Antivenom for Neuromuscular Paralysis Resulting From Snake Envenoming
Abstract
:1. Introduction
2. Neuromuscular Paralysis in Snake Envenoming
3. Neuromuscular Junction: The Primary Target Site
Neurotoxins in Snake Venoms
4. Antivenoms
4.1. Antivenom Efficacy
4.2. Antivenom Effectiveness
5. Clinical Studies of Antivenom for Neurotoxic Snake Envenoming
5.1. Randomised Controlled Trials
5.2. Non-Randomised Comparative Trials
5.3. Cohort Studies
6. Conclusions and Future Directions
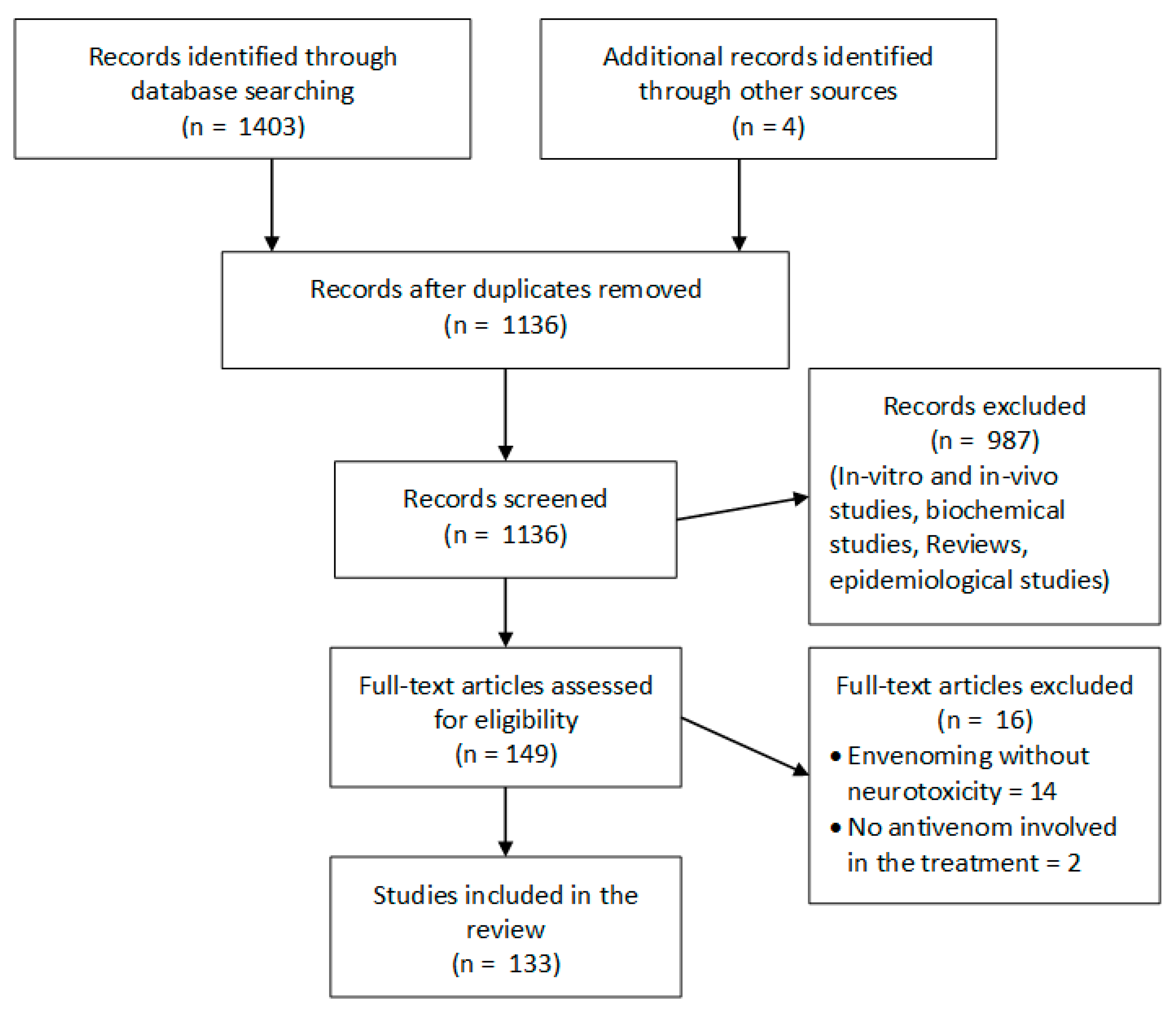
7. Methods
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kasturiratne, A.; Wickremasinghe, A.R.; De Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J. Snake-bites: Appraisal of the global situation. Bull. World Health Organ. 1998, 76, 515–524. [Google Scholar] [PubMed]
- Ranawaka, U.K.; Lalloo, D.G.; de Silva, H.J. Neurotoxicity in snakebite-the limits of our knowledge. PLoS Negl. Trop. Dis. 2013, 7, e2302. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Maduwage, K.; Sedgwick, M.; Pilapitiya, S.; Weerawansa, P.; Dahanayaka, N.J.; Buckley, N.A.; Johnston, C.; Siribaddana, S.; Isbister, G.K. Neuromuscular effects of common krait (Bungarus caeruleus) envenoming in Sri Lanka. PLoS Negl. Trop. Dis. 2016, 10, e0004368. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Maduwage, K.; Sedgwick, M.; Pilapitiya, S.; Weerawansa, P.; Dahanayake, N.; Buckley, N.A.; Siribaddana, S.; Isbister, G.K. Neurotoxicity in Russell’s viper (Daboia russelii) envenoming in Sri Lanka: A clinical and neurophysiological study. Clin. Toxicol. 2016, 54, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Warrell, D.A.; Looareesuwan, S.; White, N.J.; Theakston, R.D.G.; Warrell, M.J.; Kosakarn, W.; Reid, H.A. Severe neurotoxic envenoming by the Malayan krait Bungarus candidus (Linnaeus): Response to antivenom and anticholinesterase. Br. Med. J. 1983, 286, 678–680. [Google Scholar] [CrossRef]
- Watt, G.; Padre, L.; Tuazon, M.L.; Theakston, R.D.G.; Laughlin, L. Bites by the Philippine cobra (Naja naja philippinensis): Prominent neurotoxicity with minimal local signs. Am. J. Trop. Med. Hyg. 1988, 39, 306–311. [Google Scholar] [PubMed]
- Kularatne, S.A.M.; Budagoda, B.D.S.S.; Gawarammana, I.B.; Kularatne, W.K.S. Epidemiology, clinical profile and management issues of cobra (Naja naja) bites in Sri Lanka: First authenticated case series. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Manock, S.R.; Suarez, G.; Graham, D.; Avila-Aguero, M.L.; Warrell, D.A. Neurotoxic envenoming by South American coral snake (Micrurus lemniscatus helleri): Case report from eastern Ecuador and review. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Connolly, S.; Trevett, A.J.; Nwokolo, N.C.; Lalloo, D.G.; Naraqi, S.; Mantle, D.; Schofield, I.S.; Fawcett, P.R.W.; Harris, J.B.; Warrell, D.A. Neuromuscular effects of Papuan Taipan snake venom. Ann. Neurol. 1995, 38, 916–920. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K.; O’Leary, M.A.; Elliott, M.; Brown, S.G.A. Tiger snake (Notechis spp.) envenoming: Australian Snakebite Project (ASP-13). Med. J. Aust. 2012, 197, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.I.; O’Leary, M.A.; Brown, S.G.A.; Currie, B.J.; Halkidis, L.; Whitaker, R.; Close, B.; Isbister, G.K. Death adder envenoming causes neurotoxicity not reversed by antivenom—Australian Snakebite Project (ASP-16). PLoS Negl. Trop. Dis. 2012, 6, e1841. [Google Scholar] [CrossRef] [PubMed]
- Prasarnpun, S.; Walsh, J.; Awad, S.S.; Harris, J.B. Envenoming bites by kraits: The biological basis of treatment-resistant neuromuscular paralysis. Brain 2005, 128, 2987–2996. [Google Scholar] [CrossRef] [PubMed]
- Harris, J. Neuromuscular Junction (NMJ): A Target for Natural and Environmental Toxins in Humans. In Encyclopedia of Neuroscience; Elsevier Academic Press: Boston, MA, USA, 2009; pp. 539–549. [Google Scholar]
- Barber, C.M.; Isbister, G.K.; Hodgson, W.C. Alpha neurotoxins. Toxicon 2013, 66, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.B.; Scott-Davey, T. Secreted phospholipases A2 of snake venoms: Effects on the peripheral neuromuscular system with comments on the role of phospholipases A2 in disorders of the CNS and their uses in industry. Toxins (Basel) 2013, 1, 2533–2571. [Google Scholar] [CrossRef] [PubMed]
