Retinal Prosthetic Approaches to Enhance Visual Perception for Blind Patients
Abstract
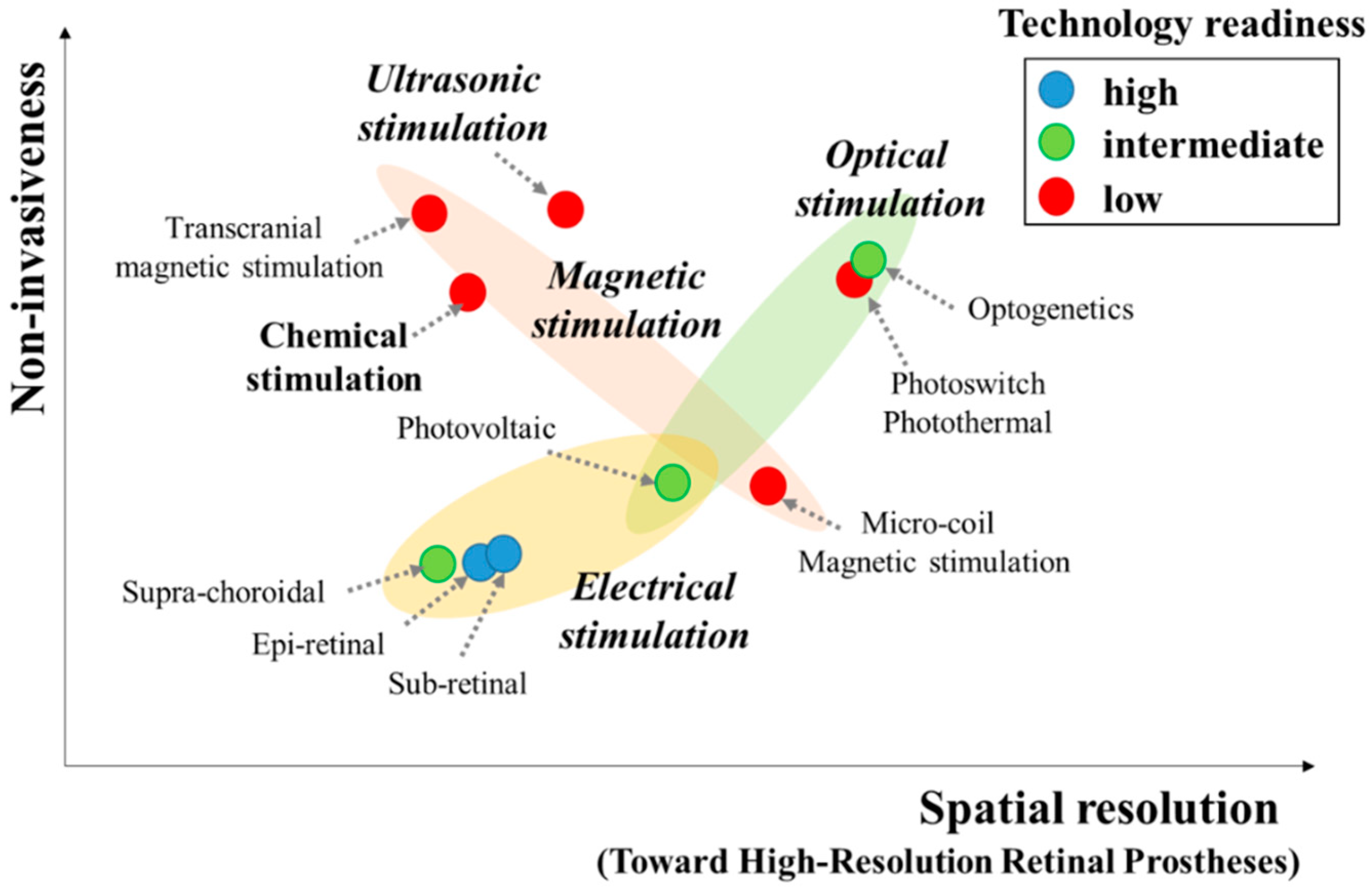
1. Introduction
2. Electrical Stimulation (ES)
2.1. Classification of ES on the Retina
2.1.1. Epi-Retinal Prostheses
2.1.2. Sub-Retinal Prostheses
2.1.3. Supra-Choroidal Prostheses
2.2. Factors Limiting Spatial Resolution of ES
2.3. Efforts to Improve Visual Acuity Using ES
2.3.1. Structural Modification of Electrodes
2.3.2. Optimization of Stimulation Patterns
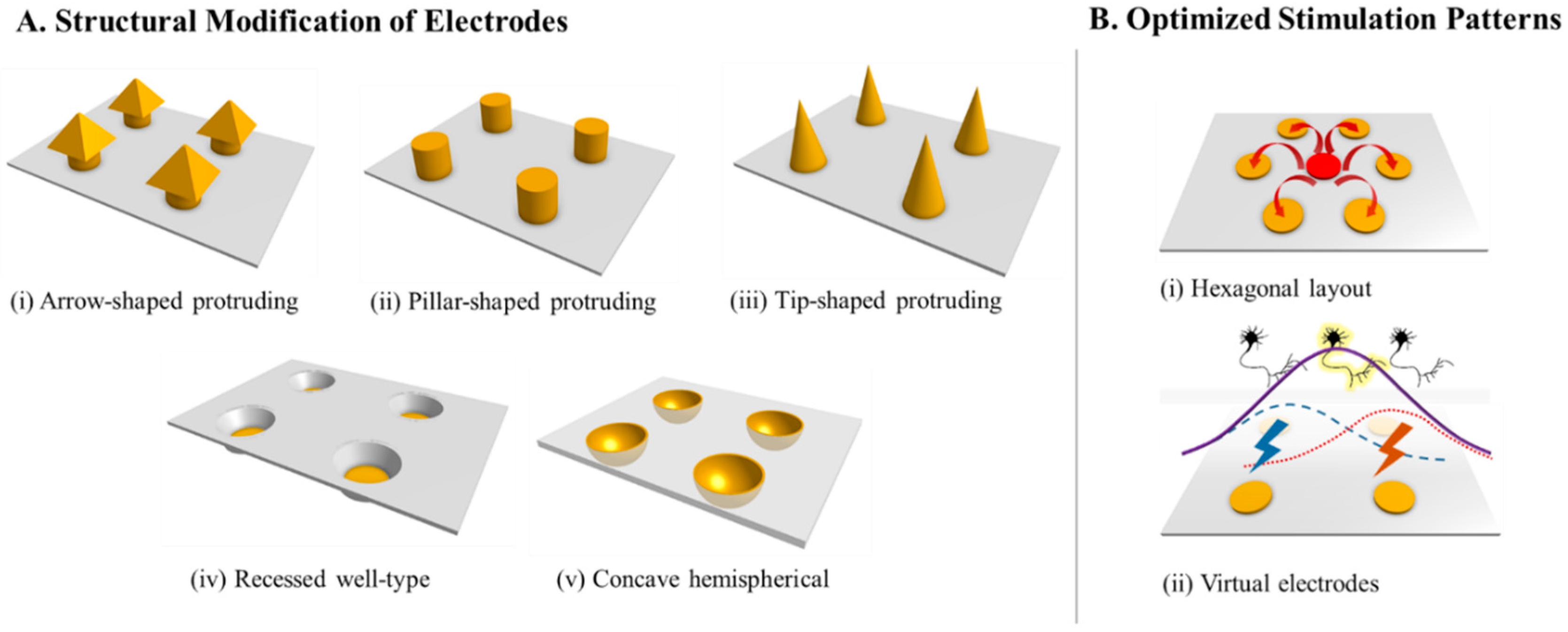
2.3.3. Optimization of Stimulation Parameters
3. Optical Stimulation (OS)
3.1. Classification of OS-Based Retinal Prostheses
3.1.1. Optogenetics-Based Retinal Prostheses
3.1.2. Photovoltaic Retinal Prostheses
3.1.3. Photoswitch-Based Retinal Prostheses
3.1.4. Photothermal Retinal Prostheses
3.2. Visual Acuity and Stimulation Methodology
| Methodology | Species | Stimulation Target | Visual Acuity | Ref. |
|---|---|---|---|---|
| Optogenetics (medium-wavelength cone-opsin) | Mouse | RGCs (in vivo) | 0.056 cpd | [146] |
| Optogenetics (CoChR) | Mouse | RGCs (in vivo) | 0.24 cpd | [147] |
| Photovoltaic - inorganic (IR light-sensitive photodiode) | Rat | Bipolar cells (in vivo) | 0.47 cpd, 20/460 | [158,159] |
| Photovoltaic - organic (ambient light-sensitive photodiode) | Rat | Bipolar cells (in vivo) | 0.62 cpd | [164] |
| Photoswitch (AAQ) | Mouse | RGCs (ex vivo) | <200 µm (receptive field of RGCs) | [166] |
4. Ultrasonic Stimulation (US)
5. Magnetic Stimulation
6. Other New Approaches
Author Contributions
Funding
Conflicts of Interest
References
- Wandell, B.A. Foundations of Vision; Sinauer Associates, Inc.: Sunderland, MA, USA, 1995; pp. 13–54. [Google Scholar]
- Oyster, C.W. The Human Eye: Structure and Function; Sinauer Associates, Inc.: Sunderland, MA, USA, 1999; pp. 545–700. [Google Scholar]
- Curcio, C.A.; Owsley, C.; Jackson, G.R. Spare the Rods, Save the Cones in Aging and Age-related Maculopathy. Investig. Ophthalmol. Vis. Sci. 2000, 41, 2015–2018. [Google Scholar]
- Busskamp, V.; Duebel, J.; Balya, D.; Fradot, M.; Viney, T.J.; Siegert, S.; Groner, A.C.; Cabuy, E.; Forster, V.; Seeliger, M.; et al. Genetic Reactivation of Cone Photoreceptors Restores Visual Responses in Retinitis Pigmentosa. Science 2010, 329, 413–417. [Google Scholar] [CrossRef]
- Yue, L.; Weiland, J.D.; Roska, B.; Humayun, M.S. Retinal stimulation strategies to restore vision: Fundamentals and systems. Prog. Retin. Eye Res. 2016, 53, 21–47. [Google Scholar] [CrossRef]
- Fahim, A. Retinitis pigmentosa: Recent advances and future directions in diagnosis and management. Curr. Opin. Pediatr. 2018, 30, 725–733. [Google Scholar] [CrossRef]
- Retinitis Pigmentosa. Available online: https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/retinitis-pigmentosa (accessed on 31 January 2020).
