Toxin and Growth Responses of the Neurotoxic Dinoflagellate Vulcanodinium rugosum to Varying Temperature and Salinity
Abstract
:1. Introduction
2. Results
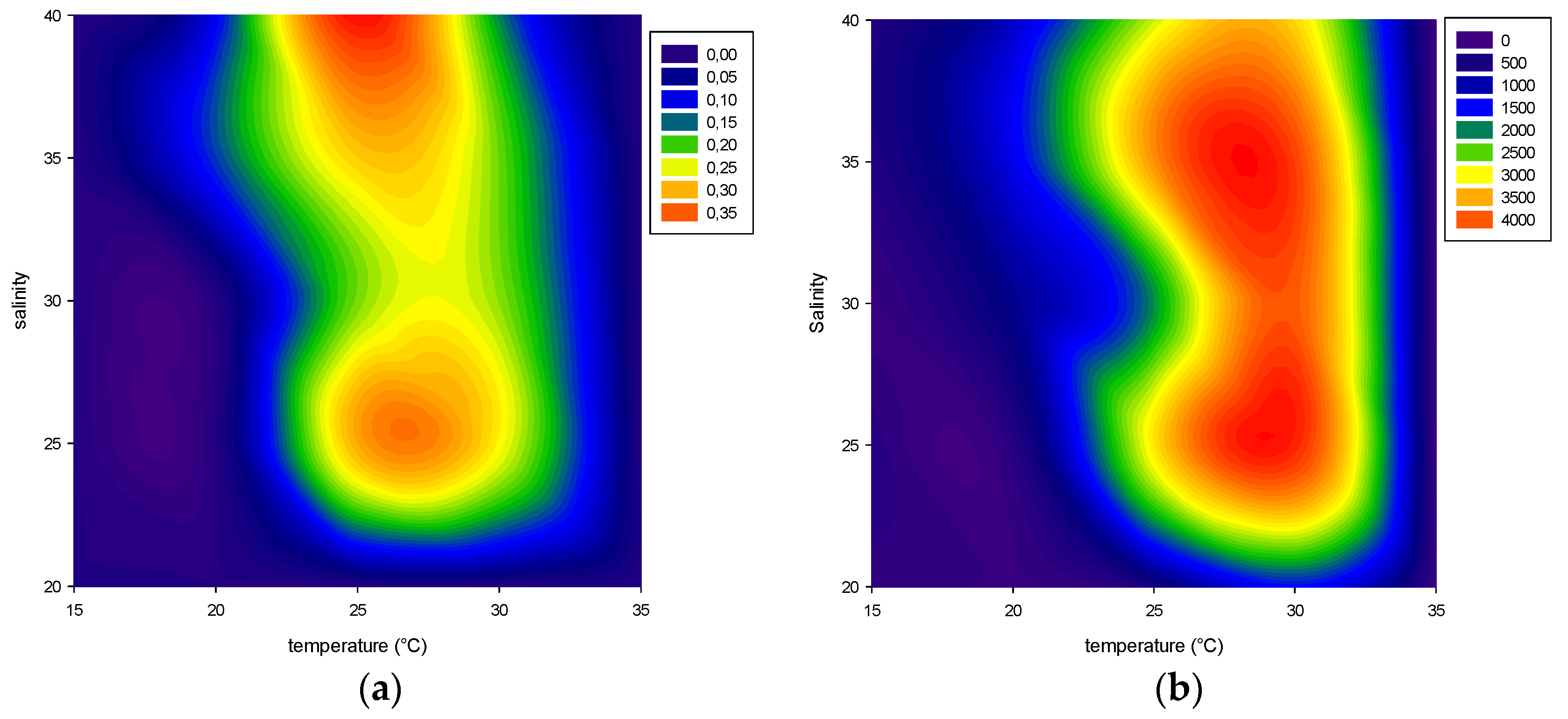
2.1. Effect of Environmental Factors on Growth
2.1.1. Temperature
2.1.2. Salinity
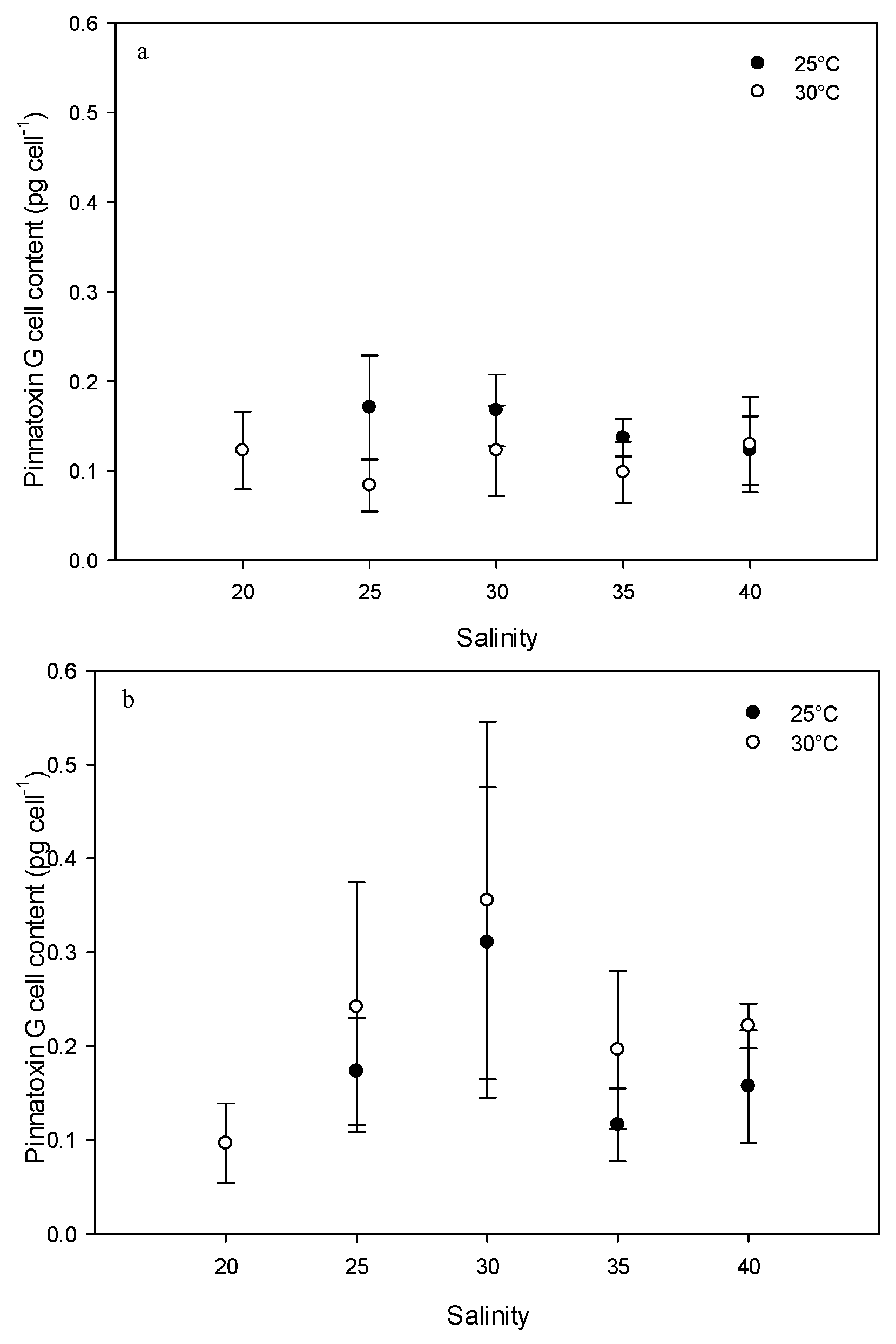
2.2. Effect of Environmental Factors on PnTX-G Production
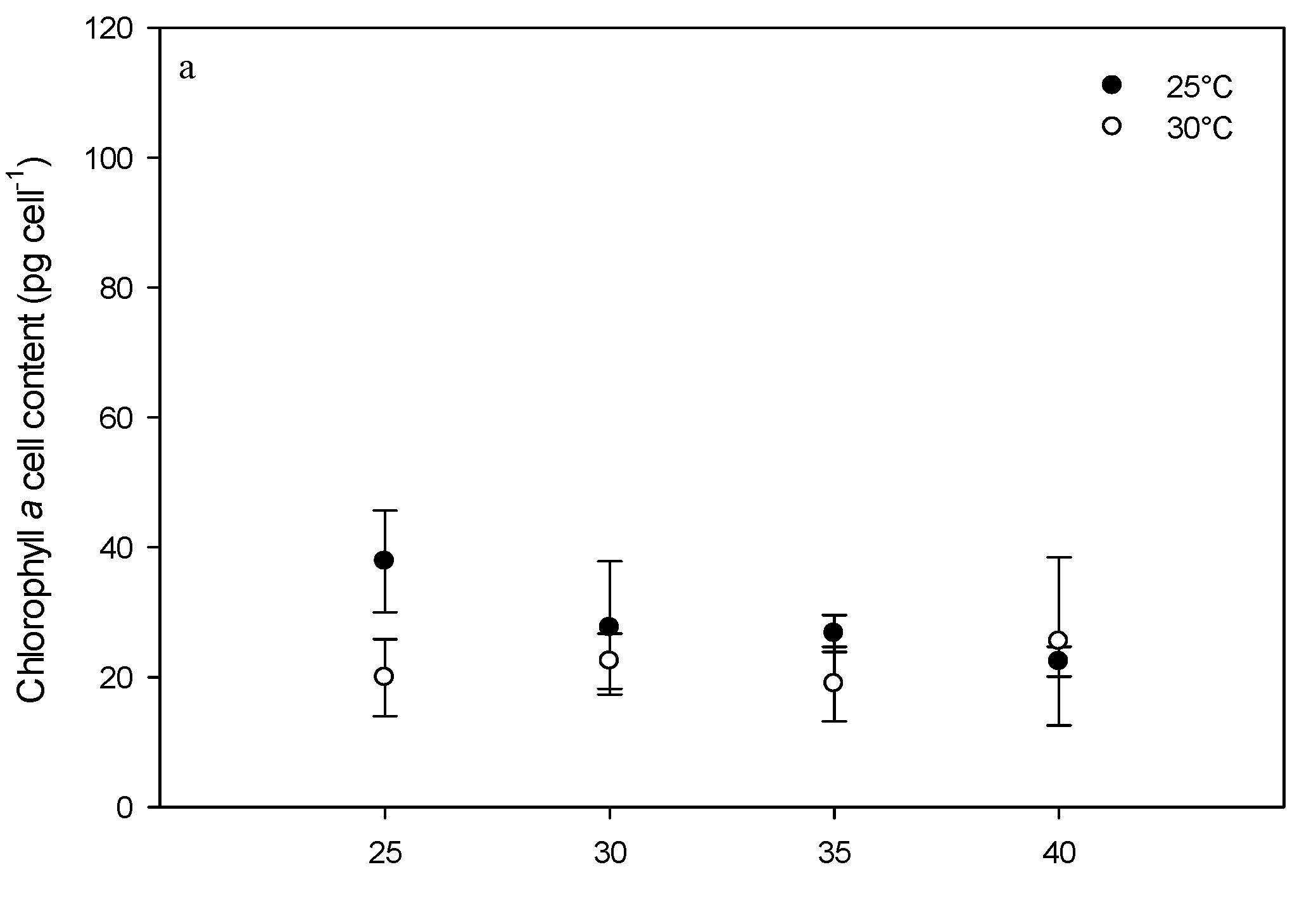
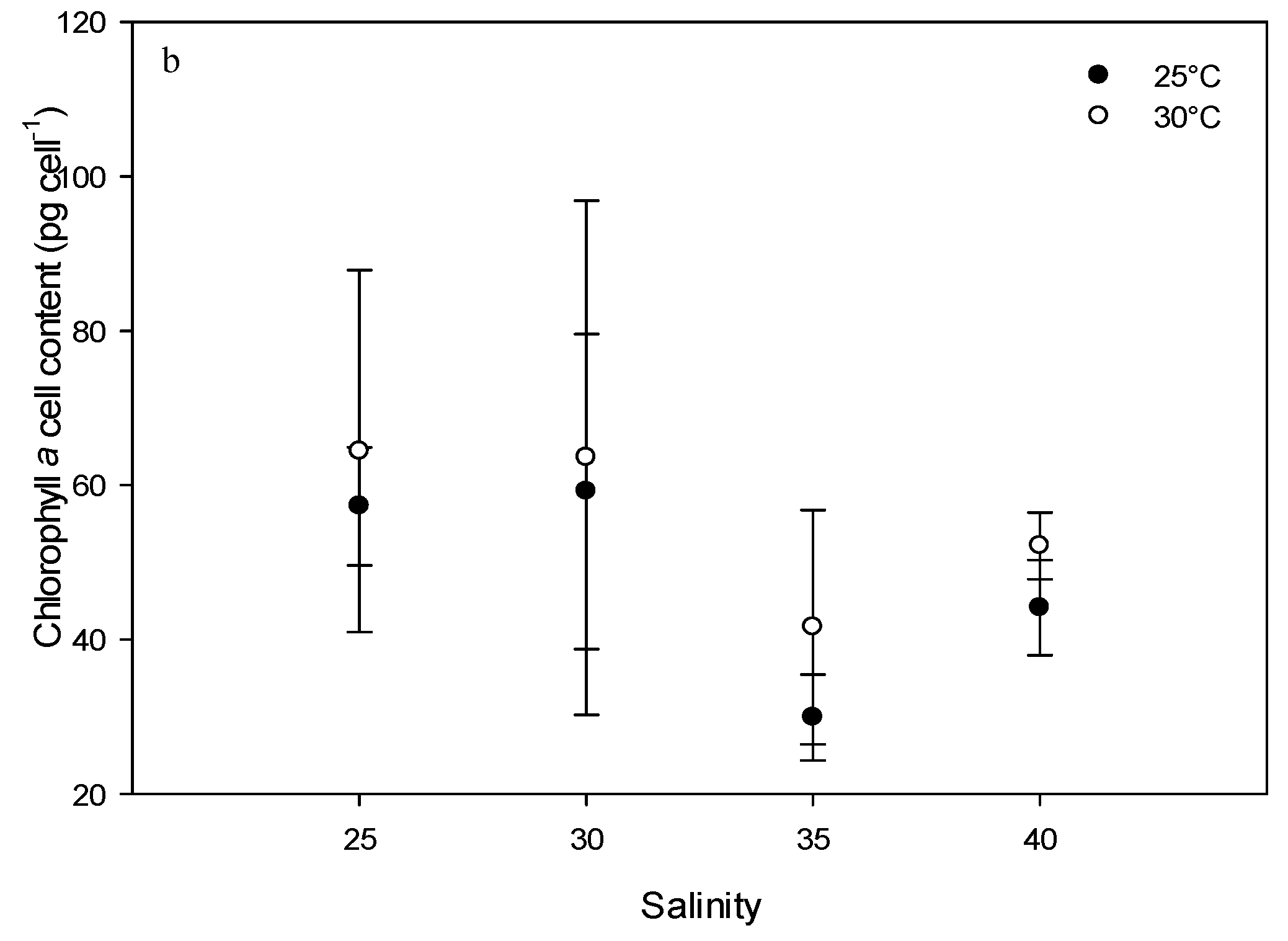
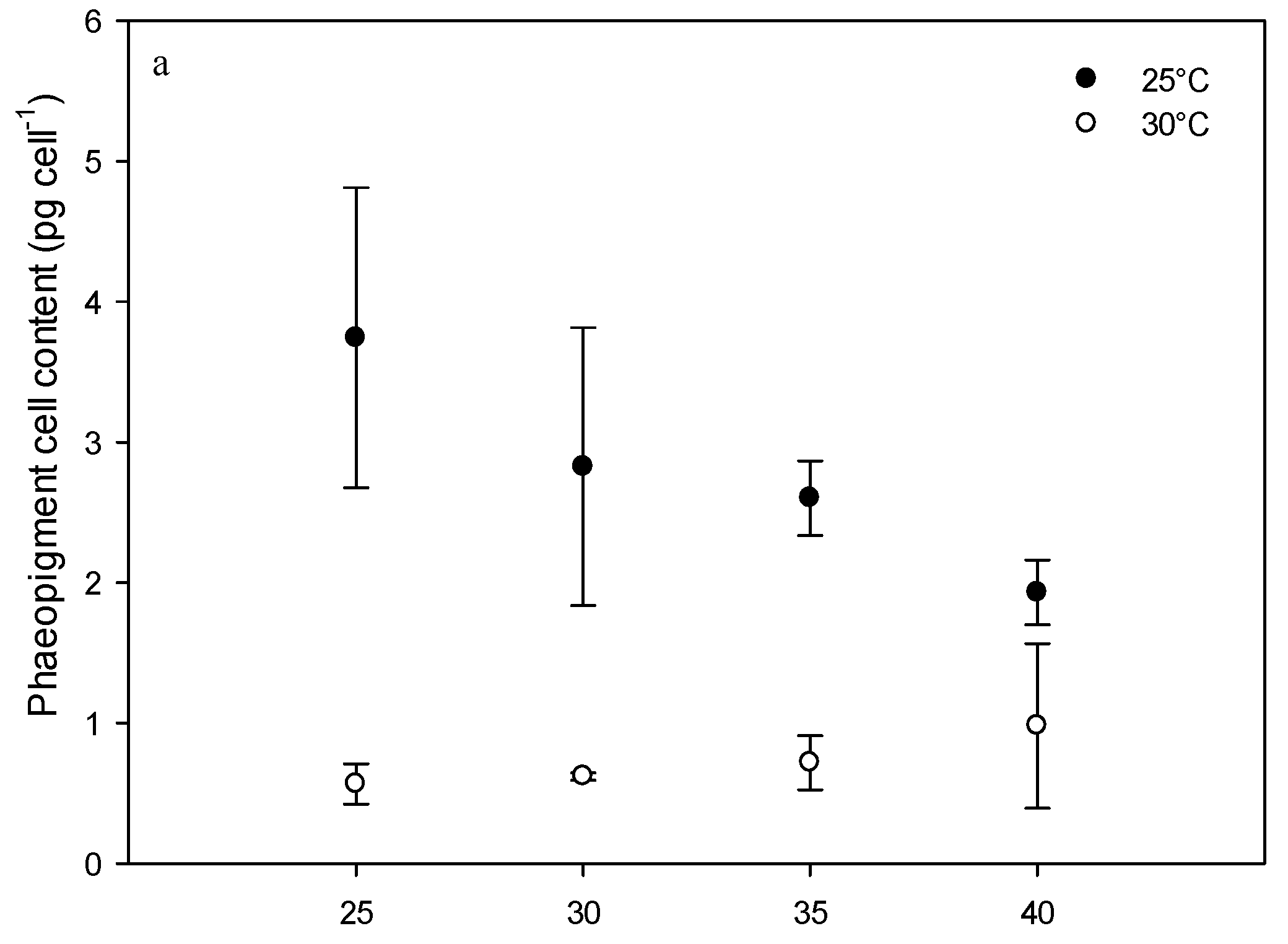
2.3. Effect of Environmental Factors on Chlorophyll Production
3. Discussion and Conclusions
3.1. Physical and Chemical Parameters Modulate Growth and Toxin Content of Vulcanodinium Rugosum
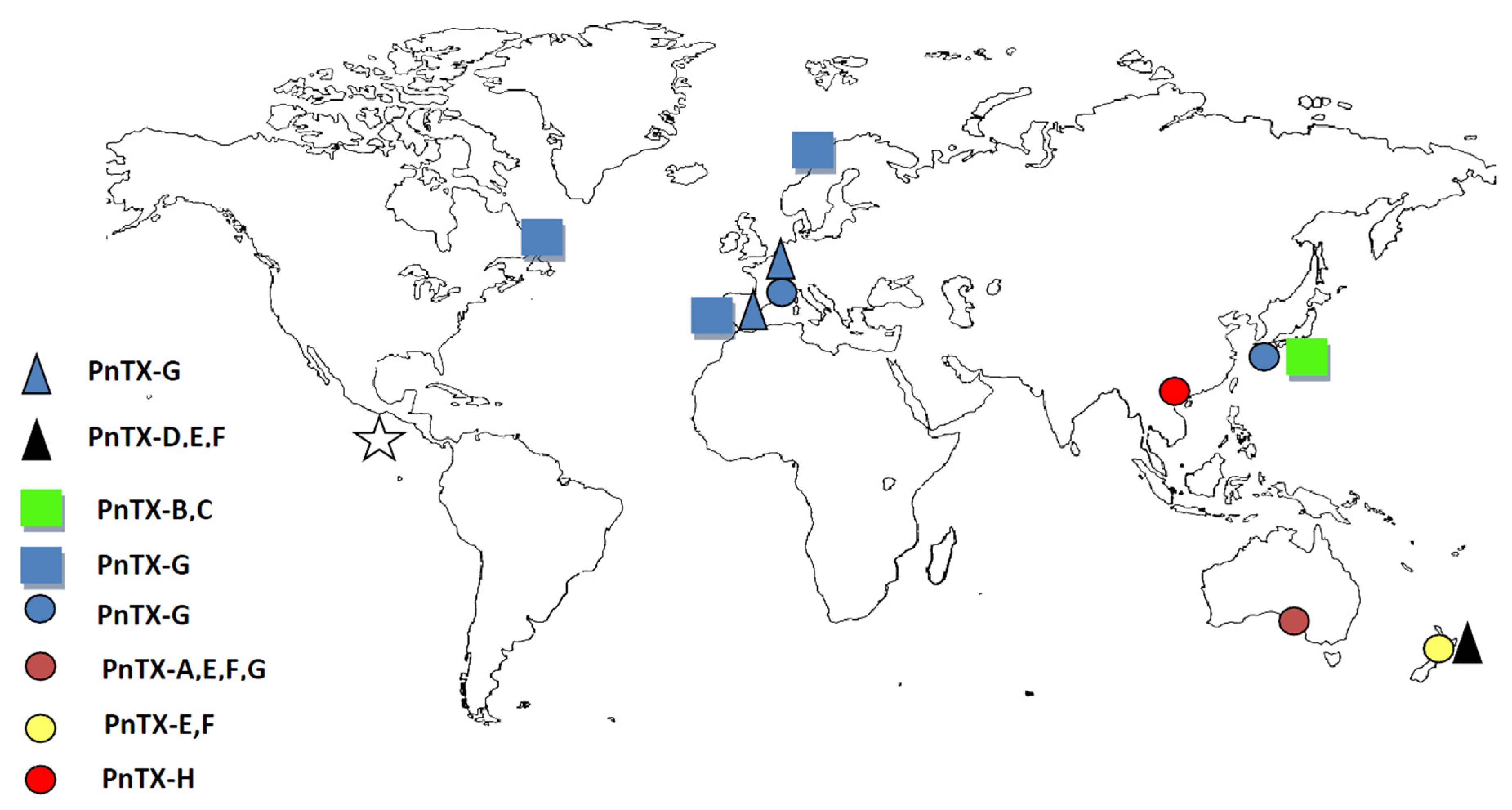
3.2. Worldwide Distribution of V. rugosum and Related Toxins
3.3. Conclusions
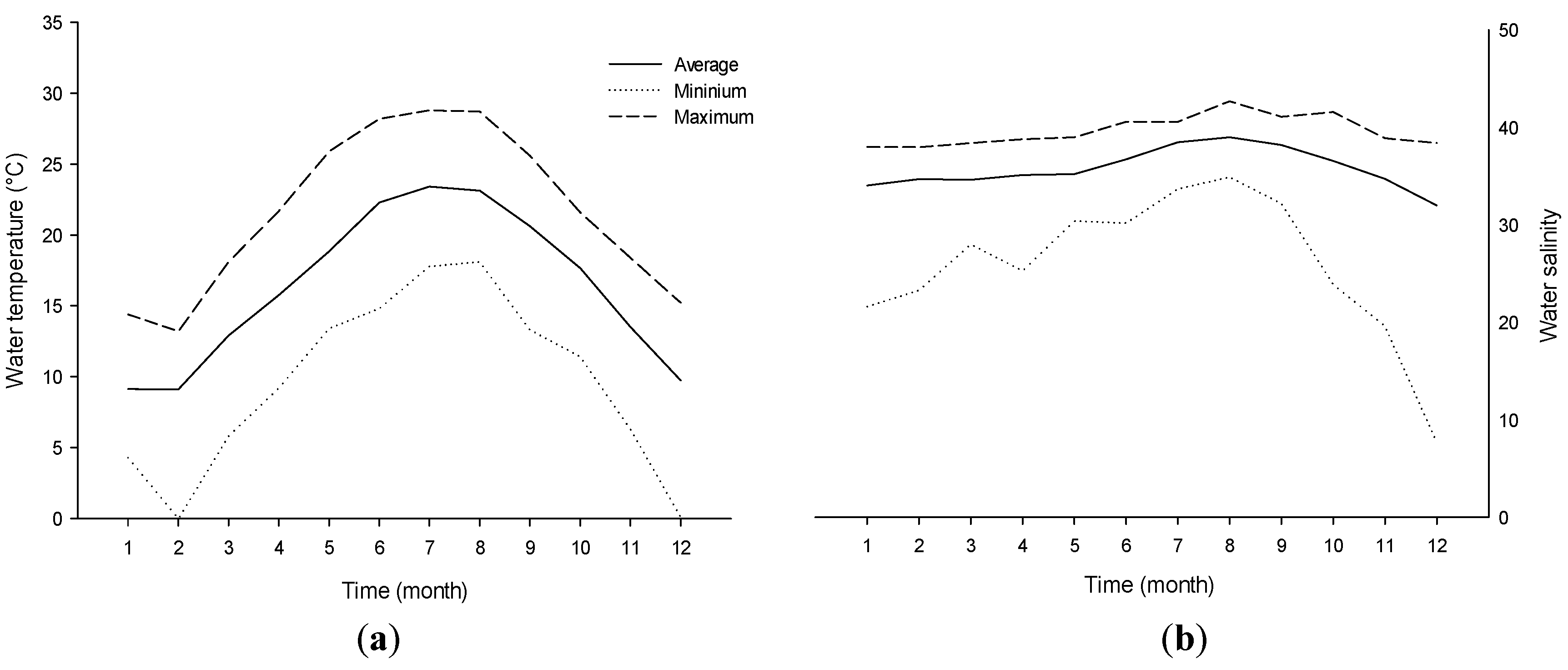
4. Experimental Section
4.1. Vulcanoidinum rugosum: Origin and Culture
4.2. Experimental Conditions
4.3. Cell Concentrations and Growth Rate Measurements
4.4. Chlorophyll a Analysis
4.5. Pinnatoxin G Measurement
4.6. Statistics
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smayda, T.J. Harmful algal blooms: Their ecophysiology and general relevance to phytoplankton blooms in the sea. Limnol. Oceanogr. 1997, 42, 1137–1153. [Google Scholar] [CrossRef]
- Laabir, M.; Jauzein, C.; Genovesi, B.; Masseret, E.; Grzebyk, D.; Cecchi, P.; Vaquer, A.; Perrin, Y.; Collos, Y. Influence of temperature, salinity and irradiance on the growth and cell yield of the harmful red tide dinoflagellate Alexandrium catenella colonizing Mediterranean waters. J. Plankton Res. 2011, 33, 1550–1563. [Google Scholar] [CrossRef]
- Abadie, E.; Amzil, Z.; Belin, C.; Comps, M.A.; Elziere-Papayanni, P.; Lassus, P.; Le Bec, C.; Marcaillou-Le Baut, C.; Nezan, E.; Poggi, R. Contamination de l’etang de Thau par Alexandrium Tamarense. Episode de Novembre a Decembre 1998; Ifremer: Plouzané, France, 1999. [Google Scholar]
- Lilly, E.L.; Halanych, K.M.; Anderson, D.M. Species boundaries and global biogeography of the Alexandrium tamarense complex (Dinophyceae). J. Phycol. 2007, 43, 1329–1338. [Google Scholar] [CrossRef]
- Genovesi, B.; Mouillot, D.; Vaquer, A.; Laabir, M.; Pastoureaud, A. Towards an optimal sampling strategy for Alexandrium catenella (Dinophyceae) benthic resting cysts. Harmful Algae 2007, 6, 837–848. [Google Scholar] [CrossRef]
