Effects of OnabotulintoxinA on Habituation of Laser Evoked Responses in Chronic Migraine
Abstract
:1. Introduction
- (1)
- Evaluate LEPs changes at the main sites of injection, as frontal and trapezius sites, with a control site as the hand dorsum, after seven days of single BontA treatment, according to PREEMPT protocol [1], in a double blind placebo controlled crossover design (first outcome: LEPs modification after seven days of BontA injection)
- (2)
- Correlate main LEPs findings with clinical outcome after one year of BontA treatment (second outcome: correlation between LEPs changes seven days after single BontA treatment with clinical improvement after one year (four treatment cycles) [1]).
2. Results
2.1. Aim 1
2.1.1. Laser Pain Perception
2.1.2. LEPs Latencies
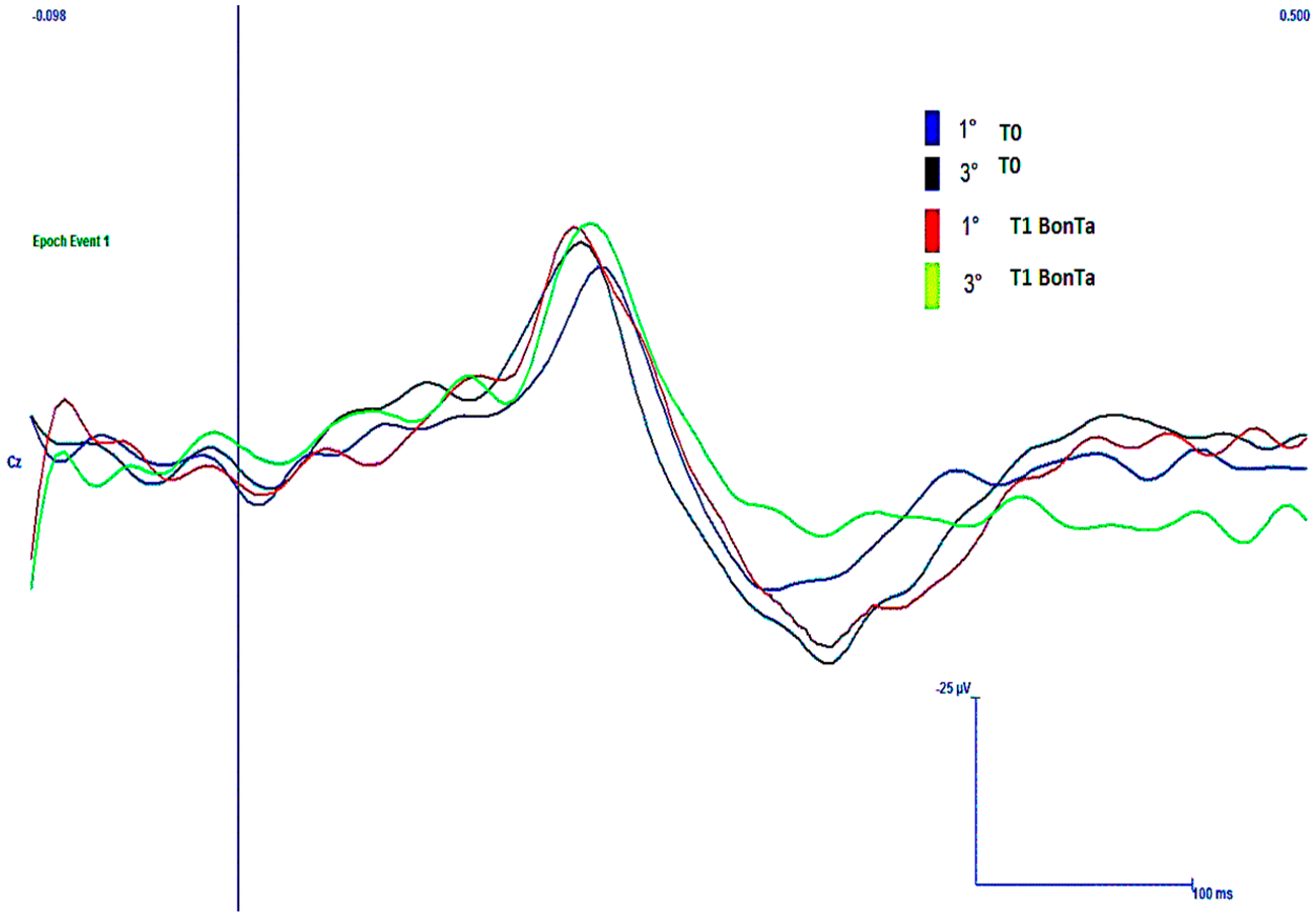
2.1.3. LEPs Amplitudes
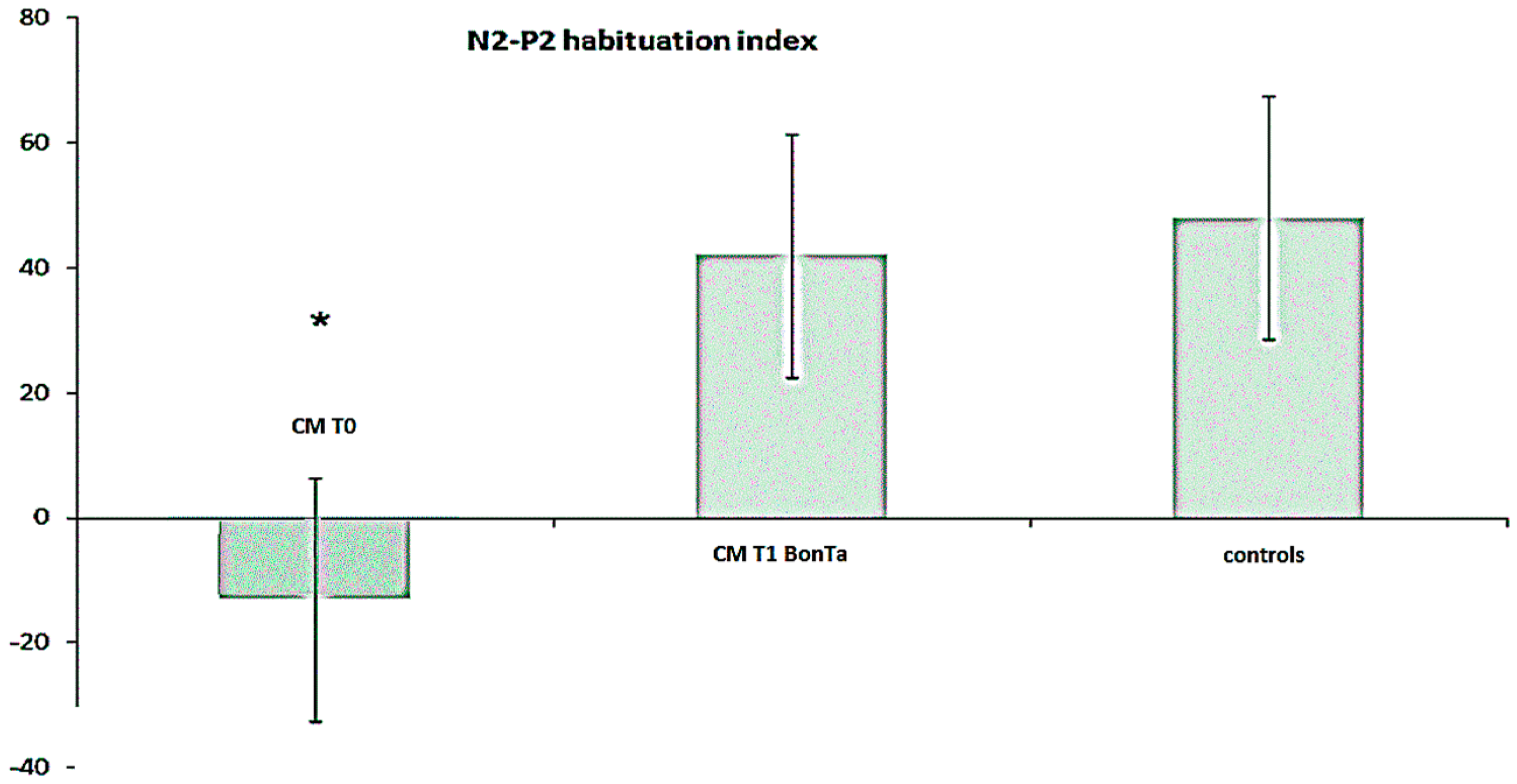
2.1.4. N2-P2 Habituation
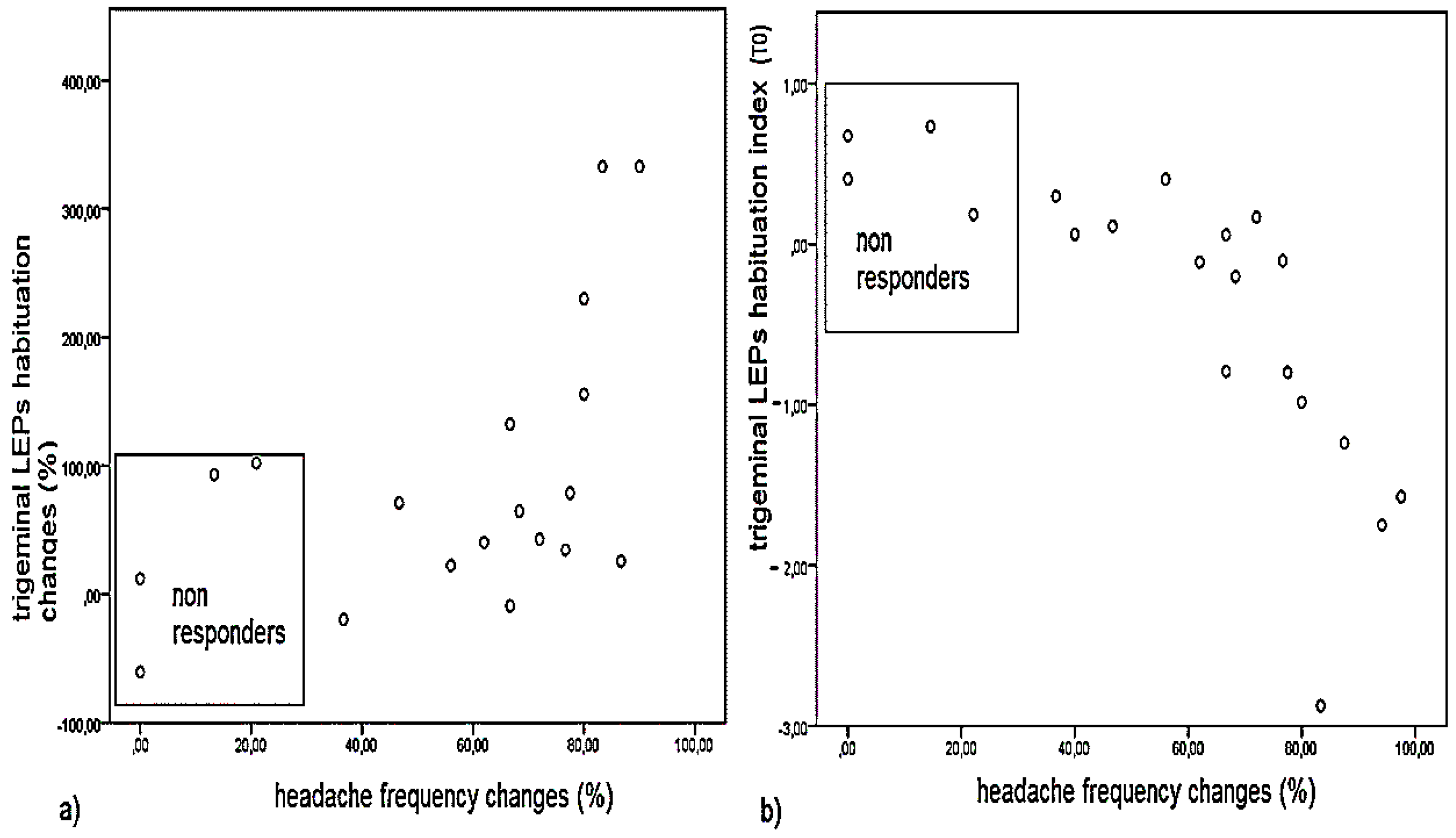
2.2. Aim 2: Clinical Outcome and Correlation with LEPs Findings
3. Discussion
3.1. Aim 1
3.1.1. Main Effects of BontA on Subjective Laser Pain and LEPs Amplitude and Latency
3.1.2. Effects of BontA on LEPs Habituation
3.2. Aim 2: Correlation between Effect of Neurotoxin on LEPs Features and Long-Term Clinical Outcome
3.3. Study Limitations
4. Conclusions
5. Materials and Methods
5.1. Patients
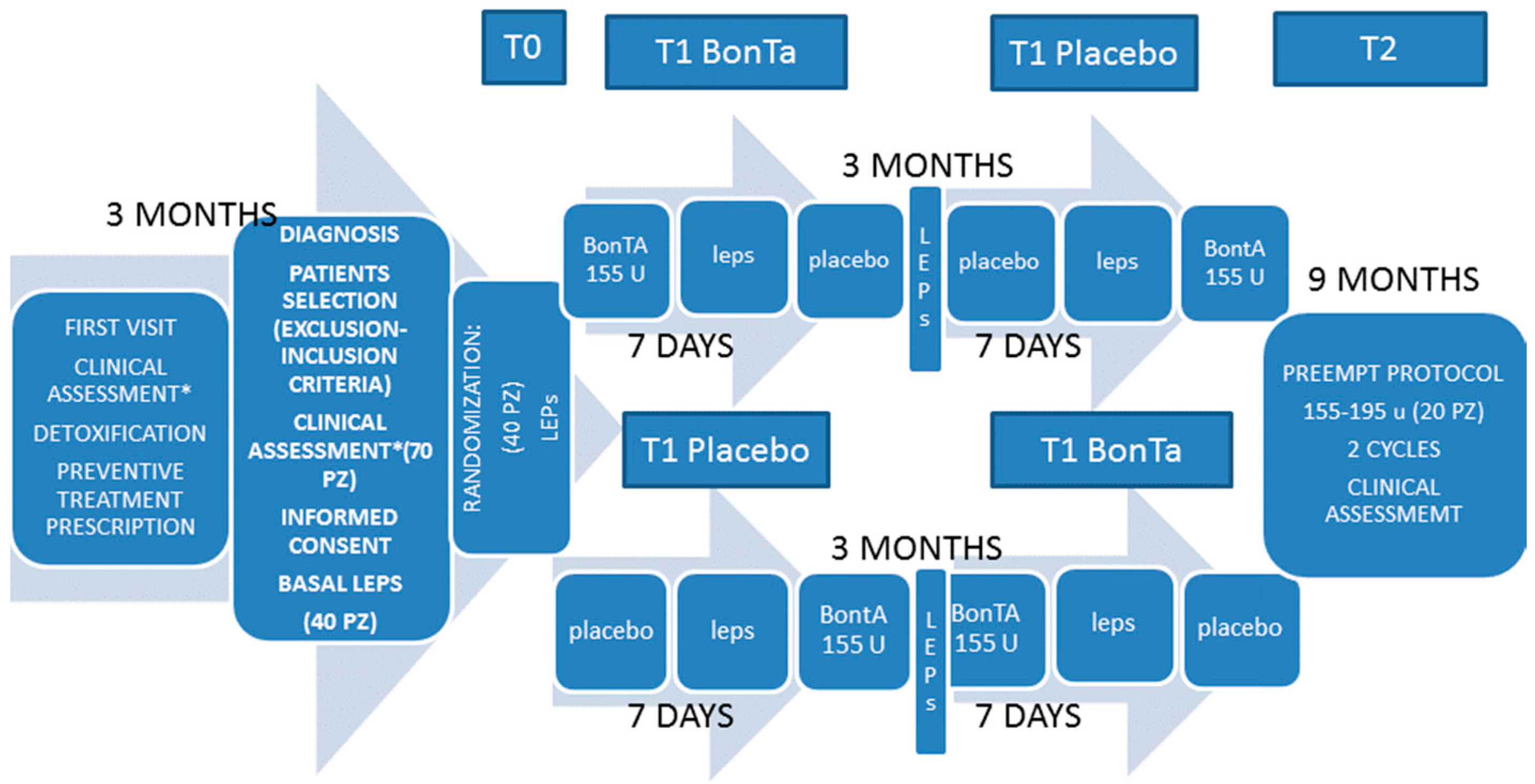
5.2. Study Design
5.3. Clinical Assessment
5.4. Aim 1
5.4.1. Laser Evoked Potentials Examination−Stimulation Procedure
5.4.2. Recording Procedure
5.4.3. Laser-Evoked Potential Analysis
5.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| LEPS | Laser Evoked Potentials |
| CM | Chronic Migraine |
| TTS | Total Tenderness Score |
| VAS | Visual Analogic Scale |
| BontA | Onabotulintoxin A |
References
- Diener, H.C.; Dodick, D.W.; Aurora, S.K.; Turkel, C.C.; DeGryse, R.E.; Lipton, R.B.; Silberstein, S.D.; Brin, M.F.; PREEMPT 2 Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia 2010, 30, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Dodick, D.W.; Turkel, C.C.; DeGryse, R.; Aurora, S.K.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F.; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache 2010, 50, 921–936. [Google Scholar] [CrossRef] [PubMed]
