Hydrolytic Fate of 3/15-Acetyldeoxynivalenol in Humans: Specific Deacetylation by the Small Intestine and Liver Revealed Using in Vitro and ex Vivo Approaches
Abstract
:1. Introduction
2. Results
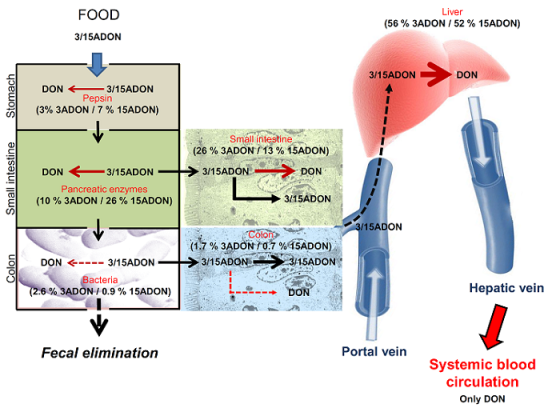
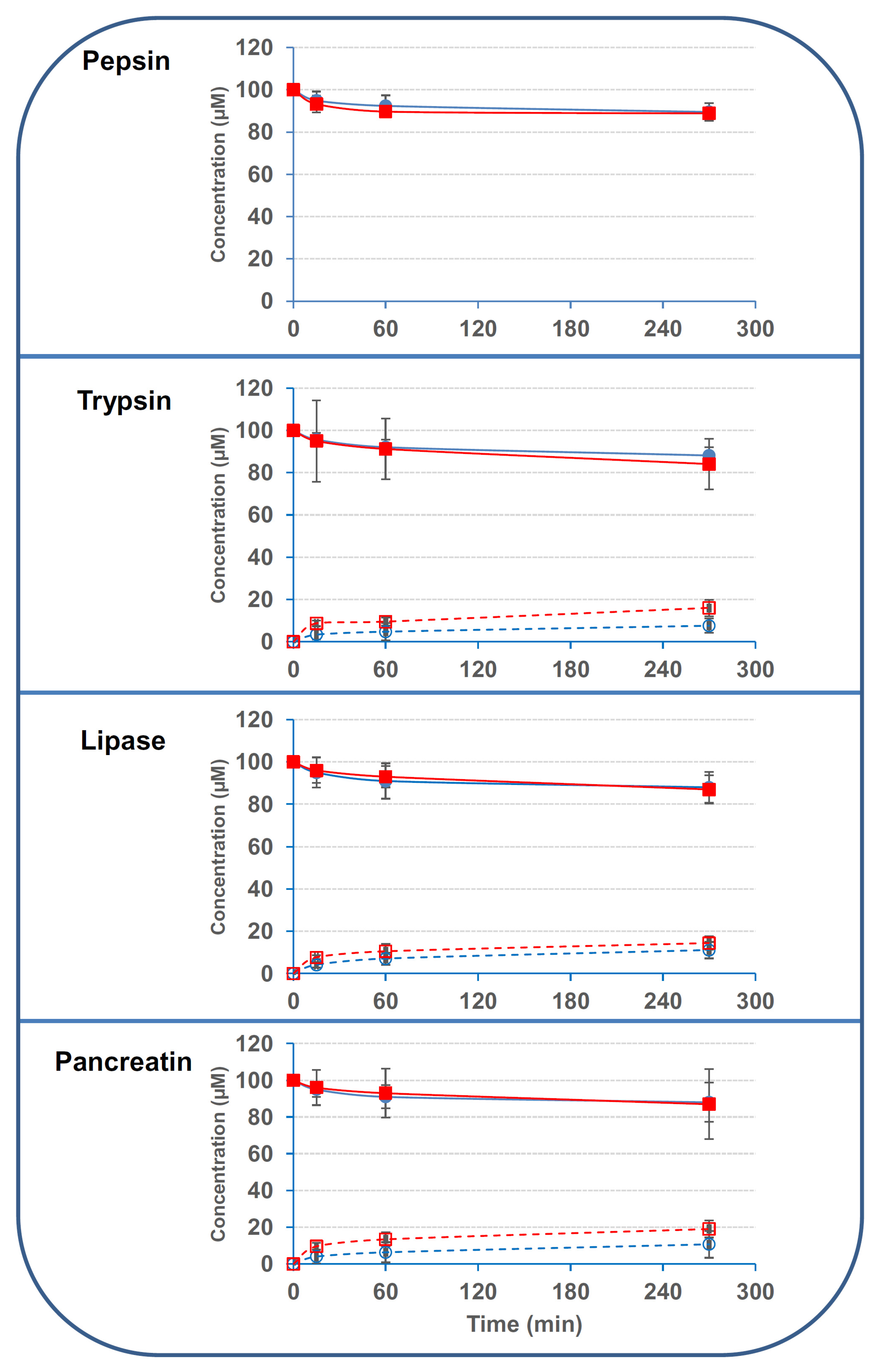
2.1. Luminal Digestive Enzymes and Fecal Bacteria Possess Limited Activity on 3/15ADON
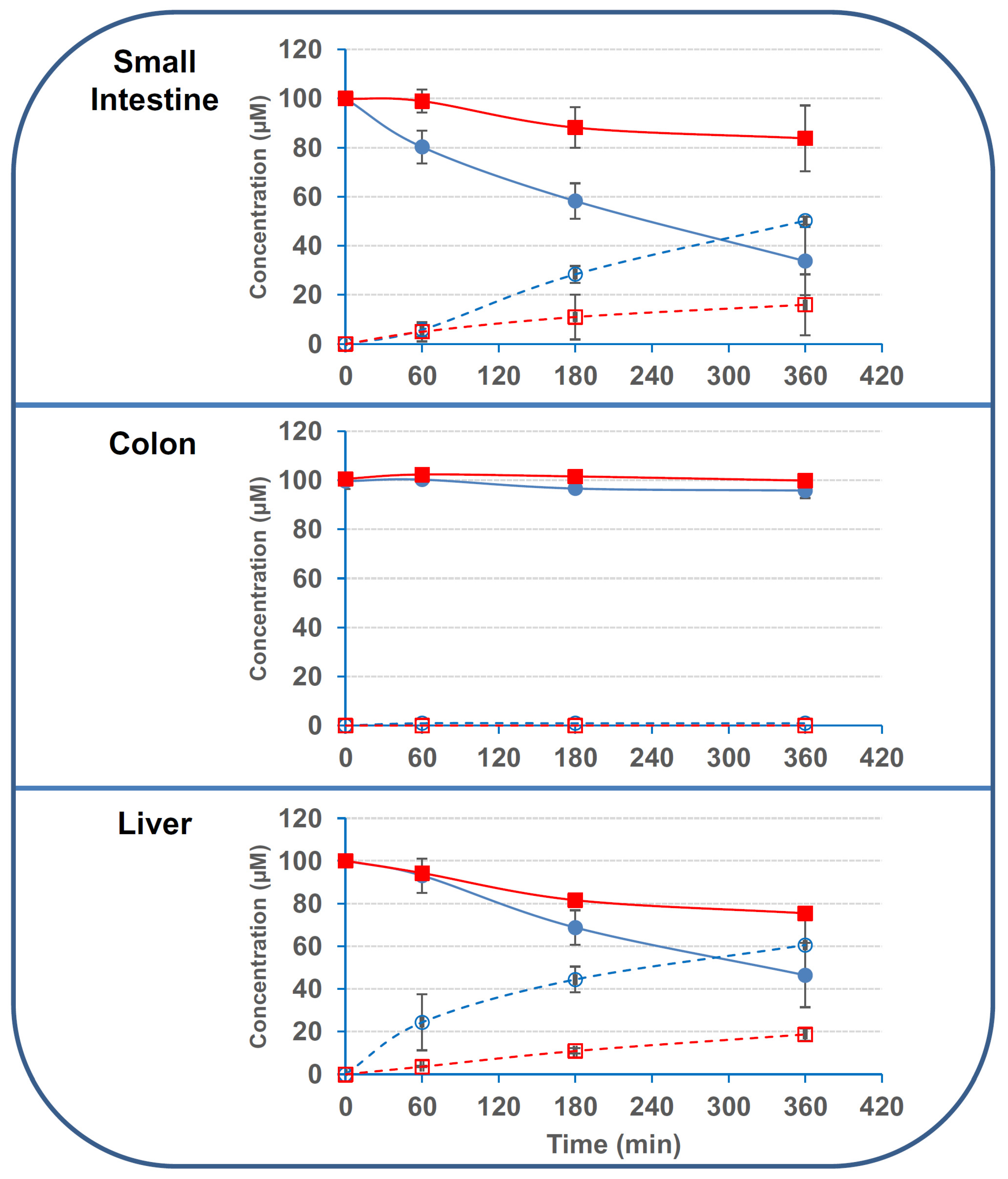
2.2. Small Intestinal and Hepatic Human Cells Display Stronger Deacetylation Activity on 3/15ADON Than Colonic or Renal Cells
2.3. The Deacetylation Activity Is Present in the S9 Fractions But Is Not Due to CES1 or CES2
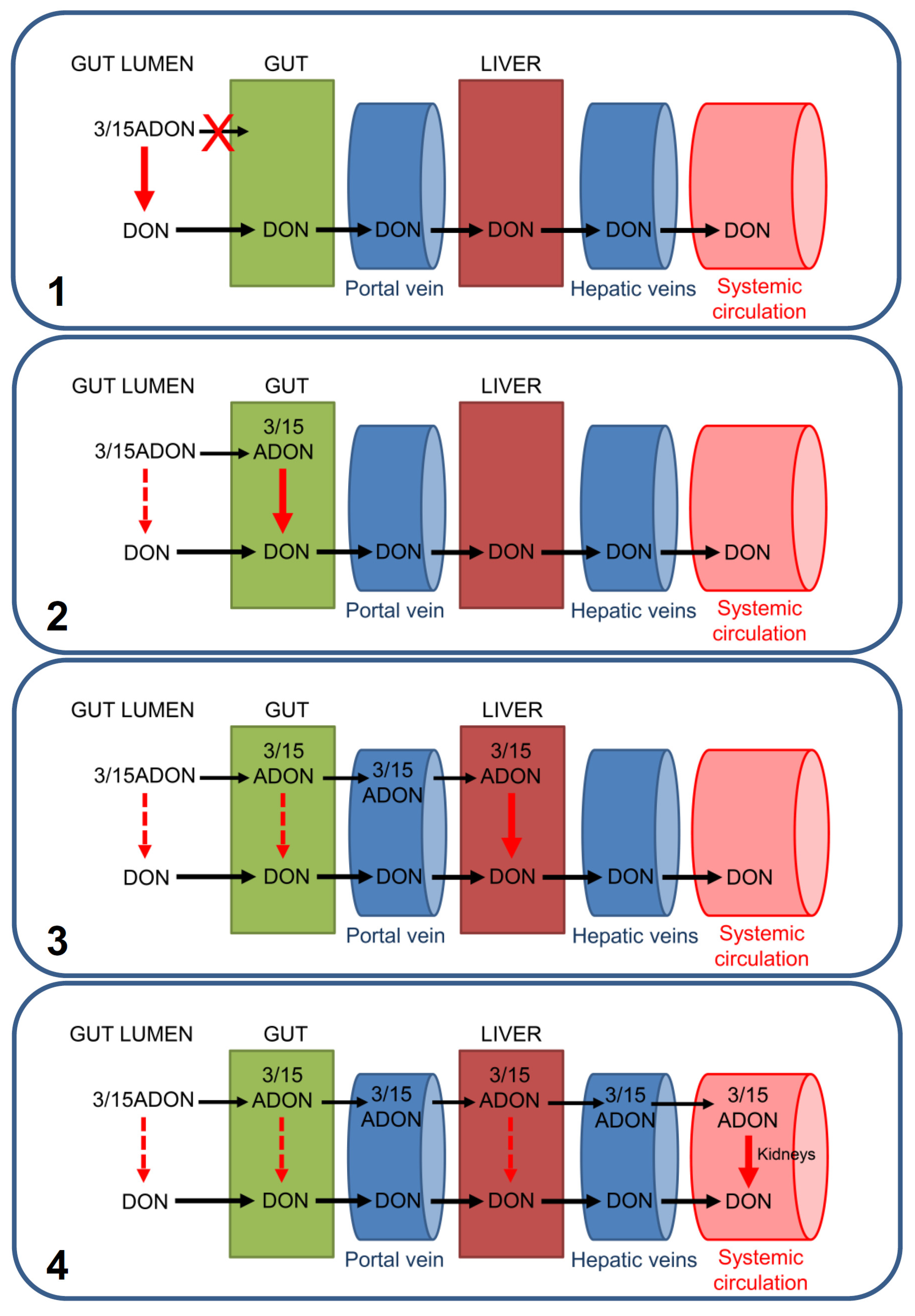
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Preparation of Human Intestinal and Hepatic Explants
4.4. Preparation of Human Fecal Bacteria
4.5. Deacetylation Assay
4.6. HPLC Analysis
4.7. Measurement of Carboxylesterases (CES) Activity
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Thrane, U.; Samson, R.A.; Pitt, J.I. Important mycotoxins and the fungi which produce them. Adv. Exp. Med. Biol. 2006, 571, 3–31. [Google Scholar] [PubMed]
- Stoev, S.D. Food safety and increasing hazard of mycotoxin occurrence in foods and feeds. Crit. Rev. Food Sci. Nutr. 2013, 53, 887–901. [Google Scholar] [CrossRef] [PubMed]
- Stoev, S.D. Foodborne mycotoxicoses, risk assessment and underestimated hazard of masked mycotoxins and joint mycotoxin effects or interaction. Environ. Toxicol. Pharmacol. 2015, 39, 794–809. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Groopman, J.D.; Pestka, J.J. Public health impacts of foodborne mycotoxins. Annu. Rev. Food Sci. Technol. 