Silver Nanoparticle-Based Fluorescence-Quenching Lateral Flow Immunoassay for Sensitive Detection of Ochratoxin A in Grape Juice and Wine
Abstract
:1. Introduction
2. Results and Discussion
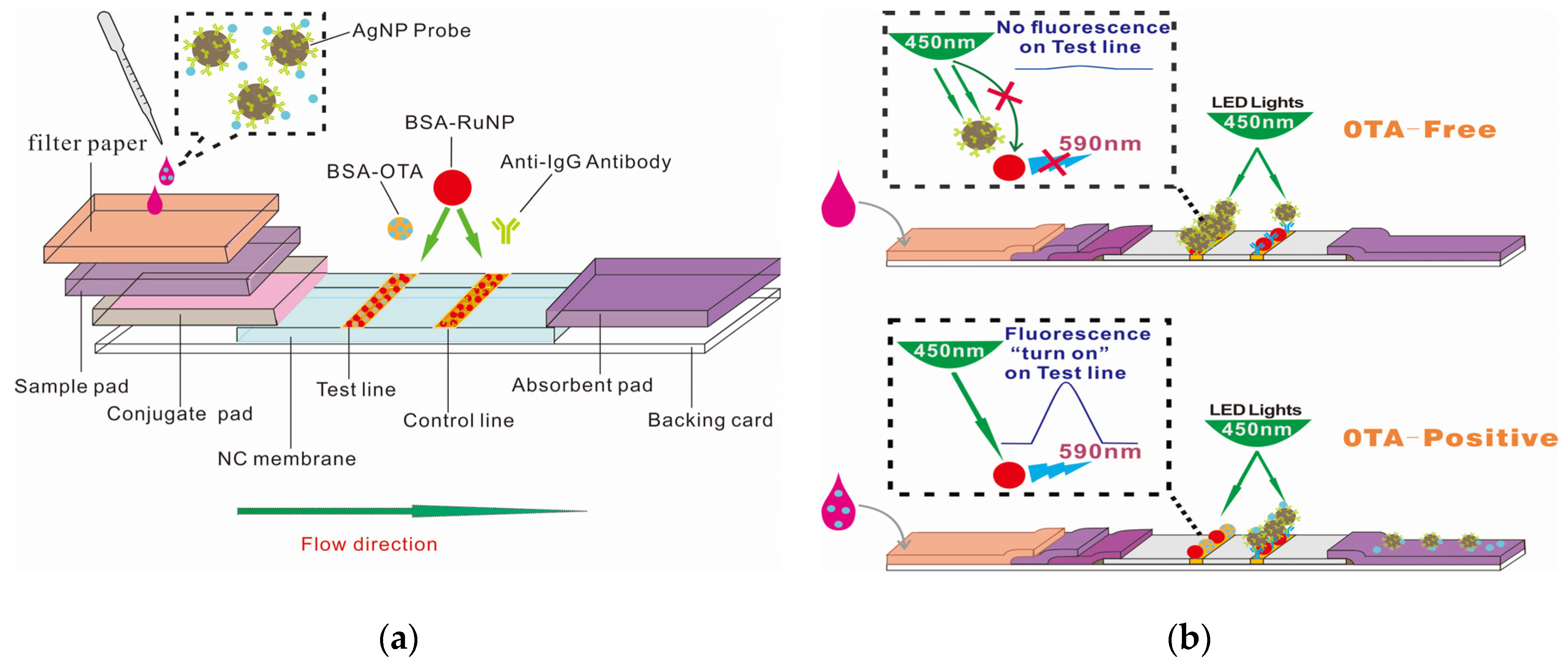
2.1. Scheme of AgNP–RuNP–cLFIA Sensor
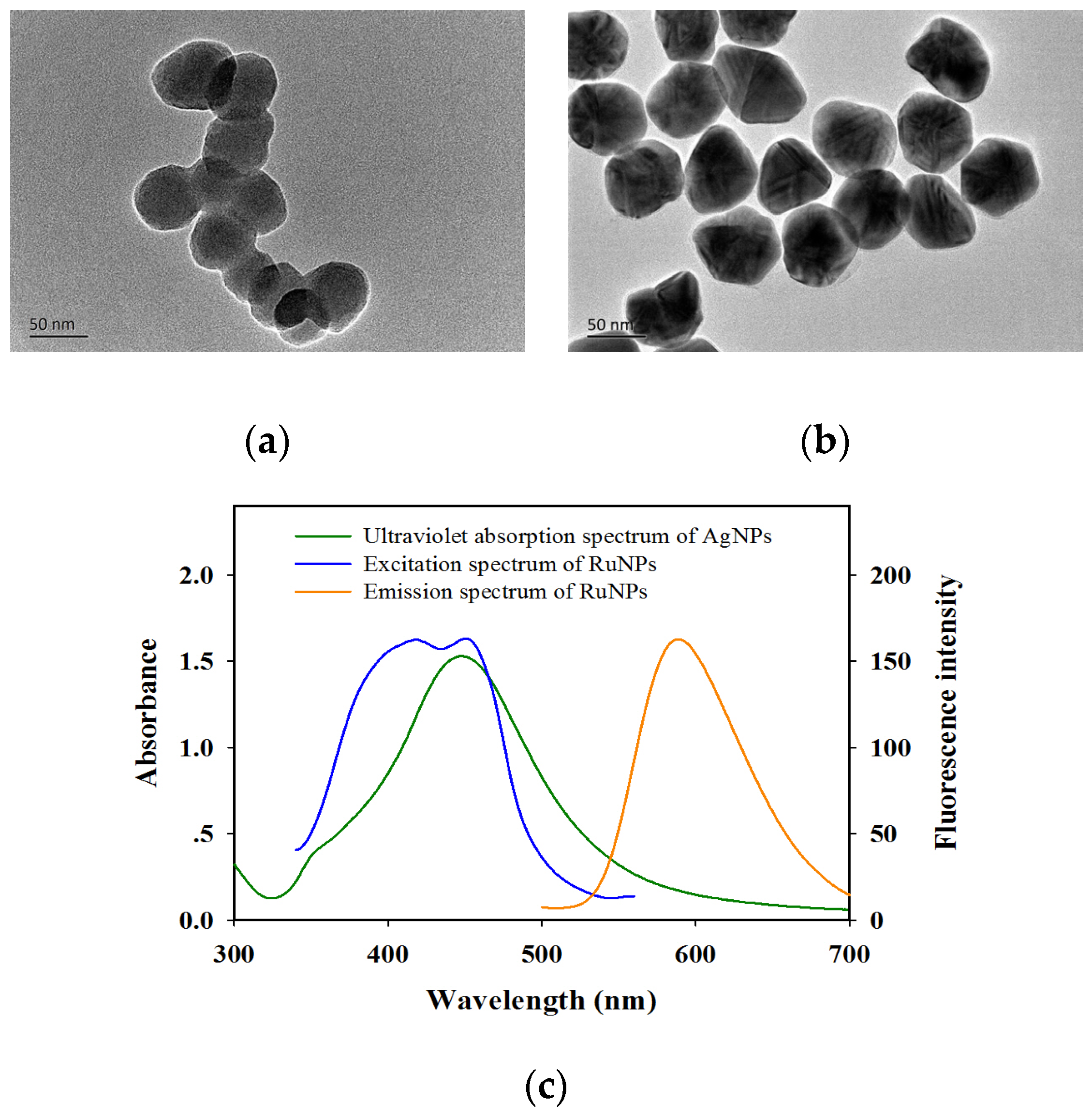
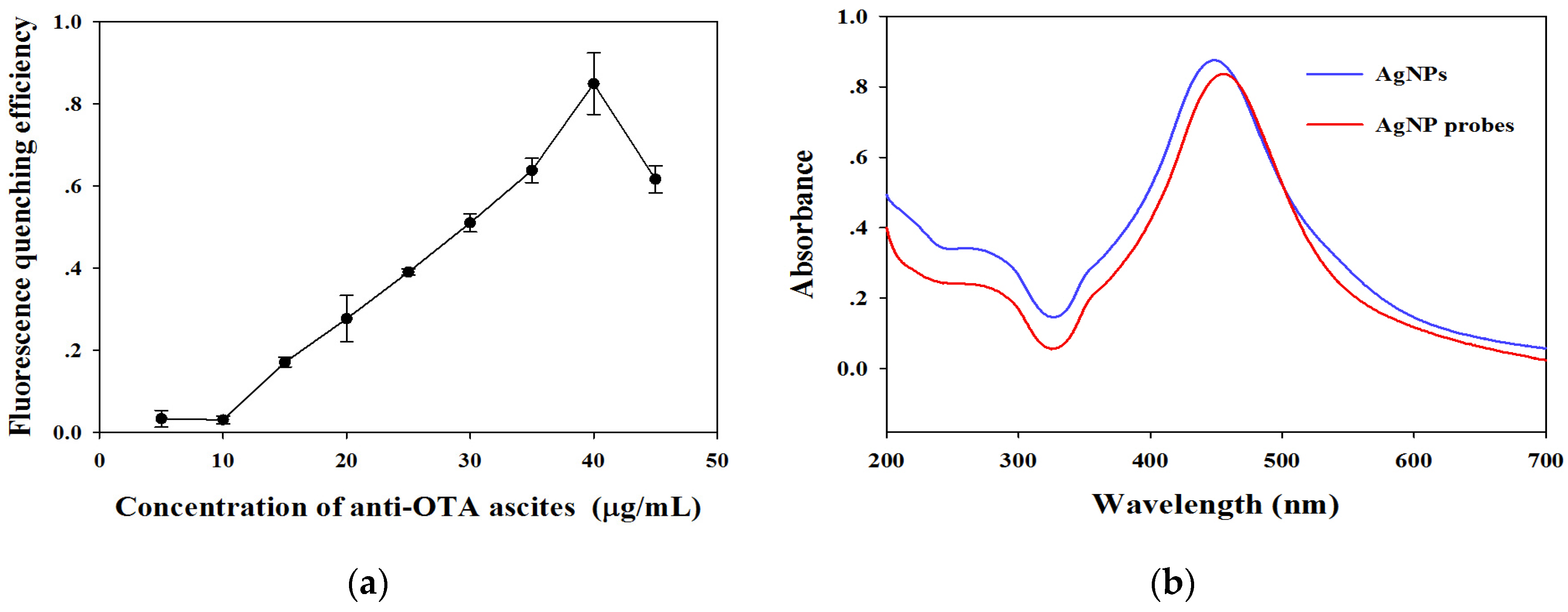
2.2. Characterization of AgNPs, RuNPs, and AgNP Probes
2.3. Parameter Optimization of AgNP–RuNP–cLFIA Sensor
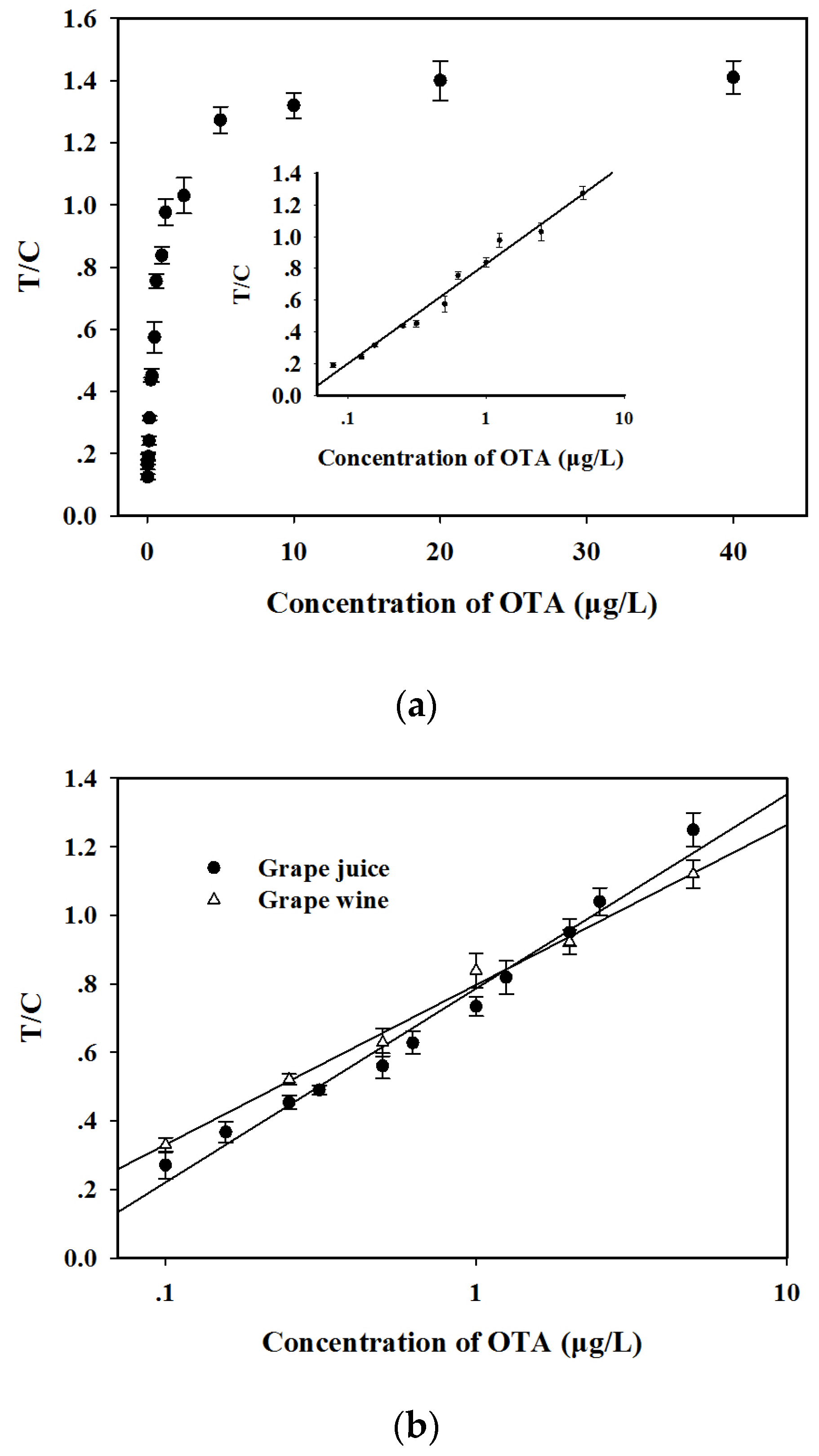
2.4. Performance Evaluation of AgNP–RuNP–cLFIA Sensor
3. Conclusions
4. Materials and Methods
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Posthuma-Trumpie, G.A.; Korf, J.; van Amerongen, A. Lateral flow (immuno) assay: Its strengths, weaknesses, opportunities and threats. A literature survey. Anal. Bioanal. Chem. 2009, 393, 569–582. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.; Fung, D.Y.C.; Yang, X.; Renrong, L.; Xiong, Y. Development of a colloidal gold strip for rapid detection of ochratoxin A with mimotope peptide. Food Control 2009, 20, 791–795. [Google Scholar] [CrossRef]
- Daniel, M.C.; Astruc, D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications toward Biology, Catalysis, and Nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Duan, H.; Xu, P.; Huang, X.; Xiong, Y. Effect of different-sized spherical gold nanoparticles grown layer by layer on the sensitivity of an immunochromatographic assay. RSC Adv. 2016, 6, 26178–26185. [Google Scholar] [CrossRef]
