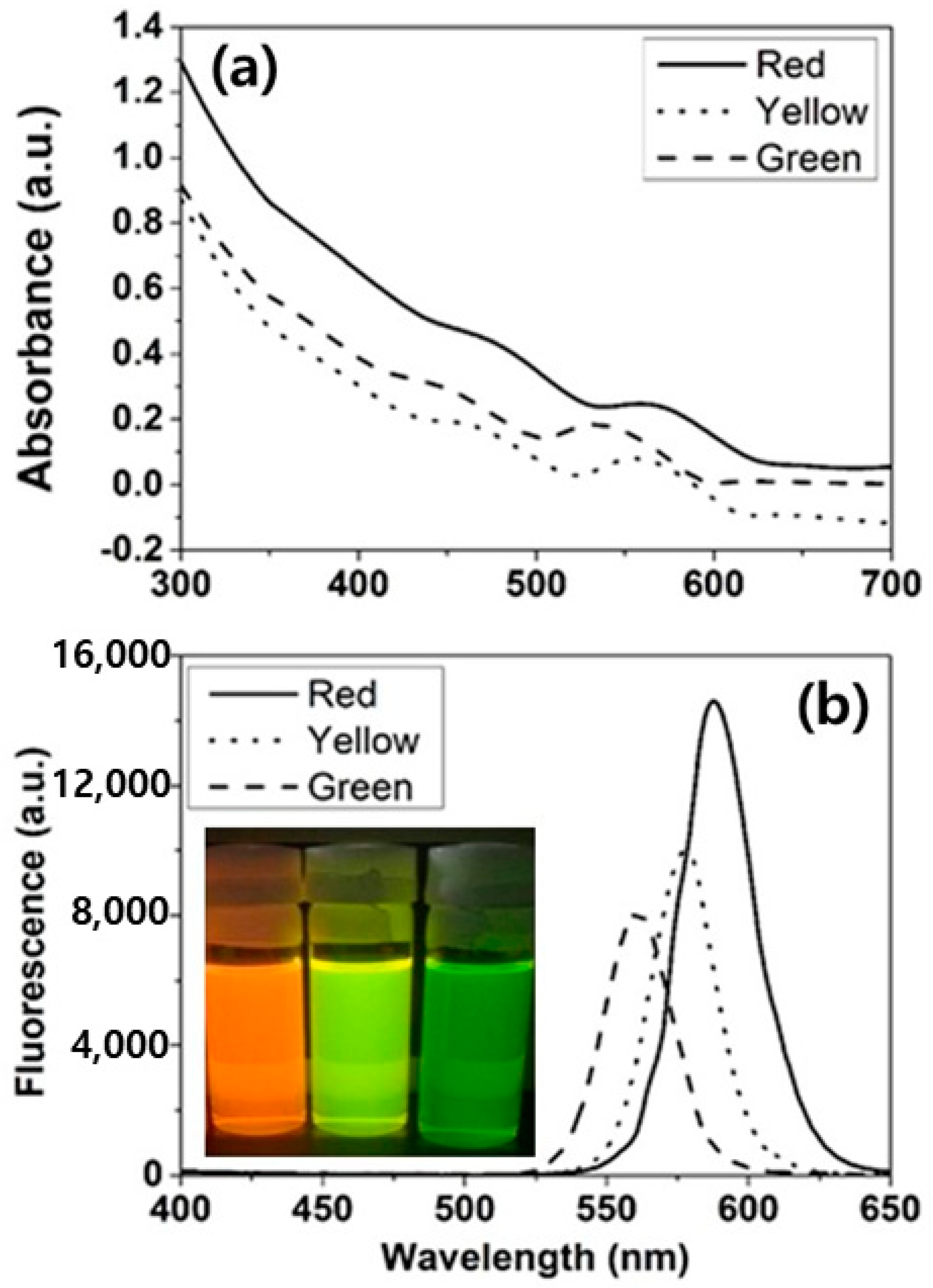
2.2. Characterization of Quantum Dots
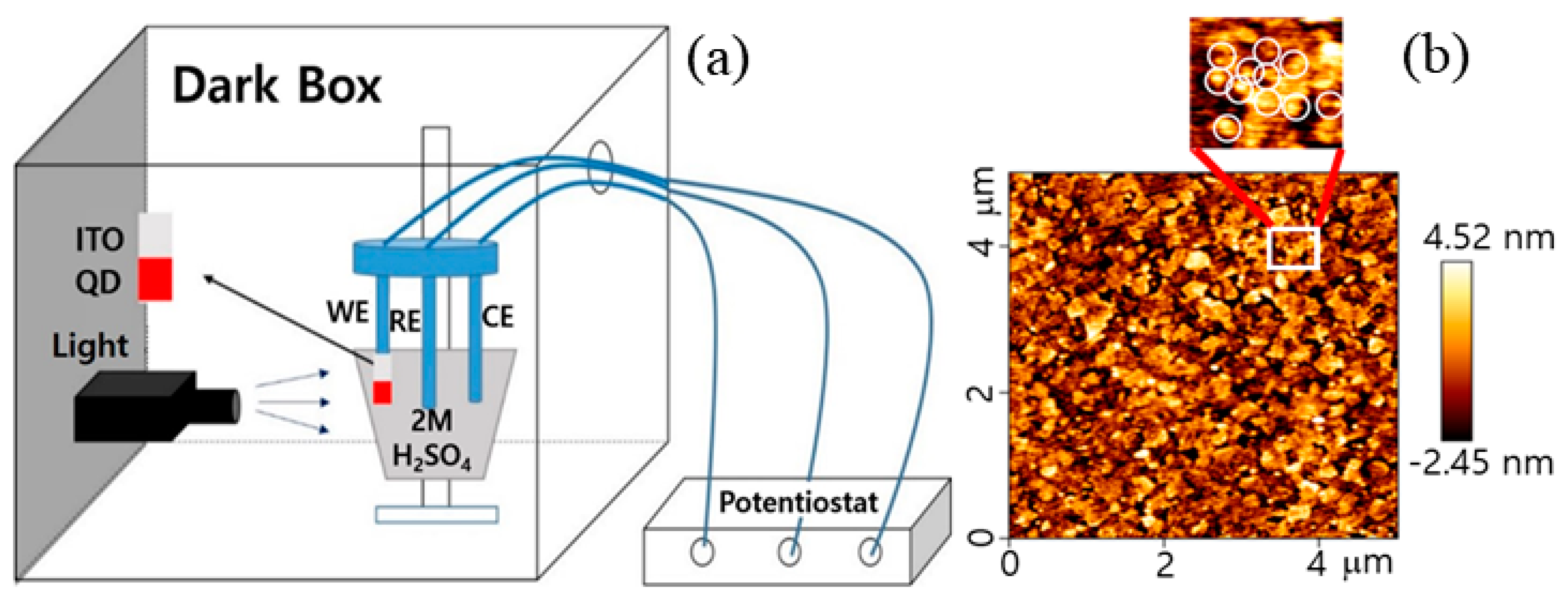
2.3. QD Electrochemical Sensor Fabrications and Measurements
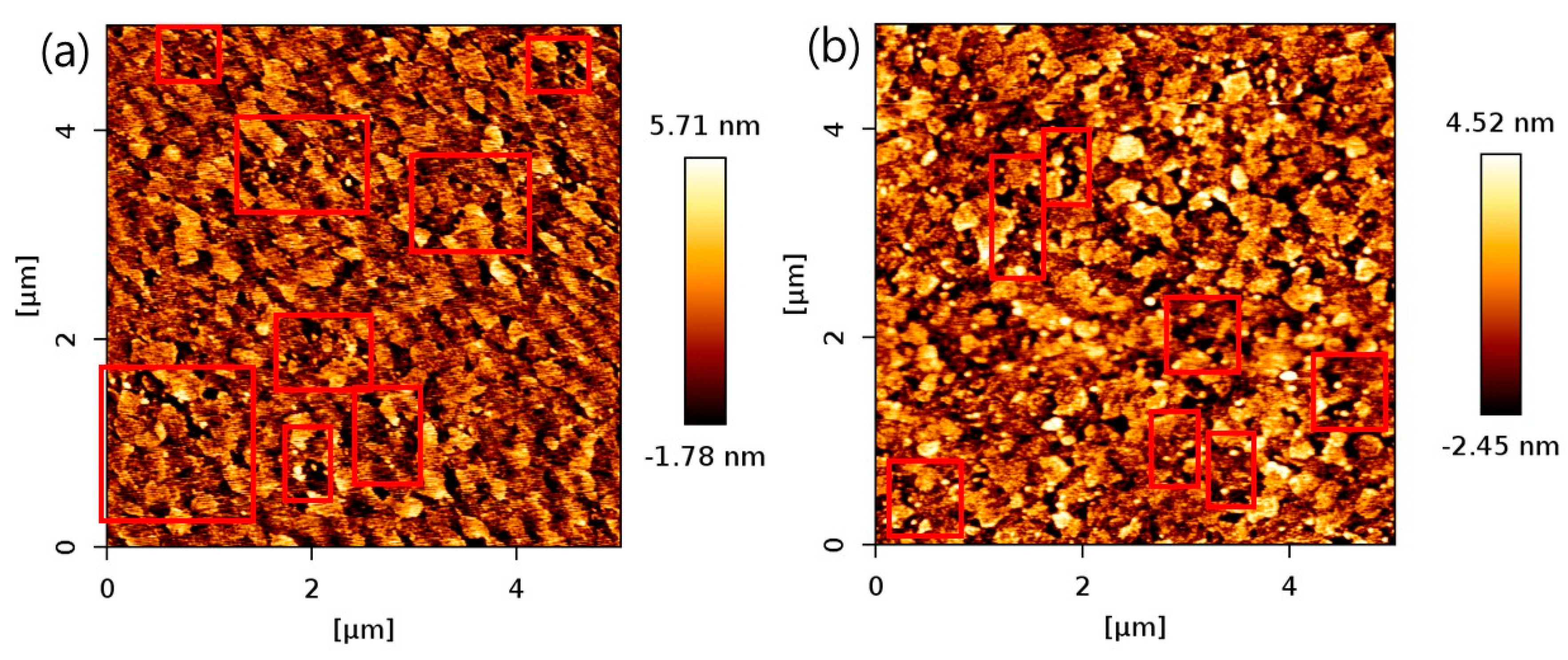
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ekimov, A.I.; Onushchenko, A.A. Quantum size effect in the optical-spectra of semiconductor micro-crystals. Sov. Phys. Semicond. -Ussr 1982, 16, 775–778. [Google Scholar]
- Brus, L.E. Electron–electron and electron-hole interactions in small semiconductor crystallites: The size dependence of the lowest excited electronic state. J. Chem. Phys. 1984, 80, 4403–4409. [Google Scholar] [CrossRef]
- Zhang, M.; Bishop, B.P.; Thompson, N.L.; Hildahl, K.; Dang, B.; Mironchuk, O.; Chen, N.; Aoki, R.; Holmberg, V.C.; Elizabeth Nance, E. Quantum dot cellular uptake and toxicity in the developing brain: Implications for use as imaging probes. Nanoscale Adv. 2019, 1, 3424–3442. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Ackerman, M.M.; Menglu Chen, M.; Guyot-Sionnest, P. Dual-band infrared imaging using stacked colloidal quantum dot photodiodes. Nat. Photonics 2019, 13, 277–282. [Google Scholar] [CrossRef]
- Moon, H.; Lee, C.; Lee, W.; Kim, J.; Chae, H. Stability of Quantum Dots, Quantum Dot Films, and Quantum Dot Light-Emitting Diodes for Display Applications. Adv. Mater. 2019, 31, 1804294. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Zhong, Q.; Chen, W.; Sang, B.; Wang, Y.; Yang, T.; Liu, Y.; Zhang, Y.; Zhang, H. Short-Chain Ligand-Passivated Stable α-CsPbI3 Quantum Dot for All-Inorganic Perovskite Solar Cells. Adv. Funct. Mater. 2019, 29, 1900991. [Google Scholar] [CrossRef]