2014, 5, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, J.L. Mixtures in the real world: The importance of plant self-defense toxicants, mycotoxins, and the human diet. Toxicol. Appl. Pharmacol. 2007, 223, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M.; Fantini, J. Some food-associated mycotoxins as potential risk factors in humans predisposed to chronic intestinal inflammatory diseases. Toxicon 2010, 56, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef] [PubMed]
- Arunachalam, C.; Doohan, F.M. Trichothecene toxicity in eukaryotes: Cellular and molecular mechanisms in plants and animals. Toxicol. Lett. 2013, 217, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Awad, W.; Ghareeb, K.; Böhm, J.; Zentek, J. The toxicological impacts of the Fusarium mycotoxin, deoxynivalenol, in poultry flocks with special reference to immunotoxicity. Toxins 2013, 5, 912–925. [Google Scholar] [CrossRef] [PubMed]
- Ghareeb, K.; Awad, W.A.; Böhm, J.; Zebeli, Q. Impacts of the feed contaminant deoxynivalenol on the intestine of monogastric animals: Poultry and swine. J. Appl. Toxicol. 2015, 35, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Graziani, F.; Pujol, A.; Nicoletti, C.; Pinton, P.; Armand, L.; Di Pasquale, E.; Oswald, I.P.; Perrier, J.; Maresca, M. The food-associated ribotoxin deoxynivalenol modulates inducible NO synthase in human intestinal cell model. Toxicol. Sci. 2015, 145, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Oswald, I.P. Effect of deoxynivalenol and other Type B trichothecenes on the intestine: A review. Toxins 2014, 6, 1615–1643. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Graziani, F.; Pujol, A.; Nicoletti, C.; Paris, O.; Ernouf, P.; Di Pasquale, E.; Perrier, J.; Oswald, I.P.; Maresca, M. Deoxynivalenol inhibits the expression by goblet cells of intestinal mucins through a PKR and MAP kinase dependent repression of the resistin-like molecule β. Mol. Nutr. Food Res. 2015, 59, 1076–1087. [Google Scholar] [CrossRef] [PubMed]