- Malina, T.; Krecsák, L.; Jelić, D.; Maretić, T.; Tóth, T.; Siško, M.; Pandak, N. First clinical experiences about the neurotoxic envenomings inflicted by lowland populations of the Balkan adder, Vipera berus bosniensis. Neurotoxicology 2011, 32, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Kularatne, S.A.M. Common krait (Bungarus caeruleus) bite in Anuradhapura, Sri Lanka: A prospective clinical study, 1996–98. Postgrad. Med. J. 2002, 78, 276–280. [Google Scholar] [CrossRef] [PubMed]
- De Silva, A.; Mendis, S.; Warrell, D.A. Neurotoxic envenoming by the Sri Lankan krait (Bungarus ceylonicus) complicated by traditional treatment and a reaction to antivenom. Trans. R. Soc. Trop. Med. Hyg. 1993, 87, 682–684. [Google Scholar] [CrossRef]
- Pillai, L.V.; Ambike, D.; Husainy, S.; Khaire, A.; Captain, A.; Kuch, U. Severe Neurotoxic Envenoming and Cardiac Complications after the Bite of a “Sind Krait” (Bungarus cf. sindanus) in Maharashtra, India. Trop. Med. Health 2012, 40, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.; Phillips, R.; Warrell, D.; Galagedara, Y.; Abeysekara, D.; Dissanayake, P.; de Silva, A.; Aloysius, D. Envenoming by the common krait (Bungarus caeruleus) and Sri Lankan cobra (Naja naja naja): Efficacy and complications of therapy with Haffkine antivenom. Trans. R. Soc. Trop. Med. Hyg. 1990, 84, 301–308. [Google Scholar] [CrossRef]
- Faiz, A.; Ghose, A.; Ahsan, F.; Rahman, R.; Amin, R.; Hassan, M.U.; Chowdhury, A.W.; Kuch, U.; Rocha, T.; Harris, J.B.; et al. The greater black krait (Bungarus niger), a newly recognized cause of neuro-myotoxic snake bite envenoming in Bangladesh. Brain 2010, 133, 3181–3193. [Google Scholar] [CrossRef] [PubMed]
- Laothong, C.; Sitprija, V. Decreased parasympathetic activities in Malayan krait (Bungarus candidus) envenoming. Toxicon 2001, 39, 1353–1357. [Google Scholar] [CrossRef]
- Kaminski, H.J.; Maas, E.; Spiegel, P.; Ruff, R.L. Why are eye muscles frequently involved in myasthenia gravis? Neurology 1990, 7, 1663–1669. [Google Scholar] [CrossRef]
- Soltys, J.; Gong, B.; Kaminski, H.; Zhou, Y.; Kusner, L. Extraocular Muscle Susceptibility to Myasthenia Gravis Unique Immunological Environment? Ann. N. Y. Acad. Sci. 2009, 1132, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Gawarammana, I.; Mendis, S.; Jeganathan, K. Acute ischemic strokes due to bites by Daboia russelii in Sri Lanka—First authenticated case series. Toxicon 2009, 54, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Mugundhan, K.; Thruvarutchelvan, K.; Sivakumar, S. Posterior circulation stroke in a young male following snake bite. J. Assoc. Physicians India 2008, 56, 713–714. [Google Scholar] [PubMed]
- Thomas, L.; Chausson, N.; Uzan, J.; Kaidomar, S.; Vignes, R.; Plumelle, Y.; Bucher, B.; Smadja, D. Thrombotic stroke following snake bites by the “Fer-de-Lance” Bothrops lanceolatus in Martinique despite antivenom treatment: A report of three recent cases. Toxicon 2006, 48, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Jeevagan, V.; Chang, T.; Gnanathasan, C.A. Acute ischemic stroke following Hump-nosed viper envenoming; first authenticated case. Thromb. J. 2012, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Narang, S.K.; Paleti, S.; Azeez Asad, M.A.; Samina, T.; Asad, M.A.A. Acute ischemic infarct in the middle cerebral artery territory following a Russell’s viper bite. Neurol. India 2009, 57, 479–480. [Google Scholar] [CrossRef] [PubMed]
- Gawarammana, I.B.; Mudiyanselage Kularatne, S.A.; Kularatne, K.; Waduge, R.; Weerasinghe, V.S.; Bowatta, S.; Senanayake, N. Deep coma and hypokalaemia of unknown aetiology following Bungarus caeruleus bites: Exploration of pathophysiological mechanisms with two case studies. J. Venom Res. 2010, 1, 71–75. [Google Scholar] [PubMed]
- Prakash, S.; Mathew, C.; Bhagat, S. Locked-in syndrome in snakebite. J. Assoc. Physicians India 2008, 56, 121–122. [Google Scholar] [PubMed]
- Sodhi, R.; Khanduri, S.; Nandha, H.; Bhasin, D.; Mandal, A.K. Brain death-think twice before labeling a patient. Am. J. Emerg. Med. 2012, 30, 1321.e1–1321.e2. [Google Scholar] [CrossRef] [PubMed]
