Antiallodynic Effects of Bee Venom in an Animal Model of Complex Regional Pain Syndrome Type 1 (CRPS-I)
Abstract
:1. Introduction
2. Results
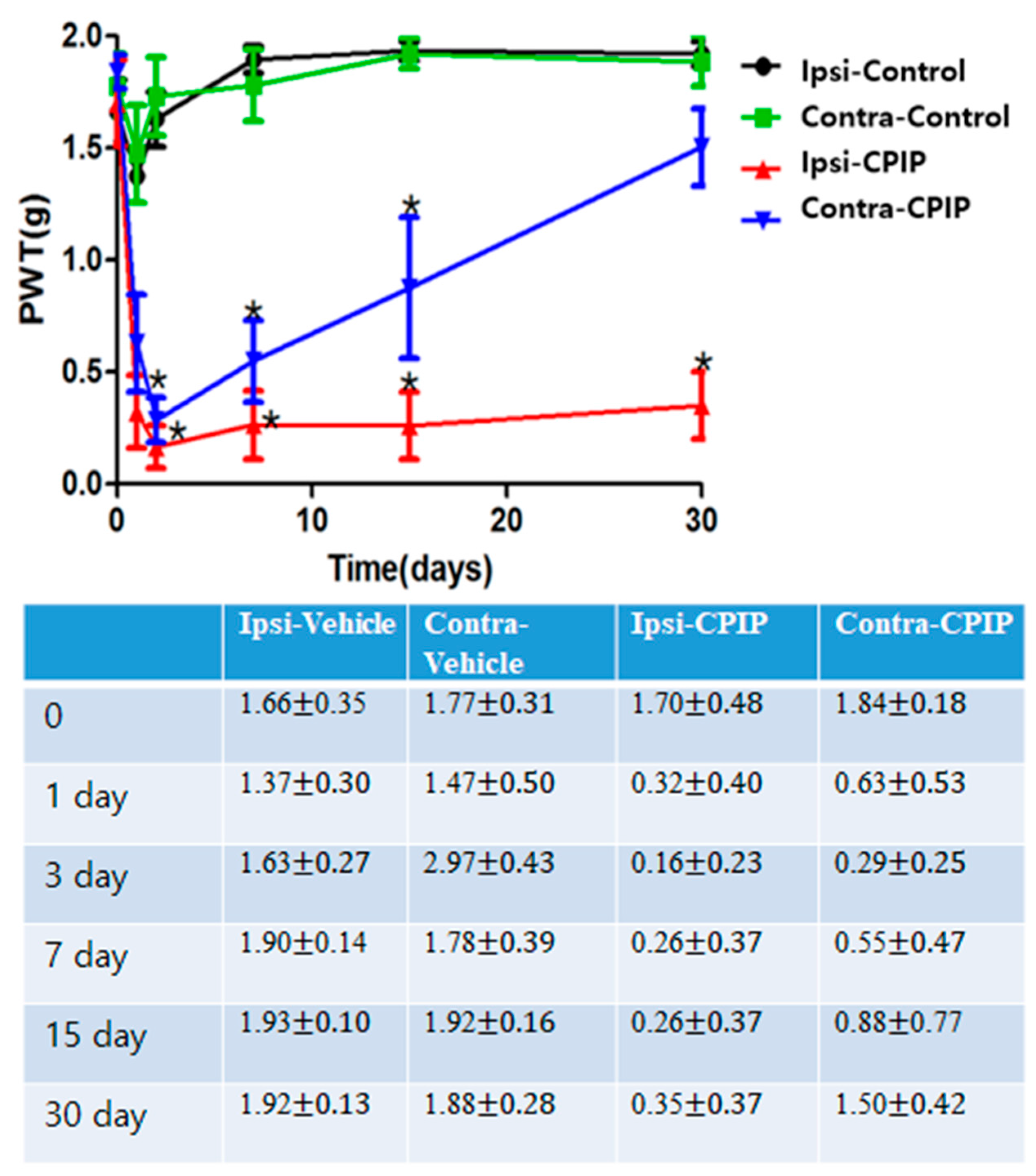
2.1. CPIP Mice Exhibited Prominent Mechano-Allodynia
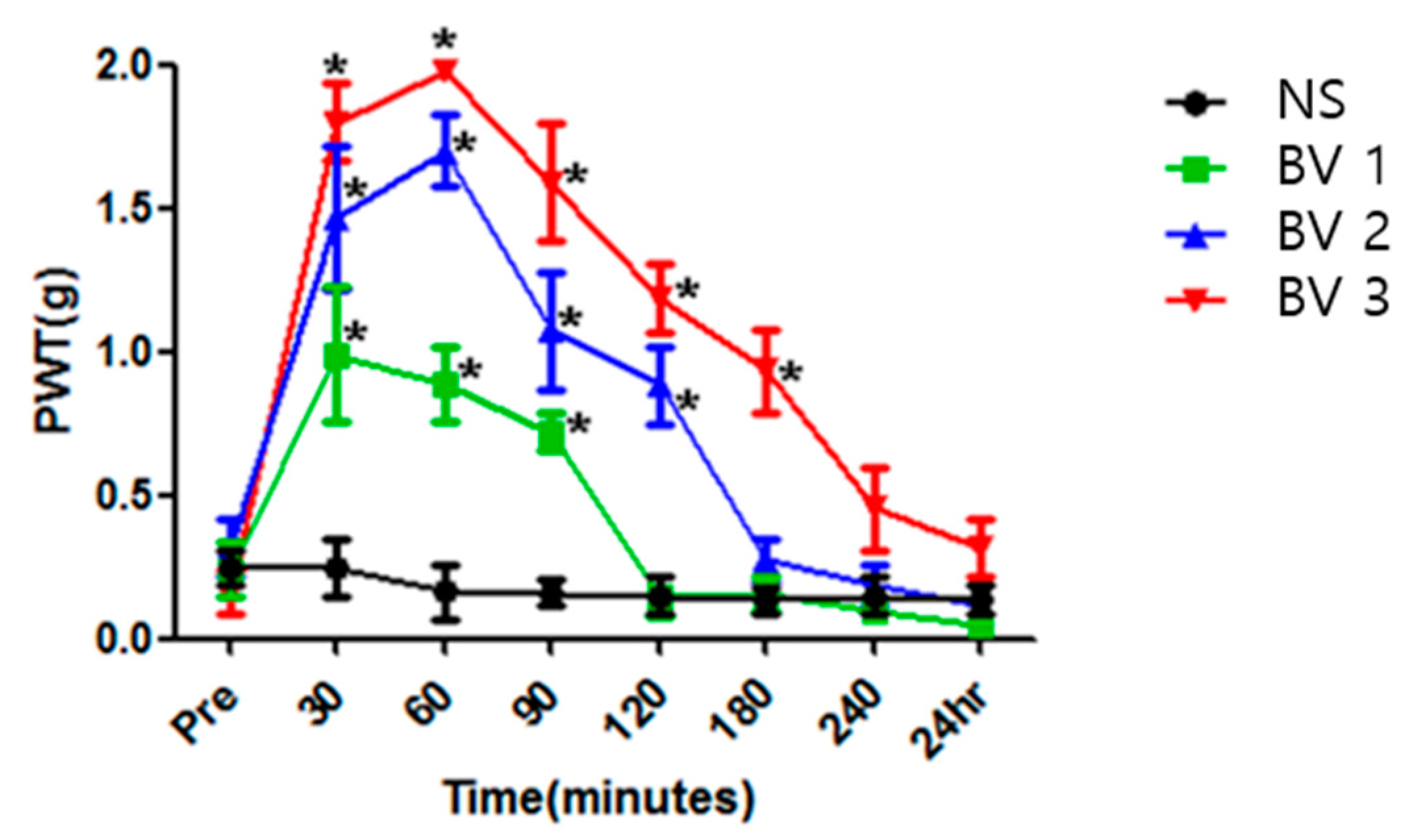
2.2. BV Attenuated Mechanical Allodynia in CPIP Mice
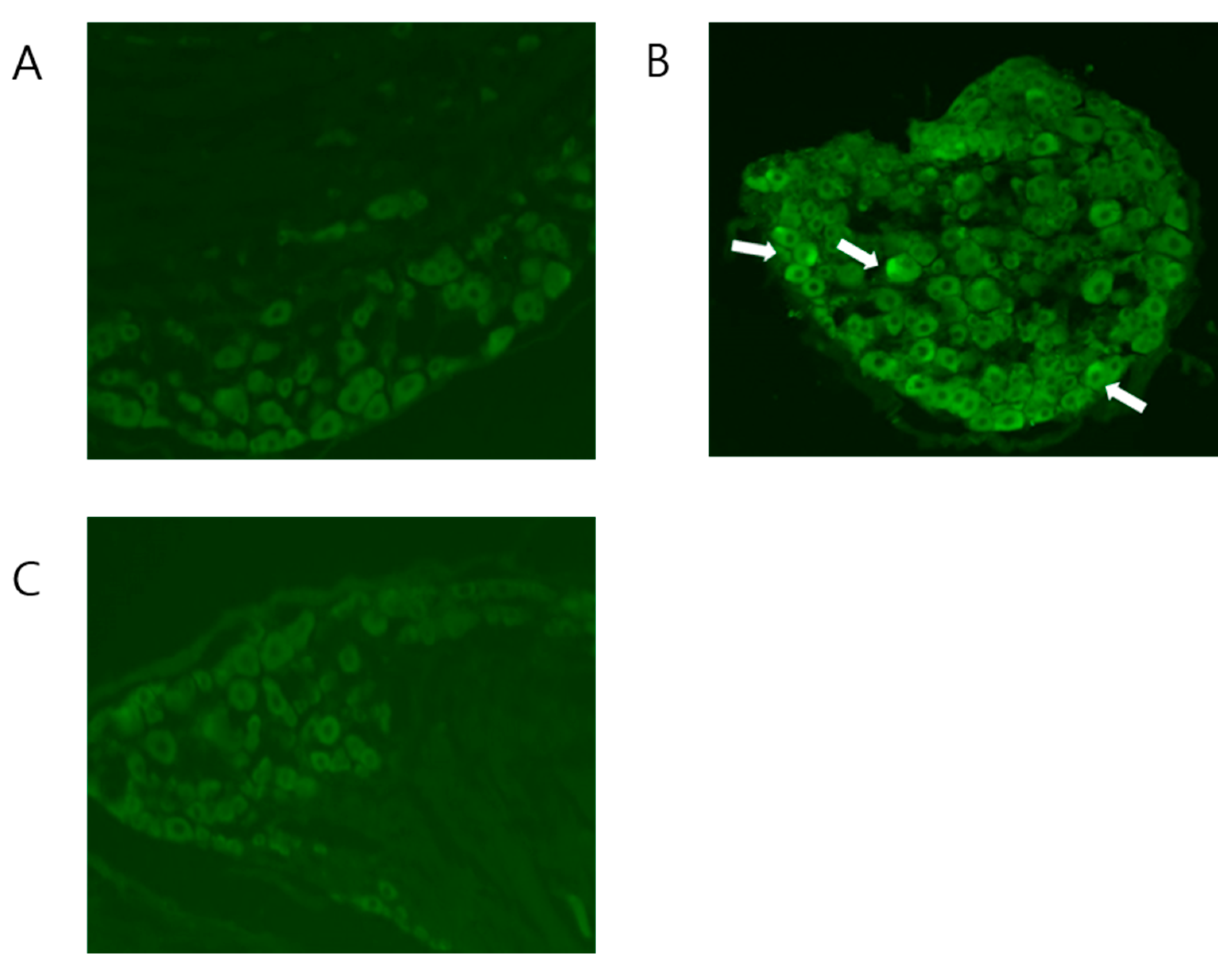
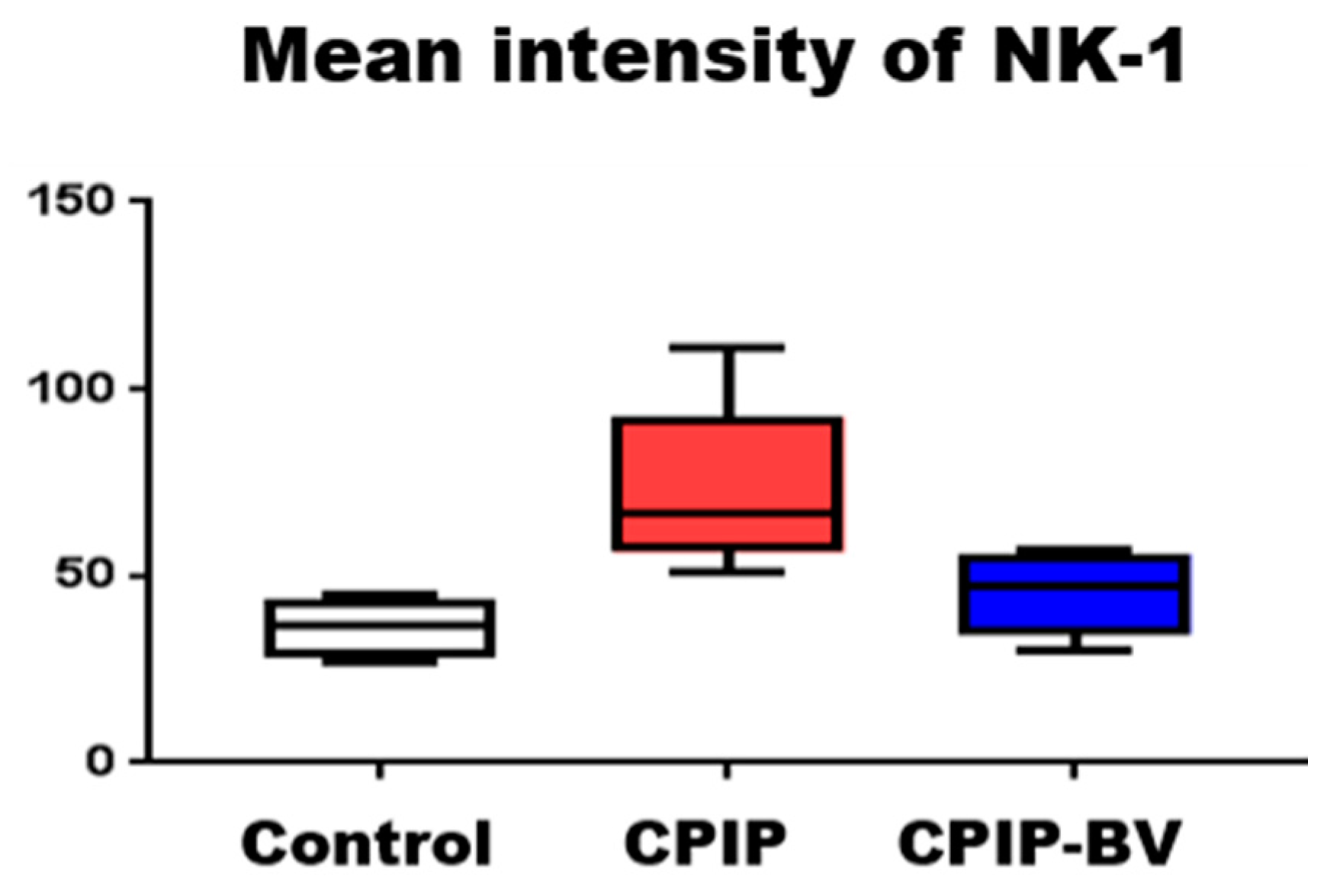
2.3. BV Attenuated the Increased Expression of NK-1 Receptors in CPIP Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. CPIP Model
4.3. Measurement of Tactile Allodynia
4.4. Drug Administration
4.5. Assessment of NK-1 Receptor Expression in DRG
4.6. Statistics
Author Contributions
Conflicts of Interest
References
- Son, D.J.; Lee, J.W.; Lee, Y.H.; Song, H.S.; Lee, C.K.; Hong, J.T. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol. Ther. 2007, 115, 246–270. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Choi, S.M.; Yang, E.J. Bee Venom Acupuncture Augments Anti-Inflammation in the Peripheral Organs of hSOD1G93A Transgenic Mice. Toxins 2015, 7, 2835–2844. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.D.; Kim, S.Y.; Kim, T.W.; Lee, S.H.; Yang, H.I.; Lee, D.I.; Lee, Y.H. Anti-inflammatory effect of bee venom on type II collagen-induced arthritis. Am. J. Chin. Med. 2004, 32, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lee, Y.; Kim, W.; Lee, K.; Bae, H.; Kim, S.K. Analgesic Effects of Bee Venom Derived Phospholipase A(2) in a Mouse Model of Oxaliplatin-Induced Neuropathic Pain. Toxins 2015, 7, 2422–2434. