ST-Producing E. coli Oppose Carcinogen-Induced Colorectal Tumorigenesis in Mice
Abstract
:1. Introduction
2. Methods and Materials
3. Results
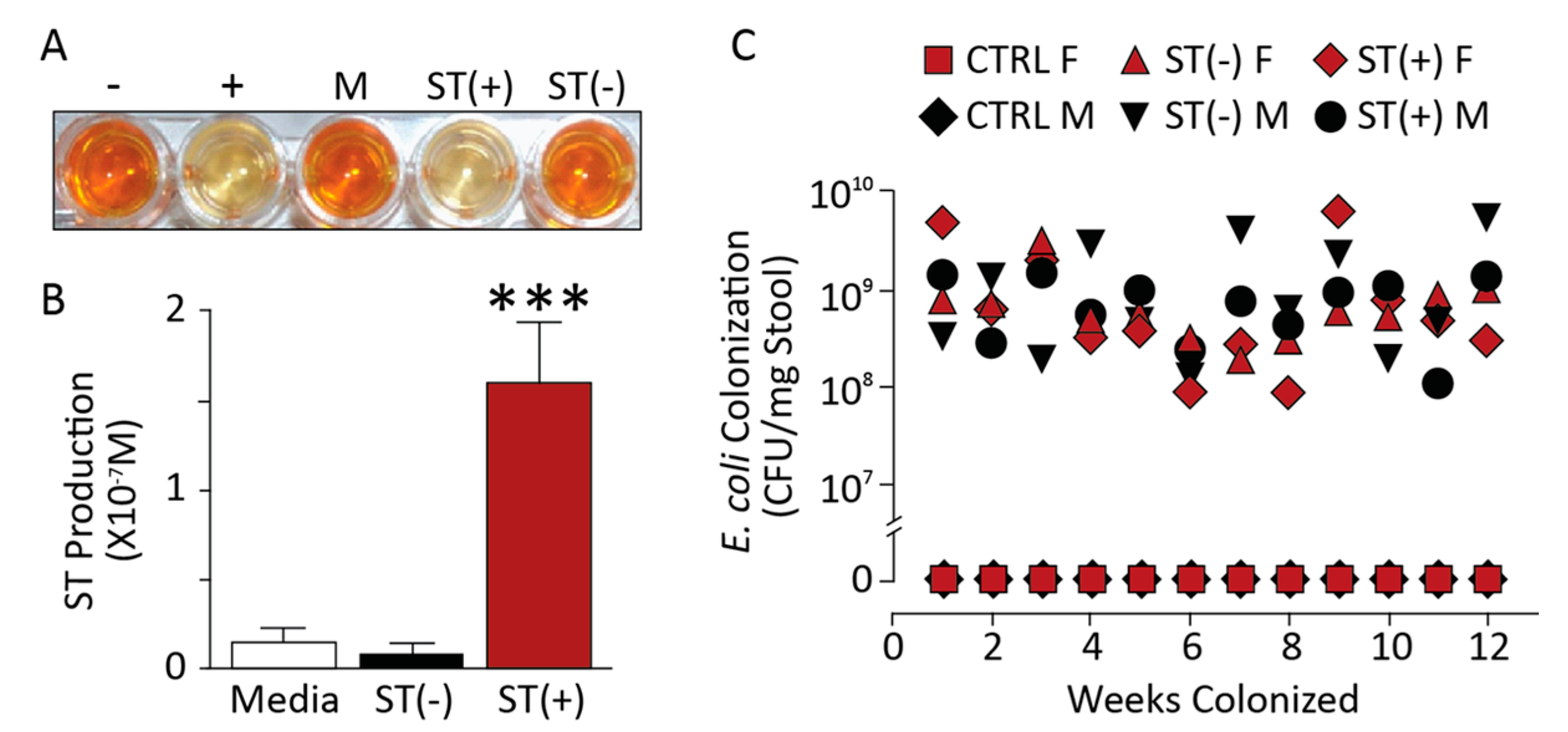
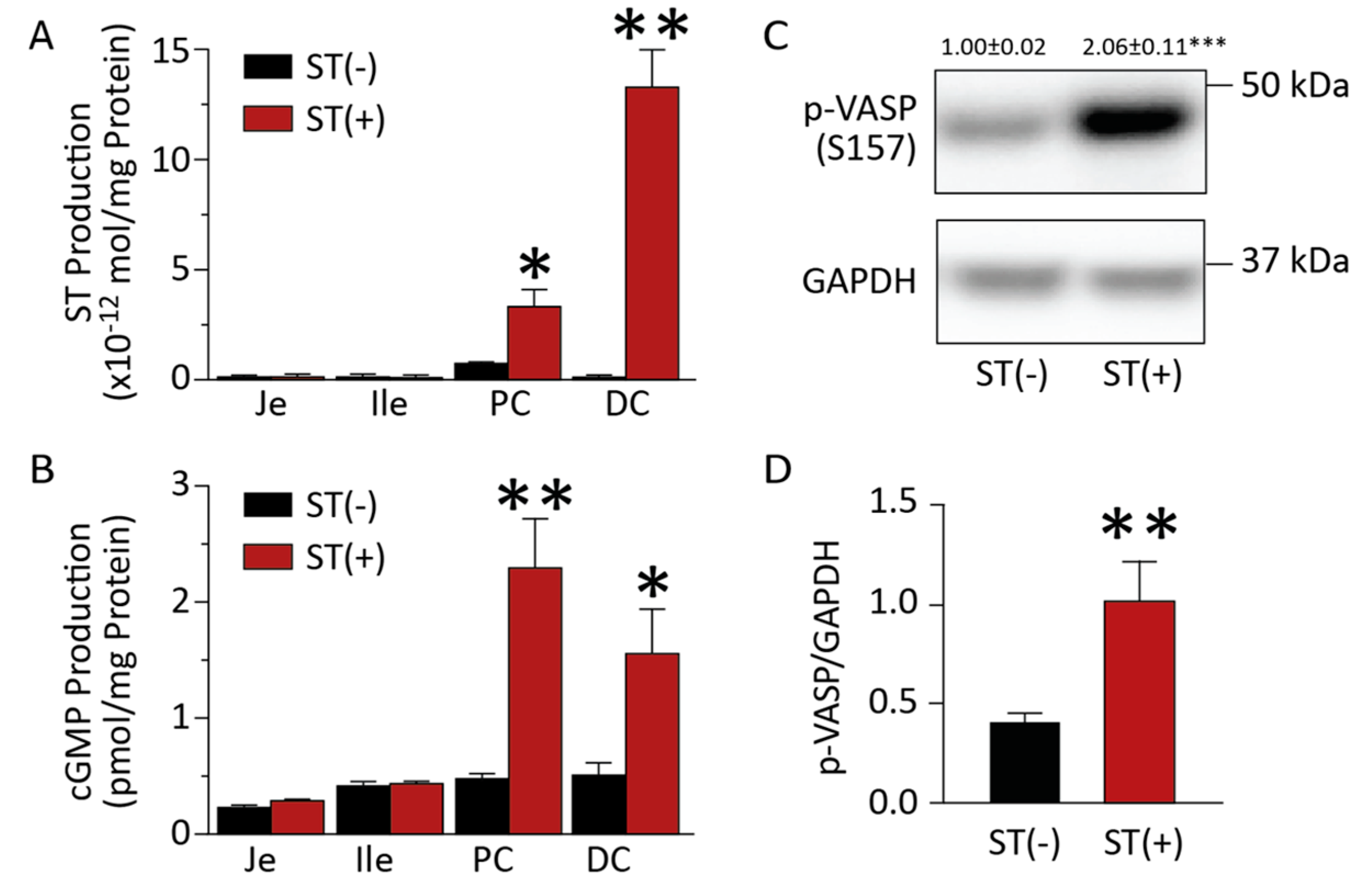
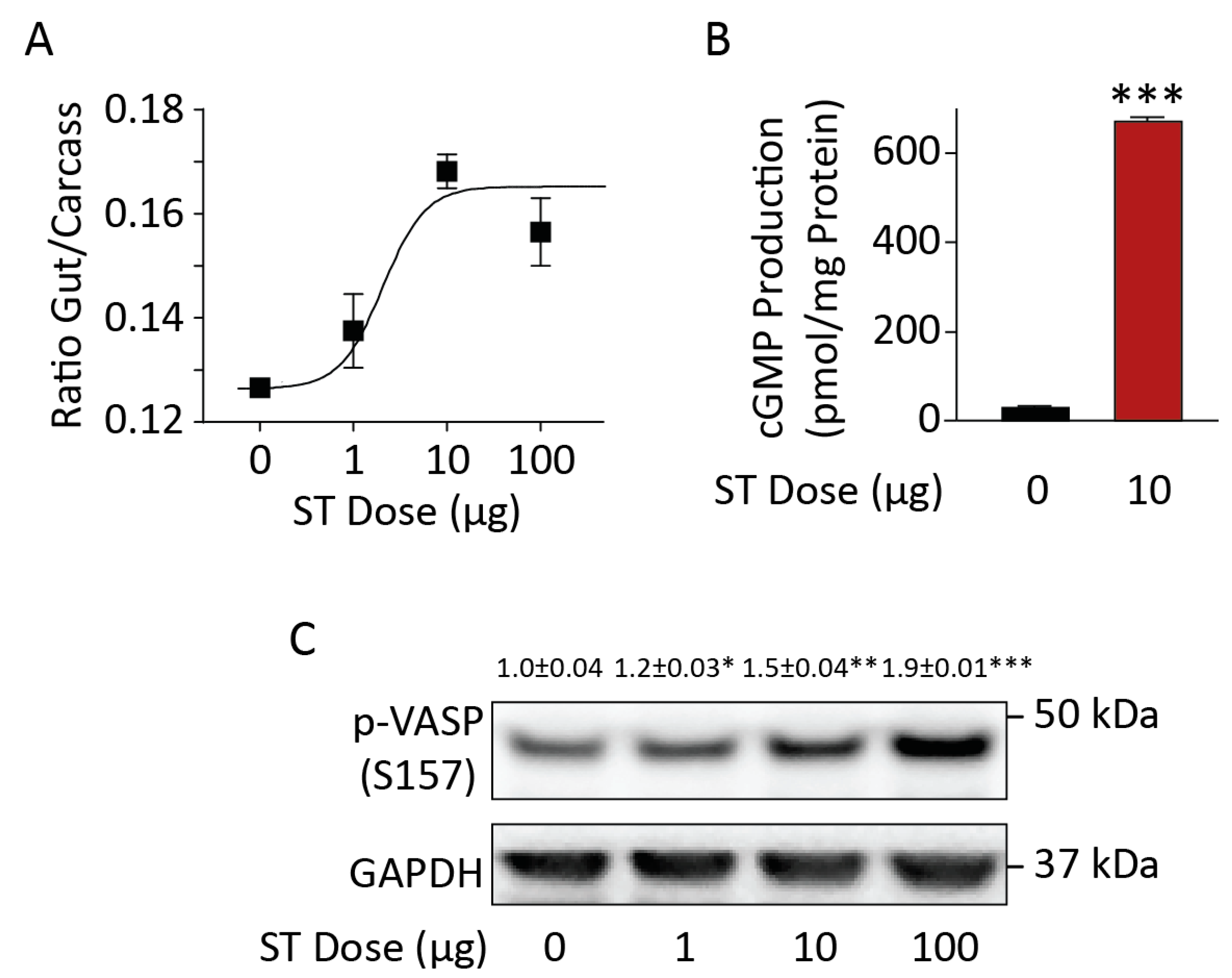
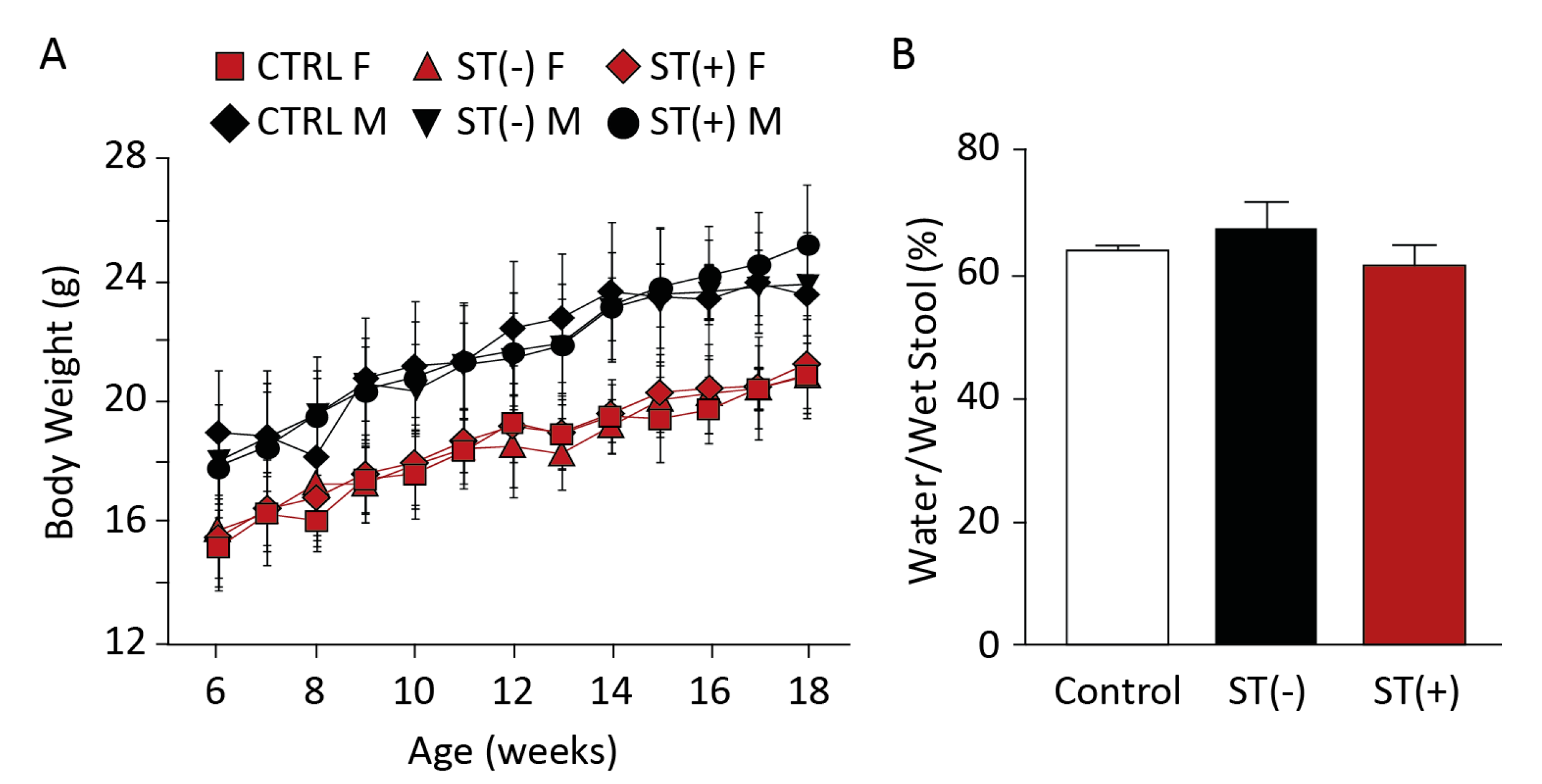
3.1. Chronic BL21 Colonization and ST Production
3.2. BL21(ST+) Colonization Opposes Carcinogen-Induced Colorectal Tumorigenesis
