Changes in the Fusarium Head Blight Complex of Malting Barley in a Three-Year Field Experiment in Italy
Abstract
:1. Introduction
2. Results
2.1. Presence of Mycotoxigenic Fungal Microorganisms in Malting Barley Grains in Relation to Climatic Conditions
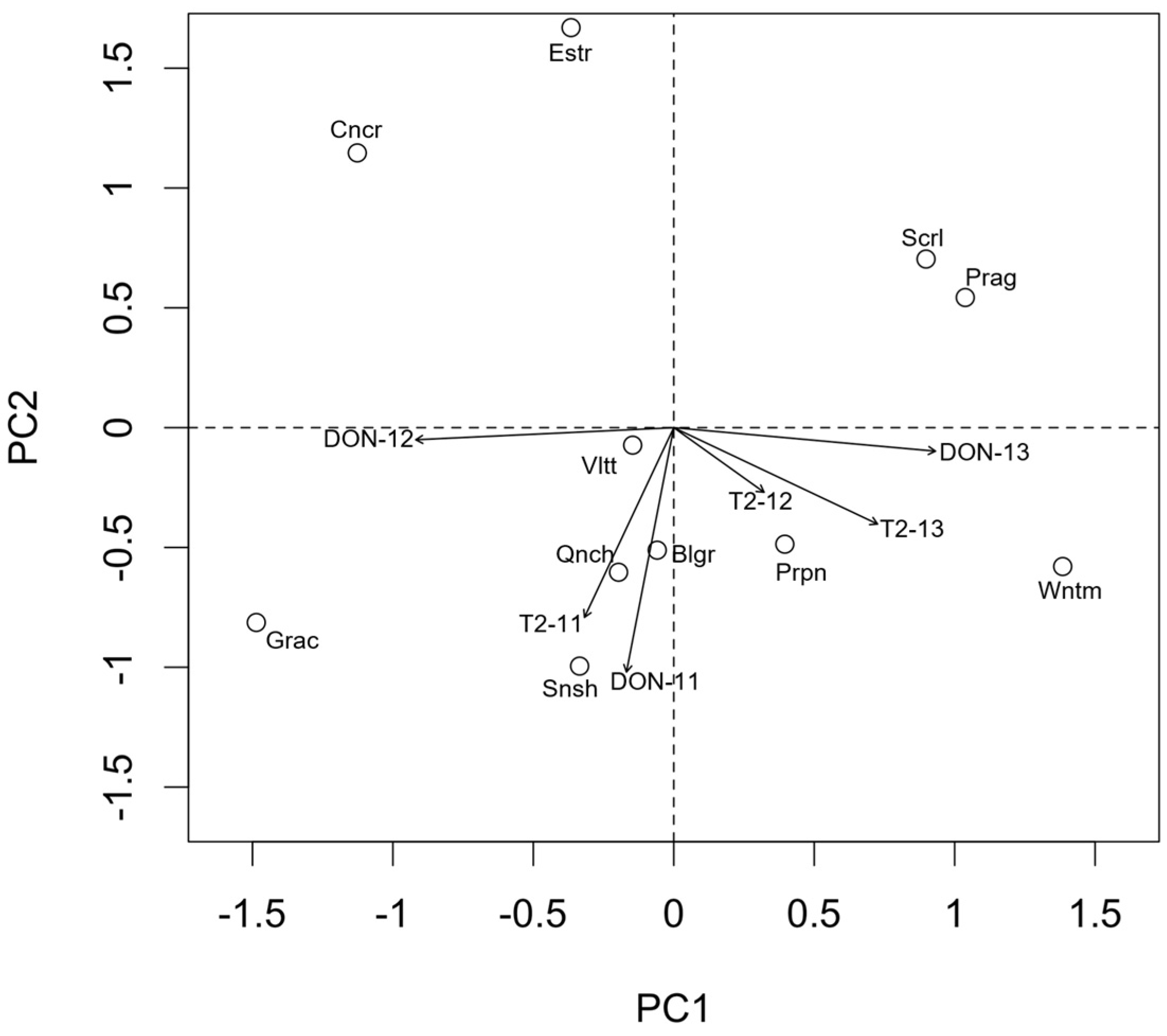
2.2. Identification of Fusarium spp. Isolated from the Kernels, Chemotype Characterization and Their Association with Malting Barley Varieties
2.3. Occurrence of Deoxynivalenol and T-2 Toxin in the Grain
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Barley Sampling, Weather Data and Anthesis Time Collection
5.2. Mycological Seed Analysis and Obtainment of Fusarium Strains
5.3. DNA Extraction, Species Identification and Chemotype Characterization
5.4. Deoxynivalenol and T-2 Toxin Analysis
5.5. Statistical Analysis
5.5.1. Presence of Fungal Microorganisms in Malting Barley Grains in Relation to Climatic Conditions
5.5.2. Identification of Fusarium spp. and Their Association with Malting Barley Varieties
5.5.3. Occurrence of Deoxynivalenol and T-2 toxin in the Grain
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food and Agriculture Organization (FAO). FAOSTAT. Statistic Division. Food and Agriculture Organization of the UN. Database 2013. Available online: http://faostat.fao.org (accessed on 28 February 2017).