- Yan, D.; Shi, T.; Zang, Z.; Zhou, T.; Liu, Z.; Zhang, Z.; Du, J.; Leng, Y.; Tang, X. Ultrastable CsPbBr3 Perovskite Quantum Dot and Their Enhanced Amplified Spontaneous Emission by Surface Ligand Modification. Small 2019, 15, 1901173. [Google Scholar] [CrossRef]
- Song, J.; Wang, O.; Shen, H.; Lin, Q.; Li, Z.; Wang, L.; Zhang, X.; Song, L. Over 30% External Quantum Efficiency Light-Emitting Diodes by Engineering Quantum Dot-Assisted Energy Level Match for Hole Transport. Layer Adv. Funct. Mater. 2019, 29, 1808377. [Google Scholar] [CrossRef]
- Ma, F.; Zhang, Q.; Zhang, C. Catalytic Self-Assembly of Quantum-Dot-Based MicroRNA Nanosensor Directed by Toehold-Mediated Strand Displacement Cascade. Nano Lett. 2019, 19, 6370–6376. [Google Scholar] [CrossRef]
- Shi, L.; Meng, L.; Jiang, F.; Ge, Y.; Li, F.; Wu, X.-G.; Zhong, H. In Situ Inkjet Printing Strategy for Fabricating Perovskite Quantum Dot Patterns. Adv. Funct. Mater. 2019, 29, 1903648. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, N.; Zeng, Z.; Lin, Q.; Zhang, F.; Tang, A.; Jia, Y.; Li, L.S.; Shen, H.; Teng, F.; et al. High-Efficiency Green InP Quantum Dot-Based Electroluminescent Device Comprising Thick-Shell Quantum Dots. Adv. Optical Mater. 2019, 7, 1801602. [Google Scholar] [CrossRef]