- Hess, P.; Abadie, E.; Herve, F.; Berteaux, T.; Sechet, V.; Araoz, R.; Molgo, J.; Zakarian, A.; Sibat, M.; Rundberget, T.; et al. Pinnatoxin G is responsible for atypical toxicity in mussels (Mytilus galloprovincialis) and clams (Venerupis decussata) from Ingril, a French Mediterranean Lagoon. Toxicon 2013, 75, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Uemura, D.; Chou, T.; Haino, T.; Nagatsu, A.; Fukuzawa, S.; Zheng, S.Z.; Chen, H.S. Pinnatoxin-A—A toxic amphoteric macrocycle from the okinawan bivalve Pinna muricata. J. Am. Chem. Soc. 1995, 117, 1155–1156. [Google Scholar] [CrossRef]
- Chou, T.; Haino, T.; Kuramoto, M.; Uemura, D. Isolation and structure of pinnatoxin D, a new shellfish poison from the okinawan bivalve Pinna muricata. Tetrahedron Lett. 1996, 37, 4027–4030. [Google Scholar] [CrossRef]
- Smith, K.F.; Rhodes, L.L.; Suda, S.; Selwood, A.I. A dinoflagellate producer of pinnatoxin G, isolated from sub-tropical Japanese waters. Harmful Algae 2011, 10, 702–705. [Google Scholar] [CrossRef]
- Nézan, E.; Chomérat, N. Vulcanodinium rugosum gen. Nov., sp. Nov. (Dinophyceae): A new marine dinoflagellate from the French Mediterranean Coast. Cryptogam. Algologie 2011, 32, 3–18. [Google Scholar] [CrossRef]
- Rhodes, L.; Smith, K.; Selwood, A.; McNabb, P.; Munday, R.; Suda, S.; Molenaar, S.; Hallegraeff, G. Dinoflagellate Vulcanodinium rugosum identified as the causative organism of pinnatoxins in Australia, New Zealand and Japan. Phycologia 2011, 50, 624–628. [Google Scholar] [CrossRef]
- Zeng, N.; Gu, H.; Smith, K.F.; Rhodes, L.L.; Selwood, A.I.; Yang, W. The first report of Vulcanodinium rugosum (Dinophyceae) from the South China Sea with a focus on the life cycle. N. Z. J. Mar. Freshw. Res. 2012, 46, 511–521. [Google Scholar] [CrossRef]
- Matsubara, T.; Nagasoe, S.; Yamasaki, Y.; Shikata, T.; Shimasaki, Y.; Oshima, Y.; Honjo, T. Effects of temperature, salinity, and irradiance on the growth of the dinoflagellate Akashiwo sanguinea. J. Exp. Mar. Biol. Ecol. 2007, 342, 226–230. [Google Scholar] [CrossRef]
- Nagasoe, S.; Kim, D.-I.; Shimasaki, Y.; Oshima, Y.; Yamaguchi, M.; Honjo, T. Effects of temperature, salinity and irradiance on the growth of the red tide dinoflagellate Gyrodinium instriatum Freudenthal et Lee. Harmful Algae 2006, 5, 20–25. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Honjo, T. Effects of temperature, salinity and irradiance on the growth of the noxious red tide flagellate Gymnodinium nagasakiense (Dinophycae). Nippon Suisan Gakk. 1989, 55, 2029–2036. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Imai, I.; Honjo, T. Effects of temperature, salinity and irradiance on the growth-rates of the noxious red tide flagellates Chattonella antiqua and C. marina (Raphidophycae). Nippon Suisan Gakk. 1991, 57, 1277–1284. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Shigeru, I.; Nagasaki, K.; Matsuyama, Y.; Uchida, T.; Imai, I. Effects of temperature and salinity on the growth of the red tide flagellates Heterocapsa circularisquama (Dinophyceae) and Chattonella verruculosa (Raphidophyceae). J. Plankton Res. 1997, 19, 1167–1174. [Google Scholar] [CrossRef]