- Oliveira, P.M.; Mauch, A.; Jacob, F.; Waters, D.M.; Arendt, E.K. Fundamental study on the influence of Fusarium infection on quality and ultrastructure of barley malt. Int. J. Food Microbiol. 2012, 156, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Laitila, A.; Kotaviita, E.; Peltola, P.; Home, S.; Wilhelmson, A. Indigenous microbial community of barley greatly influences grain germination and malt quality. J. Inst. Brew. 2007, 113, 9–20. [Google Scholar] [CrossRef]
- Sarlin, T.; Laitila, A.; Pekkarinen, A.; Haikara, A. Effects of three Fusarium species on the quality of barley and malt. J. Am. Soc. Brew. Chem. 2005, 63, 43–49. [Google Scholar]
- Wolf-Hall, C.E. Mold and mycotoxin problems encountered during malting and brewing. Int. J. Food Microbiol. 2007, 119, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Christian, M.; Titze, J.; Ilberg, V.; Jacob, F. Novel Perspectives in Gushing Analysis: A Review. J. Inst. Brew. 2011, 117, 295–313. [Google Scholar] [CrossRef]
- Oliveira, P.M.; Waters, D.M.; Arendt, E.K. The impact of Fusarium culmorum infection on the protein fractions of raw barley and malted grains. Appl. Microbiol. 2013, 97, 2053–2065. [Google Scholar] [CrossRef] [PubMed]
- Bertuzzi, T.; Rastelli, S.; Mulazzi, A.; Donadini, G.; Pietri, A. Mycotoxin occurrence in beer produced in several European countries. Food Control 2011, 22, 2059–2064. [Google Scholar] [CrossRef]
- Lancova, K.; Hajslova, J.; Poustka, J.; Krplova, A.; Zachariasova, M.; Dostalek, P.; Sachambula, L. Transfer of Fusarium mycotoxins and ‘masked’ deoxynivalenol (deoxynivalenol-3-glucoside) from field barley through malt to beer. Food Addit. Contam. 2008, 25, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (EC). Commission Regulation (EC) No. 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- Rodríguez-Carrasco, Y.; Fattore, M.; Albrizio, S.; Berrada, H.J.M. Occurrence of Fusarium mycotoxins and their dietary intake through beer consumption by the European population. Food Chem. 2015, 178, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Parry, D.W.; Jenkinson, P.; Mcleod, L. Fusarium Ear Blight (Scab) in Small-Grain Cereals—A Review. Plant Pathol. 1995, 44, 207–238. [Google Scholar] [CrossRef]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Pancaldi, D.; Tonti, S.; Prodi, A.; Salomoni, D.; Dal Pra, M.; Nipoti, P.; Alberti, I.; Pisi, A. Survey of the main causal agents of Fusarium head blight of durum wheat around Bologna, Northern Italy. Phytopathol. Mediterr. 2010, 49, 258–266. [Google Scholar]
- Pereyra, S.A.; Dill-Macky, R.; Sims, A.L. Survival and inoculum production of Gibberella zeae in wheat residue. Plant Dis. 2004, 88, 724–730. [Google Scholar] [CrossRef]
- Langseth, W.; Kosiak, B.; Clasen, P.E.; Torp, M.; Gareis, M. Toxicity and occurrence of Fusarium species and mycotoxins in late harvested and overwintered grain from Norway, 1993. J. Phytopathol. 1997, 145, 409–416. [Google Scholar] [CrossRef]
- Haidukowski, M.; Visconti, A.; Perrone, G.; Vanadia, S.; Pancaldi, D.; Covarelli, L.; Balestrazzi, R.; Pascale, M. Effect of prothioconazole-based fungicides on Fusarium head blight, grain yield and deoxynivalenol accumulation in wheat under field conditions. Phytopathol. Mediterr. 2012, 51, 236–246. [Google Scholar]
- Xu, X.M.; Nicholson, P.; Thomsett, M.A.; Simpson, D.; Cooke, B.M.; Doohan, F.M.; Brennan, J.; Monaghan, S.; Moretti, A.; Mulé, G.; et al. Relationship between the fungal complex causing Fusarium head blight of wheat and environmental conditions. Phytopathology 2008, 98, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Bernhoft, A.; Torp, M.; Clasen, P.E.; Loes, A.K.; Kristoffersen, A.B. Influence of agronomic and climatic factors on Fusarium infestation and mycotoxin contamination of cereals in Norway. Food Addit. Contam. 2012, 29, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; Beccari, G.; Prodi, A.; Generotti, S.; Etruschi, F.; Ferrer, E.; Mañes, J. Fusarium species, chemotype characterisation and trichothecene contamination of durum and soft wheat in an area of central Italy. J. Sci. Food Agric. 2015, 95, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; Beccari, G.; Prodi, A.; Generotti, S.; Etruschi, F.; Meca, G.; Juan, C.; Mañes, J. Biosynthesis of beauvericin and enniatins in vitro by wheat Fusarium species and natural grain contamination in an area of central Italy. Food Microbiol. 2015, 46, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Covarelli, L.; Beccari, G.; Colasante, V.; Mañes, J. Simultaneous analysis of twenty-six mycotoxins in durum wheat grain from Italy. Food Control 2016, 62, 322–329. [Google Scholar] [CrossRef]
- Commission Recommendation (EC). Commission Recommendation (EC) 2013/165 of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereal and cereals products (2013). Off. J. Eur. Union 2013, L91, 12–14. [Google Scholar]