- Razafimanjato, H.; Benzaria, A.; Taïeb, N.; Guo, X.J.; Vidal, N.; Di Scala, C.; Varini, K.; Maresca, M. The ribotoxin deoxynivalenol affects the viability and functions of glial cells. Glia 2011, 59, 1672–1683. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boevre, M.D.; Graniczkowska, K.; Saeger, S.D. Metabolism of modified mycotoxins studied through in vitro and in vivo models: An overview. Toxicol. Lett. 2015, 233, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Broekaert, N.; Devreese, M.; De Baere, S.; De Backer, P.; Croubels, S. Modified Fusarium mycotoxins unmasked: From occurrence in cereals to animal and human excretion. Food Chem. Toxicol. 2015, 80, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, T.; Morooka, N. Biological modification of trichothecene mycotoxins: Acetylation and deacetylation of deoxynivalenols by Fusarium spp. Appl. Microbiol. 1975, 29, 54–58. [Google Scholar] [PubMed]
- Broekaert, N.; Devreese, M.; De Mil, T.; Fraeyman, S.; Antonissen, G.; De Baere, S.; De Backer, P.; Vermeulen, A.; Croubels, S. Oral bioavailability, hydrolysis, and comparative toxicokinetics of 3-Acetyldeoxynivalenol and 15-Acetyldeoxynivalenol in broiler chickens and pigs. J. Agric. Food Chem. 2015, 63, 8734–8742. [Google Scholar] [CrossRef] [PubMed]