- Anfossi, L.; Baggiani, C.; Giovannoli, C.; D’Arco, G.; Giraudi, G. Lateral-flow immunoassays for mycotoxins and phycotoxins: A review. Anal. Bioanal. Chem. 2013, 405, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhang, Z.; Zhang, Q.; Li, P. Mycotoxin determination in foods using advanced sensors based on antibodies or aptamers. Toxins 2016, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Liang, J.; Lan, C.; Zhou, K.; Shi, C.; Tang, Y. Development of a novel dual-functional lateral-flow sensor for on-site detection of small molecule analytes. Sens. Actuators B Chem. 2014, 203, 683–689. [Google Scholar] [CrossRef]
- Fu, Q.; Tang, Y.; Shi, C.; Zhang, X.; Xiang, J.; Liu, X. A novel fluorescence-quenching immunochromatographic sensor for detection of the heavy metal chromium. Biosens. Bioelectron. 2013, 49, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Xu, Y.; Yu, J.; Li, J.; Zhou, X.; Wu, C.; Ji, Q.; Ren, Y.; Wang, L.; Huang, Z.; et al. Antigen detection based on background fluorescence quenching immunochromatographic assay. Anal. Chim. Acta 2014, 841, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Chen, M.; Liu, D.; Xiong, Y.; Feng, R.; Zhong, P.; Lai, W. Quantitative detection of β2-adrenergic agonists using fluorescence quenching by immunochromatographic assay. Anal. Meth. 2016, 8, 627–631. [Google Scholar] [CrossRef]
- Zhou, Z.; Yu, M.; Yang, H.; Huang, K.; Li, F.; Yi, T.; Huang, C. FRET-based sensor for imaging chromium (III) in living cells. Chem. Commun. 2008, 29, 3387–3389. [Google Scholar] [CrossRef] [PubMed]