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Roh, D.H.; Yoon, S.Y.; Moon, J.Y.; Kim, H.W.; Lee, H.J.; Beitz, A.J.; Lee, J.H. Repetitive treatment with diluted bee venom reduces neuropathic pain via potentiation of locus coeruleus noradrenergic neuronal activity and modulation of spinal NR1 phosphorylation in rats. J. Pain 2012, 13, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Roh, D.H.; Park, J.H.; Lee, H.J.; Lee, J.H. Activation of Spinal alpha2-Adrenoceptors Using Diluted Bee Venom Stimulation Reduces Cold Allodynia in Neuropathic Pain Rats. Evid.-Based Complement. Altern. Med. 2012, 2012, 784713. [Google Scholar] [CrossRef] [PubMed]
- Iolascon, G.; de Sire, A.; Moretti, A.; Gimigliano, F. Complex regional pain syndrome (CRPS) type I: Historical perspective and critical issues. Clin. Cases Miner. Bone Metab. 2015, 12 (Suppl. 1), 4–10. [Google Scholar] [CrossRef] [PubMed]
- Coderre, T.J.; Bennett, G.J. A hypothesis for the cause of complex regional pain syndrome-type I (reflex sympathetic dystrophy): Pain due to deep-tissue microvascular pathology. Pain Med. 2010, 11, 1224–1238. [Google Scholar] [CrossRef] [PubMed]
- Nahm, F.S.; Park, Z.Y.; Nahm, S.S.; Kim, Y.C.; Lee, P.B. Proteomic identification of altered cerebral proteins in the complex regional pain syndrome animal model. BioMed Res. Int. 2014, 2014, 498410. [Google Scholar] [CrossRef] [PubMed]
