Proteomic Characterization of the Venom of Five Bombus (Thoracobombus) Species
Abstract
1. Introduction
2. Results
2.1. 2D-PAGE Analysis of Thoracobombus Venom
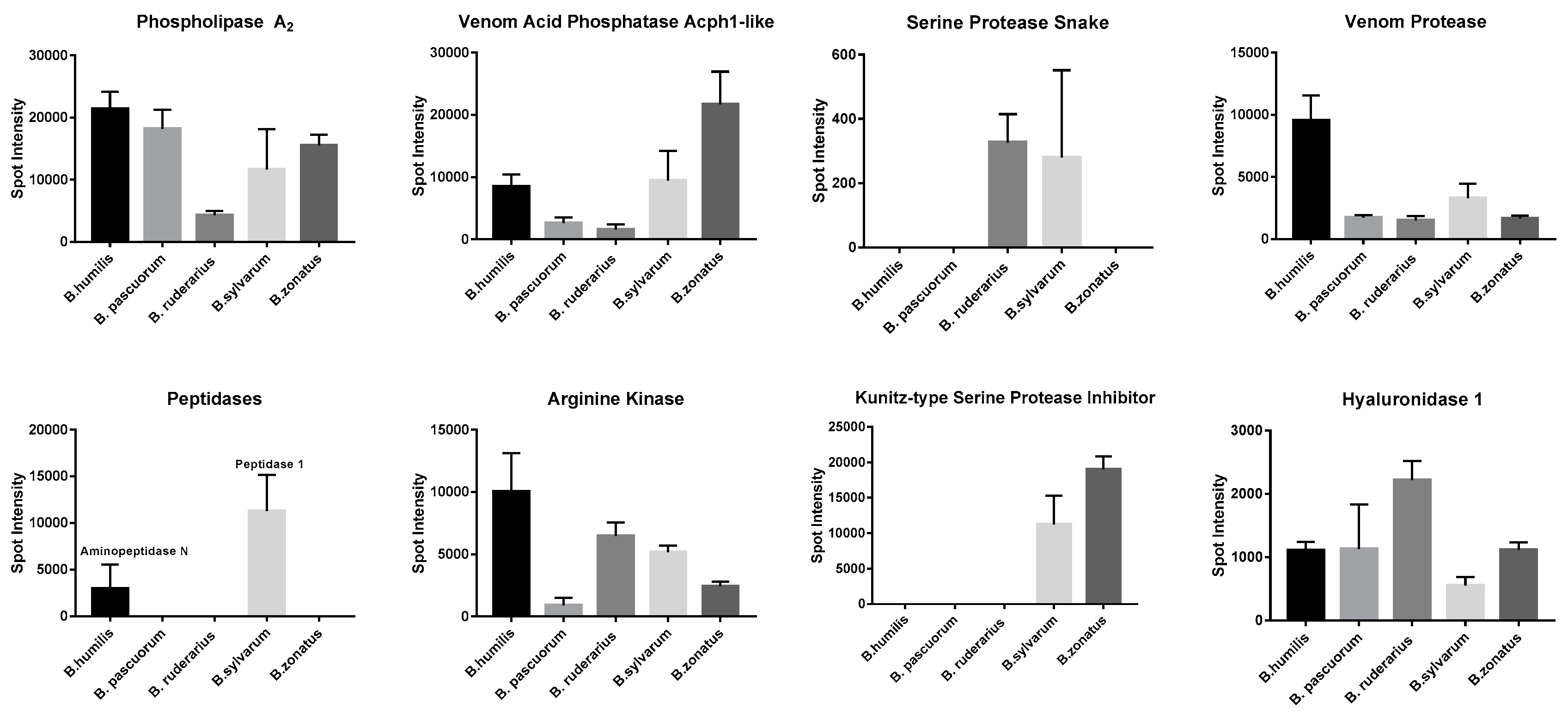
2.2. Mass-Spectrometry of Thoracobombus Venom
2.3. Species Specific Expression of Putative Venom Toxins
3. Discussion
4. Methods
4.1. Venom Extraction
4.2. Proteomic Analysis
4.2.1. Protein Quantification and Two-Dimensional Gel Electrophoresis
4.2.2. 2D-PAGE Analysis
4.2.3. In-Gel Enzymatic Digestion, Matrix-Assisted Laser Desorption Ionization/Time of Flight (MALDI-TOF) Mass Spectrometry and Peptide Mass Fingerprinting (PMF) Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Calvete, J.J. Venomics: Digging into the evolution of venomous systems and learning to twist nature to fight pathology. J. Proteom. 2009, 72, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.T. Chapter 49—Insects—Hymenoptera A2—Peterson. Michael E. In Small Animal Toxicology, 3rd ed.; Talcott, P.A., Ed.; W.B. Saunders: Saint Louis, MO, USA, 2013; pp. 573–588. [Google Scholar]
- Arbuckle, K. Evolutionary Context of Venom in Animals. In Evolution of Venomous Animals and Their Toxins; Malhotra, A., Ed.; Springer: Dordrecht, The Netherlands, 2017; pp. 3–31. [Google Scholar]
- Free, J. Insect Pollination of Crops; Academic Press: London, UK, 1993; p. 684. [Google Scholar]
- Choo, Y.M.; Lee, K.S.; Yoon, H.J.; Je, Y.H.; Lee, S.W.; Sohn, H.D.; Jin, B.R. Molecular cloning and antimicrobial activity of bombolitin, a component of bumblebee Bombus ignitus venom. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2010, 156, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Choo, Y.M.; Lee, K.S.; Yoon, H.J.; Kim, B.Y.; Sohn, M.R.; Roh, J.Y.; Je, Y.H.; Kim, N.J.; Kim, I.; Woo, S.D.; et al. Dual function of a bee venom serine protease: Prophenoloxidase-activating factor in arthropods and fibrin (ogen) olytic enzyme in mammals. PLoS ONE 2010, 5, e10393. [Google Scholar] [CrossRef] [PubMed]
- Choo, Y.M.; Lee, K.S.; Yoon, H.J.; Qiu, Y.; Wan, H.; Sohn, M.R.; Sohn, M.R.; Sohn, H.D.; Jin, B.R. Antifibrinolytic role of a bee venom serine protease inhibitor that acts as a plasmin inhibitor. PLoS ONE 2012, 7, e32269. [Google Scholar] [CrossRef] [PubMed]