- Pease, L.F.; Elliott, J.T.; Tsai, D.H.; Zachariah, M.R.; Michael, J.T. Determination of protein aggregation with differential mobility analysis: Application to IgG antibody. Biotechnol. Bioeng. 2008, 101, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Li, G.; Lv, Z.; Qiu, N.; Kong, W.; Gong, P.; Chen, G.; Xia, L.; Guo, X.; You, J.; et al. Facile and ultrasensitive fluorescence sensor platform for tumor invasive biomaker β-glucuronidase detection and inhibitor evaluation with carbon quantum dots based on inner-filter effect. Biosens. Bioelectron. 2016, 85, 358–362. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Aguilar, Z.P.; Li, H.; Lai, W.; Wei, H.; Xu, H.; Xiong, Y. Fluorescent Ru(phen)-doped silica nanoparticles-based ICTS sensor for quantitative detection of enrofloxacin residues in chicken meat. Anal. Chem. 2013, 85, 5120–5128. [Google Scholar] [CrossRef] [PubMed]
- Austin, L.A.; Mackey, M.A.; Dreaden, E.C.; El-Sayed, M.A. The optical, photothermal, and facile surface chemical properties of gold and silver nanoparticles in biodiagnostics, therapy, and drug delivery. Arch. Toxicol. 2014, 88, 1391–1417. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, R.R.; Frohlich, A.A. A review of recent advances in understanding ochratoxicosis. J. Anim. Sci. 1992, 70, 3968–3988. [Google Scholar] [CrossRef] [PubMed]