- Aurora, S.K.; Winner, P.; Freeman, M.C.; Spierings, E.L.; Heiring, J.O.; DeGryse, R.E.; VanDenburgh, A.M.; Nolan, M.E.; Turkel, C.C. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache 2011, 51, 1358–1373. [Google Scholar] [CrossRef] [PubMed]
- Matak, I.; Lacković, Z. Botulinum toxin A, brain and pain. Prog. Neurobiol. 2014, 119–120, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Durham, P.L.; Cady, R. Insights into the Mechanism of OnabotulinumtoxinA in Chronic Migraine. Headache 2011, 51, 1573–1577. [Google Scholar] [CrossRef] [PubMed]
- Humeau, Y.; Doussau, F.; Grant, N.J.; Poulain, B. How botulinum and tetanus neurotoxins block neurotransmitter release. Biochimie 2000, 82, 427–446. [Google Scholar] [CrossRef]
- Aoki, K.R. Evidence for antinociceptive activity of botulinum toxin type A in pain management. Headache 2003, 43, S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Dolly, O. Synaptic transmission: Inhibition of neurotransmitter release by botulinum toxins. Headache 2003, 43, S16–S24. [Google Scholar] [CrossRef] [PubMed]
- Simpson, L. The life history of a botulinum toxin molecule. Toxicon 2013, 68, 40–59. [Google Scholar] [CrossRef] [PubMed]
- Cairns, B.E.; Gazerani, P. Botulinum neurotoxin A for chronic migraine headaches: Does it work and how? Pain Manag. 2014, 4, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Cernuda-Morollón, E.; Ramón, C.; Martínez-Camblor, P.; Serrano-Pertierra, E.; Larrosa, D.; Pascual, J. OnabotulinumtoxinA decreases interictal CGRP plasma levels in patients with chronic migraine. Pain 2015, 156, 820–824. [Google Scholar] [CrossRef] [PubMed]
- Van Dongen, R.M.; Zielman, R.; Noga, M.; Dekkers, O.M.; Hankemeier, T.; van den Maagdenberg, A.M.; Terwindt, G.M.; Ferrari, M.D. Migraine biomarkers in cerebrospinal fluid: A systematic review and meta-analysis. Cephalalgia 2016. [Google Scholar] [CrossRef] [PubMed]