- Yamamoto, T.; Oh, S.J.; Kataoka, Y. Effects of temperature, salinity and irradiance on the growth of the toxic dinoflagellate Gymnodinium catenatum (Dinophyceae) isolated from Hiroshima Bay, Japan. Fish. Sci. 2002, 68, 356–363. [Google Scholar] [CrossRef]
- Anderson, D.M.; Cembella, A.D.; Hallegraeff, G.M. Progress in understanding harmful algal blooms: Paradigm shifts and new technologies for research, monitoring, and management. Annu. Rev. Mar. Sci. 2012, 4, 143–176. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.-X.; Zhang, Y.; Warner, M.E.; Feng, Y.; Sun, J.; Hutchins, D.A. A comparison of future increased CO2 and temperature effects on sympatric Heterosigma akashiwo and Prorocentrum minimum. Harmful Algae 2008, 7, 76–90. [Google Scholar] [CrossRef]
- Laabir, M.; Collos, Y.; Masseret, E.; Grzebyk, D.; Abadie, E.; Savar, V.; Sibat, M.; Amzil, Z. Influence of environmental factors on the paralytic shellfish toxin content and profile of Alexandrium catenella (Dinophyceae) isolated from the Mediterranean Sea. Mar. Drugs 2013, 11, 1583–1601. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Cembella, A.D.; Quilliam, M.A. Cell cycle and toxin production in the benthic dinoflagellate Prorocentrum lima. Mar. Biol. 1999, 134, 541–549. [Google Scholar] [CrossRef]
- Xu, N.; Duan, S.; Li, A.; Zhang, C.; Cai, Z.; Hu, Z. Effects of temperature, salinity and irradiance on the growth of the harmful dinoflagellate Prorocentrum donghaiense Lu. Harmful Algae 2010, 9, 13–17. [Google Scholar] [CrossRef]
- Genovesi, B.; Laabir, M.; Masseret, E.; Collos, Y.; Vaquer, A.; Grzebyk, D. Dormancy and germination features in resting cysts of Alexandrium tamarense species complex (Dinophyceae) can facilitate bloom formation in a shallow lagoon (Thau, Southern France). J. Plankton Res. 2009, 31, 1209–1224. [Google Scholar] [CrossRef]
- Triki, H.Z.; Daly-Yahia, O.K.; Malouche, D.; Komiha, Y.; Deidun, A.; Brahim, M.; Laabir, M. Distribution of resting cysts of the potentially toxic dinoflagellate Alexandrium pseudogonyaulax in recently-deposited sediment within Bizerte Lagoon (Mediterranean Coast, Tunisia). Mar. Pollut. Bull. 2014, 84, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.M.; Munoz, M.G.; Contreras, A.M. Temperature as a factor regulating growth and toxin content in the dinofiagellate Alexandrium catenella. Harmful Algae 2006, 5, 762–769. [Google Scholar] [CrossRef]
