Transcriptional and Physiological Responses to Nutrient Loading on Toxin Formation and Photosynthesis in Microcystis Aeruginosa FACHB-905
Abstract
:1. Introduction
2. Results
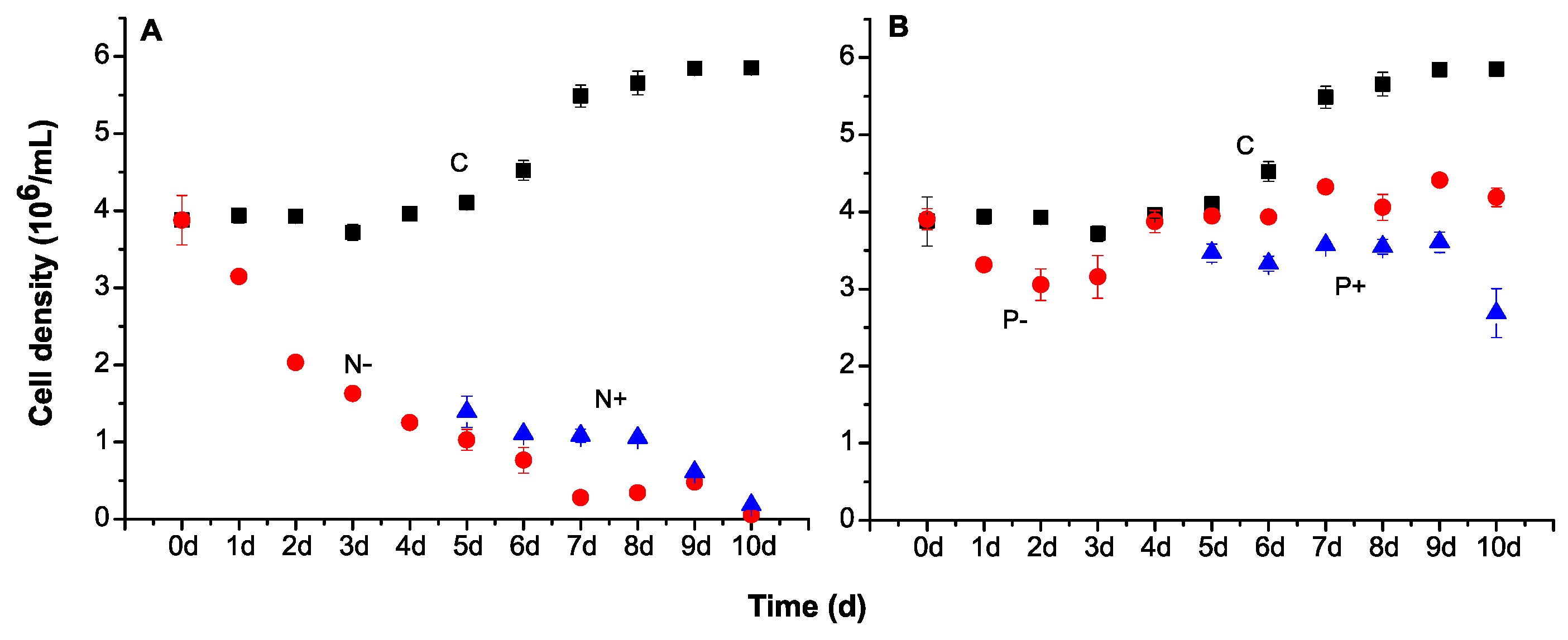
2.1. Cell Growth
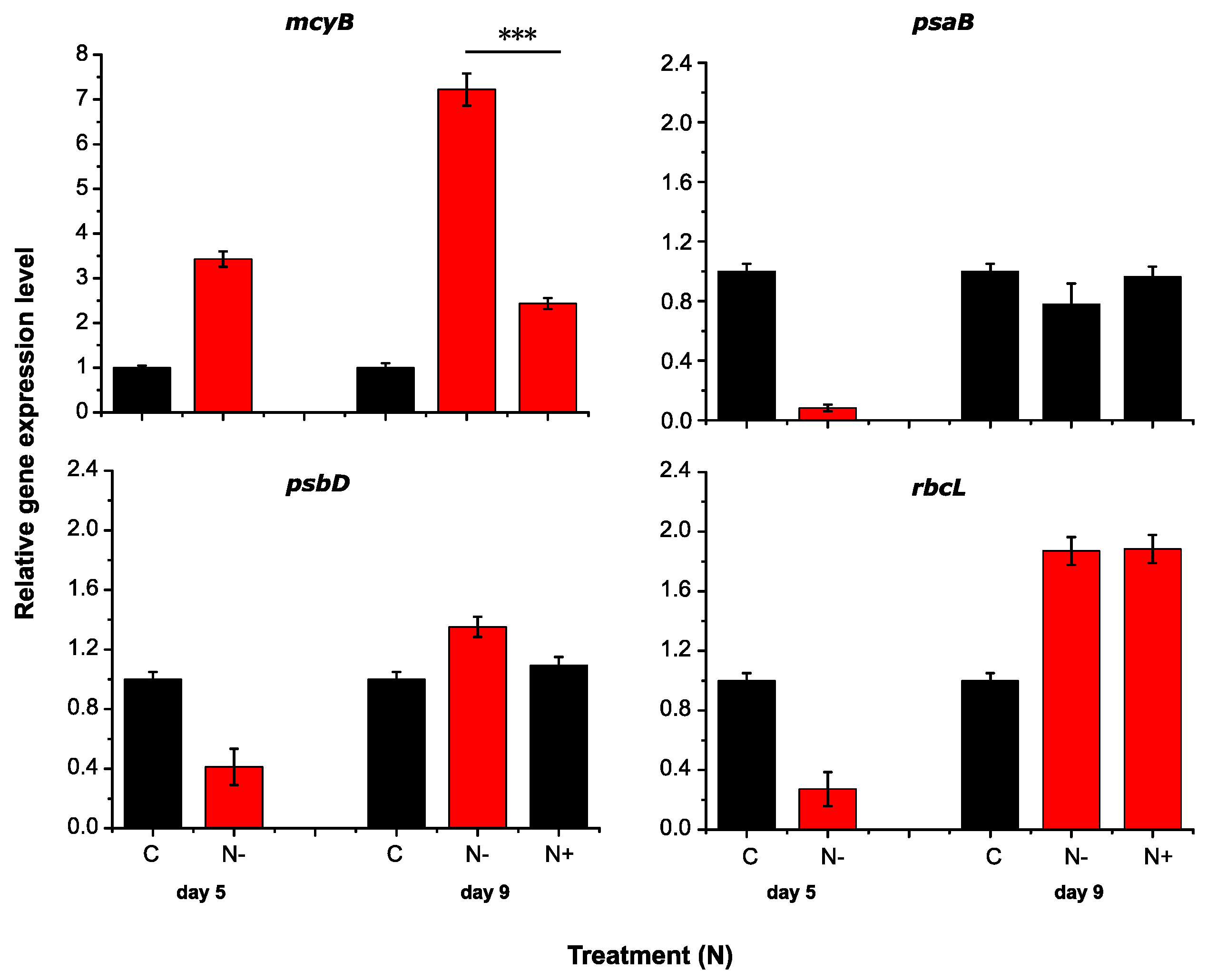
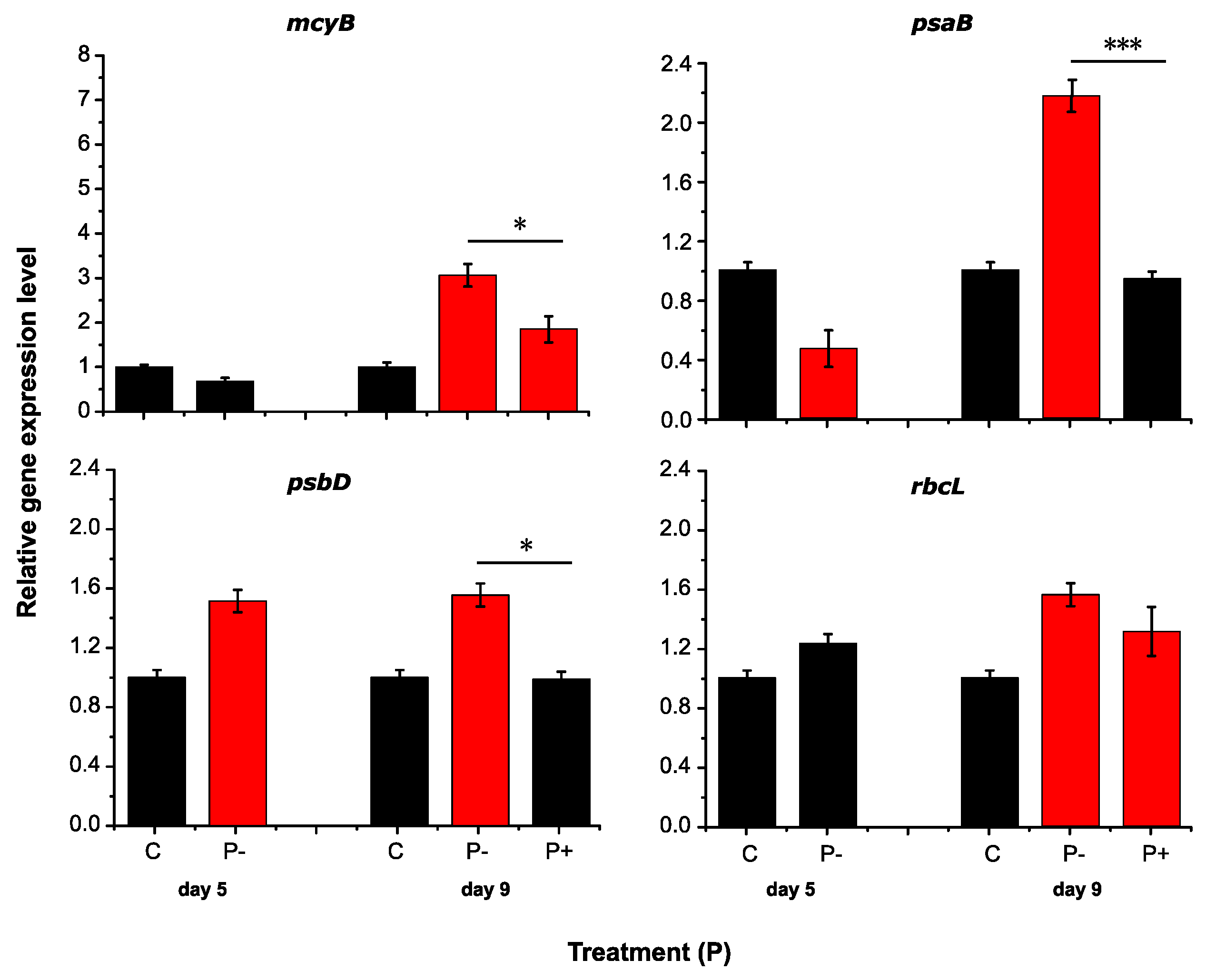
2.2. RT-qPCR Assay Performance
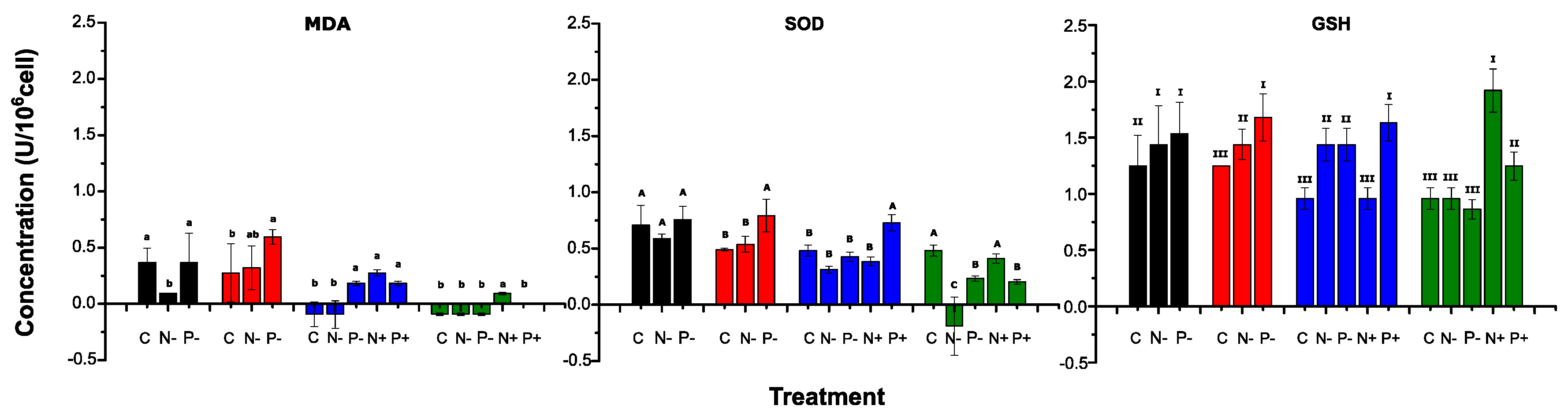
2.3. Examination of antioxidant systems
3. Discussion
4. Materials and Methods
4.1. Strain and Culture Conditions
4.2. RNA Extraction and Transcript Analysis
4.3. Relative RT-qPCR Assay
4.4. Enzyme Extraction and Assays
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Blooms Like It Hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Fleming, L.E.; Rivero, C.; Burns, J.; Williams, C.; Bean, J.A.; Shea, K.A.; Stinn, J. Blue green algal (cyanobacterial) toxins, surface drinking water, and liver cancer in Florida. Harmful Algae 2002, 1, 157–168. [Google Scholar] [CrossRef]
- Peng, G.; Fan, Z.; Wang, X.; Sui, X.; Chen, C. Photodegradation of microcystin-LR catalyzed by metal phthalocyanines immobilized on TiO2-SiO2 under visible-light irradiation. Water Sci. Technol. 2015, 72, 1824–1831. [Google Scholar] [CrossRef] [PubMed]
- Chia, M.A.; Cordeiro-Araújo, M.K.; Lorenzi, A.S.; Bittencourt-Oliveira, M.d.C. Cylindrospermopsin induced changes in growth, toxin production and antioxidant response of Acutodesmus acuminatus and Microcystis aeruginosa under differing light and nitrogen conditions. Ecotoxicology and Environmental Safety 2017, 142, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Poniedziałek, B.; Kokociński, M.; Jurczak, T.; Lipski, D.; Wiktorowicz, K. Interspecific allelopathy in cyanobacteria: Cylindrospermopsin and Cylindrospermopsis raciborskii effect on the growth and metabolism of Microcystis aeruginosa. Harmful Algae 2014, 35, 1–8. [Google Scholar] [CrossRef]
- Belisle, B.S.; Steffen, M.M.; Pound, H.L.; Watson, S.B.; Debruyn, J.M.; Bourbonniere, R.A.; Boyer, G.L.; Wilhelm, S.W. Urea in Lake Erie: Organic nutrient sources as potentially important drivers of phytoplankton biomass. J. Great Lakes Res. 2016, 42, 599–607. [Google Scholar] [CrossRef]
