Suppressive Effects of Bee Venom Acupuncture on Paclitaxel-Induced Neuropathic Pain in Rats: Mediation by Spinal α2-Adrenergic Receptor
Abstract
:1. Introduction
2. Results
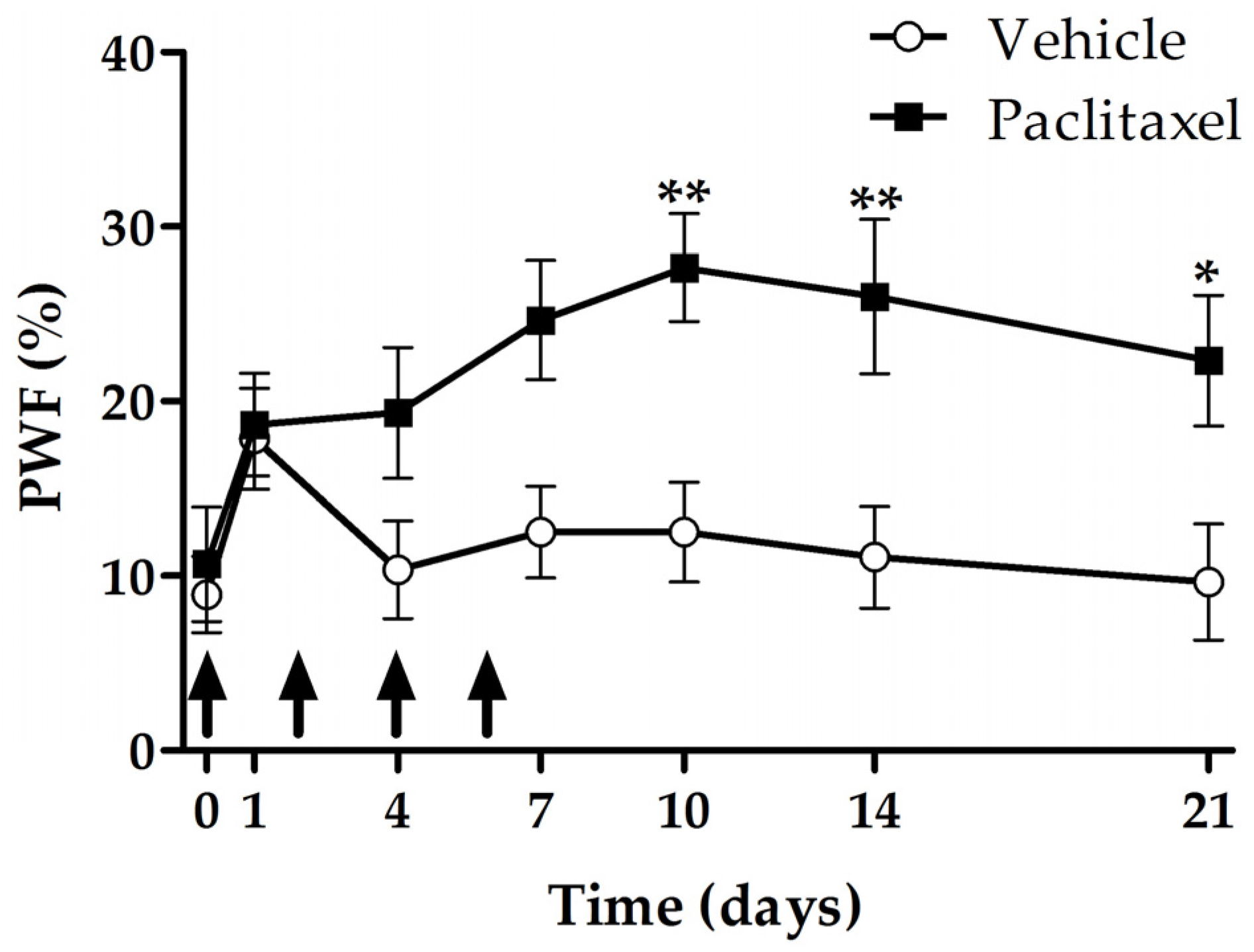
2.1. Development and Maintanance of Paclitaxel-Induced Mechanical Hyperalgesia
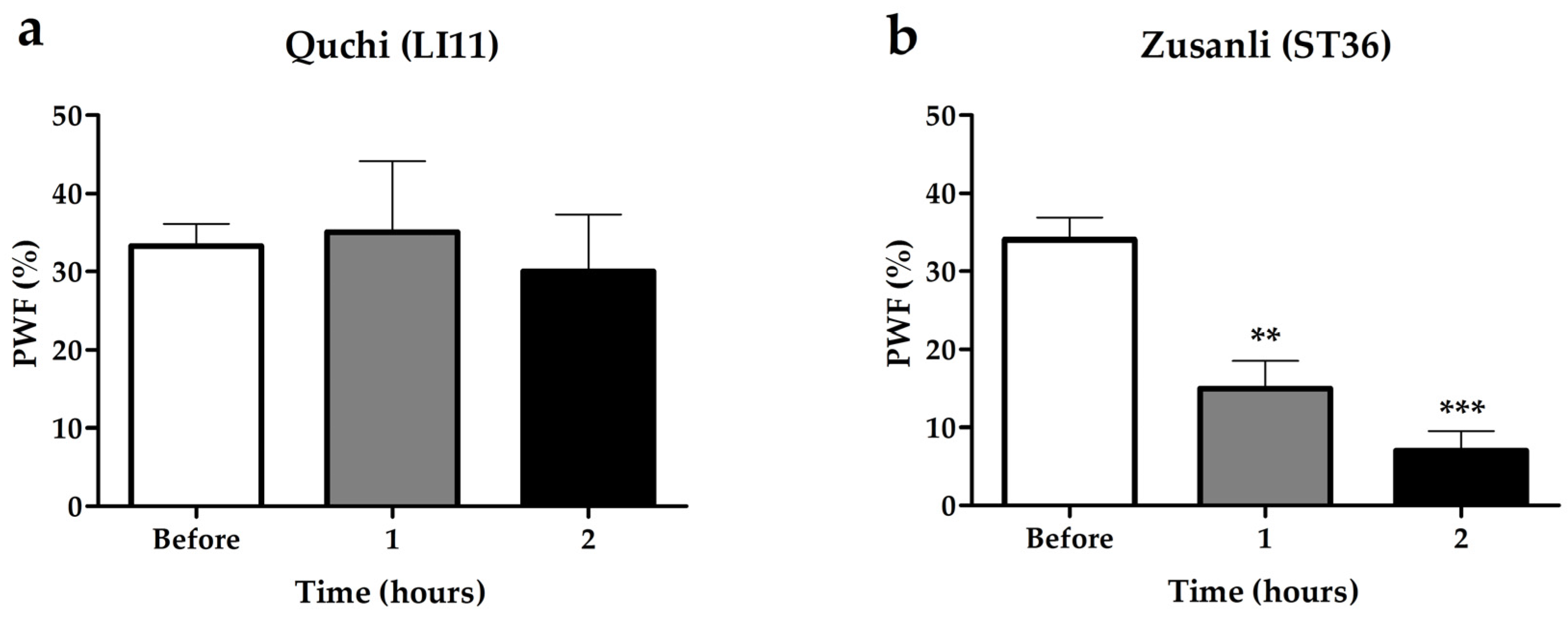
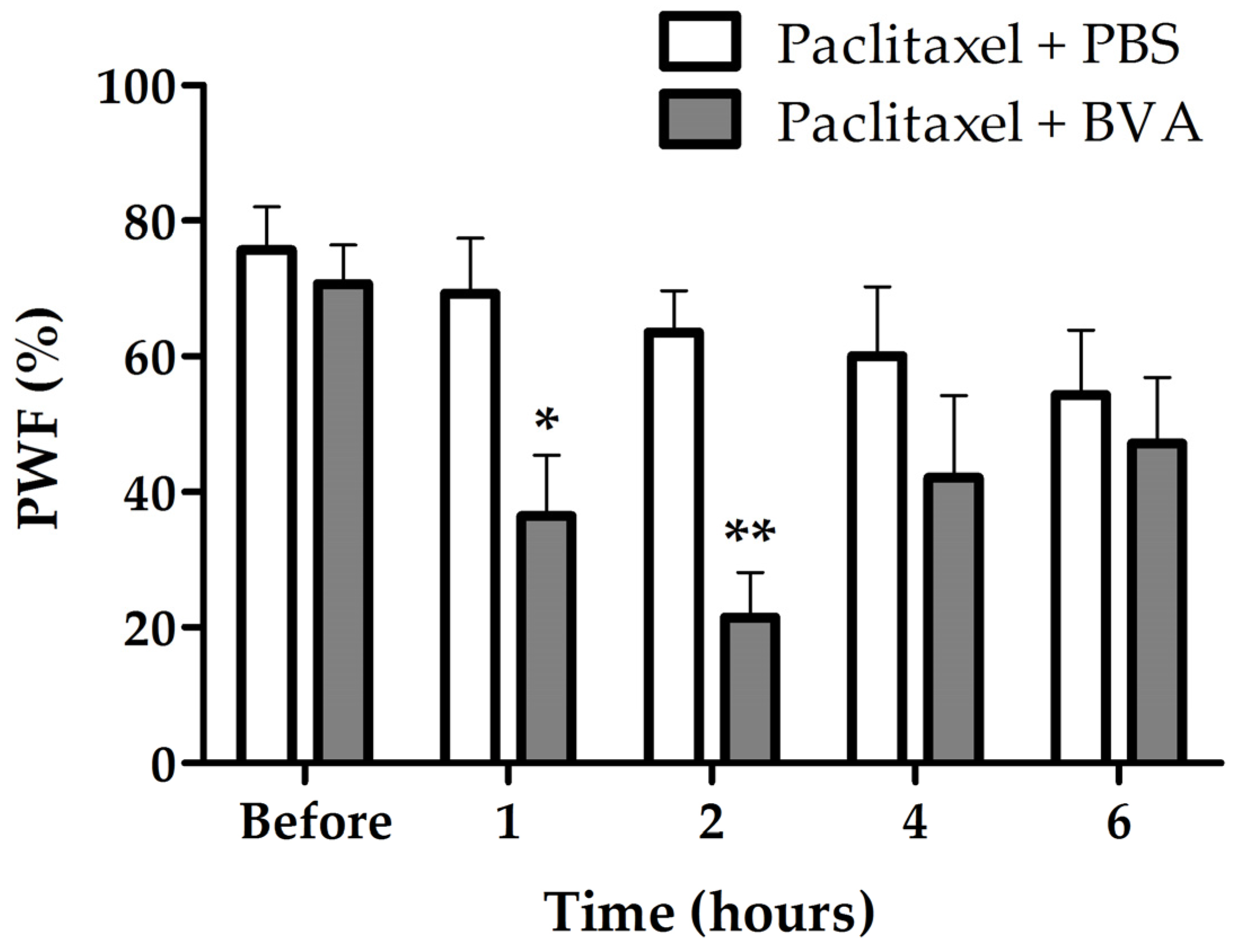
2.2. Effects of BVA on Paclitaxel-Induced Mechanical Hyperalgesia
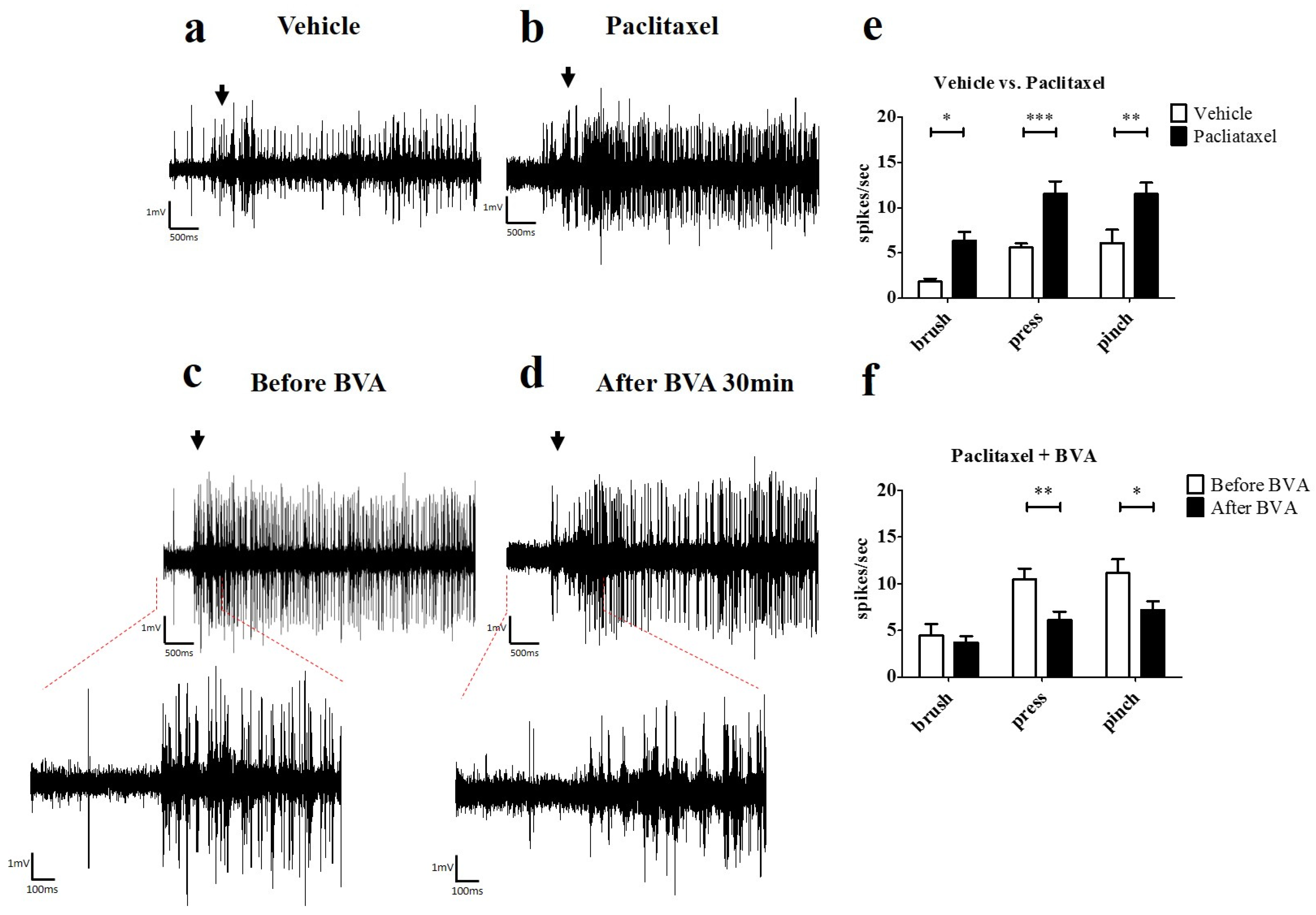
2.3. Effects of BVA on Paclitaxel-Induced Hyperexcitation in the Spinal Wide Dynamic Range (WDR) Neurons
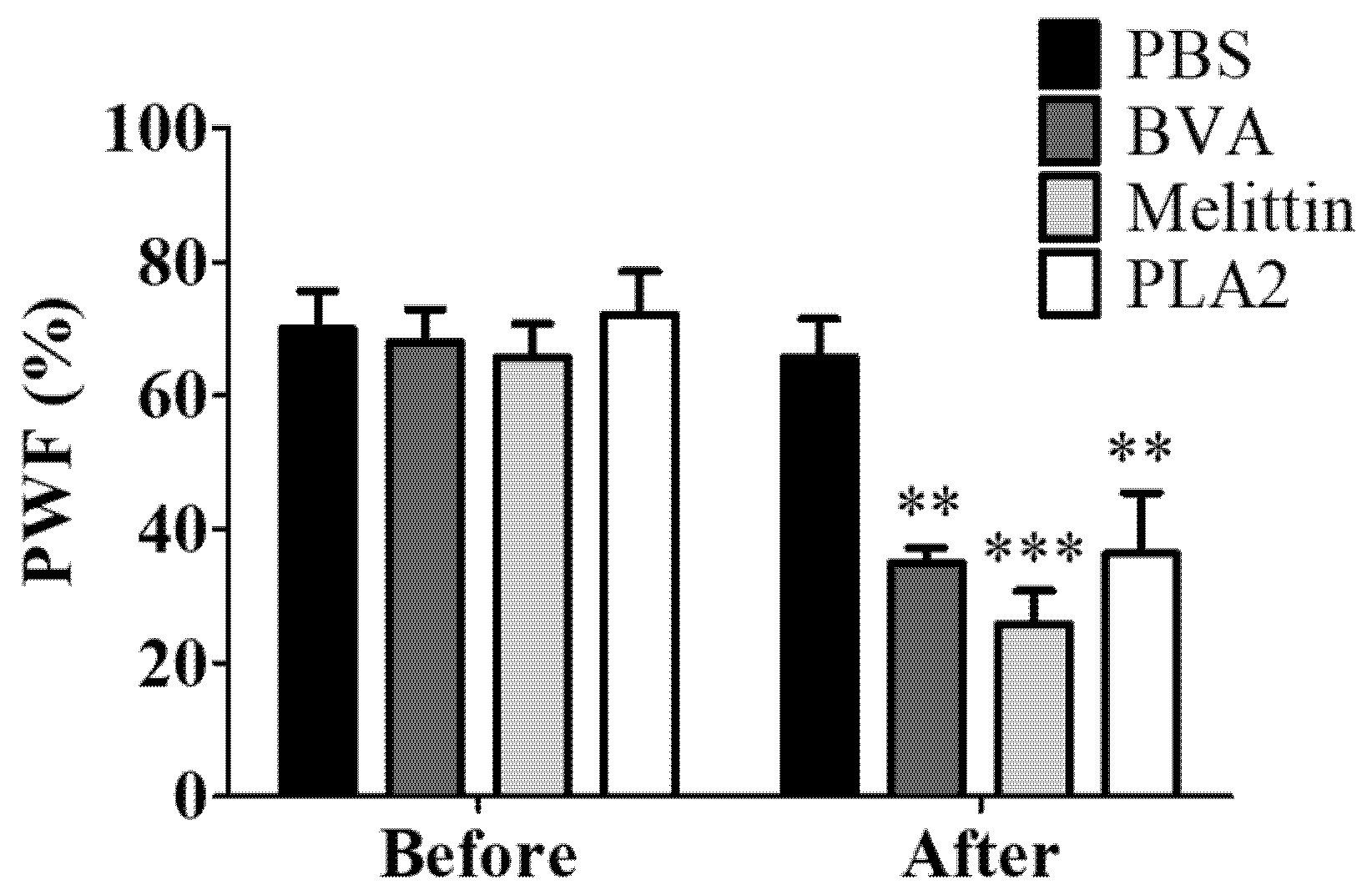
2.4. Effect of BVA, Melittin, or PLA2 on Paclitaxel-Induced Mechanical Hyperalgesia
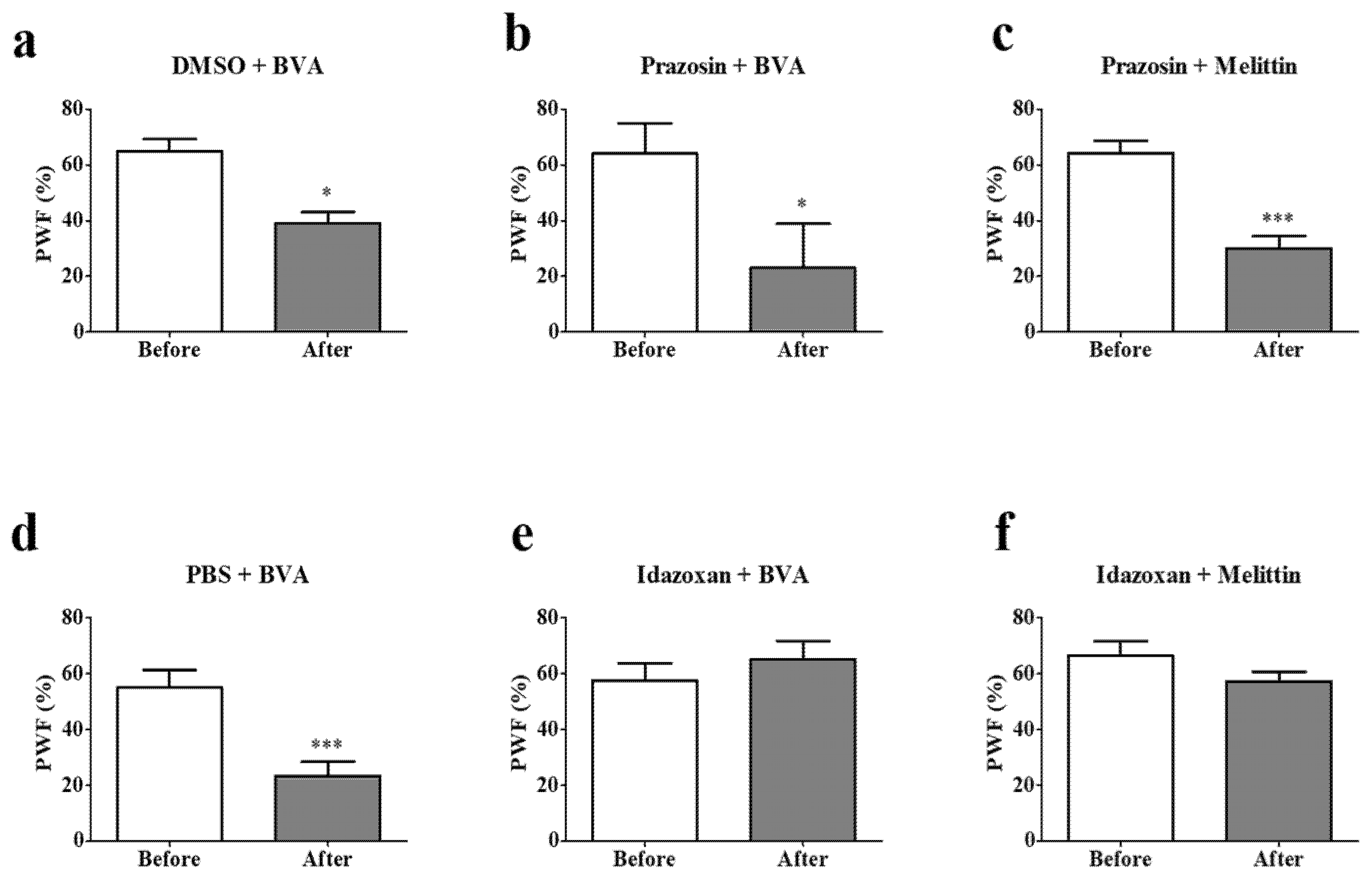
2.5. Effects of Intrathecal α-Adrenergic Receptor Subtype Antagonists on BVA- or Melittin-Induced Anti-Hyperalgesia
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animals
5.2. Administration of Paclitaxel
5.3. Behavior Tests
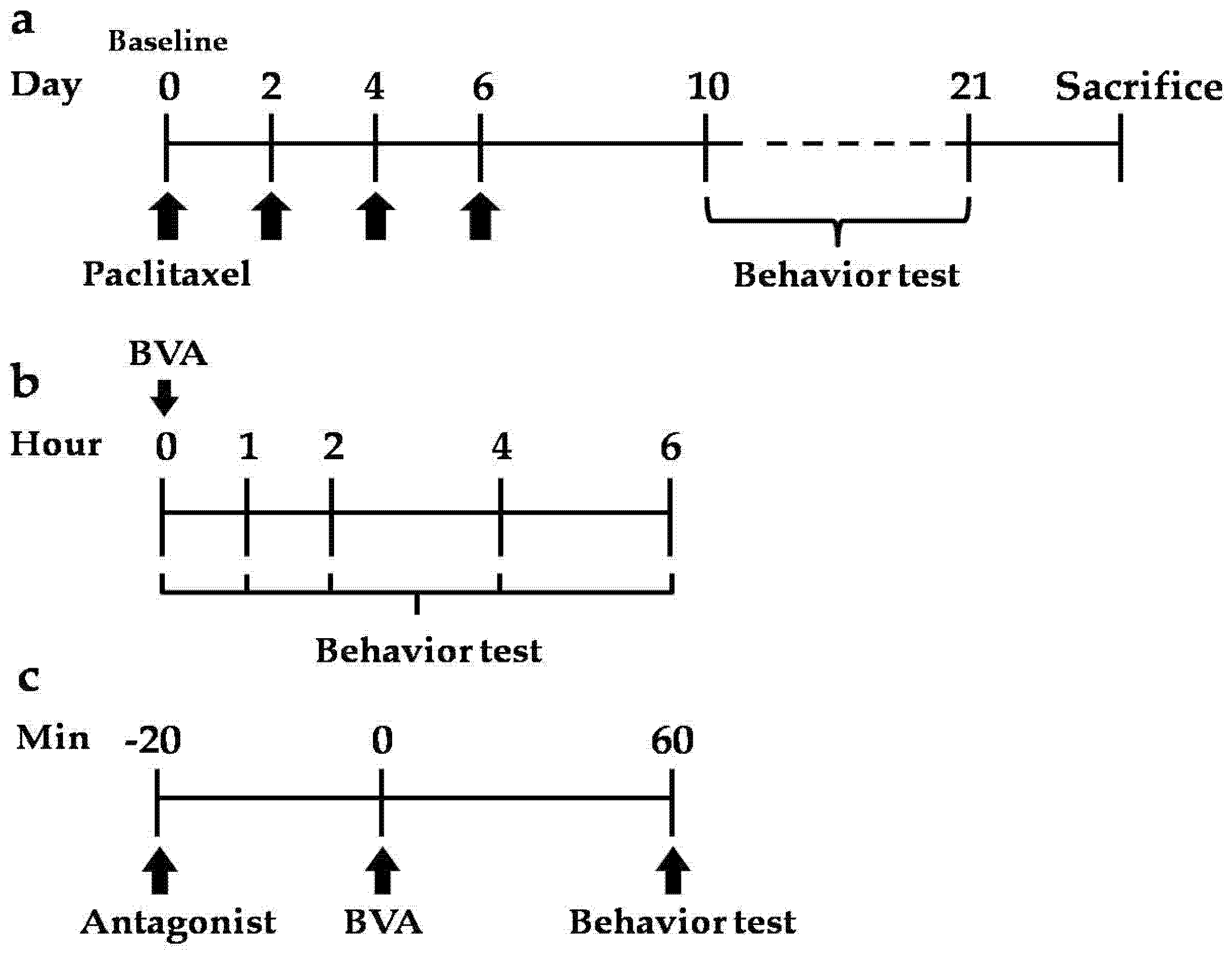
5.4. Experimental Schedule
5.5. BVA, Melittin, or PLA2 Treatment
5.6. Extracellular Recording
5.7. Antagonists
5.8. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; McPhail, A.T. Plant antitumor agents. Vi. Isolation and structure of taxol, a novel antileukemic and antitumor agent from taxus brevifolia. J. Am. Chem. Soc. 1971, 93, 2325–2327. [Google Scholar] [CrossRef] [PubMed]
- Sparano, J.A. Taxanes for breast cancer: An evidence-based review of randomized phase ii and phase iii trials. Clin. Breast Cancer 2000, 1, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Goffin, J.; Lacchetti, C.; Ellis, P.M.; Ung, Y.C.; Evans, W.K. First-line systemic chemotherapy in the treatment of advanced non-small cell lung cancer: A systematic review. J. Thorac. Oncol. 