Recombinant Phospholipase D from Loxosceles gaucho Binds to Platelets and Promotes Phosphatidylserine Exposure
Abstract
:1. Introduction
2. Results
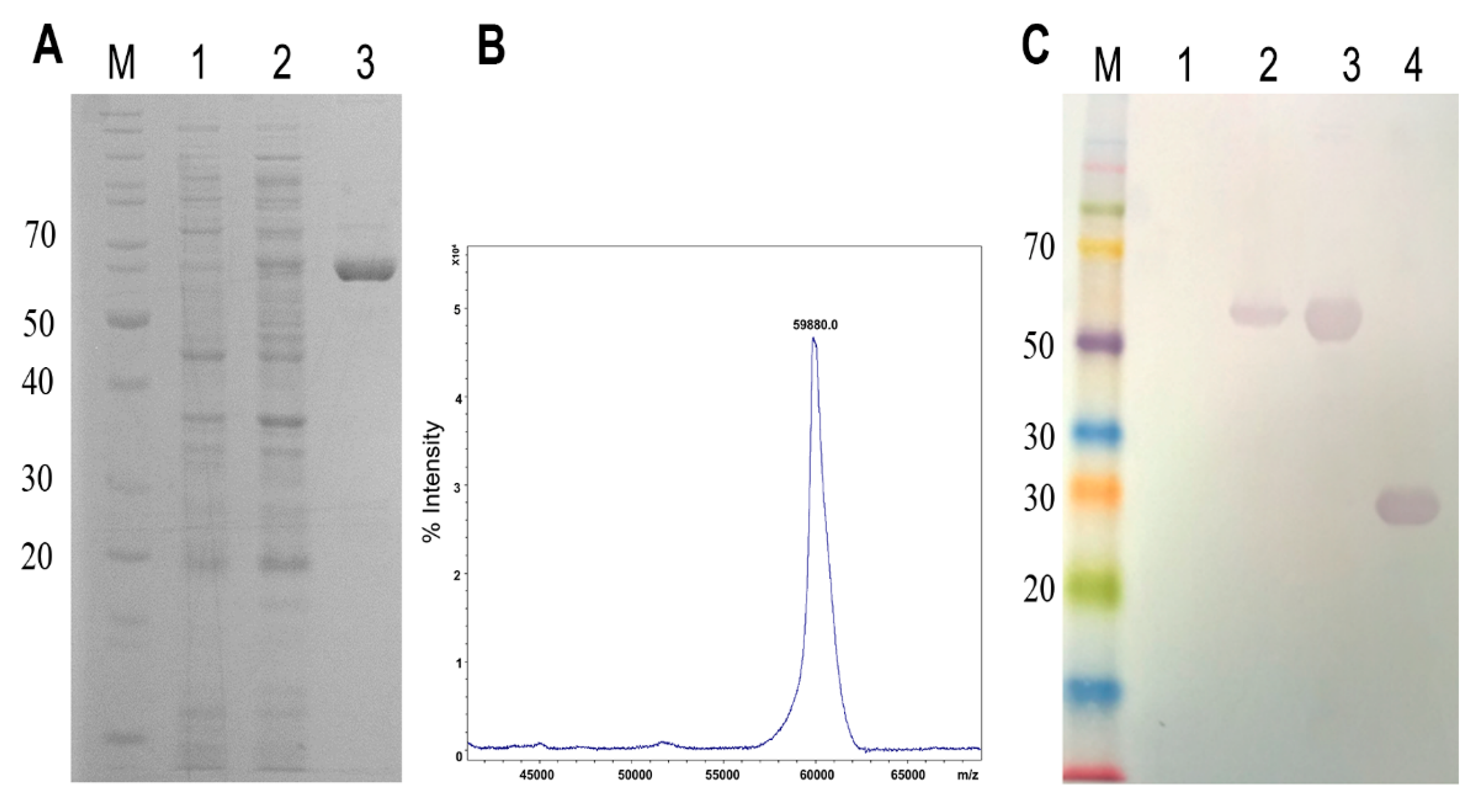
2.1. Cloning, Expression, and Purification of Phospholipase D (LgRec1) Fused to EGFP
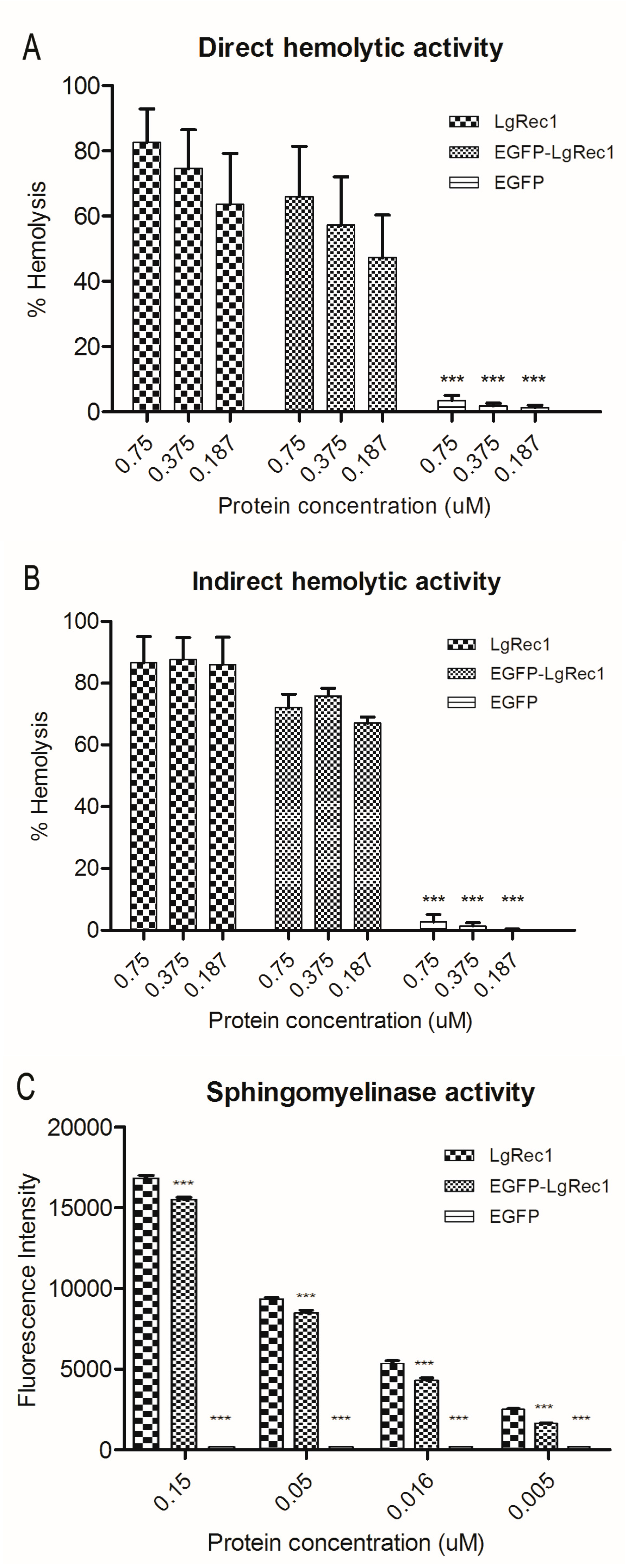
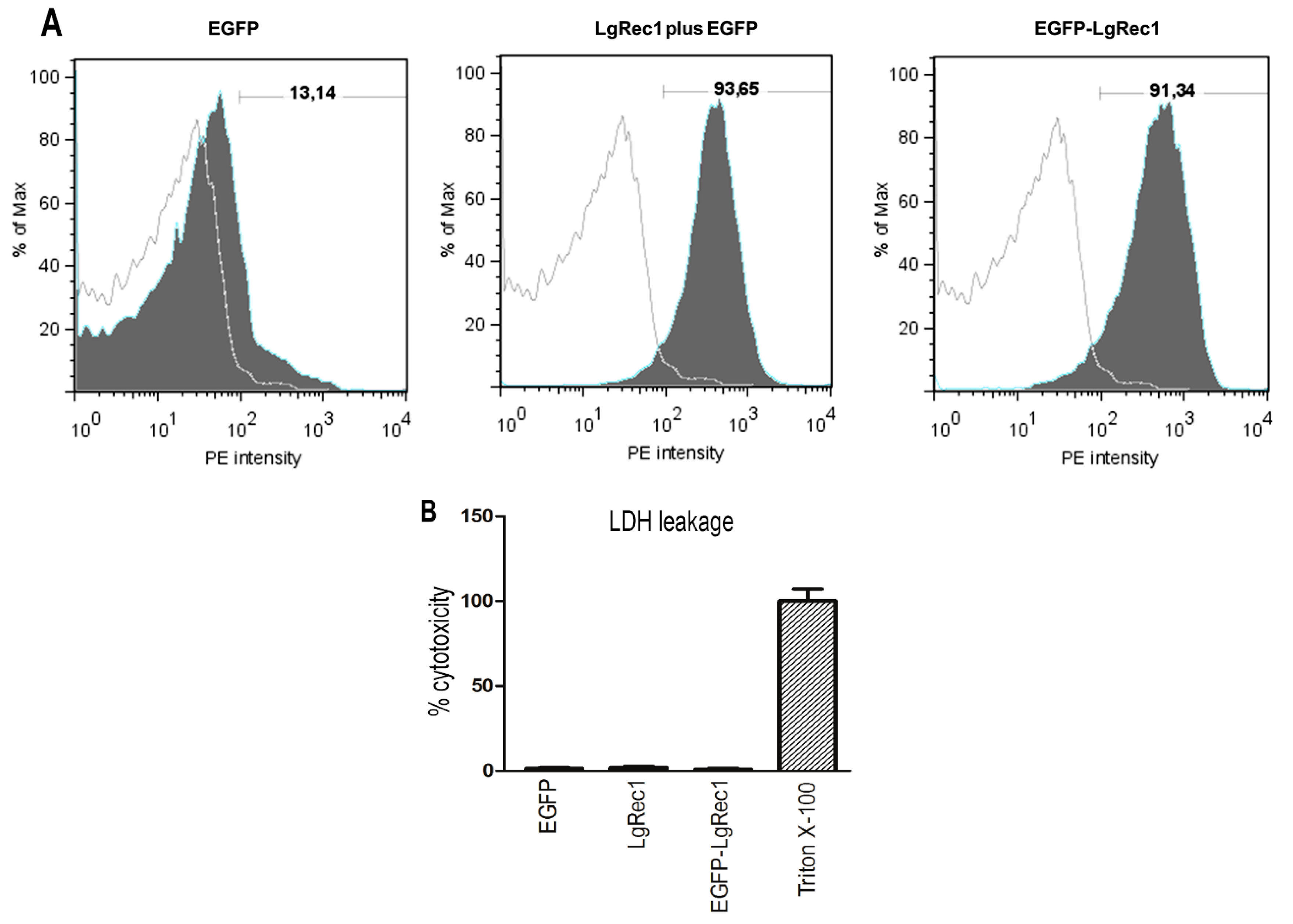
2.2. Biological Characterization of the Chimeric Toxin EGFP-LgRec1
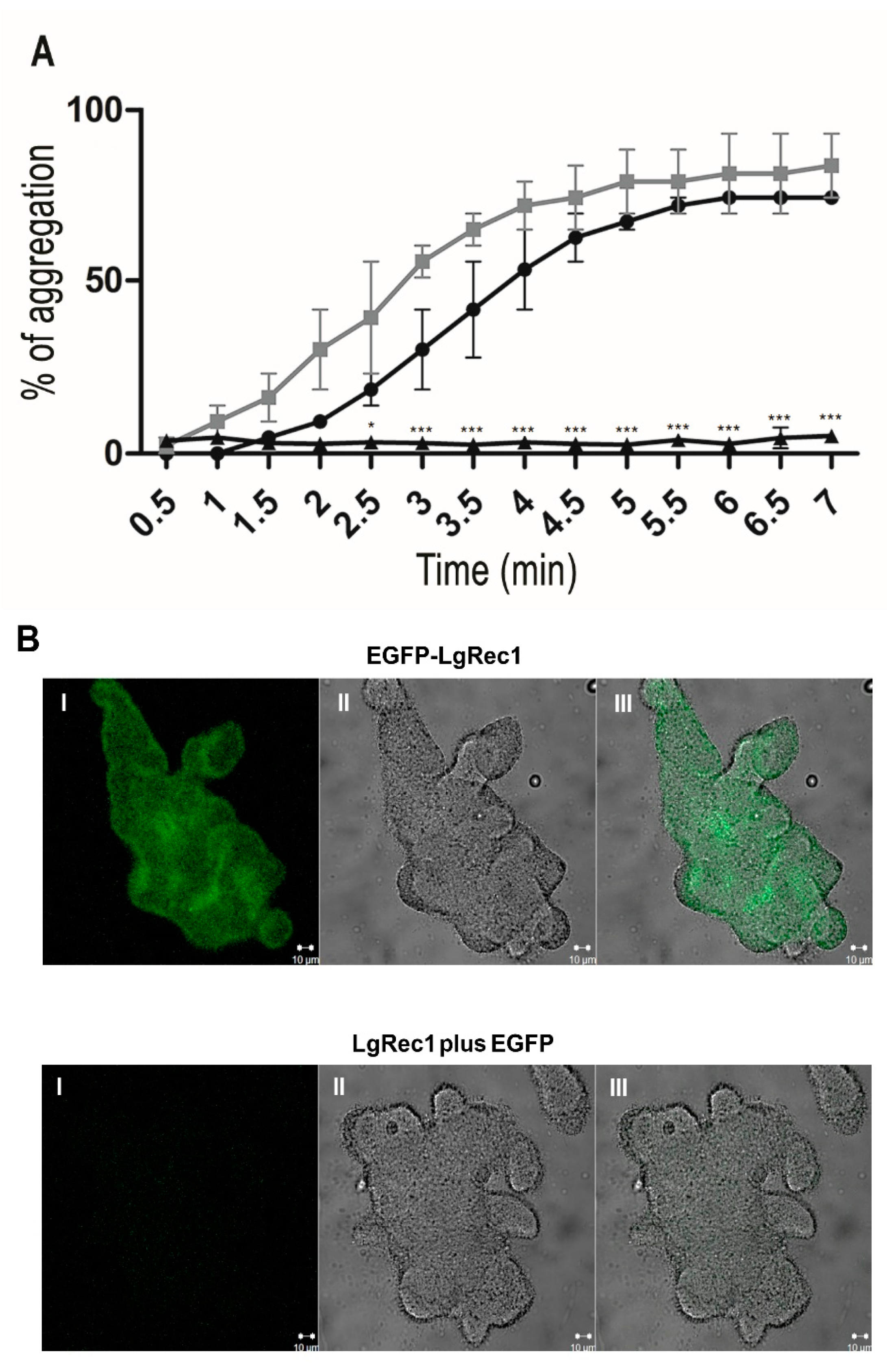
2.3. EGFP-LgRec1 Promotes Platelet Aggregation and Binding in Human Platelet-Rich Plasma
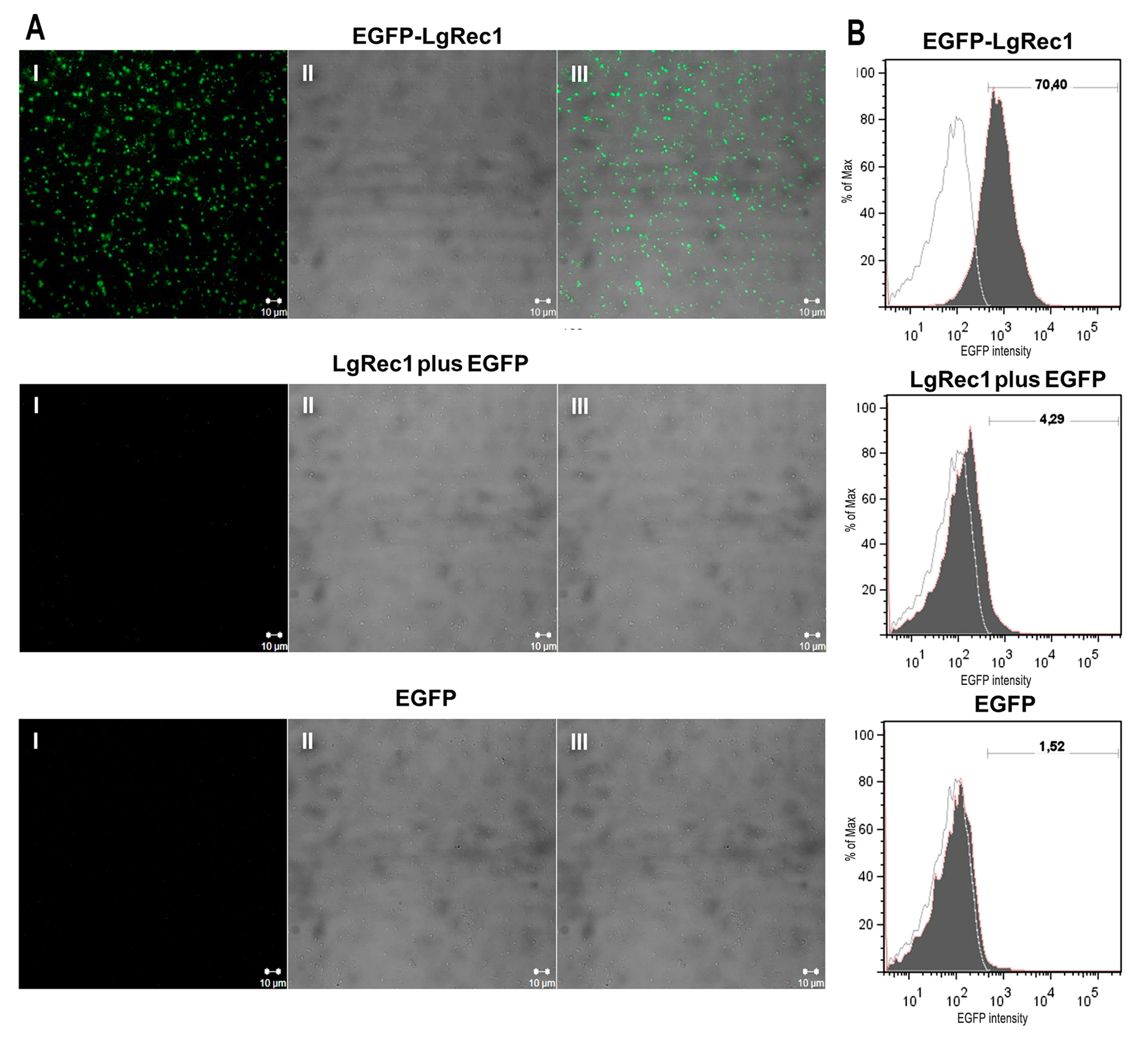
2.4. EGFP-LgRec1 Binds to Platelets in the Absence of Plasma Components
2.5. EGFP-LgRec1 Induce Exposure of Annexin-V Binding Sites in Platelets
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Ethics
5.2. Cloning, Expression, and Purification of EGFP-LgRec1
5.3. Determination of Molecular Mass by Mass Spectrometry
5.4. SDS-Polyacrylamide Gel Electrophoresis and Western Blot Analysis
5.5. Sphingomyelinase Assay
5.6. Blood Cells and Serum
5.7. Platelet Aggregation Assay
5.8. Determination of Hemolytic Activity
5.9. Confocal Immunofluorescence
5.10. Flow Cytometry
5.11. Phosphatidylserine Exposure
5.12. Lactate Dehydrogenase Leakage
5.13. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pezzi, M.; Giglio, A.M.; Scozzafava, A.; Filippelli, O.; Serafino, G.; Verre, M. Spider bite: A rare case of acute necrotic arachnidism with rapid and fatal evolution. Case Rep. Emerg. Med. 2016, 2016, 7640789. [Google Scholar] [CrossRef] [PubMed]
- Malaque, C.M.; Santoro, M.L.; Cardoso, J.L.; Conde, M.R.; Novaes, C.T.; Risk, J.Y.; França, F.O.; de Medeiros, C.R.; Fan, H.W. Clinical picture and laboratorial evaluation in human loxoscelism. Toxicon 2011, 58, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, F.A.; Amorim, F.G.; Anjolette, F.A.; Arantes, E.C. Arachnids of medical importance in Brazil: Main active compounds present in scorpion and spider venoms and tick saliva. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 24. [Google Scholar] [CrossRef] [PubMed]
- Saúde, M. Sistema de Informação Agravos de Notificação; Sinan: Brasilia, Brazil, 2016. [Google Scholar]
