Presence of Enniatins and Beauvericin in Romanian Wheat Samples: From Raw Material to Products for Direct Human Consumption
Abstract
:1. Introduction
2. Results
2.1. Presence of Emerging Mycotoxins
2.1.1. Wheat
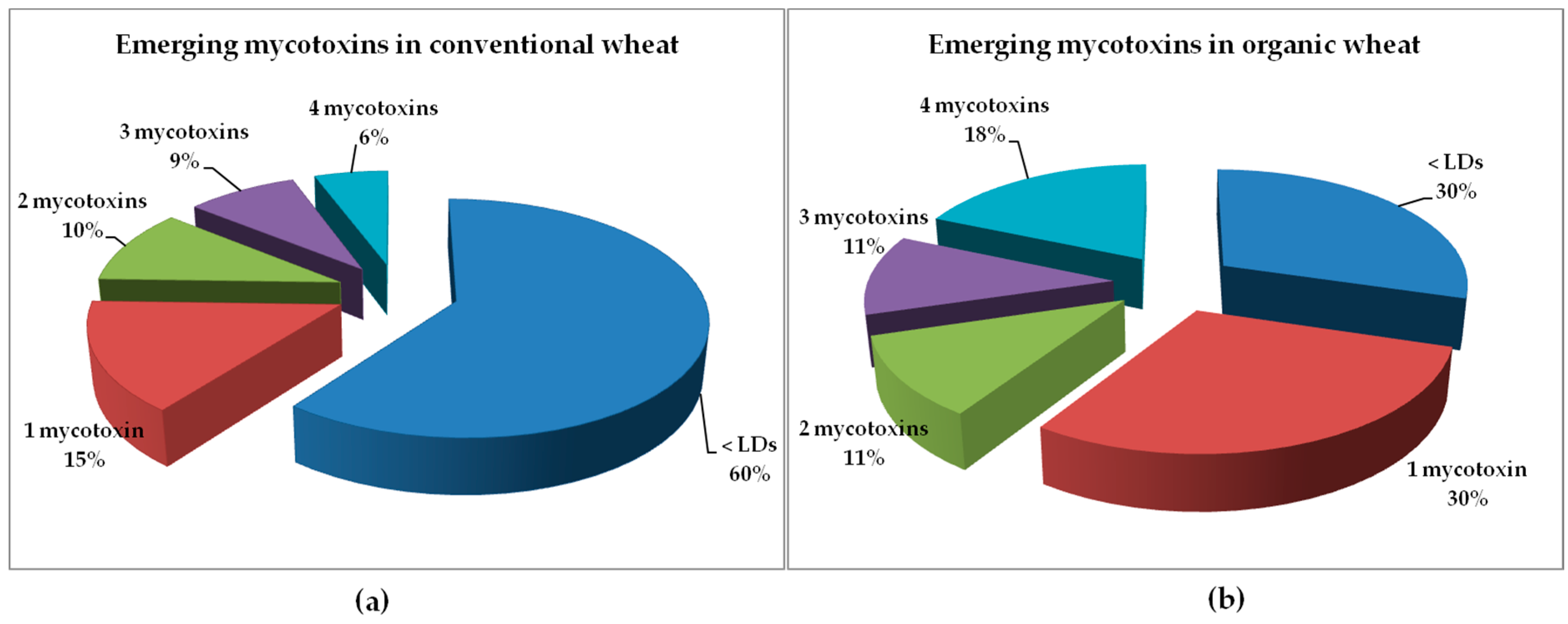
2.1.2. Conventional versus Organic Wheat
2.1.3. Wheat-Based Products
2.2. Dietary Exposure
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemical and Reagents
5.2. Sampling
5.3. Extraction
5.3.1. Wheat
5.3.2. Wheat-Based Products
5.4. Mycotoxin Analysis by HPLC-MS/MS
5.5. Statistical Analyses
5.6. Estimation of Daily Intake
Acknowledgments
Author Contributions
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization of the United Nations. Statistic Division. Download Data. Food Balance. Food Supply—Crops Primary Equivalent. Available online: http://faostat3.fao.org/compare/E (accessed on 15 August 2016).