2010, 5, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Covens, A.; Carey, M.; Bryson, P.; Verma, S.; Fung Kee Fung, M.; Johnston, M. Systematic review of first-line chemotherapy for newly diagnosed postoperative patients with stage ii, iii, or iv epithelial ovarian cancer. Gynecol. Oncol. 2002, 85, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Swain, S.M. Peripheral neuropathy induced by microtubule-stabilizing agents. J. Clin. Oncol. 2006, 24, 1633–1642. [Google Scholar] [CrossRef] [PubMed]
- Argyriou, A.A.; Koltzenburg, M.; Polychronopoulos, P.; Papapetropoulos, S.; Kalofonos, H.P. Peripheral nerve damage associated with administration of taxanes in patients with cancer. Crit. Rev. Oncol. Hematol. 2008, 66, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Koh, P.S.; Seo, B.K.; Cho, N.S.; Park, H.S.; Park, D.S.; Baek, Y.H. Clinical effectiveness of bee venom acupuncture and physiotherapy in the treatment of adhesive capsulitis: A randomized controlled trial. J. Shoulder Elbow Surg. 2013, 22, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.Y.; Shim, S.R.; Rhee, H.Y.; Park, H.J.; Jung, W.S.; Moon, S.K.; Park, J.M.; Ko, C.N.; Cho, K.H.; Park, S.U. Effectiveness of acupuncture and bee venom acupuncture in idiopathic parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-B.; Kim, J.-H.; Yoon, J.-H.; Lee, J.-D.; Han, H.-J.; Mar, W.-C.; Beitz, A.J.; Lee, J.-H. The analgesic efficacy of bee venom acupuncture for knee osteoarthritis: A comparative study with needle acupuncture. J. Chin. Med. 2001, 29, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Pittler, M.H.; Shin, B.C.; Kong, J.C.; Ernst, E. Bee venom acupuncture for musculoskeletal pain: A