- Aurora, S.K.; Gawel, M.; Brandes, J.L.; Pokta, S.; Vandenburgh, A.M. Botulinum toxin type A prophylactic treatment of episodic migraine: A randomized, double-blind, placebo-controlled exploratory study. Headache 2007, 47, 486–499. [Google Scholar] [CrossRef] [PubMed]
- Hollanda, L.; Monteiro, L.; Melo, A. Botulinum toxin type a for cephalic cutaneous allodynia in chronic migraine: A randomized, double-blinded, placebo-controlled trial. Neurol. Int. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Treede, R.D.; Lorenz, J.; Baumgärtner, U. Clinical usefulness of laser-evoked potentials. Neurophysiol. Clin. 2003, 33, 303–314. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M. Laser-evoked potentials in primary headaches and cranial neuralgias. Expert Rev. Neurother. 2008, 8, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Ambrosini, A.; Brighina, F.; Coppola, G.; Perrotta, A.; Pierelli, F.; Sandrini, G.; Valeriani, M.; Marinazzo, D.; Stramaglia, S.; et al. Altered processing of sensory stimuli in patients with migraine. Nat. Rev. Neurol. 2014, 10, 144–155. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Losito, L.; Libro, G.; Guido, M.; di Fruscolo, O.; Sardaro, M.; Sciruicchio, V.; Lamberti, P.; Livrea, P. Effects of symptomatic treatments on cutaneous hyperalgesia and laser evoked potentials during migraine attack. Cephalalgia 2005, 25, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Di Clemente, L.; Puledda, F.; Biasiotta, A.; Viganò, A.; Vicenzini, E.; Truini, A.; Gruccu, G.; di Piero, V. Topiramate modulates habituation in migraine: Evidences from nociceptive responses elicited by laser evoked potentials. J. Headache Pain 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, D.; Vollono, C.; Miliucci, R.; Virdis, D.; de Armas, L.; Pazzaglia, C.; Le Pera, D.; Tarantino, S.; Balestri, M.; di Trapani, G.; et al. Habituation to pain in ‘’medication overuse headache’’: A CO2 laser-evoked potential study. Headache 2012, 52, 792–807. [Google Scholar] [CrossRef] [PubMed]
