Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa
Abstract
:1. Introduction
2. Results and Discussion
2.1. Specimen Collection
2.2. Transcriptomics and Proteomics
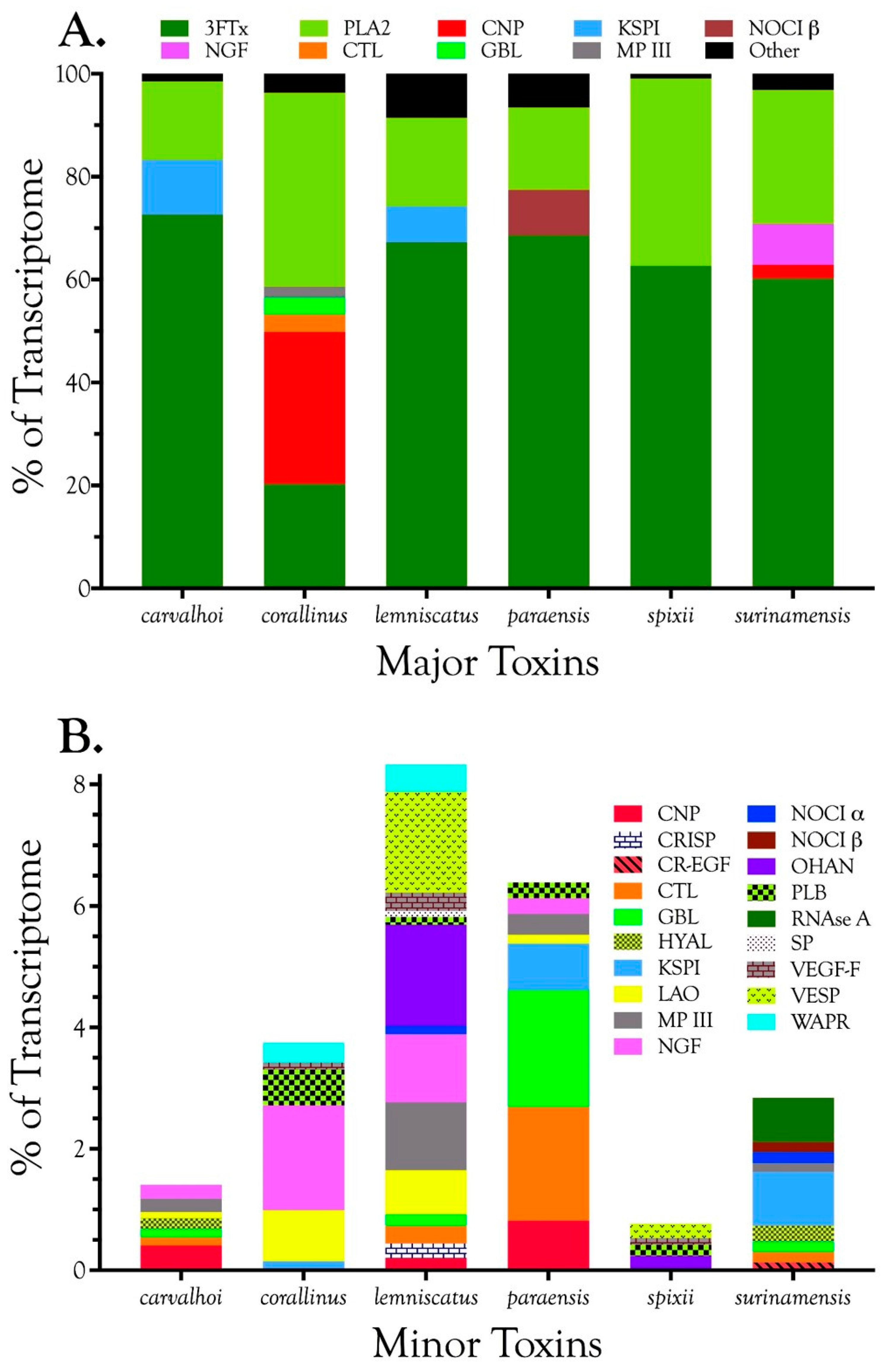
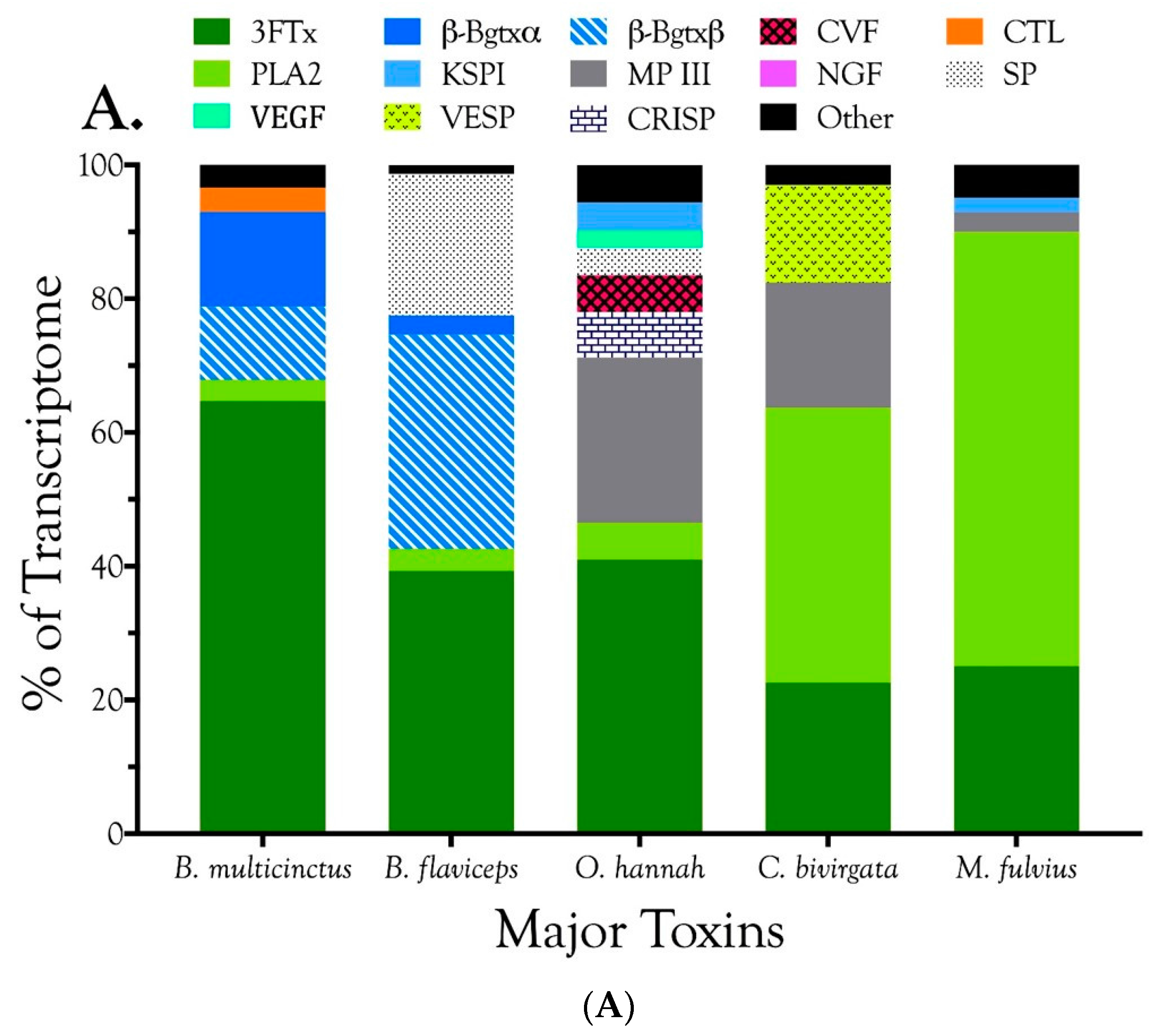
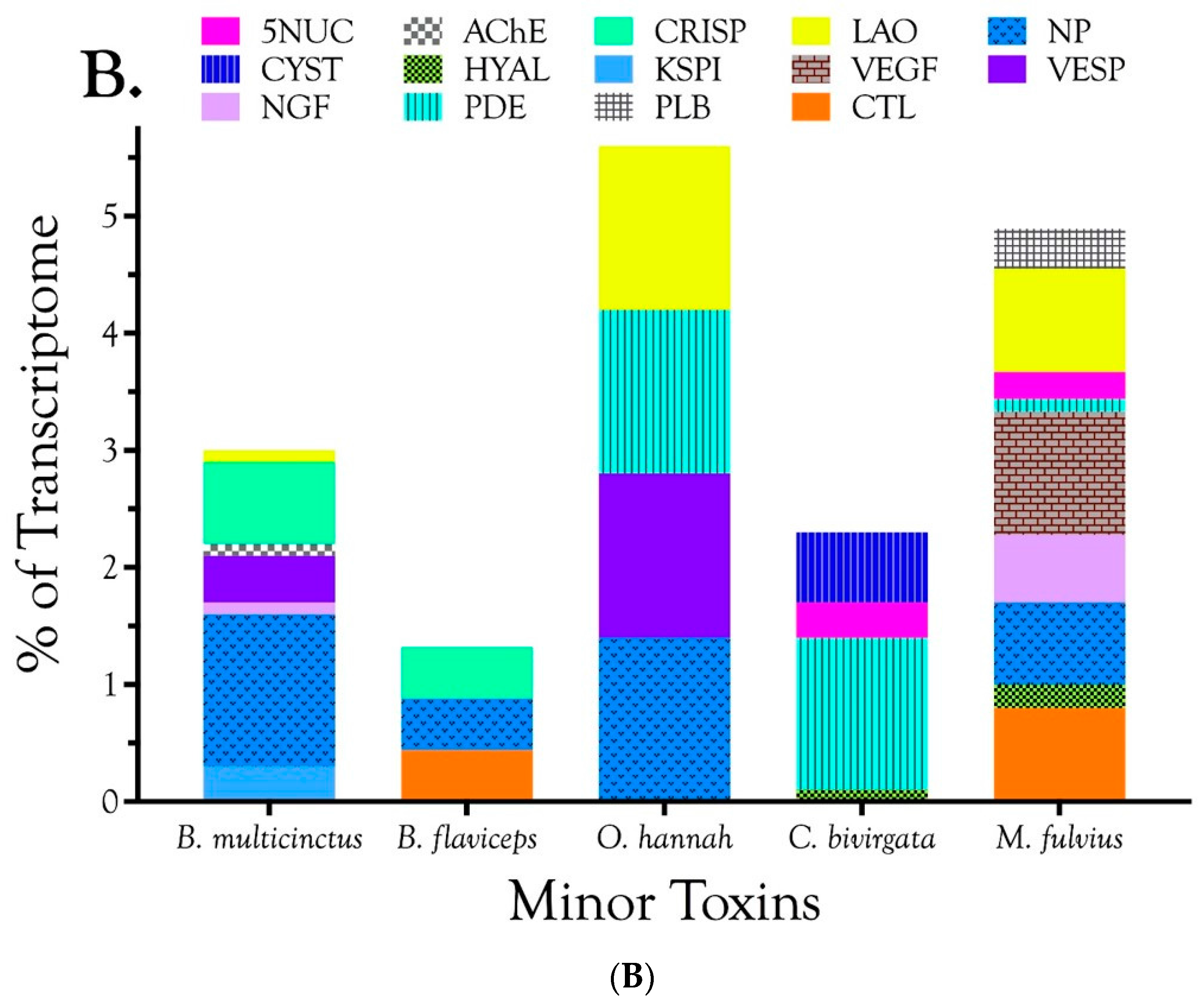
2.2.1. Characterization of Transcriptomes
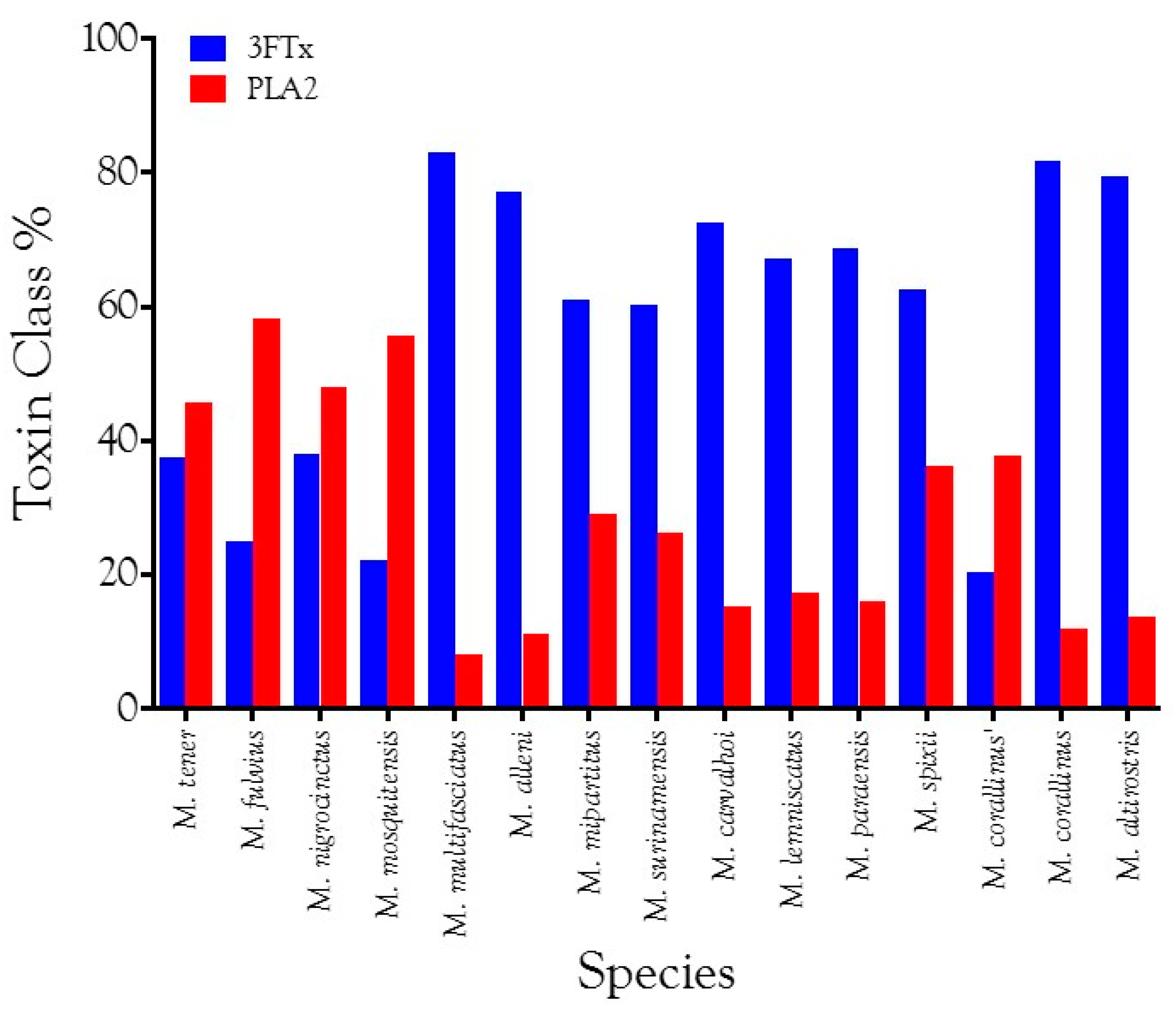
2.2.2. Characterization of Proteomes
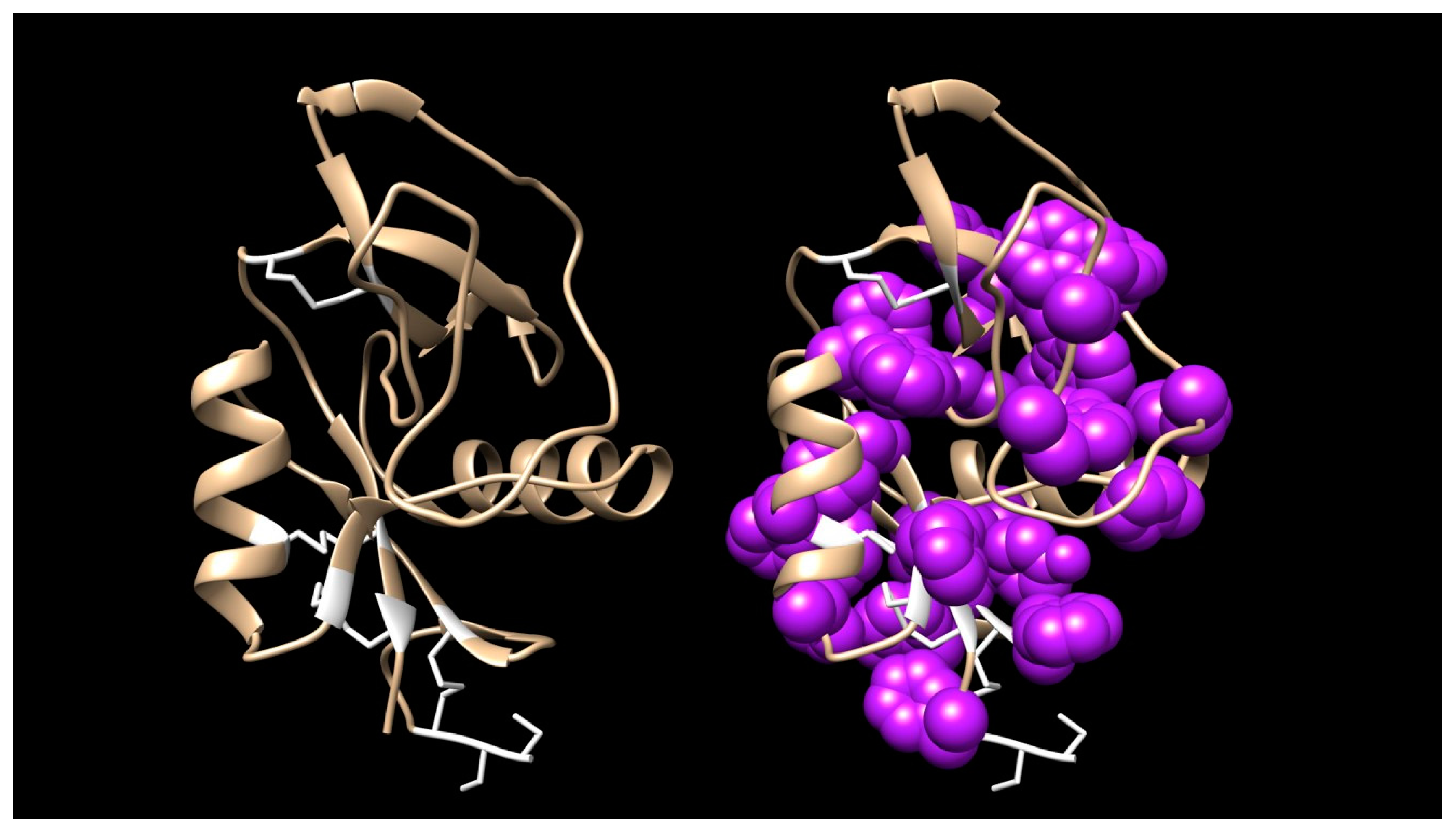
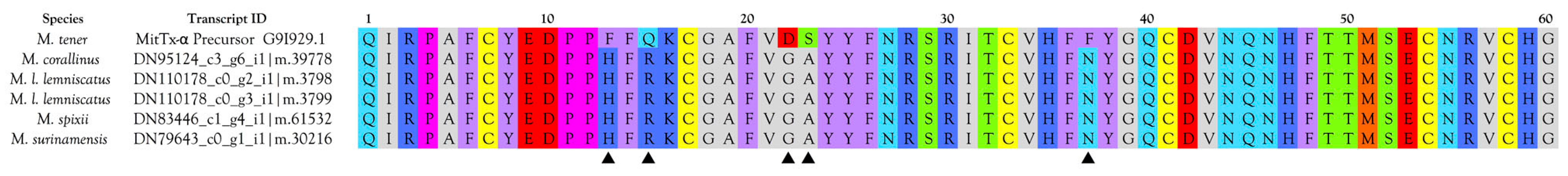
2.2.3. 3-Finger Toxins (3FTx)
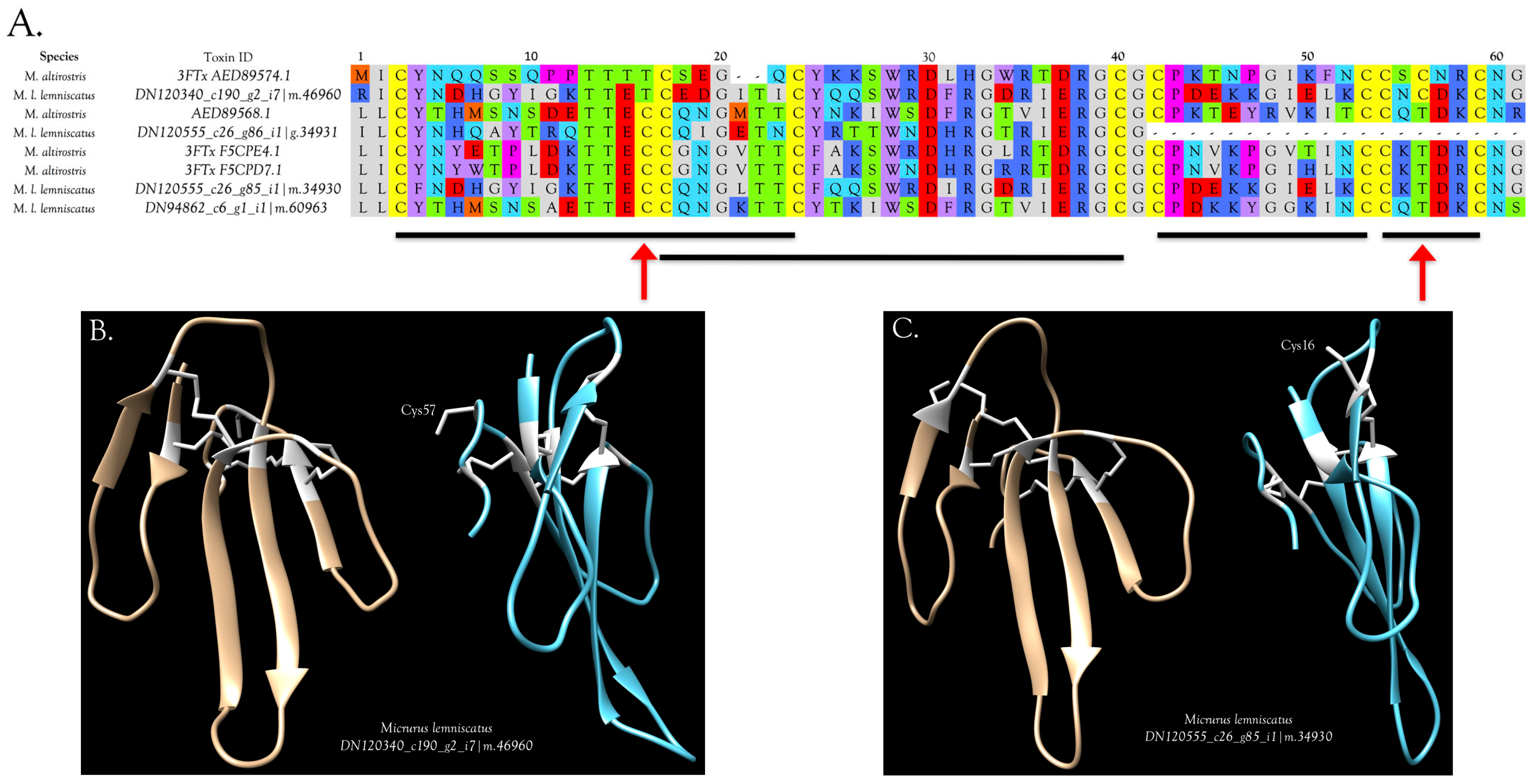
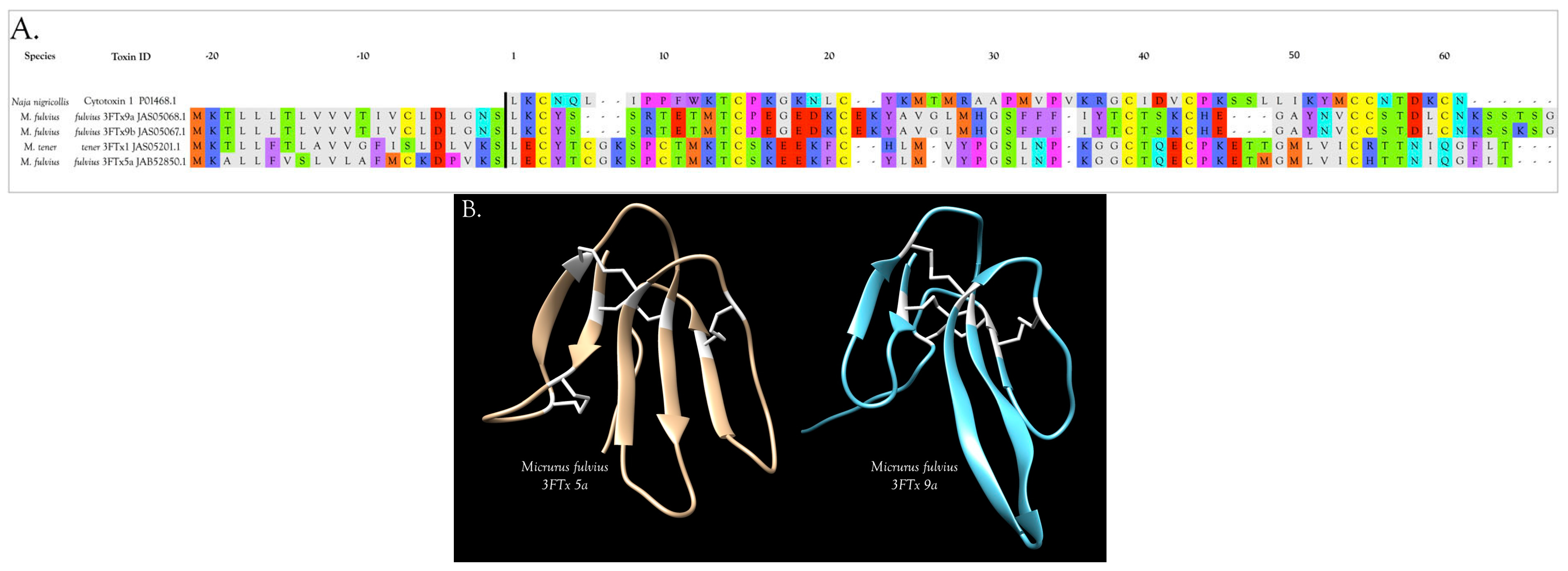
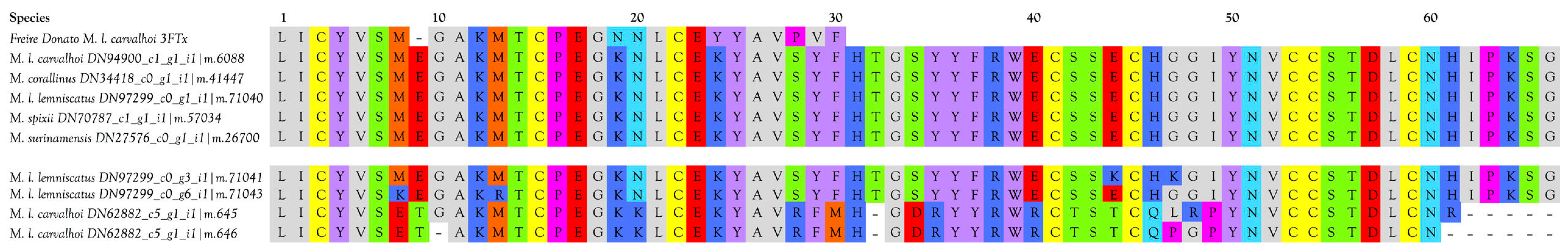
3FTxs with 10 Cysteine Residues
3FTxs and GABA Receptors
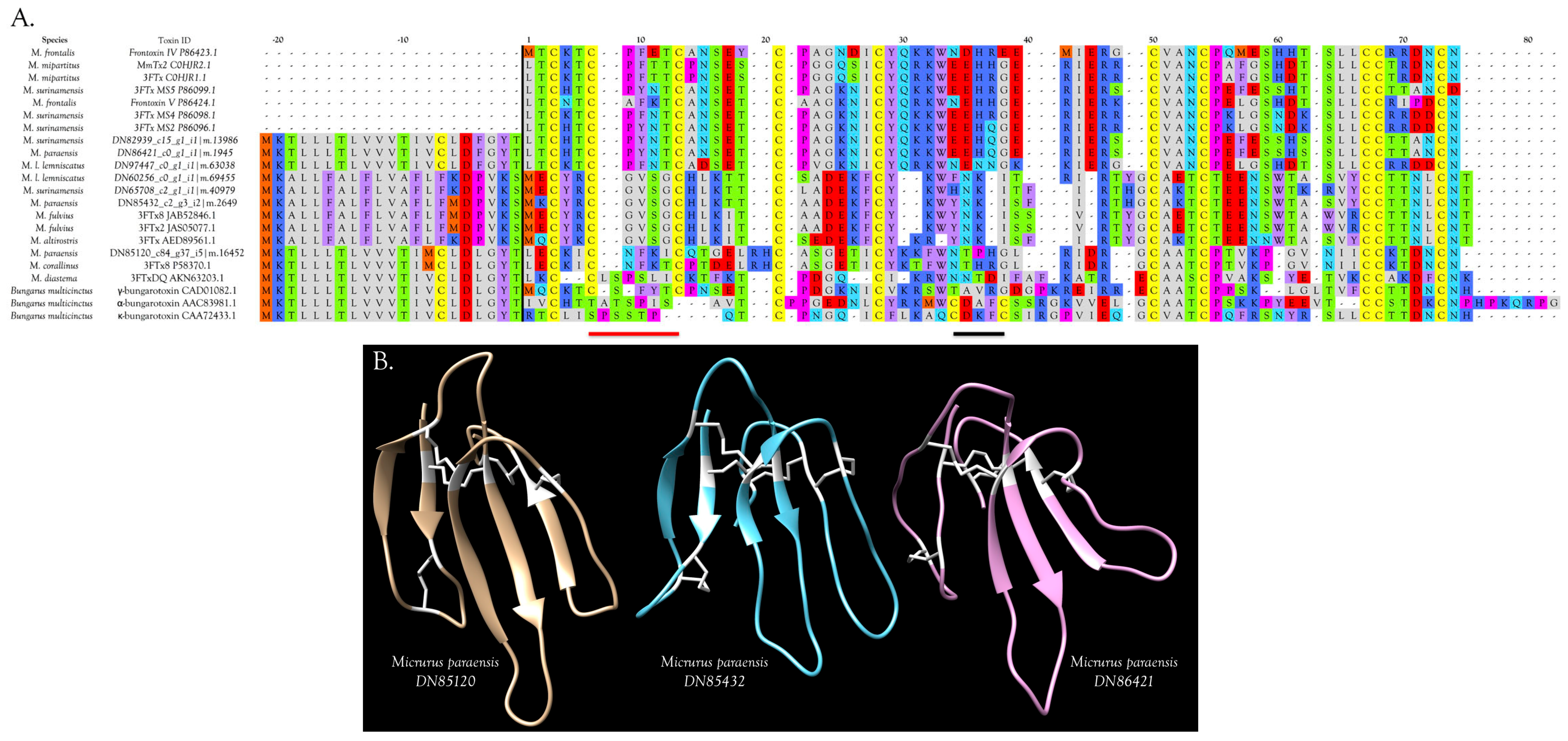
3FTxs with 8 Cysteine Residues
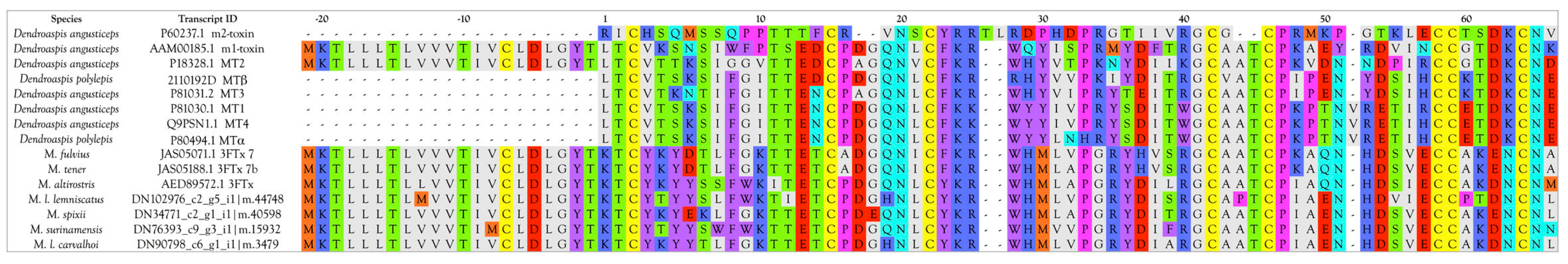
Muscarinic Toxins (MTx)
3FTxs that Impact Glutamate Receptors
Cardiotoxins/Cytotoxins/β-Cardiotoxin
Fasciculins
Calcicludine- or Calciseptine-like Toxins
2.2.4. 5′-Nucleotidase (5NUC)
2.2.5. Acetylcholinesterase (AChE)
2.2.6. Aminopeptidases A and N (APA and APN)