- Veiga, S.S.; da Silveira, R.B.; Dreyfus, J.L.; Haoach, J.; Pereira, A.M.; Mangili, O.C.; Gremski, W. Identification of high molecular weight serine-proteases in Loxosceles intermedia (brown spider) venom. Toxicon 2000, 38, 825–839. [Google Scholar] [CrossRef]
- Young, A.R.; Pincus, S.J. Comparison of enzymatic activity from three species of necrotising arachnids in Australia: Loxosceles rufescens, Badumna insignis and Lampona cylindrata. Toxicon 2001, 39, 391–400. [Google Scholar] [CrossRef]
- Da Silveira, R.B.; Chaim, O.M.; Mangili, O.C.; Gremski, W.; Dietrich, C.P.; Nader, H.B.; Veiga, S.S. Hyaluronidases in Loxosceles intermedia (brown spider) venom are endo-beta-N-acetyl-d-hexosaminidases hydrolases. Toxicon 2007, 49, 758–768. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.S.; Silvestre, F.G.; Araújo, S.C.; Gabriel, D.M.; Mangili, O.C.; Cruz, I.; Chávez-Olórtegui, C.; Kalapothakis, E. Identification and molecular cloning of insecticidal toxins from the venom of the brown spider Loxosceles intermedia. Toxicon 2004, 44, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, L.; Gremski, W.; Veiga, S.S.; Elias, M.C.; Graner, E.; Mangili, O.C.; Brentani, R.R. Detection and characterization of metalloproteinases with gelatinolytic, fibronectinolytic and fibrinogenolytic activities in brown spider (Loxosceles intermedia) venom. Toxicon 1998, 36, 1039–1051. [Google Scholar] [CrossRef]
- Tambourgi, D.; Magnoli, F.; van den Berg, C.; Morgan, B.; de Araujo, P.; Alves, E.; Da Silva, W. Sphingomyelinases in the venom of the spider Loxosceles intermedia are responsible for both dermonecrosis and complement-dependent hemolysis. Biochem. Biophys. Res. Commun. 1998, 251, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Cunha, R.B.; Barbaro, K.C.; Muramatsu, D.; Portaro, F.C.; Fontes, W.; de Sousa, M.V. Purification and characterization of loxnecrogin, a dermonecrotic toxin from Loxosceles gaucho brown spider venom. J. Protein Chem. 2003, 22, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, K.C.; Knysak, I.; Martins, R.; Hogan, C.; Winkel, K. Enzymatic characterization, antigenic cross-reactivity and neutralization of dermonecrotic activity of five loxosceles spider venoms of medical