- Gazerani, P.; Staahl, C.; Drewes, A.M.; Arendt-Nielsen, L. The effects of Botulinum Toxin type A on capsaicin-evoked pain, flare, and secondary hyperalgesia in an experimental human model of trigeminal sensitization. Pain 2006, 122, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Cumberbatch, M.J.; Hill, R.G.; Hargreaves, R.J. Rizatriptan has central antinociceptive effects against durally evoked responses. Eur. J. Pharmacol. 1997, 328, 37–40. [Google Scholar] [CrossRef]
- Akerman, S.; Holland, P.R.; Lasalandra, M.P.; Goadsby, P.J. Endocannabinoids in the brainstem modulate dural trigeminovascular nociceptive traffic via CB1 and “triptan” receptors: Implications in migraine. J. Neurosci. 2013, 33, 14869–14877. [Google Scholar] [CrossRef] [PubMed]
- Gazerani, P.; Au, S.; Dong, X.; Kumar, U.; Arendt-Nielsen, L.; Cairns, B.E. Botulinum neurotoxin type A (BontA) decreases the mechanical sensitivity of nociceptors and inhibits neurogenic vasodilation in a craniofacial muscle targeted for migraine prophylaxis. Pain 2010, 151, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.B.; Kulas, D.; Karshenas, A.; Cairns, B.E.; Bach, F.W.; Arendt-Nielsen, L.; Gazerani, P. Time course analysis of the effects of botulinum neurotoxin type A on pain and vasomotor responses evoked by glutamate injection into human temporalis muscles. Toxins 2014, 6, 592–607. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S. Botulinum toxin for the treatment of chronic migraine: The placebo effect. Headache 2011, 51, 980–984. [Google Scholar] [CrossRef] [PubMed]
- Colloca, L.; Tinazzi, M.; Recchia, S.; Le Pera, D.; Fiaschi, A.; Benedetti, F.; Valeriani, M. Learning potentiates neurophysiological and behavioral placebo analgesic responses. Pain 2008, 139, 306–314. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Brighina, F.; Fierro, B.; Francesco, V.D.; Santostasi, R.; Sciruicchio, V.; Vecchio, E.; Serpino, C.; Lamberti, P.; Livrea, P. Effects of high-frequency repetitive transcranial magnetic stimulation of primary motor cortex on laser-evoked potentials in migraine. J. Headache Pain 2010, 11, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larrea, L.; Frot, M.; Valeriani, M. Brain generators of laser-evoked potentials: From dipoles to functional significance. Neurophysiol. Clin. 2003, 33, 279–292. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Guido, M.; Libro, G.; Losito, L.; Sciruicchio, V.; Monetti, C.; Puca, F. Abnormal brain processing of cutaneous pain in migraine patients during the attack. Neurosci. Lett. 2002, 333, 29–32. [Google Scholar] [CrossRef]
- Valeriani, M.; de Tommaso, M.; Restuccia, D.; Le Pera, D.; Guido, M.; Iannetti, G.D.; Libro, G.; Truini, A.; Di Trapani, G.; Puca, F.; et al. Reduced habituation to experimental pain in migraine patients: A CO2 laser evoked potential study. Pain 2003, 105, 57–64. [Google Scholar] [CrossRef]
- De Tommaso, M.; Lo Sito, L.; di Fruscolo, O.; Sardaro, M.; Pia Prudenzano, M.; Lamberti, P.; Livrea, P. Lack of habituation of nociceptive evoked responses and pain sensitivity during migraine attack. Clin. Neurophysiol. 2005, 116, 1254–1264. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; di Lorenzo, C.; Schoenen, J.; Pierelli, F. Habituation and sensitization in primary headaches. J. Headache Pain. 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Cooke, L.; Eliasziw, M.; Becker, W.J. Cutaneous allodynia in transformed migraine patients. Headache 2007, 47, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Burstein, R.; Jakubowski, M.; Garcia-Nicas, E.; Kainz, V.; Bajwa, Z.; Hargreaves, R.; Becerra, L.; Borsook, D. Thalamic sensitization transforms localized pain into widespread allodynia. Ann. Neurol. 2010, 68, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Truini, A.; Panuccio, G.; Galeotti, F.; Maluccio, M.R.; Sartucci, F.; Avoli, M.; Cruccu, G. Laser-evoked potentials as a tool for assessing the efficacy of antinociceptive drugs. Eur. J. Pain 2010, 14, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Negro, A.; Curto, M.; Lionetto, L.; Crialesi, D.; Martelletti, P. OnabotulinumtoxinA 155 U in medication overuse headache: A two years prospective study. Springerplus 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808. [Google Scholar]