- Kortekaas, M.C.; Niehof, S.P.; Stolker, R.J.; Huygen, F.J. Pathophysiological Mechanisms Involved in Vasomotor Disturbances in Complex Regional Pain Syndrome and Implications for Therapy: A Review. Pain Prac. 2015, 16, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Daehyun Jo, R.C.; Alan, R. Light: Glial Mechanisms of Neuropathic Pain and Emerging Interventions. Korean J. Pain 2009, 22, 1–15. [Google Scholar]
- Wei, T.; Li, W.W.; Guo, T.Z.; Zhao, R.; Wang, L.; Clark, D.J.; Oaklander, A.L.; Schmelz, M.; Kingery, W.S. Post-junctional facilitation of Substance P signaling in a tibia fracture rat model of complex regional pain syndrome type I. Pain 2009, 144, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Newby, D.E.; Sciberras, D.G.; Ferro, C.J.; Gertz, B.J.; Sommerville, D.; Majumdar, A.; Lowry, R.C.; Webb, D.J. Substance P-induced vasodilatation is mediated by the neurokinin type 1 receptor but does not contribute to basal vascular tone in man. Br. J. Clin. Pharmacol. 1999, 48, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.Z.; Wei, T.; Li, W.W.; Li, X.Q.; Clark, J.D.; Kingery, W.S. Immobilization contributes to exaggerated neuropeptide signaling, inflammatory changes, and nociceptive sensitization after fracture in rats. J. Pain Off. J. Am. Pain Soc. 2014, 15, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Marchand, J.E.; Wurm, W.H.; Kato, T.; Kream, R.M. Altered tachykinin expression by dorsal root ganglion neurons in a rat model of neuropathic pain. Pain 1994, 58, 219–231. [Google Scholar] [CrossRef]