- Dayal, M.; Prakash, S.; Verma, P.K.; Pawar, M. Neurotoxin envenomation mimicking brain death in a child: A case report and review of literature. Indian J. Anaesth. 2014, 58, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, E.X.; Pereira, E.F.R.; Alkondon, M.; Rogers, S.W. Mammalian nicotinic acetylcholine receptors: From structure to function. Physiol. Rev. 2009, 89, 73–120. [Google Scholar] [CrossRef] [PubMed]
- Nirthanan, S.; Gwee, M.C.E. Three-finger-neurotoxins and the nicotinic acetylcholine receptor, forty years on. J. Pharmacol. Sci. 2004, 94, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Kuruppu, S.; Othman, I.; Goode, R.J.A.; Hodgson, W.C.; Isbister, G.K. Neurotoxicity in Sri Lankan Russell’s Viper (Daboia russelii) envenoming is primarily due to U1-viperitoxin-Dr1a, a pre-synaptic neurotoxin. Neurotox. Res. 2017, 31, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Petras, D.; Heiss, P.; Süssmuth, R.D.; Calvete, J.J. Venom proteomics of Indonesian King Cobra, Ophiophagus hannah: Integrating top-down and bottom-up approaches. J. Proteome Res. 2015, 14, 2539–2556. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Tan, K.Y.; Fung, S.Y.; Tan, N.H. Venom-gland transcriptome and venom proteome of the Malaysian king cobra (Ophiophagus hannah). BMC Genom. 2015, 16, 687. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-W.; Liu, B.; Chien, K.-Y.; Chiang, L.-C.; Huang, S.-Y.; Sung, W.-C.; Wu, W.-G. Cobra venom proteome and glycome determined from individual snakes of Naja atra reveal medically important dynamic range and systematic geographic variation. J. Proteom. 2015, 128, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Petras, D.; Sanz, L.; Segura, Á.; Herrera, M.; Villalta, M.; Solano, D.; Vargas, M.; León, G.; Warrell, D.A.; Theakston, R.D.G.; et al. Snake venomics of African spitting cobras: Toxin composition and assessment of congeneric cross-reactivity of the Pan-African EchiTAb-Plus-ICP antivenom by antivenomics and neutralization approaches. J. Proteome Res. 2011, 10, 1266–1280. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Fung, S.Y.; Yap, M.K.K.; Leong, P.K.; Liew, J.L.; Tan, N.H. Unveiling the elusive and exotic: Venomics of the Malayan blue coral snake (Calliophis bivirgata flaviceps). J. Proteom. 2015, 132, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Prasarnpun, S.; Walsh, J.; Harris, J.B. Beta-bungarotoxin-induced depletion of synaptic vesicles at the mammalian neuromuscular junction. Neuropharmacology 2004, 47, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.W.; Harris, J.B. Nerve Terminal Damage by β-Bungarotoxin. Am. J. Pathol. 1999, 154, 447–455. [Google Scholar] [CrossRef]
- Hodgson, W.C.; Wickramaratna, J.C. In Vitro neuromuscular activity of snake venoms. Clin. Exp. Pharmacol. Physiol. 2002, 29, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Rigoni, M.; Paoli, M.; Milanesi, E.; Caccin, P.; Rasola, A.; Bernardi, P.; Montecucco, C. Snake phospholipase A2 neurotoxins enter neurons, bind specifically to mitochondria, and open their transition pores. J. Biol. Chem. 2008, 283, 34013–34020. [Google Scholar] [CrossRef] [PubMed]
- Rigoni, M.; Schiavo, G.; Weston, A.E.; Caccin, P.; Allegrini, F.; Pennuto, M.; Valtorta, F.; Montecucco, C.; Rossetto, O. Snake presynaptic neurotoxins with phospholipase A2 activity induce punctate swellings of neurites and exocytosis of synaptic vesicles. J. Cell Sci. 2004, 117, 3561–3570. [Google Scholar] [CrossRef] [PubMed]
- Pe, T.; Myint, T.; Htut, A.; Htut, T.; Myint, A.A.; Aung, N.N.; Unit, I.C.; Hospital, Y.G.; Division, B.; Township, M. Envenoming by Chinese krait (Bungarus multicinctus) and banded krait (B. fasciatus) in Myanmar. Trans. R. Soc. Trop. Med. Hyg. 1997, 91, 686–688. [Google Scholar] [CrossRef]
- Isbister, G.K.; Brown, S.G.A. Bites in Australian snake handlers—Australian snakebite project (ASP-15). QJM 2012, 105, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Servent, D.; Antil-Delbeke, S.; Gaillard, C.; Corringer, P.; Changeux, J.; Ménez, A. Molecular characterization of the specificity of interactions of various neurotoxins on two distinct nicotinic acetylcholine receptors. Eur. J. Pharmacol. 2000, 393, 197–204. [Google Scholar] [CrossRef]