- Band-Schmidt, C.J.; Bustillos-Guzman, J.J.; Hernandez-Sandoval, F.E.; Nunez-Vasquez, E.J.; Lopez-Cortés, D.J. Effect of temperature on growth and paralytic toxin profiles in isolates of Gymnodinium catenatum (Dinophyceae) from the Pacific Coast of Mexico. Toxicon 2014, 90, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Giacobbe, M.G.; Oliva, F.D.; Maimone, G. Environmental factors and seasonal occurrence of the dinoflagellate Alexandrium minutum, a PSP potential producer, in a Mediterranean Lagoon. Estuar. Coast. Shelf Sci. 1996, 42, 539–549. [Google Scholar] [CrossRef]
- Grzebyk, D.; Bechemin, C.; Ward, C.J.; Verite, C.; Codd, G.A.; Maestrini, S.Y. Effects of salinity and two coastal waters on the growth and toxin content of the dinoflagellate Alexandrium minutum. J. Plankton Res. 2003, 25, 1185–1199. [Google Scholar] [CrossRef]
- Hwang, D.F.; Lu, Y.H. Influence of environmental and nutritional factors on growth, toxicity, and toxin profile of dinoflagellate Alexandrium minutum. Toxicon 2000, 38, 1491–1503. [Google Scholar] [CrossRef]
- Kim, D.-I.; Matsuyama, Y.; Nagasoe, S.; Yamaguchi, M.; Yoon, Y.-H.; Oshima, Y.; Imada, N.; Honjo, T. Effects of temperature, salinity and irradiance on the growth of the harmful red tide dinoflagellate Cochlodinium polykrikoides margalef (Dinophyceae). J. Plankton Res. 2004, 26, 61–66. [Google Scholar] [CrossRef]
- Parkhill, J.P.; Cembella, A.D. Effects of salinity, light and inorganic nitrogen on growth and toxigenicity of the marine dinoflagellate Alexandrium tamarense from Northeastern Canada. J. Plankton Res. 1999, 21, 939–955. [Google Scholar] [CrossRef]
- Wang, D.-Z.; Hsieh, D.P.H. Growth and toxin production in batch cultures of a marine dinoflagellate Alexandrium tamarense HK9301 isolated from the South China Sea. Harmful Algae 2005, 4, 401–410. [Google Scholar] [CrossRef]
- Kibler, S.R.; Litaker, R.W.; Holland, W.C.; Vandersea, M.W.; Tester, P.A. Growth of eight Gambierdiscus (Dinophyceae) species: Effects of temperature, salinity and irradiance. Harmful Algae 2012, 19, 1–14. [Google Scholar] [CrossRef]
- Yoshimatsu, T.; Yamaguchi, H.; Iwamoto, H.; Nishimura, T.; Adachi, M. Effects of temperature, salinity and their interaction on growth of Japanese Gambierdiscus spp. (Dinophyceae). Harmful Algae 2014, 35, 29–37. [Google Scholar] [CrossRef]
- Aguilera-Belmonte, A.; Inostroza, I.; Carrillo, K.S.; Franco, J.M.; Riobo, P.; Gomez, P.I. The combined effect of salinity and temperature on the growth and toxin content of four chilean strains of Alexandrium catenella (Whedon and Kofoid) balech 1985 (Dinophyceae) isolated from an outbreak occurring in Southern Chile in 2009. Harmful Algae 2013, 23, 55–59. [Google Scholar] [CrossRef]