- Wells, J.; Wroblewski, J.; Keen, J.; Inglehearn, C.; Jubb, C.; Eckstein, A.; Jay, M.; Arden, G.; Bhattacharya, S.; Fitzke, F.; et al. Mutations in the human retinal degeneration slow (RDS) gene can cause either retinitis pigmentosa or macular dystrophy. Nat. Genet. 1993, 3, 213–218. [Google Scholar] [CrossRef]
- Hartong, D.T.; Berson, E.L.; Dryja, T.P. Retinitis pigmentosa. Lancet 2006, 368, 1795–1809. [Google Scholar] [CrossRef]
- Quillen, D.A. Common Causes of Vision Loss in Elderly Patients. Am. Fam. Physician 1999, 60, 99–108. [Google Scholar] [PubMed]
- Jackson, G.R.; Owsley, C.; Curcio, C.A. Photoreceptor degeneration and dysfunction in aging and age-related maculopathy. Ageing Res. Rev. 2002, 1, 381–396. [Google Scholar] [CrossRef]
- Age-Related Macular Degeneration. Available online: https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/age-related-macular-degeneration (accessed on 2 February 2020).
- Bressler, N.M. Age-Related Macular Degeneration Is the Leading Cause of Blindness. Jama 2004, 291, 1900–1901. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.S.; O’Colmain, B.J.; Muñoz, B.; Tomany, S.C.; McCarty, C.; de Jong, P.T.V.M.; Nemesure, B.; Mitchell, P.; Kempen, J.; Congdon, N. Prevalence of Age-Related Macular Degeneration in the United States. Arch. Ophthalmol. 2004, 122, 564–572. [Google Scholar] [PubMed]
- Age-Related Macular Degeneration (AMD) Data and Statistics. Available online: https://www.nei.nih.gov/learn-about-eye-health/resources-for-health-educators/eye-health-data-and-statistics/age-related-macular-degeneration-amd-data-and-statistics (accessed on 2 February 2020).
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Heier, J.S.; Brown, D.M.; Chong, V.; Korobelnik, J.F.; Kaiser, P.K.; Nguyen, Q.D.; Kirchhof, B.; Ho, A.; Ogura, Y.; Yancopoulos, G.D.; et al. Intravitreal Aflibercept (VEGF Trap-Eye) in Wet Age-related Macular Degeneration. Ophthalmology 2012, 119, 2537–2548. [Google Scholar] [CrossRef] [PubMed]
- Stieger, K.; Chauveau, C.; Rolling, F. Preclinical Studies on Specific Gene Therapy for Recessive Retinal Degenerative Diseases. Curr. Gene Ther. 2010, 10, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Enzmann, V.; Ildstad, S.T. Stem Cell-Based Therapeutic Applications in Retinal Degenerative Diseases. Stem Cell Rev. Rep. 2011, 7, 434–445. [Google Scholar] [CrossRef]
- Wong, I.Y.H.; Poon, M.W.; Pang, R.T.W.; Lian, Q.; Wong, D. Promises of stem cell therapy for retinal degenerative diseases. Graef. Arch. Clin. Exp. Ophthalmol. 2011, 249, 1439. [Google Scholar] [CrossRef]
- Beltran, W.A.; Cideciyan, A.V.; Lewin, A.S.; Iwabe, S.; Khanna, H.; Sumaroka, A.; Chiodo, V.A.; Fajardo, D.S.; Román, A.J.; Deng, W.T.; et al. Gene therapy rescues photoreceptor blindness in dogs and paves the way for treating human X-linked retinitis pigmentosa. Proc. Natl. Acad. Sci. USA 2012, 109, 2132–2137. [Google Scholar] [CrossRef]
- Wiley, L.A.; Burnight, E.R.; Songstad, A.E.; Drack, A.V.; Mullins, R.F.; Stone, E.M.; Tucker, B.A. Patient-specific induced pluripotent stem cells (iPSCs) for the study and treatment of retinal degenerative diseases. Prog. Retin. Eye Res. 2015, 44, 15–35. [Google Scholar] [CrossRef]
- Rakoczy, E.P.; Lai, C.M.; Magno, A.L.; Wikstrom, M.E.; French, M.A.; Pierce, C.M.; Schwartz, S.D.; Blumenkranz, M.S.; Chalberg, T.W.; Degli-Esposti, M.A.; et al. Gene therapy with recombinant adeno-associated vectors for neovascular age-related macular degeneration: 1 year follow-up of a phase 1 randomised clinical trial. Lancet 2015, 386, 2395–2403. [Google Scholar] [CrossRef]
- Takahashi, V.K.L.; Takiuti, J.T.; Jauregui, R.; Tsang, S.H. Gene therapy in inherited retinal degenerative diseases, a review. Ophthalmic Genet. 2018, 39, 560–568. [Google Scholar] [CrossRef]
- Bloch, E.; Luo, Y.; da Cruz, L. Advances in retinal prosthesis systems. Ther. Adv. Ophthalmol. 2019, 11, 2515841418817501. [Google Scholar] [CrossRef]
- Petersen-Jones, S.M.; Occelli, L.M.; Winkler, P.A.; Lee, W.; Sparrow, J.R.; Tsukikawa, M.; Boye, S.L.; Chiodo, V.; Capasso, J.E.; Becirovic, E.; et al. Patients and animal models of CNGβ1-deficient retinitis pigmentosa support gene augmentation approach. J. Clin. Investig. 2018, 128, 190–206. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.S.; Issa, P.C.; Butler, R.; Martin, C.; Lipinski, D.M.; Sekaran, S.; Barnard, A.R.; MacLaren, R.E. Reversal of end-stage retinal degeneration and restoration of visual function by photoreceptor transplantation. Proc. Natl. Acad. Sci. USA 2013, 110, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.D.; Regillo, C.D.; Lam, B.L.; Eliott, D.; Rosenfeld, P.J.; Gregori, N.Z.; Hubschman, J.P.; Davis, J.L.; Heilwell, G.; Spirn, M.; et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt’s macular dystrophy: Follow-up of two open-label phase 1/2 studies. Lancet 2015, 385, 509–516. [Google Scholar] [CrossRef]
- Weiland, J.D.; Liu, W.; Humayun, M.S. Retinal Prosthesis. Annu. Rev. Biomed. Eng. 2005, 7, 361–401. [Google Scholar] [CrossRef]
- Weiland, J.D.; Humayun, M.S. Retinal Prosthesis. IEEE Trans. Biomed. Eng. 2014, 61, 1412–1424. [Google Scholar] [CrossRef]
- Santos, A.; Humayun, M.S.; de Juan, E., Jr.; Greenburg, R.J.; Marsh, M.J.; Klock, I.B.; Milam, A.H. Preservation of the Inner Retina in Retinitis Pigmentosa: A Morphometric Analysis. Arch. Ophthalmol. 1997, 115, 511–515. [Google Scholar] [CrossRef]
- Humayun, M.S.; Prince, M.; de Juan , E., Jr.; Barron, Y.; Moskowitz, M.; Klock, I.B.; Milam, A.H. Morphometric analysis of the extramacular retina from postmortem eyes with retinitis pigmentosa. Investig. Ophthalmol. Vis. Sci. 1999, 40, 143–148. [Google Scholar]
- Medeiros, N.E.; Curcio, C.A. Preservation of Ganglion Cell Layer Neurons in Age-Related Macular Degeneration. Investig. Ophthalmol. Vis. Sci. 2001, 42, 795–803. [Google Scholar]
- Kim, S.Y.; Sadda, S.; Pearlman, J.; Humayun, M.S.; de Juan, E., Jr.; Melia, B.M.; Green, W.R. Morphometric analysis of the macula in eyes with disciform age-related macular degeneration. Retina 2002, 22, 471–477. [Google Scholar] [CrossRef]
- Klauke, S.; Goertz, M.; Rein, S.; Hoehl, D.; Thomas, U.; Eckhorn, R.; Bremmer, F.; Wachtler, T. Stimulation with a Wireless Intraocular Epiretinal Implant Elicits Visual Percepts in Blind Humans. Investig. Ophthalmol. Vis. Sci. 2011, 52, 449–455. [Google Scholar] [CrossRef]
- Fujikado, T.; Kamei, M.; Sakaguchi, H.; Kanda, H.; Morimoto, T.; Ikuno, Y.; Nishida, K.; Kishima, H.; Maruo, T.; Konoma, K.; et al. Testing of Semichronically Implanted Retinal Prosthesis by Suprachoroidal-Transretinal Stimulation in Patients with Retinitis Pigmentosa. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4726–4733. [Google Scholar] [CrossRef] [PubMed]
- Stingl, K.; Bartz-Schmidt, K.U.; Besch, D.; Braun, A.; Bruckmann, A.; Gekeler, F.; Greppmaier, U.; Hipp, S.; Hörtdörfer, G.; Kernstock, C.; et al. Artificial vision with wirelessly powered subretinal electronic implant alpha-IMS. Proc. R. Soc. B 2013, 280, 20130077. [Google Scholar] [CrossRef] [PubMed]
