Alternagin-C (ALT-C), a Disintegrin-Like Cys-Rich Protein Isolated from the Venom of the Snake Rhinocerophis alternatus, Stimulates Angiogenesis and Antioxidant Defenses in the Liver of Freshwater Fish, Hoplias malabaricus
Abstract
1. Introduction
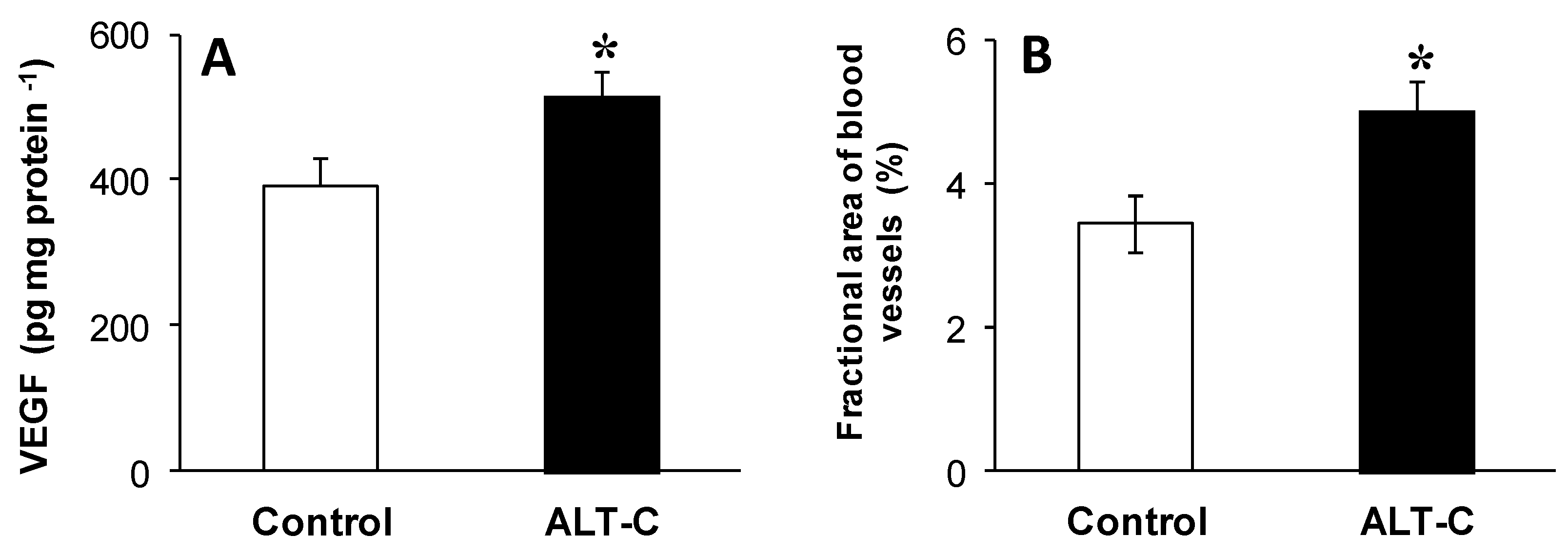
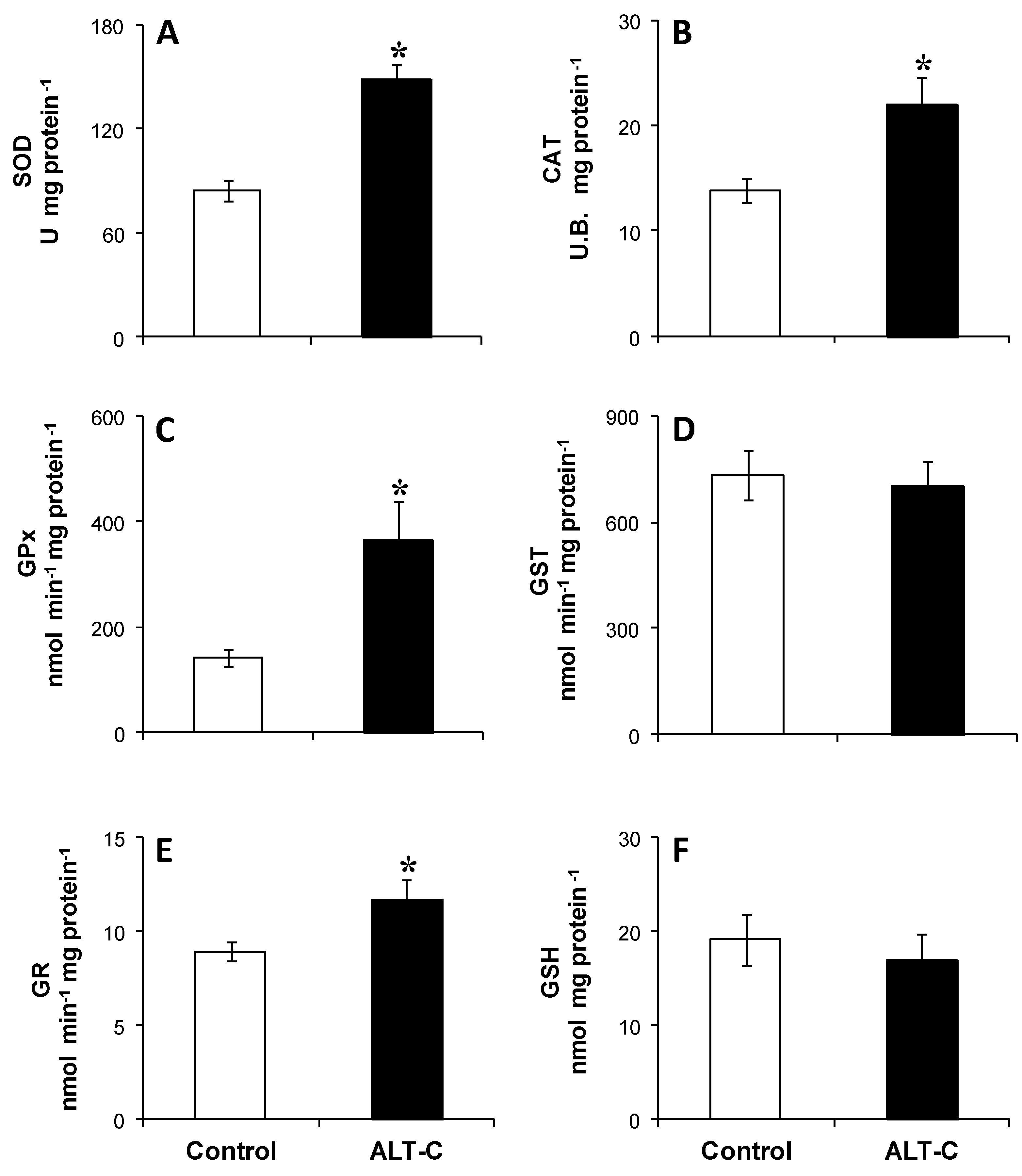
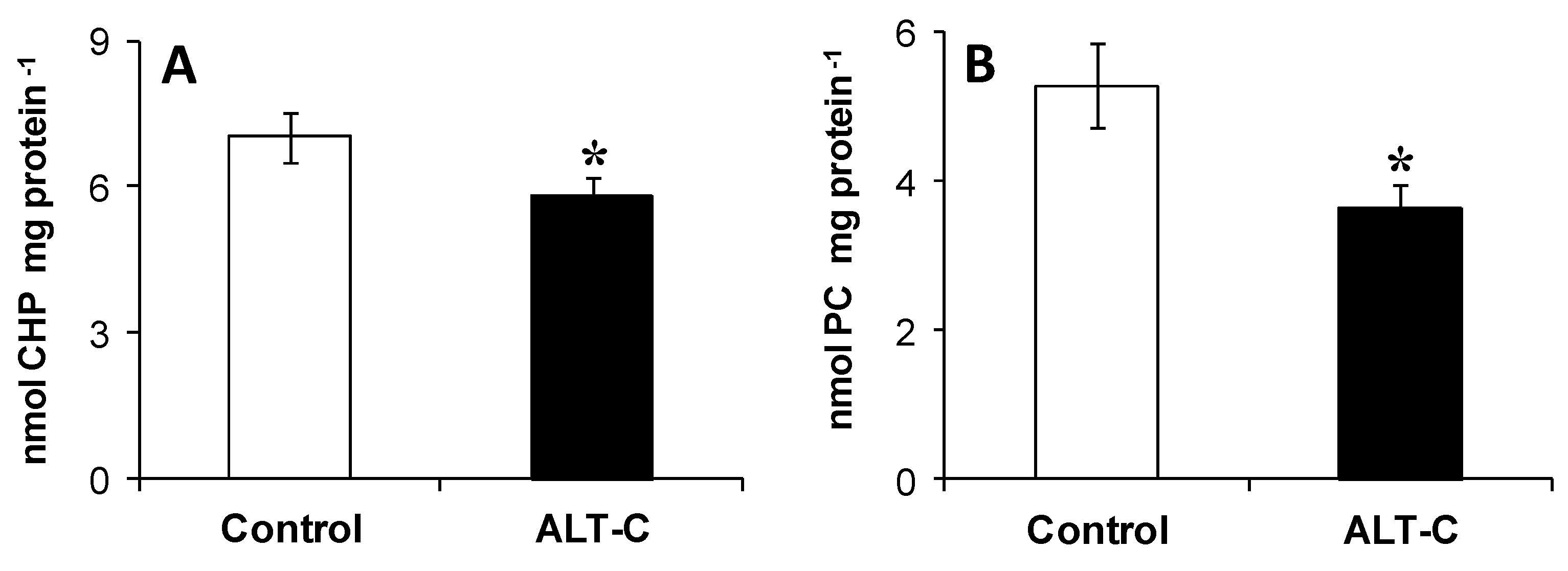
2. Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animals
5.2. Alternagin-C (ALT-C)
5.3. Experimental Design and Treatment
5.4. Liver Morphological Analysis
5.5. Quantification of VEGF
5.6. Biomarkers for Antioxidant Defense and Oxidative Damage
5.6.1. Antioxidant Defenses
5.6.2. Oxidative Damage
5.7. Total Protein
5.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Huancahuire-Vega, S.; Ponce-Soto, L.A.; Martins-de-Souza, D.; Marangoni, S. Biochemical and pharmacological characterization of PhTX-I a new myotoxic phospholipase A isolated from snake venom. Comp. Biochem. Physiol. C 2011, 154, 108–119. [Google Scholar] [CrossRef]
- Ramos, O.H.P.; Selistre-de-Araujo, H.S. Snake venom metalloproteases—Structure and function of catalytic and disintegrin domains. Comp. Biochem. Physiol. C 2006, 142, 328–346. [Google Scholar] [CrossRef] [PubMed]
- Selistre-de-Araujo, H.S.; Pontes, C.L.; Montenegro, C.F.; Martin, A.C. Snake venom disintegrins and cell migration. Toxins 2010, 2, 2606–2621. [Google Scholar] [CrossRef] [PubMed]
- Ramos, O.H.; Terruggi, C.H.; Ribeiro, J.U.; Cominetti, M.R.; Figueiredo, C.C.; Berard, M.; Crepin, M.; Morandi, V.; Selistre-de-Araujo, H.S. Modulation of in vitro and in vivo angiogenesis by alternagin-C, a disintegrin-like protein from Bothrops alternatus snake venom and by a peptide derived from its sequence. Arch. Biochem. Biophys. 