importance in the americas. Toxicon 2005, 45, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Gremski, L.H.; da Silveira, R.B.; Chaim, O.M.; Probst, C.M.; Ferrer, V.P.; Nowatzki, J.; Weinschutz, H.C.; Madeira, H.M.; Gremski, W.; Nader, H.B.; et al. A novel expression profile of the Loxosceles intermedia spider venomous gland revealed by transcriptome analysis. Mol. Biosyst. 2010, 6, 2403–2416. [Google Scholar] [CrossRef] [PubMed]
- Fernandes-Pedrosa, M.F.; Junqueira-de-Azevedo, I.L.; Gonçalves-de-Andrade, R.M.; Kobashi, L.S.; Almeida, D.D.; Ho, P.L.; Tambourgi, D.V. Transcriptome analysis of loxosceles laeta (araneae, sicariidae) spider venomous gland using expressed sequence tags. BMC Genom. 2008, 9, 279. [Google Scholar] [CrossRef] [PubMed]
- Paludo, K.S.; Biscaia, S.M.; Chaim, O.M.; Otuki, M.F.; Naliwaiko, K.; Dombrowski, P.A.; Franco, C.R.; Veiga, S.S. Inflammatory events induced by brown spider venom and its recombinant dermonecrotic toxin: A pharmacological investigation. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 149, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Kusma, J.; Chaim, O.M.; Wille, A.C.; Ferrer, V.P.; Sade, Y.B.; Donatti, L.; Gremski, W.; Mangili, O.C.; Veiga, S.S. Nephrotoxicity caused by brown spider venom phospholipase-D (dermonecrotic toxin) depends on catalytic activity. Biochimie 2008, 90, 1722–1736. [Google Scholar] [CrossRef] [PubMed]
- Tambourgi, D.; Paixao-Cavalcante, D.; de Andrade, R.; Fernandes-Pedrosa, M.; Magnoli, F.; Morgan, B.; van den Berg, C. Loxosceles sphingomyelinase induces complement-dependent dermonecrosis, neutrophil infiltration, and endogenous gelatinase expression. J. Investig. Dermatol. 2005, 124, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Chaim, O.; da Silveira, R.; Trevisan-Silva, D.; Ferrer, V.; Sade, Y.; Boia-Ferreira, M.; Gremski, L.; Gremski, W.; Senff-Ribeiro, A.; Takahashi, H.; et al. Phospholipase-D activity and inflammatory response induced by brown spider dermonecrotic toxin: Endothelial cell membrane phospholipids as targets for toxicity. Biochim. Biophys. Acta 2011, 1811, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Tambourgi, D.V.; Morgan, B.P.; de Andrade, R.M.; Magnoli, F.C.; van Den Berg, C.W. Loxosceles intermedia spider envenomation induces activation of an endogenous metalloproteinase, resulting in cleavage of glycophorins from the erythrocyte surface and facilitating complement-mediated lysis. Blood 2000, 95, 683–691. [Google Scholar] [PubMed]