- Chicheportiche, R.; Vincent, J.P.; Kopeyan, C.; Schweitz, H.; Lazdunski, M. Structure-function relationship in the binding of snake neurotoxins to the torpedo membrane receptor. Biochemistry 1975, 14, 2081–2091. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.Y.; Tan, C.H.; Fung, S.Y.; Tan, N.H. Venomics, lethality and neutralization of Naja kaouthia (monocled cobra) venoms from three different geographical regions of Southeast Asia. J. Proteom. 2015, 120, 105–125. [Google Scholar] [CrossRef] [PubMed]
- Sintiprungrat, K.; Watcharatanyatip, K.; Senevirathne, W.D.S.T.; Chaisuriya, P.; Chokchaichamnankit, D.; Srisomsap, C.; Ratanabanangkoon, K. A comparative study of venomics of Naja naja from India and Sri Lanka, clinical manifestations and antivenomics of an Indian polyspecific antivenom. J. Proteom. 2016, 132, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Marchot, P.; Prowse, C.N.; Kanter, J.; Camp, S.; Ackermann, E.J.; Radic, Z.; Bougis, P.E.; Taylor, P. Expression and Activity of Mutants of Fasciculin, a Peptidic Acetylcholinesterase Inhibitor from Mamba Venom. J. Biol. Chem. 1997, 272, 3502–3510. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.J.; Harvey, A.L. Effects of the potassium channel blocking dendrotoxins on acetylcholine release and motor nerve terminal activity. Br. J. Pharmacol. 1988, 1985, 215–221. [Google Scholar] [CrossRef]
- Chippaux, J.-P.; Goyffon, M. Venoms, antivenoms and immunotherapy. Toxicon 1998, 36, 823–846. [Google Scholar] [CrossRef]
- Maduwage, K.; Isbister, G.K. Current treatment for venom-induced consumption coagulopathy resulting from snakebite. PLoS Negl. Trop. Dis. 2014, 8, e3220. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; León, G.; Lomonte, B. Pharmacokinetic-pharmacodynamic relationships of immunoglobulin therapy for envenomation. Clin. Pharmacokinet. 2003, 42, 721–741. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, D.F.; Nato, F.; England, P.; Ferreira, M.L.; Vaughan, T.J.; Mota, I.; Mazie, J.C.; Choumet, V.; Lafaye, P. Neutralizing human anti crotoxin scFv isolated from a nonimmunized phage library. Scand. J. Immunol. 2000, 51, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Richard, G.; Meyers, A.J.; McLean, M.D.; Arbabi-Ghahroudi, M.; MacKenzie, R.; Hall, J.C. In Vivo neutralization of α-cobratoxin with high-affinity llama single-domain antibodies (VHHs) and a VHH-Fc antibody. PLoS ONE 2013, 8, e69495. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K. Antivenom efficacy or effectiveness: The Australian experience. Toxicology 2010, 268, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K.; O’Leary, M.A.; Schneider, J.J.; Brown, S.G.A.; Currie, B.J. Efficacy of antivenom against the procoagulant effect of Australian brown snake (Pseudonaja sp.) venom: In vivo and in vitro studies. Toxicon 2007, 49, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Maduwage, K.; Silva, A.; O’Leary, M.A.; Hodgson, W.C.; Isbister, G.K. Efficacy of Indian polyvalent snake antivenoms against Sri Lankan snake venoms: Lethality studies or clinically focussed in vitro studies. Sci. Rep. 2016, 6, 26778. [Google Scholar] [CrossRef] [PubMed]
- Pla, D.; Gutiérrez, J.M.; Calvete, J.J. Second generation snake antivenomics: Comparing immunoaffinity and immunodepletion protocols. Toxicon 2012, 60, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Rusmili, M.; Yee, T.; Mustafa, M.; Othman, I.; Hodgson, W. In Vitro Neurotoxicity of Two Malaysian Krait Species (Bungarus candidus and Bungarus fasciatus) Venoms: Neutralization by Monovalent and Polyvalent Antivenoms from Thailand. Toxins (Basel) 2014, 6, 1036–1048. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Hodgson, W.; Isbister, G. Cross-neutralisation of in vitro neurotoxicity of Asian and Australian snake neurotoxins and venoms by different antivenoms. Toxins (Basel) 2016, 8, 302. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.C.; Kuruppu, S.; Smith, A.I.; Reeve, S.; Hodgson, W.C. Isolation and pharmacological characterisation of hostoxin-1, a postsynaptic neurotoxin from the venom of the Stephen’s banded snake (Hoplocephalus stephensi). Neuropharmacology 2006, 51, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Kuruppu, S.; Reeve, S.; Banerjee, Y.; Kini, R.M.; Smith, A.I.; Hodgson, W.C. Isolation and pharmacological characterization of cannitoxin, a presynaptic neurotoxin from the venom of the Papuan Taipan (Oxyuranus scutellatus canni). J. Pharmacol. Exp. Ther. 2005, 315, 1196–1202. [Google Scholar] [CrossRef] [PubMed]