2007, 461, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Teklemariam, T.; Seoane, A.I.; Ramos, C.J.; Sanchez, E.E.; Lucena, S.E.; Perez, J.C.; Mandal, S.A.; Soto, J.G. Functional analysis of a recombinant PIII-SVMP, GST-acocostatin; an apoptotic inducer of HUVEC and HeLa, but not SK-Mel-28 cells. Toxicon 2011, 57, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Cominetti, M.R.; Terruggi, C.H.; Ramos, O.H.; Fox, J.W.; Mariano-Oliveira, A.; De Freitas, M.S.; Figueiredo, C.C.; Morandi, V.; Selistre-de-Araujo, H.S. Alternagin-C, a disintegrin-like protein, induces vascular endothelial cell growth factor (VEGF) expression and endothelial cell proliferation in vitro. J. Biol. Chem. 2004, 279, 18247–18255. [Google Scholar] [CrossRef] [PubMed]
- Selistre-de-Araujo, H.S.; Cominetti, M.R.; Terruggi, C.H.B.; Mariano-Oliveira, A.; De Freitas, M.S.; Crepin, M.; Figueiredo, C.C.; Morandi, V. Alternagin-C, a disintegrin-like protein from the venom of Bothrops alternatus, modulates α2β1 integrin-mediated cell adhesion, migration and proliferation. Braz. J. Med. Biol. Res. 2005, 38, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Hinton, D.E.; Segner, H.; Au, D.W.T.; Kullman, S.W.; Hardman, R.C. Liver toxicology. In The Toxicology of Fishes; Di Giulio, R.T., Hinton, D.E., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 327–400. [Google Scholar]
- Vliegenthart, A.D.B.; Tucker, C.S.; Del Pozo, J.; Dear, J.W. Zebrafish as model organisms for studying drug-induced liver injury. Br. J. Clin. Pharmacol. 2014, 78, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Bocca, C.; Novo, E.; Miglietta, A.; Parola, M. Angiogenesis and fibrogenesis in chronic liver diseases. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Souza, D.H.; Iemma, M.R.; Ferreira, L.L.; Faria, J.P.; Oliva, M.L.; Zingali, R.B.; Niewiarowski, S.; Selistre-de-Araujo, H.S. The disintegrin-like domain of the snake venom metalloprotease alternagin inhibits alpha2beta1 integrin-mediated cell adhesion. Arch. Biochem. Biophys. 2000, 384, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, M. Vascular endothelial growth factor (VEGF) and its receptor (VEGFR) signaling in angiogenesis: A crucial target for anti- and pro-angiogenic therapies. Genes Cancer 2011, 2, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Sant’Ana, E.M.; Gouvea, C.M.; Nakaie, C.R.; Selistre-de-Araujo, H.S. Angiogenesis and growth factor modulation induced by alternagin C, a snake venom disintegrin-like, cysteine-rich protein on a rat skin wound model. Arch. Biochem. Biophys. 2008, 479, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Sant’Ana, E.M.; Gouvêa, C.M.; Durigan, J.L.; Cominetti, M.R.; Pimentel, E.R.; Selistre-de-Araújo, H.S. Rat skin wound healing induced by alternagin-C, a disintegrin-like, Cys-rich protein from Bothrops alternatus venom. Int. Wound J. 2011, 8, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Senger, D.R.; Perruzzi, C.A.; Streit, M.; Koteliansky, V.E.; de Fougerolles, A.R.; Detmar, M. The α1β1 and α2β1 integrins provide critical support for vascular endothelial growth factor signaling, endothelial cell migration, and tumor angiogenesis. Am. J. Pathol. 2002, 160, 195–204. [Google Scholar] [CrossRef]
- Monteiro, D.A.; Kalinin, A.L.; Selistre-de-Araujo, H.S.; Vasconcelos, E.S.; Rantin, F.T. Alternagin-C (ALT-C), a disintegrin-like protein from snake venom promotes positive inotropism and chronotropism in fish heart. Toxicon 2016, 110, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Schaeck, M.; Van den Broeck, W.; Hermans, K.; Decostere, A. Fish as research tools: Alternatives to in vivo experiments. Altern. Lab. Anim. 2013, 41, 219–229. [Google Scholar] [PubMed]