- Beccari, G.; Caproni, L.; Tini, F.; Uhlig, S.; Covarelli, L. Presence of Fusarium species and other toxigenic fungi in malting barley and multi-mycotoxin analysis by liquid chromatography-high-resolution mass spectrometry. J. Agric. Food Chem. 2016, 64, 4390–4399. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez Pereyra, M.L.; Rosa, C.A.R.; Dalcero, A.M.; Cavaglieri, L.R. Mycobiota and mycotoxins in malted barley and brewer’s spent grain from Argentinean breweries. Lett. Appl. Microbiol. 2011, 53, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Andersen, B.; Thrane, U.; Svendsen, A.; Rasmussen, I.A. Associated field mycobiota on malt barley. Can. J. Bot. 1996, 74, 854–858. [Google Scholar] [CrossRef]
- Rohacik, T.; Hudec, K. Fungal infection of malt barley kernels in Slovak Republic. Plant. Prot. Sci. 2007, 43, 86–93. [Google Scholar]
- Medina, A.; Valle-Algarra, F.M.; Mateo, R.; Gimeno-Adelantado, J.V.; Mateo, F.; Jiménez, M. Survey of the mycobiota of Spanish malting barley and evaluation of the mycotoxin producing potential of species of Alternaria, Aspergillus and Fusarium. Int. J. Food Microbiol. 2006, 108, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Moschini, R.C.; Fortugno, C. Predicting wheat head blight incidence using models based on meteorological factors in Pergamino, Argentina. Eur. J. Plant Pathol. 1996, 102, 211–218. [Google Scholar] [CrossRef]
- Lacey, J.; Bateman, G.L.; Mirocha, C.J. Effects of infection time and moisture on development of ear blight and deoxynivalenol production by Fusarium spp. in wheat. Ann. Appl. Biol. 1999, 134, 277–283. [Google Scholar] [CrossRef]
- De Wolf, E.D.; Madden, L.V.; Lipps, P.E. Risk assessment models for wheat Fusarium head blight epidemics based on within-season weather data. Phytopathology 2003, 93, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.M.; Monger, W.; Ritieni, A.; Nicholson, P. Effect of temperature and duration of wetness during initial infection periods on disease development, fungal biomass and mycotoxin concentrations on wheat inoculated with single, or combinations of, Fusarium species. Plant Pathol. 2007, 56, 943–956. [Google Scholar] [CrossRef]
- Rossi, V.; Ravanetti, A.; Pattori, E.; Giosuè, S. Influence of temperature and humidity on the infection of wheat spikes by some fungi causing Fusarium head blight. J. Plant Pathol. 2001, 83, 189–198. [Google Scholar]
- Xu, X.M. Effects of environmental conditions on the development of Fusarium ear blight. Eur. J. Plant Pathol. 2003, 109, 683–689. [Google Scholar] [CrossRef]
- Brennan, J.M.; Egan, D.; Cooke, B.M.; Doohan, F.M. Effect of temperature on head blight of wheat caused by Fusarium culmorum and F. graminearum. Plant Pathol. 2005, 54, 156–160. [Google Scholar] [CrossRef]
- Nielsen, L.K.; Cook, D.J.; Edwards, S.G.; Ray, R.V. The prevalence and impact of Fusarium head blight pathogens and mycotoxins on malting barley quality in UK. Int. J. Food Microbiol. 2014, 179, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Brennan, J.M.; Fagan, B.; van Maanen, A.; Cooke, B.M.; Doohan, F.M. Studies on in vitro growth and pathogenicity of European Fusarium fungi. Eur. J. Plant Pathol. 2003, 109, 577–587. [Google Scholar] [CrossRef]
- Yli-Mattila, T.; Paavanen-Huhtala, S.; Parikka, P.; Konstantinova, P.; Gagkaeva, T.Y. Molecular and morphological diversity of Fusarium species in Finland and north-western Russia. Eur. J. Plant Pathol. 2004, 110, 573–585. [Google Scholar] [CrossRef]
- Uhlig, S.; Jestoi, M.; Parikka, P. Fusarium avenaceum—The North European situation. Int. J. Food Microbiol. 2007, 119, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Lysoe, E.; Harris, L.J.; Walkowiak, S.; Subramaniam, R.; Divon, H.H.; Riiser, E.S.; Llorens, C.; Gabaldon, T.; Kistler, H.C.; Jonkers, W.; et al. The genome of the generalist plant pathogen Fusarium avenaceum is enriched with genes involved in redox, signaling and secondary metabolism. PLoS ONE 2014, 9, e112703. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.M.; Nicholson, P. Community ecology of fungal pathogens causing wheat head blight. Annu. Rev. Phytopathol. 2009, 47, 83–103. [Google Scholar] [CrossRef] [PubMed]
- Thrane, U.; Adler, A.; Clasen, P.E.; Galvano, F.; Langseth, W.; Lew, H.; Logrieco, A.; Frog Nielsen, K.; Ritieni, A. Diversity in metabolite production by Fusarium langsethiae, Fusarium poae and Fusarium sporotrichioides. Int. J. Food Microbiol. 2004, 95, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Jestoi, M.N.; Paavanen-Huntala, S.; Parikka, P.; Yli-Mattila, T. In vitro and in vivo mycotoxin production of Fusarium species isolated from Finnish grains. Food Addit. Contam. 2008, 41, 545–558. [Google Scholar]
- Schutt, F.; Nirenberg, H.I.; Deml, G. Moniliformin production in the genus Fusarium. Mycotoxins Res. 1998, 14, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Rizzo, A.; Ferracane, R.; Ritieni, A. Occurrence of beauvericin and enniatins in wheat affected by Fusarium avenaceum head blight. Appl. Environ. Microbiol. 2002, 68, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.M. Recent research on fumonisins: A review. Food Addit. Contam. 2012, 29, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Prodi, A.; Purahong, W.; Tonti, S.; Salomoni, D.; Nipoti, P.; Covarelli, L.; Pisi, A. Difference in chemotype composition of Fusarium graminearum populations isolated from durum wheat in adjacent areas separated by the Apennines in Northern-Central Italy. Plant Pathol. J. 2011, 27, 354–359. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Ciasca, B.; Terzi, V.; Ghizzoni, R.; McCormick, S.P.; Pascale, M. Study of the natural occurrence of T-2 and HT-2 toxins and their glucosyl derivatives from field barley to malt by high-resolution Orbitrap mass spectrometry. Food Addit. Contam. 2015, 32, 2647–1655. [Google Scholar] [CrossRef] [PubMed]