- De Boevre, M.; Jacxsens, L.; Lachat, C.; Eeckhout, M.; Di Mavungu, J.D.; Audenaert, K.; Maene, P.; Haesaert, G.; Kolsteren, P.; De Meulenaer, B.; et al. Human exposure to mycotoxins and their masked forms through cereal-based foods in Belgium. Toxicol. Lett. 2013, 218, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Final Report SCOOP Task 3.2.10: Collection of Occurrence Data of Fusarium Toxins in Food and Assessment of Dietary Intake by the Population of EU Member States. Available online: http://ec.europa.eu/food/fs/scoop/task3210.pdf (accessed on 15 February 2016).
- Broekaert, N.; Devreese, M.; De Mil, T.; Fraeyman, S.; De Baere, S.; De Saeger, S.; De Backer, P.; Croubels, S. Development and validation of an LC-MS/MS method for the toxicokinetic study of deoxynivalenol and its acetylated derivatives in chicken and pig plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 971, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Kadota, T.; Furusawa, H.; Hirano, S.; Tajima, O.; Kamata, Y.; Sugita-Konishi, Y. Comparative study of deoxynivalenol, 3-acetyldeoxynivalenol, and 15-acetyldeoxynivalenol on intestinal transport and IL-8 secretion in the human cell line Caco-2. Toxicol. In Vitro 2013, 27, 1888–1895. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, G.S.; Pettersson, H.; Lindberg, J.E. Absorption, metabolism and excretion of 3-acetyl DON in pigs. Arch. Anim. Nutr. 2003, 57, 335–345. [Google Scholar] [CrossRef]
- Awad, W.A.; Hess, M.; Twarużek, M.; Grajewski, J.; Kosicki, R.; Böhm, J.; Zentek, J. The impact of the Fusarium mycotoxin deoxynivalenol on the health and performance of broiler chickens. Int. J. Mol. Sci. 2011, 12, 7996–8012. [Google Scholar] [CrossRef] [PubMed]