- Weiland, J.D.; Cho, A.K.; Humayun, M.S. Retinal Prostheses: Current Clinical Results and Future Needs. Ophthalmology 2011, 118, 2227–2237. [Google Scholar] [CrossRef] [PubMed]
- Lam, B.L.; Gregori, N.Z. Successes and Challenges of Retinal Implants for Profound Visual Loss from Outer Retinal Degeneration. JAMA Ophthalmol. 2019, 137, 903–904. [Google Scholar] [CrossRef] [PubMed]
- DiFrancesco, D. Pacemaker mechanisms in cardiac tissue. Annu. Rev. Physiol. 1993, 55, 455–472. [Google Scholar] [CrossRef]
- Benabid, A.L. Deep brain stimulation for Parkinson’s disease. Curr. Opin. Neurobiol. 2003, 13, 696–706. [Google Scholar] [CrossRef]
- Perlmutter, J.S.; Mink, J.W. Deep Brain Stimulation. Annu. Rev. Neurosci. 2006, 29, 229–257. [Google Scholar] [CrossRef]
- Zeng, F.G.; Rebscher, S.; Harrison, W.; Sun, X.; Feng, H. Cochlear Implants: System Design, Integration, and Evaluation. IEEE Rev. Biomed. Eng. 2008, 1, 115–142. [Google Scholar] [CrossRef]
- Baruscotti, M.; Barbuti, A.; Bucchi, A. The cardiac pacemaker current. J. Mol. Cell. Cardiol. 2010, 48, 55–64. [Google Scholar] [CrossRef]
- Gaylor, J.M.; Raman, G.; Chung, M.; Lee, J.; Rao, M.; Lau, J.; Poe, D.S. Cochlear Implantation in Adults: A Systematic Review and Meta-analysis. JAMA Otolaryngol. Head Neck Surg. 2013, 139, 265–272. [Google Scholar] [CrossRef]
- Luo, Y.H.L.; da Cruz, L. A review and update on the current status of retinal prostheses (bionic eye). Br. Med. Bull. 2014, 109, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Brindley, G.S.; Lewin, W.S. The sensations produced by electrical stimulation of the visual cortex. J. Physiol. 1968, 196, 479–493. [Google Scholar] [CrossRef] [PubMed]
- Potts, A.M.; Inoue, J. The electrically evoked response (EER) of the visual system: II. Effect of adaptation and retinitis pigmentosa. Investig. Ophthalmol. Vis. Sci. 1969, 8, 605–612. [Google Scholar]
- Dobelle, W.H.; Mladejovsky, M.G.; Girvin, J.P. Artificial Vision for the Blind: Electrical Stimulation of Visual Cortex Offers Hope for a Functional Prosthesis. Science 1974, 183, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, J.F., III; Wyatt, J. REVIEW: Prospects for a Visual Prosthesis. Neuroscientist 1997, 3, 251–262. [Google Scholar] [CrossRef]
- Veraart, C.; Raftopoulos, C.; Mortimer, J.T.; Delbeke, J.; Pins, D.; Michaux, G.; Vanlierde, A.; Parrini, S.; Wanet-Defalque, M.C. Visual sensations produced by optic nerve stimulation using an implanted self-sizing spiral cuff electrode. Brain Res. 1998, 813, 181–186. [Google Scholar] [CrossRef]
- Troyk, P.; Bak, M.; Berg, J.; Bradley, D.; Cogan, S.; Erickson, R.; Kufta, C.; McCreery, D.; Schmidt, E.; Towle, V. A Model for Intracortical Visual Prosthesis Research. Artif. Organs 2003, 27, 1005–1015. [Google Scholar] [CrossRef]
- Loudin, J.D.; Simanovskii, D.M.; Vijayraghavan, K.; Sramek, C.K.; Butterwick, A.F.; Huie, P.; McLean, G.Y.; Palanker, D.V. Optoelectronic retinal prosthesis: System design and performance. J. Neural Eng. 2007, 4, S72. [Google Scholar] [CrossRef]
- Pezaris, J.S.; Eskandar, E.N. Getting signals into the brain: Visual prosthetics through thalamic microstimulation. Neurosurg. Focus 2009, 27, E6. [Google Scholar] [CrossRef]
- Lewis, P.M.; Ackland, H.M.; Lowery, A.J.; Rosenfeld, J.V. Restoration of vision in blind individuals using bionic devices: A review with a focus on cortical visual prostheses. Brain Res. 2015, 1595, 51–73. [Google Scholar] [CrossRef]
- Gaillet, V.; Cutrone, A.; Artoni, F.; Vagni, P.; Pratiwi, A.M.; Romero, S.A.; di Paola, D.L.; Micera, S.; Ghezzi, D. Spatially selective activation of the visual cortex via intraneural stimulation of the optic nerve. Nat. Biomed. Eng. 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Humayun, M.S.; de Juan, E., Jr.; Weiland, J.D.; Dagnelie, G.; Katona, S.; Greenberg, R.; Suzuki, S. Pattern electrical stimulation of the human retina. Vis. Res. 1999, 39, 2569–2576. [Google Scholar] [CrossRef]
- Wandell, B.A.; Wade, A.R. Functional imaging of the visual pathways. Neurol. Clin. 2003, 21, 417–443. [Google Scholar] [CrossRef]
- Rizzo, J.F., III; Wyatt, J.; Loewenstein, J.; Kelly, S.; Shire, D. Perceptual Efficacy of Electrical Stimulation of Human Retina with a Microelectrode Array during Short-Term Surgical Trials. Investig. Ophthalmol. Vis. Sci. 2003, 44, 5362–5369. [Google Scholar] [CrossRef]
- Zeff, B.W.; White, B.R.; Dehghani, H.; Schlaggar, B.L.; Culver, J.P. Retinotopic mapping of adult human visual cortex with high-density diffuse optical tomography. Proc. Natl. Acad. Sci. USA 2007, 104, 12169–12174. [Google Scholar] [CrossRef]
- Kitiratschky, V.B.D.; Stingl, K.; Wilhelm, B.; Peters, T.; Besch, D.; Sachs, H.; Gekeler, F.; Bartz-Schmidt, K.U.; Zrenner, E. Safety evaluation of “retina implant alpha IMS”-a prospective clinical trial. Graef. Arch. Clin. Exp. Ophthalmol. 2015, 253, 381–387. [Google Scholar] [CrossRef]
- Luo, Y.H.L.; da Cruz, L. The Argus® II Retinal Prosthesis System. Prog. Retin. Eye Res. 2016, 50, 89–107. [Google Scholar] [CrossRef]
- Rizzo, J.F., III. Update on retinal prosthetic research: The Boston Retinal Implant Project. J. Neuro-Ophth. 2011, 31, 160–168. [Google Scholar] [CrossRef]
- Shepherd, R.K.; Shivdasani, M.N.; Nayagam, D.A.; Williams, C.E.; Blamey, P.J. Visual prostheses for the blind. Trends Biotechnol. 2013, 31, 562–571. [Google Scholar] [CrossRef]
- Chuang, A.T.; Margo, C.E.; Greenberg, P.B. Retinal implants: A systematic review. Br. J. Ophthalmol. 2014, 98, 852–856. [Google Scholar] [CrossRef]
- Stingl, K.; Bartz-Schmidt, K.U.; Besch, D.; Chee, C.K.; Cottriall, C.L.; Gekeler, F.; Groppe, M.; Jackson, T.L.; MacLaren, R.E.; Koitschev, A.; et al. Subretinal visual implant alpha IMS-clinical trial interim report. Vis. Res. 2015, 111, 149–160. [Google Scholar] [CrossRef]
- Humayun, M.S.; Dorn, J.D.; da Cruz, L.; Dagnelie, G.; Sahel, J.A.; Stanga, P.E.; Cideciyan, A.V.; Duncan, J.L.; Eliott, D.; Filley, E.; et al. Interim Results from the International Trial of Second Sight’s Visual Prosthesis. Ophthalmology 2012, 119, 779–788. [Google Scholar] [CrossRef]
- Ho, A.C.; Humayun, M.S.; Dorn, J.D.; da Cruz, L.; Dagnelie, G.; Handa, J.; Barale, P.O.; Sahel, J.A.; Stanga, P.E.; Hafezi, F.; et al. Long-Term Results from an Epiretinal Prosthesis to Restore Sight to the Blind. Ophthalmology 2015, 122, 1547–1554. [Google Scholar] [CrossRef]
- Richard, G.; Hornig, R.; Keserü, M.; Feucht, M. Chronic Epiretinal Chip Implant in Blind Patients with Retinitis Pigmentosa: Long-Term Clinical Results. Investig. Ophthalmol. Vis. Sci. 2007, 48, 666. [Google Scholar]
- Hornig, R.; Zehnder, T.; Velikay-Parel, M.; Laube, T.; Feucht, M.; Richard, G. The IMI Retinal Implant System. In Artificial Sight: Basic Research, Biomedical Engineering, and Clinical Advances; Humayun, M.S., Weiland, J.D., Chader, G., Greenbaum, E., Eds.; Springer: New York, NY, USA, 2007; pp. 111–128. [Google Scholar]