- RINS. Producţia Vegetală la Principalele Culturi în Anul 2015—Date Provizorii [Agricultural Vegetal Production for Main Crops in Romania for 2015 Harvesting Year]; Romanian National Institute of Statistcs: Bucharest, Romania, 2016; Available online: http://www.insse.ro/cms/files/statistici/comunicate/com_anuale/Prod_veg/prod_veg_r15.pdf (accessed on 14 January 2017).
- Schmidt, M.; Horstmann, S.; De Colli, L.; Danaher, M.; Speer, K.; Zannini, E.; Arendt, E.K. Impact of fungal contamination of wheat on grain quality criteria. J. Cereal Sci. 2016, 69, 95–103. [Google Scholar] [CrossRef]
- Stanciu, O.; Banc, R.; Cozma, A.; Filip, L.; Miere, D.; Mañes, J.; Loghin, F. Occurence of Fusarium mycotoxins in wheat from Europe—A review. Acta Univ. Cibiniensis Ser. E FOOD Technol. 2015, 19, 35–60. [Google Scholar] [CrossRef]
- Garcia, J.M.; Teixeira, P. Organic versus conventional food: A comparison regarding food safety. Food Rev. Int. 2017, 33, 424–446. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- Sirot, V.; Fremy, J.M.; Leblanc, J.C. Dietary exposure to mycotoxins and health risk assessment in the second French total diet study. Food Chem. Toxicol. 2013, 52, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jestoi, M. Emerging Fusarium-Mycotoxins Fusaproliferin, Beauvericin, Enniatins, and Moniliformin: A Review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef] [PubMed]
- Vaclavikova, M.; Malachova, A.; Veprikova, Z.; Dzuman, Z.; Zachariasova, M.; Hajslova, J. “Emerging” mycotoxins in cereals processing chains: Changes of enniatins during beer and bread making. Food Chem. 2013, 136, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; Beccari, G.; Prodi, A.; Generotti, S.; Etruschi, F.; Meca, G.; Juan, C.; Mañes, J. Biosynthesis of beauvericin and enniatins invitro by wheat Fusarium species and natural grain contamination in an area of central Italy. Food Microbiol. 2015, 46, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Gruber-Dorninger, C.; Novak, B.; Nagl, V.; Berthiller, F. Emerging mycotoxins: Beyond traditionally determined food contaminants. J. Agric. Food Chem. 2016. [Google Scholar] [CrossRef] [PubMed]
- Escrivá, L.; Font, G.; Manyes, L. In vivo toxicity studies of fusarium mycotoxins in the last decade: A review. Food Chem. Toxicol. 2015, 78, 185–206. [Google Scholar] [CrossRef] [PubMed]
- Juan-Garcia, A.; Ruiz, M.J.; Font, G.; Manyes, L. Enniatin A1, enniatin B1 and beauvericin on HepG2: Evaluation of toxic effects. Food Chem. Toxicol. 2015, 84, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Xu, L. Beauvericin, a bioactive compound produced by fungi: A short review. Molecules 2012, 17, 2367–2377. [Google Scholar] [CrossRef] [PubMed]
- Juan-García, A.; Manyes, L.; Ruiz, M.-J.; Font, G. Involvement of enniatins-induced cytotoxicity in human HepG2 cells. Toxicol. Lett. 2013, 218, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Prosperini, A.; Font, G.; Ruiz, M.J. Interaction effects of Fusarium enniatins (A, A1, B and B1) combinations on in vitro cytotoxicity of Caco-2 cells. Toxicol. In Vitro 2014, 28, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Fernández-Franzón, M.; Font, G.; Ruiz, M.J. Toxicity evaluation of individual and mixed enniatins using an in vitro method with CHO-K1 cells. Toxicol. In Vitro 2013, 27, 672–680. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain. Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J. 2014, 12, 3802. [Google Scholar]