- Visconti, A.; Pascale, M.; Centonze, G. Determination of ochratoxin A in wine by means of immunoaffinity column clean-up and high-performance liquid chromatography. J. Chromatogr. A 1999, 864, 89–101. [Google Scholar] [CrossRef]
- Solfrizzo, M.; Piemontese, L.; Gambacorta, L.; Zivoli, R.; Longobardi, F. Food coloring agents and plant food supplements derived from Vitis vinifera: A new source of human exposure to ochratoxin A. J. Agric. Food Chem. 2015, 63, 3609–3614. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.; Mankotia, M.; Pantazopoulos, P.; Neil, R.J.; Scott, P.M. Ochratoxin A in wine and grape juice sold in Canada. Food Addit. Contam. 2004, 21, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.; Strub, C.; Hilaire, F.; Schorr-Galindo, S. First report: Penicillium adametzioides, a potential biocontrol agent for ochratoxin-producing fungus in grapes, resulting from natural product pre-harvest treatment. Food Control 2015, 51, 23–30. [Google Scholar] [CrossRef]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Covarelli, L.; Beccari, G.; Marini, A.; Tosi, L. A review on the occurrence and control of ochratoxigenic fungal species and ochratoxin A in dehydrated grapes, non-fortified dessert wines and dried vine fruit in the Mediterranean area. Food Control 2012, 26, 347–356. [Google Scholar] [CrossRef]
- Caba Es, F.J.; Accensi, F.; Bragulat, M.R.; Abarca, M.L.; Castellá, G.; Minguez, S.; Pons, A. What is the source of ochratoxin A in wine? Int. J. Food Microbiol. 2002, 79, 213–215. [Google Scholar] [CrossRef]