2.2.7. C-Type Lectin-like Proteins (CTLs)
2.2.8. Cysteine-Rich Secretory Proteins (CRISPs)
2.2.9. Cysteine-Rich Secretory Proteins with EGF Domains (CRISP-EGF)
2.2.10. Cystatin (CYST)
2.2.11. Dipeptidyl Peptidase IV (DPP IV)
2.2.12. Galactose-Binding Lectins (GBL)
2.2.13. Guanylyl Cyclase (QC)
2.2.14. Hyaluronidase (HYAL)
2.2.15. Kunitz Serine Protease Inhibitors (KSPI)
2.2.16. L-Amino Acid Oxidase (LAO)
2.2.17. Metalloproteases, SVMP Type III (P-III)
2.2.18. Natriuretic Peptides (NP)
2.2.19. Nerve Growth Factor (NGF)
2.2.20. Nociceptive Toxins (NOCI)
2.2.21. Phosphodiesterase (PDE)
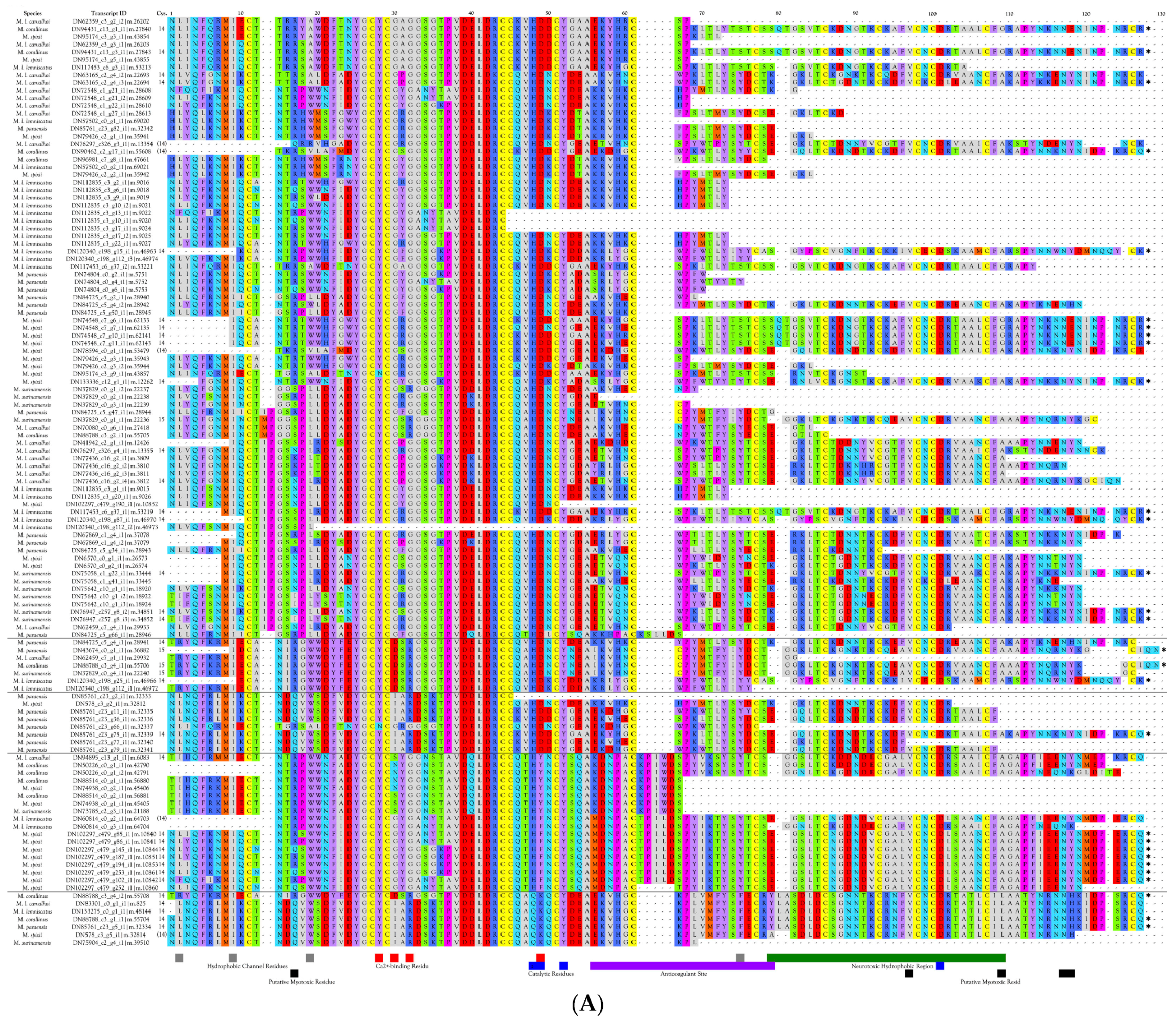
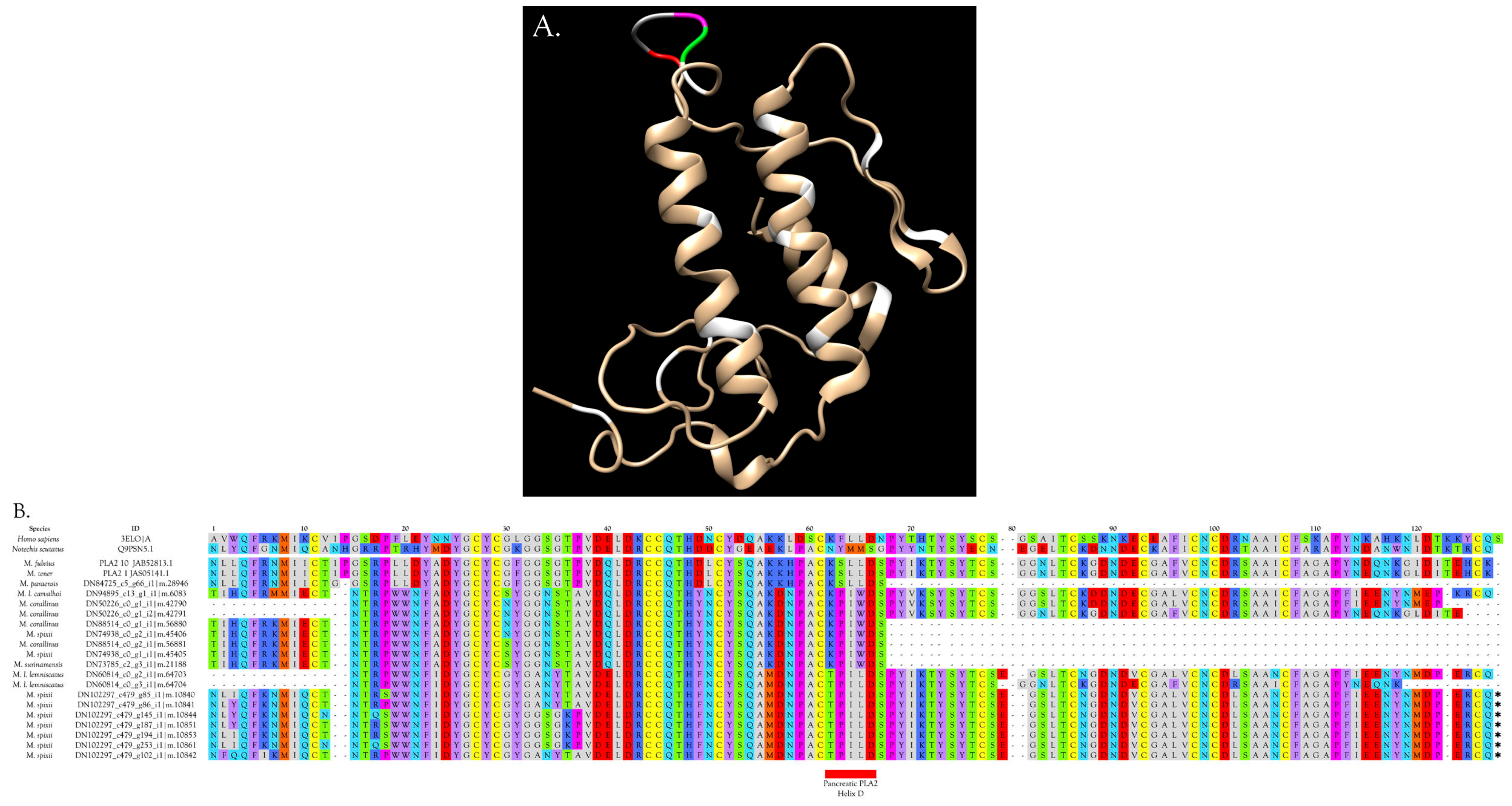
2.2.22. Phospholipases A2 (PLA2)
Overview and Diversity of PLA2s
Myotoxic PLA2s
Hemorrhagic PLA2s
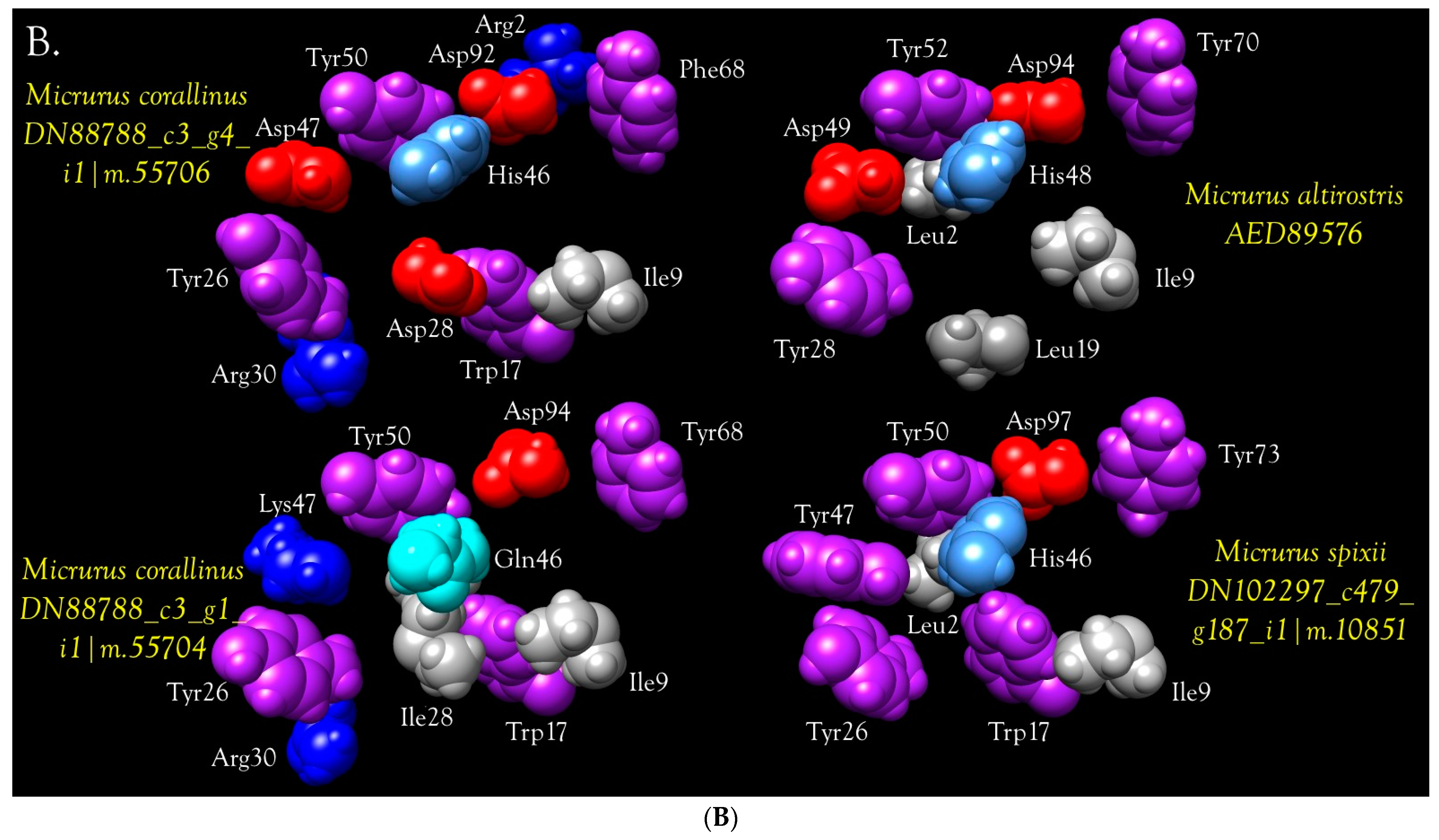
Neurotoxic PLA2s
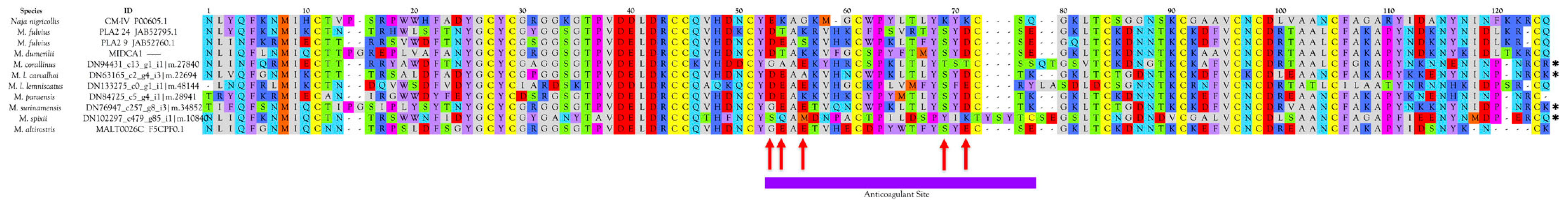
Anticoagulant PLA2s
- negatively charged Glu-54 (53) is replaced by neutral residues;
- positively charged Lys-55 (54) is replaced by negatively charged Glu;
- uncharged Gly-57 (56) is replaced by negatively charged Glu;