- JECFA. Evaluation of Certain Mycotoxins in Food; WHO Technical Report Series, 56th Report; WHO: Geneve, Switzerland, 2002. [Google Scholar]
- SCF. Opinion of the Scientific Committee on Food on Fusarium Toxins. Part 6: Group Evaluation of T-2 Toxin, HT-2 Toxin, Nivalenol and Deoxynivalenol; European Commission: Brusells, Belgium, 2002. [Google Scholar]
- Alkadri, D.; Rubert, J.; Prodi, A.; Pisi, A.; Mañes, J.; Soler, C. Natural co-occurrence of mycotoxins in wheat grains from Italy and Syria. Food Chem. 2014, 157, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Covarelli, L.; Beccari, G.; Colasante, V.; Mañes, J. Simultaneous analysis of twenty-six mycotoxins in durum wheat grain from Italy. Food Control 2016, 62, 322–329. [Google Scholar] [CrossRef]
- Bryła, M.; Waśkiewicz, A.; Podolska, G.; Szymczyk, K.; Jędrzejczak, R.; Damaziak, K.; Sułek, A. Occurrence of 26 Mycotoxins in the Grain of Cereals Cultivated in Poland. Toxins 2016, 8, 160. [Google Scholar] [CrossRef] [PubMed]
- Jestoi, M.; Rokka, M.; Parikka, P.; Rizzo, A.; Peltonen, K. Presence and concentrations of the Fusarium-related mycotoxins beauvericin, enniatins and moniliformin in Finnish grain samples. Food Addit. Contam. 2004, 21, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.B.; Font, G.; Mañes, J.; Ferrer, E. Emerging Fusarium mycotoxins in organic and conventional pasta collected in Spain. Food Chem. Toxicol. 2013, 51, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.G. Fusarium mycotoxin content of UK organic and conventional wheat. Food Addit. Contam. Part A 2009, 26, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Bernhoft, A.; Torp, M.; Clasen, P.-E.-E.; Løes, A.-K.; Kristoffersen, A.B. Influence of agronomic and climatic factors on Fusarium infestation and mycotoxin contamination of cereals in Norway. Food Addit. Contam. Part A 2012, 29, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Lazzaro, I.; Moretti, A.; Giorni, P.; Brera, C.; Battilani, P. Organic vs. conventional farming: Differences in infection by mycotoxin-producing fungi on maize and wheat in Northern and Central Italy. Crop Prot. 2015, 72, 22–30. [Google Scholar] [CrossRef]
- Vrček, I.V.; Čepo, D.V.; Rašić, D.; Peraica, M.; Žuntar, I.; Bojić, M.; Mendaš, G.; Medić-Šarić, M. A comparison of the nutritional value and food safety of organically and conventionally produced wheat flours. Food Chem. 2014, 143, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Kirinčič, S.; Škrjanc, B.; Kos, N.; Kozolc, B.; Pirnat, N.; Tavčar-Kalcher, G. Mycotoxins in cereals and cereal products in Slovenia—Official control of foods in the years 2008–2012. Food Control 2015, 50, 157–165. [Google Scholar] [CrossRef]
- Czaban, J.; Wróblewska, B.; Sułek, A.; Mikos, M.; Boguszewska, E.; Podolska, G.; Nieróbca, A. Colonisation of winter wheat grain by Fusarium spp. and mycotoxin content as dependent on a wheat variety, crop rotation, a crop management system and weather conditions. Food Addit. Contam. Part A 2015, 32, 874–910. [Google Scholar] [CrossRef] [PubMed]
- Gagiu, A.; Mateescu, E. Evaluarea Riscului Privind Contaminarea cu Micotoxine a Producţiilor Anuale de Grâu din România [Risk Assessment Concerning Mycotoxin Contamination of Annual Wheat Production from Romania]; National Institute of Research and Development for Food Bioresources—IBA Bucharest: Bucharest, Romania, 2012; Available online: http://www.indal.bioresurse.ro/docs/et2/raport2.pdf (accessed on 27 November 2016).