- Gao, L.; Laude, K.; Cai, H. Mitochondrial pathophysiology, reactive oxygen species, and cardiovascular diseases. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Cheng, D.; Ma, Y.; Zhou, W.; Wang, Y. The relationship between oxygen concentration, reactive oxygen species and the biological characteristics of human bone marrow hematopoietic stem cells. Transplant. Proc. 2011, 43, 2755–2761. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tan, H.Y.; Wang, N.; Zhang, Z.J.; Lao, L.; Wong, C.W.; Feng, Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef] [PubMed]
- Scandalios, J.G. Oxidative stress: Molecular perception and transduction of signals triggering antioxidant gene defenses. Braz. J. Med. Biol. Res. 2005, 38, 995–1014. [Google Scholar] [CrossRef] [PubMed]
- Storey, K.B. Oxidative stress: Animal adaptation in nature. Braz. J. Med. Res. 1996, 29, 1715–1733. [Google Scholar]
- Halliwell, B. Free radicals, antioxidants, and human disease: Curiosity, cause, or consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Pandey, S.; Parvez, S.; Sayeed, I.; Haque, R.; Bin-Hafeez, B.; Raisuddin, S. Biomarkers of oxidative stress: A comparative study of river Yamuna fish Wallago attu (Bl. & Schn.). Sci. Total Environ. 2003, 309, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Regoli, F.; Winston, G.W.; Gorbi, S.; Frenzilli, G.; Nigro, M.; Corsi, I.; Focardi, S. Integrating enzymatic responses to organic chemical exposure with total oxyradical absorbing capacity and DNA damage in the European eel Anguilla anguilla. Environ. Toxicol. Chem. 2003, 22, 2120–2129. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, D.A.; Rantin, F.T.; Kalinin, A.L. Inorganic mercury exposure: Toxicological effects, oxidative stress biomarkers and bioaccumulation in the tropical freshwater fish matrinxã, Brycon amazonicus (Spix and Agassiz, 1829). Ecotoxicology 2010, 19, 105–123. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.R. The glutathione peroxidases. Cell. Mol. Life Sci. 1994, 57, 1825–1835. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Koh, Y.H.; Park, Y.S.; Fujiwara, N.; Sakiyama, H.; Misonou, Y.; Ookawara, T.; Suzuki, K.; Honke, K.; Taniguchi, N. Oxidative stress caused by inactivation of glutathione peroxidase and adaptive responses. Biol. Chem. 2003, 384, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Van der Oost, R.; Beyer, J.; Vermeulen, N.P.E. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ. Toxicol. Pharmacol. 2003, 13, 57–149. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, H.; Wang, X.; Wu, J.; Xue, Y. Effects of chronic exposure of 2,4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 2004, 55, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Zhang, H.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Aspects Med. 2009, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pierrick, L.; Haoues, A.; Luc, D.; Nicole, P.; Mylène, W. Evolution of resistance to insecticide in disease vectors. In Genetics and Evolution of Infectious Disease; Tibayrenc, M., Ed.; Elsevier: London, UK, 2011; pp. 363–409. [Google Scholar] [CrossRef]