- Berguete, E.M. Fusarium spp. Mycotoxins in the European Malting Barley Chain; RIKILT Wageningen UR: Wageningen, The Netherlands, 2012; p. 45. [Google Scholar]
- Van der Fels-Klerx, H.J.; Stratakou, I. T-2 toxin and HT-2 toxin in grain and grain-based commodities in Europe occurrence, factors affecting occurrence, co-occurrence and toxicological effects. World Mycotoxin J. 2010, 3, 349–367. [Google Scholar] [CrossRef]
- Malachova, A.; Cerkal, R.; Ehrenbergerova, J.; Dzuman, Z.; Vaculova, K.; Hajslova, J. Fusarium mycotoxins in various barley cultivars and their transfer into malt. J. Sci. Food Agric. 2010, 90, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V.M.T.; Ciasca, B.; Haidukowski, M.; Infantino, A.; Visconti, A.; Pascale, M. Mycotoxin profile of Fusarium langsethiae isolated from wheat in Italy: Production of type-A trichothecenes and relevant glucosyl derivatives. J. Mass. Spectrom. 2013, 48, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- Strub, C.; Pocaznoi, D.; Lebrihi, A.; Fournier, R.; Mathieu, F. Influence of barley malting operating parameters on T-2 and HT-2 toxinogenesis of Fusarium langsethiae, a worrying contaminant of malting barley in Europe. Food Addit. Contam. 2010, 27, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.K.; Jensen, J.D.; Nielsen, G.C.; Jensen, J.E.; Spliid, N.H.; Thomsen, I.K.; Justesen, A.F.; Collinge, D.B.; Jorgensen, L.N. Fusarium Head Blight of Cereals in Denmark: Species Complex and Related Mycotoxins. Phytopathology 2011, 101, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; University of Perugia, Perugia, Italy; Beccari, G.; University of Perugia, Perugia, Italy. Personal communication, 2014.
- Muller, H.M.; Reimann, J.; Schumacher, U.; Schwadorf, K. Occurrence of Fusarium toxins in barley harvested during five years in an area of southwest Germany. Mycopathologia 1997, 137, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Ibanez-Vea, M.; Lizarraga, E.; Gonzalez-Penas, E.; de Cerain, A.L. Co-occurrence of type-A and type-B trichothecenes in barley from a northern region of Spain. Food Control 2012, 25, 81–88. [Google Scholar] [CrossRef]
- Langseth, W.; Rundberget, T. The occurrence of HT-2 toxin and other trichothecenes in Norwegian cereals. Mycopathologia 1999, 147, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Mankeviciene, A.; Butkute, B.; Gaurilcikiene, I.; Dabkevicius, Z.; Suproniene, S. Risk assessment of Fusarium mycotoxins in Lithuanian small cereal grains. Food Control 2011, 22, 970–976. [Google Scholar] [CrossRef]
- Perkowski, J.; Kiecana, I.; Kaczmarek, Z. Natural occurrence and distribution of Fusarium toxins in contaminated barley cultivars. Eur. J. Plant Pathol. 2003, 109, 331–339. [Google Scholar] [CrossRef]
- Edwards, S.G. Fusarium mycotoxin content of UK organic and conventional barley. Food Addit. Contam. 2009, 26, 1185–1190. [Google Scholar] [CrossRef]
- Balmas, V.; Santori, A.; Corazza, L. Le specie di Fusarium più comuni in Italia. Petria 2000, 10 (Suppl. 1), 14–15. [Google Scholar]
- Nicholson, P.; Simpson, D.R.; Weston, G.; Rezanoor, H.N.; Lees, A.K.; Parry, D.W.; Joyce, D. Detection and quantification of Fusarium culmorum and Fusarium graminearum in cereals using PCR assays. Physiol. Mol. Plant Pathol. 1998, 53, 17–37. [Google Scholar] [CrossRef]
- Doohan, F.M.; Parry, D.W.; Jenkinson, P.; Nicholson, P. The use of species specific PCR based assays to analyse Fusarium ear blight of wheat. Plant Pathol. 1998, 47, 197–205. [Google Scholar] [CrossRef]
- Jurado, M.; Vazquez, C.; Patino, B.; Gonzalez-Jaen, M.T. PCR detection assays for the trichothecene-producing species Fusarium graminearum, Fusarium culmorum, Fusarium poae, Fusarium equiseti and Fusarium sporotrichioides. Syst. Appl. Microbiol. 2005, 28, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Simpson, D.; Chandler, E.; Jennings, P.; Nicholson, P. Development of PCR assays for the detection and differentiation of Fusarium sporotrichioides and Fusarium langsethiae. FEMS Microbiol. Lett. 2004, 233, 69–76. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, K.; Kistler, H.C.; Cigelnik, E.; Ploetz, R.C. Multiple evolutionary origins of the fungus causing Panama disease of banana: Concordant evidence from nuclear and mitochondrial gene genealogies. Proc. Natl. Acad. Sci. USA 1998, 95, 2044–2049. [Google Scholar] [CrossRef] [PubMed]
- Geiser, D.M.; Jimenez-Gasco, M.D.; Kang, S.C.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’Donnell, K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479. [Google Scholar]
- FUSARIUM ID. Available online: http://isolate.fusariumdb.org/ (accessed on 20 April 2015).