- Richard, G.; Keserue, M.; Feucht, M.; Post, N.; Hornig, R. Visual Perception After Long-Term Implantation of a Retinal Implant. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1786. [Google Scholar]
- Keserue, M.; Post, N.; Hornig, R.; Zeitz, O.; Richard, G. Long Term Tolerability of the First Wireless Implant for Electrical Epiretinal Stimulation. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4226. [Google Scholar]
- Hornig, R.; Dapper, M.; Le Joliff, E.; Hill, R.; Ishaque, K.; Posch, C.; Benosman, R.; LeMer, Y.; Sahel, J.A.; Picaud, S. Pixium Vision: First Clinical Results and Innovative Developments. In Artificial Vision; Gabel, V.P., Ed.; Springer: New York, NY, USA, 2017; pp. 99–113. [Google Scholar]
- Roessler, G.; Laube, T.; Brockmann, C.; Kirschkamp, T.; Mazinani, B.; Goertz, M.; Koch, C.; Krisch, I.; Sellhaus, B.; Trieu, H.K.; et al. Implantation and Explantation of a Wireless Epiretinal Retina Implant Device: Observations during the EPIRET3 Prospective Clinical Trial. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3003–3008. [Google Scholar] [CrossRef]
- Stingl, K.; Schippert, R.; Bartz-Schmidt, K.U.; Besch, D.; Cottriall, C.L.; Edwards, T.L.; Gekeler, F.; Greppmaier, U.; Kiel, K.; Koitschev, A.; et al. Interim Results of a Multicenter Trial with the New Electronic Subretinal Implant Alpha AMS in 15 Patients Blind from Inherited Retinal Degenerations. Front. Neurosci. 2017, 11, 445. [Google Scholar] [CrossRef]
- Edwards, T.L.; Cottriall, C.L.; Xue, K.; Simunovic, M.P.; Ramsden, J.D.; Zrenner, E.; MacLaren, R.E. Assessment of the Electronic Retinal Implant Alpha AMS in Restoring Vision to Blind Patients with End-Stage Retinitis Pigmentosa. Ophthalmology 2018, 125, 432–443. [Google Scholar] [CrossRef]
- Ayton, L.N.; Blamey, P.J.; Guymer, R.H.; Luu, C.D.; Nayagam, D.A.; Sinclair, N.C.; Shivdasani, M.N.; Yeoh, J.; McCombe, M.F.; Briggs, R.J.; et al. First-in-Human Trial of a Novel Suprachoroidal Retinal Prosthesis. PLoS ONE 2014, 9, e115239. [Google Scholar] [CrossRef]
- Opie, N.L.; Burkitt, A.N.; Meffin, H.; Grayden, D.B. Heating of the Eye by a Retinal Prosthesis: Modeling, Cadaver and In Vivo Study. IEEE Trans. Biomed. Eng. 2012, 59, 339–345. [Google Scholar] [CrossRef] [PubMed]
- da Cruz, L.; Dorn, J.D.; Humayun, M.S.; Dagnelie, G.; Handa, J.; Barale, P.O.; Sahel, J.A.; Stanga, P.E.; Hafezi, F.; Safran, A.B.; et al. Five-Year Safety and Performance Results from the Argus II Retinal Prosthesis System Clinical Trial. Ophthalmology 2016, 123, 2248–2254. [Google Scholar] [CrossRef] [PubMed]
- Humayun, M.S.; Dorn, J.D.; Ahuja, A.K.; Caspi, A.; Filley, E.; Dagnelie, G.; Salzmann, J.; Santos, A.; Duncan, J.; da Cruz, L.; et al. Preliminary 6 month results from the argustm ii epiretinal prosthesis feasibility study. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 4566–4568. [Google Scholar]
- Ahuja, A.K.; Dorn, J.D.; Caspi, A.; McMahon, M.J.; Dagnelie, G.; da Cruz, L.; Stanga, P.; Humayun, M.S.; Greenberg, R.J.; Group, A.I.S. Blind subjects implanted with the Argus II retinal prosthesis are able to improve performance in a spatial-motor task. Br. J. Ophthalmol. 2011, 95, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Dorn, J.D.; Ahuja, A.K.; Caspi, A.; da Cruz, L.; Dagnelie, G.; Sahel, J.A.; Greenberg, R.J.; McMahon, M.J.; Group, A.I.S. The Detection of Motion by Blind Subjects with the Epiretinal 60-Electrode (Argus II) Retinal Prosthesis. JAMA Ophthalmol. 2013, 131, 183–189. [Google Scholar] [CrossRef]
- da Cruz, L.; Coley, B.F.; Dorn, J.; Merlini, F.; Filley, E.; Christopher, P.; Chen, F.K.; Wuyyuru, V.; Sahel, J.; Stanga, P.; et al. The Argus II epiretinal prosthesis system allows letter and word reading and long-term function in patients with profound vision loss. Br. J. Ophthalmol. 2013, 97, 632–636. [Google Scholar] [CrossRef]
- Kotecha, A.; Zhong, J.; Stewart, D.; da Cruz, L. The Argus II prosthesis facilitates reaching and grasping tasks: A case series. BMC Ophthalmol. 2014, 14, 71. [Google Scholar] [CrossRef]
- Luo, Y.H.L.; Zhong, J.J.; da Cruz, L. The use of Argus® II retinal prosthesis by blind subjects to achieve localisation and prehension of objects in 3-dimensional space. Graef. Arch. Clin. Exp. Ophthalmol. 2015, 253, 1907–1914. [Google Scholar] [CrossRef]
- Dagnelie, G.; Christopher, P.; Arditi, A.; da Cruz, L.; Duncan, J.L.; Ho, A.C.; Olmos de Koo, L.C.; Sahel, J.A.; Stanga, P.E.; Thumann, G.; et al. Performance of real-world functional vision tasks by blind subjects improves after implantation with the Argus® II retinal prosthesis system. Clin. Exp. Ophthalmol. 2017, 45, 152–159. [Google Scholar] [CrossRef]
- Rathbun, D.L.; Shivdasani, M.N.; Guo, T.; Fried, S.; Lovell, N.H.; Hessburg, P. The eye and the chip 2019-Conference Report. J. Neural Eng. 2020, 17, 010401. [Google Scholar] [CrossRef]
- Second Sight Announces Employee Layoffs, Intent to Wind Down Operations and Appointment of Matthew Pfeffer as Acting CEO. Available online: http://investors.secondsight.com/news-releases/news-release-details/second-sight-announces-employee-layoffs-intent-wind-down (accessed on 30 April 2020).
- Lorach, H.; Benosman, R.; Marre, O.; Ieng, S.H.; Sahel, J.A.; Picaud, S. Artificial retina: The multichannel processing of the mammalian retina achieved with a neuromorphic asynchronous light acquisition device. J. Neural Eng. 2012, 9, 066004. [Google Scholar] [CrossRef]
- Pixium Vision Announces its 2018 Annual Results and Provides Business Update. Available online: https://www.globenewswire.com/news-release/2019/02/08/1713837/0/en/Pixium-Vision-announces-its-2018-annual-results-and-provides-business-update.html (accessed on 3 May 2020).
- Waschkowski, F.; Hesse, S.; Rieck, A.C.; Lohmann, T.; Brockmann, C.; Laube, T.; Bornfeld, N.; Thumann, G.; Walter, P.; Mokwa, W.; et al. Development of very large electrode arrays for epiretinal stimulation (VLARS). Biomed. Eng. Online 2014, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Zrenner, E.; Bartz-Schmidt, K.U.; Benav, H.; Besch, D.; Bruckmann, A.; Gabel, V.P.; Gekeler, F.; Greppmaier, U.; Harscher, A.; Kibbel, S.; et al. Subretinal electronic chips allow blind patients to read letters and combine them to words. Proc. R. Soc. B 2010, 278, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Wilke, R.; Gabel, V.P.; Sachs, H.; Bartz Schmidt, K.U.; Gekeler, F.; Besch, D.; Szurman, P.; Stett, A.; Wilhelm, B.; Peters, T.; et al. Spatial Resolution and Perception of Patterns Mediated by a Subretinal 16-Electrode Array in Patients Blinded by Hereditary Retinal Dystrophies. Investig. Ophthalmol. Vis. Sci. 2011, 52, 5995–6003. [Google Scholar] [CrossRef] [PubMed]
- Retina Implant. Available online: https://www.retina-implant.de/en (accessed on 1 May 2020).