- Kuruppu, S.; Reeve, S.; Smith, A.I.; Hodgson, W.C. Isolation and pharmacological characterisation of papuantoxin-1, a postsynaptic neurotoxin from the venom of the Papuan black snake (Pseudechis papuanus). Biochem. Pharmacol. 2005, 70, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Blacklow, B.; Escoubas, P.; Nicholson, G.M. Characterisation of the heterotrimeric presynaptic phospholipase A2 neurotoxin complex from the venom of the common death adder (Acanthophis antarcticus). Biochem. Pharmacol. 2010, 80, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Alape-Giron, A.; Stiles, B.G.; Gutierrez, J.M. Antibody-mediated neutralization and binding-reversal studies on alpha-neurotoxins from Micrurus nigrocinctus nigrocinctus (coral snake) venom. Toxicon 1996, 34, 369–380. [Google Scholar] [CrossRef]
- Boulain, J.C.; Ménez, A. Neurotoxin-specific immunoglobulins accelerate dissociation of the neurotoxin-acetylcholine receptor complex. Science 1982, 217, 732–733. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO). Guidelines for the Production Control and Regulation of Snake Antivenom Immunoglobulins; World Health Organisation (WHO): Geneva, Switzerland, 2010; pp. 1–134. [Google Scholar]
- Ishikawa, Y.; Kano, M.; Tamiya, N.; Shimada, Y. Acetylcholine receptors of human skeletal muscle: A species difference detected by snake neurotoxins. Brain Res. 1985, 346, 82–88. [Google Scholar] [CrossRef]
- Leong, P.K.; Tan, N.H.; Fung, S.Y.; Sim, S.M. Cross neutralisation of Southeast Asian cobra and krait venoms by Indian polyvalent antivenoms. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Kornhauser, R.; Isbister, G.K.; O’Leary, M.A.; Mirtschin, P.; Dunstan, N.; Hodgson, W.C. Cross-Neutralisation of the neurotoxic effects of Egyptian cobra venom with commercial tiger snake antivenom. Basic Clin. Pharmacol. Toxicol. 2013, 112, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K. Snake antivenom research: The importance of case definition. Emerg. Med. J. 2005, 22, 399–400. [Google Scholar] [CrossRef] [PubMed]
- Ariaratnam, A.; Sjostrom, L.; Raziek, Z.; Kularatne, S.; Kodikara Arachchi, R.W.K.; Sheriff, M.; Theakston, R.; Warrell, D. An open, randomized comparative trial of two antivenoms for the treatment of envenoming by Sri Lankan Russell’s viper (Daboia russelii russelii). Trans. R. Soc. Trop. Med. Hyg. 2001, 95, 74–80. [Google Scholar] [CrossRef]
- Dart, R.C.; Seifert, S.A.; Boyer, L.V.; Clark, R.F.; Hall, E.; McKinney, P.; McNally, J.; Kitchens, C.S.; Curry, S.C.; Bogdan, G.M.; et al. A randomized multicenter trial of crotalinae polyvalent immune Fab (Ovine) antivenom for the treatment for crotaline snakebite in the United States. Arch. Intern. Med. 2001, 161, 2031–2036. [Google Scholar] [CrossRef]
- Holstege, C.P.; Miller, M.; Wermuth, M.; Furbee, B.; Curry, S.C. Crotalid snake envenomation. Crit. Care Clin. 1997, 13, 889–921. [Google Scholar] [CrossRef]
- Tariang, D.; Phillip, P.; Alexander, G.; Jeyaseelan, L.; Peter, J.; Cherian, A. Randomized controlled trial on the effective dose of anti-snake venom in cases of snake bite with systemic envenomation. J. Assoc. Physicians India 1999, 47, 369–371. [Google Scholar] [PubMed]
- Sellahewa, K.; Kumararatne, M.; Dassanayake, P.; Wijesundara, A. Intravenous immunoglobulin in the treatment of snake bite envenoming. Ceylon Med. J. 1994, 39, 173–175. [Google Scholar] [PubMed]
- Watt, G.; Meade, B.D.; Theakston, R.D.G.; Padre, L.P.; Tuazonl, M.L.; Calubaquib, C.; Santiago, E.; Ranoa, C.P.; Naval, U.S.; No, U.; et al. Comparison of Tensilon and antivenom for the treatment of cobra bite paralysis. Trans. R. Soc. Trop. Med. Hyg. 1989, 83, 570–573. [Google Scholar] [CrossRef]