- Ashkenazi, A.; Silberstein, S.; Jakubowski, M.; Burstein, R. Improved identification of allodynic migraine patients using a questionnaire. Cephalalgia 2007, 27, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Sarchielli, P.; Granella, F.; Prudenzano, M.P.; Pini, L.A.; Guidetti, V.; Bono, G.; Pinessi, L.; Alessandri, M.; Antonaci, F.; Fanciullacci, M.; et al. Italian guidelines for primary headaches: 2012 revised version. J. Headache Pain 2012, 13, S31–S70. [Google Scholar] [CrossRef] [PubMed]
- Martelletti, P.; Katsarava, Z.; Lampl, C.; Magis, D.; Bendtsen, L.; Negro, A.; Russell, M.B.; Mitsikostas, D.D.; Jensen, R.H. Refractory chronic migraine: A consensus statement on clinical definition from the European Headache Federation. J. Headache Pain 2014, 28. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, D.; Mosconi, P.; Genco, S.; Usai, S.; Prudenzano, A.M.; Grazzi, L.; Leone, M.; Puca, F.M.; Bussone, G. The Migraine Disability Assessment (MIDAS) questionnaire: Translation and reliability of the Italian version. Cephalalgia 2001, 21, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Langermark, M.; Olesen, J. Pericranial tenderness in tension headache. A blind, controlled study. Cephalalgia 1987, 7, 249–255. [Google Scholar] [CrossRef]
- De Tommaso, M.; Sardaro, M.; Serpino, C.; Vecchio, E.; Franco, G.; Sardaro, M.; Delussi, M.; Livrea, P. Fibromyalgia comorbidity in primary headaches. Cephalalgia 2009, 29, 453–464. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Libro, G.; Guido, M.; Losito, L.; Lamberti, P.; Livrea, P. Habituation of single CO2 laser-evoked responses during interictal phase of migraine. J. Headache Pain 2005, 6, 195–198. [Google Scholar] [CrossRef] [PubMed]
- De Tommaso, M.; Sciruicchio, V.; Ricci, K.; Montemurno, A.; Gentile, F.; Vecchio, E.; Barbaro, M.G.; Simeoni, M.; Goffredo, M.; Livrea, P. Laser-evoked potential habituation and central sensitization symptoms in childhood migraine. Cephalalgia 2016, 36, 463–473. [Google Scholar] [CrossRef] [PubMed]
| Site | T0 | T1 BontA | T1 Placebo |
|---|---|---|---|
| Hand | 47.43 | 44.61 | 48.39 |
| 17.13 | 20.53 | 19.31 | |
| ANOVA | - | - | - |