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AOM | azoxymethane |
| ETEC | enterotoxigenic E. coli |
| STs | heat-stable enterotoxins |
| LT | heat-liable enterotoxin |
| GUCY2C | guanylyl cyclase C |
| cGMP | cyclic GMP |
| PKG | cGMP-dependent protein kinase |
| CFTR | cystic fibrosis transmembrane conductance regulator |
| EIA | enzyme immunoassay |
| IACUC | institutional animal care and use committee |
| LB broth | Luria-Bertani broth |
References
- Bolin, I.; Wiklund, G.; Qadri, F.; Torres, O.; Bourgeois, A.L.; Savarino, S.; Svennerholm, A.M. Enterotoxigenic escherichia coli with sth and stp genotypes is associated with diarrhea both in children in areas of endemicity and in travelers. J. Clin. Microbiol. 2006, 44, 3872–3877. [Google Scholar] [CrossRef] [PubMed]
- Donowitz, M.; Alpers, D.H.; Binder, H.J.; Brewer, T.; Carrington, J.; Grey, M.J. Translational approaches for pharmacotherapy development for acute diarrhea. Gastroenterology 2012, 142, e1–e9. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diarrhoeal Disease Fact Sheet, 2009. World Health Organization Media Centre. Available online: http://www.who.int/mediacentre/factsheets/fs330/en/ (accessed on 7 September 2017).