- positively charged Lys-75 (69) is replaced by Ser or Thr;
- positively charged Lys-77 (71) is replaced by negatively charged Glu or Asp
- Half of micrurine PLA2s (49.7%) have Asp in position 54 (position 53 in Figure 14), like CM-IV. Gly or Ala occupy this position in 37.8%, while 9.8% have a hydroxylated amino acid, and 2.6% have Asn. See Figure 12, Figure 14, and Figure S22 for all PLA2s;
- No micrurine PLA2s have Lys in position 53 (position 54 in Figure 14). All have Glu (41.8%), Gln (8.9%), Asp (6.8%), or Ala (13.1%), Thr (29.5%), like the weak anticoagulants;
- Most Micrurus PLA2s (53.2%) have Glu in position 57 instead of Gly (Figure 14). 30% have Lys; 4.2% have Ser, and 0.4% have Tyr. An additional 11% have an aliphatic/hydrophobic residue (Ile, Ala, Met, or Val);
- No micrurine PLA2s have Lys in position 75 (position 69 in Figure 14). Overwhelmingly they have Ser (80.4%). An additional 14.2% have Thr and 5.4% have Ile;
- Only 5% of micrurine PLA2s have Lys in position 77 (71 in Figure 14), whereas 50% have Asp. Other substitutions include Thr (27.7%), Tyr (4.0%), Glu (10.4%), and Asn (3.0%).