- Peng, G.; Fan, Z.; Wang, X.; Chen, C. Photosynthetic response to nitrogen source and different ratios of nitrogen and phosphorus in toxic cyanobacteria, Microcystis aeruginosa FACHB-905. J. Limnol. 2016, 75, 560–570. [Google Scholar] [CrossRef]
- Steffen, M.M.; Dearth, S.P.; Dill, B.D.; Li, Z.; Larsen, K.M.; Campagna, S.R.; Wilhelm, S.W. Nutrients drive transcriptional changes that maintain metabolic homeostasis but alter genome architecture in Microcystis. The ISME J. 2014, 8, 2080–2092. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.H. Low nitrogen to phosphorus ratios favor dominance by blue-green algae in lake phytoplankton. Science 1983, 221, 669–671. [Google Scholar] [CrossRef] [PubMed]
- Likens, G.E.; Bartsch, A.F.; Lauff, G.H.; Hobbie, J.E. Nutrients and Eutrophication. Science 1971, 172, 873–874. [Google Scholar] [CrossRef] [PubMed]
- Yue, D.; Peng, Y.; Yin, Q.; Xiao, L. Proteomic analysis of Microcystis aeruginosa in response to nitrogen and phosphorus starvation. J. Appl. Phycol. 2015, 27, 1–10. [Google Scholar] [CrossRef]
- Kuniyoshi, T.M.; Sevilla, E.; Bes, M.T.; Fillat, M.F.; Peleato, M.L. Phosphate deficiency (N/P 40:1) induces mcyD transcription and microcystin synthesis in Microcystis aeruginosa PCC7806. Plant Physiol. Biochem. 2013, 65, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.M.; Lee, S.J.; Jang, M.H.; Yoon, B.D. Microcystin production by Microcystis aeruginosa in a phosphorus-limited chemostat. Appl. Environ. Microbiol. 2000, 66, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Downing, T.G.; Sember, C.S.; Gehringer, M.M.; Leukes, W. Medium N:P ratios and specific growth rate comodulate microcystin and protein content in Microcystis aeruginosa PCC7806 and M. aeruginosa UV027. Microb. Ecol. 2005, 49, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Jang, M.H.; Kim, H.S.; Yoon, B.D.; Oh, H.M. Variation of microcystin content of Microcystis aeruginosa relative to medium N:P ratio and growth stage. Eur. J. Appl. Microbiol. 2000, 89, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Utkilen, H.; Gjølme, N. Iron-stimulated toxin production in Microcystis aeruginosa. Appl. Environ. Microbiol. 1995, 61, 797–800. [Google Scholar] [PubMed]