- Mahnine, N.; Meca, G.; Elabidi, A.; Fekhaoui, M.; Saoiabi, A.; Font, G.; Mañes, J.; Zinedine, A. Further data on the levels of emerging Fusarium mycotoxins enniatins (A, A1, B, B1), beauvericin and fusaproliferin in breakfast and infant cereals from Morocco. Food Control 2011, 124, 481–485. [Google Scholar] [CrossRef]
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions: A review. LWT Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Juan, C.; Mañes, J.; Raiola, A.; Ritieni, A. Evaluation of beauvericin and enniatins in Italian cereal products and multicereal food by liquid chromatography coupled to triple quadrupole mass spectrometry. Food Chem. 2013, 140, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Capriotti, A.L.; Cavaliere, C.; Foglia, P.; Samperi, R.; Stampachiacchiere, S.; Ventura, S.; Laganà, A. Multiclass analysis of mycotoxins in biscuits by high performance liquid chromatography-tandem mass spectrometry. Comparison of different extraction procedures. J. Chromatogr. A 2014, 1343, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, O.; Juan, C.; Miere, D.; Loghin, F.; Mañes, J. Occurrence and co-occurrence of Fusarium mycotoxins in wheat grains and wheat flour from Romania. Food Control 2017, 73, 147–155. [Google Scholar] [CrossRef]
- Tolosa, J.; Graziani, G.; Gaspari, A.; Chianese, D.; Ferrer, E.; Mañes, J.; Ritieni, A. Multi-Mycotoxin Analysis in Durum Wheat Pasta by Liquid Chromatography Coupled to Quadrupole Orbitrap Mass Spectrometry. Toxins 2017, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.M.; Saladino, F.; Mañes, J.; Fernández-Franzón, M.; Meca, G. Occurrence of mycotoxins in refrigerated pizza dough and risk assessment of exposure for the Spanish population. Food Chem. Toxicol. 2016, 94, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Fernández-Franzón, M.; Mañes, J.; Manyes, L. Multi-mycotoxin contamination of couscous semolina commercialized in Morocco. Food Chem. 2017, 214, 440–446. [Google Scholar] [CrossRef] [PubMed]
- JECFA. Evaluation of Certain Contaminants in Food; WHO Technical Report Series, 72th Report; WHO: Geneve, Switzerland, 2011. [Google Scholar]
- Saladino, F.; Quiles, J.M.; Mañes, J.; Fernández-Franzón, M.; Luciano, F.B.; Meca, G. Dietary exposure to mycotoxins through the consumption of commercial bread loaf in Valencia, Spain. LWT Food Sci. Technol. 2017, 75, 697–701. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Union 2006, L70, 12–34. [Google Scholar]
- Stanciu, O.; Juan, C.; Miere, D.; Loghin, F.; Mañes, J. Analysis of enniatins and beauvericin by LC-MS/MS in wheat-based products. CyTA J. Food 2017, 15, 433–440. [Google Scholar] [CrossRef]
