A Novel Toxin from Haplopelma lividum Selectively Inhibits the NaV1.8 Channel and Possesses Potent Analgesic Efficacy
Abstract
:1. Introduction
2. Results
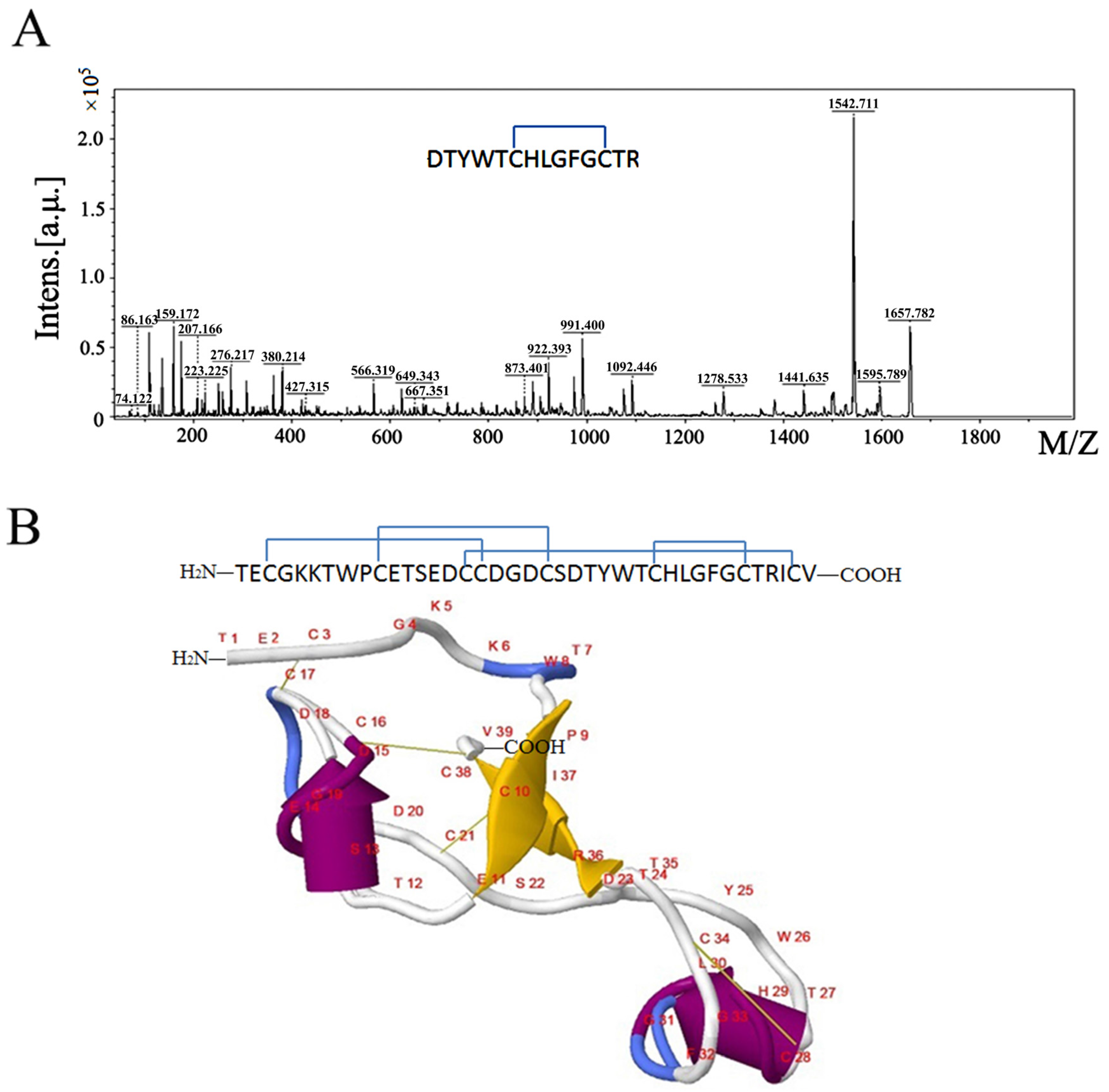
2.1. Sequence Analysis of µ-TRTX-Hl1a
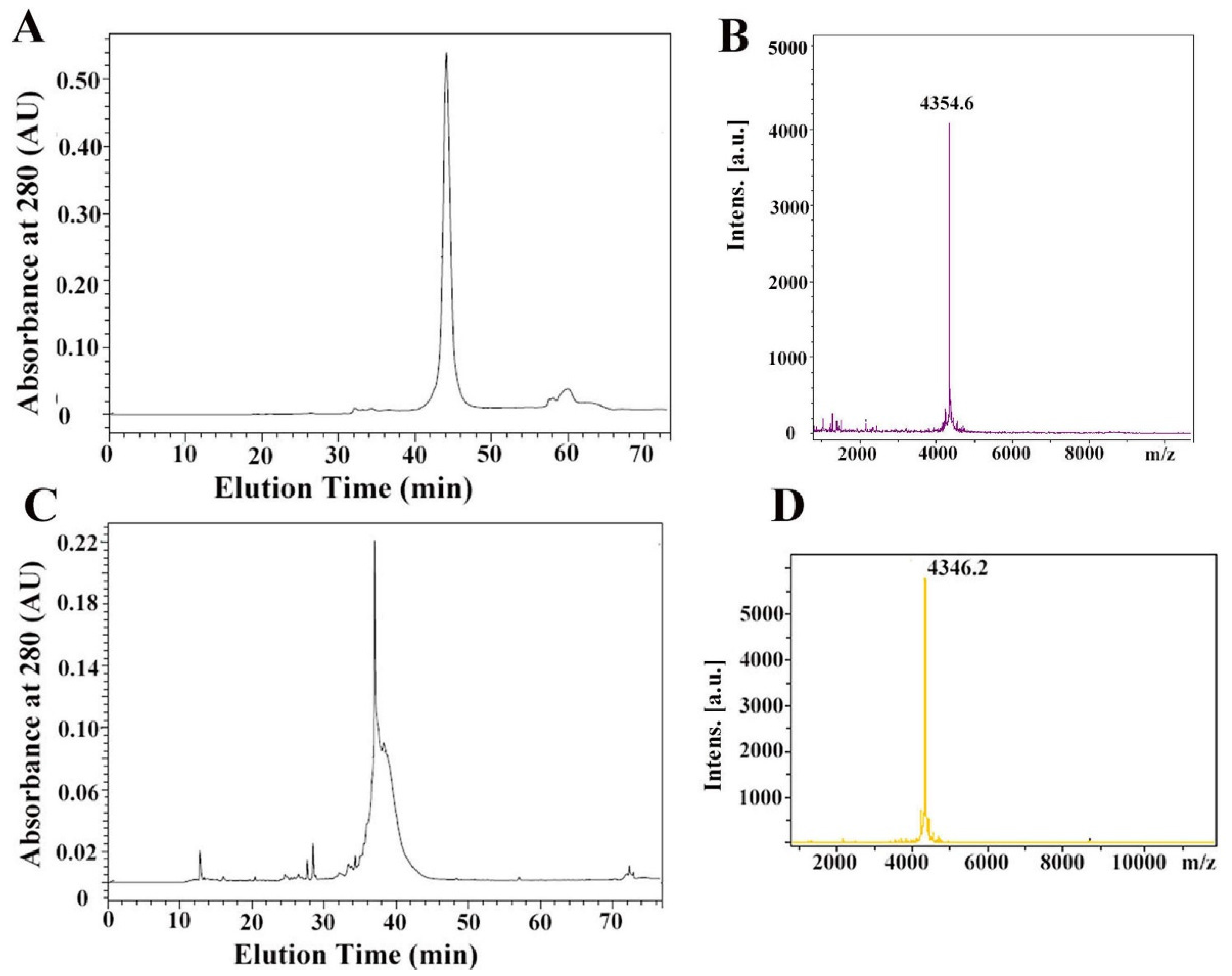
2.2. Synthesis and Refolding of µ-TRTX-Hl1a
2.3. The Secondary Structure and Disulfide Bond of µ-TRTX-Hl1a
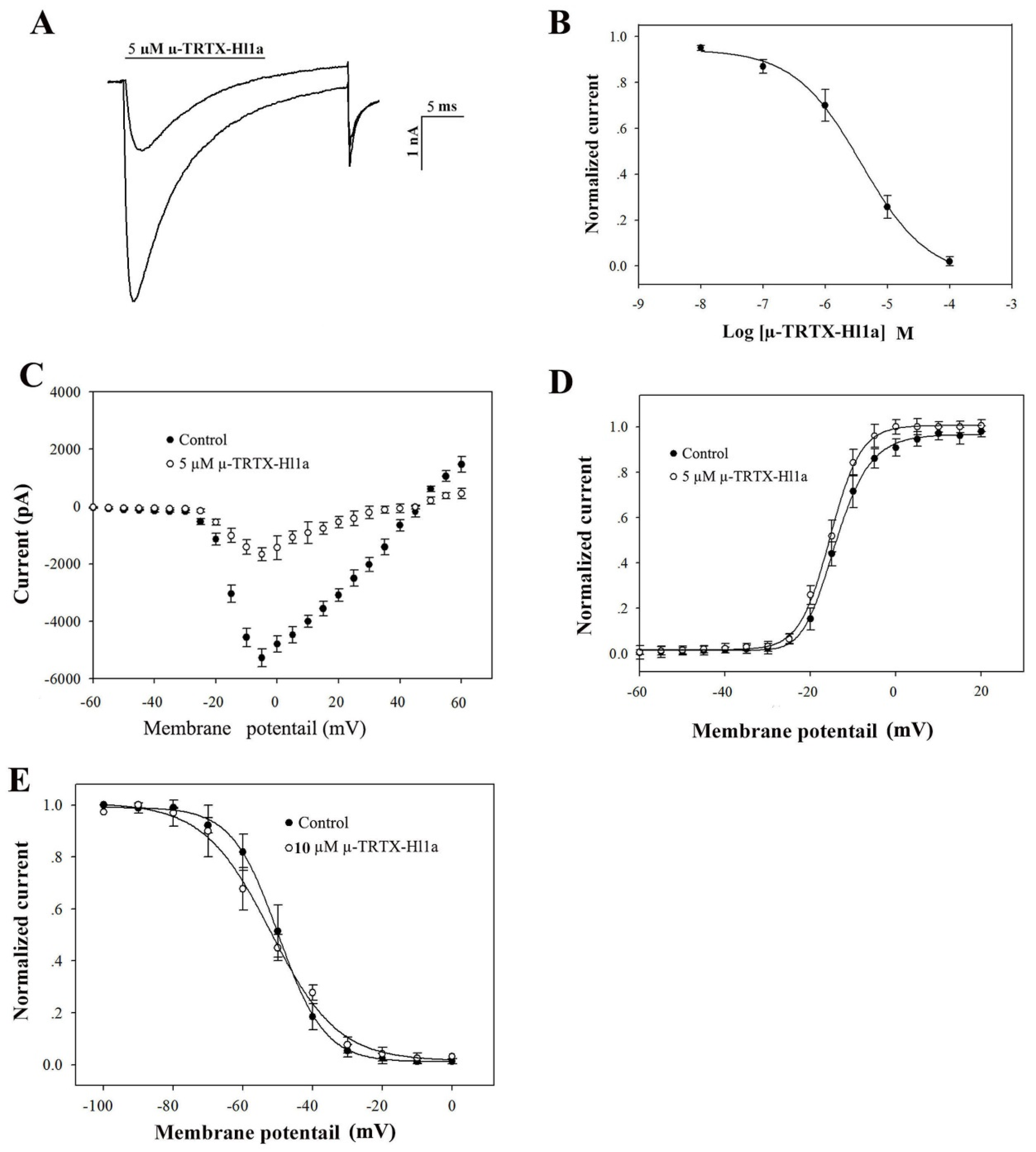
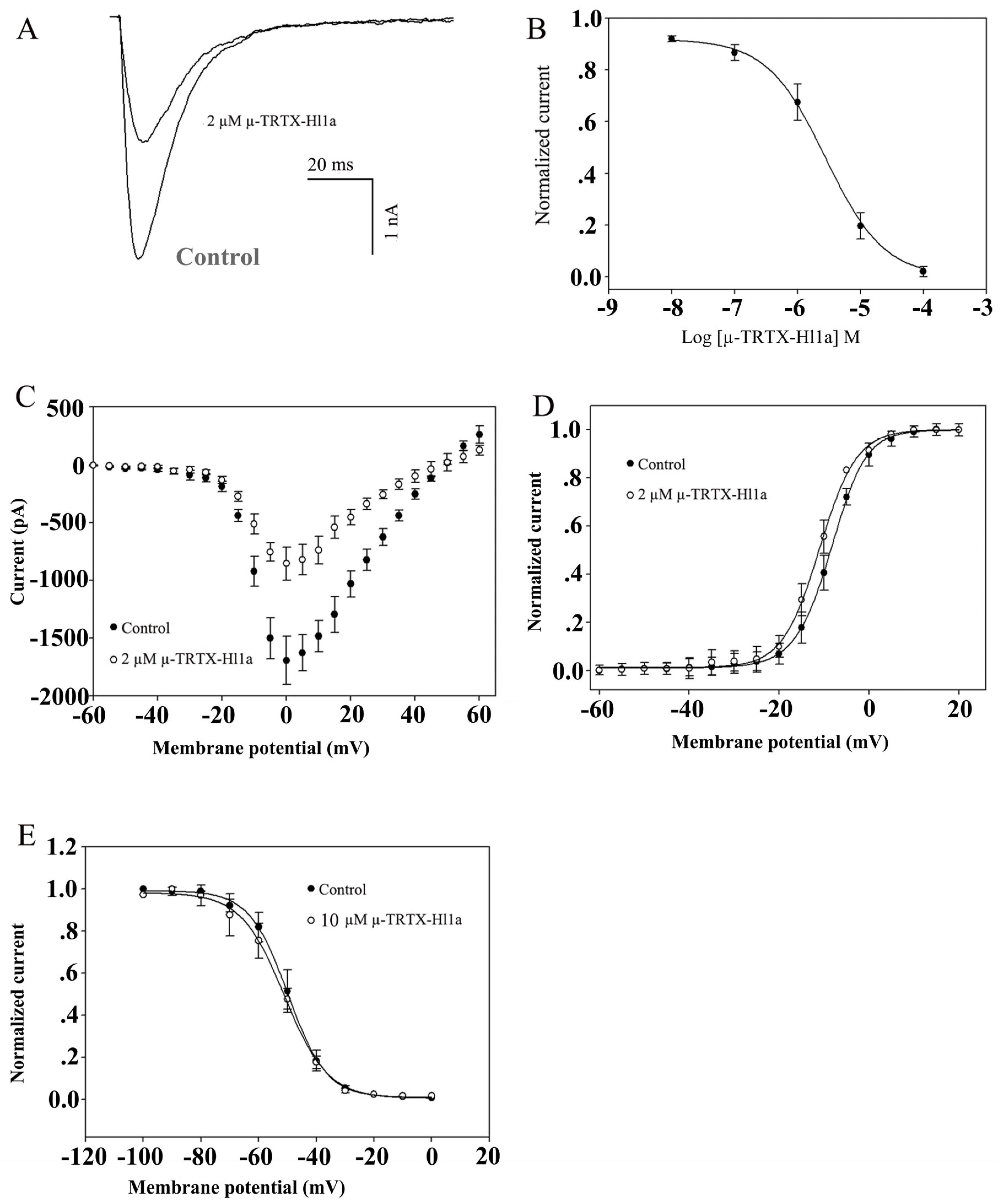
2.4. Effects of µ-TRTX-Hl1a on VGSCs
2.5. Effects of µ-TRTX-Hl1a on NaV1.8
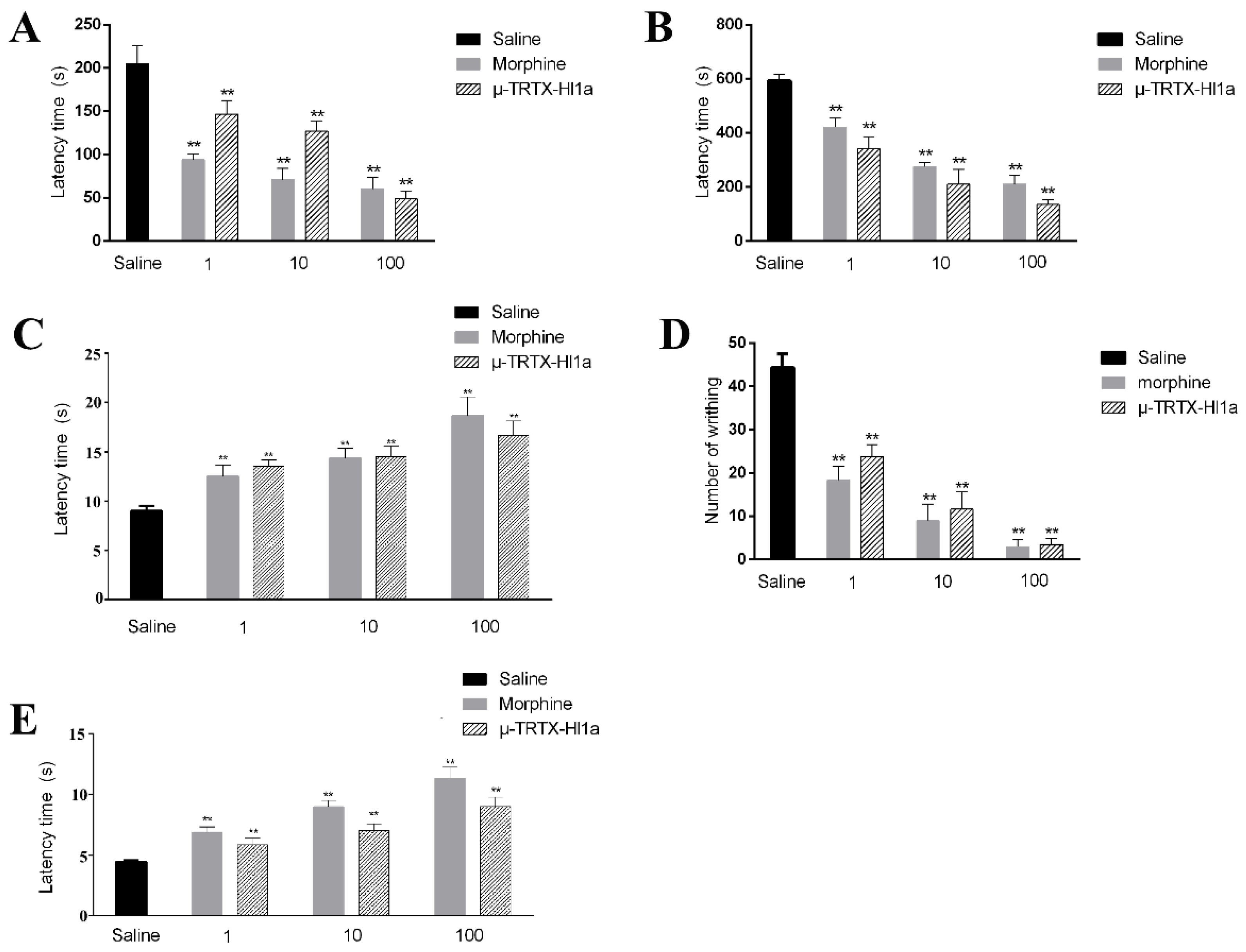
2.6. Effects of µ-TRTX-Hl1a on Pain
3. Discussion
4. Materials and Methods
4.1. cDNA Synthesis
4.2. cDNA Sequence and Bioinformatics Analysis
4.3. Peptide Synthesis and Refolding
4.4. Mass Spectrometric Analysis
4.5. Digestion of the Peptide
4.6. MALDI-TOF/TOF-MSMS
4.7. Structure Prediction and Modeling