- Jähnichen, S.; Long, B.M.; Petzoldt, T. Microcystin production by Microcystis aeruginosa Direct regulation by multiple environmental factors. Harmful Algae 2011, 12, 95–104. [Google Scholar] [CrossRef]
- Watanabe, M.F.; Oishi, S. Effects of environmental factors on toxicity of a cyanobacterium (Microcystis aeruginosa) under culture conditions. Appl. Environ. Microbiol. 1985, 49, 1342–1344. [Google Scholar] [PubMed]
- Orr, P.T.; Jones, G.J. Relationship between microcystin production and cell division rates in nitrogen-limited Microcystis aeruginosa cultures. Limnol. Oceanogr. 1998, 43, 1604–1614. [Google Scholar] [CrossRef]
- Sevilla, E.; Martinluna, B.; Vela, L.; Bes, M.T.; Peleato, M.L.; Fillat, M.F. Microcystin-LR synthesis as response to nitrogen: transcriptional analysis of the mcyD gene in Microcystis aeruginosa PCC7806. Ecotoxicology 2010, 19, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; Davis, T.W.; Coyne, K.J.; Boyer, G.L. Interactive influences of nutrient loading, zooplankton grazing, and microcystin synthetase gene expression on cyanobacterial bloom dynamics in a eutrophic New York lake. Harmful Algae 2007, 6, 119–133. [Google Scholar] [CrossRef]
- Flores, E.; Herrero, A. Nitrogen assimilation and nitrogen control in cyanobacteria. Biochem. Soc. Trans. 2005, 33, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Horst, G.P.; Sarnelle, O.; White, J.D.; Hamilton, S.K.; Kaul, R.R.B.; Bressie, J.D. Nitrogen availability increases the toxin quota of a harmful cyanobacterium, Microcystis aeruginosa. Water Res. 2014, 54, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Downing, T.; Meyer, C.; Gehringer, M.; Van de Venter, M. Microcystin content of Microcystis aeruginosa is modulated by nitrogen uptake rate relative to specific growth rate or carbon fixation rate. Environ. Toxicol. 2005, 20, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Ginn, H.; Pearson, L.; Neilan, B. NtcA from Microcystis aeruginosa PCC 7806 is autoregulatory and binds to the microcystin promoter. Appl. Environ. Microbiol. 2010, 76, 4362–4368. [Google Scholar] [CrossRef] [PubMed]