- Starkey, D.E.; Ward, T.J.; Aoki, T.; Gale, L.R.; Kistler, H.C.; Geiser, D.M.; Suga, H.; Toth, B.; Varga, J.; O’Donnell, K. Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fungal Genet. Biol. 2007, 44, 1191–1204. [Google Scholar] [CrossRef] [PubMed]
- Ward, T.J.; Bielawski, J.P.; Kistler, H.C.; Sullivan, E.; O’Donnell, K. Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proc. Nat. Acad. Sci. USA 2002, 99, 9278–9283. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, K.; Sutton, D.A.; Rinaldi, M.G.; Gueidan, C.; Crous, P.W.; Geiser, D.M. Novel multilocus sequence typing scheme reveals high genetic diversity of human pathogenic members of the Fusarium incarnatum-F. equiseti and F. chlamydosporum species complexes within the United States. J. Clin. Microbiol. 2009, 47, 3851–3861. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.0–10. Available online: http://CRAN.R-project.org/package=vegan (accessed on 7 March 2015).
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2011; Available online: http://www. R-project.org (accessed on 7 March 2015).
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier Science: Amsterdam, The Netherlands, 1998; p. 969. [Google Scholar]
- Gauch, H.G.J. Statistical Analysis of Regional Yield Trials: AMMI Analysis of Factorial Designs; Elsevier Science: Amsterdam, The Netherlands, 1992; p. 278. [Google Scholar]
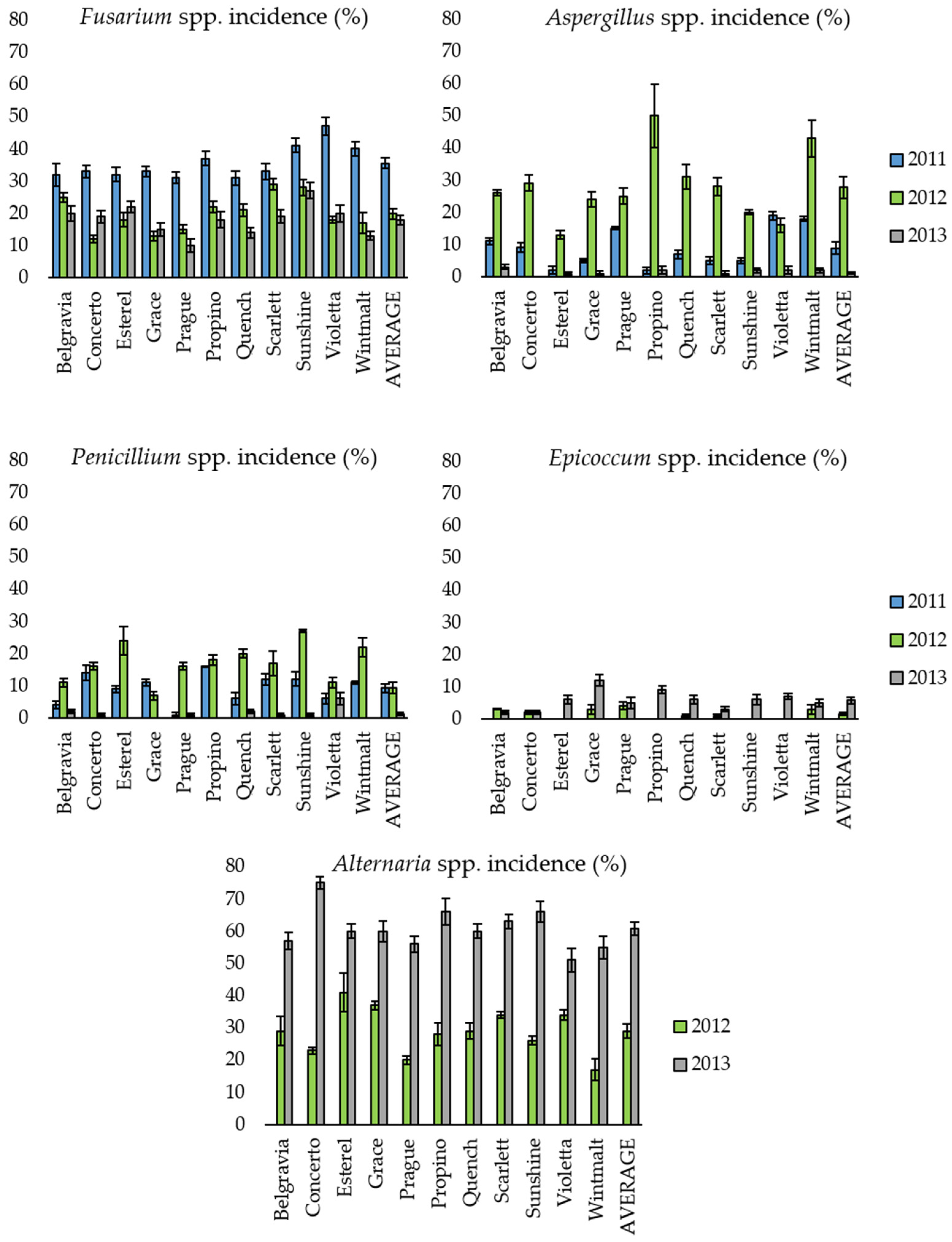
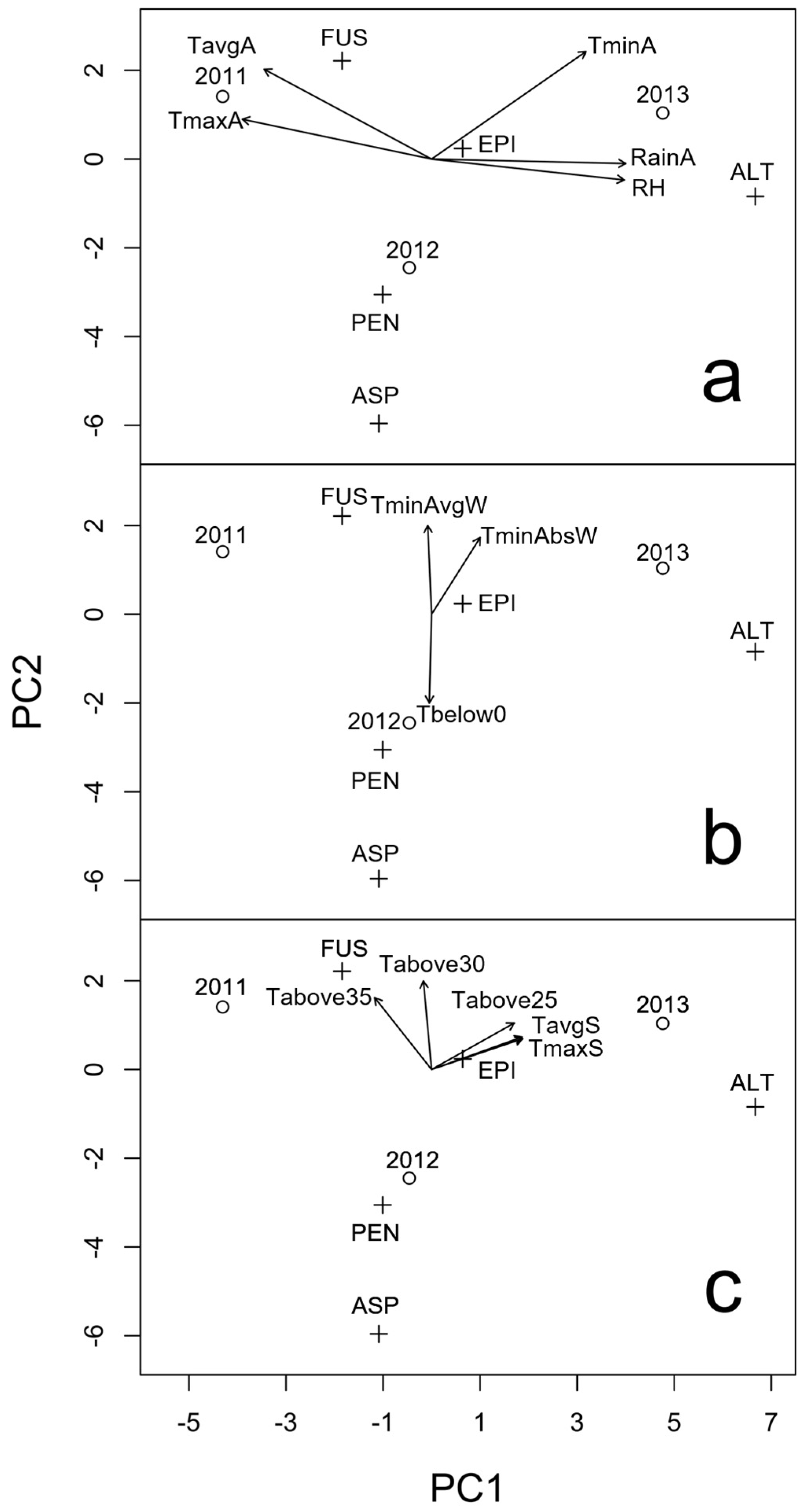
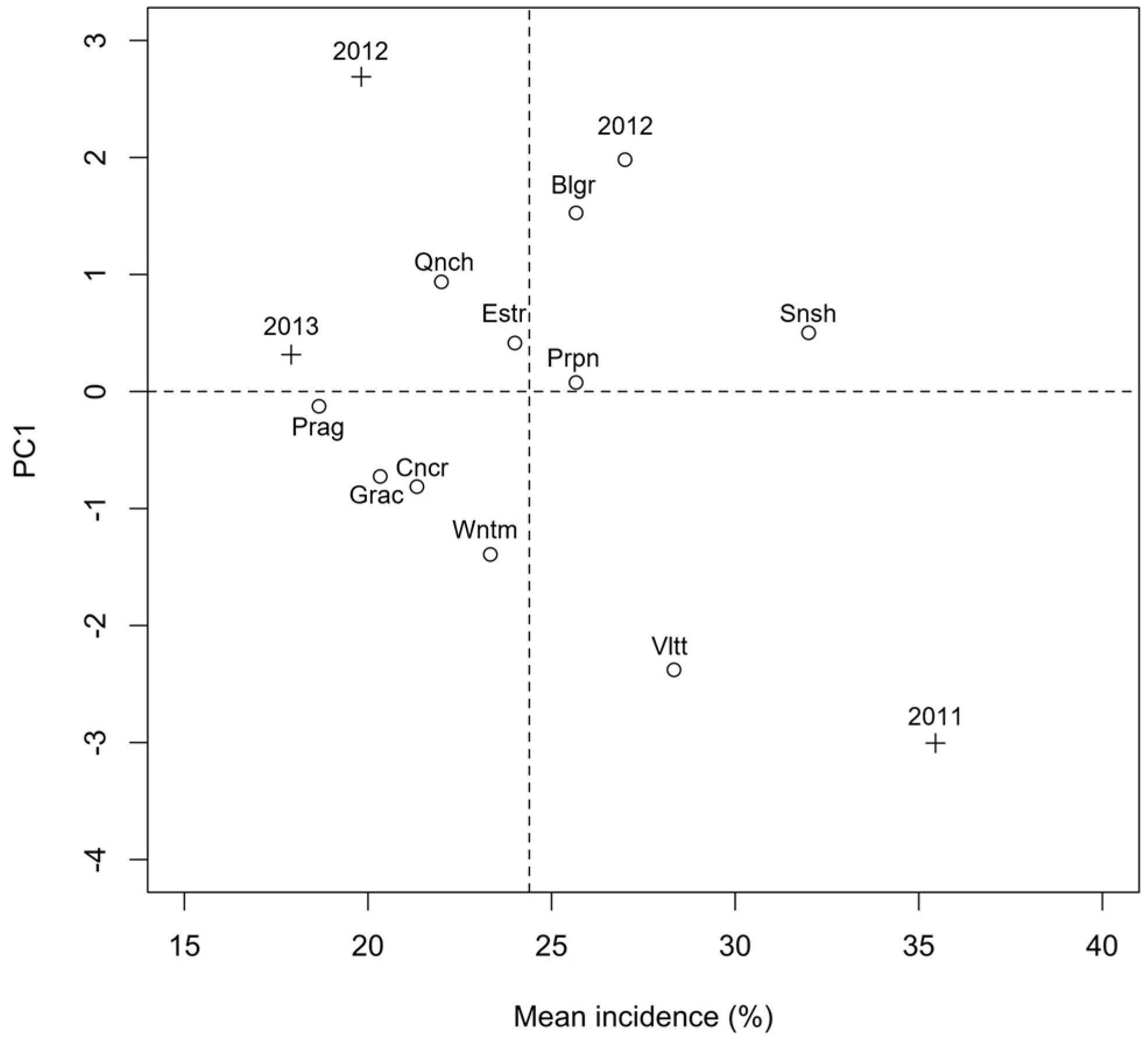
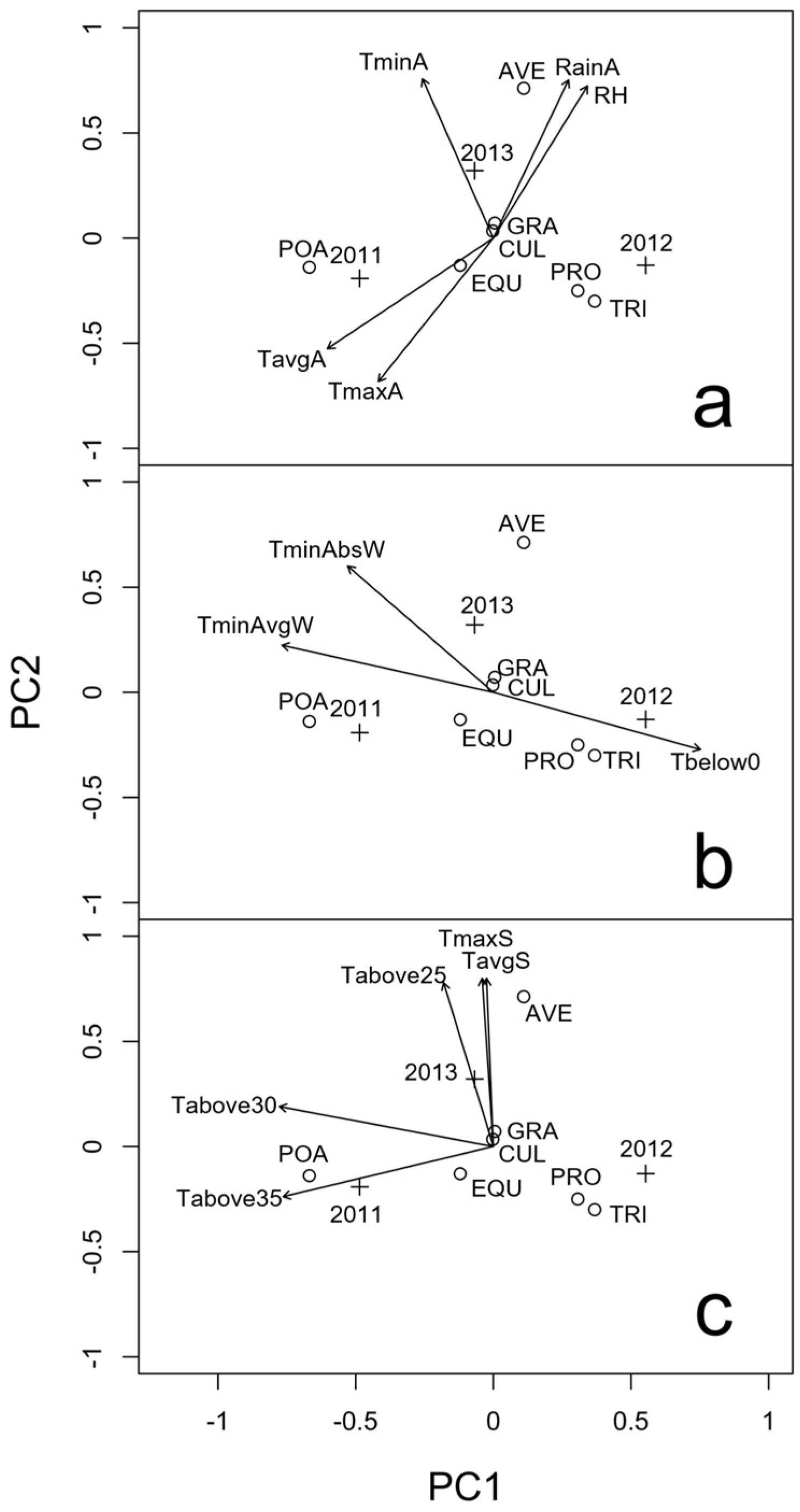
| Time | Variable a | 2011 | 2012 | 2013 |
|---|---|---|---|---|
| Anthesis (May) | TminA (°C) | 8.6 | 8.1 | 10.3 |
| TavgA (°C) | 17.4 | 15.7 | 15.5 | |
| TmaxA (°C) | 25.3 | 23.0 | 21.7 | |
| RH (%) | 65.0 | 74.2 | 82.3 | |
| RainA (mm) | 0.9 | 2.3 | 3.5 | |
| Winter (December–February) | TminAvgW (°C) | 1.0 | −1.1 | 0.7 |
| TminAbsW (°C) | −8.3 | −12.4 | −5.3 | |
| Tbelow0 (n.) | 41 | 55 | 42 | |
| Summer (June–August) b | TmaxS (°C) | 30.5 | 30.8 | 33.4 |
| TavgS (°C ) | 22.6 | 22.8 | 24.5 | |
| Tabove25 (n.) | 79 | 78 | 86 | |
| Tabove30 (n.) | 53 | 31 | 49 | |
| Tabove35 (n.) | 13 | 4 | 7 |
| Year | F. poae | F. avenaceum | F. tricinctum | F. graminearum | F. culmorum | FIESC a | F. proliferatum |
|---|---|---|---|---|---|---|---|
| 2011 | 33 | 1 | 0 | 6 | 0 | 6 | 0 |
| 2012 | 1 | 6 | 12 | 4 | 0 | 0 | 10 |
| 2013 | 23 | 26 | 0 | 8 | 1 | 1 | 0 |
| Varieties | DON b | T-2 Toxin c | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2011 | 2012 | 2013 | 2011 | 2012 | 2013 | |||||||