4.8. Patch Clamp Recording on Rat Dorsal Root Ganglion (DRG) Neurons
4.9. Patch Clamp Recording on Sodium Channel Subtypes
4.10. Formalin-Induced Paw Licking
4.11. Tail-Flicking Test
4.12. Abdominal Writhing Response Caused by Acetic Acid
4.13. Hot Plate
4.14. Statistics
4.14.1. Data analysis for Patch Clamp
4.14.2. Data Analysis of the Animal Model
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Herzig, V.; Wood, D.L.; Newell, F.; Chaumeil, P.A.; Kaas, Q.; Binford, G.J.; Nicholson, G.M.; Gorse, D.; King, G.F. Arachnoserver 2.0, an updated online resource for spider toxin sequences and structures. Nucleic Acids Res. 2011, 39, D653–D657. [Google Scholar] [CrossRef] [PubMed]
- Klint, J.K.; Senff, S.; Rupasinghe, D.B.; Er, S.Y.; Herzig, V.; Nicholson, G.M.; King, G.F. Spider-venom peptides that target voltage-gated sodium channels: Pharmacological tools and potential therapeutic leads. Toxicon 2012, 60, 478–491. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, M.; Nakajima, S.; Sakae, H.; Nakamura, T.; Tohma, Y.; Shiono, S.; Tabuse, H. Tarantulas bite: Two case reports of finger bite from Haplopelma lividum. Chudoku Kenkyu 2001, 14, 247–250. [Google Scholar] [PubMed]
- Gaskin, D.J.; Richard, P. The economic costs of pain in the united states. J. Pain 2012, 13, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.I. Actions of atx-ii and other gating-modifiers on Na(+) currents in HEK-293 cells expressing WT and deltaKPQ hNa(v) 1.5 Na(+) channels. Toxicon 2009, 53, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Catterall, W.A.; Perez-Reyes, E.; Snutch, T.P.; Striessnig, J. International union of pharmacology. Xlviii. Nomenclature and structure-function relationships of voltage-gated calcium channels. Pharmacol. Rev. 2005, 57, 411–425. [Google Scholar] [CrossRef] [PubMed]