- Ha, T.H.; Höjer, J.; Trinh, X.K.; Nguyen, T. Du A controlled clinical trial of a novel antivenom in patients envenomed by Bungarus multicinctus. J. Med. Toxicol. 2010, 6, 393–397. [Google Scholar] [PubMed]
- Pochanugool, C.; Limthongkul, S.; Wilde, H. Management of Thai cobra bites with a single bolus of antivenin. Wilderness Environ. Med. 1997, 8, 20–23. [Google Scholar] [CrossRef]
- Agarwal, R.; Aggarwal, A.N.; Gupta, D.; Behera, D.; Jindal, S.K. Low dose of snake antivenom is as effective as high dose in patients with severe neurotoxic snake envenoming. Emerg. Med. J. 2005, 22, 397–399. [Google Scholar] [CrossRef] [PubMed]
- Ariaratnam, C.A.; Sheriff, M.H.R.; Theakston, R.D.G.; Warrell, D.A. Distinctive epidemiologic and clinical features of common krait (Bungarus caeruleus) bites in Sri Lanka. Am. J. Trop. Med. Hyg. 2008, 79, 458–462. [Google Scholar] [PubMed]
- Singh, G.; Pannu, H.S.S.; Chawla, P.S.S.; Malhotra, S. Neuromuscular transmission failure due to common krait (Bungarus caeruleus) envenomation. Muscle Nerve 1999, 22, 1637–1643. [Google Scholar] [CrossRef]
- Hung, T.H.; Du Thi, N.; Hojer, J. Clinical features of 60 consecutive ICU-treated patients envenomed by Bungarus multicinctus. Southeast Asian J. Trop. Med. Public Health 2009, 40, 518–524. [Google Scholar] [PubMed]
- Trinh, K.X.; Khac, Q.L.; Trinh, L.X.; Warrell, D.A.; Xuan, K.; Le, Q.; Xuan, L. Hyponatraemia, rhabdomyolysis, alterations in blood pressure and persistent mydriasis in patients envenomed by Malayan kraits (Bungarus candidus) in southern Viet Nam. Toxicon 2010, 56, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Wongtongkam, N.; Wilde, H.; Sitthi-Amorn, C.; Ratanabanangkoon, K. A study of Thai cobra (Naja kaouthia) bites in Thailand. Mil. Med. 2005, 170, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Viravan, C.; Veeravat, U.; Warrell, M.J.; Theakstow, R.D.G.; Warrell, D. ELISA confirmation of acute and past envenoming by the Monocellate Thai Cobra (Naja kaouthia). Am. J. Trop. Med. Hyg. 1986, 35, 173–181. [Google Scholar] [PubMed]
- Trishnananda, M.; Oonsombat, P.; Dumavibhat, B.; Yongchaiyudha, S.; Boonyapisit, V. Clinical manifestations of cobra bite in the Thai farmer. Am. J. Trop. Med. Hyg. 1979, 28, 165–166. [Google Scholar] [PubMed]
- Faiz, M.A.; Ahsan, M.F.; Ghose, A.; Rahman, M.R.; Amin, R.; Hossain, M.; Tareq, M.N.; Jalil, M.A.; Kuch, U.; Theakston, R.D.; et al. Bites by the monocled cobra, Naja kaouthia, in Chittagong division, Bangladesh: Epidemiology, clinical features of envenoming and management of 70 identified cases. Am. J. Trop. Med. Hyg. 2017. pii: 16-0842. [Google Scholar] [CrossRef] [PubMed]
- Kitchens, C.S.; Van Mierop, L.H. Envenomation by the Eastern coral snake (Micrurus fulvius fulvius). A study of 39 victims. JAMA 1987, 258, 1615–1618. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.I.; Ryan, N.M.; O’Leary, M.A.; Brown, S.G.A.; Isbister, G.K. Australian taipan (Oxyuranus spp.) envenoming: Clinical effects and potential benefits of early antivenom therapy—Australian Snakebite Project (ASP-25). Clin. Toxicol. 2017, 55, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Trevett, A.J.; Lalloo, D.G.; Nwokolo, N.C.; Naraqi, S.; Kevau, I.H.; Theakston, R.D.G.; Warrell, D.A. The efficacy of antivenom in the treatment of bites by the Papuan taipan (Oxyuranus scutellatus canni). Trans. R. Soc. Trop. Med. Hyg. 1995, 89, 322–325. [Google Scholar] [CrossRef]
- Lalloo, D.G.; Treveti, A.J.; Korinhona, A.; Laurenson, I.A.N.F.; Paul, M.; Black, J.; Naraqi, S.; Mavo, B.; Sawer, A.; Hutfon, R.O.N.A.; et al. Snake bites by the Papuan Taipan (Oxyuranus scutellatus canni): Paralysis, Haemostatic and Electrocardiographic abnormalities, and effects of antivenom. Am. J. Trop. Med. Hyg. 1995, 52, 525–531. [Google Scholar] [PubMed]
- Scop, J.; Little, M.; Jelinek, G.A.; Daly, F.F.S. Sixteen years of severe tiger snake (Notechis) envenoming in Perth, Western Australia. Anaesth. Intensive Care 2009, 37, 613–618. [Google Scholar] [PubMed]