| F 0.66 | - | - | - |
| P 0.53 | - | - | - |
| Face | 54.78 | 50.22 | 55.08 |
| 21.84 | 17.99 | 19.53 | |
| ANOVA | - | - | - |
| F 1.76 | - | - | - |
| P 0.2 | - | - | - |
| Trapezius | 54.45 | 51.41 | 55.32 |
| 16.04 | 14.55 | 19.71 | |
| ANOVA | - | - | - |
| F 0.83 | - | - | - |
| P 0.83 | - | - | - |
| Site | N1 T0 | N1 T1 BontA | N1 T1 Placebo | N2 T0 | N2 T1 BontA | N2 T1 Placebo | P2 T0 | P2 T1 BontA | P2 T1 Placebo |
|---|---|---|---|---|---|---|---|---|---|
| Hand (msec) | 159.11 | 169.54 | 159.23 | 206.1.2 | 228.45 | 200.18 | 339.34 | 362.45 | 325.29 |
| 21.8 | 26.4 | 29.23 | 27.6 | 26.5 | 25.5 | 9.41 | 10.18 | 9.9 | |
| ANOVA | F 0.83 | - | - | F 4.97 | - | - | F 3.94 | - | - |
| P 0.45 | - | - | P 0.019 | - | - | P 0.038 | - | - | |
| - | - | - | Bonferroni T1 BontA vs T0 and T1 placebo p < 0.05 | - | - | Bonferroni T1 BontA vs T0 and T1 placebo p < 0.05 | - | - | |
| Face (msec) | 135.23 | 137.43 | 138.96 | 185.34 | 187.18 | 190.38 | 304.44 | 307.14 | 309.41 |
| 32.12 | 29.9 | 32.12 | 35.5 | 15.5 | 24.7 | 29.1 | 30.2 | 45.3 | |
| ANOVA | F 0.25 | - | - | F 0.69 | - | - | F 0.1 | - | - |
| P 0.78 | - | - | P 0.37 | - | - | P 0.9 | - | - | |
| Trapezius (msec) | 155.11 | 160.23 | 156.43 | 202.34 | 203.21 | 208.37 | 335.34 | 344.23 | 337.16 |
| 22.3 | 25.7 | 15.8 | 32.12 | 37.45 | 30.17 | 32.4 | 36.2 | 41.1 | |
| ANOVA | F 0.39 | - | - | F 0.20 | - | - | F 0.33 | - | - |
| P 0.68 | - | - | P 0.82 | - | - | P 0.72 | - | - |
| Site | N1 T0 (uV) | N1 T1 BontA (uV) | N1 T1 Placebo (uV) | N2-P2 T0 (uV) | N2-P2 T1 BontA (uV) | N2-P2 T1 Placebo (uV) | HAB-n2p2 T0% | HAB-n2p2 T1 BontA% | HAB-n2p2 T1 Placebo% |
|---|---|---|---|---|---|---|---|---|---|
| Hand | 5.5 | 4.1 | 4.5 | 13.86 | 15.32 | 13.42 | 1.60 | 18.1 | 8.2 |
| 3.9 | 4 | 2.9 | 9.92 | 11.25 | 11.39 | - | - | - | |
| ANOVA | F 0.23 | - | - | F 0.32 | - | - | F 132 | - | - |
| P 0.79 | - | - | P 0.72 | - | - | P 0.29 | - | - | |
| Face | 7.26 | 8.28 | 6.91 | 16 | 22.77 | 20.13 | −13.12 | 42.1 | −2.81 |
| 5.1 | 4.2 | 4.5 | 12.21 | 18.12 | 15.13 | 6.7 | 23.3 | 4.5 | |
| ANOVA | F 0.63 | - | - | F 0.91 | - | - | F 5.58 | - | - |