- Choo, Y.M.; Qiu, Y.; Yoon, H.J.; Lee, K.Y.; Sohn, H.D.; Jin, B.R. Enzymatic properties of a bee venom serine protease from the bumblebee Bombus ignitus. J. Asia Pac. Entomol. 2011, 14, 249–251. [Google Scholar] [CrossRef]
- Choo, Y.M.; Yoon, H.J.; Jin, B.R. Effects of the bumblebee (Bombus ignitus) venom serine protease inhibitor on serine protease and phospholipase A 2 of B. ignitus venom. J. Asia Pac. Entomol. 2012, 15, 543–545. [Google Scholar] [CrossRef]
- Kim, J.S.; Choi, J.Y.; Lee, J.H.; Park, J.B.; Fu, Z.; Liu, Q.; Tao, X.; Jin, B.R.; Skinner, M.; Parker, B.L.; et al. Bumblebee venom serine protease increases fungal insecticidal virulence by inducing insect melanization. PLoS ONE 2013, 8, e62555. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Choo, Y.M.; Yoon, H.J.; Jia, J.; Cui, Z.; Wang, D.; Kim, D.H.; Sohn, H.D.; Jin, B.R. Fibrin (ogen) olytic activity of bumblebee venom serine protease. Toxicol. Appl. Pharmacol. 2011, 255, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Choo, Y.M.; Yoon, H.J.; Jin, B.R. Molecular cloning and antibacterial activity of bombolitin isolated from the venom of a bumblebee, Bombus terrestris. J. Asia Pac. Entomol. 2012, 15, 21–25. [Google Scholar] [CrossRef]
- Qiu, Y.; Choo, Y.M.; Yoon, H.J.; Jin, B.R. Molecular cloning and fibrin (ogen) olytic activity of a bumblebee (Bombus hypocrita sapporoensis) venom serine protease. J. Asia Pac. Entomol. 2012, 15, 79–82. [Google Scholar] [CrossRef]
- Qiu, Y.; Lee, K.S.; Choo, Y.M.; Kong, D.; Yoon, H.J.; Jin, B.R. Molecular cloning and antifibrinolytic activity of a serine protease inhibitor from bumblebee (Bombus terrestris) venom. Toxicon 2013, 63, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Kim, B.Y.; Lee, K.S.; Yoon, H.J.; Lee, K.Y.; Jin, B.R. A bumblebee (Bombus ignitus) venom serine protease inhibitor that acts as a microbial serine protease inhibitor. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2014, 167, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Favreau, P.; Menin, L.; Michalet, S.; Perret, F.; Cheneval, O.; Stöcklin, M.; Bulet, P.; Stöcklin, R. Mass spectrometry strategies for venom mapping and peptide sequencing from crude venoms: Case applications with single arthropod specimen. Toxicon 2006, 47, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Argiolas, A.; Pisano, J.J. Bombolitins, a new class of mast cell degranulating peptides from the venom of the bumblebee Megabombus pennsylvanicus. J. Biol. Chem. 1985, 260, 1437–1444. [Google Scholar] [PubMed]
- Hoffman, D.R.; El-Choufani, S.E.; Smith, M.M.; De Groot, H. Occupational allergy to bumblebees: Allergens of Bombus terrestris. J. Allergy Clin. Immunol. 2001, 108, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.R.; Jacobson, R.S. Allergens in hymenoptera venom XXVII: Bumblebee venom allergy and allergens. J. Allergy Clin. Immunol. 1996, 97, 812–821. [Google Scholar] [CrossRef]
- Van Vaerenbergh, M.; Debyser, G.; Smagghe, G.; Devreese, B.; de Graaf, D.C. Unraveling the venom proteome of the bumblebee (Bombus terrestris) by integrating a combinatorial peptide ligand library approach with FT-ICR MS. Toxicon 2015, 102, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Barkan, N.P.; Demiralp, D.O.; Aytekin, A.M. Comparison of Venom Extraction Methods of Bombus (Bombus) terrestris (Hymenoptera: Apidae: Bombus) by Using Bottom-up Proteomic Strategies. Curr. Proteom. 2015, 12, 80–86. [Google Scholar] [CrossRef]
- Piek, T. Venoms of the Hymenoptera: Biochemical, Pharmacological and Behavioural Aspects; Elsevier: London, UK, 2013. [Google Scholar]
- Fenton, A.W.; West, P.R.; Odell, G.V.; Hudiburg, S.M.; Ownby, C.L.; Mills, J.N.; Scroggins, B.T.; Shannon, S.B. Arthropod venom citrate inhibits phospholipase A2. Toxicon 1995, 33, 763–770. [Google Scholar] [CrossRef]
- Rees, J.A.; Moniatte, M.; Bulet, P. Novel antibacterial peptides isolated from a European bumblebee, Bombus pascuorum (Hymenoptera, Apoidea). Insect Biochem. Mol. Biol. 1997, 27, 413–422. [Google Scholar] [CrossRef]
- Piek, T.; Veldsema-Currie, R.; Spanjer, W.; Mantel, P. Acetylcholine and an unidentified, muscle-contracting factor in the venom of the bumblebee, Bombus terrestris L. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1983, 75, 351–356. [Google Scholar] [CrossRef]