Pro-Inflammatory PLA2s
2.2.23. Phospholipase B (PLB)
2.2.24. Ribonuclease (RNAse A)
2.2.25. Serine Proteases (SP)
2.2.26. Serine Protease Inhibitors (Serpins)
2.2.27. Vascular Endothelial Growth Factors (VEGF)
2.2.28. Vespryns (VESP)
2.2.29. Waprins (WAPR)
2.2.30. Phylogenetic Conclusions
3. Conclusions
4. Materials and Methods
4.1. Collection of Snakes and Venom Samples
4.2. Collection of Tissue Samples
4.3. Transcriptomics
4.3.1. Removal of Venom Glands
4.3.2. Isolation of Total mRNA from Venom Glands and Muscle
4.3.3. mRNA Isolation and First and Second Strand cDNA Synthesis
4.3.4. Library Sequencing
4.4. Proteomics
4.4.1. Protein Sequencing by Liquid Chromatography-Mass Spectrometry
4.4.2. Protein Identification
4.4.3. Sequence and Structural Analyses
4.5. Bioinformatics
Transcriptome Assembly and Quantitation
4.6. Phylogenetics
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Data Accessibility
Appendix A
Appendix A.1. First Strand cDNA Synthesis
Appendix A.2. Second Strand cDNA Synthesis
Appendix A.3. Size Selection of the Pooled cDNA Libraries
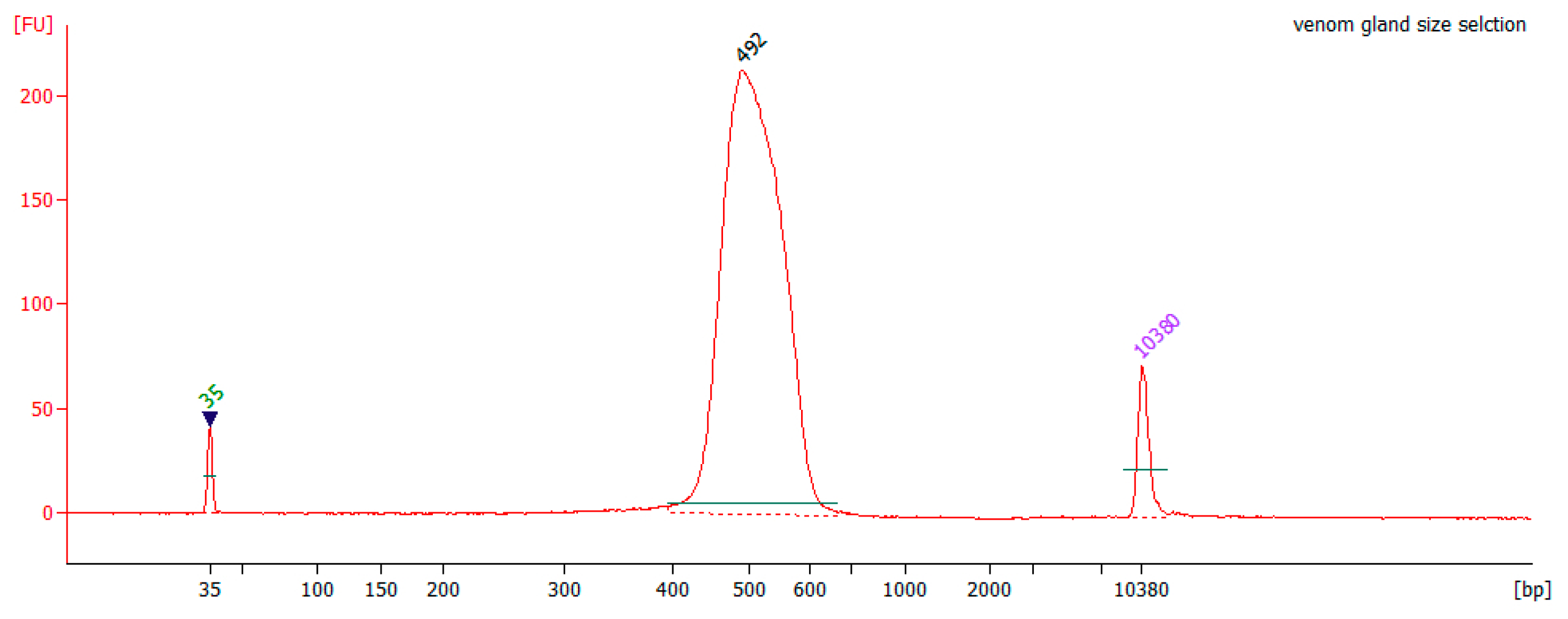
Appendix A.4. Reduction and Alkylation of Snake Venoms Prior to Enzymatic Digestion
Appendix A.5. Tryptic Hydrolyses
Appendix A.6. Chymotryptic Hydrolyses
Appendix A.7. Formic Acid Hydrolyses
Appendix A.8. Digestion and Extraction of Peptides from SDS PAGE Gels
References
- Margres, M.J.; Aronow, K.; Loyacano, J.; Rokyta, D.R. The venom-gland transcriptome of the eastern coral snake (Micrurus fulvius) reveals high venom complexity in the intragenomic evolution of venoms. BMC Genom. 2013, 14, 531. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Margres, M.J.; Calvin, K. Post-transcriptional Mechanisms Contribute Little to Phenotypic Variation in Snake Venoms. G3 2015, 5, 2375–2382. [Google Scholar] [CrossRef] [PubMed]
- Cunha, O.; Nascimento, F.P. Ofídios da Amazônia XIV—As espécies de Micrurus, Bothrops, Lachesis e Crotalus do sul do Pará e oeste do Maranhão, incluindo áreas de cerrado deste estado (Ophidia: Elapidae e Viperidae). Bol. Mus. Para. Emílio Goeldi Zool. 1982, 112, 1–58. [Google Scholar]
- Da Silva, N.J., Jr. Novas ocorrências de Micrurus braziliensis Roze, 1967 (Serpentes: Elapidae) em áreas de tensão ambiental no Centro-Oeste Brazileiro. Estudos 2007, 34, 931–956. [Google Scholar]
- Rodrigues de Almeida, P.C.; Costa Prudente, A.L.; Curcio, F.F.; Urbano Rodrigues, M.T. Biologia e História Natural. In As Cobras Corais do Brasil: Biologia, Taxonomia, Veneno e Envenenamentos; Silva, N.J., Jr., Ed.; Editora PUC Goiás: Goiânia, Brazil, 2016. [Google Scholar]
- Da Silva, N.J., Jr. Capítulo 3: Características Morfológicas das Cobras-Corais do Novo Mundo. In As Cobras-Corais do Brazil: Biologia, Taxonomia, Venenos e Envenenamentos; da Silva, N.J., Jr., Ed.; Editora PUC Goiás: Goiânia, Brazil, 2016. [Google Scholar]
- Da Silva, N.J., Jr.; Pessoa, H.L.R. Micrurus surinamensis (Surinam Coralsnake) geographic distribution. Herpetol. Rev. 2008, 39, 372. [Google Scholar]
- Suto, K.; Yamazaki, Y.; Morita, T.; Mizuno, H. Crystal structures of novel vascular endothelial growth factors (VEGF) from snake venoms: Insight into selective VEGF binding to kinase insert domain-containing receptor but not to fms-like tyrosine kinase-1. J. Biol. Chem. 2005, 280, 2126–2131. [Google Scholar] [CrossRef] [PubMed]
- Gartner, T.K.; Ogilvie, M.L. Isolation and characterization of three Ca2+-dependent beta-galactoside-specific lectins from snake venoms. Biochem. J. 1984, 224, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; Watanabe, Y.; Villar-Briones, A.; Roy, M.C.; Terada, K.; Mikheyev, A.S. Quantitative high-throughput profiling of snake venom gland transcriptomes and proteomes (Ovophis okinavensis and Protobothrops flavoviridis). BMC Genom. 2013, 14, 790. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; Aggarwal, S.; Villar-Briones, A.; Tin, M.M.-Y.; Terada, K.; Mikheyev, A.S. Snake venoms are integrated systems, but abundant venom proteins evolve more rapidly. BMC Genom. 2015, 2015, 647. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; da Silva, N.J., Jr. Comparative enzymatic composition of Brazilian coral snake (Micrurus) venoms. Comp. Biochem. Physiol. B 1991, 99, 287–294. [Google Scholar] [CrossRef]
- Perez-Polo, J.R.; Bomar, H.; Beck, C.; Hall, K. Nerve growth factor from Crotalus adamanteus snake venom. J. Biol. Chem. 1978, 253, 6140–6148. [Google Scholar] [PubMed]
- Siigur, E.; Neuman, T.; Järve, V.; Tara, A.; Siigur, J. Isolation and characterization of nerve growth factor from Vipera lebetina (snake) venom. Comp. Biochem. Physiol. B 1985, 81, 211–215. [Google Scholar] [CrossRef]
- Kostiza, T.; Meier, J. Nerve growth factors from snake venoms: Chemical properties, mode of action and biological significance. Toxicon 1996, 34, 787–806. [Google Scholar] [CrossRef]
- Silva, N.J., Jr.; Griffin, P.R.; Aird, S.D. Primary structure of a short postsynaptic neurotoxin from the venom of Micrurus surinamensis surinamensis. Toxicon 1993, 31, 108. [Google Scholar]
- Olamendi-Portugal, T.; Batista, C.V.F.; Restano-Cassulini, R.; Pando, V.; Villa-Hernandez, O.; Zavaleta-Martínez-Vargas, A.; Salas-Arruz, M.C.; Rodríguez de la Vega, R.C.; Becerril, B.; Possani, L.D. Proteomic analysis of the venom from the fish eating coral snake Micrurus surinamensis: Novel toxins, their function and phylogeny. Proteomics 2008, 8, 1919–1932. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Wootton, J.C.; Gertz, E.M.; Agarwala, R.; Morgulis, A.; Schäffer, A.A.; Yu, Y.-K. Protein database searches using compositionally adjusted substitution matrices. FEBS J. 2005, 272, 5101–5109. [Google Scholar] [CrossRef] [PubMed]
- Samejima, Y.; Aoki-Tomomatsu, Y.; Yanagisawa, M.; Mebs, D. Amino acid sequence of two neurotoxins from the venom of the Egyptian black snake (Walterinnesia aegyptia). Toxicon 1997, 35, 151–157. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; Womble, G.C.; Yates, J.R., 3rd; Griffin, P.R. Primary structure of γ-bungarotoxin, a new postsynaptic neurotoxin from venom of Bungarus multicinctus. Toxicon 1999, 37, 609–625. [Google Scholar] [CrossRef]
- Chang, L.S.; Lin, J. cDNA sequence analysis of a novel neurotoxin homolog from Taiwan banded krait. Biochem. Mol. Biol. Int. 1997, 43, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Nirthanan, S.; Gopalakrishnakone, P.; Gwee, M.C.E.; Khoo, H.E.; Kini, R.M. Non-conventional toxins from Elapid venoms. Toxicon 2003, 41, 397–407. [Google Scholar] [CrossRef]
- Utkin, Y.N.; Kukhtina, V.V.; Kryukova, E.V.; Chiodini, F.; Bertrand, D.; Methfessel, C.; Tsetlin, V.I. “Weak toxin” from Naja kaouthia is a nontoxic antagonist of alpha 7 and muscle-type nicotinic acetylcholine receptors. J. Biol. Chem. 2001, 276, 15810–15815. [Google Scholar] [CrossRef] [PubMed]
- Rzhevskiĭ, D.I.; Murashev, A.N.; Kukhtina, V.V.; Tsetlin, V.I.; Utkin, I.N. The weak neurotoxin from Naja kaouthia Cobra venom decreases the arterial blood pressure in rats. Bioorg. Khim 2001, 27, 221–223. [Google Scholar] [PubMed]
- Servent, D.; Winckler-Dietrich, V.; Hu, H.-Y.; Kessler, P.; Drevet, P.; Bertrand, D.; Ménez, A. Only snake curaremimetic toxins with a fifth disulfide bond have high affinity for the neuronal α7 nicotinic receptor. J. Biol. Chem. 1997, 272, 24279–24286. [Google Scholar] [CrossRef] [PubMed]
- Rosso, J.-P.; Schwarz, J.R.; Diaz-Bustamante, M.; Céard, B.; Gutiérrez, J.M.; Kneussel, M.; Pongs, O.; Bosmans, F.; Bougis, P.E. MmTX1 and MmTX2 from coral snake venom potently modulate GABAA receptor activity. Proc. Natl. Acad. Sci. USA 2015, 112, E891–E900. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.C.; Yang, H.J.; Chiu, R.H. The position of disulfide bonds in cobrotoxin. Biochim. Biophys. Acta 1970, 214, 355–363. [Google Scholar] [CrossRef]
- Endo, Y.; Sato, S.; Ishii, S.; Tamiya, N. The disulphide bonds of erabutoxin a, a neurotoxic protein of a sea-snake (Laticauda semifasciata) venom. Biochem. J. 1971, 122, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Fryklund, L.; Eaker, D. Complete covalent structure of a cardiotoxin from the venom of Naja nigricollis (African black-necked spitting cobra). Biochemistry 1975, 14, 2865–2871. [Google Scholar] [CrossRef] [PubMed]
- Dörje, F.; Wess, J.; Lambrecht, G.; Tacke, R.; Mutschler, E.; Brann, M.R. Antagonist binding profiles of five cloned human muscarinic receptor subtypes. J. Pharmacol. Exp. Ther. 1991, 256, 727–733. [Google Scholar] [PubMed]
- Max, S.I.; Liang, J.S.; Potter, L.T. Purification and properties of m1-toxin, a specific antagonist of m1 muscarinic receptors. J. Neurosci. 1993, 13, 4293–4300. [Google Scholar] [PubMed]
- Jerusalinsky, D.; Kornisiuk, E.; Alfaro, P.; Quillfeldt, J.; Alonso, M.; Verde, E.R.; Cerveñansky, C.; Harvey, A. Muscarinic toxin selective for m4 receptors impairs memory in the rat. Neuroreport 1998, 9, 1407–1411. [Google Scholar] [CrossRef] [PubMed]
- Bradley, K.N. Muscarinic toxins from the green mamba. Pharmacol. Ther. 2000, 85, 87–109. [Google Scholar] [CrossRef]
- Jolkkonen, M.; Oras, A.; Toomela, T.; Karlsson, E.; Järv, J.; Åkerman, K.E.O. Kinetic evidence for different mechanisms of interaction of black mamba toxins MTα and MTβ with muscarinic receptors. Toxicon 2001, 39, 377–382. [Google Scholar] [CrossRef]
- Adem, A.; Asblom, A.; Johansson, G.; Mbugua, P.M.; Karlsson, E. Toxins from the venom of the green mamba Dendroaspis angusticeps that inhibit the binding of quinuclidinyl benzilate to muscarinic acetylcholine receptors. Biochim. Biophys. Acta 1988, 968, 340–345. [Google Scholar] [CrossRef]
- Ducancel, F.; Rowan, E.G.; Cassar, E.; Harvey, A.L.; Ménez, A.; Boulain, J.C. Amino acid sequence of a muscarinic toxin deduced from the cDNA nucleotide sequence. Toxicon 1991, 29, 516–520. [Google Scholar] [CrossRef]
- Karlsson, E.; Risinger, C.; Jolkkonen, M.; Wernstedt, C.; Adem, A. Amino acid sequence of a snake venom toxin that binds to the muscarinic acetylcholine receptor. Toxicon 1991, 29, 521–526. [Google Scholar] [CrossRef]
- Karlsson, E.; Jolkkonen, M.; Satyapan, N.; Adem, A.; Kumlin, E.; Hellman, U.; Wernstedt, C. Protein toxins that bind to muscarinic acetylcholine receptors. Ann. N. Y. Acad. Sci. 1994, 710, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Walch, L.; Brink, C.; Norel, X. The muscarinic receptor subtypes in human blood vessels. Therapie 2001, 56, 223–226. [Google Scholar] [PubMed]
- Lamping, K.G.; Wess, J.; Cui, Y.; Nuno, D.W.; Faraci, F.M. Muscarinic (M) receptors in coronary circulation: Gene-Targeted mice define the role of M2 and M3 receptors in response to acetylcholine. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.D. Muscarinic Receptor Agonists and Antagonists: Effects on Cardiovascular Function. In Handbook of Experimental Pharmacology; Springer: Berlin, Germany, 2011; pp. 299–316. [Google Scholar]
- Jolkkonen, M.; Van Giersbergen, P.L.; Hellman, U.; Wernstedt, C.; Oras, A.; Satyapan, N.; Adem, A.; Karlsson, E. Muscarinic toxins from the black mamba Dendroaspis polylepis. Eur. J. Biochem. 1995, 234, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Ryberg, A.T.; Selberg, H.; Soukup, O.; Gradin, K.; Tobin, G. Cholinergic submandibular effects and muscarinic receptor expression in blood vessels of the rat. Arch. Oral Biol. 2008, 53, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D. Ophidian envenomation strategies and the role of purines. Toxicon 2002, 40, 335–393. [Google Scholar] [CrossRef]
- Wangai, J.; Ng’ang’a, J.N.; Mungai, S.; Muthaura, C.N.; Thairu, K.; Telang, B.V. Mechanisms of centrally induced vasodepressor response in cats after lateral ventricular administration of whole venom of Dendroaspis angusticeps-Part I. Agressologie 1982, 23, 301–304. [Google Scholar] [PubMed]
- Medina, A.; Bodick, N.; Goldberger, A.L.; Mac Mahon, M.; Lipsitz, L.A. Effects of central muscarinic-1 receptor stimulation on blood pressure regulation. Hypertension 1997, 29, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Brezenoff, H.E.; Vargas, H.; Xiao, Y.F. Blockade of brain M2 muscarinic receptors lowers blood pressure in spontaneously hypertensive rats. Pharmacology 1988, 36, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Pazos, A.; Wiederhold, K.H.; Palacios, J.M. Central pressor effects induced by muscarinic receptor agonists: Evidence for a predominant role of the M2 receptor subtype. Eur. J. Pharmacol. 1986, 125, 63–70. [Google Scholar] [CrossRef]
- Kubo, T. Cholinergic mechanism and blood pressure regulation in the central nervous system. Brain Res. Bull. 1998, 46, 475–481. [Google Scholar] [CrossRef]
- Ozkutlu, U.; Onat, F.; Aslan, A.N.; Oktay, S. Central muscarinic M2 cholinoceptors involved in cholinergic hypertension. Eur. J. Pharmacol. 1993, 250, 349–354. [Google Scholar] [CrossRef]