| P 0.54 | - | - | P 0.43 | - | - | P 0.03 | - | - | |
| - | - | - | - | - | - | Bonferroni T1 BontA vs. T0 p < 0.002 T1 BontA vs. T1 placebo p < 0.01 | - | - | |
| Trapezius | 4.88 | 6.32 | 5.79 | 13.12 | 15.13 | 13.67 | 16.4 | 12.8 | 14.8 |
| 4.1 | 5.5 | 3.9 | 9.12 | 9.8 | 7.8 | 11.5 | 12.1 | 12.1 | |
| ANOVA | F 0.65 | - | - | F 0.23 | - | - | F 0.40 | - | - |
| P 0.53 | - | - | P 0.69 | - | - | P 0.67 | - | - |
| Chronic Migraine Patients N° 20 | Age | Sex | Preventive Concomitant Treatments | Headache Frequency | Allodynia | Total Tenderness Score | MIDAS | Headache Intensity |
|---|---|---|---|---|---|---|---|---|
| basal | 45.45 ± 11.7 | 18 f 2 m | 10 topiramate (75–100 mg/die) 6 amitriptiline 10–25 mg/die 4 sodium vaproate (500–1000/die) | 25.7 ± 3.7 | 3.65 ± 2.83 | 17.5 ± 4.94 | 79.4 ± 25.5 | 2.9 ± 0.14 |
| one year observation (BontA treatment) | - | - | - | 10.5 ± 7.3 | 2.25 ± 1.97 | 15.5 ± 8.7 | 29.55 ± 30.3 | 1.2 ± 0.5 |
| t-test | - | - | - | 7.97 | 3.39 | 1.72 | 3.85 | 7.96 |
| p | - | - | - | 0.0001 | 0.003 | 0.1 | 0.001 | 0.0001 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Tommaso, M.; Delussi, M.; Ricci, K.; Montemurno, A.; Carbone, I.; Vecchio, E. Effects of OnabotulintoxinA on Habituation of Laser Evoked Responses in Chronic Migraine. Toxins 2016, 8, 163. https://doi.org/10.3390/toxins8060163
De Tommaso M, Delussi M, Ricci K, Montemurno A, Carbone I, Vecchio E. Effects of OnabotulintoxinA on Habituation of Laser Evoked Responses in Chronic Migraine. Toxins. 2016; 8(6):163. https://doi.org/10.3390/toxins8060163
Chicago/Turabian StyleDe Tommaso, Marina, Marianna Delussi, Katia Ricci, Anna Montemurno, Irene Carbone, and Eleonora Vecchio. 2016. "Effects of OnabotulintoxinA on Habituation of Laser Evoked Responses in Chronic Migraine" Toxins 8, no. 6: 163. https://doi.org/10.3390/toxins8060163
APA StyleDe Tommaso, M., Delussi, M., Ricci, K., Montemurno, A., Carbone, I., & Vecchio, E. (2016). Effects of OnabotulintoxinA on Habituation of Laser Evoked Responses in Chronic Migraine. Toxins, 8(6), 163. https://doi.org/10.3390/toxins8060163