- Sadd, B.M.; Barribeau, S.M.; Bloch, G.; de Graaf, D.C.; Dearden, P.; Elsik, C.G.; Gadau, J.; Grimmelikhuijzen, C.J.; Hasselmann, M.; Lozier, J.D.; et al. The genomes of two key bumblebee species with primitive eusocial organization. Genome Biol. 2015, 16, 76. [Google Scholar] [CrossRef] [PubMed]
- Williams, P. The distribution of bumblebee colour patterns worldwide: Possible significance for thermoregulation, crypsis, and warning mimicry. Biol. J. Linn. Soc. 2007, 92, 97–118. [Google Scholar] [CrossRef]
- Barkan, N.P.; Aytekin, A.M. Systematical studies on the species of the subgenus Bombus (Thoracobombus) (Hymenoptera: Apidae: Bombus Latreille) in Turkey. Zootaxa 2013, 3737, 167–183. [Google Scholar] [CrossRef] [PubMed]
- Terzo, M.; Valterová, I.; Rasmont, P. Atypical Secretions of the Male Cephalic Labial Glands in Bumblebees: The Case of Bombus (Rhodobombus) mesomelasGerstaecker (Hymenoptera, Apidae). Chem. Biodivers. 2007, 4, 1466–1471. [Google Scholar] [CrossRef] [PubMed]
- Terzo, M.; Valterova, I.; Urbanova, K.; Rasmont, P. De la nécessité de redécrire les phéromones sexuelles des mâles de bourdons [Hymenoptera: Apidae, Bombini] publiées avant 1996 pour leur utilisation en analyse phylogénétique. Phytoprotection 2003, 84, 39–49. [Google Scholar] [CrossRef]
- Urbanová, K.; Valterová, I.; Rasmont, P.; Terzo, M. Znaèkovací feromony: Slovení sekretu labiální vlázy samcù Bombus magnus. [Marking pheromones of bumblebees: Composition of the labial gland secretion of males of Bombus magnus]. Chemické Listy 2002, 96, 918–919. [Google Scholar]
- Hovorka, O.; Valterova, I.; Rasmont, P.; Terzo, M. Male cephalic labial gland secretions of two bumblebee species of the subgenus Cullumanobombus (Hymenoptera: Apidae: Bombus latreille) and their distribution in central Europe. Chem. Biodivers. 2006, 3, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.; Hines, H.; Williams, P. A comprehensive phylogeny of the bumble bees (Bombus). Biol. J. Linn. Soc. 2007, 91, 161–188. [Google Scholar] [CrossRef]