- Rizzo, J.F.; Wyatt, J.; Loewenstein, J.; Kelly, S.; Shire, D. Methods and Perceptual Thresholds for Short-Term Electrical Stimulation of Human Retina with Microelectrode Arrays. Investig. Ophthalmol. Vis. Sci. 2003, 44, 5355–5361. [Google Scholar] [CrossRef]
- Rizzo, J.F.; Shire, D.B.; Kelly, S.K.; Troyk, P.; Gingerich, M.; McKee, B.; Priplata, A.; Chen, J.; Drohan, W.; Doyle, P.; et al. Development of the boston retinal prosthesis. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 3135–3138. [Google Scholar]
- Kelly, S.K.; Shire, D.B.; Chen, J.; Doyle, P.; Gingerich, M.D.; Cogan, S.F.; Drohan, W.A.; Behan, S.; Theogarajan, L.; Wyatt, J.L.; et al. A Hermetic Wireless Subretinal Neurostimulator for Vision Prostheses. IEEE Trans. Biomed. Eng. 2011, 58, 3197–3205. [Google Scholar] [CrossRef]
- Kelly, S.K.; Shire, D.B.; Chen, J.; Gingerich, M.D.; Cogan, S.F.; Drohan, W.; Ellersick, W.; Krishnan, A.; Behan, S.; Wyatt, J.L.; et al. Developments on the Boston 256-channel retinal implant. In Proceedings of the 2013 IEEE International Conference on Multimedia and Expo Workshops (ICMEW), San Jose, CA, USA, 15–19 July 2013; pp. 1–6. [Google Scholar]
- Yamauchi, Y.; Franco, L.M.; Jackson, D.J.; Naber, J.F.; Ziv, R.O.; Rizzo, J.F.; Kaplan, H.J.; Enzmann, V. Comparison of electrically evoked cortical potential thresholds generated with subretinal or suprachoroidal placement of a microelectrode array in the rabbit. J. Neural Eng. 2005, 2, S48. [Google Scholar] [CrossRef]
- Shivdasani, M.N.; Sinclair, N.C.; Dimitrov, P.N.; Varsamidis, M.; Ayton, L.N.; Luu, C.D.; Perera, T.; McDermott, H.J.; Blamey, P.J. Factors Affecting Perceptual Thresholds in a Suprachoroidal Retinal Prosthesis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6467–6481. [Google Scholar] [CrossRef]
- Ayton, L.N.; Suaning, G.J.; Lovell, N.H.; Petoe, M.A.; Nayagam, D.A.; Brawn, T.L.E.; Burkitt, A.N. Suprachoroidal Retinal Prostheses. In Artificial Vision; Gabel, V.P., Ed.; Springer: New York, NY, USA, 2017; pp. 125–138. [Google Scholar]
- Abbott, C.J.; Nayagam, D.A.; Luu, C.D.; Epp, S.B.; Williams, R.A.; Salinas-LaRosa, C.M.; Villalobos, V.; McGowan, C.; Shivdasani, M.N.; Burns, O.; et al. Safety Studies for a 44-Channel Suprachoroidal Retinal Prosthesis: A Chronic Passive Study. Investig. Ophthalmol. Vis. Sci. 2018, 59, 1410–1424. [Google Scholar] [CrossRef]
- Fujikado, T.; Kamei, M.; Sakaguchi, H.; Kanda, H.; Endo, T.; Hirota, M.; Morimoto, T.; Nishida, K.; Kishima, H.; Terasawa, Y.; et al. One-Year Outcome of 49-Channel Suprachoroidal–Transretinal Stimulation Prosthesis in Patients with Advanced Retinitis Pigmentosa. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6147–6157. [Google Scholar] [CrossRef]
- Endo, T.; Fujikado, T.; Hirota, M.; Kanda, H.; Morimoto, T.; Nishida, K. Light localization with low-contrast targets in a patient implanted with a suprachoroidal–transretinal stimulation retinal prosthesis. Graef. Arch. Clin. Exp. Ophthalmol. 2018, 256, 1723–1729. [Google Scholar] [CrossRef]
- Stronks, H.C.; Dagnelie, G. The functional performance of the Argus II retinal prosthesis. Expert Rev. Med. Devices 2014, 11, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Koo, K.; Lee, S.; Ban, J.; Jeong, H.; Park, H.; Ha, S.; Seo, J.M.; Chung, H.; Cho, D. Arrowhead-shaped micro-electrode array on polyimide substrate for retinal prostheses enabling close approach to target cells. In Proceedings of the TRANSDUCERS 2009–2009 International Solid-State Sensors, Actuators and Microsystems Conference, Denver, CO, USA, 21–25 June 2009; pp. 342–345. [Google Scholar]
- Butterwick, A.; Huie, P.; Jones, B.W.; Marc, R.E.; Marmor, M.; Palanker, D. Effect of shape and coating of a subretinal prosthesis on its integration with the retina. Exp. Eye Res. 2009, 88, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Huang, X.; Liu, G.; Wang, W.; Dong, F.; Li, Z. Fabrication and Characterization of a Parylene-Based Three-Dimensional Microelectrode Array for Use in Retinal Prosthesis. J. Microelectromech. Sys. 2010, 19, 367–374. [Google Scholar] [CrossRef]
- Yanovitz, L.; Raz-Prag, D.; Eden, K.O.; Saini, R.; Hanein, Y.; Gefen, R.A. Retinal responses evoked and recorded with 3D electrodes designated for a novel prosthetic device. Investig. Ophthalmol. Vis. Sci. 2014, 55, 1808. [Google Scholar]
- Nano Retina. Available online: https://www.nano-retina.com (accessed on 1 May 2020).
- Djilas, M.; Oles, C.; Lorach, H.; Bendali, A.; Degardin, J.; Dubus, E.; Lissorgues-Bazin, G.; Rousseau, L.; Benosman, R.; Ieng, S.H.; et al. Three-dimensional electrode arrays for retinal prostheses: Modeling, geometry optimization and experimental validation. J. Neural Eng. 2011, 8, 046020. [Google Scholar] [CrossRef][Green Version]
- Bendali, A.; Rousseau, L.; Lissorgues, G.; Scorsone, E.; Djilas, M.; Dégardin, J.; Dubus, E.; Fouquet, S.; Benosman, R.; Bergonzo, P.; et al. Synthetic 3D diamond-based electrodes for flexible retinal neuroprostheses: Model, production and in vivo biocompatibility. Biomaterials 2015, 67, 73–83. [Google Scholar] [CrossRef]
- Cao, X.; Sui, X.; Lyu, Q.; Li, L.; Chai, X. Effects of different three-dimensional electrodes on epiretinal electrical stimulation by modeling analysis. J. Neuroeng. Rehabil. 2015, 12, 73. [Google Scholar] [CrossRef]
- Cicione, R.; Shivdasani, M.N.; Fallon, J.B.; Luu, C.D.; Allen, P.J.; Rathbone, G.D.; Shepherd, R.K.; Williams, C.E. Visual cortex responses to suprachoroidal electrical stimulation of the retina: Effects of electrode return configuration. J. Neural Eng. 2012, 9, 036009. [Google Scholar] [CrossRef]
- Moghaddam, G.K.; Lovell, N.H.; Wilke, R.G.; Suaning, G.J.; Dokos, S. Performance optimization of current focusing and virtual electrode strategies in retinal implants. Comput. Meth. Prog. Bio. 2014, 117, 334–342. [Google Scholar] [CrossRef]
- Palanker, D.; Vankov, A.; Huie, P.; Baccus, S. Design of a high-resolution optoelectronic retinal prosthesis. J. Neural Eng. 2005, 2, S105. [Google Scholar] [CrossRef]
- Habib, A.G.; Cameron, M.A.; Suaning, G.J.; Lovell, N.H.; Morley, J.W. Spatially restricted electrical activation of retinal ganglion cells in the rabbit retina by hexapolar electrode return configuration. J. Neural Eng. 2013, 10, 036013. [Google Scholar] [CrossRef] [PubMed]
- Dumm, G.; Fallon, J.B.; Williams, C.E.; Shivdasani, M.N. Virtual Electrodes by Current Steering in Retinal Prostheses. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8077–8085. [Google Scholar] [CrossRef] [PubMed]
- Spencer, T.C.; Fallon, J.B.; Thien, P.C.; Shivdasani, M.N. Spatial Restriction of Neural Activation Using Focused Multipolar Stimulation with a Retinal Prosthesis. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3181–3191. [Google Scholar] [CrossRef][Green Version]
- Spencer, T.C.; Fallon, J.B.; Shivdasani, M.N. Creating virtual electrodes with 2D current steering. J. Neural Eng. 2018, 15, 035002. [Google Scholar] [CrossRef]