- Varga, J.; Kozakiewicz, Z. Ochratoxin A in grapes and grape-derived products. Trends Food Sci. Tech. 2006, 17, 72–81. [Google Scholar] [CrossRef]
- Anfossi, L.; Giovannoli, C.; Giraudi, G.; Biagioli, F.; Passini, C.; Baggiani, C. A lateral flow immunoassay for the rapid detection of ochratoxin A in wine and grape must. J. Agric. Food Chem. 2012, 60, 11491–11497. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; D'Arco, G.; Baggiani, C.; Giovannoli, C.; Giraudi, G. A lateral flow immunoassay for measuring ochratoxin A: Development of a single system for maize, wheat and durum wheat. Food Control 2011, 22, 1965–1970. [Google Scholar]
- Sun, Y.; Xing, G.; Yang, J.; Wang, F.; Deng, R.; Zhang, G.; Hu, X.; Zhang, Y. Development of an immunochromatographic test strip for simultaneous qualitative and quantitative detection of ochratoxin A and zearalenone in cereal. J. Sci. Food Agric. 2016, 96, 3673–3678. [Google Scholar] [CrossRef] [PubMed]
- Bastús, N.G.; Merkoçi, F.; Piella, J.; Puntes, V. Synthesis of highly monodisperse citrate-stabilized silver nanoparticles of up to 200 nm: Kinetic control and catalytic properties. Chem. Mater. 2014, 26, 2836–2846. [Google Scholar] [CrossRef]
- Wilson, P.K.; Szymanski, M.; Porter, R. Standardisation of metalloimmunoassay protocols for assessment of silver nanoparticle antibody conjugates. J. Immunol. Meth. 2013, 387, 303–307. [Google Scholar] [CrossRef] [PubMed]

| Sample | Spiking Levels (μg/L) | Dilution Factors | Mean Value (μg/L) * | SD % | CV % | Recovery % |