- Cameron, S.A.; Hines, H.M.; Williams, P.H. Molecular phylogeny of the bumble bee subgenus Pyrobombus (Hymenoptera: Apidae: Bombus) with insights into gene utility for lower-level analysis. Invertebr. Syst. 2006, 20, 289–303. [Google Scholar]
- Cameron, S.A.; Williams, P.H. Phylogeny of bumble bees in the New World subgenus Fervidobombus (Hymenoptera: Apidae): Congruence of molecular and morphological data. Mol. Phylogenet. Evol. 2003, 28, 552–563. [Google Scholar] [CrossRef]
- Kawakita, A.; Sota, T.; Ascher, J.S.; Ito, M.; Tanaka, H.; Kato, M. Evolution and phylogenetic utility of alignment gaps within intron sequences of three nuclear genes in bumble bees (Bombus). Mol. Biol. Evol 2003, 20, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, A.; Sota, T.; Ito, M.; Ascher, J.S.; Tanaka, H.; Kato, M.; Roubik, D.W. Phylogeny. historical biogeography, and character evolution in bumble bees (Bombus: Apidae) based on simultaneous analysis of three nuclear gene sequences. Mol. Phylogenet. Evol. 2004, 31, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Bi, G.; Zhao, E.; Yang, J.; Zhang, Z.; Liu, G. Complete mitochondrial genome of Bombus terrestris (Hymenoptera: Apidae). Mitochondrial DNA 2016, 27, 4455–4456. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.H.; An, J.; Brown, M.J.F.; Carolan, J.C.; Goulson, D.; Huang, J.; Ito, M. Cryptic Bumblebee Species: Consequences for Conservation and the Trade in Greenhouse Pollinators. PLoS ONE 2012, 7, e32992. [Google Scholar] [CrossRef] [PubMed]
- Van Vaerenbergh, M.; Debyser, G.; Devreese, B.; de Graaf, D.C. Exploring the hidden honeybee (Apis mellifera) venom proteome by integrating a combinatorial peptide ligand library approach with FTMS. J. Proteom. 2014, 99, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.L.; Sanz, L.; Chiucchi, J.E.; Farrell, T.M.; Calvete, J.J. Proteomic analysis of ontogenetic and diet-related changes in venom composition of juvenile and adult Dusky Pigmy rattlesnakes (Sistrurus miliarius barbouri). J. Proteom. 2011, 74, 2169–2179. [Google Scholar] [CrossRef] [PubMed]