- Chaves-Moreira, D.; Chaim, O.M.; Sade, Y.B.; Paludo, K.S.; Gremski, L.H.; Donatti, L.; de Moura, J.; Mangili, O.C.; Gremski, W.; da Silveira, R.B.; et al. Identification of a direct hemolytic effect dependent on the catalytic activity induced by phospholipase-D (dermonecrotic toxin) from brown spider venom. J. Cell. Biochem. 2009, 107, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Fernandes Pedrosa, M.F.; Junqueira de Azevedo, I.L.; Gonçalves-de-Andrade, R.M.; van den Berg, C.W.; Ramos, C.R.; Ho, P.L.; Tambourgi, D.V. Molecular cloning and expression of a functional dermonecrotic and haemolytic factor from Loxosceles laeta venom. Biochem. Biophys. Res. Commun. 2002, 298, 638–645. [Google Scholar] [CrossRef]
- Kurpiewski, G.; Forrester, L.J.; Barrett, J.T.; Campbell, B.J. Platelet aggregation and sphingomyelinase D activity of a purified toxin from the venom of Loxosceles reclusa. Biochim. Biophys. Acta 1981, 678, 467–476. [Google Scholar] [CrossRef]
- Gates, C.A.; Rees, R.S. Serum amyloid P component: Its role in platelet activation stimulated by sphingomyelinase D purified from the venom of the brown recluse spider (Loxosceles reclusa). Toxicon 1990, 28, 1303–1315. [Google Scholar] [CrossRef]
- Da Silveira, R.B.; Pigozzo, R.B.; Chaim, O.M.; Appel, M.H.; Silva, D.T.; Dreyfuss, J.L.; Toma, L.; Dietrich, C.P.; Nader, H.B.; Veiga, S.S.; et al. Two novel dermonecrotic toxins LiRecDT4 and LiRecDT5 from brown spider (Loxosceles intermedia) venom: From cloning to functional characterization. Biochimie 2007, 89, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Tavares, F.L.; Peichoto, M.E.; Rangel, D.E.M.; Barbaro, K.C.; Cirillo, M.C.; Santoro, M.L.; Sano-Martins, I.S. Loxosceles gaucho spider venom and its sphingomyelinase fraction trigger the main functions of human and rabbit platelets. Hum. Exp. Toxicol. 