- Kuhn, M. Molecular physiology of membrane guanylyl cyclase receptors. Physiol. Rev. 2016, 96, 751–804. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A. Clinical practice. Irritable bowel syndrome. N. Engl. J. Med. 2008, 358, 1692–1699. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhou, W.; Velculescu, V.E.; Kern, S.E.; Hruban, R.H.; Hamilton, S.R.; Vogelstein, B.; Kinzler, K.W. Gene expression profiles in normal and cancer cells. Science 1997, 276, 1268–1272. [Google Scholar] [CrossRef] [PubMed]
- Steinbrecher, K.A.; Tuohy, T.M.; Heppner Goss, K.; Scott, M.C.; Witte, D.P.; Groden, J.; Cohen, M.B. Expression of guanylin is downregulated in mouse and human intestinal adenomas. Biochem. Biophys. Res. Commun. 2000, 273, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Notterman, D.A.; Alon, U.; Sierk, A.J.; Levine, A.J. Transcriptional gene expression profiles of colorectal adenoma, adenocarcinoma, and normal tissue examined by oligonucleotide arrays. Cancer Res. 2001, 61, 3124–3130. [Google Scholar] [PubMed]
- Shailubhai, K.; Yu, H.H.; Karunanandaa, K.; Wang, J.Y.; Eber, S.L.; Wang, Y.; Joo, N.S.; Kim, H.D.; Miedema, B.W.; Abbas, S.Z.; et al. Uroguanylin treatment suppresses polyp formation in the apc(min/+) mouse and induces apoptosis in human colon adenocarcinoma cells via cyclic gmp. Cancer Res. 2000, 60, 5151–5157. [Google Scholar] [PubMed]
- Li, P.; Schulz, S.; Bombonati, A.; Palazzo, J.P.; Hyslop, T.M.; Xu, Y.; Barab, A.A.; Siracusa, L.D.; Pitari, G.M.; Waldman, S.A. Guanylyl cyclase c suppresses intestinal tumorigenesis by restricting proliferation and maintaining genomic integrity. Gastroenterology 2007, 133, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.E.; Colon-Gonzalez, F.; Blomain, E.; Kim, G.W.; Aing, A.; Stoecker, B.; Rock, J.; Snook, A.E.; Zhan, T.; Hyslop, T.M.; et al. Obesity-induced colorectal cancer is driven by caloric silencing of the guanylin-gucy2c paracrine signaling axis. Cancer Res. 2016, 76, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.E.; Li, P.; Snook, A.E.; Schulz, S.; Dasgupta, A.; Hyslop, T.M.; Gibbons, A.V.; Marszlowicz, G.; Pitari, G.M.; Waldman, S.A. The hormone receptor gucy2c suppresses intestinal tumor formation by inhibiting akt signaling. Gastroenterology 2010, 138, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Pitari, G.M.; Di Guglielmo, M.D.; Park, J.; Schulz, S.; Waldman, S.A. Guanylyl cyclase c agonists regulate progression through the cell cycle of human colon carcinoma cells. Proc. Natl. Acad. Sci. USA 2001, 98, 7846–7851. [Google Scholar] [CrossRef] [PubMed]
- Pitari, G.M.; Zingman, L.V.; Hodgson, D.M.; Alekseev, A.E.; Kazerounian, S.; Bienengraeber, M.; Hajnoczky, G.; Terzic, A.; Waldman, S.A. Bacterial enterotoxins are associated with resistance to colon cancer. Proc. Natl. Acad. Sci. USA 2003, 100, 2695–2699. [Google Scholar] [CrossRef] [PubMed]
- Blomain, E.; Pattison, A.; Waldman, S.A. Gucy2c ligand replacement to prevent colorectal cancer. Cancer Biol. Ther. 2016, 17, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, D.S.; Lin, J.E.; Foster, N.R.; Della’Zanna, G.; Umar, A.; Seisler, D.; Kraft, W.K.; Kastenberg, D.M.; Katz, L.C.; Limburg, P.J.; et al. Bioactivity of oral linaclotide in human colorectum for cancer chemoprevention. Cancer Prev. Res. 2017, 10, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M.; Whelan, S.L.; Ferlay, J.; Teppo, L.; Thomas, D.B. Cancer Incidence in Five Continents; IARC Scientific Publication: Lyon, France, 2002; Volume IX[M]. [Google Scholar]