review. J. Pain 2008, 9, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Kwon, Y.B.; Han, H.J.; Yang, I.S.; Beitz, A.J.; Lee, J.H. Antinociceptive mechanisms associated with diluted bee venom acupuncture (apipuncture) in the rat formalin test: Involvement of descending adrenergic and serotonergic pathways. Pharmacol. Res. 2005, 51, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Kim, M.; Go, D.; Min, B.-I.; Na, H.; Kim, S. Combined effects of bee venom acupuncture and morphine on oxaliplatin-induced neuropathic pain in mice. Toxins 2016, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-B.; Kang, M.-S.; Han, H.-J.; Beitz, A.J.; Lee, J.-H. Visceral antinociception produced by bee venom stimulation of the zhongwan acupuncture point in mice: Role of α2 adrenoceptors. Neurosci. Lett. 2001, 308, 133–137. [Google Scholar] [CrossRef]
- Lim, B.S.; Moon, H.J.; Li, D.X.; Gil, M.; Min, J.K.; Lee, G.; Bae, H.; Kim, S.K.; Min, B.I. Effect of bee venom acupuncture on oxaliplatin-induced cold allodynia in rats. Evid. Based Complement. Altern. Med. eCAM 2013, 2013, 369324. [Google Scholar] [CrossRef] [PubMed]
- Roh, D.H.; Kwon, Y.B.; Kim, H.W.; Ham, T.W.; Yoon, S.Y.; Kang, S.Y.; Han, H.J.; Lee, H.J.; Beitz, A.J.; Lee, J.H. Acupoint stimulation with diluted bee venom (apipuncture) alleviates thermal hyperalgesia in a rodent neuropathic pain model: Involvement of spinal alpha 2-adrenoceptors. J. Pain 2004, 5, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Jeon, J.H.; Yoon, J.; Jung, T.Y.; Kwon, K.R.; Cho, C.K.; Lee, Y.W.; Sagar, S.; Wong, R.; Yoo, H.S. Effects of sweet bee venom pharmacopuncture treatment for chemotherapy-induced peripheral neuropathy: A case series. Integr. Cancer Ther. 2012, 11, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Jeon, J.H.; Lee, Y.W.; Cho, C.K.; Kwon, K.R.; Shin, J.E.; Sagar, S.; Wong, R.; Yoo, H.S. Sweet bee venom pharmacopuncture for chemotherapy-induced peripheral neuropathy. J. Acupunct. Meridian Stud. 2012, 5, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Li, D.X.; Yoon, H.; Go, D.; Quan, F.S.; Min, B.I.; Kim, S.K. Serotonergic mechanism of the relieving effect of bee venom acupuncture on oxaliplatin-induced neuropathic cold allodynia in rats. BMC Complement. Altern. Med. 2014, 14, 471. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Kim, M.J.; Yoon, I.; Li, D.X.; Bae, H.; Kim, S.K. Nicotinic acetylcholine receptors mediate the suppressive effect of an injection of diluted bee venom into the gv3 acupoint on oxaliplatin-induced neuropathic cold allodynia in rats. Biol. Pharm. Bull. 2015, 38, 710–714. [Google Scholar] [CrossRef] [PubMed]
- Baek, Y.H.; Huh, J.E.; Lee, J.D.; Choi, D.Y.; Park, D.S. Antinociceptive effect and the mechanism of bee venom acupuncture (apipuncture) on inflammatory pain in the rat model of collagen-induced arthritis: Mediation by alpha2-adrenoceptors. Brain Res. 2006, 1073–1074, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Eze, O.B.; Nwodo, O.F.; Ogugua, V.N. Therapeutic effect of honey bee venom. Proteins (enzymes) 2016, 1, 2. [Google Scholar]
- Gilron, I.; Bailey, J.M.; Tu, D.; Holden, R.R.; Weaver, D.F.; Houlden, R.L. Morphine, gabapentin, or their combination for neuropathic pain. N. Engl. J. Med. 2005, 352, 1324–1334. [Google Scholar] [CrossRef] [PubMed]
- Ormseth, M.J.; Scholz, B.A.; Boomershine, C.S. Duloxetine in the management of diabetic peripheral neuropathic pain. Patient Prefer. Adher. 2011, 5, 343–356. [Google Scholar]