- Long, B.M.; Jones, G.J.; Orr, P.T. Cellular microcystin content in N-limited Microcystis aeruginosa can be predicted from growth rate. Appl. Environ. Microbiol. 2001, 67, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, J.S.M.; Giani, A. Microcystin production and regulation under nutrient stress conditions in toxic Microcystis strains. Appl. Environ. Microbiol. 2014, 80, 5836–5843. [Google Scholar] [CrossRef] [PubMed]
- Asada, K. Chloroplasts: Formation of active oxygen and its scavenging. Methods Enzymol. 1984, 105, 422–429. [Google Scholar]
- Kebeish, R.; El-Ayouty, Y.; Husain, A. Effect of copper on growth, bioactive metabolites, antioxidant enzymes and photosynthesis-related gene transcription in Chlorella vulgaris. World J. Biol. Biol. Sci. 2014, 2, 34–43. [Google Scholar]
- Hu, Z.; Liu, Y.; Li, D.; Dauta, A. Growth and antioxidant system of the cyanobacterium Synechococcus elongatus in response to microcystin-RR. Hydrobiologia 2005, 534, 23–29. [Google Scholar] [CrossRef]
- Eldridge, S.L.C.; Wood, T.M.; Echols, K.R.; Topping, B.R. Microcystins, nutrient dynamics, and other environmental factors during blooms of non-microcystin-producing Aphanizomenon flos-aquae in Upper Klamath Lake, Oregon, 2009. Lake Reservoir Manage. 2013, 29, 68–81. [Google Scholar] [CrossRef]
- Paul, M.J.; Foyer, C.H. Sink regulation of photosynthesis. J. Exp. Bot. 2001, 52, 1383–1400. [Google Scholar] [CrossRef] [PubMed]
- Harke, M.J.; Gobler, C.J. Global transcriptional responses of the toxic cyanobacterium, Microcystis aeruginosa, to nitrogen stress, phosphorus stress, and growth on organic matter. PLoS ONE 2013, 8, e69834. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, R.; Forchhammer, K. Acclimation of unicellular cyanobacteria to macronutrient deficiency: emergence of a complex network of cellular responses. Microbiology 2005, 151, 2503–2514. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.M. Cyanobacterial Cell Inclusions. Annu. Rev. Microbiol. 1984, 38, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Qin, B.; Xu, H.; Wang, X. Effects of sediment and turbulence on alkaline phosphatase activity and photosynthetic activity of phytoplankton in the shallow hyper-eutrophic Lake Taihu, China. Environ. Sci. Pollut. Res. Int. 2016, 23, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Baldia, S.; Evangelista, A.; Aralar, E.; Santiago, A. Nitrogen and phosphorus utilization in the cyanobacterium Microcystis aeruginosa isolated from Laguna de Bay, Philippines. J. Appl. Phycol. 2007, 19, 607–613. [Google Scholar] [CrossRef]
- Jacobson, L.; Halmann, M. Polyphosphate metabolism in the blue-green alga Microcystis aeruginosa. J. Plankton Res. 1982, 4, 481–488. [Google Scholar] [CrossRef]
- Shi, X.; Yang, L.; Niu, X.; Xiao, L.; Kong, Z.; Qin, B.; Gao, G. Intracellular phosphorus metabolism of Microcystis aeruginosa under various redox potential in darkness. Microbiol. Res. 2003, 158, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Zilliges, Y.; Kehr, J.-C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef] [PubMed]
- Ramasubramanian, T.; Wei, T.F.; Oldham, A.K.; Golden, J.W. Transcription of the Anabaena sp. strain PCC 7120 NtcA gene: multiple transcripts and NtcA binding. J. Bacteriol. 1996, 178, 922–926. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Xiao, X.; Ghadouani, A.; Wu, J.; Nie, Z.; Peng, C.; Xu, X.; Shi, J. Effects of natural flavonoids on photosynthetic activity and cell integrity in Microcystis aeruginosa. Toxins 2015, 7, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Qian, Y.; Li, H.; Cheng, Y.; Zhao, M. The reduced bioavailability of copper by nano-TiO2 attenuates the toxicity to Microcystis aeruginosa. Environ. Sci. Pollut. Res. Int. 