- Bruning, T.A.; Hendriks, M.G.; Chang, P.C.; Kuypers, E.A.; van Zwieten, P.A. In vivo characterization of vasodilating muscarinic-receptor subtypes in humans. Circ. Res. 1994, 74, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, M.G.; Pfaffendorf, M.; Van Zwieten, P.A. Characterization of the muscarinic receptor subtype mediating vasodilation in the rat perfused mesenteric vascular bed preparation. J. Auton. Pharmacol. 1992, 12, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Gericke, A.; Sniatecki, J.J.; Mayer, V.G.A.; Goloborodko, E.; Patzak, A.; Wess, J.; Pfeiffer, N. Role of M1, M3, and M5 muscarinic acetylcholine receptors in cholinergic dilation of small arteries studied with gene-targeted mice. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H1602–H1608. [Google Scholar] [CrossRef] [PubMed]
- Höglund, A.U.; Baghdoyan, H.A. M2, M3 and M4, but not M1, muscarinic receptor subtypes are present in rat spinal cord. J. Pharmacol. Exp. Ther. 1997, 281, 470–477. [Google Scholar] [PubMed]
- Yamada, M.; Lamping, K.G.; Duttaroy, A.; Zhang, W.; Cui, Y.; Bymaster, F.P.; McKinzie, D.L.; Felder, C.C.; Deng, C.X.; Faraci, F.M.; et al. Cholinergic dilation of cerebral blood vessels is abolished in M(5) muscarinic acetylcholine receptor knockout mice. Proc. Natl. Acad. Sci. USA 2001, 98, 14096–14101. [Google Scholar] [CrossRef] [PubMed]
- Jolkkonen, M. Muscarinic Toxins from Dendroaspis (mamba) Venoms. Peptides Selective for Subtypes of Acetylcholine Muscarinic Receptors. Ph.D. Thesis, University of Uppsala, Uppsala, Sweden, 1996. [Google Scholar]
- Liang, J.S.; Carsi-Gabrenas, J.; Krajewski, J.L.; McCafferty, J.M.; Purkerson, S.L.; Santiago, M.P.; Strauss, W.L.; Valentine, H.H.; Potter, L.T. Anti-muscarinic toxins from Dendroaspis angusticeps. Toxicon 1996, 34, 1257–1267. [Google Scholar] [CrossRef]
- Carsi, J.M.; Valentine, H.H.; Potter, L.T. m2-toxin: A selective ligand for M2 muscarinic receptors. Mol. Pharmacol. 1999, 56, 933–937. [Google Scholar] [PubMed]
- Jolkkonen, M.; Adem, A.; Hellman, U.; Wernstedt, C.; Karlsson, E. A snake toxin against muscarinic acetylcholine receptors: Amino acid sequence, subtype specificity and effect on guinea-pig ileum. Toxicon 1995, 33, 399–410. [Google Scholar] [CrossRef]
- Jolkkonen, M.; van Giersbergen, P.L.; Hellman, U.; Wernstedt, C.; Karlsson, E. A toxin from the green mamba Dendroaspis angusticeps: Amino acid sequence and selectivity for muscarinic m4 receptors. FEBS Lett. 1994, 352, 91–94. [Google Scholar] [CrossRef]
- Ségalas, I.; Roumestand, C.; Zinn-Justin, S.; Gilquin, B.; Ménez, R.; Ménez, A.; Toma, F. Solution structure of a green mamba toxin that activates muscarinic acetylcholine receptors, as studied by nuclear magnetic resonance and molecular modeling. Biochemistry 1995, 34, 1248–1260. [Google Scholar] [CrossRef] [PubMed]
- Servent, D.; Blanchet, G.; Mourier, G.; Marquer, C.; Marcon, E.; Fruchart-Gaillard, C. Muscarinic toxins. Toxicon 2011, 58, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, D.C.; de Medeiros, W.A.A.; Batista, I.D.F.C.; Pimenta, D.C.; Lebrun, I.; Abdalla, F.M.F.; Sandoval, M.R.L. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011, 89, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Dhein, S.; van Koppen, C.J.; Brodde, O.E. Muscarinic receptors in the mammalian heart. Pharmacol. Res. 2001, 44, 161–182. [Google Scholar] [CrossRef] [PubMed]
- Montandon, A.C. Avaliação da liberação de L-glutamato por sinaptosomas cérebro-corticais de rato pela ação da peçonha da serpente coral Sul-Americana, Micrurus lemniscatus (Linnaeus, 1758), e por algumas de suas fraçOões semi-purificadas. Master’s Thesis, Universidade Federal de Minas Gerais: Belo Horizonte, Minas Gerais, Brazil, 2013. [Google Scholar]
- Freire Donato, M. Peçonha da serpente Micrurus l. lemniscatus (Roze, 1967) Caracterização Parcial das Propriedades Bioquímica e Farmacológicas: Neurotoxicidade e Atividade pré Sináptica de Toxinas Três-Dígitos. Ph.D. Thesis, Federal University of Minas Gerais, Belo Horizonte, Minas Gerais, Brazil, 2014. [Google Scholar]
- Vital Brazil, O. Coral snake venoms: Mode of action and pathophysiology of experimental envenomation. Rev. Inst. Med. Trop. Sao Paulo 1987, 29, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, N.J., Jr.; Griffin, P.R.; Aird, S.D. Comparative chromatography of Brazilian coral snake (Micrurus) venoms. Comp. Biochem. Physiol. B 1991, 100, 117–126. [Google Scholar] [CrossRef]
- Rajagopalan, N.; Pung, Y.F.; Zhu, Y.Z.; Wong, P.T.H.; Kumar, P.P.; Kini, R.M. Beta-cardiotoxin: A new three-finger toxin from Ophiophagus hannah (king cobra) venom with beta-blocker activity. FASEB J. 2007, 21, 3685–3695. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Rejent, T.A.; Elliott, W.B. Anticholinesterase activity of elapid venoms. Toxicon 1973, 11, 131–138. [Google Scholar] [CrossRef]
- De Weille, J.R.; Schweitz, H.; Maes, P.; Tartar, A.; Lazdunski, M. Calciseptine, a peptide isolated from black mamba venom, is a specific blocker of the L-type calcium channel. Proc. Natl. Acad. Sci. USA 1991, 88, 2437–2440. [Google Scholar] [CrossRef] [PubMed]
- Schweitz, H.; Heurteaux, C.; Bois, P.; Moinier, D.; Romey, G.; Lazdunski, M. Calcicludine, a venom peptide of the Kunitz-type protease inhibitor family, is a potent blocker of high-threshold Ca2+ channels with a high affinity for L-type channels in cerebellar granule neurons. Proc. Natl. Acad. Sci. USA 1994, 91, 878–882. [Google Scholar] [CrossRef] [PubMed]
- Raina, R.K.; Ng’ang’a, J.N.; Njoroge, D.K.; Telang, B.V. Further studies on the mechanism of vasodepressor response in cats after intravenous administration of venom from the snake Dendroaspis jamesoni (Jameson’s mamba). Toxicon 1977, 15, 561–570. [Google Scholar] [CrossRef]
- Wangai, J.; Ng’ang’a, J.N.; Njoroge, D.; Thairu, K.; Telang, B.V. Centrally acting hypotensive fraction in the venom of Dendroaspis angusticeps. Experientia 1978, 34, 874–876. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L.; Karlsson, E. Dendrotoxin from the venom of the green mamba, Dendroaspis angusticeps. A neurotoxin that enhances acetylcholine release at neuromuscular junction. Naunyn-Schmiedeberg’s Arch. Pharmacol. 1980, 312, 1–6. [Google Scholar] [CrossRef]
- Wangai, J.; Thairu, K.; Bharaj, B.S.; Telang, B.V. Identification and isolation of three acetylcholinesterase inactivating fractions in the venom of Dendroaspis angusticeps. Acta Physiol. Acad. Sci. Hung. 1982, 60, 83–88. [Google Scholar] [PubMed]
- Rodríguez-Ithurralde, D.; Silveira, R.; Barbeito, L.; Dajas, F. Fasciculin, a powerful anticholinesterase polypeptide from Dendroaspis angusticeps venom. Neurochem. Int. 1983, 5, 267–274. [Google Scholar] [CrossRef]
- Karlsson, E.; Mbugua, P.M.; Rodriguez-Ithurralde, D. Fasciculins, anticholinesterase toxins from the venom of the green mamba Dendroaspis angusticeps. J. Physiol. Paris 1984, 79, 232–240. [Google Scholar] [PubMed]
- Orawski, A.T.; Susz, J.P.; Simmons, W.H. Metabolism of bradykinin by multiple coexisting membrane-bound peptidases in lung: Techniques for investigating the role of each peptidase using specific inhibitors. Adv. Exp. Med. Biol. 1989, 247, 355–364. [Google Scholar]
- Kokkonen, J.O.; Kuoppala, A.; Saarinen, J.; Lindstedt, K.A.; Kovanen, P.T. Kallidin- and Bradykinin-Degrading Pathways in Human Heart : Degradation of Kallidin by Aminopeptidase M Like Activity and Bradykinin by Neutral Endopeptidase. Circulation 1999, 99, 1984–1990. [Google Scholar] [CrossRef] [PubMed]
- Bausback, H.H.; Churchill, L.; Ward, P.E. Angiotensin metabolism by cerebral microvascular aminopeptidase A. Biochem. Pharmacol. 1988, 37, 155–160. [Google Scholar] [CrossRef]
- Ward, P.E.; Benter, I.F.; Dick, L.; Wilk, S. Metabolism of vasoactive peptides by plasma and purified renal aminopeptidase M. Biochem. Pharmacol. 1990, 40, 1725–1732. [Google Scholar] [CrossRef]
- Holzer, P.; Guth, P.H. Neuropeptide control of rat gastric mucosal blood flow. Increase by calcitonin gene-related peptide and vasoactive intestinal polypeptide, but not substance P and neurokinin A. Circ. Res. 1991, 68, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Figini, M.; Emanueli, C.; Geppetti, P.; Grady, E.F.; Gerard, N.P.; Ansell, J.; Payan, D.G.; Gerard, C.; Bunnett, N. The control of microvascular permeability and blood pressure by neutral endopeptidase. Nat. Med. 1997, 3, 904–907. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Murayama, N.; Fujita, Y.; Yanoshita, R. Characterization and cDNA cloning of aminopeptidase A from the venom of Gloydius blomhoffi brevicaudus. Toxicon 2007, 49, 1172–1181. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Wüster, W. Assembling an arsenal: Origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences. Mol. Biol. Evol. 2004, 21, 870–883. [Google Scholar] [CrossRef] [PubMed]
- Drickamer, K. C-type lectin-like domains. Curr. Opin. Struct. Biol. 1999, 9, 585–590. [Google Scholar] [CrossRef]
- Morita, T. C-type lectin-related proteins from snake venoms. Curr. Drug Targets Cardiovasc. Haematol. Disord. 2004, 4, 357–373. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Clemetson, J.M.; Clemetson, K.J. Snake venoms and hemostasis. J. Thromb. Haemost. 2005, 3, 1791–1799. [Google Scholar] [CrossRef] [PubMed]
- Zha, H.G.; Lee, W.H.; Zhang, Y. Cloning of cDNAs encoding C-type lectins from Elapidae snakes Bungarus fasciatus and Bungarus multicinctus. Toxicon 2001, 39, 1887–1892. [Google Scholar] [CrossRef]
- Earl, S.T.H.; Robson, J.; Trabi, M.; de Jersey, J.; Masci, P.P.; Lavin, M.F. Characterisation of a mannose-binding C-type lectin from Oxyuranus scutellatus snake venom. Biochimie 2011, 93, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Corrêa-Netto, C.; Junqueira-de-Azevedo Ide, L.; Silva, D.A.; Ho, P.L.; Leitao-de-Araujo, M.; Alves, M.L.; Sanz, L.; Foguel, D.; Zingali, R.B.; Calvete, J.J. Snake venomics and venom gland transcriptomic analysis of Brazilian coral snakes, Micrurus altirostris and M. corallinus. J. Proteom. 2011, 74, 1795–1809. [Google Scholar] [CrossRef] [PubMed]
- Drickamer, K. Engineering galactose-binding activity into a C-type mannose-binding protein. Nature 1992, 360, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Morita, T. Structure and function of snake venom cysteine-rich secretory proteins. Toxicon 2004, 44, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Koike, H.; Sugiyama, Y.; Motoyoshi, K.; Wada, T.; Hishinuma, S.; Mita, M.; Morita, T. Cloning and characterization of novel snake venom proteins that block smooth muscle contraction. Eur. J. Biochem. 2002, 269, 2708–2715. [Google Scholar] [CrossRef] [PubMed]
- Peichoto, M.E.; Mackessy, S.P.; Teibler, P.; Tavares, F.L.; Burckhardt, P.L.; Breno, M.C.; Acosta, O.; Santoro, M.L. Purification and characterization of a cysteine-rich secretory protein from Philodryas patagoniensis snake venom. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 150, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Lemmon, A.R.; Margres, M.J.; Aronow, K. The venom-gland transcriptome of the eastern diamondback rattlesnake (Crotalus adamanteus). BMC Genom. 2012, 13, 312. [Google Scholar] [CrossRef] [PubMed]
- Argraves, W.S.; Tran, H.; Burgess, W.H.; Dickerson, K. Fibulin is an extracellular matrix and plasma glycoprotein with repeated domain structure. J. Cell Biol. 1990, 111, 3155–3164. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.; Urdea, M.; Quiroga, M.; Sanchez-Pescador, R.; Fong, N.; Selby, M.; Rutter, W.J.; Bell, G.I. Structure of a mouse submaxillary messenger RNA encoding epidermal growth factor and seven related proteins. Science 1983, 221, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Engel, J.; Jürgen, E. EGF-like domains in extracellular matrix proteins: Localized signals for growth and differentiation? FEBS Lett. 1989, 251, 1–7. [Google Scholar] [CrossRef]
- Tran, H.; Tanaka, A.; Litvinovich, S.V.; Medved, L.V.; Haudenschild, C.C.; Argraves, W.S. The interaction of fibulin-1 with fibrinogen. A potential role in hemostasis and thrombosis. J. Biol. Chem. 1995, 270, 19458–19464. [Google Scholar] [PubMed]
- Godyna, S.; Diaz-Ricart, M.; Argraves, W.S. Fibulin-1 mediates platelet adhesion via a bridge of fibrinogen. Blood 1996, 88, 2569–2577. [Google Scholar] [PubMed]
- Aspberg, A.; Adam, S.; Kostka, G.; Timpl, R.; Heinegård, D. Fibulin-1 is a ligand for the C-type lectin domains of aggrecan and versican. J. Biol. Chem. 1999, 274, 20444–20449. [Google Scholar] [CrossRef] [PubMed]
- Eagle, H. The coagulation of blood by snake venoms and its physiologic significance. J. Exp. Med. 1937, 65, 613–639. [Google Scholar] [CrossRef] [PubMed]
- Keppler, D. Towards novel anti-cancer strategies based on cystatin function. Cancer Lett. 2006, 235, 159–176. [Google Scholar] [CrossRef] [PubMed]
- Abrahamson, M. Cystatins. Methods Enzymol. 1994, 244, 685–700. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Kanai-Azuma, M.; Akimoto, Y.; Kawakami, H.; Yanoshita, R. Exosome-like vesicles in Gloydius blomhoffii blomhoffii venom. Toxicon 2008, 51, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, N.J., Jr.; Aird, S.D. Prey specificity, comparative lethality and compositional differences of coral snake venoms. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2001, 128, 425–456. [Google Scholar] [CrossRef]