- Veršilovskis, A.; Huybrecht, B.; Tangni, E.K.; Pussemier, L.; De Saeger, S.; Callebaut, A. Cross-reactivity of some commercially available deoxynivalenol (DON) and zearalenone (ZEN) immunoaffinity columns to DON- and ZEN-conjugated forms and metabolites. Food Addit. Contam. A 2011, 28, 1687–1693. [Google Scholar] [CrossRef] [PubMed]
- Hänninen, O.; Lindström-Seppä, P.; Pelkonen, K. Role of gut in xenobiotic metabolism. Arch. Toxicol. 1987, 60, 34–36. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, M.J.; Tsurkan, L.; Garrett, M.; Shaver, T.M.; Hyatt, J.L.; Edwards, C.C.; Hicks, L.D.; Potter, P.M. Organ-specific carboxylesterase profiling identifies the small intestine and kidney as major contributors of activation of the anticancer prodrug CPT-11. Biochem. Pharmacol. 2011, 81, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, M.J.; Potter, P.M. Carboxylesterase inhibitors. Expert Opin. Ther. Pat. 2011, 21, 1159–1171. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, G.S.; Pettersson, H. Lack of de-epoxidation of type B trichothecenes in incubates with human faeces. Food Addit. Contam. 2003, 20, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Juan-García, A.; Juan, C.; König, S.; Ruiz, M.J. Cytotoxic effects and degradation products of three mycotoxins: Alternariol, 3-acetyl-deoxynivalenol and 15-acetyl-deoxynivalenol in liver hepatocellular carcinoma cells. Toxicol. Lett. 2015, 235, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Schmeitzl, C.; Warth, B.; Fruhmann, P.; Michlmayr, H.; Malachová, A.; Berthiller, F.; Schuhmacher, R.; Krska, R.; Adam, G. The metabolic fate of deoxynivalenol and its acetylated derivatives in a wheat suspension culture: Identification and detection of DON-15-O-Glucoside, 15-Acetyl-DON-3-O-Glucoside and 15-Acetyl-DON-3-Sulfate. Toxins 2015, 7, 3112–3126. [Google Scholar] [CrossRef] [PubMed]