- Payandeh, J.; Scheuer, T.; Zheng, N.; Catterall, W.A. The crystal structure of a voltage-gated sodium channel. Nature 2011, 475, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Shields, S.D.; Ahn, H.S.; Yang, Y.; Han, C.; Seal, R.P.; Wood, J.N.; Waxman, S.G.; Dib-Hajj, S.D. Nav1.8 expression is not restricted to nociceptors in mouse peripheral nervous system. Pain 2012, 153, 2017–2030. [Google Scholar] [CrossRef] [PubMed]
- Faber, C.G.; Lauria, G.; Merkies, I.S.; Cheng, X.; Han, C.; Ahn, H.S.; Persson, A.K.; Hoeijmakers, J.G.; Gerrits, M.M.; Pierro, T.; et al. Gain-of-function nav1.8 mutations in painful neuropathy. Proc. Natl. Acad. Sci. USA 2012, 109, 19444–19449. [Google Scholar] [CrossRef] [PubMed]
- Garrison, S.R.; Weyer, A.D.; Barabas, M.E.; Beutler, B.A.; Stucky, C.L. A gain-of-function voltage-gated sodium channel 1.8 mutation drives intense hyperexcitability of a- and c-fiber neurons. Pain 2014, 155, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Bierhaus, A.; Fleming, T.; Stoyanov, S.; Leffler, A.; Babes, A.; Neacsu, C.; Sauer, S.K.; Eberhardt, M.; Schnolzer, M.; Lasitschka, F.; et al. Methylglyoxal modification of nav1.8 facilitates nociceptive neuron firing and causes hyperalgesia in diabetic neuropathy. Nat. Med. 2012, 18, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, M.F.; Honore, P.; Shieh, C.C.; Chapman, M.; Joshi, S.; Zhang, X.F.; Kort, M.; Carroll, W.; Marron, B.; Atkinson, R.; et al. A-803467, a potent and selective nav1.8 sodium channel blocker, attenuates neuropathic and inflammatory pain in the rat. Proc. Natl. Acad. Sci. USA 2007, 104, 8520–8525. [Google Scholar] [CrossRef] [PubMed]
- King, G.F.; Gentz, M.C.; Escoubas, P.; Nicholson, G.M. A rational nomenclature for naming peptide toxins from spiders and other venomous animals. Toxicon 2008, 52, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Deng, M.; He, Q.; Meng, E.; Jiang, L.; Liao, Z.; Rong, M.; Liang, S. Molecular diversity and evolution of cystine knot toxins of the tarantula Chilobrachys jingzhao. Cell. Mol. Life Sci. CMLS 2008, 65, 2431–2444. [Google Scholar] [CrossRef] [PubMed]
- Skinner, W.S.; Dennis, P.A.; Li, J.P.; Quistad, G.B. Identification of insecticidal peptides from venom of the trap-door spider, Aptostichus schlingeri (Ctenizidae). Toxicon 1992, 30, 1043–1050. [Google Scholar] [CrossRef]
- Bende, N.S.; Kang, E.; Herzig, V.; Bosmans, F.; Nicholson, G.M.; Mobli, M.; King, G.F. The insecticidal neurotoxin aps iii is an atypical knottin peptide that potently blocks insect voltage-gated sodium channels. Biochem. Pharmacol. 2013, 85, 1542–1554. [Google Scholar] [CrossRef] [PubMed]
- Phyre 2 tool. Available online: http://www.sbg.bio.ic.ac.uk/phyre2 (accessed on 15 June 2016).