- Gan, M.; O’Leary, M.A.; Brown, S.G.A.; Jacoby, T.; Spain, D.; Tankel, A.; Gavaghan, C.; Garrett, P.; Isbister, G.K. Envenoming by the rough-scaled snake (Tropidechis carinatus): A series of confirmed cases. Med. J. Aust. 2009, 191, 183–186. [Google Scholar] [PubMed]
- Lalloo, D.G.; Trevett, A.J.; Black, J.; Mapao, J.; Saweri, A.; Naraqi, S.; Owens, D.; Kamiguti, A.S.; Hutton, R.A.; Theakston, R.D.; et al. Neurotoxicity, anticoagulant activity and evidence of rhabdomyolysis in patients bitten by death adders (Acanthophis sp.) in southern Papua New Guinea. QJM 1996, 89, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Lalloo, D.; Trevett1, A.; Black, J.; Mapao, J.; Naraqi, S.; Owens, D.; Hutton, R.; Theakston, D.; Warrell, D. Neurotoxicity and haemostatic disturbances in patients envenomed by the Papuan Black Snake (Pseudechis papuanus). Toxicon 1994, 32, 927–936. [Google Scholar] [CrossRef]
- Phillips, R.E.; Theakston, R.D.G.; Warrell, D.A.; Galigedara, Y.; Aloysius, D.J. Paralysis, rhabdomyolysis and haemolysis caused by bites of Russell’s viper (Vipera russelli pulchella) in Sri Lanka: Failure of Indian (Haffkine) antivenom. Q. J. Med. 1988, 68, 691–716. [Google Scholar] [PubMed]
- Kularatne, S.A.M. Epidemiology and clinical picture of the Russell’s viper (Daboia russelli russelli) bite in Anuradhapura, Sri Lanka: A prospective study of 336 patients. Southeast Asian J. Trop. Med. Public Health 2000, 34, 855–862. [Google Scholar]
- Kularatne, K.; Budagoda, S.; Maduwage, K.; Naser, K.; Kumarasiri, R.; Kularatne, S. Parallels between Russell’s viper (Daboia russelii) and hump-nosed viper (Hypnale species) bites in the central hills of Sri Lanka amidst the heavy burden of unidentified snake bites. Asian Pac. J. Trop. Med. 2011, 4, 564–567. [Google Scholar] [CrossRef]
- Azevedo-Marques, M.M.; Hering, S.E.; Cupo, P. Evidence that Crotalus durissus terrificus (South American rattlesnake) envenomation in humans causes myolysis rather than hemolysis. Toxicon 1987, 25, 1163–1168. [Google Scholar] [CrossRef]
- Cupo, P.; de Azevedo-Marques, M.M.; Hering, S.E. Absence of myocardial involvement in children victims of Crotalus durissus terrificus envenoming. Toxicon 2003, 42, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Hardy, D.L. Envenomation by the Mojave rattlesnake (Crotalus scutulatus scutulatus) in southern Arizona, U.S.A. Toxicon 1983, 21, 111–118. [Google Scholar] [CrossRef]
- Dart, R.C.; Seifert, S.A.; Carroll, L.; Clark, R.F.; Hall, E.; Boyer-Hassen, L.V.; Curry, S.C.; Kitchens, C.S.; Garcia, R.A. Affinity-purified, mixed monospecific crotalid antivenom ovine Fab for the treatment of crotalid venom poisoning. Ann. Emerg. Med. 1997, 30, 33–39. [Google Scholar] [CrossRef]
- Agarwal, R.; Singh, A.P.; Aggarwal, A.N.; Agarwal, R.; Singh, A.P.; Aggarwal, A.N. Pulmonary oedema complicating snake bite due to Bungarus caeruleus. Singap. Med. J. 2007, 48, 227–230. [Google Scholar]
- Blaylock, R.S.; Lichtman, A.R.; Potgieter, P.D. Clinical manifestations of Cape cobra (Naja nivea) bites. A report of 2 cases. S. Afr. Med. J. 1985, 68, 342–344. [Google Scholar] [PubMed]
- Warrell, D.A.; Barnes, H.J.; Piburn, M.F. Neurotoxic effects of bites by the Egyptian cobra (Naja haje) in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 1976, 70, 78–79. [Google Scholar] [CrossRef]
- Ronan-Bentle, S.E.; Bryant, S.M.; Williams, J.B. Naja kaouthia envenomation in the Midwest. Vet. Hum. Toxicol. 2004, 46, 181–182. [Google Scholar] [PubMed]
- Pochanugool, C.; Wilde, H.; Jitapunkul, S. Spontaneous recovery from severe neurotoxic envenoming by a Malayan krait Bungarus candidus (Linnaeus) in Thailand. Wilderness Environ. Med. 1997, 8, 223–225. [Google Scholar] [CrossRef]
- Leeprasert, W.; Kaojarem, S. Specific Antivenom for Bungarus candidus. J. Med. Assoc. Thail. 2007, 90, 1467–1476. [Google Scholar]
- Kanchanapongkul, J. Neurotoxic Envenoming Following Bites by the Malayan Krait (Bungarus candidus). J. Med. Assoc. Thail. 2002, 85, 945–947. [Google Scholar]
- Pettigrew, L.C.; Glass, J.P. Neurologic complications of a coral snake bite. Neurology 1985, 35, 589–592. [Google Scholar] [CrossRef] [PubMed]