- Yuan, J.; Bi, C.; Wang, S.; Guo, R.; Shen, T.; Zhang, L.; Tian, J. Spray-Coated Colloidal Perovskite Quantum Dot Films for Highly Efficient Solar Cells. Adv. Funct. Mater. 2019, 29, 1906615. [Google Scholar] [CrossRef]
- Chebrolu, V.T.; Kim, H.-J. Recent progress in quantum dot sensitized solar cells: An inclusive review of photoanode, sensitizer, electrolyte, and the counter electrode. J. Mater. Chem. C 2019, 7, 4911–4933. [Google Scholar] [CrossRef]
- Li, X.; Lin, Q.; Song, J.; Shen, H.; Zhang, H.; Li, L.S.; Li, X.; Du, Z. Quantum-Dot Light-Emitting Diodes for Outdoor Displays with High Stability at High Brightness. Adv. Opt. Mater. 2020, 8, 1901145. [Google Scholar] [CrossRef]
- Sun, Y.; Su, Q.; Zhang, H.; Wang, F.; Zhang, S.; Chen, S. Investigation on Thermally Induced Efficiency Roll-Off: Toward Efficient and Ultrabright Quantum-Dot Light-Emitting Diodes. ACS Nano 2019, 13, 11433–11442. [Google Scholar] [CrossRef]
- Ouellette, O.; Lesage-Landry, A.; Scheffel, B.; Hoogland, S.; de Pelayo García Arquer, F.; Sargent, E.H. Spatial Collection in Colloidal Quantum Dot Solar Cells. Adv. Funct. Mater. 2020, 30, 1908200. [Google Scholar] [CrossRef]
- Norris, D.J.; Bawendi, M.G. Measurement and assignment of the size-dependent optical spectrum in CdSe quantum dots. Phys. Rev. B 1996, 53, 16338. [Google Scholar] [CrossRef] [PubMed]
- Alivisatos, A.P. Perspectives on the physical chemistry of semiconductor nanocrystals. J. Phys. Chem. 1996, 100, 13226–13239. [Google Scholar] [CrossRef]