- Deng, M.; Kuang, F.; Sun, Z.; Tao, H.; Cai, T.; Zhong, L.; Chen, Z.; Xiao, Y.; Liang, S. Jingzhaotoxin-ix, a novel gating modifier of both sodium and potassium channels from Chinese tarantula Chilobrachys jingzhao. Neuropharmacology 2009, 57, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Saez, N.J.; Senff, S.; Jensen, J.E.; Er, S.Y.; Herzig, V.; Rash, L.D.; King, G.F. Spider-venom peptides as therapeutics. Toxins 2010, 2, 2851–2871. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Xiao, Y.; Kang, D.; Liu, J.; Li, Y.; Undheim, E.A.; Klint, J.K.; Rong, M.; Lai, R.; King, G.F. Discovery of a selective Nav1.7 inhibitor from centipede venom with analgesic efficacy exceeding morphine in rodent pain models. Proc. Natl. Acad. Sci. USA 2013, 110, 17534–17539. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.; Smyth, W.F.; Gault, V.A.; O’Kane, E.; McClean, S. Mass spectrometric characterisation and quantitation of selected low molecular mass compounds from the venom of Haplopelma lividum (Theraphosidae). RCM 2009, 23, 1747–1755. [Google Scholar] [PubMed]
- Okumura, M.; Shimamoto, S.; Hidaka, Y. Chemical methods for producing disulfide bonds in peptides and proteins to study folding regulation. Curr. Protoc. Protein Sci. 2014, 76. [Google Scholar] [CrossRef]
- Middleton, R.E.; Warren, V.A.; Kraus, R.L.; Hwang, J.C.; Liu, C.J.; Dai, G.; Brochu, R.M.; Kohler, M.G.; Gao, Y.D.; Garsky, V.M.; et al. Two tarantula peptides inhibit activation of multiple sodium channels. Biochemistry 2002, 41, 14734–14747. [Google Scholar] [CrossRef] [PubMed]
- Bosmans, F.; Rash, L.; Zhu, S.; Diochot, S.; Lazdunski, M.; Escoubas, P.; Tytgat, J. Four novel tarantula toxins as selective modulators of voltage-gated sodium channel subtypes. Mol. Pharmacol. 2006, 69, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Luo, X.; Jiang, L.; Chen, H.; Wang, J.; He, H.; Liang, S. Synthesis and biological characterization of synthetic analogs of huwentoxin-iv (mu-theraphotoxin-hh2a), a neuronal tetrodotoxin-sensitive sodium channel inhibitor. Toxicon 2013, 71, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Blumenthal, K.; Cummins, T.R. Gating-pore currents demonstrate selective and specific modulation of individual sodium channel voltage-sensors by biological toxins. Mol. Pharmacol. 2014, 86, 159–167. [Google Scholar] [CrossRef] [PubMed]
- SignalP 4.1 program. Available online: http://www.cbs.dtu.dk/services/SignalP/ (accessed on 8 February 2014).