- Serpell, M.G. Gabapentin in neuropathic pain syndromes: A randomised, double-blind, placebo-controlled trial. Pain 2002, 99, 557–566. [Google Scholar] [CrossRef]
- Vinik, A.I.; Casellini, C.M. Guidelines in the management of diabetic nerve pain: Clinical utility of pregabalin. Diabetes Metab. Syndr. Obes. Targets Ther. 2013, 6, 57–78. [Google Scholar] [CrossRef] [PubMed]
- Son, D.J.; Lee, J.W.; Lee, Y.H.; Song, H.S.; Lee, C.K.; Hong, J.T. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol. Ther. 2007, 115, 246–270. [Google Scholar] [CrossRef] [PubMed]
- Ling, B.; Coudore, F.; Decalonne, L.; Eschalier, A.; Authier, N. Comparative antiallodynic activity of morphine, pregabalin and lidocaine in a rat model of neuropathic pain produced by one oxaliplatin injection. Neuropharmacology 2008, 55, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Baron, R.; Binder, A.; Wasner, G. Neuropathic pain: Diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. 2010, 9, 807–819. [Google Scholar] [CrossRef]
- Cata, J.P.; Weng, H.R.; Chen, J.H.; Dougherty, P.M. Altered discharges of spinal wide dynamic range neurons and down-regulation of glutamate transporter expression in rats with paclitaxel-induced hyperalgesia. Neuroscience 2006, 138, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lee, Y.; Kim, W.; Lee, K.; Bae, H.; Kim, S.K. Analgesic effects of bee venom derived phospholipase a2 in a mouse model of oxaliplatin-induced neuropathic pain. Toxins 2015, 7, 2422–2434. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ke, T.; He, C.; Cao, W.; Wei, M.; Zhang, L.; Zhang, J.-X.; Wang, W.; Ma, J.; Wang, Z.