- Heymann, E.; Mentlein, R. Has dipeptidyl peptidase IV an effect on blood pressure and coagulation? Klin. Wochenschr. 1984, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.A.; Schünke, M.; Tillmann, B. Histochemical visualization of lymphatic capillaries in the rat: A comparison of methods demonstrated at the posterior pharyngeal surface. Arch. Histol. Jpn. 1987, 50, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Wang, L.; Ward, P.E. Dipeptidyl(amino)peptidase IV and aminopeptidase M metabolize circulating substance P in vivo. J. Pharmacol. Exp. Ther. 1992, 260, 1257–1261. [Google Scholar] [PubMed]
- Zukowska-Grojec, Z.; Haass, M.; Bayorh, M.A. Neuropeptide Y and peptide YY mediate nonadrenergic vasoconstriction and modulate sympathetic responses in rats. Regul. Pept. 1986, 15, 99–110. [Google Scholar] [CrossRef]
- Mentlein, R.; Dahms, P.; Grandt, D.; Krüger, R. Proteolytic processing of neuropeptide Y and peptide YY by dipeptidyl peptidase IV. Regul. Pept. 1993, 49, 133–144. [Google Scholar] [CrossRef]
- Aird, S.D. Snake venom dipeptidyl peptidase IV: Taxonomic distribution and quantitative variation. Comp. Biochem. Physiol. B 2008, 150, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Yuko, O.; Yoshie, M.; Nobuhiro, M.; Ryohei, Y. Characterization and cDNA cloning of dipeptidyl peptidase IV from the venom of Gloydius blomhoffi brevicaudus. Comp. Biochem. Physiol. B 2006, 145, 35–42. [Google Scholar] [CrossRef] [PubMed]
- St. Pierre, L.; Birrell, G.W.; Earl, S.T.; Wallis, T.P.; Gorman, J.J.; de Jersey, J.; Masci, P.P.; Lavin, M.F. Diversity of toxic components from the venom of the evolutionarily distinct black whip snake, Demansia vestigiata. J. Proteome Res. 2007, 6, 3093–3107. [Google Scholar] [CrossRef] [PubMed]
- Gartner, T.K.; Stocker, K.; Williams, D.C. Thrombolectin: A lectin isolated from Bothrops atrox venom. FEBS Lett. 1980, 117, 13–16. [Google Scholar] [CrossRef]
- Ogilvie, M.L.; Dockter, M.E.; Wenz, L.; Gartner, T.K. Isolation and characterization of lactose-binding lectins from the venoms of the snakes Lachesis muta and Dendroaspis jamesonii. J. Biochem. 1986, 100, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Mastro, A.M.; Hurley, D.J.; Winning, R.K.; Filipowski, R.; Ogilvie, M.L.; Gartner, T.K. Mitogenic activity of snake venom lectins. Cell Tissue Kinet. 1986, 19, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Rojas, G.; Gutiérrez, J.M.; Ramírez, G. Isolation of a galactose-binding lectin from the venom of the snake Bothrops godmani (Godmann’s pit viper). Toxicon 1990, 28, 75–81. [Google Scholar] [CrossRef]
- Aird, S.D.; Kaiser, I.I.; Lewis, R.V.; Kruggel, W.G. Rattlesnake presynaptic neurotoxins: Primary structure and evolutionary origin of the acidic subunit. Biochemistry 1985, 24, 7054–7058. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; Yates, J.R., 3rd; Martino, P.A.; Shabanowitz, J.; Hunt, D.F.; Kaiser, I.I. The amino acid sequence of the acidic subunit B-chain of crotoxin. Biochim. Biophys. Acta 1990, 1040, 217–224. [Google Scholar] [CrossRef]
- Tu, A.T.; Hendon, R.R. Characterization of lizard venom hyaluronidase and evidence for its action as a spreading factor. Comp. Biochem. Physiol. B 1983, 76, 377–383. [Google Scholar] [CrossRef]
- Girish, K.S.; Mohanakumari, H.P.; Nagaraju, S.; Vishwanath, B.S.; Kemparaju, K. Hyaluronidase and protease activities from Indian snake venoms: Neutralization by Mimosa pudica root extract. Fitoterapia 2004, 75, 378–380. [Google Scholar] [CrossRef] [PubMed]
- Zupunski, V.; Kordis, D.; Gubensek, F. Adaptive evolution in the snake venom Kunitz/BPTI protein family. FEBS Lett. 2003, 547, 131–136. [Google Scholar] [CrossRef]
- Sigle, R.; Hackett, M.; Aird, S.D. Primary structures of four trypsin inhibitor E homologs from venom of Dendroaspis angusticeps: Structure-function comparisons with other dendrotoxin homologs. Toxicon 2002, 40, 297–308. [Google Scholar] [CrossRef]
- Tytgat, J.; Jan, T.; Isabel, V.; Chris, U.; Van Beeumen, J. New polypeptide components purified from mamba venom. FEBS Lett. 2001, 491, 217–221. [Google Scholar] [CrossRef]
- Strydom, D.J.; Joubert, F.J. The amino acid sequence of a weak trypsin inhibitor B from Dendroaspis polylepis polylepis (black mamba) venom. Hoppe Seylers Z. Physiol. Chem. 1981, 362, 1377–1384. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Twenty years of dendrotoxins. Toxicon 2001, 39, 15–26. [Google Scholar] [CrossRef]
- Dufton, M.J. Proteinase inhibitors and dendrotoxins. Sequence classification, structural prediction and structure/activity. Eur. J. Biochem. 1985, 153, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.W.; Serrano, S.M.T. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon 2005, 45, 969–985. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.W.; Serrano, S.M.T. Insights into and speculations about snake venom metalloproteinase (SVMP) synthesis, folding and disulfide bond formation and their contribution to venom complexity. FEBS J. 2008, 275, 3016–3030. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Takeya, H.; Iwanaga, S. Snake venom metalloproteinases: Structure, function and relevance to the mammalian ADAM/ADAMTS family proteins. Biochim. Biophys. Acta 2012, 1824, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, J.; Zhang, X.; Ren, Y.; Wang, N.; Zhao, K.; Chen, X.; Zhao, C.; Li, X.; Shao, J.; et al. Proteomic characterization of two snake venoms: Naja naja atra and Agkistrodon halys. Biochem. J. 2004, 384, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.-H.; Goh, K.-S.; Davamani, F.; Wu, P.-L.; Huang, Y.-W.; Jeyakanthan, J.; Wu, W.-G.; Chen, C.-J. Structures of two elapid snake venom metalloproteases with distinct activities highlight the disulfide patterns in the D domain of ADAMalysin family proteins. J. Struct. Biol. 2010, 169, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Sudoh, T.; Minamino, N.; Kangawa, K.; Matsuo, H. C-type natriuretic peptide (CNP): A new member of natriuretic peptide family identified in porcine brain. Biochem. Biophys. Res. Commun. 1990, 168, 863–870. [Google Scholar] [CrossRef]
- Schweitz, H.; Vigne, P.; Moinier, D.; Frelin, C.; Lazdunski, M. A new member of the natriuretic peptide family is present in the venom of the green mamba (Dendroaspis angusticeps). J. Biol. Chem. 1992, 267, 13928–13932. [Google Scholar] [PubMed]
- Murayama, N.; Hayashi, M.A.; Ohi, H.; Ferreira, L.A.; Hermann, V.V.; Saito, H.; Fujita, Y.; Higuchi, S.; Fernandes, B.L.; Yamane, T.; et al. Cloning and sequence analysis of a Bothrops jararaca cDNA encoding a precursor of seven bradykinin-potentiating peptides and a C-type natriuretic peptide. Proc. Natl. Acad. Sci. USA 1997, 94, 1189–1193. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.L.; Soares, M.B.; Maack, T.; Gimenez, I.; Puorto, G.; Furtado, M.F.; Raw, I. Cloning of an unusual natriuretic peptide from the South American coral snake Micrurus corallinus. Eur. J. Biochem. 1997, 250, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Wickramaratana, J.C.; Lemme, S.; Beuve, A.; Garbers, D.; Hodgson, W.C.; Alewood, P. Novel natriuretic peptides from the venom of the inland taipan (Oxyuranus microlepidotus): Isolation, chemical and biological characterisation. Biochem. Biophys. Res. Commun. 2005, 327, 1011–1015. [Google Scholar] [CrossRef] [PubMed]
- St Pierre, L.; Flight, S.; Masci, P.P.; Hanchard, K.J.; Lewis, R.J.; Alewood, P.F.; de Jersey, J.; Lavin, M.F. Cloning and characterisation of natriuretic peptides from the venom glands of Australian elapids. Biochimie 2006, 88, 1923–1931. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, S.; Murayama, N.; Saguchi, K.-I.; Ohi, H.; Fujita, Y.; da Silva, N.J., Jr.; de Siqueira, R.J.B.; Lahlou, S.; Aird, S.D. A novel peptide from the ACEI/BPP-CNP precursor in the venom of Crotalus durissus collilineatus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 144, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Kostiza, T.; Dahinden, C.A.; Rihs, S.; Otten, U.; Meier, J. Nerve growth factor from the venom of the Chinese cobra Naja naja atra: Purification and description of non-neuronal activities. Toxicon 1995, 33, 1249–1261. [Google Scholar] [CrossRef]
- Guo, L.Y.; Zhu, J.F.; Wu, X.F.; Zhou, Y.C. Cloning of a cDNA encoding a nerve growth factor precursor from the Agkistrodon halys Pallas. Toxicon 1999, 37, 465–470. [Google Scholar] [CrossRef]
- Orenstein, N.S.; Dvorak, H.F.; Blanchard, M.H.; Young, M. Nerve growth factor: A protease that can activate plasminogen. Proc. Natl. Acad. Sci. USA 1978, 75, 5497–5500. [Google Scholar] [CrossRef] [PubMed]
- Young, M. Proteolytic activity of nerve growth factor: A case of autocatalytic activation. Biochemistry 1979, 18, 3050–3055. [Google Scholar] [CrossRef] [PubMed]
- Bohlen, C.J.; Chesler, A.T.; Sharif-Naeini, R.; Medzihradszky, K.F.; Zhou, S.; King, D.; Sanchez, E.E.; Burlingame, A.L.; Basbaum, A.I.; Julius, D. A heteromeric Texas coral snake toxin targets acid-sensing ion channels to produce pain. Nature 2011, 479, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Kocholaty, W.F.; Ledford, E.B.; Daly, J.G.; Billings, T.A. Toxicity and some enzymatic properties and activities in the venoms of Crotalidae, Elapidae and Viperidae. Toxicon 1971, 9, 131–138. [Google Scholar] [CrossRef]
- Ramsey, H.W.; Snyder, G.K.; Kitchen, H.; Taylor, W.J. Fractionation of coral snake venom. Toxicon 1972, 10, 67–72. [Google Scholar] [CrossRef]
- Possani, L.D.; Alagon, A.C.; Fletcher, P.L., Jr.; Varela, M.J.; Julia, J.Z. Purification and characterization of a phospholipase A2 from the venom of the coral snake, Micrurus fulvius microgalbineus (Brown and Smith). Biochem. J. 1979, 179, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Vergara, I.; Pedraza-Escalona, M.; Paniagua, D.; Restano-Cassulini, R.; Zamudio, F.; Batista, C.V.F.; Possani, L.D.; Alagón, A. Eastern coral snake Micrurus fulvius venom toxicity in mice is mainly determined by neurotoxic phospholipases A2. J. Proteom. 2014, 105, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, J.; Alape-Giron, A.; Angulo, Y.; Sanz, L.; Gutierrez, J.M.; Calvete, J.J.; Lomonte, B. Venomic and antivenomic analyses of the Central American coral snake, Micrurus nigrocinctus (Elapidae). J. Proteome Res. 2011, 10, 1816–1827. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, A.L.; Marcussi, S.; Silveira, L.B.; Borja-Oliveira, C.R.; Rodrigues-Simioni, L.; Amara, S.; Stabeli, R.G.; Giglio, J.R.; Arantes, E.C.; Soares, A.M. Biological and enzymatic activities of Micrurus sp. (Coral) snake venoms. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2005, 140, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Maraganore, J.M.; Merutka, G.; Cho, W.; Welches, W.; Kézdy, F.J.; Heinrikson, R.L. A new class of phospholipases A2 with lysine in place of aspartate 49. Functional consequences for calcium and substrate binding. J. Biol. Chem. 1984, 259, 13839–13843. [Google Scholar] [PubMed]
- Lomonte, B.; Gutiérrez, J.M. A new muscle damaging toxin, myotoxin II, from the venom of the snake Bothrops asper (terciopelo). Toxicon 1989, 27, 725–733. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Lomonte, B.; Cerdas, L. Isolation and partial characterization of a myotoxin from the venom of the snake Bothrops nummifer. Toxicon 1986, 24, 885–894. [Google Scholar] [CrossRef]
- Montecucco, C.; Gutiérrez, J.M.; Lomonte, B. Cellular pathology induced by snake venom phospholipase A2 myotoxins and neurotoxins: Common aspects of their mechanisms of action. Cell. Mol. Life Sci. 2008, 65, 2897–2912. [Google Scholar] [CrossRef] [PubMed]
- Kopper, R.A.; Harper, G.R.; Zimmerman, S.; Hook, J. Comparison of total protein and phospholipase A(2) levels in individual coralsnake venoms. Toxicon 2013, 76, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Weis, R.; McIsaac, R.J. Cardiovascular and muscular effects of venom from coral snake, Micrurus fulvius. Toxicon 1971, 9, 219–228. [Google Scholar] [CrossRef]
- Maeno, H.; Mitsuhashi, S.; Okonogi, T.; Hoshi, S.; Homma, M. Studies on Habu snake venom. (V). Myolysis caused by phospholipase A in Habu snake venom. Jpn. J. Exp. Med. 1962, 32, 55–64. [Google Scholar] [PubMed]
- Kurashige, S.; Hara, Y.; Kawakami, M.; Mitsuhashi, S. Studies on Habu snake venom. VII. Heat-stable myolytic factor and development of its activity by addition of phospholipase A. Jpn. J. Microbiol. 1966, 10, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.B.; Johnson, M.A.; Karlsson, E. Pathological responses of rat skeletal muscle to a single subcutaneous injection of a toxin isolated from the venom of the Australian tiger snake, Notechis scutatus scutatus. Clin. Exp. Pharmacol. Physiol. 1975, 2, 383–404. [Google Scholar] [CrossRef]
- Kensil, C.R.; Dennis, E.A. Action of cobra venom phospholipase A2 on the gel and liquid crystalline states of dimyristoyl and dipalmitoyl phosphatidylcholine vesicles. J. Biol. Chem. 1979, 254, 5843–5848. [Google Scholar] [PubMed]
- Napias, C.; Heilbronn, E. Phospholipase A2 activity and substrate specificity of snake venom presynaptic toxins. Biochemistry 1980, 19, 1146–1151. [Google Scholar] [CrossRef] [PubMed]
- Radvanyi, F.; Saliou, B.; Bon, C.; Strong, P.N. The interaction between the presynaptic phospholipase neurotoxins beta-bungarotoxin and crotoxin and mixed detergent-phosphatidylcholine micelles. A comparison with non-neurotoxic snake venom phospholipases A2. J. Biol. Chem. 1987, 262, 8966–8974. [Google Scholar] [PubMed]