- Yi, D.K.; Selvan, S.T.; Lee, S.S.; Papaefthymiou, G.C.; Kundaliya, D.; Ying, J.Y. Silica-coated nanocomposites of magnetic nanoparticles and quantum dots. J. Am. Chem. Soc. 2005, 127, 4990–4991. [Google Scholar] [CrossRef]
- Liu, H.; Li, M.; Voznyy, O.; Hu, L.; Fu, Q.; Zhou, D.; Xia, Z.; Sargent, E.H.; Tang, J. Physically flexible, rapid-response gas sensor based on colloidal quantum dot solids. Adv. Mater. 2014, 26, 2718–2724. [Google Scholar] [CrossRef]
- Zhou, J.; Yan, H.; Zheng, Y.; Wu, H.X. Highly Fluorescent Poly (dimethylsiloxane) for On-Chip Temperature Measurements. Adv. Funct. Mater. 2009, 19, 324–329. [Google Scholar] [CrossRef]
- Selvan, S.T.; Tan, T.T.Y.; Yi, D.K.; Jana, N.R. Functional and multifunctional nanoparticles for bioimaging and biosensing. Langmuir 2010, 26, 11631–11641. [Google Scholar] [CrossRef] [PubMed]
- Kazes, M.; Saraidarov, T.; Reisfeld, R.; Banin, U. Organic–inorganic sol–gel composites incorporating semiconductor nanocrystals for optical gain applications. Adv. Mater. 2009, 21, 1716–1720. [Google Scholar] [CrossRef]
- Song, Z.; Huang, Z.; Liu, J.; Hu, Z.; Zhang, J.; Zhang, G.; Yi, F.; Jiang, S.; Lian, J.; Yan, J.; et al. Fully Stretchable and Humidity-Resistant Quantum Dot Gas Sensors. ACS Sens. 2018, 3, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Raeyani, D.; Shojaei, S. Optical graphene quantum dots gas sensors: Experimental study. Mater. Res. Express 2020, 7, 015608. [Google Scholar] [CrossRef]