- Schmeitzl, C.; Varga, E.; Warth, B.; Kugler, K.G.; Malachová, A.; Michlmayr, H.; Wiesenberger, G.; Mayer, K.F.; Mewes, H.W.; Krska, R.; et al. Identification and characterization of carboxylesterases from brachypodium distachyon deacetylating trichothecene mycotoxins. Toxins 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Sanghani, S.P.; Sanghani, P.C.; Schiel, M.A.; Bosron, W.F. Human carboxylesterases: An update on CES1, CES2 and CES3. Protein Pept. Lett. 2009, 16, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M.; Mahfoud, R.; Garmy, N.; Fantini, J. The mycotoxin deoxynivalenol affects nutrient absorption in human intestinal epithelial cells. J. Nutr. 2002, 132, 2723–2731. [Google Scholar] [PubMed]
- Benzaria, A.; Maresca, M.; Taieb, N.; Dumay, E. Interaction of curcumin with phosphocasein micelles processed or not by dynamic high-pressure. Food Chem. 2013, 138, 2327–2337. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Razafimanjato, H.; Garmy, N.; Guo, X.J.; Varini, K.; Di Scala, C.; Di Pasquale, E.; Taïeb, N.; Maresca, M. The food-associated fungal neurotoxin ochratoxin A inhibits the absorption of glutamate by astrocytes through a decrease in cell surface expression of the excitatory amino-acid transporters GLAST and GLT-1. Neurotoxicology 2010, 31, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Varini, K.; Benzaria, A.; Taïeb, N.; Di Scala, C.; Azmi, A.; Graoudi, S.; Maresca, M. Mislocalization of the exitatory amino-acid transporters (EAATs) in human astrocytoma and non-astrocytoma cancer cells: Effect of the cell confluence. J. Biomed. Sci. 2012, 19. [Google Scholar] [CrossRef] [PubMed]



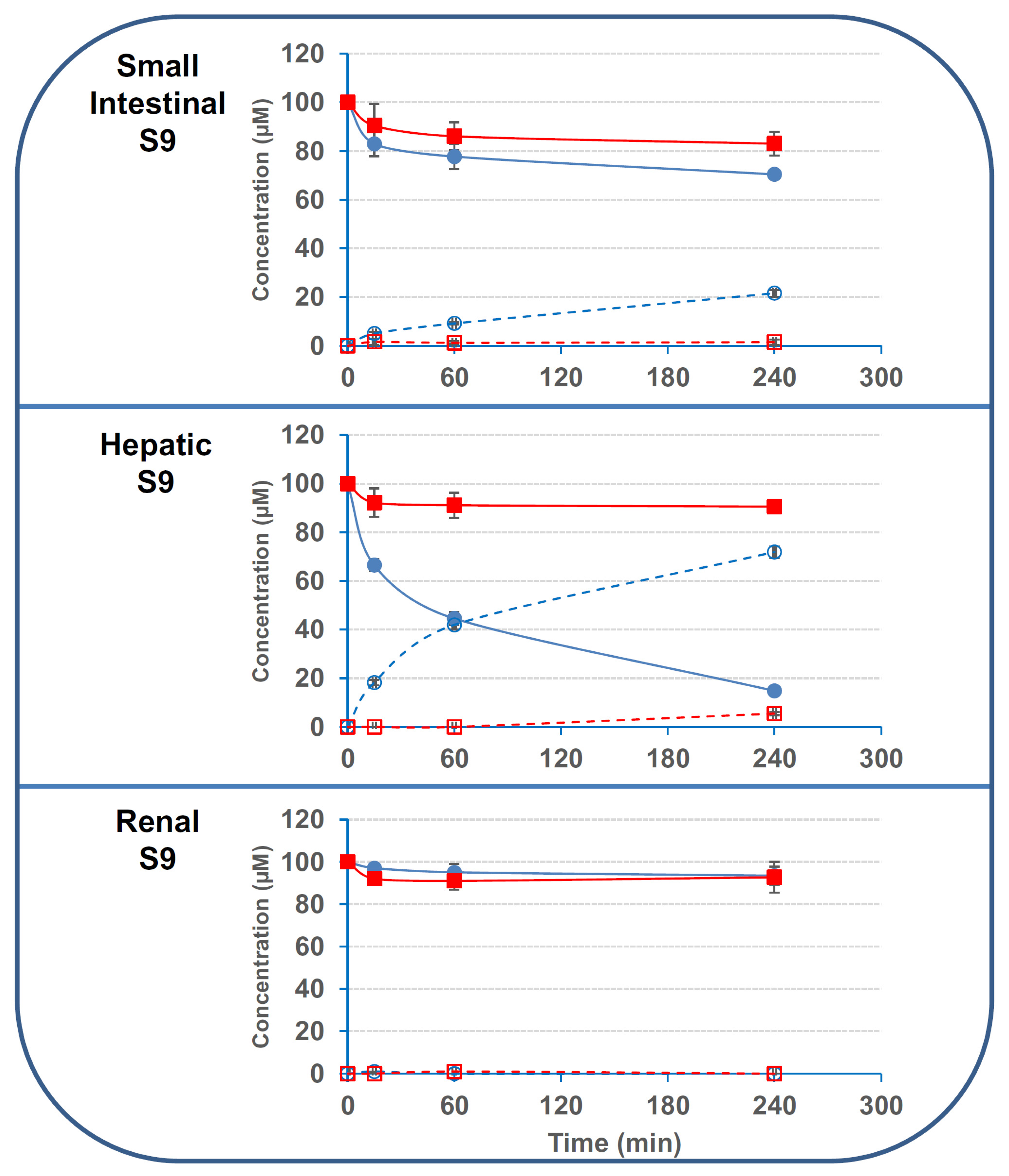
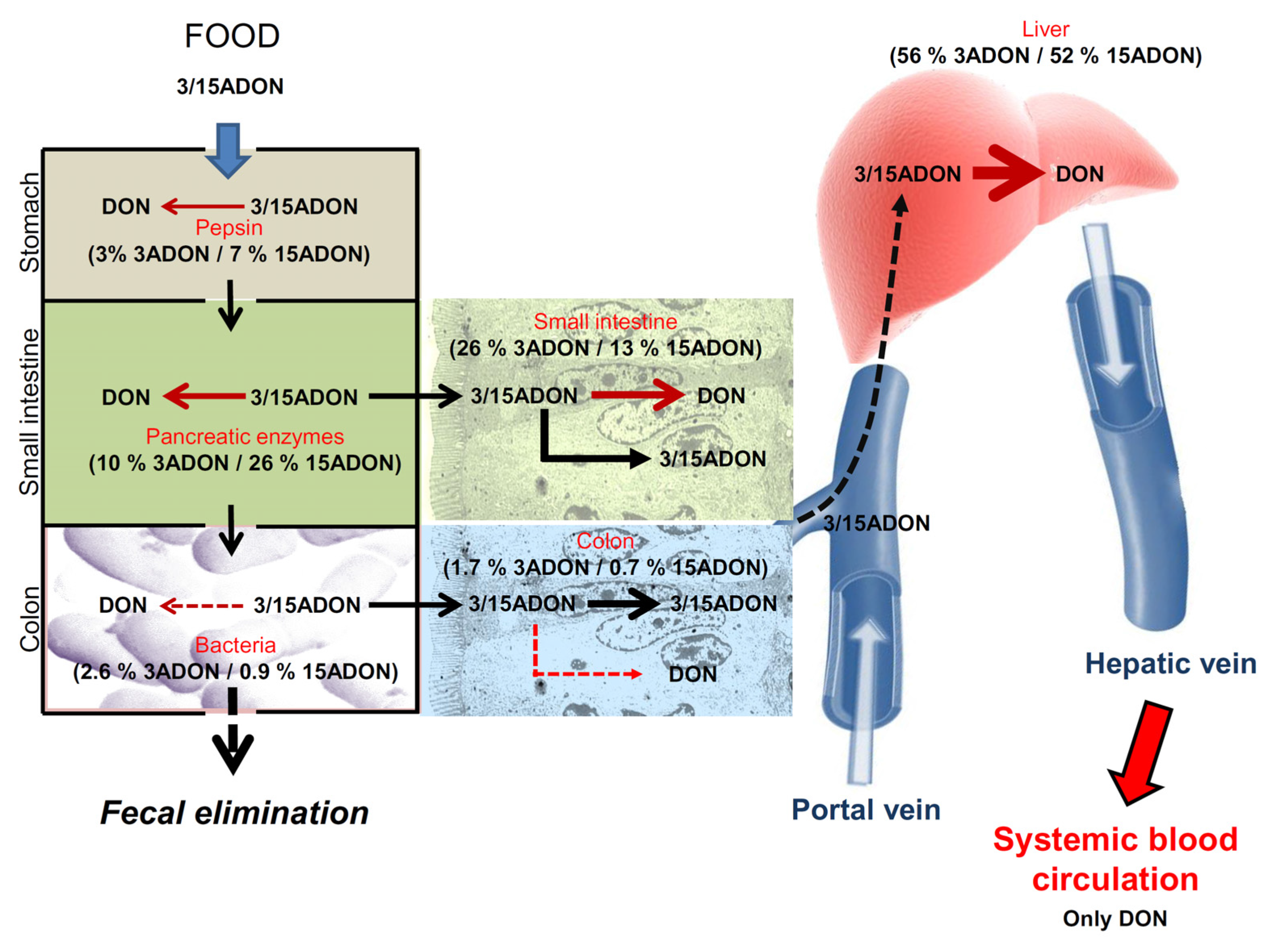