- Bray, F.; Ferlay, J.; Laversanne, M.; Brewster, D.H.; Gombe Mbalawa, C.; Kohler, B.; Pineros, M.; Steliarova-Foucher, E.; Swaminathan, R.; Antoni, S.; et al. Cancer incidence in five continents: Inclusion criteria, highlights from volume x and the global status of cancer registration. Int. J. Cancer 2015, 137, 2060–2071. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Haggar, F.A.; Boushey, R.P. Colorectal cancer epidemiology: Incidence, mortality, survival, and risk factors. Clin. Colon Rectal Surg. 2009, 22, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Fekete, P.Z.; Brzuszkiewicz, E.; Blum-Oehler, G.; Olasz, F.; Szabo, M.; Gottschalk, G.; Hacker, J.; Nagy, B. DNA sequence analysis of the composite plasmid ptc conferring virulence and antimicrobial resistance for porcine enterotoxigenic escherichia coli. Int. J. Med. Microbiol. 2012, 302, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Rhee, K.J.; Albesiano, E.; Rabizadeh, S.; Wu, X.; Yen, H.R.; Huso, D.L.; Brancati, F.L.; Wick, E.; McAllister, F.; et al. A human colonic commensal promotes colon tumorigenesis via activation of t helper type 17 t cell responses. Nat. Med. 2009, 15, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Schulz, S.; Lopez, M.J.; Kuhn, M.; Garbers, D.L. Disruption of the guanylyl cyclase-c gene leads to a paradoxical phenotype of viable but heat-stable enterotoxin-resistant mice. J. Clin. Investig. 1997, 100, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Waldman, S.A.; Barber, M.; Pearlman, J.; Park, J.; George, R.; Parkinson, S.J. Heterogeneity of guanylyl cyclase c expressed by human colorectal cancer cell lines in vitro. Cancer Epidemiol. Biomark. Prev. 1998, 7, 505–514. [Google Scholar]
- Li, P.; Lin, J.E.; Chervoneva, I.; Schulz, S.; Waldman, S.A.; Pitari, G.M. Homeostatic control of the crypt-villus axis by the bacterial enterotoxin receptor guanylyl cyclase c restricts the proliferating compartment in intestine. Am. J. Pathol. 2007, 171, 1847–1858. [Google Scholar] [CrossRef] [PubMed]
- Zuzga, D.S.; Pelta-Heller, J.; Li, P.; Bombonati, A.; Waldman, S.A.; Pitari, G.M. Phosphorylation of vasodilator-stimulated phosphoprotein ser239 suppresses filopodia and invadopodia in colon cancer. Int. J. Cancer 2012, 130, 2539–2548. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Future directions for research on enterotoxigenic escherichia coli vaccines for developing countries. Wkly. Epidemiol. Rec. 2006, 81, 97–104. [Google Scholar]
- Huilan, S.; Zhen, L.G.; Mathan, M.M.; Mathew, M.M.; Olarte, J.; Espejo, R.; Khin Maung, U.; Ghafoor, M.A.; Khan, M.A.; Sami, Z.; et al. Etiology of acute diarrhoea among children in developing countries: A multicentre study in five countries. Bull. World Health Organ. 1991, 69, 549–555. [Google Scholar] [PubMed]
- Kotloff, K.L. The burden and etiology of diarrheal illness in developing countries. Pediatr. Clin. N. Am. 2017, 64, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Boyle, P.; Ferlay, J. Mortality and survival in breast and colorectal cancer. Nat. Clin. Pract. Oncol. 2005, 2, 424–425. [Google Scholar] [CrossRef] [PubMed]
- Janout, V.; Kollarova, H. Epidemiology of colorectal cancer. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czechoslov. 2001, 145, 5–10. [Google Scholar] [CrossRef]