- Townsend, D.M.; Manevich, Y.; He, L.; Hutchens, S.; Pazoles, C.J.; Tew, K.D. Novel role for glutathione S-transferase pi. Regulator of protein S-Glutathionylation following oxidative and nitrosative stress. J. Biol. Chem. 2009, 284, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Manautou, J.E. Molecular mechanisms underlying chemical liver injury. Expert Rev. Mol. Med. 2012, 14, e4. [Google Scholar] [CrossRef] [PubMed]
- Kalgutkar, A.S. Handling reactive metabolite positives in drug discovery: What has retrospective structure-toxicity analyses taught us? Chem. Biol. Interact. 2011, 192, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Izuta, H.; Matsunaga, N.; Shimazawa, M.; Sugiyama, T.; Ikeda, T.; Hara, H. Proliferative diabetic retinopathy and relations among antioxidant activity, oxidative stress, and VEGF in the vitreous body. Mol. Vis. 2010, 16, 130–136. [Google Scholar] [PubMed]
- Kweider, N.; Fragoulis, A.; Rosen, C.; Pecks, U.; Rath, W.; Pufe, T.; Wruck, C.J. Interplay between vascular endothelial growth factor (VEGF) and nuclear factor erythroid 2-related factor-2 (Nrf2): Implications for preeclampsia. J. Biol. Chem. 2011, 286, 42863–42872. [Google Scholar] [CrossRef] [PubMed]
- Kleinow, K.M.; Nichols, J.W.; Hayton, W.L.; McKim, J.M.; Barron, M.G. Toxicokinetics, in fishes. In The Toxicology of Fishes; Di Giulio, R.T., Hinton, D.E., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 55–152. [Google Scholar]
- Wolf, J.C.; Wolfe, M.J. A brief overview of nonneoplastic hepatic toxicity in fish. Toxicol. Pathol. 2005, 33, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, M.; Fritsche, R. Cannulation techniques. In Analytical Techniques. Biochemistry and Molecular Biology of Fishes; Hochachka, P.W., Mommsen, T.P., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1994; pp. 17–36. [Google Scholar]
- Flohé, L.; Ötting, F. Superoxide dismutase assays. Methods Enzymol. 1984, 105, 93–105. [Google Scholar] [PubMed]