- Kumar, Y.R.; Deshmukh, K.; Sadasivunic, K.; Khadheer Pasha, S.K. Graphene quantum dot based materials for sensing, bio-imaging and energy storage applications: A review. RSC Adv. 2020, 10, 23861. [Google Scholar] [CrossRef]
- Bae, J.; Paik, U.; Yi, D.K. Novel semiconducting CdSe quantum dot based electrochemical capacitors. Mater. Lett. 2016, 162, 230–234. [Google Scholar] [CrossRef]
- Konstantatos, G.; Howard, I.; Fischer, A.; Hoogland, S.; Clifford, J.; Klem, E.; Levina, L.; Sargent, E.H. Ultrasensitive solution-cast quantum dot photodetectors. Nature 2006, 442, 180–183. [Google Scholar] [CrossRef]
- Fischer, M.; Georges, J. Fluorescence quantum yield of rhodamine 6G in ethanol as a function of concentration using thermal lens spectrometry. Chem. Phys. Lett. 1996, 260, 115–118. [Google Scholar] [CrossRef]
- Liu, Z.; Park, J.; Li, B.; Chan, H.P.; Yi, D.K.; Lee, E.-C. Performance improvement of organic bulk-heterojunction solar cells using complementary plasmonic gold nanorods. Org. Electron. 2020, 84, 105802. [Google Scholar] [CrossRef]
- Khanam, J.J.; Simon, Y.; Foo, S.Y.; Yu, Z.; Liu, T.; Mao, P. Efficient, Stable, and Low-Cost PbS Quantum Dot Solar Cells with Cr–Ag Electrodes. Nanomaterials 2019, 9, 1205. [Google Scholar] [CrossRef] [PubMed]

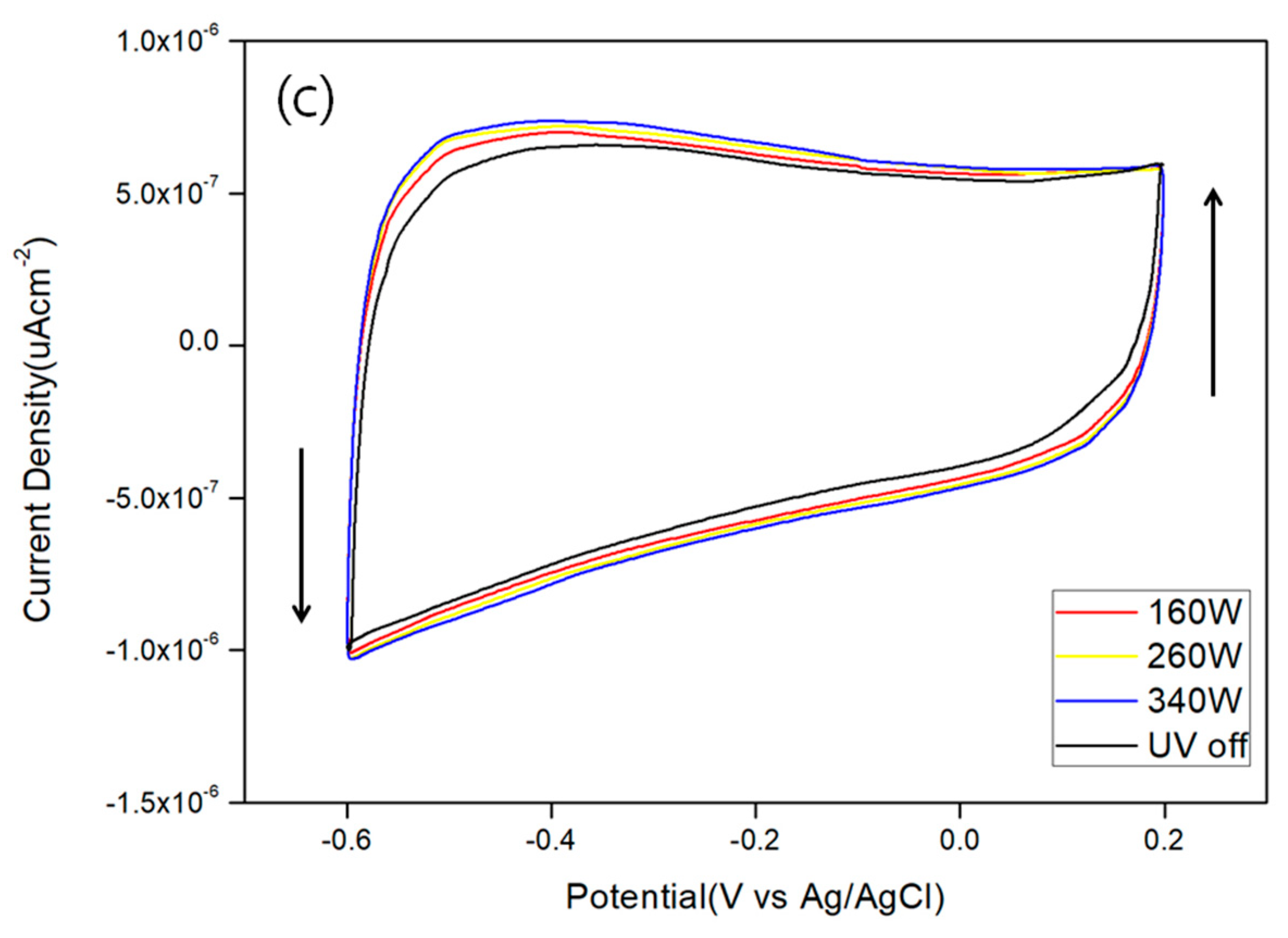
| μAcm−2 | 160 W | 260 W | 340 W | Light off |
|---|---|---|---|---|
| Red | 7.00 × 10−7 | 7.20 × 10−7 | 7.39 × 10−7 | 6.53 × 10−7 |