- Etheridge, S.M.; Roesler, C.S. Effects of temperature, irradiance, and salinity on photosynthesis, growth rates, total toxicity, and toxin composition for Alexandrium fundyense isolates from the gulf of maine and bay of fundy. Deep Sea Res. Part II Top. Stud. Oceanogr. 2005, 52, 2491–2500. [Google Scholar] [CrossRef]
- Tawong, W.; Yoshimatsu, T.; Yamaguchi, H.; Adachi, M. Effects of temperature, salinity and their interaction on growth of benthic dinoflagellates Ostreopsis spp. from Thailand. Harmful Algae 2015, 44, 37–45. [Google Scholar] [CrossRef]
- Pistocchi, R.; Pezzolesi, L.; Guerrini, F.; Vanucci, S.; Dell’Aversano, C.; Fattorusso, E. A review on the effects of environmental conditions on growth and toxin production of Ostreopsis ovata. Toxicon 2011, 57, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, F.; Pezzolesi, L.; Feller, A.; Riccardi, M.; Ciminiello, P.; Dell’Aversano, C.; Tartaglione, L.; Iacovo, E.D.; Fattorusso, E.; Forino, M.; et al. Comparative growth and toxin profile of cultured Ostreopsis ovata from the Tyrrhenian and Adriatic Seas. Toxicon 2010, 55, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Morton, S.L.; Norris, D.R.; Bomber, J.W. Effect of temperature, salinity and light-intensity on the growth and seasonality of toxic dinoflagellates associeted with ciguatera. J. Exp. Mar. Biol. Ecol. 1992, 157, 79–90. [Google Scholar] [CrossRef]
- Vale, P.; Veloso, V.; Amorim, A. Toxin composition of a Prorocentrum lima strain isolated from the Portuguese Coast. Toxicon 2009, 54, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, K.; Horie, M.; Tokimitsu, S.; Toda, T.; Taguchi, S. Variability in toxicity of the dinoflagellate Alexandrium tamarense isolated from hiroshima bay, western japan, as a reflection of changing environmental conditions. J. Plankton Res. 2001, 23, 271–278. [Google Scholar] [CrossRef]
- Lee, T.C.-H.; Kwok, O.-T.; Ho, K.-C.; Lee, F.W.-F. Effects of different nitrate and phosphate concentrations on the growth and toxin production of an Alexandrium tamarense strain collected from drake passage. Mar. Environ. Res. 2012, 81, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.C.Y.; Murata, A.; Nagashima, Y.; Taguchi, S. Variability in toxicity of the dinoflagellate Alexandrium tamarense in response to different nitrogen sources and concentrations. Toxicon 2004, 43, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Lim, P.-T.; Leaw, C.-P.; Kobiyama, A.; Ogata, T. Growth and toxin production of tropical Alexandrium minutum halim (Dinophyceae) under various nitrogen to phosphorus ratios. J. Appl. Phycol. 2010, 22, 203–210. [Google Scholar] [CrossRef]