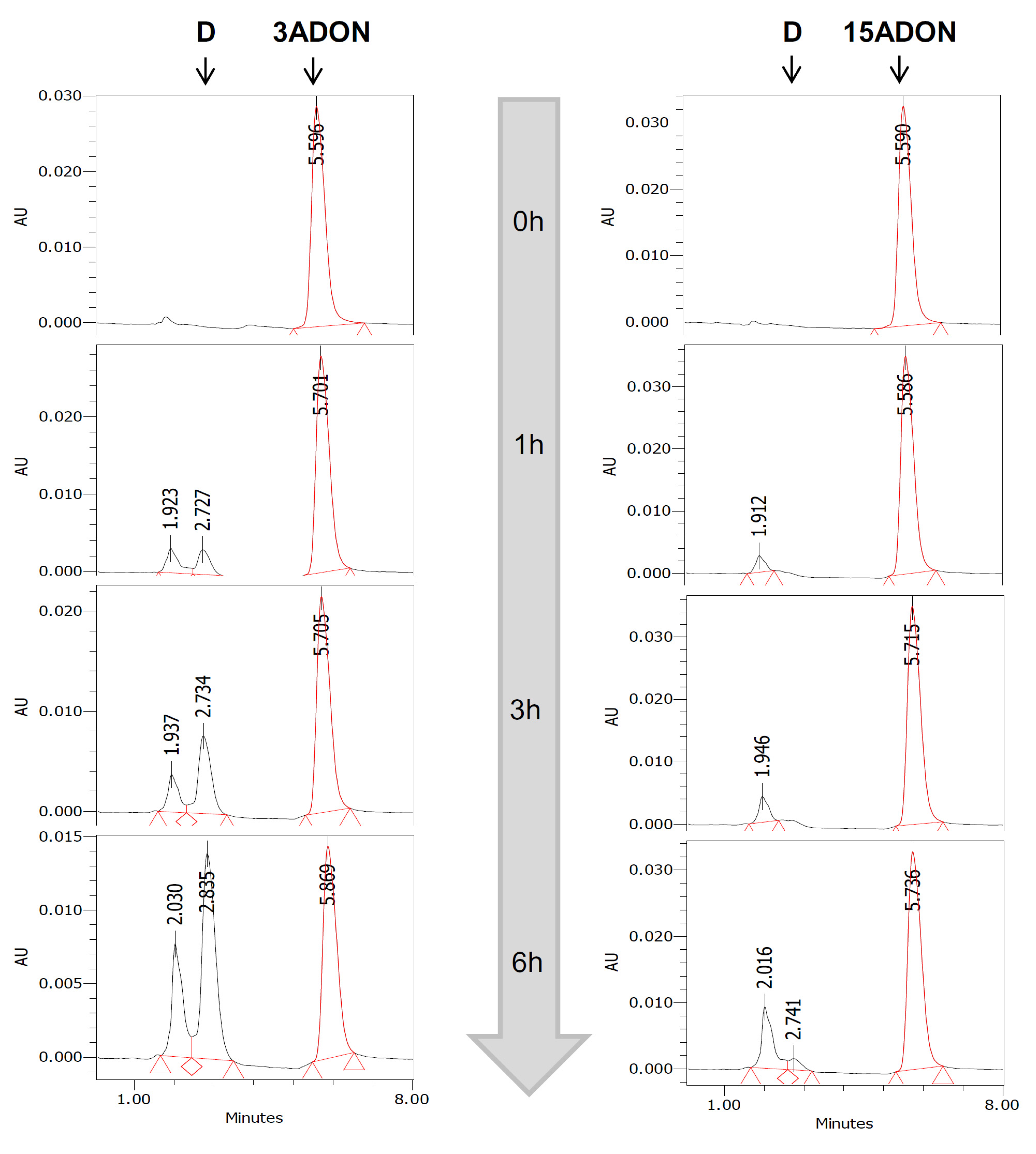
| Conditions | Disappearance of 3ADON | Disappearance of 15ADON | Formation of DON from 3ADON | Formation of DON from 15ADON |
|---|---|---|---|---|
| S9 Intestine | 1166 ± 110 | 208 ± 37 | 916 ± 57 | 250 ± 54 |
| S9 Intestine + Benzil | 875 ± 83 | 171 ± 79 | 791 ± 6.6 | 191 ± 37 |
| S9 Liver | 1729 ± 27 | 166 ± 3.5 | 1791 ± 48 | 104 ± 12 |
| S9 Liver + Benzil | 1583 ± 39 | 125 ± 15 | 1395 ± 18 | 79 ± 29 |
| Conditions | Without Benzil | With Benzil | % of Inhibition |
|---|---|---|---|
| Caco-2 | 6.2 ± 1.8 | 1.1 ± 0.1 | 83 ± 16 |
| T84 | 6.6 ± 0.7 | 0.9 ± 0.2 | 85 ± 14 |
| HepG2 | 15.7 ± 1.1 | 2.1 ± 0.1 | 87 ± 12 |
| A498 | 6.2 ± 1.8 | 1.1 ± 0.1 | 83 ± 16 |
| S9 IntestineT84 | 55 ± 9.9 | 8.3 ± 2.7 | 84 ± 15 |
| S9 Liver | 142 ± 4.6 | 21 ± 2.2 | 85 ± 14 |
| S9 Kidney | 69 ± 7.6 | 8.1 ± 1.4 | 88 ± 11 |
| CES1 | 4965 ± 197 | 167 ± 10 | 96 ± 3.3 |
| CES2 | 5671 ± 207 | 214 ± 43 | 96 ± 3.7 |
| Conditions | Disappearance of 3ADON | Disappearance of 15ADON | Formation of DON from 3ADON | Formation of DON from 15ADON |
|---|---|---|---|---|
| Pepsin | 34 ± 7.5 | 40 ± 4.5 | ND | ND |
| Trypsin | 57 ± 19 | 94 ± 62 | 36 ± 17 | 99 ± 19 |
| Lipase | 48 ± 21 | 42 ± 18 | 34 ± 11 | 42 ± 8.8 |
| Pancreatin | 24 ± 16 | 26 ± 17 | 20 ± 15 | 22 ± 9.7 |
| Fecal bacteria | 34 ± 0.9 | 5.8 ± 1.3 | 37 ± 1.6 | 14 ± 0.6 |
| Caco-2 | 479 ± 97 | 152 ± 38 | 298 ± 48 | 97 ± 27 |
| T84 | 80 ± 24 | 98 ± 29 | 72 ± 11 | 91 ± 30 |
| HepG2 | 902 ± 78 | 295 ± 41 | 920 ± 34 | 312 ± 52 |
| A498 | 79 ± 46 | 0 ± 3.1 | 63 ± 39 | 0 ± 4.7 |
| Small intestinal explant | 338 ± 69 | 85 ± 65 | 252 ± 7.5 | 80 ± 62 |
| Colonic explant | 22 ±13 | 4.5 ± 5.6 | 14 ± 9.3 | 2.1 ± 4.1 |