- Spencer, T.C.; Fallon, J.B.; Abbott, C.J.; Allen, P.J.; Brandli, A.; Luu, C.D.; Epp, S.B.; Shivdasani, M.N. Electrical Field Shaping Techniques in a Feline Model of Retinal Degeneration. In Proceedings of the 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Honolulu, HI, USA, 18–21 July 2018; pp. 1222–1225. [Google Scholar]
- Fried, S.I.; Hsueh, H.A.; Werblin, F.S. A Method for Generating Precise Temporal Patterns of Retinal Spiking Using Prosthetic Stimulation. J. Neurophysiol. 2006, 95, 970–978. [Google Scholar] [CrossRef]
- Margalit, E.; Thoreson, W.B. Inner Retinal Mechanisms Engaged by Retinal Electrical Stimulation. Investig. Ophthalmol. Vis. Sci. 2006, 47, 2606–2612. [Google Scholar] [CrossRef]
- Freeman, D.K.; Eddington, D.K.; Rizzo, J.F., III; Fried, S.I. Selective Activation of Neuronal Targets with Sinusoidal Electric Stimulation. J. Neurophysiol. 2010, 104, 2778–2791. [Google Scholar] [CrossRef]
- Nanduri, D.; Fine, I.; Horsager, A.; Boynton, G.M.; Humayun, M.S.; Greenberg, R.J.; Weiland, J.D. Frequency and Amplitude Modulation Have Different Effects on the Percepts Elicited by Retinal Stimulation. Investig. Ophthalmol. Vis. Sci. 2012, 53, 205–214. [Google Scholar] [CrossRef]
- Weitz, A.C.; Nanduri, D.; Behrend, M.R.; Gonzalez-Calle, A.; Greenberg, R.J.; Humayun, M.S.; Chow, R.H.; Weiland, J.D. Improving the spatial resolution of epiretinal implants by increasing stimulus pulse duration. Sci. Transl. Med. 2015, 7, 318ra203. [Google Scholar] [CrossRef]
- Im, M.; Werginz, P.; Fried, S.I. Electric stimulus duration alters network-mediated responses depending on retinal ganglion cell type. J. Neural Eng. 2018, 15, 036010. [Google Scholar] [CrossRef]
- Lee, J.I.; Im, M. Optimal Electric Stimulus Amplitude Improves the Selectivity Between Responses of ON Versus OFF Types of Retinal Ganglion Cells. IEEE Trans. Neural Sys. Rehabil. Eng. 2019, 27, 2015–2024. [Google Scholar] [CrossRef]
- Curcio, C.A.; Allen, K.A. Topography of ganglion cells in human retina. J. Comp. Neurol. 1990, 300, 5–25. [Google Scholar] [CrossRef]
- Martin, P.R.; Grünert, U. Spatial density and immunoreactivity of bipolar cells in the macaque monkey retina. J. Comp. Neurol. 1992, 323, 269–287. [Google Scholar] [CrossRef]
- Boyden, E.S.; Zhang, F.; Bamberg, E.; Nagel, G.; Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8, 1263–1268. [Google Scholar] [CrossRef]
- Han, X.; Boyden, E.S. Multiple-Color Optical Activation, Silencing, and Desynchronization of Neural Activity, with Single-Spike Temporal Resolution. PLoS ONE 2007, 2, e299. [Google Scholar] [CrossRef]
- Busskamp, V.; Roska, B. Optogenetic approaches to restoring visual function in retinitis pigmentosa. Curr. Opin. Neurobiol. 2011, 21, 942–946. [Google Scholar] [CrossRef]
- Busskamp, V.; Picaud, S.; Sahel, J.A.; Roska, B. Optogenetic therapy for retinitis pigmentosa. Gene Ther. 2012, 19, 169–175. [Google Scholar] [CrossRef]
- Duebel, J.; Marazova, K.; Sahel, J.A. Optogenetics. Curr. Opin. Ophthalmol. 2015, 26, 226. [Google Scholar] [CrossRef]
- Jones, B.W.; Pfeiffer, R.L.; Ferrell, W.D.; Watt, C.B.; Marmor, M.; Marc, R.E. Retinal remodeling in human retinitis pigmentosa. Exp. Eye Res. 2016, 150, 149–165. [Google Scholar] [CrossRef]
- Bi, A.; Cui, J.; Ma, Y.P.; Olshevskaya, E.; Pu, M.; Dizhoor, A.M.; Pan, Z.H. Ectopic Expression of a Microbial-Type Rhodopsin Restores Visual Responses in Mice with Photoreceptor Degeneration. Neuron 2006, 50, 23–33. [Google Scholar] [CrossRef]
- Lagali, P.S.; Balya, D.; Awatramani, G.B.; Münch, T.A.; Kim, D.S.; Busskamp, V.; Cepko, C.L.; Roska, B. Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nat. Neurosci. 2008, 11, 667. [Google Scholar] [CrossRef]
- Lin, B.; Koizumi, A.; Tanaka, N.; Panda, S.; Masland, R.H. Restoration of visual function in retinal degeneration mice by ectopic expression of melanopsin. Proc. Natl. Acad. Sci. USA 2008, 105, 16009–16014. [Google Scholar] [CrossRef]
- Gaub, B.M.; Berry, M.H.; Holt, A.E.; Isacoff, E.Y.; Flannery, J.G. Optogenetic Vision Restoration Using Rhodopsin for Enhanced Sensitivity. Mol. Ther. 2015, 23, 1562–1571. [Google Scholar] [CrossRef]
- Berry, M.H.; Holt, A.; Levitz, J.; Broichhagen, J.; Gaub, B.M.; Visel, M.; Stanley, C.; Aghi, K.; Kim, Y.J.; Cao, K.; et al. Restoration of patterned vision with an engineered photoactivatable G protein-coupled receptor. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Ivanova, E.; Pan, Z.H. Evaluation of the adeno-associated virus mediated long-term expression of channelrhodopsin-2 in the mouse retina. Mol. Vis. 2009, 15, 1680. [Google Scholar]
- Kleinlogel, S.; Feldbauer, K.; Dempski, R.E.; Fotis, H.; Wood, P.G.; Bamann, C.; Bamberg, E. Ultra light-sensitive and fast neuronal activation with the Ca2+-permeable channelrhodopsin CatCh. Nat. Neurosci. 2011, 14, 513–518. [Google Scholar] [CrossRef]
- Zhang, F.; Vierock, J.; Yizhar, O.; Fenno, L.E.; Tsunoda, S.; Kianianmomeni, A.; Prigge, M.; Berndt, A.; Cushman, J.; Polle, J.; et al. The Microbial Opsin Family of Optogenetic Tools. Cell 2011, 147, 1446–1457. [Google Scholar] [CrossRef]
- Chaffiol, A.; Caplette, R.; Jaillard, C.; Brazhnikova, E.; Desrosiers, M.; Dubus, E.; Duhamel, L.; Macé, E.; Marre, O.; Benoit, P.; et al. A New Promoter Allows Optogenetic Vision Restoration with Enhanced Sensitivity in Macaque Retina. Mol. Ther. 2017, 25, 2546–2560. [Google Scholar] [CrossRef]
- Berry, M.H.; Holt, A.; Salari, A.; Veit, J.; Visel, M.; Levitz, J.; Aghi, K.; Gaub, B.M.; Sivyer, B.; Flannery, J.G.; et al. Restoration of high-sensitivity and adapting vision with a cone opsin. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Ganjawala, T.H.; Lu, Q.; Fenner, M.D.; Abrams, G.W.; Pan, Z.H. Improved CoChR Variants Restore Visual Acuity and Contrast Sensitivity in a Mouse Model of Blindness under Ambient Light Conditions. Mol. Ther. 2019, 27, 1195–1205. [Google Scholar] [CrossRef]
- Klapoetke, N.C.; Murata, Y.; Kim, S.S.; Pulver, S.R.; Birdsey-Benson, A.; Cho, Y.K.; Morimoto, T.K.; Chuong, A.S.; Carpenter, E.J.; Tian, Z.; et al. Independent optical excitation of distinct neural populations. Nat. Methods 2014, 11, 338. [Google Scholar] [CrossRef]
- Chuong, A.S.; Miri, M.L.; Busskamp, V.; Matthews, G.A.; Acker, L.C.; Sørensen, A.T.; Young, A.; Klapoetke, N.C.; Henninger, M.A.; Kodandaramaiah, S.B.; et al. Noninvasive optical inhibition with a red-shifted microbial rhodopsin. Nat. Neurosci. 2014, 17, 1123. [Google Scholar] [CrossRef]
- RetroSense Therapeutics Doses First Patient in Phase I/II Clinical Trial for Lead Compound RST-001. Available online: https://rbvcapital.com/en/news/retrosense-therapeutics-doses-first-patient-in-phase-i/ii-clinical-trial-for-lead-compound-rst-001.html (accessed on 15 May 2020).
- GS030 for Retinitis Pigmentosa. Available online: https://www.gensight-biologics.com/product/gs030-for-retinitis-pigmentosa/?cn-reloaded=1 (accessed on 15 May 2020).