| Yellow | 7.83 × 10−8 | 9.14 × 10−8 | 1.58 × 10−7 | 5.01 × 10−8 |
| Green | 5.62 × 10−7 | 6.12 × 10−7 | 6.40 × 10−7 | 4.83 × 10−7 |
| μA-V/cm2 | Light Off | 160 W | 260 W | 340 W |
|---|---|---|---|---|
| Red | 7.60 | 8.65 | 9.10 | 9.48 |
| Yellow | 0.76 | 1.15 | 1.37 | 2.21 |
| Green | 8.57 | 9.26 | 9.57 | 9.78 |
| Ratio | 160 W/Light Off | 260 W/Light Off | 340 W/Light Off |
|---|---|---|---|
| Red | 1.13 | 1.20 | 1.25 |
| Yellow | 1.51 | 1.80 | 2.91 |
| Green | 1.08 | 1.12 | 1.14 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nanda, S.S.; Kim, M.; Yoo, S.J.; Papaefthymiou, G.C.; Yi, D.K. Monolayer Quantum-Dot Based Light-Sensor by a Photo-Electrochemical Mechanism. Micromachines 2020, 11, 817. https://doi.org/10.3390/mi11090817
Nanda SS, Kim M, Yoo SJ, Papaefthymiou GC, Yi DK. Monolayer Quantum-Dot Based Light-Sensor by a Photo-Electrochemical Mechanism. Micromachines. 2020; 11(9):817. https://doi.org/10.3390/mi11090817
Chicago/Turabian StyleNanda, Sitansu Sekhar, Minjik Kim, Sung Jong Yoo, Georgia C. Papaefthymiou, and Dong Kee Yi. 2020. "Monolayer Quantum-Dot Based Light-Sensor by a Photo-Electrochemical Mechanism" Micromachines 11, no. 9: 817. https://doi.org/10.3390/mi11090817
APA StyleNanda, S. S., Kim, M., Yoo, S. J., Papaefthymiou, G. C., & Yi, D. K. (2020). Monolayer Quantum-Dot Based Light-Sensor by a Photo-Electrochemical Mechanism. Micromachines, 11(9), 817. https://doi.org/10.3390/mi11090817