-R. The anti-arthritic effects of synthetic melittin on the complete freund’s adjuvant-induced rheumatoid arthritis model in rats. Am. J. Chin. Med. 2010, 38, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lao, L.; Ren, K.; Berman, B.M. Mechanisms of acupuncture–electroacupuncture on persistent pain. Anesthesiology 2014, 120, 482–503. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lariviere, W.R. The nociceptive and anti-nociceptive effects of bee venom injection and therapy: A double-edged sword. Prog. Neurobiol. 2010, 92, 151–183. [Google Scholar] [CrossRef] [PubMed]
- Gim, G.-T.; Lee, J.-h.; Park, E.; Sung, Y.-H.; Kim, C.-J.; Hwang, W.-w.; Chu, J.-P.; Min, B.-I. Electroacupuncture attenuates mechanical and warm allodynia through suppression of spinal glial activation in a rat model of neuropathic pain. Brain Res. Bull. 2011, 86, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-C.; Feldman, S.C.; Cook, D.B.; Hung, D.-L.; Xu, T.; Kalnin, A.J.; Komisaruk, B.R. Fmri study of acupuncture-induced periaqueductal gray activity in humans. Neuroreport 2004, 15, 1937–1940. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-b.; Kang, M.-s.; Ahn, C.-j.; Han, H.-j.; Ahn, B.-c.; Lee, J.-h. Effect of high or low frequency electroacupuncture on the cellular actitivy of catecholaminergic neurons in the brain stem. Acupunct. Electro Ther. Res. 2000, 25, 27–36. [Google Scholar] [CrossRef]
- Young Bae, K.; Ho Jae, H.; Alvin, J.B.; Jang Hern, L. Bee venom acupoint stimulation increases fos expression in catecholaminergic neurons in the rat brain. Mol. Cells 2004, 17, 329–333. [Google Scholar]
- Kwon, Y.B.; Yoon, S.Y.; Kim, H.W.; Roh, D.H.; Kang, S.Y.; Ryu, Y.H.; Choi, S.M.; Han, H.J.; Lee, H.J.; Kim, K.W.; et al. Substantial role of locus coeruleus-noradrenergic activation and capsaicin-insensitive primary afferent fibers in bee venom’s anti-inflammatory effect. Neurosci. Res. 2006, 55, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-W.; Kwon, Y.