- Chow, A.Y.; Pardue, M.T.; Chow, V.Y.; Peyman, G.A.; Liang, C.; Perlman, J.I.; Peachey, N.S. Implantation of silicon chip microphotodiode arrays into the cat subretinal space. IEEE Trans. Neural Sys. Rehabil. Eng. 2001, 9, 86–95. [Google Scholar] [CrossRef]
- Chow, A.Y.; Chow, V.Y.; Packo, K.H.; Pollack, J.S.; Peyman, G.A.; Schuchard, R. The Artificial Silicon Retina Microchip for the Treatment of Vision Loss from Retinitis Pigmentosa. Arch. Ophthalmol. 2004, 122, 460–469. [Google Scholar] [CrossRef]
- Chow, A.Y.; Bittner, A.K.; Pardue, M.T. The Artificial Silicon Retina in Retinitis Pigmentosa Patients (An American Ophthalmological Association thesis). Trans. Am. Ophthalmol. Soc. 2010, 108, 120–154. [Google Scholar] [PubMed]
- Pardue, M.T.; Phillips, M.J.; Yin, H.; Sippy, B.D.; Webb-Wood, S.; Chow, A.Y.; Ball, S.L. Neuroprotective Effect of Subretinal Implants in the RCS Rat. Investig. Ophthalmol. Vis. Sci. 2005, 46, 674–682. [Google Scholar] [CrossRef][Green Version]
- Mathieson, K.; Loudin, J.; Goetz, G.; Huie, P.; Wang, L.; Kamins, T.I.; Galambos, L.; Smith, R.; Harris, J.S.; Sher, A.; et al. Photovoltaic retinal prosthesis with high pixel density. Nat. Photonics 2012, 6, 391. [Google Scholar] [CrossRef]
- Mandel, Y.; Goetz, G.; Lavinsky, D.; Huie, P.; Mathieson, K.; Wang, L.; Kamins, T.; Galambos, L.; Manivanh, R.; Harris, J.; et al. Cortical responses elicited by photovoltaic subretinal prostheses exhibit similarities to visually evoked potentials. Nat. Commun. 2013, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lorach, H.; Goetz, G.; Smith, R.; Lei, X.; Mandel, Y.; Kamins, T.; Mathieson, K.; Huie, P.; Harris, J.; Sher, A.; et al. Photovoltaic restoration of sight with high visual acuity. Nat. Med. 2015, 21, 476. [Google Scholar] [CrossRef]
- Palanker, D.; Le Mer, Y.; Mohand-Said, S.; Muqit, M.M.K.; Sahel, J.A. Photovoltaic Restoration of Central Vision in Atrophic Age-Related Macular Degeneration. Ophthalmology 2020. [Google Scholar] [CrossRef]
- Bosse, B.; Damle, S.; Akinin, A.; Jing, Y.; Bartsch, D.U.; Cheng, L.; Oesch, N.; Lo, Y.H.; Cauwenberghs, G.; Freeman, W.R. In Vivo Photovoltaic Performance of a Silicon Nanowire Photodiode–Based Retinal Prosthesis. Investig. Ophthalmol. Vis. Sci. 2018, 59, 5885–5892. [Google Scholar] [CrossRef] [PubMed]
- Nanovision. Available online: https://nanovisionbio.com (accessed on 3 May 2020).
- Tang, J.; Qin, N.; Chong, Y.; Diao, Y.; Wang, Z.; Xue, T.; Jiang, M.; Zhang, J.; Zheng, G. Nanowire arrays restore vision in blind mice. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, D.; Antognazza, M.R.; Maccarone, R.; Bellani, S.; Lanzarini, E.; Martino, N.; Mete, M.; Pertile, G.; Bisti, S.; Lanzani, G.; et al. A polymer optoelectronic interface restores light sensitivity in blind rat retinas. Nat. Photonics 2013, 7, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Maya-Vetencourt, J.F.; Ghezzi, D.; Antognazza, M.R.; Colombo, E.; Mete, M.; Feyen, P.; Desii, A.; Buschiazzo, A.; Di Paolo, M.; Di Marco, S.; et al. A fully organic retinal prosthesis restores vision in a rat model of degenerative blindness. Nat. Mater. 2017, 16, 681–689. [Google Scholar] [CrossRef]
- Fortin, D.L.; Banghart, M.R.; Dunn, T.W.; Borges, K.; Wagenaar, D.A.; Gaudry, Q.; Karakossian, M.H.; Otis, T.S.; Kristan, W.B.; Trauner, D.; et al. Photochemical control of endogenous ion channels and cellular excitability. Nat. Methods 2008, 5, 331–338. [Google Scholar] [CrossRef]
- Polosukhina, A.; Litt, J.; Tochitsky, I.; Nemargut, J.; Sychev, Y.; De Kouchkovsky, I.; Huang, T.; Borges, K.; Trauner, D.; Van Gelder, R.N.; et al. Photochemical Restoration of Visual Responses in Blind Mice. Neuron 2012, 75, 271–282. [Google Scholar] [CrossRef]
- Benfenati, F.; Lanzani, G. New technologies for developing second generation retinal prostheses. Lab Anim. 2018, 47, 71–75. [Google Scholar] [CrossRef]
- Albert, E.S.; Bec, J.M.; Desmadryl, G.; Chekroud, K.; Travo, C.; Gaboyard, S.; Bardin, F.; Marc, I.; Dumas, M.; Lenaers, G.; et al. TRPV4 channels mediate the infrared laser-evoked response in sensory neurons. J. Neurophysiol. 2012, 107, 3227–3234. [Google Scholar] [CrossRef]
- Porter, J.; Queener, H.M.; Lin, J.E.; Thorn, K.; Awwal, A. Adaptive Optics for Vision Science: Principles, Practices, Design, and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006. [Google Scholar]
- Al-Atabany, W.; McGovern, B.; Mehran, K.; Berlinguer-Palmini, R.; Degenaar, P. A Processing Platform for Optoelectronic/Optogenetic Retinal Prosthesis. IEEE Trans. Biomed. Eng. 2011, 60, 781–791. [Google Scholar] [CrossRef]
- Strettoi, E. A Survey of Retinal Remodeling. Front. Cell. Neurosci. 2015, 9, 494. [Google Scholar] [CrossRef]
- Im, M.; Fried, S.I. Indirect activation elicits strong correlations between light and electrical responses in ON but not OFF retinal ganglion cells. J. Physiol. 2015, 593, 3577–3596. [Google Scholar] [CrossRef] [PubMed]
- Im, M.; Kim, S.W. Neurophysiological and medical considerations for better-performing microelectronic retinal prostheses. J. Neural Eng. 2020. [Google Scholar] [CrossRef] [PubMed]
- Fry, W.J.; Mosberg, W.H.; Barnard, J.W.; Fry, F.J. Production of Focal Destructive Lesions in the Central Nervous System with Ultrasound. J. Neurosurg. 1954, 11, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Gavrilov, L.R.; Tsirulnikov, E.M.; Davies, I.A.I. Application of focused ultrasound for the stimulation of neural structures. Ultrasound Med. Biol. 1996, 22, 179–192. [Google Scholar] [CrossRef]
- Naor, O.; Krupa, S.; Shoham, S. Ultrasonic neuromodulation. J. Neural Eng. 2016, 13, 031003. [Google Scholar] [CrossRef] [PubMed]
- Blackmore, J.; Shrivastava, S.; Sallet, J.; Butler, C.R.; Cleveland, R.O. Ultrasound Neuromodulation: A Review of Results, Mechanisms and Safety. Ultrasound Med. Biol. 2019, 45, 1509–1536. [Google Scholar] [CrossRef]
- Tufail, Y.; Matyushov, A.; Baldwin, N.; Tauchmann, M.L.; Georges, J.; Yoshihiro, A.; Tillery, S.I.H.; Tyler, W.J. Transcranial Pulsed Ultrasound Stimulates Intact Brain Circuits. Neuron 2010, 66, 681–694. [Google Scholar] [CrossRef]
- Tufail, Y.; Yoshihiro, A.; Pati, S.; Li, M.M.; Tyler, W.J. Ultrasonic neuromodulation by brain stimulation with transcranial ultrasound. Nat. Protoc. 2011, 6, 1453–1470. [Google Scholar] [CrossRef]
- Qiu, W.; Zhou, J.; Chen, Y.; Su, M.; Li, G.; Zhao, H.; Gu, X.; Meng, D.; Wang, C.; Xiao, Y.; et al. A Portable Ultrasound System for Non-Invasive Ultrasonic Neuro-Stimulation. IEEE Trans. Neural Sys. Rehabil. Eng. 2017, 25, 2509–2515. [Google Scholar] [CrossRef]
- Darrow, D.P. Focused Ultrasound for Neuromodulation. Neurotherapeutics 2019, 16, 88–99. [Google Scholar] [CrossRef]
- Hertzberg, Y.; Naor, O.; Volovick, A.; Shoham, S. Towards multifocal ultrasonic neural stimulation: Pattern generation algorithms. J. Neural Eng. 2010, 7, 056002. [Google Scholar] [CrossRef] [PubMed]
- Naor, O.; Hertzberg, Y.; Zemel, E.; Kimmel, E.; Shoham, S. Towards multifocal ultrasonic neural stimulation II: Design considerations for an acoustic retinal prosthesis. J. Neural Eng. 2012, 9, 026006. [Google Scholar] [CrossRef] [PubMed]
- Menz, M.D.; Oralkan, Ö.; Khuri-Yakub, P.T.; Baccus, S.A. Precise Neural Stimulation in the Retina Using Focused Ultrasound. J. Neurosci. 2013, 33, 4550–4560. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Kumar, M. An ultrasound-based noninvasive neural interface to the retina. In Proceedings of the IEEE International Ultrasonics Symposium, Chicago, IL, USA, 3–6 September 2014; pp. 2623–2626. [Google Scholar]