| µg/kg | ±SE d | µg/kg | ±SE | µg/kg | ±SE | μg/kg | ±SE | μg/kg | ±SE | μg/kg | ±SE | |
| Belgravia | <LOQ a | - | 380.9 | 60.2 | <LOQ | - | 100.3 | 31.8 | 423.7 | 1.2 | 144.3 | 56.4 |
| Concerto | <LOQ | - | 480.5 | 40.3 | <LOQ | - | <LOQ | - | 115 | 4.9 | 87.7 | 11.8 |
| Esterel | <LOQ | - | 370.1 | 60.6 | <LOQ | - | <LOQ | - | <LOQ | - | 54.1 | 0.9 |
| Grace | 270.5 | 30.5 | 470.2 | 70.2 | <LOQ | - | 244.5 | 198.1 | <LOQ | - | 108.1 | 11.1 |
| Prague | <LOQ | - | 300.2 | 20.5 | 700.2 | 60.2 | 133.3 | 91.2 | 199 | 0.7 | 100.7 | 17.4 |
| Propino | <LOQ | - | 370.3 | 60.6 | 500.5 | 40.4 | 147.8 | 7.4 | 63.3 | 8.2 | 163.4 | 29.9 |
| Quench | 280.4 | 10.4 | 440 | 60.2 | 460.6 | 20.2 | 125.4 | 44.1 | 196.8 | 3.6 | 115.4 | 17.7 |
| Scarlett | <LOQ | - | 300.2 | 20.4 | 410.3 | 20.7 | <LOQ | - | 114.1 | 6.9 | 157.6 | 24.4 |
| Sunshine | 280.7 | 40.3 | 320.8 | 20.3 | 320.4 | 40.3 | 285.9 | 123.6 | 117.4 | 0.3 | 62.7 | 8 |
| Violetta | 300.7 | 20.3 | 400.3 | 90.6 | 440.5 | 20.6 | <LOQ | - | <LOQ | - | 104.3 | 23.4 |
| Wintmalt | 250.2 | 60.4 | 340.7 | 50.5 | 610.7 | 10.2 | <LOQ | - | 162.2 | 6.9 | 236.1 | 61.6 |
| Positive (%) | 45 | 100 | 63 | 54 | 73 | 100 | ||||||
| Average (µg/kg) | 125.6 | 43.7 | 379.4 | 19.1 | 313.0 | 80.5 | 94.3 | 31.5 | 126.5 | 37.2 | 121.3 | 15.6 |