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Guan, Y.; Gao, B.; Yue, Q. Antioxidant responses and degradation of two antibiotic contaminants in Microcystis aeruginosa. Ecotoxicol. Environ. Saf. 2012, 86, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Guo, P.; Peng, X.; Wen, K. Effect of erythromycin exposure on the growth, antioxidant system and photosynthesis of Microcystis flos-aquae. J. Hazard. Mater. 2015, 283, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Wu, F.; Yuan, M.; Li, Q.; Gu, Y.; Wang, Y.; Liu, Q. Antioxidant responses of triangle sail mussel hyriopsis cumingii exposed to harmful algae Microcystis aeruginosa and hypoxia. Chemosphere 2015, 139, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, S.; Zhang, J.; Gao, B. Nitrogen-regulated interactions between Microcystis aeruginosa and spiramycin contaminant. Water Air Soil Pollut. 2015, 226, 1–11. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D. Molecular Cloning: A Laboratory Manual (3-Volume Set). Immunol. 2001, 49, 895–909. [Google Scholar]
- Edwards, U.; Rogall, T.; Blöcker, H.; Emde, M.; Böttger, E.C. Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res. 1989, 17, 7843–7853. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wu, H.; Chen, L.; Xie, S.; Zang, H.; Borriss, R.; Gao, X. Bacilysin from Bacillus amyloliquefaciens FZB42 has specific bactericidal activity against harmful algal bloom species. Appl. Environ. Microbiol. 2014, 80, 7512–7520. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Jie, X.; Wang, P.; Li, S.; Wang, G.; Li, Y.; Li, Y.; Acharya, K. Effect of linoleic acid sustained-release microspheres on Microcystis aeruginosa antioxidant enzymes activity and microcystins production and release. Chemosphere 2015, 121, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xie, P. Antioxidant enzyme activities of Microcystis aeruginosa in response to nonylphenols and degradation of nonylphenols by M. aeruginosa. Environ. Geochem. Health. 2007, 29, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Placer, Z.A.; Cushman, L.L.; Johnson, B.C. Estimation of product of lipid peroxidation (malonyl dialdehyde) in biochemical systems. Anal. Biochem. 1966, 16, 359–364. [Google Scholar] [CrossRef]