- Xu, J.; Ho, A.Y.T.; He, L.; Yin, K.; Hung, C.; Choi, N.; Lam, P.K.S.; Wu, R.S.S.; Anderson, D.M.; Harrison, P.J. Effects of inorganic and organic nitrogen and phosphorus on the growth and toxicity of two Alexandrium species from Hong Kong. Harmful Algae 2012, 16, 89–97. [Google Scholar] [CrossRef]
- Abadie, E.; Kaci, L.; Berteaux, T.; Hess, P.; Sechet, V.; Masseret, E.; Rolland, J.; Laabir, M. Effect of nitrate, ammonium and urea on growth and pinnatoxin G production of Vulcanodinium rugosum. Mar. Drugs 2015, 13, 5642. [Google Scholar] [CrossRef] [PubMed]
- Gedaria, A.I.; Luckas, B.; Reinhardt, K.; Azanza, R.V. Growth response and toxin concentration of cultured Pyrodinium bahamense var. Compressum to varying salinity and temperature conditions. Toxicon 2007, 50, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; Smith, K.; Selwood, A.; McNabb, P.; Molenaar, S.; Munday, R.; Wilkinson, C.; Hallegraeff, G. Production of pinnatoxins E, F and G by scrippsielloid dinoflagellates isolated from Franklin Harbour, South Australia. N. Z. J. Mar. Freshw. Res. 2011, 45, 703–709. [Google Scholar] [CrossRef]
- Hansen, G.; Daugbjerg, N.; Franco, J.M. Morphology, toxin composition and lsu rdna phylogeny of Alexandrium minutum (Dinophyceae) from Denmark, with some morphological observations on other European strains. Harmful Algae 2003, 2, 317–335. [Google Scholar] [CrossRef]
- Martins, C.A.; Kulis, D.; Franca, S.; Anderson, D.M. The loss of psp toxin production in a formerly toxic Alexandrium lusitanicum clone. Toxicon 2004, 43, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Satta, C.T.; Anglès, S.; Lugliè, A.; Guillén, J.; Sechi, N.; Camp, J.; Garcés, E. Studies on dinoflagellate cyst assemblages in two estuarine Mediterranean Bays: A useful tool for the discovery and mapping of harmful algal species. Harmful Algae 2013, 24, 65–79. [Google Scholar] [CrossRef]
- Rhodes, L.; Smith, K.; Selwood, A.; McNabb, P.; van Ginkel, R.; Holland, P.; Munday, R. Production of pinnatoxins by a peridinoid dinoflagellate isolated from Northland, New Zealand. Harmful Algae 2010, 9, 384–389. [Google Scholar] [CrossRef]
- Selwood, A.I.; Wilkins, A.L.; Munday, R.; Shi, F.; Rhodes, L.L.; Holland, P.T. Portimine: A bioactive metabolite from the benthic dinoflagellate Vulcanodinium rugosum. Tetrahedron Lett. 2013, 54, 4705–4707. [Google Scholar] [CrossRef]
- Selwood, A.I.; Wilkins, A.L.; Munday, R.; Gu, H.F.; Smith, K.F.; Rhodes, L.L.; Rise, F. Pinnatoxin H: A new pinnatoxin analogue from a South China Sea Vulcanodinium rugosum isolate. Tetrahedron Letters 2014, 55, 5508–5510. [Google Scholar] [CrossRef]