- IPCS. Chapter 6: Dietary exposure assessment of chemicals in food. In Principles and Methods for the Risk Assessment of Chemicals in Food; FAO/WHO: Geneve, Switzerland, 2009; pp. 1–98. [Google Scholar]
- European Food Safety Authority. Management of left-censored data in dietary exposure assessment of chemical substances. EFSA J. 2010, 8, 1557. [Google Scholar]
| Mycotoxin | Parameter | Conventional | Organic | Total |
|---|---|---|---|---|
| (n = 106) | (n = 27) | (n = 133) | ||
| BEA | Incidence | 3 | 0 | 3 |
| LD-LQ | 2 | 0 | 2 | |
| Frequency (%) | 2 | 0 | 2 | |
| Mean (μg kg−1) | 0.07 | n.q. | 0.07 | |
| Max. (μg kg−1) | 9.1 | n.q. | 9.1 | |
| ENA | Incidence | 6 | 5 | 11 |
| LD-LQ | 0 | 2 | 2 | |
| Frequency (%) | 6 | 19 | 8 | |
| Mean (μg kg−1) | 3.8 | 7.0 | 4.5 | |
| Max. (μg kg−1) | 140 | 96.6 | 140 | |
| ENA1 | Incidence | 19 | 8 | 27 |
| LD-LQ | 0 | 0 | 0 | |
| Frequency (%) | 18 | 30 | 20 | |
| Mean (μg kg−1) | 11.2 | 23.7 | 13.7 | |
| Max. (μg kg−1) | 356 | 272 | 356 | |
| ENB | Incidence | 36 | 19 | 55 |
| LD-LQ | 0 | 1 | 1 | |
| Frequency (%) | 34 | 70 | 41 | |
| Mean (μg kg−1) | 42.9 | 102 | 54.8 | |
| Max. (μg kg−1) | 815 | 487 | 815 | |
| ENB1 | Incidence | 25 | 11 | 36 |
| LD-LQ | 1 | 0 | 1 | |
| Frequency (%) | 24 | 41 | 27 | |
| Mean (μg kg−1) | 21.5 | 66.0 | 30.5 | |
| Max. (μg kg−1) | 510 | 510 | 510 |
| Mycotoxins | Conventional Wheat | Organic Wheat | Wheat Products |
|---|---|---|---|
| (n = 106) | (n = 27) | (n = 111) | |
| Two mycotoxins | |||
| ENA1 + ENB | 1 | 0 | 0 |
| ENA1 + ENB1 | 3 | 0 | 0 |
| ENB + ENB1 | 7 | 3 | 16 |
| Three mycotoxins | |||
| ENA1 + ENB + ENB1 | 9 | 3 | 1 |
| BEA + ENB + ENB1 | 0 | 0 | 1 |
| Four mycotoxins | |||
| BEA + ENA1 + ENB + ENB1 | 1 | 0 | 0 |
| ENA + ENA1 + ENB + ENB1 | 5 | 5 | 0 |
| Variety | ENA | ENA1 | ENB | ENB1 | Overall Occurrence | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Occurrence | Mean | Max. | Occurrence | Mean | Max. | Occurrence | Mean | Max. | Occurrence | Mean | Max. | ||
| (μg kg−1) | (μg kg−1) | (μg kg−1) | (μg kg−1) | ||||||||||
| Alcantara | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Alex | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Altigo | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 1/1 | 157.4 | 157 | 1/1 | 12.4 | 12.4 | 1/1 |
| Arezzo | 0/3 | n.q. | n.q. | 1/3 | 3.9 | 11.8 | 0/3 | n.q. | n.q. | 1/3 | 3.8 | 11.6 | 1/3 |
| Arieşan | 0/7 | n.q. | n.q. | 1/7 | 3.6 | 25.5 | 6/7 | 57.7 | 149 | 2/7 | 15.2 | 71.1 | 6/7 |