- Winterbourn, C.C.; Hawkins, R.E.; Brian, M.; Carrell, R. The estimation of red cell superoxide dismutase activity. J. Lab. Clin. Med. 1975, 85, 337–341. [Google Scholar] [PubMed]
- Hafeman, D.; Sunde, R.; Hoekstra, W. Effect of dietary selenium on erythrocyte and liver glutathione peroxidase in the rat. J. Nutr. 1974, 104, 580–587. [Google Scholar] [PubMed]
| Pearson’s Correlation Analysis | p Value | Pearson’s R | Adj. R-Square |
|---|---|---|---|
| MDA vs. SOD | 0.002 | 0.7261 | 0.4908 |
| MDA vs. GSH | 0.039 | 0.5363 | 0.2328 |
| Primer | Nucleotide Sequence (5′ to 3′) | Amplification Size (bp) | Reference |
|---|---|---|---|
| 16S F | GGACGGGTGAGTAACGCGTA | 74 | [52] |
| 16S R | CCCATTGCGGAAAATTCCCC | ||
| mcyB F | CCTACCGAGCGCTTGGG | 77 | This study |
| mcyB R | GAAAATCCCCAAAGATTCCTGAGT | ||
| psaB F | CGGTGACTGGGGTGTGTATG | 119 | This study |
| psaB R | ACTCGGTTTGGGGATGGA | ||
| psbD F | TCTTCGGCATCGCTTTCTC | 90 | This study |
| psbD R | CACCCACAGCACTCATCCA | ||
| rbcL F | CGTTTCCCCGTCGCTTT | 122 | This study |
| rbcL R | CCGAGTTTGGGTTTGATGGT |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, G.; Lin, S.; Fan, Z.; Wang, X. Transcriptional and Physiological Responses to Nutrient Loading on Toxin Formation and Photosynthesis in Microcystis Aeruginosa FACHB-905. Toxins 2017, 9, 168. https://doi.org/10.3390/toxins9050168
Peng G, Lin S, Fan Z, Wang X. Transcriptional and Physiological Responses to Nutrient Loading on Toxin Formation and Photosynthesis in Microcystis Aeruginosa FACHB-905. Toxins. 2017; 9(5):168. https://doi.org/10.3390/toxins9050168
Chicago/Turabian StylePeng, Guotao, Sijie Lin, Zhengqiu Fan, and Xiangrong Wang. 2017. "Transcriptional and Physiological Responses to Nutrient Loading on Toxin Formation and Photosynthesis in Microcystis Aeruginosa FACHB-905" Toxins 9, no. 5: 168. https://doi.org/10.3390/toxins9050168
APA StylePeng, G., Lin, S., Fan, Z., & Wang, X. (2017). Transcriptional and Physiological Responses to Nutrient Loading on Toxin Formation and Photosynthesis in Microcystis Aeruginosa FACHB-905. Toxins, 9(5), 168. https://doi.org/10.3390/toxins9050168