2011, 30, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, G.S.; Caporrino, M.C.; Della-Casa, M.S.; Kimura, L.F.; Prezotto-Neto, J.P.; Fukuda, D.A.; Portes-Junior, J.A.; Neves-Ferreira, A.G.; Santoro, M.L.; Barbaro, K.C. Cloning, expression and characterization of a phospholipase D from Loxosceles gaucho venom gland. Biochimie 2013, 95, 1773–1783. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Moreira, D.; Senff-Ribeiro, A.; Wille, A.C.; Gremski, L.H.; Chaim, O.M.; Veiga, S.S. Highlights in the knowledge of brown spider toxins. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 6. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Moreira, D.; Souza, F.N.; Fogaça, R.T.; Mangili, O.C.; Gremski, W.; Senff-Ribeiro, A.; Chaim, O.M.; Veiga, S.S. The relationship between calcium and the metabolism of plasma membrane phospholipids in hemolysis induced by brown spider venom phospholipase-d toxin. J. Cell. Biochem. 2011, 112, 2529–2540. [Google Scholar] [CrossRef] [PubMed]
- Gremski, L.H.; Trevisan-Silva, D.; Ferrer, V.P.; Matsubara, F.H.; Meissner, G.O.; Wille, A.C.; Vuitika, L.; Dias-Lopes, C.; Ullah, A.; de Moraes, F.R.; et al. Recent advances in the understanding of brown spider venoms: From the biology of spiders to the molecular mechanisms of toxins. Toxicon 2014, 83, 91–120. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lynch, K.R. Brown recluse spider (Loxosceles reclusa) venom phospholipase D (PLD) generates lysophosphatidic acid (LPA). Biochem. J. 2005, 391, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Van Meeteren, L.; Frederiks, F.; Giepmans, B.; Pedrosa, M.; Billington, S.; Jost, B.; Tambourgi, D.; Moolenaar, W. Spider and bacterial sphingomyelinases D target cellular lysophosphatidic acid receptors by hydrolyzing lysophosphatidylcholine. J. Biol. Chem. 2004, 279, 10833–10836. [Google Scholar] [CrossRef] [PubMed]
- Wille, A.C.; Chaves-Moreira, D.; Trevisan-Silva, D.; Magnoni, M.G.; Boia-Ferreira, M.; Gremski, L.H.; Gremski, W.; Chaim, O.M.; Senff-Ribeiro, A.; Veiga, S.S. Modulation of membrane phospholipids, the cytosolic calcium influx and cell proliferation following treatment of B16-F10 cells with recombinant phospholipase-d from Loxosceles intermedia (brown spider) venom. Toxicon 2013, 67, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Tambourgi, D.; Da Silva, M.; Billington, S.; De Andrade, R.; Magnoli, F.; Songer, J.; Van den Berg, C. Mechanism of induction of complement susceptibility of erythrocytes by spider and bacterial sphingomyelinases. Immunology 2002, 107, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Rees, R.S.; Gates, C.; Timmons, S.; Des Prez, R.M.; King, L.E. Plasma components are required for platelet activation by the toxin of Loxosceles reclusa. Toxicon 1988, 26, 1035–1045. [Google Scholar] [CrossRef]
- Da Silveira, R.B.; Pigozzo, R.B.; Chaim, O.M.; Appel, M.H.; Dreyfuss, J.L.; Toma, L.; Mangili, O.C.; Gremski, W.; Dietrich, C.P.; Nader, H.B.; et al. Molecular cloning and functional characterization of two isoforms of dermonecrotic toxin from Loxosceles intermedia (brown spider) venom gland. Biochimie 2006, 88, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Appel, M.H.; da Silveira, R.B.; Chaim, O.M.; Paludo, K.S.; Silva, D.T.; Chaves, D.M.; da Silva, P.H.; Mangili, O.C.; Senff-Ribeiro, A.; Gremski, W.; et al. Identification, cloning and functional characterization of a novel dermonecrotic toxin (phospholipase D) from brown spider (Loxosceles intermedia) venom. Biochim. Biophys. Acta 2008, 1780, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Tavares, F.L.; Sousa-e-Silva, M.C.; Santoro, M.L.; Barbaro, K.C.; Rebecchi, I.M.; Sano-Martins, I.S. Changes in hematological, hemostatic and biochemical parameters induced experimentally in rabbits by Loxosceles gaucho spider venom. Hum. Exp. Toxicol. 2004, 23, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Tavares, F.L.; Peichoto, M.E.; Marcelino, J.R.; Barbaro, K.C.; Cirillo, M.C.; Santoro, M.L.; Sano-Martins, I.S. Platelet participation in the pathogenesis of dermonecrosis induced by Loxosceles gaucho venom. Hum. Exp. Toxicol. 2016, 35, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Bevers, E.M.; Williamson, P.L. Getting to the outer leaflet: Physiology of phosphatidylserine exposure at the plasma membrane. Physiol. Rev. 2016, 96, 605–645. [Google Scholar] [CrossRef] [PubMed]