- Rong, M.; Chen, J.; Tao, H.; Wu, Y.; Jiang, P.; Lu, M.; Su, H.; Chi, Y.; Cai, T.; Zhao, L.; et al. Molecular basis of the tarantula toxin jingzhaotoxin-iii (beta-trtx-cj1alpha) interacting with voltage sensors in sodium channel subtype nav1.5. FASEB J. 2011, 25, 3177–3185. [Google Scholar] [CrossRef] [PubMed]
- Owoyele, V.B.; Adediji, J.O.; Soladoye, A.O. Anti-inflammatory activity of aqueous leaf extract of Chromolaena odorata. Inflammopharmacology 2005, 13, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Dong, L.; Zhao, T.; You, D.; Liu, R.; Liu, H.; Yang, H.; Lai, R. Analgesic and anti-inflammatory effects of the amphibian neurotoxin, anntoxin. Biochimie 2011, 93, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.A.; Arruda, A.; Silva, M.A.; Cardoso, C.A.; Vieira Mdo, C.; Kassuya, C.A.; Arena, A.C. Anti-inflammatory effects and acute toxicity of hydroethanolic extract of jacaranda decurrens roots in adult male rats. J. Ethnopharmacol. 2012, 144, 802–805. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, Z.; Liu, H.; He, X.; Zhang, Y.; Jin, J.; Che, J.; Li, C.; Chen, W.; Lai, R.; et al. Novel analgesic peptides from the tree frog of Hyla japonica. Biochimie 2014, 99, 38–43. [Google Scholar] [CrossRef] [PubMed]

| Buffer condition | Temperature (℃) | Duration (h) | pH | Folding Yield % |
|---|---|---|---|---|
| 0.1 mol/L Tris-HCl, 0.1 mol/L NaCl, 0.3 mmol/L GSSG, 3 mmol/L GSH | 25 | 24 | 7.2 | 6.04 |
| 4 | 24 | 7.2 | 7.08 | |
| 4 | 24 | 6.8 | 8.52 | |
| 4 | 48 | 6.8 | 8.46 | |
| 0.1 mol/L Tris-HCl, 0.1 mol/L NaCl, 0.3 mmol/L GSSG, 3 mmol/L GSH, 1 mol/L L-Arg | 4 | 24 | 7.6 | 8.13 |
| 4 | 24 | 7.4 | 8.53 | |
| 4 | 24 | 7.2 | 9.39 | |
| 4 | 24 | 6.8 | 10.38 | |
| 0.05 mol/L Tris-HCl, 0.05 mol/L NaCl, 0.15 mmol/L GSSG, 1.5 mmol/L GSH | 4 | 24 | 7.2 | 10.20 |
| 4 | 24 | 6.8 | 13.46 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, P.; Huang, H.; Wang, G.; Yang, S.; Lu, Q.; Liu, J.; Lai, R.; Rong, M. A Novel Toxin from Haplopelma lividum Selectively Inhibits the NaV1.8 Channel and Possesses Potent Analgesic Efficacy. Toxins 2017, 9, 7. https://doi.org/10.3390/toxins9010007
Meng P, Huang H, Wang G, Yang S, Lu Q, Liu J, Lai R, Rong M. A Novel Toxin from Haplopelma lividum Selectively Inhibits the NaV1.8 Channel and Possesses Potent Analgesic Efficacy. Toxins. 2017; 9(1):7. https://doi.org/10.3390/toxins9010007
Chicago/Turabian StyleMeng, Ping, Honggang Huang, Gan Wang, Shilong Yang, Qiuming Lu, Jingze Liu, Ren Lai, and Mingqiang Rong. 2017. "A Novel Toxin from Haplopelma lividum Selectively Inhibits the NaV1.8 Channel and Possesses Potent Analgesic Efficacy" Toxins 9, no. 1: 7. https://doi.org/10.3390/toxins9010007
APA StyleMeng, P., Huang, H., Wang, G., Yang, S., Lu, Q., Liu, J., Lai, R., & Rong, M. (2017). A Novel Toxin from Haplopelma lividum Selectively Inhibits the NaV1.8 Channel and Possesses Potent Analgesic Efficacy. Toxins, 9(1), 7. https://doi.org/10.3390/toxins9010007