| Range (µg/kg) | <LOQ–300.7 | 300.2–480.5 | <LOQ–700 | <LOQ–285.9 | <LOQ–423.7 | 54.1–236.1 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beccari, G.; Prodi, A.; Tini, F.; Bonciarelli, U.; Onofri, A.; Oueslati, S.; Limayma, M.; Covarelli, L. Changes in the Fusarium Head Blight Complex of Malting Barley in a Three-Year Field Experiment in Italy. Toxins 2017, 9, 120. https://doi.org/10.3390/toxins9040120
Beccari G, Prodi A, Tini F, Bonciarelli U, Onofri A, Oueslati S, Limayma M, Covarelli L. Changes in the Fusarium Head Blight Complex of Malting Barley in a Three-Year Field Experiment in Italy. Toxins. 2017; 9(4):120. https://doi.org/10.3390/toxins9040120
Chicago/Turabian StyleBeccari, Giovanni, Antonio Prodi, Francesco Tini, Umberto Bonciarelli, Andrea Onofri, Souheib Oueslati, Marwa Limayma, and Lorenzo Covarelli. 2017. "Changes in the Fusarium Head Blight Complex of Malting Barley in a Three-Year Field Experiment in Italy" Toxins 9, no. 4: 120. https://doi.org/10.3390/toxins9040120
APA StyleBeccari, G., Prodi, A., Tini, F., Bonciarelli, U., Onofri, A., Oueslati, S., Limayma, M., & Covarelli, L. (2017). Changes in the Fusarium Head Blight Complex of Malting Barley in a Three-Year Field Experiment in Italy. Toxins, 9(4), 120. https://doi.org/10.3390/toxins9040120