| Liver explant | 714 ± 198 | 330 ± 34 | 793 ± 231 | 238 ± 38 |
| S9 Small intestine | 1250 ± 79 | 166 ± 20 | 875 ± 54 | 128 ± 45 |
| S9 Liver | 1791 ± 29 | 208 ± 41 | 1479 ± 50 | 125 ± 33 |
| S9 Kidney | 33 ± 8.3 | 16 ± 5.8 | 12 ± 16 | 8.5 ± 11 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajandouz, E.H.; Berdah, S.; Moutardier, V.; Bege, T.; Birnbaum, D.J.; Perrier, J.; Di Pasquale, E.; Maresca, M. Hydrolytic Fate of 3/15-Acetyldeoxynivalenol in Humans: Specific Deacetylation by the Small Intestine and Liver Revealed Using in Vitro and ex Vivo Approaches. Toxins 2016, 8, 232. https://doi.org/10.3390/toxins8080232
Ajandouz EH, Berdah S, Moutardier V, Bege T, Birnbaum DJ, Perrier J, Di Pasquale E, Maresca M. Hydrolytic Fate of 3/15-Acetyldeoxynivalenol in Humans: Specific Deacetylation by the Small Intestine and Liver Revealed Using in Vitro and ex Vivo Approaches. Toxins. 2016; 8(8):232. https://doi.org/10.3390/toxins8080232
Chicago/Turabian StyleAjandouz, El Hassan, Stéphane Berdah, Vincent Moutardier, Thierry Bege, David Jérémie Birnbaum, Josette Perrier, Eric Di Pasquale, and Marc Maresca. 2016. "Hydrolytic Fate of 3/15-Acetyldeoxynivalenol in Humans: Specific Deacetylation by the Small Intestine and Liver Revealed Using in Vitro and ex Vivo Approaches" Toxins 8, no. 8: 232. https://doi.org/10.3390/toxins8080232
APA StyleAjandouz, E. H., Berdah, S., Moutardier, V., Bege, T., Birnbaum, D. J., Perrier, J., Di Pasquale, E., & Maresca, M. (2016). Hydrolytic Fate of 3/15-Acetyldeoxynivalenol in Humans: Specific Deacetylation by the Small Intestine and Liver Revealed Using in Vitro and ex Vivo Approaches. Toxins, 8(8), 232. https://doi.org/10.3390/toxins8080232