- de Roodt, A.R.; Lago, N.R.; Stock, R.P. Myotoxicity and nephrotoxicity by Micrurus venoms in experimental envenomation. Toxicon 2012, 59, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Leao, L.I.; Ho, P.L.; Junqueira-de-Azevedo Ide, L. Transcriptomic basis for an antiserum against Micrurus corallinus (coral snake) venom. BMC Genom. 2009, 10, 112. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Vargas-Vargas, N.; Pla, D.; Sasa, M.; Rey-Suárez, P.; Sanz, L.; Gutiérrez, J.M.; Calvete, J.J.; Lomonte, B. Snake venomics of Micrurus alleni and Micrurus mosquitensis from the Caribbean region of Costa Rica reveals two divergent compositional patterns in New World elapids. Toxicon 2015, 107, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-H.; Wang, Y.-M.; Hseu, M.-J.; Tsai, I.-H. Molecular evolution and structure-function relationships of crotoxin-like and asparagine-6-containing phospholipases A2 in pit viper venoms. Biochem. J. 2004, 381, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Kini, R.M.; Iwanaga, S. Structure-function relationships of phospholipases. I: Prediction of presynaptic neurotoxicity. Toxicon 1986, 24, 527–541. [Google Scholar] [CrossRef]
- Kini, R.M. Structure–function relationships and mechanism of anticoagulant phospholipase A2 enzymes from snake venoms. Toxicon 2005, 45, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Kini, R.M.; Iwanaga, S. Structure-function relationships of phospholipases. II: Charge density distribution and the myotoxicity of presynaptically neurotoxic phospholipases. Toxicon 1986, 24, 895–905. [Google Scholar] [CrossRef]
- Alape-Girón, A.; Persson, B.; Cederlund, E.; Flores-Díaz, M.; Gutiérrez, J.M.; Thelestam, M.; Bergman, T.; Jörnvall, H. Elapid venom toxins: Multiple recruitments of ancient scaffolds. Eur. J. Biochem. 1999, 259, 225–234. [Google Scholar] [CrossRef] [PubMed]
- White, S.P.; Scott, D.L.; Otwinowski, Z.; Gelb, M.H.; Sigler, P.B. Crystal structure of cobra-venom phospholipase A2 in a complex with a transition-state analogue. Science 1990, 250, 1560–1563. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.; JoséMaría, G.; Bruno, L.; Elsa, P.; Luis, C.; Ermila, R. Local effects induced by coral snake venoms: Evidence of myonecrosis after experimental inoculations of venoms from five species. Toxicon 1983, 21, 777–783. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Arroyo, O.; Chaves, F.; Lomonte, B.; Cerdas, L. Pathogenesis of myonecrosis induced by coral snake (Micrurus nigrocinctus) venom in mice. Br. J. Exp. Pathol. 1986, 67, 1–12. [Google Scholar] [PubMed]
- Wrogemann, K.; Pena, S.D. Mitochondrial calcium overload: A general mechanism for cell-necrosis in muscle diseases. Lancet 1976, 1, 672–674. [Google Scholar] [CrossRef]
- Arroyo, O.; Rosso, J.P.; Vargas, O.; Gutierrez, J.M.; Cerdas, L. Skeletal muscle necrosis induced by a phospholipase A2 isolated from the venom of the coral snake Micrurus nigrocinctus nigrocinctus. Comp. Biochem. Physiol. B 1987, 87, 949–952. [Google Scholar] [CrossRef]
- Goularte, F.C.; da Cruz-Höfling, M.A.; Corrado, A.P.; Rodrigues-Simioni, L. Electrophysiological and ultrastructural analysis of the neuromuscular blockade and miotoxicity induced by the Micrurus nigrocinctus snake venom. Acta Physiol. Pharmacol. Ther. Latinoam. 1999, 49, 290–296. [Google Scholar] [PubMed]
- Terra, A.L.; Moreira-Dill, L.S.; Simoes-Silva, R.; Monteiro, J.R.; Cavalcante, W.L.; Gallacci, M.; Barros, N.B.; Nicolete, R.; Teles, C.B.; Medeiros, P.S.; et al. Biological characterization of the Amazon coral Micrurus spixii snake venom: Isolation of a new neurotoxic phospholipase A2. Toxicon 2015, 103, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, N.D.; Garcia, R.C.; Ferreira, A.K.; Batista, D.R.; Cassola, A.C.; Maria, D.; Lebrun, I.; Carneiro, S.M.; Afeche, S.C.; Marcourakis, T.; et al. Neurotoxicity of coral snake phospholipases A2 in cultured rat hippocampal neurons. Brain Res. 2014, 1552, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lind, P.; Peter, L.; David, E. Amino acid sequence of a lethal myotoxic phospholipase A2 from the venom of the common sea snake (Enhydrina schistosa). Toxicon 1981, 19, 11–24. [Google Scholar] [CrossRef]
- Lin, W.W.; Chang, P.L.; Lee, C.Y.; Joubert, F.J. Pharmacological study on phospholipases A2 isolated from Naja mossambica mossambica venom. Proc. Natl. Sci. Counc. Repub. China B 1987, 11, 155–163. [Google Scholar] [PubMed]
- Lambeau, G.; Schmid-Alliana, A.; Lazdunski, M.; Barhanin, J. Identification and purification of a very high affinity binding protein for toxic phospholipases A2 in skeletal muscle. J. Biol. Chem. 1990, 265, 9526–9532. [Google Scholar] [PubMed]
- Halpert, J.; Eaker, D. Isolation and amino acid sequence of a neurotoxic phospholipase A from the venom of the Australian tiger snake Notechis scutatus scutatus. J. Biol. Chem. 1976, 251, 7343–7347. [Google Scholar] [PubMed]
- Dixon, R.W.; Harris, J.B. Myotoxic activity of the toxic phospholipase, notexin, from the venom of the Australian tiger snake. J. Neuropathol. Exp. Neurol. 1996, 55, 1230–1237. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Ownby, C.L. Skeletal muscle degeneration induced by venom phospholipases A2: Insights into the mechanisms of local and systemic myotoxicity. Toxicon 2003, 42, 915–931. [Google Scholar] [CrossRef] [PubMed]
- Casais-e-Silva, L.L.; Teixeira, C.F.P.; Lebrun, I.; Lomonte, B.; Alape-Girón, A.; Gutiérrez, J.M. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2. Toxicol. Lett. 2016, 257, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Alberto Ponce-Soto, L.; Marangoni, S.; Lomonte, B. Systemic and local myotoxicity induced by snake venom group II phospholipases A2: Comparison between crotoxin, crotoxin B and a Lys49 PLA2 homologue. Toxicon 2008, 51, 80–92. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, S.C.; Hyslop, S.; Fontes, M.R.M.; Prado-Franceschi, J.; Zambelli, V.O.; Magro, A.J.; Brigatte, P.; Gutierrez, V.P.; Cury, Y. Crotoxin: Novel activities for a classic beta-neurotoxin. Toxicon 2010, 55, 1045–1060. [Google Scholar] [CrossRef] [PubMed]
- Nunez, C.E.; Angulo, Y.; Lomonte, B. Identification of the myotoxic site of the Lys49 phospholipase A2 from Agkistrodon piscivorus piscivorus snake venom: Synthetic C-terminal peptides from Lys49, but not from Asp49 myotoxins, exert membrane-damaging activities. Toxicon 2001, 39, 1587–1594. [Google Scholar] [CrossRef]
- Francis, B.; Brian, F.; Williams, E.S.; Corrine, S.; Kaiser, I.I. Proteins isolated from the venom of the common tiger snake (Notechis scutatus scutatus) promote hypotension and hemorrhage. Toxicon 1993, 31, 447–458. [Google Scholar] [CrossRef]
- Francis, B.; Coffield, J.A.; Simpson, L.L.; Kaiser, I.I. Amino Acid Sequence of a New Type of Toxic Phospholipase A2 from the Venom of the Australian Tiger Snake (Notechis scutatus scutatus). Arch. Biochem. Biophys. 1995, 318, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Francis, B.R.; da Silva Junior, N.J.; Seebart, C.; Casais e Silva, L.L.; Schmidt, J.J.; Kaiser, I.I. Toxins isolated from the venom of the Brazilian coral snake (Micrurus frontalis frontalis) include hemorrhagic type phospholipases A2 and postsynaptic neurotoxins. Toxicon 1997, 35, 1193–1203. [Google Scholar] [CrossRef]
- Marks, S.L.; Mannella, C.; Schaer, M. Coral snake envenomation in the dog: Report of four cases and review of the literature. J. Am. Anim. Hosp. Assoc. 1990, 26, 629–634. [Google Scholar]
- Peterson, M.E. Snake bite: Pit vipers. Clin. Tech. Small Anim. Pract. 2006, 21, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.L.; Fox, K.; Schaer, M. A retrospective evaluation of coral snake envenomation in dogs and cats: 20 cases (1996–2011). J. Vet. Emerg. Crit. Care 2012, 22, 682–689. [Google Scholar] [CrossRef] [PubMed]
- Arce-Bejarano, R.; Lomonte, B.; Gutiérrez, J.M. Intravascular hemolysis induced by the venom of the Eastern coral snake, Micrurus fulvius, in a mouse model: Identification of directly hemolytic phospholipases A2. Toxicon 2014, 90, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Lambeau, G.; Ancian, P.; Nicolas, J.P.; Beiboer, S.H.; Moinier, D.; Verheij, H.; Lazdunski, M. Structural elements of secretory phospholipases A2 involved in the binding to M-type receptors. J. Biol. Chem. 1995, 270, 5534–5540. [Google Scholar] [CrossRef] [PubMed]
- Belo, C.A.; Toyama, M.H.; Toyama Dde, O.; Marangoni, S.; Moreno, F.B.; Cavada, B.S.; Fontana, M.D.; Hyslop, S.; Carneiro, E.M.; Boschero, A.C. Determination of the amino acid sequence of a new phospholipase A(2) (MIDCA1) isolated from Micrurus dumerilii carinicauda venom. Protein J. 2005, 24, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Carbajal-Saucedo, A.; Floriano, R.S.; Dal Belo, C.A.; Olvera-Rodriguez, A.; Alagon, A.; Rodrigues-Simioni, L. Neuromuscular activity of Micrurus laticollaris (Squamata: Elapidae) venom in vitro. Toxins 2014, 6, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Alape-Girón, A.; Alberto, A.-G.; Bruno, L.; Bjorn, G.; Da Silva, N.J.; Monica, T. Electrophoretic and immunochemical studies of Micrurus snake venoms. Toxicon 1994, 32, 713–723. [Google Scholar] [CrossRef]
- Alape-Girón, A.; Stiles, B.; Schmidt, J.; Girón-Cortes, M.; Thelestam, M.; Jörnvall, H.; Bergman, T. Characterization of multiple nicotinic acetylcholine receptor-binding proteins and phospholipases A2 from the venom of the coral snake Micrurus nigrocinctus. FEBS Lett. 1996, 380, 29–32. [Google Scholar] [CrossRef]
- Goularte, F.C.; Cruz-Höfling, M.A.; Cogo, J.C.; Gutiérrez, J.M.; Rodrigues-Simioni, L. The ability of specific antivenom and low temperature to inhibit the myotoxicity and neuromuscular block induced by Micrurus nigrocinctus venom. Toxicon 1995, 33, 679–689. [Google Scholar] [CrossRef]
- Oliveira, D.A.; Harasawa, C.; Seibert, C.S.; Casais e Silva, L.L.; Pimenta, D.C.; Lebrun, I.; Sandoval, M.R. Phospholipases A2 isolated from Micrurus lemniscatus coral snake venom: Behavioral, electroencephalographic, and neuropathological aspects. Brain Res. Bull. 2008, 75, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Camargo, T.M.; de Roodt, A.R.; da Cruz-Höfling, M.A.; Rodrigues-Simioni, L. The neuromuscular activity of Micrurus pyrrhocryptus venom and its neutralization by commercial and specific coral snake antivenoms. J. Venom Res. 2011, 2, 24–31. [Google Scholar] [PubMed]
- Bénard-Valle, M.; Carbajal-Saucedo, A.; de Roodt, A.; Lopez-Vera, E.; Alagon, A. Biochemical characterization of the venom of the coral snake Micrurus tener and comparative biological activities in the mouse and a reptile model. Toxicon 2013, 77, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Condrea, E.; Rapuano, B.E.; Fletcher, J.E.; Yang, C.C.; Rosenberg, P. Effects of arginine modification of Naja nigricollis and Naja naja atra snake venom phospholipases A2 on enzymatic activity, lethality and anticoagulant action. Toxicon 1981, 19, 721–725. [Google Scholar] [CrossRef]
- Rosenberg, P.; Condrea, E.; Rapuano, B.E.; Soons, K.R.; Yang, C.C. Dissociation of pharmacological and enzymatic activities of snake venom phospholipases A2 by modification of carboxylate groups. Biochem. Pharmacol. 1983, 32, 3525–3530. [Google Scholar] [CrossRef]
- Rosenberg, P.; Condrea, E.; Fletcher, J.E.; Rapuano, B.E. Dissociation between enzymatic activity and pharmacological properties of snake venom phospholipases A2. Toxicon 1983, 21, 371–375. [Google Scholar] [CrossRef]
- Kini, R.M.; Evans, H.J. A model to explain the pharmacological effects of snake venom phospholipases A2. Toxicon 1989, 27, 613–635. [Google Scholar] [CrossRef]
- Kini, R.M. Excitement ahead: Structure, function and mechanism of snake venom phospholipase A2 enzymes. Toxicon 2003, 42, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Salazar, A.M.; Vivas, J.; Sánchez, E.E.; Rodríguez-Acosta, A.; Ibarra, C.; Gil, A.; Carvajal, Z.; Girón, M.E.; Estrella, A.; Navarrete, L.F.; et al. Hemostatic and toxinological diversities in venom of Micrurus tener tener, Micrurus fulvius fulvius and Micrurus isozonus coral snakes. Toxicon 2011, 58, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Evans, H.J.; Franson, R.; Qureshi, G.D.; Moo-Penn, W.F. Isolation of anticoagulant proteins from cobra venom (Naja nigricollis). Identity with phospholipases A2. J. Biol. Chem. 1980, 255, 3793–3797. [Google Scholar] [PubMed]
- Yuan, Y.; Jackson, S.P.; Mitchell, C.A.; Salem, H.H. Purification and characterisation of a snake venom phospholipase A2: A potent inhibitor of platelet aggregation. Thromb. Res. 1993, 70, 471–481. [Google Scholar] [CrossRef]
- Rudrammaji, L.M.; Machiah, K.D.; Kantha, T.P.; Gowda, T.V. Role of catalytic function in the antiplatelet activity of phospholipase A2 cobra (Naja naja naja) venom. Mol. Cell. Biochem. 2001, 219, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Sundell, I.B.; Theakston, R.D.; Kamiguti, A.S.; Harris, R.J.; Treweeke, A.T.; Laing, G.D.; Fox, J.W.; Warrell, D.A.; Zuzel, M. The inhibition of platelet aggregation and blood coagulation by Micropechis ikaheka venom. Br. J. Haematol. 2001, 114, 852–860. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-L.; Peng, L.-S.; Zhong, X.-F.; Wei, J.-W.; Jiang, X.-Y.; Ye, L.-T.; Zou, L.; Tu, H.-B.; Wu, W.-Y.; Xu, A.-L. Functional expression and characterization of a recombinant phospholipase A2 from sea snake Lapemis hardwickii as a soluble protein in E. coli. Toxicon 2003, 41, 713–721. [Google Scholar] [CrossRef]