- King, G.F. Venoms as a platform for human drugs: Translating toxins into therapeutics. Expert Opin. Biol. Ther. 2011, 11, 1469–1484. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Igci, N.; Demiralp, D.O. A preliminary investigation into the venom proteome of Macrovipera lebetina obtusa (Dwigubsky, 1832) from Southeastern Anatolia by MALDI-TOF mass spectrometry and comparison of venom protein profiles with Macrovipera lebetina lebetina (Linnaeus, 1758) from Cyprus by 2D-PAGE. Arch. Toxicol. 2012, 86, 441–451. [Google Scholar] [PubMed]
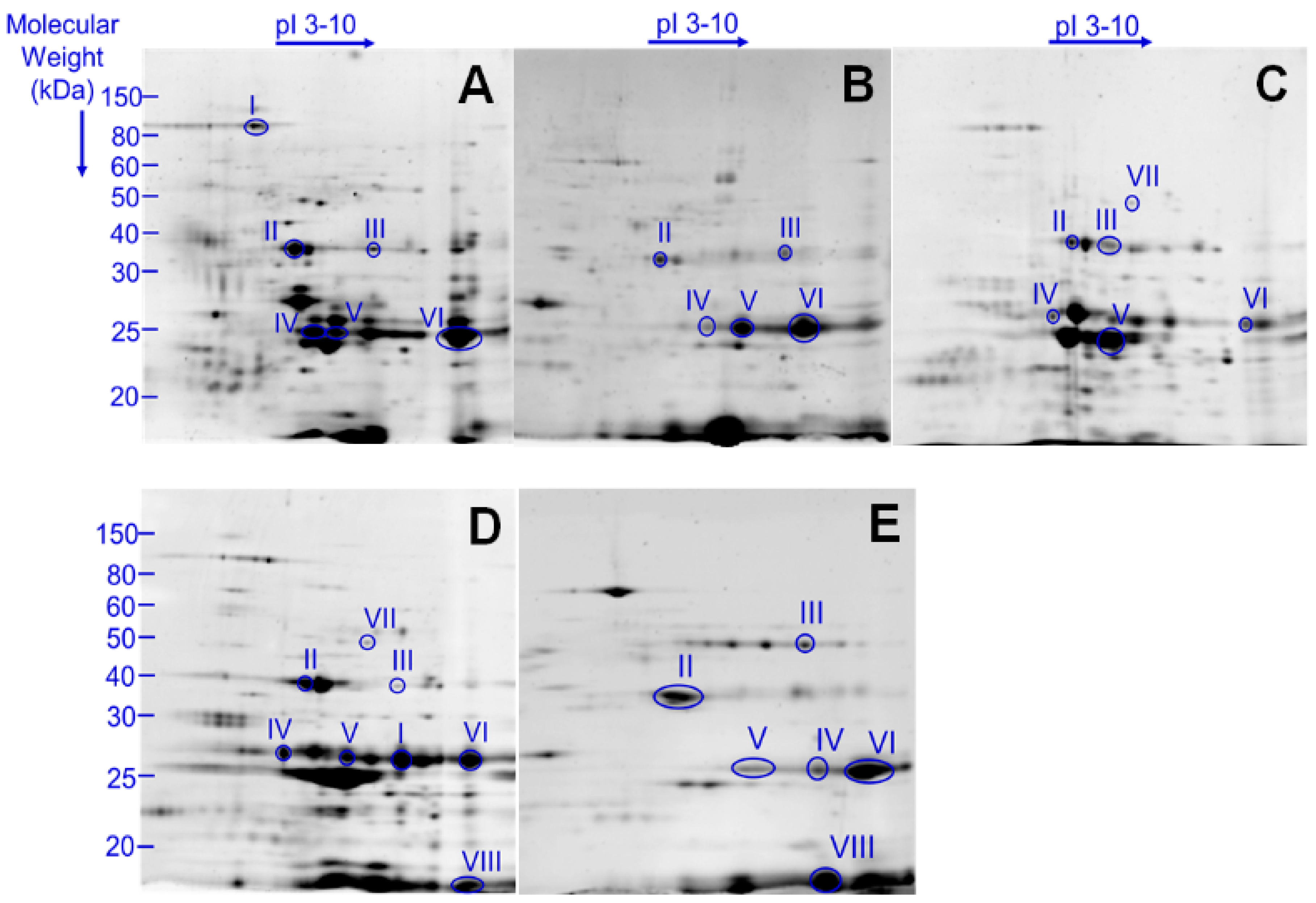
| Putative Toxin Family | Spot | Toxin Name | B. terrestris | A. mellifera |
|---|---|---|---|---|
| Peptidase/protease | I | Peptidase I *, Aminopeptidase N ** | Dipeptidyl peptidase IV, Serine carboxypeptidase, Prolylcarboxypeptidase | Dipeptidyl peptidase IV, Serine carboxypeptidase, Prolylcarboxypeptidase |
| IV | Venom protease | |||
| VII | Serine protease snake | Serine protease 1–6 CUB serine protease | Serine protease snake, CLIP serine protease, CUB serine protease | |
| Esterase | II | Venom acid phosphatase Acph1-like | Acid phosphatase | Acid phosphatase 1 |
| V | Arginine Kinase | |||
| VI | Phospholipase A2 | Phospholipase A2-1, Phospholipase A2-2 | Phospholipase A2-1, Phospholipase A2-2 | |
| Carbohdyrate metabolism | III | Hyaluronidase 1 | Hyaluronidase | Hyaluronidase |
| Serine protease inhibitor | VIII | Kunitz-type serine protease inhibitor | Serpin 1–3 | Serpin 1–2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barkan, N.P.; Bayazit, M.B.; Ozel Demiralp, D. Proteomic Characterization of the Venom of Five Bombus (Thoracobombus) Species. Toxins 2017, 9, 362. https://doi.org/10.3390/toxins9110362
Barkan NP, Bayazit MB, Ozel Demiralp D. Proteomic Characterization of the Venom of Five Bombus (Thoracobombus) Species. Toxins. 2017; 9(11):362. https://doi.org/10.3390/toxins9110362
Chicago/Turabian StyleBarkan, Nezahat Pınar, Mustafa Bilal Bayazit, and Duygu Ozel Demiralp. 2017. "Proteomic Characterization of the Venom of Five Bombus (Thoracobombus) Species" Toxins 9, no. 11: 362. https://doi.org/10.3390/toxins9110362
APA StyleBarkan, N. P., Bayazit, M. B., & Ozel Demiralp, D. (2017). Proteomic Characterization of the Venom of Five Bombus (Thoracobombus) Species. Toxins, 9(11), 362. https://doi.org/10.3390/toxins9110362