- Kulawickrama, S.; O’Leary, M.A.; Hodgson, W.C.; Brown, S.G.; Jacoby, T.; Davern, K.; Isbister, G.K. Development of a sensitive enzyme immunoassay for measuring taipan venom in serum. Toxicon 2010, 55, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
| Study | Number in Each Arm | Snake Species | Authentication of Snake | Trial Arms | Blinded | Randomisation | Allocation Concealment | AV ** Dose Defined | Primary Outcome Defined | Neurotoxicity Measures for Outcome | Conclusion |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ariaratnam et al. 2001 | 23/20 | Daboia russelii | Identification of snake specimen and ELISA * (n = 43); clinical features (n = 3) | 2 different AV (2 arms) | No | Yes | Good | Yes | Multiple outcomes | Duration of neurological signs (ptosis, diplopia) | No difference between groups |
| Dart et al. 2001 | 16/15 | North American Crotalids | Not stated | 2 doses of AV (2 arms) | No | Yes | Good | Yes | Yes (Improvement of a defined severity score) | Weakness Fasciculations Dizziness Paraesthesia (included to the severity score) | Both doses equally effective |
| Sellahewa et al. | 8/7 | Naja naja (n = 3) Daboia russelli (n = 11) Hypnale hypnale (n = 1) | Identification of specimen (n = 10), clinical features (n = 5) | AV vs. AV + Intravenous immunoglobulin (IVIG) | No | Yes | Good | Yes | No | Duration of neurotoxic features (ptosis and ophthalmoplegia) | No clear difference except more re-dosing in AV group. |
| Tariang et al. 1999 | 31/29 | ‘Cobra’, ‘viper’ | Not stated | Two doses of AV | Yes | Yes | Not described | Yes | No | Not defined | Lower dose is effective than higher dose |
| Watt et al. 1989 | 2/2/4/8 (Cross-over) | Philippine cobra (Naja philippinensis) | ELISA (n = 5); Identification from pictures (n = 2); clinical features (n = 1) | Three different doses of AV and all patients received edrophonium | No | No | No | Yes | Multiple outcomes | Improvement of neurological signs | Tensilon is effective compared to AV |
| Study | Number in Each Group | Snake Species | Authentication of Snake | Study Groups | Antivenom Dose Defined | Neurotoxicity Measures | Conclusion | Remarks |
|---|---|---|---|---|---|---|---|---|
| Agarwal et al. 2005 | 28/27 | Not defined | Not defined. | High vs. low dose AV | Yes | Duration of the mechanical ventilation, duration of ICU stay | No difference in outcome between the two groups | Same AV loading dose was given to both groups. The difference was only the maintenance dose. |
| Hung et al. 2010 | 27/54 | Bungarus multicinctus | Not defined | AV vs. no AV | Yes | Number of patients requiring mechanical ventilation, duration of mechanical ventilation, length of stay in the ICU, duration of a defined degree of muscle paralysis | AV group had shorter duration of ventilation, ICU stay and other neurological signs. No difference between the number of patients requiring ventilation. | The no AV group is a historical group; AV dose varied within the AV group; Bite-to-AV delay is 19 ± 9 h (range: 5–38 h). |
| Pochanugool et al. 1997 | 27/41 | Naja kaouthia Naja sumatrana | Patient’s description of snake or physician’s identification of the snake | No AV vs. AV (three unbalanced dose groups within AV group) | Yes | Duration of respiratory failure | Two dose groups of antivenom (100 and 200 mL) had significantly lower duration of respiratory failure compared to no AV | The no AV group is a historical group; No definition of ‘respiratory paralysis’. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, A.; Hodgson, W.C.; Isbister, G.K. Antivenom for Neuromuscular Paralysis Resulting From Snake Envenoming. Toxins 2017, 9, 143. https://doi.org/10.3390/toxins9040143
Silva A, Hodgson WC, Isbister GK. Antivenom for Neuromuscular Paralysis Resulting From Snake Envenoming. Toxins. 2017; 9(4):143. https://doi.org/10.3390/toxins9040143
Chicago/Turabian StyleSilva, Anjana, Wayne C. Hodgson, and Geoffrey K. Isbister. 2017. "Antivenom for Neuromuscular Paralysis Resulting From Snake Envenoming" Toxins 9, no. 4: 143. https://doi.org/10.3390/toxins9040143
APA StyleSilva, A., Hodgson, W. C., & Isbister, G. K. (2017). Antivenom for Neuromuscular Paralysis Resulting From Snake Envenoming. Toxins, 9(4), 143. https://doi.org/10.3390/toxins9040143