- Satish, S.; Tejaswini, J.; Krishnakantha, T.P.; Gowda, T.V. Purification of a Class B1 platelet aggregation inhibitor phospholipase A2 from Indian cobra (Naja naja) venom. Biochimie 2004, 86, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Kini, R.M.; Evans, H.J. Structure-function relationships of phospholipases. The anticoagulant region of phospholipases A2. J. Biol. Chem. 1987, 262, 14402–14407. [Google Scholar] [PubMed]
- Doery, H.M.; Pearson, J.E. Phospholipase B in snake venoms and bee venom. Biochem. J. 1964, 92, 599–602. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Chen, S.; Gu, J. Identification and characterization of Harobin, a novel fibrino(geno)lytic serine protease from a sea snake (Lapemis hardwickii). FEBS Lett. 2007, 581, 2965–2973. [Google Scholar] [CrossRef] [PubMed]
- Rau, J.C.; Beaulieu, L.M.; Huntington, J.A.; Church, F.C. Serpins in thrombosis, hemostasis and fibrinolysis. J. Thromb. Haemost. 2007, 5 (Suppl. 1), 102–115. [Google Scholar] [CrossRef] [PubMed]
- Bock, P.E.; Olson, S.T.; Björk, I. Inactivation of thrombin by antithrombin is accompanied by inactivation of regulatory exosite I. J. Biol. Chem. 1997, 272, 19837–19845. [Google Scholar] [CrossRef] [PubMed]
- Huntington, J.A.; Read, R.J.; Carrell, R.W. Structure of a serpin-protease complex shows inhibition by deformation. Nature 2000, 407, 923–926. [Google Scholar] [CrossRef] [PubMed]
- Meekins, D.A.; Kanost, M.R.; Michel, K. Serpins in arthropod biology. Semin. Cell Dev. Biol. 2017, 62, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.R.; James, A.A. Isolation and characterization of the gene encoding a novel factor Xa-directed anticoagulant from the yellow fever mosquito, Aedes aegypti. J. Biol. Chem. 1998, 273, 20802–20809. [Google Scholar] [CrossRef] [PubMed]
- Calvo, E.; Mizurini, D.M.; Sá-Nunes, A.; Ribeiro, J.M.C.; Andersen, J.F.; Mans, B.J.; Monteiro, R.Q.; Kotsyfakis, M.; Francischetti, I.M.B. Alboserpin, a factor Xa inhibitor from the mosquito vector of yellow fever, binds heparin and membrane phospholipids and exhibits antithrombotic activity. J. Biol. Chem. 2011, 286, 27998–28010. [Google Scholar] [CrossRef] [PubMed]
- Prevot, P.-P.; Adam, B.; Boudjeltia, K.Z.; Brossard, M.; Lins, L.; Cauchie, P.; Brasseur, R.; Vanhaeverbeek, M.; Vanhamme, L.; Godfroid, E. Anti-hemostatic effects of a serpin from the saliva of the tick Ixodes ricinus. J. Biol. Chem. 2006, 281, 26361–26369. [Google Scholar] [CrossRef] [PubMed]
- Chmelar, J.; Oliveira, C.J.; Rezacova, P.; Francischetti, I.M.B.; Kovarova, Z.; Pejler, G.; Kopacek, P.; Ribeiro, J.M.C.; Mares, M.; Kopecky, J.; et al. A tick salivary protein targets cathepsin G and chymase and inhibits host inflammation and platelet aggregation. Blood 2011, 117, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Ibelli, A.M.G.; Kim, T.K.; Hill, C.C.; Lewis, L.A.; Bakshi, M.; Miller, S.; Porter, L.; Mulenga, A. A blood meal-induced Ixodes scapularis tick saliva serpin inhibits trypsin and thrombin, and interferes with platelet aggregation and blood clotting. Int. J. Parasitol. 2014, 44, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Tirloni, L.; Kim, T.K.; Coutinho, M.L.; Ali, A.; Seixas, A.; Termignoni, C.; Mulenga, A.; da Silva Vaz, I., Jr. The putative role of Rhipicephalus microplus salivary serpins in the tick-host relationship. Insect Biochem. Mol. Biol. 2016, 71, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Lew-Tabor, A.; Rodriguez-Valle, M. Effective inhibition of thrombin by Rhipicephalus microplus serpin-15 (RmS-15) obtained in the yeast Pichia pastoris. Ticks Tick Borne Dis. 2016, 7, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Roy, H.; Bhardwaj, S.; Ylä-Herttuala, S. Biology of vascular endothelial growth factors. FEBS Lett. 2006, 580, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- Karkkainen, M.J.; Haiko, P.; Sainio, K.; Partanen, J.; Taipale, J.; Petrova, T.V.; Jeltsch, M.; Jackson, D.G.; Talikka, M.; Rauvala, H.; et al. Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins. Nat. Immunol. 2004, 5, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Pung, Y.F.; Wong, P.T.H.; Kumar, P.P.; Hodgson, W.C.; Kini, R.M. Ohanin, a novel protein from king cobra venom, induces hypolocomotion and hyperalgesia in mice. J. Biol. Chem. 2005, 280, 13137–13147. [Google Scholar] [CrossRef] [PubMed]
- St Pierre, L.; Fischer, H.; Adams, D.J.; Schenning, M.; Lavidis, N.; de Jersey, J.; Masci, P.P.; Lavin, M.F. Distinct activities of novel neurotoxins from Australian venomous snakes for nicotinic acetylcholine receptors. Cell. Mol. Life Sci. 2007, 64, 2829–2840. [Google Scholar] [CrossRef] [PubMed]
- Junqueira-de-Azevedo, I.L.M. Lachesis muta (Viperidae) cDNAs Reveal Diverging Pit Viper Molecules and Scaffolds Typical of Cobra (Elapidae) Venoms: Implications for Snake Toxin Repertoire Evolution. Genetics 2006, 173, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; Lemmon, A.R.; Lemmon, E.M.; Caudle, S.B. A high-throughput venom-gland transcriptome for the Eastern Diamondback Rattlesnake (Crotalus adamanteus) and evidence for pervasive positive selection across toxin classes. Toxicon 2011, 57, 657–671. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, A.D.; Swain, M.T.; Logan, D.W.; Mulley, J.F. Testing the Toxicofera: Comparative reptile transcriptomics casts doubt on the single, early evolution of the reptile venom system. Toxicon 2014, 92, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Tomee, J.F.; Koëter, G.H.; Hiemstra, P.S.; Kauffman, H.F. Secretory leukoprotease inhibitor: A native antimicrobial protein presenting a new therapeutic option? Thorax 1998, 53, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Nathan, C.; Jin, W.; Sim, D.; Ashcroft, G.S.; Wahl, S.M.; Lacomis, L.; Erdjument-Bromage, H.; Tempst, P.; Wright, C.D.; et al. Conversion of proepithelin to epithelins: Roles of SLPI and elastase in host defense and wound repair. Cell 2002, 111, 867–878. [Google Scholar] [CrossRef]
- Pahari, S.; Mackessy, S.P.; Manjunatha Kini, R. The venom gland transcriptome of the Desert Massasauga Rattlesnake (Sistrurus catenatus edwardsii): Towards an understanding of venom composition among advanced snakes (Superfamily Colubroidea). BMC Mol. Biol. 2007, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Sanz, L.; Pla, D.; Pérez, A.; Rodríguez, Y.; Zavaleta, A.; Salas, M.; Lomonte, B.; Calvete, J.J. Venomic Analysis of the Poorly Studied Desert Coral Snake, Micrurus tschudii tschudii, Supports the 3FTx/PLA₂ Dichotomy across Micrurus Venoms. Toxins 2016, 8, 178. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Sasa, M.; Rey-Suárez, P.; Bryan, W.; Gutiérrez, J.M. Venom of the Coral Snake Micrurus clarki: Proteomic Profile, Toxicity, Immunological Cross-Neutralization, and Characterization of a Three-Finger Toxin. Toxins 2016, 8, 138. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, N.J., Jr.; Sites, J.W., Jr. Phylogeny of South American triad coral snakes (Elapidae: Micrurus) based on molecular characters. Herpetologica 2001, 57, 1–22. [Google Scholar]
- Slowinski, J.B. A Phylogenetic Analysis of the New World Coral Snakes (Elapidae: Leptomicrurus, Micruroides, and Micrurus) Based on Allozymic and Morphological Characters. J. Herpetol. 1995, 29, 325–338. [Google Scholar] [CrossRef]
- Vonk, F.J.; Casewell, N.R.; Henkel, C.V.; Heimberg, A.M.; Jansen, H.J.; McCleary, R.J.; Kerkkamp, H.M.; Vos, R.A.; Guerreiro, I.; Calvete, J.J.; et al. The king cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc. Natl. Acad. Sci. USA 2013, 110, 20651–20656. [Google Scholar] [CrossRef] [PubMed]
- Siang, A.S.; Doley, R.; Vonk, F.J.; Kini, R.M. Transcriptomic analysis of the venom gland of the red-headed krait (Bungarus flaviceps) using expressed sequence tags. BMC Mol. Biol. 2010, 11, 24. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Li, Y.; Lee, W.; Xu, X.; Zhang, Y.; Zhao, R.; Zhang, Y.; Wang, W. Venom gland transcriptomes of two elapid snakes (Bungarus multicinctus and Naja atra) and evolution of toxin genes. BMC Genom. 2011, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Fung, S.Y.; Yap, M.K.K.; Leong, P.K.; Liew, J.L.; Tan, N.H. Unveiling the elusive and exotic: Venomics of the Malayan blue coral snake (Calliophis bivirgata flaviceps). J. Proteom. 2016, 132, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Tin, M.M.-Y.; Economo, E.P.; Mikheyev, A.S. Sequencing degraded DNA from non-destructively sampled museum specimens for RAD-tagging and low-coverage shotgun phylogenetics. PLoS ONE 2014, 9, e96793. [Google Scholar] [CrossRef] [PubMed]
- Transdecoder. Available online: http://transdecoder.sf.net, version r20131110 (accessed on 5 June 2017).
- Common Repository of Adventitious Proteins. Available online: http://www.thegpm.org/crap/ (accessed on 5 June 2017).
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Arnold, K.; Bordoli, L.; Kopp, J.; Schwede, T. The SWISS-MODEL workspace: A web-based environment for protein structure homology modelling. Bioinformatics 2006, 22, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Li, L. OrthoMCL: Identification of Ortholog Groups for Eukaryotic Genomes. Genome Res. 2003, 13, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- bioPython v1.64. Available online: http://biopython.org/wiki/Download (accessed on 7 June 2017).
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 113. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML-VI-HPC: Maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 2006, 22, 2688–2690. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Köster, J.; Rahmann, S. Snakemake—A scalable bioinformatics workflow engine. Bioinformatics 2012, 28, 2520–2522. [Google Scholar] [CrossRef] [PubMed]
- Github Repository. Available online: https://github.com/migrau/phylopipe (accessed on 5 June 2017).
- Ogawa, Y.; Murayama, N.; Yanoshita, R. Molecular cloning and characterization of ecto-5′-nucleotidase from the venoms of Gloydius blomhoffi. Toxicon 2009, 54, 408–412. [Google Scholar] [CrossRef] [PubMed]



| mAChR Class | Tissue Target | Agent Action | Organism | Effect | Reference | Toxin | Reference | Action | Probable Effect |
|---|---|---|---|---|---|---|---|---|---|
| M1 | Rostral ventrolateral medulla | Agonist | Humans | Vasoconstriction, Tachycardia | Medina et al., 1997 | m1-Toxin; MT7 | Max et al., 1993; Jolkkonen, 1996; Liang et al., 1996 | Antagonist | Hypotension |
| M1 | Submandibular & jugular vein endothelium | Agonist | Rats | Vasoconstriction | Ryberg et al., 2008 | ||||
| M2 | Cerebral ventricles | Antagonist | SH rats | Hypotension | Brezenoff et al., 1988 | m2-toxin | Carsi et al., 1999 | Antagonist | Hypotension |
| M2 | Cerebral ventricles | Agonist | Rats | Hypertension, Tachycardia | Pazos et al., 1986; Kubo, 1998; Ozkutlu et al., 1993 | ||||
| M2 | Cardiac Atrium | Mice | Negative Inotropy | Nishimaru et al., 2000 | |||||
| M3 | Aorta | Agonist | Mice | Vasodilation | Khurana et al, 2004 | MT2; MTα; MTβ | Bradley, 2000; Jolkkonen et al., 1995 | Agonist | Hypotension |
| M3 | Resistance vessels | Agonist | Mice | Vasodilation | Bruning et al., 1994; Gericke et al., 2011 | ||||
| M3 | Mesenteric vessels | Agonist | Rat | Vasodilation | Hendriks et al., 1992 | ||||
| M3 | Coronary arteries | Agonist | Mice | Vasodilation | Lamping et al., 2004 | ||||
| M4 | Submandibular & jugular vein endothelium | Agonist | Rats | Vasoconstriction | Ryberg et al., 2008 | m4-toxin; MT3; MT2; MT7 | Jolkkonen et al., 1994; Liang et al., 1996; Bradley, 2000 | Antagonist | Hypotension |
| M4 | Spinal Cord | Binding only | Rats | Not determined | Höglund & Baghdoyan, 1997 | ||||
| M5 | Cerebral blood vessels | Agonist | Mice | Vasodilation | Yamada et al., 2001 | MTα; MTβ | Jolkkonen et al., 1995 | Agonist | Hypotension |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aird, S.D.; Da Silva, N.J.; Qiu, L.; Villar-Briones, A.; Saddi, V.A.; Pires de Campos Telles, M.; Grau, M.L.; Mikheyev, A.S. Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa. Toxins 2017, 9, 187. https://doi.org/10.3390/toxins9060187
Aird SD, Da Silva NJ, Qiu L, Villar-Briones A, Saddi VA, Pires de Campos Telles M, Grau ML, Mikheyev AS. Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa. Toxins. 2017; 9(6):187. https://doi.org/10.3390/toxins9060187
Chicago/Turabian StyleAird, Steven D., Nelson Jorge Da Silva, Lijun Qiu, Alejandro Villar-Briones, Vera Aparecida Saddi, Mariana Pires de Campos Telles, Miguel L. Grau, and Alexander S. Mikheyev. 2017. "Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa" Toxins 9, no. 6: 187. https://doi.org/10.3390/toxins9060187
APA StyleAird, S. D., Da Silva, N. J., Qiu, L., Villar-Briones, A., Saddi, V. A., Pires de Campos Telles, M., Grau, M. L., & Mikheyev, A. S. (2017). Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa. Toxins, 9(6), 187. https://doi.org/10.3390/toxins9060187