- Van Genderen, H.O.; Kenis, H.; Hofstra, L.; Narula, J.; Reutelingsperger, C.P. Extracellular annexin A5: Functions of phosphatidylserine-binding and two-dimensional crystallization. Biochim. Biophys. Acta 2008, 1783, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.; Chaim, O.; da Silveira, R.; Gremski, L.; Sade, Y.; Paludo, K.; Senff-Ribeiro, A.; de Moura, J.; Chavez-Olortegui, C.; Gremski, W.; et al. Biological and structural comparison of recombinant phospholipase d toxins from Loxosceles intermedia (brown spider) venom. Toxicon 2007, 50, 1162–1174. [Google Scholar] [CrossRef] [PubMed]
- Levin, C.; Bonstein, L.; Lauterbach, R.; Mader, R.; Rozemman, D.; Koren, A. Immune-mediated mechanism for thrombocytopenia after Loxosceles spider bite. Pediatr. Blood Cancer 2014, 61, 1466–1468. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Ye, H.; Locklin, R.M.; Ferguson, D.J.; Cui, Z.; Triffitt, J.T. Efficient characterisation of human cell-bioceramic interactions In Vitro and In Vivo by using enhanced GFP-labelled mesenchymal stem cells. Biomaterials 2005, 26, 5790–5800. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, A.; Sawano, A.; Kogure, T. Lighting up cells: Labelling proteins with fluorophores. Nat. Cell Biol. 2003, S1–S7. [Google Scholar]
- Denny, W.F.; Dillaha, C.J.; Morgan, P.N. Hemotoxic effect of Loxosceles reclusa venom: In vivo and In Vitro studies. J. Lab. Clin. Med. 1964, 64, 291–298. [Google Scholar] [PubMed]
- Rees, R.S. Platelet activation stimulated by the toxin of the brown recluse spider requires serum amyloid P component, not C-reactive protein. Toxicon 1989, 27, 953–954. [Google Scholar] [PubMed]
- Rother, E.; Brandl, R.; Baker, D.L.; Goyal, P.; Gebhard, H.; Tigyi, G.; Siess, W. Subtype-selective antagonists of lysophosphatidic acid receptors inhibit platelet activation triggered by the lipid core of atherosclerotic plaques. Circulation 2003, 108, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Huber, S.M.; Szabo, I.; Gulbins, E. Plasma membrane ion channels in suicidal cell death. Arch. Biochem. Biophys. 2007, 462, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Mitsutake, S.; Igarashi, Y. Transbilayer movement of ceramide in the plasma membrane of live cells. Biochem. Biophys. Res. Commun. 