| Balaton | 0/2 | n.q. | n.q. | 2/2 | 27.0 | 43.0 | 2/2 | 40.5 | 65.1 | 1/2 | 37.0 | 74.0 | 2/2 |
| Boema | 0/4 | n.q. | n.q. | 0/4 | n.q. | n.q. | 1/4 | 0.5 | 2.1 | 0/4 | n.q. | n.q. | 1/4 |
| Dropia | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Exotic | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 1/1 | 55.7 | 55.7 | 1/1 | 4.5 | 4.5 | 1/1 |
| Felix | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Glosa | 0/10 | n.q. | n.q. | 1/10 | 1.8 | 17.5 | 3/10 | 3.9 | 33.9 | 1/10 | 2.0 | 20.2 | 3/10 |
| Hyfi | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| ITC-20 | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Izvor | 0/7 | n.q. | n.q. | 1/7 | 9.4 | 65.6 | 1/7 | 22.7 | 159 | 1/7 | 16.6 | 116 | 1/7 |
| Kontrast | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 1/1 | 5.2 | 5.2 | 0/1 | n.q. | n.q. | 1/1 |
| Litera | 0/5 | n.q. | n.q. | 1/5 | 2.8 | 14.2 | 1/5 | 8.0 | 40.1 | 1/5 | 7.4 | 37.2 | 1/5 |
| Lukulus | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Miranda | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 |
| Ponomicus | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 | n.q. | n.q. | 0/2 |
| Solehio | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Soxenos | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 1/1 | 12.6 | 12.6 | 0/1 | n.q. | n.q. | 1/1 |
| Urbanus | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 | n.q. | n.q. | 0/1 |
| Other types | 6/51 | 7.9 | 140 | 12/51 | 19.5 | 356 | 18/51 | 70.4 | 815 | 16/51 | 37.1 | 510 | 23/51 |
| Mycotoxin | Parameter | Flour | Pasta | Breakfast Cereals | Biscuits | Total |
|---|---|---|---|---|---|---|
| (n = 41) | (n = 40) | (n = 7) | (n = 23) | (n = 111) | ||
| BEA | Incidence | 0 | 0 | 0 | 1 | 1 |
| LD-LQ | 0 | 0 | 0 | 1 | 1 | |
| Frequency (%) | 0 | 0 | 0 | 4 | 1 | |
| Mean (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| Max. (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| ENA | Incidence | 0 | 0 | 0 | 0 | 0 |
| LD-LQ | 0 | 0 | 0 | 0 | 0 | |
| Frequency (%) | 0 | 0 | 0 | 0 | 0 | |
| Mean (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| Max. (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| ENA1 | Incidence | 0 | 1 | 0 | 0 | 1 |
| LD-LQ | 0 | 1 | 0 | 0 | 1 | |
| Frequency (%) | 0 | 3 | 0 | 0 | 1 | |
| Mean (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| Max. (μg kg−1) | n.q. | n.q. | n.q. | n.q. | n.q. | |
| ENB | Incidence | 12 | 11 | 2 | 10 | 35 |
| LD-LQ | 1 | 0 | 0 | 3 | 4 | |
| Frequency (%) | 29 | 28 | 29 | 43 | 32 | |
| Mean (μg kg−1) | 1.8 | 10.4 | 1.9 | 1.7 | 4.9 | |