- Hernandez-Becerril, D.U.; Rodriguez-Palacio, M.C.; Lozano-Ramirez, C. Morphology and life stages of the potentially pinnatoxin-producing thecate dinoflagellate Vulcanodinium rugosum from the tropical Mexican Pacific. Bot. Mar. 2013, 56, 535–540. [Google Scholar] [CrossRef]
- Munday, R.; Selwood, A.I.; Rhodes, L. Acute toxicity of pinnatoxins E, F and G to mice. Toxicon 2012, 60, 995–999. [Google Scholar] [CrossRef] [PubMed]
- Hallegraeff, G.M. A review of harmful algal blooms and their apparent global increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef]
- Anderson, D.M.; Kulis, D.M.; Sullivan, J.J.; Hall, S.; Lee, C. Dynamics and physiology of saxitoxin production by the dinoflagellates Alexandrium spp. Mar. Biol. 1990, 104, 511–524. [Google Scholar] [CrossRef]
- Garcia-Altares, M.; Casanova, A.; Bane, V.; Diogène, J.; Furey, A.; de la Iglesia, P. Confirmation of pinnatoxins and spirolides in shellfish and passive samplers from Catalonia (Spain) by liquid chromatography coupled with triple quadrupole and high-resolution hybrid tandem mass spectrometry. Mar. Drugs 2014, 12, 3706–3732. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, L.A.; Selwood, A.I.; McNabb, P.; Rhodes, L. Benthic dinoflagellate toxins in two warm-temperate estuaries: Rangaunu and Parengarenga Harbours, Northland, New Zealand. Harmful Algae 2011, 10, 559–566. [Google Scholar] [CrossRef]
- Takada, N.; Umemura, N.; Suenaga, K.; Chou, T.; Nagatsu, A.; Haino, T.; Yamada, K.; Uemura, D. Pinnatoxins B and C, the most toxic components in the pinnatoxin series from the okinawan bivalve Pinna muricata. Tetrahedron Lett. 2001, 42, 3491–3494. [Google Scholar] [CrossRef]
- McCarron, P.; Rourke, W.A.; Hardstaff, W.; Pooley, B.; Quilliam, M.A. Identification of pinnatoxins and discovery of their fatty acid ester metabolites in mussels (Mytilus edulis) from Eastern Canada. J. Agric. Food Chem. 2012, 60, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Rundberget, T.; Aasen, J.A.B.S.; Selwood, A.I.; Miles, C.O. Pinnatoxins and spirolides in norwegian blue mussels and seawater. Toxicon 2011, 58, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Selwood, A.I.; Miles, C.O.; Wilkins, A.L.; van Ginkel, R.; Munday, R.; Rise, F.; McNabb, P. Isolation, structural determination and acute toxicity of pinnatoxins E, F and G. J. Agric. Food Chem. 2010, 58, 6532–6542. [Google Scholar] [CrossRef] [PubMed]
- Abadie, E.; Rhodes, L.; Chomerat, N.; Chiantella, C.; Crottier, A.; Laabir, M. Occurrence of the neurotoxic dinoflagellate Vulcanodinium rugosum in mediterranean: What are the driving environnemental factors of its dynamic. J. Plankton Res. Submission in progress.