- Millecamps, M.; Laferriere, A.; Ragavendran, J.V.; Stone, L.S.; Coderre, T.J. Role of peripheral endothelin receptors in an animal model of complex regional pain syndrome type 1 (CRPS-I). Pain 2010, 151, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Coderre, T.J.; Xanthos, D.N.; Francis, L.; Bennett, G.J. Chronic post-ischemia pain (CPIP): A novel animal model of complex regional pain syndrome-type I (CRPS-I; reflex sympathetic dystrophy) produced by prolonged hindpaw ischemia and reperfusion in the rat. Pain 2004, 112, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.Y.; Yeo, J.H.; Han, S.D.; Bong, D.J.; Oh, B.; Roh, D.H. Diluted bee venom injection reduces ipsilateral mechanical allodynia in oxaliplatin-induced neuropathic mice. Biol. Pharm. Bull. 2013, 36, 1787–1793. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.U.; Choi, S.S.; Lee, J.H.; Lee, S.H.; Lee, S.K.; Lee, Y.K.; Leem, J.G.; Song, J.G.; Shin, J.W. Perineural pretreatment of bee venom attenuated the development of allodynia in the spinal nerve ligation injured neuropathic pain model; an experimental study. BMC Complement. Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.B.; Kang, M.S.; Kim, H.W.; Ham, T.W.; Yim, Y.K.; Jeong, S.H.; Park, D.S.; Choi, D.Y.; Han, H.J.; Beitz, A.J.; et al. Antinociceptive effects of bee venom acupuncture (apipuncture) in rodent animal models: A comparative study of acupoint versus non-acupoint stimulation. Acupunct. Electrother. Res. 2001, 26, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Jang, M.; Choi, J.; Lee, G.; Min, H.J.; Chung, W.S.; Kim, J.I.; Jee, Y.; Chae, Y.; Kim, S.H.; et al. Bee Venom Acupuncture Alleviates Experimental Autoimmune Encephalomyelitis by Upregulating Regulatory T Cells and Suppressing Th1 and Th17 Responses. Mol. Neurobiol. 2016, 53, 1419–1445. [Google Scholar] [CrossRef] [PubMed]