-B.; Ham, T.-W.; Roh, D.-H.; Yoon, S.-Y.; Lee, H.-J.; Han, H.-J.; Yang, I.-S.; Beitz, A.J.; Lee, J.-H. Acupoint stimulation using bee venom attenuates formalin-induced pain behavior and spinal cord fos expression in rats. J. Vet. Med. Sci. 2003, 65, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.L. Chapter 29—descending noradrenergic influences on pain. In Progress in Brain Research; Barnes, C.D., Pompeiano, O., Eds.; Elsevier: Amsterdam, The Netherlands, 1991; Volume 88, pp. 381–394. [Google Scholar]
- Yoon, S.Y.; Roh, D.H.; Kwon, Y.B.; Kim, H.W.; Seo, H.S.; Han, H.J.; Lee, H.J.; Beitz, A.J.; Lee, J.H. Acupoint stimulation with diluted bee venom (apipuncture) potentiates the analgesic effect of intrathecal clonidine in the rodent formalin test and in a neuropathic pain model. J. Pain 2009, 10, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhu, B.-P.; Cai, L. Therapeutic effect of melittin on a rat model of chronic prostatitis induced by complete freund’s adjuvant. Biomed. Pharmacother. 2017, 90, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar] [CrossRef]
- Flatters, S.J.; Bennett, G.J. Ethosuximide reverses paclitaxel- and vincristine-induced painful peripheral neuropathy. Pain 2004, 109, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Polomano, R.C.; Mannes, A.J.; Clark, U.S.; Bennett, G.J. A painful peripheral neuropathy in the rat produced by the chemotherapeutic drug, paclitaxel. Pain 2001, 94, 293–304. [Google Scholar] [CrossRef]