- Gao, M.; Yu, Y.; Zhao, H.; Li, G.; Jiang, H.; Wang, C.; Cai, F.; Chan, L.L.H.; Chiu, B.; Qian, W.; et al. Simulation Study of an Ultrasound Retinal Prosthesis With a Novel Contact-Lens Array for Noninvasive Retinal Stimulation. IEEE Trans. Neural Sys. Rehabil. Eng. 2017, 25, 1605–1611. [Google Scholar] [CrossRef]
- Jiang, Q.; Li, G.; Zhao, H.; Sheng, W.; Yue, L.; Su, M.; Weng, S.; Chan, L.L.H.; Zhou, Q.; Humayun, M.S.; et al. Temporal Neuromodulation of Retinal Ganglion Cells by Low-Frequency Focused Ultrasound Stimulation. IEEE Trans. Neural Sys. Rehabil. Eng. 2018, 26, 969–976. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Z.; Cai, F.; Su, M.; Jiang, Q.; Zhou, Q.; Humayun, M.S.; Qiu, W.; Zheng, H. A Novel Racing Array Transducer for Noninvasive Ultrasonic Retinal Stimulation: A Simulation Study. Sensors 2019, 19, 1825. [Google Scholar] [CrossRef]
- Geddes, L.A. Optimal stimulus duration for extracranial cortical stimulation. Neurosurgery 1987, 20, 94–99. [Google Scholar] [CrossRef]
- Grandori, F.; Ravazzani, P. Magnetic stimulation of the motor cortex-theoretical considerations. IEEE Trans. Biomed. Eng. 1991, 38, 180–191. [Google Scholar] [CrossRef]
- Rossini, P.M.; Barker, A.T.; Berardelli, A.; Caramia, M.D.; Caruso, G.; Cracco, R.Q.; Dimitrijević, M.R.; Hallett, M.; Katayama, Y.; Lücking, C.H.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord and roots: Basic principles and procedures for routine clinical application. Report of an IFCN committee. Electroen. Clin. Neuro. 1994, 91, 79–92. [Google Scholar] [CrossRef]
- Kobayashi, M.; Pascual-Leone, A. Transcranial magnetic stimulation in neurology. Lancet Neurol. 2003, 2, 145–156. [Google Scholar] [CrossRef]
- Rotenstreich, Y.; Tzameret, A.; Zangen, A. Transcranial Magnetic Stimulation Improves Retinal Function in an Animal Model of Retinal Dystrophy. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5562. [Google Scholar]
- Marg, E. Magnetostimulation of vision: Direct noninvasive stimulation of the retina and the visual brain. Optometry Vis. Sci. 1991, 68, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Lövsund, P.; Nilsson, S.E.G.; Öberg, P.A. Influence on frog retina of alternating magnetic fields with special reference to ganglion cell activity. Med. Biol. Eng. Comput. 1981, 19, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.Y.; Ahn, J.H.; Pi, K.; Goo, Y.S.; Cho, D.I.D. Electrodeless, Non-Invasive Stimulation of Retinal Neurons Using Time-Varying Magnetic Fields. IEEE Sens. J. 2016, 16, 8832–8839. [Google Scholar] [CrossRef]
- Basham, E.; Sivaprakasam, M.; Liu, W. Functional Magnetic Stimulation for Implantable Epiretinal Prosthesis. Investig. Ophthalmol. Vis. Sci. 2005, 46, 1485. [Google Scholar]
- Basham, E.; Liu, W.; Yang, Z. In vitro magnetic stimulation of unmyelinated nerves. In Proceedings of the 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 2385–2388. [Google Scholar]
- Basham, E.; Yang, Z.; Liu, W. Circuit and Coil Design for In-Vitro Magnetic Neural Stimulation Systems. IEEE Trans. Biomed. Circ. Sys. 2009, 3, 321–331. [Google Scholar] [CrossRef]
- Lee, S.W.; Bonmassar, G.; Fried, S.I. Activation of Retinal Ganglion Cells By Microcoil-Induced Magnetic Stimulation. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5530. [Google Scholar]
- Bonmassar, G.; Lee, S.W.; Freeman, D.K.; Polasek, M.; Fried, S.I.; Gale, J.T. Microscopic magnetic stimulation of neural tissue. Nat. Commun. 2012, 3, 1–10. [Google Scholar] [CrossRef]
- Lee, S.W.; Fallegger, F.; Casse, B.D.; Fried, S.I. Implantable microcoils for intracortical magnetic stimulation. Sci. Adv. 2016, 2, e1600889. [Google Scholar] [CrossRef]
- Finlayson, P.G.; Iezzi, R. Glutamate Stimulation of Retinal Ganglion Cells in Normal and S334ter-4 Rat Retinas: A Candidate for a Neurotransmitter-Based Retinal Prosthesis. Investig. Ophthalmol. Vis. Sci. 2010, 51, 3619–3628. [Google Scholar] [CrossRef]
- Inayat, S.; Rountree, C.M.; Troy, J.B.; Saggere, L. Chemical stimulation of rat retinal neurons: Feasibility of an epiretinal neurotransmitter-based prosthesis. J. Neural Eng. 2014, 12, 016010. [Google Scholar] [CrossRef] [PubMed]
- Bareket, L.; Waiskopf, N.; Rand, D.; Lubin, G.; David-Pur, M.; Ben-Dov, J.; Roy, S.; Eleftheriou, C.; Sernagor, E.; Cheshnovsky, O.; et al. Semiconductor Nanorod–Carbon Nanotube Biomimetic Films for Wire-Free Photostimulation of Blind Retinas. Nano Lett. 2014, 14, 6685–6692. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, D. Retinal prostheses: Progress toward the next generation implants. Front. Neurosci. 2015, 9, 290. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriou, C.G.; Zimmermann, J.B.; Kjeldsen, H.D.; David-Pur, M.; Hanein, Y.; Sernagor, E. Carbon nanotube electrodes for retinal implants: A study of structural and functional integration over time. Biomaterials 2017, 112, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.W.; Yu, Z.Y.; Cheng, S.J.; Chung, J.H.; Liu, X.; Wu, C.Y.; Lin, S.F.; Chen, G.Y. Graphene Oxide–Based Nanomaterials: An Insight into Retinal Prosthesis. Int. J. Mol. Sci. 2020, 21, 2957. [Google Scholar] [CrossRef]
- Parashar, K.; Prajapati, D.; Mcintyre, R.; Kandasubramanian, B. Advancements in Biological Neural Interfaces using Conducting Polymer: A Review. Ind. Eng. Chem. Res. 2020. [Google Scholar] [CrossRef]

| Type | Device Name | Electrodes | Clinical Trial Results | Ref. | ||||
|---|---|---|---|---|---|---|---|---|
| Number | Size (μm) | Pitch (μm) | Number of Subjects | Visual Field | Visual Acuity 8 | |||
| Epi-retinal | Argus II | 60 | Ø 200 | 525 | 30 | ~20° | 20/1262 (grating) | [67,68] |
| IMI 1 | 49 | Ø 250 | 120 | 20 6, 7 7 | - | - | [69,70,71,72] | |
| IRIS 2 | 150 | - | - | 20 | - | - | [73] | |
| EPI-RET3 | 25 | Ø 100 5 | 500 | 6 | - | - | [35,74] | |
| Sub-retinal | Alpha IMS | 1500 | 50 × 50 | 70 | 29 | 11° × 11° | 20/546 (Landolt-C) | [37,66] |
| Alpha AMS | 1600 | - | - | 15 | ~15° | - | [75,76] | |
| Supra-choroidal | BVA 3 | 33 | Ø 400, Ø 600 | 1000 | 3 | ~12° | 20/4451 (Landolt-C) | [77] |
| STS 4 | 49 | Ø 500 | 700 | 2 | 20° × 16° | - | [36] | |
| Transducer | Device Placement | Stimulation Target | Acoustic Frequency (MHz) | Average Intensity (W/cm2) | Spatial Resolution (mm) | Ref. |
|---|---|---|---|---|---|---|
| Phased array | External to the cornea 2 | RGCs 3 (simulation, in vivo) | 0.5 | 0.12–0.42 | 0.40–0.53 (estimated) | [182,183] |
| 1.0 | 5.15–8.52 | |||||
| Single transducer | - | RGCs 4 (in vitro) | 43 | 10.0–30.0 | ~0.10 | [184] |
| 2D CMUT 1 | In front of the eye | Retina (simulation) | 40 | - | - | [185] |
| Racing array | Attached on the cornea like a contact lens | RGCs 5 (simulation) | 2.5 | 0.20–0.60 | 1.30 | [186,188] |
| 5.0 | 0.60 | |||||
| 10 | 0.26 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shim, S.; Eom, K.; Jeong, J.; Kim, S.J. Retinal Prosthetic Approaches to Enhance Visual Perception for Blind Patients. Micromachines 2020, 11, 535. https://doi.org/10.3390/mi11050535
Shim S, Eom K, Jeong J, Kim SJ. Retinal Prosthetic Approaches to Enhance Visual Perception for Blind Patients. Micromachines. 2020; 11(5):535. https://doi.org/10.3390/mi11050535
Chicago/Turabian StyleShim, Shinyong, Kyungsik Eom, Joonsoo Jeong, and Sung June Kim. 2020. "Retinal Prosthetic Approaches to Enhance Visual Perception for Blind Patients" Micromachines 11, no. 5: 535. https://doi.org/10.3390/mi11050535
APA StyleShim, S., Eom, K., Jeong, J., & Kim, S. J. (2020). Retinal Prosthetic Approaches to Enhance Visual Perception for Blind Patients. Micromachines, 11(5), 535. https://doi.org/10.3390/mi11050535