- Aebi, H. Catalase. In Methods of Enzymatic Analysis; Bergmayer, H.U., Ed.; Academic Press: London, UK, 1874; pp. 671–684. [Google Scholar]
- Bergmeyer, H.U. Measurement of catalase activity. Biochem. Z. 1955, 327, 255–258. [Google Scholar] [PubMed]
- Nakamura, W.; Hosoda, S.; Hayashi, K. Purification and properties of rat liver glutathione peroxidase. Biochem. Biophys. Acta 1974, 358, 251–261. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferase, the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, I.; Mannervik, B. Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J. Biol. Chem. 1975, 250, 5475–5480. [Google Scholar] [PubMed]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar] [PubMed]
- Jiang, Z.Y.; Hunt, J.V.; Wolff, S.P. Ferrous ion oxidation in the presence of xylenol orange for detection of lipid hydroperoxide in low-density lipoprotein. Anal. Biochem. 1992, 202, 384–389. [Google Scholar] [CrossRef]
- Reznick, A.Z.; Packer, L. Oxidative damage to proteins: Spectrophotometric method for carbonyl assay. Methods Enzymol. 1994, 233, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M.A. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 218–251. [Google Scholar] [CrossRef]
- Kruger, N.J. The Bradford method for protein quantification. Meth. Mol. Biol. 1994, 32, 9–15. [Google Scholar] [CrossRef]
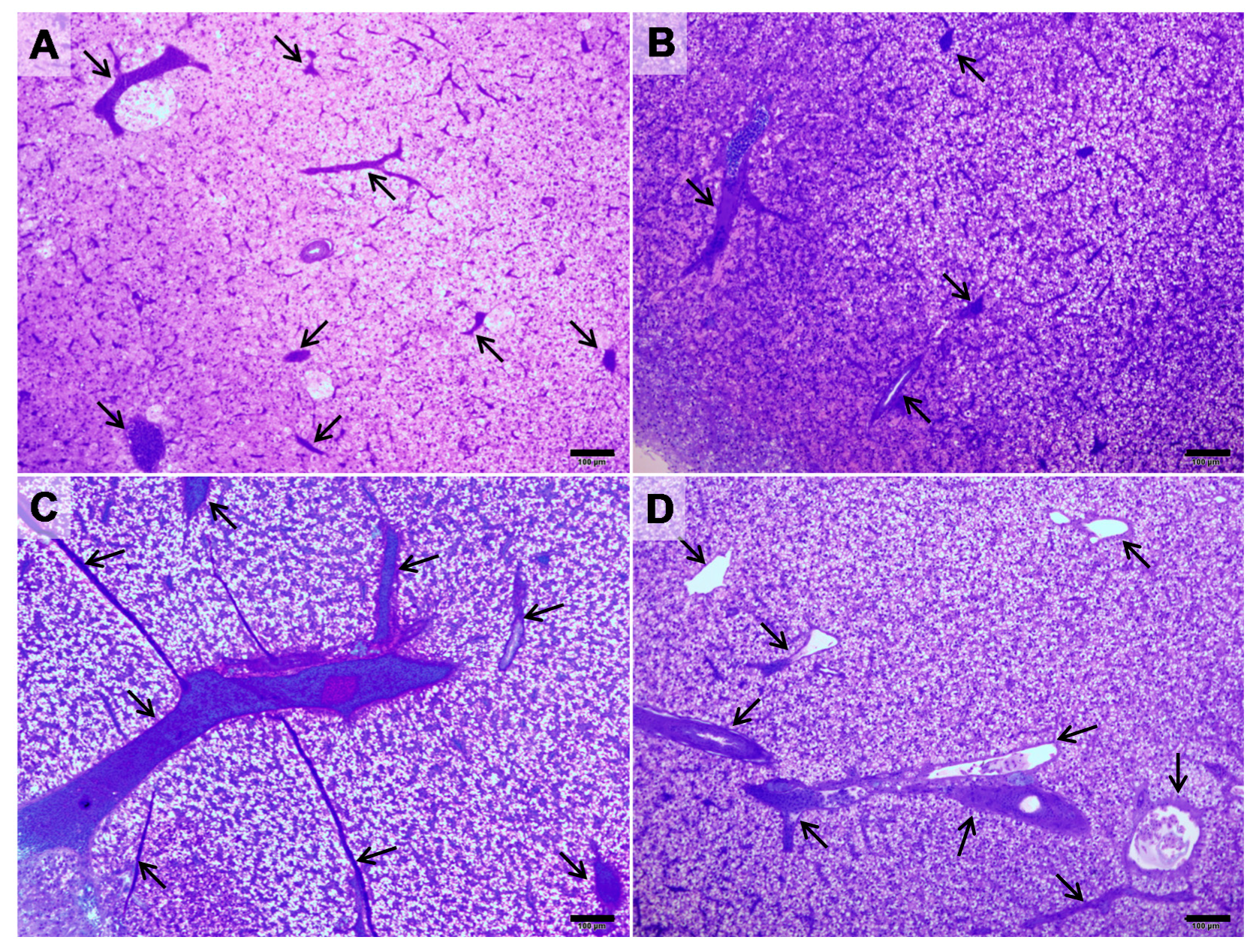
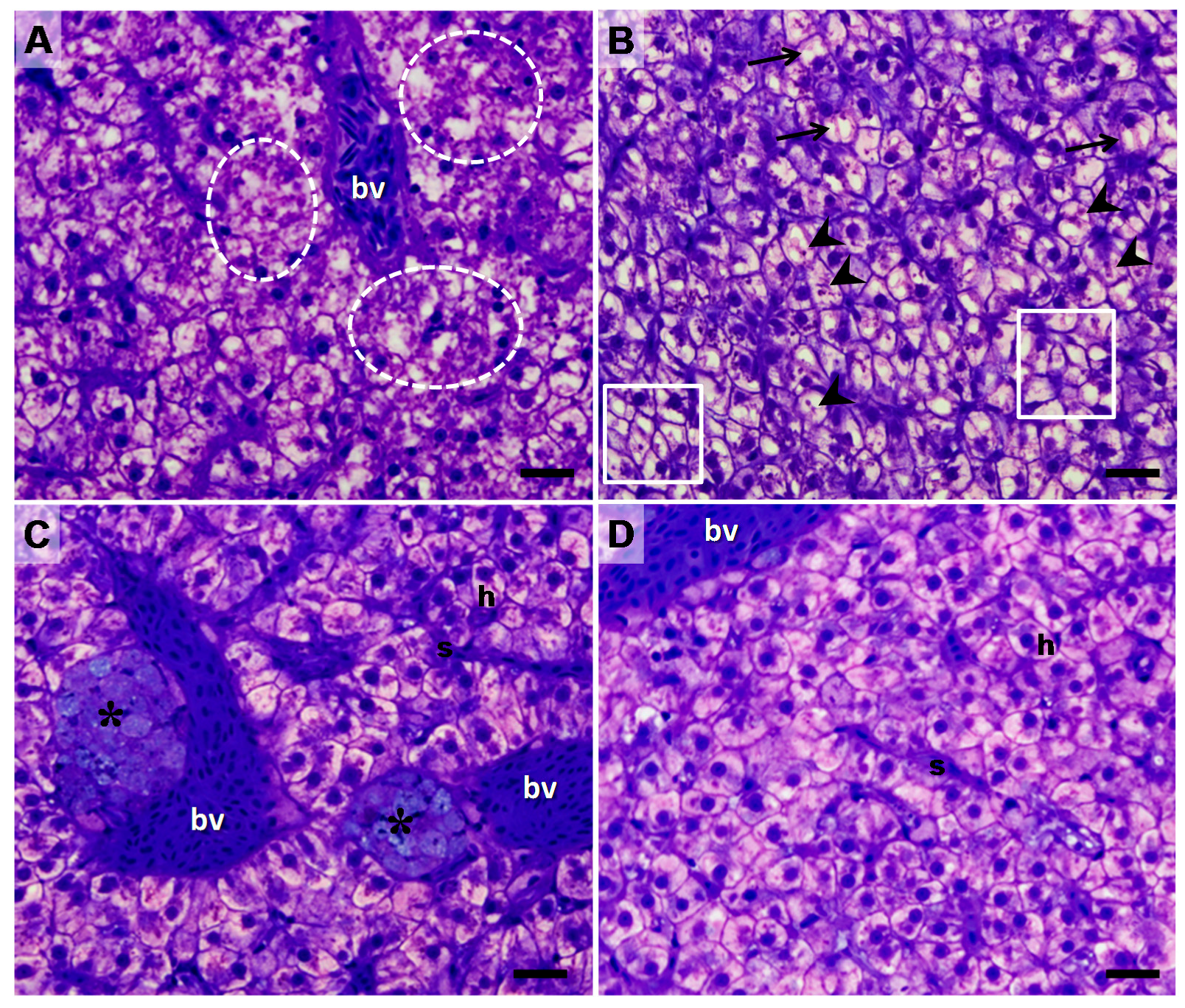
| Lesion | Control | ALT-C |
|---|---|---|
| Aneurism/Haemorrhage/Hyperaemia | A | A |
| Architectural and structural alterations | + | 0+ |
| Atrophy | + | A |
| Cytoplasmic vacuoles | 0+ | 0+ |
| Cytoplasmic degeneration | ++ | + |
| Eosinophilic granules in cytoplasm | 0+ | 0+ |
| Hypertrophy | A | A |