2007, 359, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Schoenwaelder, S.M.; Yuan, Y.; Josefsson, E.C.; White, M.J.; Yao, Y.; Mason, K.D.; O’Reilly, L.A.; Henley, K.J.; Ono, A.; Hsiao, S.; et al. Two distinct pathways regulate platelet phosphatidylserine exposure and procoagulant function. Blood 2009, 114, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, J.W.; Mattheij, N.J.; Cosemans, J.M. Platelet-based coagulation: Different populations, different functions. J. Thromb. Haemost. 2013, 11, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, J.W.; Bevers, E.M.; Lindhout, T. Platelet activation and blood coagulation. Thromb. Haemost. 2002, 88, 186–193. [Google Scholar] [PubMed]
- Butera, D.; Piazza, R.M.; McLane, M.A.; Chammas, R.; da Silva, A.M. Molecular engineering of an egfp/disintegrin-based integrin marker. Toxicon 2005, 46, 178–184. [Google Scholar] [CrossRef] [PubMed]
- mMass Open Source Mass Spectrometry Tool (Martin Strohalm©). Available online: www.mmass.org (accessed on 10 May 2017).
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukuda, D.A.; Caporrino, M.C.; Barbaro, K.C.; Della-Casa, M.S.; Faquim-Mauro, E.L.; Magalhaes, G.S. Recombinant Phospholipase D from Loxosceles gaucho Binds to Platelets and Promotes Phosphatidylserine Exposure. Toxins 2017, 9, 191. https://doi.org/10.3390/toxins9060191
Fukuda DA, Caporrino MC, Barbaro KC, Della-Casa MS, Faquim-Mauro EL, Magalhaes GS. Recombinant Phospholipase D from Loxosceles gaucho Binds to Platelets and Promotes Phosphatidylserine Exposure. Toxins. 2017; 9(6):191. https://doi.org/10.3390/toxins9060191
Chicago/Turabian StyleFukuda, Daniel A., Maria C. Caporrino, Katia C. Barbaro, Maisa S. Della-Casa, Eliana L. Faquim-Mauro, and Geraldo S. Magalhaes. 2017. "Recombinant Phospholipase D from Loxosceles gaucho Binds to Platelets and Promotes Phosphatidylserine Exposure" Toxins 9, no. 6: 191. https://doi.org/10.3390/toxins9060191
APA StyleFukuda, D. A., Caporrino, M. C., Barbaro, K. C., Della-Casa, M. S., Faquim-Mauro, E. L., & Magalhaes, G. S. (2017). Recombinant Phospholipase D from Loxosceles gaucho Binds to Platelets and Promotes Phosphatidylserine Exposure. Toxins, 9(6), 191. https://doi.org/10.3390/toxins9060191