- Moore, S.K.; Mantua, N.J.; Hickey, B.M.; Trainer, V.L. The relative influences of el niño-southern oscillation and pacific decadal oscillation on paralytic shellfish toxin accumulation in Northwest Pacific shellfish. Limnol. Oceanogr. 2010, 55, 2262–2274. [Google Scholar] [CrossRef]
- Moore, S.K.; Mantua, N.J.; Hickey, B.M.; Trainer, V.L. Recent trends in paralytic shellfish toxins in puget sound, relationships to climate, and capacity for prediction of toxic events. Harmful Algae 2009, 8, 463–477. [Google Scholar] [CrossRef]
- Hiscock, K.; Southward, A.; Tittley, I.; Hawkins, S. Effects of changing temperature on benthic marine life in Britain and Ireland. Aquat. Conserv. Mar. Freshw. Ecosyst. 2004, 14, 333–362. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Philippart, C.J.M.; Anadon, R.; Danovaro, R.; Dippner, J.W.; Drinkwater, K.F.; Hawkins, S.J.; Oguz, T.; O’Sullivan, G.; Reid, P.C. Impacts of climate change on european marine ecosystems: Observations, expectations and indicators. J. Exp. Mar. Biol. Ecol. 2011, 400, 52–69. [Google Scholar] [CrossRef]
- Wernberg, T.; Smale, D.A.; Thomsen, M.S. A decade of climate change experiments on marine organisms: Procedures, patterns and problems. Glob. Chang. Biol. 2012, 18, 1491–1498. [Google Scholar] [CrossRef]
- Harrison, P.J.; Waters, R.E.; Taylor, F.J.R. A broad-spectrum artificial seawater medium for coastal and open ocean phytoplankton. J. Phycol. 1980, 16, 28–35. [Google Scholar] [CrossRef]
- Guillard, R.R.L. Division Rates. In Handbook of Phycological Methods: Culture Methods and Growth Measurements; Janet R. Stein, Cambridge University Press: Cambridge, United Kingdom, 1973; Volume 1, pp. 289–311. [Google Scholar]
- Neuveux, J.; Lantoine, F. Spectrofluorometric assay for chlorophylls phaeopigments using the least squares approximation technique. Deep Sea Res. 1993, 40, 1747–1765. [Google Scholar] [CrossRef]


| Strain and Studied Area | Temperature (°C) | Salinity | Irradiance (µmol photons m−2 s−1) | Culture Medium | Detected Toxins (Amount in pg cell−1) | Toxicity (LD 50 mice) | Reference |
|---|---|---|---|---|---|---|---|
| Mediterranean waters | |||||||
| IFR-VRU-01(Ingril Lagoon France) | 25–30 | 30–35 | 100 | ENSW | PnTX-G (0.14–0.36 ) Portimine | This study, Abadie et al. (2015) [48] | |
| IFR-VRU-01(Ingril Lagoon France) | 18 | 38 | 200 | L1 | PnTX-G (4.7) | Hess et al. (2013) [6] | |
| ** (Ebre Delta Spain) | ** | ** | ** | ** | Satta et al. (2013) [53] | ||
| Pacific waters | |||||||
| CAWD163 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (0.8) PnTX-F (5.1) | Rhodes et al. (2010) [54] | |
| CAWD166 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (3.7) PnTX-F (20.1) | Rhodes et al. (2010) [54] | |
| CAWD167 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (1.4) PnTX-F (8.4) | Rhodes et al. (2010) [54] | |
| CAWD168 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (0.8) PnTX-F (4.6) | IP 1.33 mg/kg gavage 2.33 mg/kg | Rhodes et al. (2010) [54] |
| CAWD170 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (2.4) PnTX-F (13.6) | Rhodes et al. (2010) [54] | |
| CAWD171 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (0.5) PnTX-F (3.5) | Rhodes et al. (2010) [54] | |
| CAWD178 (Rangaunu Harbour NZ) | 25 | ** | 70–100 | K | PnTX-E (0.4) PnTX-F (2.3) | Rhodes et al. (2010) [54] | |
| ** (Rangaunu Harbour NZ) | * | ** | * | K | Portimine | IP: 1570 µg/kg | Selwood et al. (2013) [55] |
| CAWD188 (Ishigakijima Island Okinawa Japan) | * | ** | ** | K | PnTX-G (11.9) | Smith et al. (2011) [9] | |
| CAWD190 (Ishigakijima Island Okinawa Japan) | * | ** | ** | K | PnTX-G (15) | Smith et al. (2011) [9] | |
| G65 (South China Sea) | 20 | ** | 90 | f2 | new PnTX (20) | Zeng et al. (2012) [12] | |
| CAWD198 (South China Sea) | 25 | ** | 100 | K | PnTX-H | IP 67 µg/kg gavage 163 µg/kg | Selwood et al. (2014) [56] |
| ** (Lazaro Cardenas Michoacan Mexico) | 18–20 | ** | 90–167 | L1SE | ** | Hernandez-Becerril et al. (2013) [57] | |
| Indian Ocean | |||||||
| CAWD180 (Franklin Harbour Australia) | 25 | K | PnTX-G (87) PnTX-E (10) PnTX-F (41) PnTX-A (1.3) | Rhodes et al. (2011) [50] | |||
| CAWD180 (Franklin Harbour Australia) | 25 | K | PnTX-G (13) | Rhodes et al. (2011) [50] | |||
| CAWD183 (Franklin Harbour Australia) | 25 | ** | ** | K | PnTX-G | IP Fed: 48.0 µg/kg IP Fasted: 42.7 µg/kg | Munday et al. (2012) [58] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abadie, E.; Muguet, A.; Berteaux, T.; Chomérat, N.; Hess, P.; Roque D’OrbCastel, E.; Masseret, E.; Laabir, M. Toxin and Growth Responses of the Neurotoxic Dinoflagellate Vulcanodinium rugosum to Varying Temperature and Salinity. Toxins 2016, 8, 136. https://doi.org/10.3390/toxins8050136
Abadie E, Muguet A, Berteaux T, Chomérat N, Hess P, Roque D’OrbCastel E, Masseret E, Laabir M. Toxin and Growth Responses of the Neurotoxic Dinoflagellate Vulcanodinium rugosum to Varying Temperature and Salinity. Toxins. 2016; 8(5):136. https://doi.org/10.3390/toxins8050136
Chicago/Turabian StyleAbadie, Eric, Alexia Muguet, Tom Berteaux, Nicolas Chomérat, Philipp Hess, Emmanuelle Roque D’OrbCastel, Estelle Masseret, and Mohamed Laabir. 2016. "Toxin and Growth Responses of the Neurotoxic Dinoflagellate Vulcanodinium rugosum to Varying Temperature and Salinity" Toxins 8, no. 5: 136. https://doi.org/10.3390/toxins8050136
APA StyleAbadie, E., Muguet, A., Berteaux, T., Chomérat, N., Hess, P., Roque D’OrbCastel, E., Masseret, E., & Laabir, M. (2016). Toxin and Growth Responses of the Neurotoxic Dinoflagellate Vulcanodinium rugosum to Varying Temperature and Salinity. Toxins, 8(5), 136. https://doi.org/10.3390/toxins8050136