- Wilmink, A.B. Overview of the epidemiology of colorectal cancer. Dis. Colon Rectum 1997, 40, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Schulz, S.; Green, C.K.; Yuen, P.S.; Garbers, D.L. Guanylyl cyclase is a heat-stable enterotoxin receptor. Cell 1990, 63, 941–948. [Google Scholar] [CrossRef]
- Currie, M.G.; Fok, K.F.; Kato, J.; Moore, R.J.; Hamra, F.K.; Duffin, K.L.; Smith, C.E. Guanylin: An endogenous activator of intestinal guanylate cyclase. Proc. Natl. Acad. Sci. USA 1992, 89, 947–951. [Google Scholar] [CrossRef] [PubMed]
- Hamra, F.K.; Forte, L.R.; Eber, S.L.; Pidhorodeckyj, N.V.; Krause, W.J.; Freeman, R.H.; Chin, D.T.; Tompkins, J.A.; Fok, K.F.; Smith, C.E.; et al. Uroguanylin: Structure and activity of a second endogenous peptide that stimulates intestinal guanylate cyclase. Proc. Natl. Acad. Sci. USA 1993, 90, 10464–10468. [Google Scholar] [CrossRef] [PubMed]
- Kita, T.; Smith, C.E.; Fok, K.F.; Duffin, K.L.; Moore, W.M.; Karabatsos, P.J.; Kachur, J.F.; Hamra, F.K.; Pidhorodeckyj, N.V.; Forte, L.R.; et al. Characterization of human uroguanylin: A member of the guanylin peptide family. Am. J. Physiol. 1994, 266, F342–F348. [Google Scholar] [PubMed]
- Cuthbert, A.W.; Hickman, M.E.; MacVinish, L.J.; Evans, M.J.; Colledge, W.H.; Ratcliff, R.; Seale, P.W.; Humphrey, P.P. Chloride secretion in response to guanylin in colonic epithelial from normal and transgenic cystic fibrosis mice. Br. J. Pharmacol. 1994, 112, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Field, M.; Graf, L.H., Jr.; Laird, W.J.; Smith, P.L. Heat-stable enterotoxin of escherichia coli: In vitro effects on guanylate cyclase activity, cyclic gmp concentration, and ion transport in small intestine. Proc. Natl. Acad. Sci. USA 1978, 75, 2800–2804. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.M.; Murad, F.; Chang, B.; Guerrant, R.L. Role of cyclic gmp in the action of heat-stable enterotoxin of escherichia coli. Nature 1978, 271, 755–756. [Google Scholar] [CrossRef] [PubMed]
- Date, Y.; Nakazato, M.; Yamaguchi, H.; Miyazato, M.; Matsukura, S. Tissue distribution and plasma concentration of human guanylin. Int. Med. 1996, 35, 171–175. [Google Scholar] [CrossRef]
- Birkenkamp-Demtroder, K.; Lotte Christensen, L.; Harder Olesen, S.; Frederiksen, C.M.; Laiho, P.; Aaltonen, L.A.; Laurberg, S.; Sorensen, F.B.; Hagemann, R.; Orntoft, T.F. Gene expression in colorectal cancer. Cancer Res. 2002, 62, 4352–4363. [Google Scholar] [PubMed]
- Cohen, M.B.; Hawkins, J.A.; Witte, D.P. Guanylin mrna expression in human intestine and colorectal adenocarcinoma. Lab. Investig. 1998, 78, 101–108. [Google Scholar] [PubMed]
- Camilleri, M. Guanylate cyclase c agonists: Emerging gastrointestinal therapies and actions. Gastroenterology 2015, 148, 483–487. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, P.; Lin, J.E.; Snook, A.E.; Waldman, S.A. ST-Producing E. coli Oppose Carcinogen-Induced Colorectal Tumorigenesis in Mice. Toxins 2017, 9, 279. https://doi.org/10.3390/toxins9090279
Li P, Lin JE, Snook AE, Waldman SA. ST-Producing E. coli Oppose Carcinogen-Induced Colorectal Tumorigenesis in Mice. Toxins. 2017; 9(9):279. https://doi.org/10.3390/toxins9090279
Chicago/Turabian StyleLi, Peng, Jieru E. Lin, Adam E. Snook, and Scott A. Waldman. 2017. "ST-Producing E. coli Oppose Carcinogen-Induced Colorectal Tumorigenesis in Mice" Toxins 9, no. 9: 279. https://doi.org/10.3390/toxins9090279
APA StyleLi, P., Lin, J. E., Snook, A. E., & Waldman, S. A. (2017). ST-Producing E. coli Oppose Carcinogen-Induced Colorectal Tumorigenesis in Mice. Toxins, 9(9), 279. https://doi.org/10.3390/toxins9090279