- Leis, S.; Weber, M.; Isselmann, A.; Schmelz, M.; Birklein, F. Substance-P-induced protein extravasation is bilaterally increased in complex regional pain syndrome. Exp. Neurol. 2003, 183, 197–204. [Google Scholar] [CrossRef]
- Wei, T.; Guo, T.Z.; Li, W.W.; Kingery, W.S.; Clark, J.D. Acute versus chronic phase mechanisms in a rat model of CRPS. J. Neuroinflamm. 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Darwish, S.F.; El-Bakly, W.M.; Arafa, H.M.; El-Demerdash, E. Targeting TNF-alpha and NF-kappaB activation by bee venom: Role in suppressing adjuvant induced arthritis and methotrexate hepatotoxicity in rats. PLoS ONE 2013, 8, e79284. [Google Scholar] [CrossRef] [PubMed]
- Mashaghi, A.; Marmalidou, A.; Tehrani, M.; Grace, P.M.; Pothoulakis, C.; Dana, R. Neuropeptide substance P and the immune response. Cell. Mol. Life Sci. 2016, 73, 4249–4264. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, J.V.; Blum, A.; Metwali, A.; Elliott, D.; Arsenescu, R. IL-18 and IL-12 signal through the NF-kappa B pathway to induce NK-1R expression on T cells. J. Immunol. 2003, 170, 5003–5007. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Pittler, M.H.; Shin, B.C.; Kong, J.C.; Ernst, E. Bee venom acupuncture for musculoskeletal pain: A review. J. Pain 2008, 9, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; Lee, S.H. Effectiveness of bee venom acupuncture in alleviating post-stroke shoulder pain: A systematic review and meta-analysis. J. Integr. Med. 2015, 13, 241–247. [Google Scholar] [CrossRef]