| Max. (μg kg−1) | 38.2 | 170 | 7.8 | 9.7 | 170 | |
| ENB1 | Incidence | 2 | 9 | 1 | 6 | 18 |
| LD-LQ | 0 | 2 | 0 | 3 | 5 | |
| Frequency (%) | 5 | 23 | 14 | 26 | 16 | |
| Mean (μg kg−1) | 0.5 | 1.9 | 0.5 | 0.7 | 1.0 | |
| Max. (μg kg−1) | 16.6 | 44.8 | 3.6 | 6.2 | 44.8 |
| Mycotoxin | EDI (ng/kg bw/day) | %TDI | ||
|---|---|---|---|---|
| LB | UB | LB | UB | |
| BEA | 0 | 5.3 | n.c. | n.c. |
| ENA | 0 | 31.7 | 0 | 3.17 |
| ENA1 | 0 | 10.7 | 0 | 1.07 |
| ENB | 25.8 | 27.8 | 2.58 | 2.78 |
| ENB1 | 5.4 | 10.3 | 0.54 | 1.03 |
| Sum of ENs | 31.2 | 80.5 | 3.12 | 8.05 |
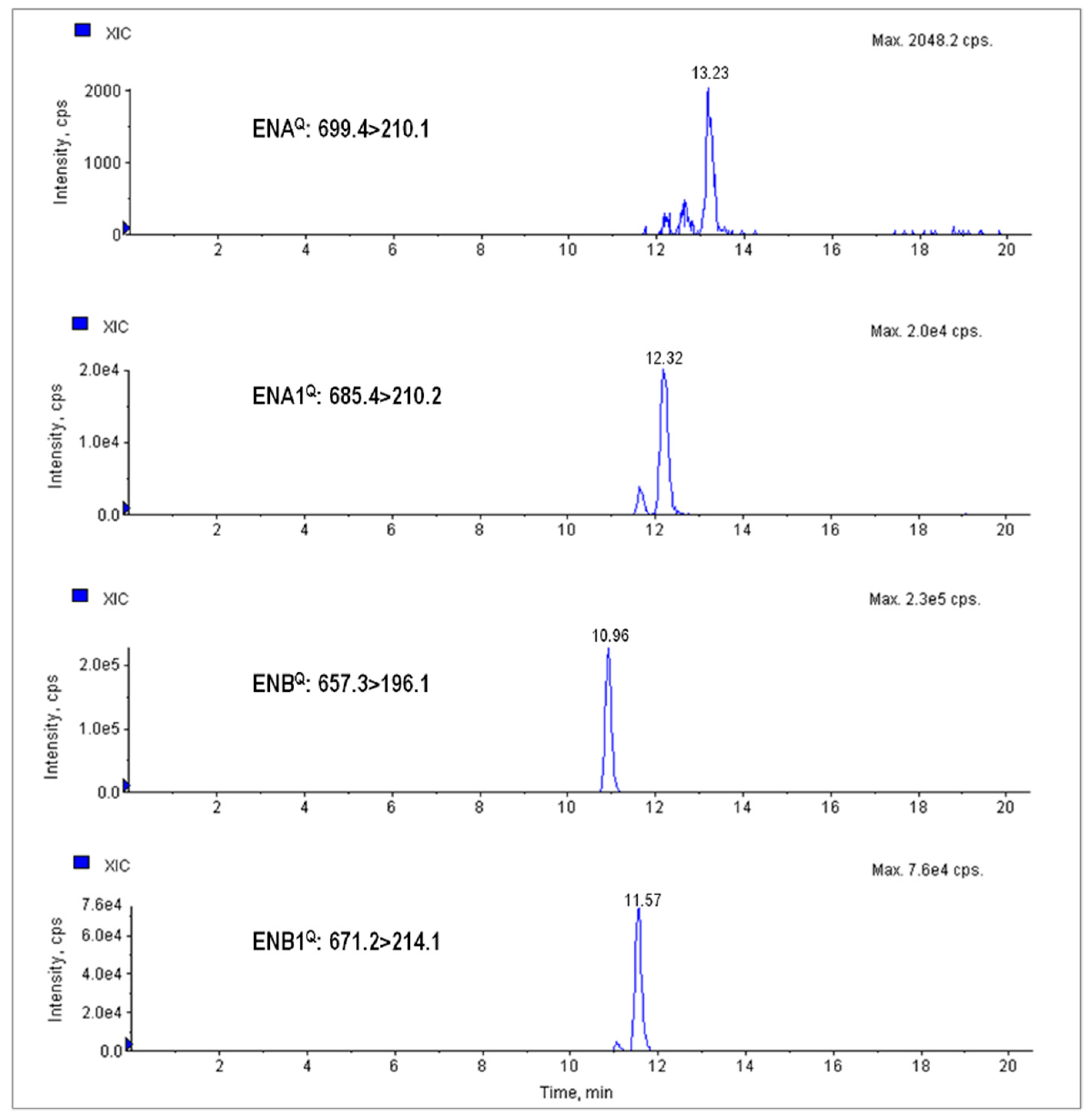
| Analyte | Rt | Mw | MS/MS Detection Parameters | Method Sensitivity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Precursor Ion | Product Ions | DP | CEP | CE | CXP | Wheat | Products | |||||
| LD | LQ | LD | LQ | |||||||||
| (min) | (g/mol) | (m/z) | (V) | (μg kg−1) | ||||||||
| BEA | 11.33 | 783.95 | 801.2 | 784.1Q | 116 | 33 | 27 | 10 | 4 | 8 | 1 | 2 |
| [M + NH4]+ | 244.1q | 39 | 6 | |||||||||
| ENA | 13.23 | 681.90 | 699.4 | 210.1Q | 76 | 30 | 35 | 14 | 6 | 12 | 6 | 12 |
| [M + NH4]+ | 228.2q | 59 | 16 | |||||||||
| ENA1 | 12.32 | 667.87 | 685.4 | 210.2Q | 66 | 29 | 37 | 8 | 3 | 6 | 2 | 4 |
| [M + NH4]+ | 214.2q | 59 | 10 | |||||||||
| ENB | 10.96 | 639.82 | 657.3 | 196.1Q | 51 | 28 | 39 | 8 | 1 | 2 | 0.5 | 1 |
| [M + NH4]+ | 214.0q | 59 | 10 | |||||||||
| ENB1 | 11.57 | 653.85 | 671.2 | 214.1Q | 66 | 29 | 61 | 10 | 1 | 2 | 1 | 2 |
| [M + NH4]+ | 228.1q | 57 | 12 | |||||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanciu, O.; Juan, C.; Miere, D.; Loghin, F.; Mañes, J. Presence of Enniatins and Beauvericin in Romanian Wheat Samples: From Raw Material to Products for Direct Human Consumption. Toxins 2017, 9, 189. https://doi.org/10.3390/toxins9060189
Stanciu O, Juan C, Miere D, Loghin F, Mañes J. Presence of Enniatins and Beauvericin in Romanian Wheat Samples: From Raw Material to Products for Direct Human Consumption. Toxins. 2017; 9(6):189. https://doi.org/10.3390/toxins9060189
Chicago/Turabian StyleStanciu, Oana, Cristina Juan, Doina Miere, Felicia Loghin, and Jordi Mañes. 2017. "Presence of Enniatins and Beauvericin in Romanian Wheat Samples: From Raw Material to Products for Direct Human Consumption" Toxins 9, no. 6: 189. https://doi.org/10.3390/toxins9060189
APA StyleStanciu, O., Juan, C., Miere, D., Loghin, F., & Mañes, J. (2017). Presence of Enniatins and Beauvericin in Romanian Wheat Samples: From Raw Material to Products for Direct Human Consumption. Toxins, 9(6), 189. https://doi.org/10.3390/toxins9060189