- Flatters, S.J.L.; Xiao, W.-H.; Bennett, G.J. Acetyl-l-carnitine prevents and reduces paclitaxel-induced painful peripheral neuropathy. Neurosci. Lett. 2006, 397, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.S.; Jeong, H.S.; Park, H.J.; Baik, Y.; Yoon, M.H.; Choi, C.B.; Koh, H.G. A proposed transpositional acupoint system in a mouse and rat model. Res. Vet. Sci. 2008, 84, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Yamada, A.; Kim, W.; Kim, S.K.; Furue, H. Noradrenergic inhibition of spinal hyperexcitation elicited by cutaneous cold stimuli in rats with oxaliplatin-induced allodynia: Electrophysiological and behavioral assessments. J. Physiol. Sci. JPS 2016. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.; Jeon, C.; Lee, J.H.; Jang, J.U.; Quan, F.S.; Lee, K.; Kim, W.; Kim, S.K. Suppressive Effects of Bee Venom Acupuncture on Paclitaxel-Induced Neuropathic Pain in Rats: Mediation by Spinal α2-Adrenergic Receptor. Toxins 2017, 9, 351. https://doi.org/10.3390/toxins9110351
Choi J, Jeon C, Lee JH, Jang JU, Quan FS, Lee K, Kim W, Kim SK. Suppressive Effects of Bee Venom Acupuncture on Paclitaxel-Induced Neuropathic Pain in Rats: Mediation by Spinal α2-Adrenergic Receptor. Toxins. 2017; 9(11):351. https://doi.org/10.3390/toxins9110351
Chicago/Turabian StyleChoi, Jiho, Changhoon Jeon, Ji Hwan Lee, Jo Ung Jang, Fu Shi Quan, Kyungjin Lee, Woojin Kim, and Sun Kwang Kim. 2017. "Suppressive Effects of Bee Venom Acupuncture on Paclitaxel-Induced Neuropathic Pain in Rats: Mediation by Spinal α2-Adrenergic Receptor" Toxins 9, no. 11: 351. https://doi.org/10.3390/toxins9110351
APA StyleChoi, J., Jeon, C., Lee, J. H., Jang, J. U., Quan, F. S., Lee, K., Kim, W., & Kim, S. K. (2017). Suppressive Effects of Bee Venom Acupuncture on Paclitaxel-Induced Neuropathic Pain in Rats: Mediation by Spinal α2-Adrenergic Receptor. Toxins, 9(11), 351. https://doi.org/10.3390/toxins9110351