| Melano-macrophages centers | + | 0+ |
| Necrosis | A | A |
| Nuclear alterations | A | A |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monteiro, D.A.; Selistre-de-Araújo, H.S.; Tavares, D.; Fernandes, M.N.; Kalinin, A.L.; Rantin, F.T. Alternagin-C (ALT-C), a Disintegrin-Like Cys-Rich Protein Isolated from the Venom of the Snake Rhinocerophis alternatus, Stimulates Angiogenesis and Antioxidant Defenses in the Liver of Freshwater Fish, Hoplias malabaricus. Toxins 2017, 9, 307. https://doi.org/10.3390/toxins9100307
Monteiro DA, Selistre-de-Araújo HS, Tavares D, Fernandes MN, Kalinin AL, Rantin FT. Alternagin-C (ALT-C), a Disintegrin-Like Cys-Rich Protein Isolated from the Venom of the Snake Rhinocerophis alternatus, Stimulates Angiogenesis and Antioxidant Defenses in the Liver of Freshwater Fish, Hoplias malabaricus. Toxins. 2017; 9(10):307. https://doi.org/10.3390/toxins9100307
Chicago/Turabian StyleMonteiro, Diana Amaral, Heloisa Sobreiro Selistre-de-Araújo, Driele Tavares, Marisa Narciso Fernandes, Ana Lúcia Kalinin, and Francisco Tadeu Rantin. 2017. "Alternagin-C (ALT-C), a Disintegrin-Like Cys-Rich Protein Isolated from the Venom of the Snake Rhinocerophis alternatus, Stimulates Angiogenesis and Antioxidant Defenses in the Liver of Freshwater Fish, Hoplias malabaricus" Toxins 9, no. 10: 307. https://doi.org/10.3390/toxins9100307
APA StyleMonteiro, D. A., Selistre-de-Araújo, H. S., Tavares, D., Fernandes, M. N., Kalinin, A. L., & Rantin, F. T. (2017). Alternagin-C (ALT-C), a Disintegrin-Like Cys-Rich Protein Isolated from the Venom of the Snake Rhinocerophis alternatus, Stimulates Angiogenesis and Antioxidant Defenses in the Liver of Freshwater Fish, Hoplias malabaricus. Toxins, 9(10), 307. https://doi.org/10.3390/toxins9100307