|---|---|---|---|---|---|---|
| Grape wine | 0.40 | 4 | 0.11 | 0.94 | 8.54 | 110.0 |
| 2.00 | 4 | 0.44 | 3.01 | 6.84 | 88.0 | |
| 5.00 | 4 | 1.26 | 2.28 | 1.81 | 100.8 | |
| Grape juice | 0.40 | 2 | 0.22 | 0.76 | 3.45 | 110.0 |
| 2.00 | 2 | 0.92 | 3.59 | 3.90 | 92.0 | |
| 5.00 | 2 | 2.56 | 4.80 | 1.87 | 102.4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, H.; Li, X.; Xiong, Y.; Pei, K.; Nie, L.; Xiong, Y. Silver Nanoparticle-Based Fluorescence-Quenching Lateral Flow Immunoassay for Sensitive Detection of Ochratoxin A in Grape Juice and Wine. Toxins 2017, 9, 83. https://doi.org/10.3390/toxins9030083
Jiang H, Li X, Xiong Y, Pei K, Nie L, Xiong Y. Silver Nanoparticle-Based Fluorescence-Quenching Lateral Flow Immunoassay for Sensitive Detection of Ochratoxin A in Grape Juice and Wine. Toxins. 2017; 9(3):83. https://doi.org/10.3390/toxins9030083
Chicago/Turabian StyleJiang, Hu, Xiangmin Li, Ying Xiong, Ke Pei, Lijuan Nie, and Yonghua Xiong. 2017. "Silver Nanoparticle-Based Fluorescence-Quenching Lateral Flow Immunoassay for Sensitive Detection of Ochratoxin A in Grape Juice and Wine" Toxins 9, no. 3: 83. https://doi.org/10.3390/toxins9030083
APA StyleJiang, H., Li, X., Xiong, Y., Pei, K., Nie, L., & Xiong, Y. (2017). Silver Nanoparticle-Based Fluorescence-Quenching Lateral Flow Immunoassay for Sensitive Detection of Ochratoxin A in Grape Juice and Wine. Toxins, 9(3), 83. https://doi.org/10.3390/toxins9030083