- Jeon, Y. Cell based therapy for the management of chronic pain. Korean J. Anesthesiol. 2011, 60, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Bonin, R.P.; Bories, C.; De Koninck, Y. A simplified up-down method (SUDO) for measuring mechanical nociception in rodents using von Frey filaments. Mol. Pain 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1994, 53, 55–63. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.H.; Lee, J.M.; Kim, Y.H.; Choi, J.H.; Jeon, S.H.; Kim, D.K.; Jeong, H.D.; Lee, Y.J.; Park, H.J. Antiallodynic Effects of Bee Venom in an Animal Model of Complex Regional Pain Syndrome Type 1 (CRPS-I). Toxins 2017, 9, 285. https://doi.org/10.3390/toxins9090285
Lee SH, Lee JM, Kim YH, Choi JH, Jeon SH, Kim DK, Jeong HD, Lee YJ, Park HJ. Antiallodynic Effects of Bee Venom in an Animal Model of Complex Regional Pain Syndrome Type 1 (CRPS-I). Toxins. 2017; 9(9):285. https://doi.org/10.3390/toxins9090285
Chicago/Turabian StyleLee, Sung Hyun, Jae Min Lee, Yun Hong Kim, Jung Hyun Choi, Seung Hwan Jeon, Dong Kyu Kim, Hyeon Do Jeong, You Jung Lee, and Hue Jung Park. 2017. "Antiallodynic Effects of Bee Venom in an Animal Model of Complex Regional Pain Syndrome Type 1 (CRPS-I)" Toxins 9, no. 9: 285. https://doi.org/10.3390/toxins9090285
APA StyleLee, S. H., Lee, J. M., Kim, Y. H., Choi, J. H., Jeon, S. H., Kim, D. K., Jeong, H. D., Lee, Y. J., & Park, H. J. (2017). Antiallodynic Effects of Bee Venom in an Animal Model of Complex Regional Pain Syndrome Type 1 (CRPS-I). Toxins, 9(9), 285. https://doi.org/10.3390/toxins9090285




